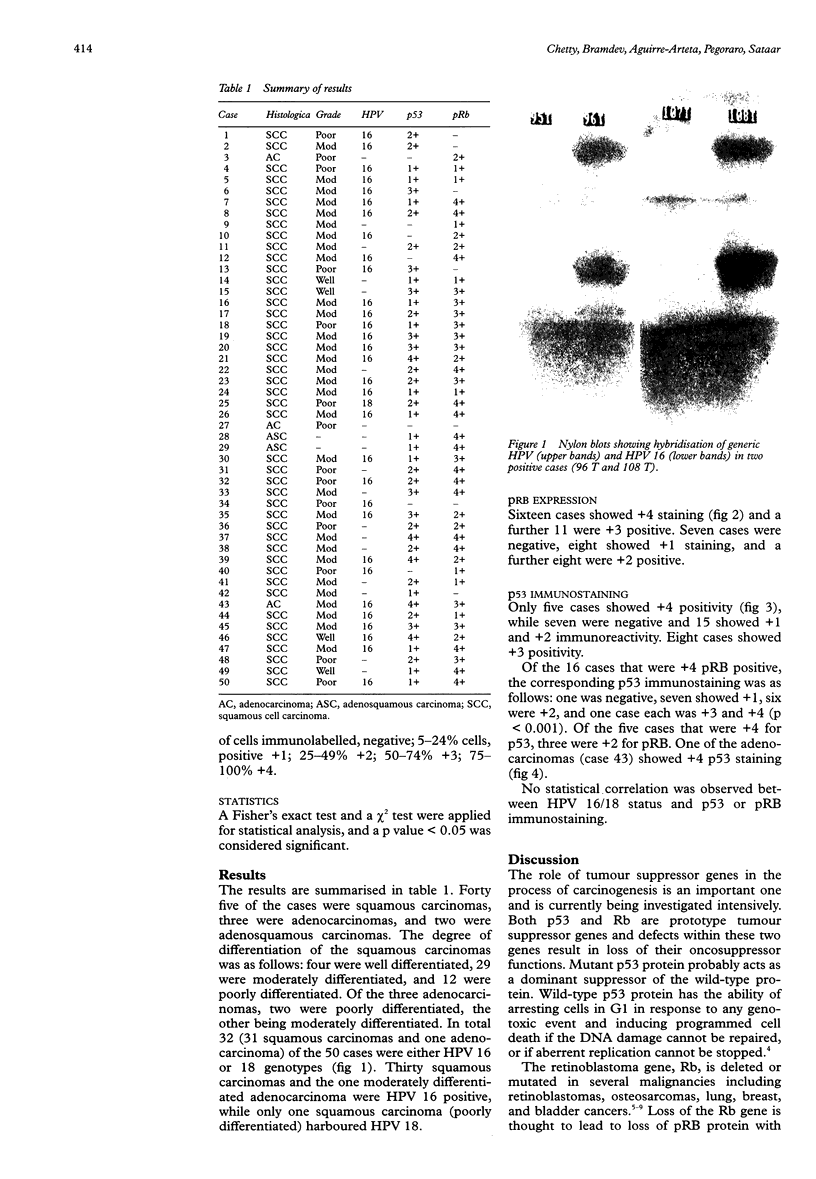
Abstract
AIM: To ascertain the extent of retinoblastoma protein (pRB) expression in comparison to p53 protein and human papilloma viruses (HPV) 16/18 status in cervical carcinomas. METHODS: Fifty cases of invasive cervical carcinoma were HPV typed for genotypes 16 and 18 using consensus primers by polymerase chain reaction (PCR). Immunohistochemistry for pRB and p53 was done on formalin fixed tissue using microwave antigen retrieval and commercially available antibodies. RESULTS: Forty five cases were squamous carcinomas, three were adenocarcinomas, and two were adenosquamous carcinomas. Thirty one cases were HPV 16 positive and one was HPV 18. Sixteen cases showed +4 pRB expression and a further 11 were +3 positive. Seven cases were negative. Only five cases (10%) showed +4 p53 immunostaining, while seven were negative and 15 were +1. Of the 16 pRB +4 positive cases, one was negative for p53 and a further seven were +1 positive. This inverse pattern of staining between pRB and p53 had a p value of < 0.001. No correlation was observed between HPV 16/18 status and p53 and/or pRB staining. CONCLUSIONS: pRB is expressed in the majority of cases of cervical cancer (86%), with more than 75% (+4) of the tumour cell population being positive in 16 cases (32%). There appears to be a general inverse pattern of staining between pRB (high) and p53 (low) in cervical cancer. The expression of both pRB and p53 proteins is independent of the HPV 16/18 status of the tumour.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Busby-Earle R. M., Steel C. M., Williams A. R., Cohen B., Bird C. C. p53 mutations in cervical carcinogenesis--low frequency and lack of correlation with human papillomavirus status. Br J Cancer. 1994 Apr;69(4):732–737. doi: 10.1038/bjc.1994.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper K., Herrington C. S., Evans M. F., Gatter K. C., McGee J. O. p53 antigen in cervical condylomata, intraepithelial neoplasia, and carcinoma: relationship to HPV infection and integration. J Pathol. 1993 Sep;171(1):27–34. doi: 10.1002/path.1711710107. [DOI] [PubMed] [Google Scholar]
- Cooper K., Herrington C. S., Stickland J. E., Evans M. F., McGee J. O. Episomal and integrated human papillomavirus in cervical neoplasia shown by non-isotopic in situ hybridisation. J Clin Pathol. 1991 Dec;44(12):990–996. doi: 10.1136/jcp.44.12.990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullen A. P., Reid R., Campion M., Lörincz A. T. Analysis of the physical state of different human papillomavirus DNAs in intraepithelial and invasive cervical neoplasm. J Virol. 1991 Feb;65(2):606–612. doi: 10.1128/jvi.65.2.606-612.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das B. C., Sharma J. K., Gopalakrishna V., Luthra U. K. Analysis by polymerase chain reaction of the physical state of human papillomavirus type 16 DNA in cervical preneoplastic and neoplastic lesions. J Gen Virol. 1992 Sep;73(Pt 9):2327–2336. doi: 10.1099/0022-1317-73-9-2327. [DOI] [PubMed] [Google Scholar]
- Dyson N., Howley P. M., Münger K., Harlow E. The human papilloma virus-16 E7 oncoprotein is able to bind to the retinoblastoma gene product. Science. 1989 Feb 17;243(4893):934–937. doi: 10.1126/science.2537532. [DOI] [PubMed] [Google Scholar]
- Friend S. H., Bernards R., Rogelj S., Weinberg R. A., Rapaport J. M., Albert D. M., Dryja T. P. A human DNA segment with properties of the gene that predisposes to retinoblastoma and osteosarcoma. Nature. 1986 Oct 16;323(6089):643–646. doi: 10.1038/323643a0. [DOI] [PubMed] [Google Scholar]
- Hansen M. F., Koufos A., Gallie B. L., Phillips R. A., Fodstad O., Brøgger A., Gedde-Dahl T., Cavenee W. K. Osteosarcoma and retinoblastoma: a shared chromosomal mechanism revealing recessive predisposition. Proc Natl Acad Sci U S A. 1985 Sep;82(18):6216–6220. doi: 10.1073/pnas.82.18.6216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrington C. S. Human papillomaviruses and cervical neoplasia. I. Classification, virology, pathology, and epidemiology. J Clin Pathol. 1994 Dec;47(12):1066–1072. doi: 10.1136/jcp.47.12.1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehn H., Villa L. L., Marziona F., Hilgarth M., Hillemans H. G., Sauer G. Physical state and biological activity of human papillomavirus genomes in precancerous lesions of the female genital tract. J Gen Virol. 1988 Jan;69(Pt 1):187–196. doi: 10.1099/0022-1317-69-1-187. [DOI] [PubMed] [Google Scholar]
- Logothetis C. J., Xu H. J., Ro J. Y., Hu S. X., Sahin A., Ordonez N., Benedict W. F. Altered expression of retinoblastoma protein and known prognostic variables in locally advanced bladder cancer. J Natl Cancer Inst. 1992 Aug 19;84(16):1256–1261. doi: 10.1093/jnci/84.16.1256. [DOI] [PubMed] [Google Scholar]
- Mansur C. P., Androphy E. J. Cellular transformation by papillomavirus oncoproteins. Biochim Biophys Acta. 1993 Dec 23;1155(3):323–345. doi: 10.1016/0304-419x(93)90013-3. [DOI] [PubMed] [Google Scholar]
- Miller S. A., Dykes D. D., Polesky H. F. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988 Feb 11;16(3):1215–1215. doi: 10.1093/nar/16.3.1215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittnacht S., Weinberg R. A. G1/S phosphorylation of the retinoblastoma protein is associated with an altered affinity for the nuclear compartment. Cell. 1991 May 3;65(3):381–393. doi: 10.1016/0092-8674(91)90456-9. [DOI] [PubMed] [Google Scholar]
- Moran E. Interaction of adenoviral proteins with pRB and p53. FASEB J. 1993 Jul;7(10):880–885. doi: 10.1096/fasebj.7.10.8344487. [DOI] [PubMed] [Google Scholar]
- Scheffner M., Münger K., Byrne J. C., Howley P. M. The state of the p53 and retinoblastoma genes in human cervical carcinoma cell lines. Proc Natl Acad Sci U S A. 1991 Jul 1;88(13):5523–5527. doi: 10.1073/pnas.88.13.5523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiffman M. H. Recent progress in defining the epidemiology of human papillomavirus infection and cervical neoplasia. J Natl Cancer Inst. 1992 Mar 18;84(6):394–398. doi: 10.1093/jnci/84.6.394. [DOI] [PubMed] [Google Scholar]
- T'Ang A., Varley J. M., Chakraborty S., Murphree A. L., Fung Y. K. Structural rearrangement of the retinoblastoma gene in human breast carcinoma. Science. 1988 Oct 14;242(4876):263–266. doi: 10.1126/science.3175651. [DOI] [PubMed] [Google Scholar]
- Vousden K. Interactions of human papillomavirus transforming proteins with the products of tumor suppressor genes. FASEB J. 1993 Jul;7(10):872–879. doi: 10.1096/fasebj.7.10.8393818. [DOI] [PubMed] [Google Scholar]
- Yokota J., Akiyama T., Fung Y. K., Benedict W. F., Namba Y., Hanaoka M., Wada M., Terasaki T., Shimosato Y., Sugimura T. Altered expression of the retinoblastoma (RB) gene in small-cell carcinoma of the lung. Oncogene. 1988 Oct;3(4):471–475. [PubMed] [Google Scholar]