Introduction
Data from epidemiologic and genome-wide association studies support the role of inherited predisposition in lung carcinogenesis, even after adjustment for age, sex, and smoking habits.1,2 Because lung cancer is most often diagnosed at advanced stages when there are no curative therapies, there is a critical need to identify patients who are at inherited risk. Identification of such patients is especially important because screening tests have been shown to reduce death from lung cancer in high-risk individuals.3 Germline epidermal growth factor receptor (EGFR) T790M is a rare mutation, which is associated with familial lung cancer; only 10 cases have thus far been reported, usually in association with concurrent somatic gain-of-function EGFR mutations.4–9 We describe 2 contrasting presentations of germline EGFR T790M mutation, including a previously undescribed patient with a concurrent KRAS mutation, and discuss the potential pathogenesis of lung cancer in these patients.
Materials and Methods
In the course of clinical molecular profiling of patients with lung cancer in the Thoracic Oncology Clinic, Center for Cancer Research, National Cancer Institute, we identified 2 patients with germline T790M mutations. Screening for EGFR and KRAS mutations was performed on all patients using pyrosequencing (PyroMark Q24, Qiagen, Valencia, CA) to detect the most common point mutations, and capillary gene electrophoresis was used to detect small deletions/insertions. Our assay covers EGFR point mutations involving exon 18 (codon 719), exon 20 (codon 790), and exon 21 (codons 858, 861, and 863); insertions involving exon 20; and deletions involving exon 19. For KRAS, all potential mutations involving codons 12, 13 and 61 are assessed. Paraffin-embedded tumors were macrodissected to ensure that the tumor percentage was at least 20% for all assays. For cases in which germline mutations were suspected, normal adjacent lung was macrodissected and analyzed, and peripheral blood mononuclear cells were obtained to confirm the tissue results.
Case Report
Case 1
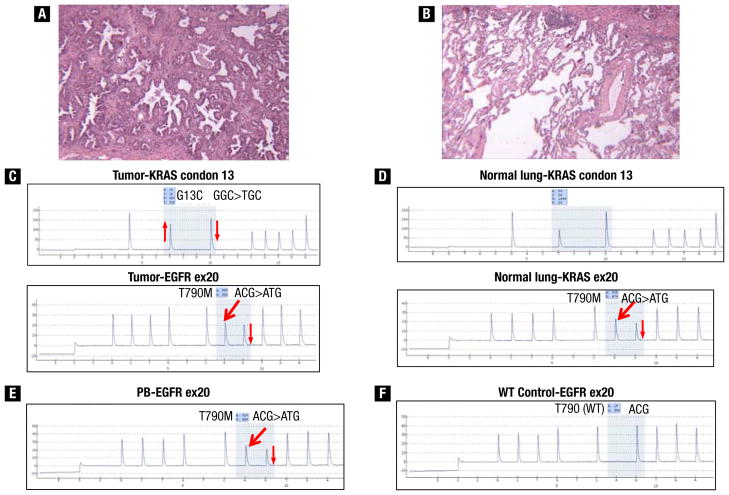
A 70-year-old white woman with a 60-pack-year smoking history presented with metastatic lung adenocarcinoma. Seventeen years before presentation she underwent right upper lobectomy for stage IA disease, and 2 years ago underwent right pneumonectomy followed by adjuvant chemotherapy for recurrent disease. Her family history was notable for her father who was a smoker and died of lung cancer in his 60s. The patient’s younger brother was also a smoker and had throat cancer at age 62 years. Pathologic examination of pneumonectomy specimens showed moderately differentiated adenocarcinoma with bronchoalveolar differentiation (Figure 1A). Pyrosequencing detected an EGFR exon 20 T790M mutation in a 1:1 ratio with the wild-type allele and a KRAS G13C mutation (Figure 1C), but no gain-of-function mutations were identified. Because it is unusual to have T790M mutations in the absence of exposure to EGFR tyrosine kinase inhibitor (TKI) or gain-of-function mutations, the possibility of germline EGFR T790M mutation was assessed. We found EGFR T790M with identical allelic frequency in microdissected normal lung, peripheral blood mononuclear cells, and tumor, indicating a germline mutation (Figure 1B–E). A KRAS G13C mutation was found only in the tumor. The patient achieved a minimal response to pemetrexed and sirolimus as part of a phase I trial and remained in the study for more than a year.
Figure 1.
(A and B) Histopathologic sections (patient 1) showing moderately differentiated adenocarcinoma with bronchoalveolar differentiation and microdissected normal lung. (C) Pyrograms from adenocarcinoma showing KRAS G13C mutation and EGFR exon 20 T790M mutation. (D) Pyrograms from adjacent normal lung sample showing wild-type KRAS sequence and EGFR exon 20 T790M mutation, indicating that the KRAS mutation is somatic, whereas the EGFR T790M mutation is occurring in the germline. (E) Pyrogram from peripheral blood showing the EGFR exon 20 T790M mutation, confirming its germline origin. (F) Pyrogram from a placental control demonstrating the wild-type (WT) T790 sequence for comparison with the mutant sequences present. The thick red arrows indicate new signals in the pyrograms caused by the presence of the mutant allele, and the thin red arrows indicate relative changes (up or down) in signals in the pyrograms caused by the presence of mutant alleles.
Abbreviations: ex = exon; PB = peripheral blood.
Case 2
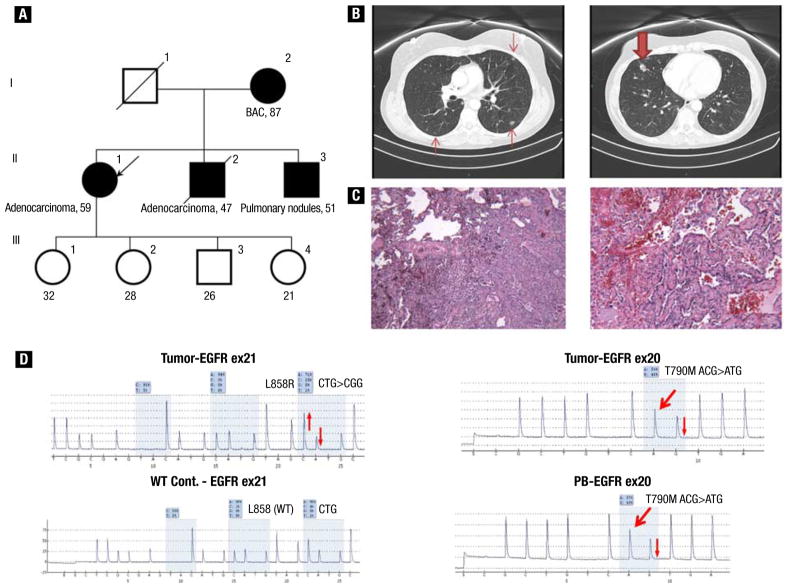
A 58-year-old white woman with a 3-pack-year smoking history was evaluated in view of a family history of lung cancer (Figure 2A). The patient’s mother (Figure 2A, I-2), a never-smoker, was known to have had lung nodules since her 70s and had biopsy-proven bronchioalveolar carcinoma. The patient’s younger brother (Figure 2A, II-2), a never-smoker, was diagnosed with invasive lung adenocarcinoma at age 45 years and died 2 years later. The patient’s youngest brother (Figure 2A, II-3) was a never-smoker who had bilateral lung nodules of uncertain cause, which were being followed radiographically. Two of the patient’s children (Figure 2A, III-1 and III-4) had normal pulmonary imaging studies. The patient underwent chest computed tomography, which showed bilateral ground-glass opacities and nodules (Figure 2B). Pathologic examination of specimens from multiple wedge resections of the right lung showed invasive moderately differentiated adenocarcinoma, predominantly of acinar morphologic type, with focal papillary and bronchoalveolar patterns (Figure 2C). Corresponding pyrograms showed a tumor-derived EGFR exon 21 L858R mutation and a germline EGFR T790M mutation (Figure 2D). Given the indolent nature of her disease, the patient was followed radiographically and continues to have stable disease 15 months after initial diagnosis.
Figure 2.
(A) Pedigree of family (case 2) with multiple cases of lung adenocarcinoma. (B) Computed tomographic scan of chest at diagnosis, which showed right middle lobe nodule measuring 1.6 cm (bold arrow) and bilateral ground-glass opacities (arrows). (C) Pathologic examination showed predominantly acinar moderately differentiated adenocarcinoma, with focal papillary and bronchoalveolar patterns. (D) Pyrograms showing somatic EGFR exon 21 L858R mutation, wild-type EGFR exon 21 L858 control (WT Cont.) for comparison, and EGFR T790M mutations present in tumor and peripheral blood (PB), indicating its germline origin. The thick red arrows indicate new signals in the pyrograms caused by the presence of the mutant allele, whereas the thin red arrows indicate relative changes (up or down) in signals in the pyrograms caused by the presence of mutant alleles.
Discussion
We describe 2 cases of a rare germline mutation, EGFR T790M, that is associated with familial lung cancer. In patient 1, a germline EGFR T790M mutation was found concurrently with a KRAS mutation in the absence of a gain-of-function EGFR mutation, a presentation that has not been described previously.
EGFR T790M mutations occur in exon 20, which encodes part of the kinase domain and results in an amino acid substitution at position 790 from threonine to methionine. Somatic EGFR T790M is known to occur as a “second-site mutation” in more than 50% of patients in whom acquired resistance to EGFR TKI develops.10,11
Germline EGFR T790M mutations are rare. Bell et al reported a family with multiple cases of lung cancer associated with germline transmission of the EGFR T790M mutation.4 After the initial description, only a limited number of additional cases have been identified from cohorts of never-smokers, lung cancer family probands, or patients with bronchoalveolar carcinoma or lung adenocarcinoma, or those with a pretreatment EGFR T790M mutation.5–9 Table 1 shows the clinical and molecular characteristics of previously reported cases with germline EGFR T790M mutations.4,5,7,9 Based on available data, germline EGFR T790M mutations are commonly associated with a family history of lung cancer and the presence of somatic gain-of-function EGFR kinase domain mutations (commonly exon 19 deletions or exon 21 L858R point mutations), a presentation exemplified by patient 2. Although EGFR T790M mutations have been reported in small fractions of tumor cells with concurrent KRAS mutations using highly sensitive mutation detection technology,12 to our knowledge concurrent KRAS and EGFR germline T790M mutations (patient 1), have not been described previously.
Table 1.
Characteristics of Previously Reported EGFR Germline T790M Cases
| Variable | n (%) |
|---|---|
|
| |
| Number of Patients | 10 |
|
| |
| Age (y) | |
| Median (range) | 64 (44–73) |
|
| |
| Sex | |
|
| |
| Male | 3 (30) |
|
| |
| Female | 4 (40) |
|
| |
| Not reported | 3 (30) |
|
| |
| Ethnicity | |
|
| |
| European | 5 (50) |
|
| |
| Asian-Indian | 1 (10) |
|
| |
| Not reported | 4 (40) |
|
| |
| Smoking History | |
|
| |
| Never-smoker | 7 (70) |
|
| |
| Former/current-smoker | 2 (20) |
|
| |
| Not reported | 1 (10) |
|
| |
| Stage | |
|
| |
| I | 1 (10) |
|
| |
| II | 0 (0) |
|
| |
| III | 1 (10) |
|
| |
| IV | 7 (70) |
|
| |
| Not reported | 1 (10) |
|
| |
| Histologic Type | |
|
| |
| Adenocarcinoma | 8 (80) |
|
| |
| Bronchoalveolar carcinoma | 1 (10) |
|
| |
| Poorly differentiated carcinoma | 1 (10) |
|
| |
| Concurrent Somatic EGFR Gain-of-Function Mutation | |
|
| |
| Yes | 8 (83) |
|
| |
| L858R | 2 (33) |
|
| |
| Exon 19 del | 3 (25) |
|
| |
| Different mutations in different lesions | 2 (17) |
|
| |
| G719A | 1 (8) |
|
| |
| No | (17) |
The precise mechanisms of EGFR germline T790M-mediated predisposition to lung cancer are unknown. In human bronchial epithelial cell lines, overexpression of EGFR T790M confers increased phosphorylation of tyrosine residues and is associated with a proliferative advantage over wild-type EGFR.5 In global phosphoproteomic analyses, the analogous substitution to T790M in bcr-abl, T315I, substantially increases kinase activity.13,14 Genetically engineered mice that express an EGFR T790M transgene develop lung tumors in the absence of additional gain-of-function EGFR kinase domain mutations, albeit with decreased incidence and longer latency than animals expressing EGFR L858R/T790M or even EGFR L858R alone.13 Although no additional EGFR kinase domain mutations were documented in the tumors of these EGFR T790M transgenic mice, the possibility that other cooperating oncogenes were present cannot be excluded.
Available data suggest a synergistic effect between EGFR T790M and other gain-of-function EGFR kinase domain mutations resulting in significantly enhanced catalytic phosphorylating activity of EGFR kinase.15 Because of its relatively weak oncogenic activity, additional mutations in EGFR or possibly other genes may be needed in addition to EGFR T790M to induce lung tumorigenesis,13 which was likely the case in patient 2 who had concurrent EGFR L858R and germline EGFR T790M. In patient 1, the extensive tobacco exposure suggests that mutant KRAS, rather than EGFR T790M, is the primary driver mutation. In this case, although we cannot exclude that EGFR T790M may be contributing to carcinogenesis by concurrent activation of the EGFR signaling pathway, it may simply be a passenger mutation.5
The clinical implications of germline EGFR T90M mutation are currently unknown. Unanswered questions include the risk conferred by such mutations, the patterns of familial involvement, clinical course, and benefits of screening asymptomatic individuals harboring the mutations. The main impediment to better understanding the clinical implications of this mutation is the limited number of families that are known to be affected. Germline testing of patients who are found to have somatic EGFR T790M mutation before treatment with EGFR TKIs could enhance the rate of detection of germline EGFR T790M mutations.9 Future studies should assess whether radiographic surveillance, lifestyle modification, or chemoprevention could decrease the risk of death from lung cancer in healthy individuals with germline EGFR T790M mutations.
Conclusion
We report 2 cases of germline EGFR T790M mutations: a previously undescribed presentation with concurrent KRAS mutation and a more typical presentation with a concurrent somatic EGFR exon 21 L858R mutation. In the first case, EGFR T790M may be a passenger mutation or it may be contributing to carcinogenesis by concurrent activation of the EGFR signaling pathway. Regardless, the assessment of tumors in patients with germline EGFR T790M mutations should not be limited to the classic EGFR-activating mutations and should include other somatic mutations associated with lung cancer, including KRAS.
Clinical Practice Points.
Germline epidermal growth factor receptor (EGFR) T790M is a rare mutation associated with familial lung cancer. Among the limited number of cases reported to date, germline EGFR T790M has been associated only with somatic gain-of-function EGFR mutations.
We report 2 presentations of germline EGFR T790M mutations, including a unique case with a previously undescribed associated KRAS mutation, and a second more typical presentation with a concurrent somatic EGFR exon 21 L858R mutation.
Although the major oncogenic pathway to lung cancer in patients with T790M germline mutations appears to be activation of EGFR through secondary somatic mutations, these patients may also be at risk for the development of tumors through mutational activation of other oncogenes such as KRAS, and screening of tumors that occur in these families should not be limited to EGFR-activating mutations.
Footnotes
Disclosure
This research was supported by the Intramural Research Program of the National Institutes of Health (National Cancer Institute).
The authors have stated that they have no conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Gao Y, Goldstein AM, Consonni D, et al. Family history of cancer and nonmalignant lung diseases as risk factors for lung cancer. Int J Cancer. 2009;125:146–52. doi: 10.1002/ijc.24283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yang P, Li YF, Jiang RX, et al. A rigorous and comprehensive validation: common genetic variations and lung cancer. Cancer Epidemiol Biomarkers Prev. 2010;19:240–4. doi: 10.1158/1055-9965.EPI-09-0710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Aberle DR, Adams AM, Berg CD, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365:395–409. doi: 10.1056/NEJMoa1102873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bell DW, Gore I, Okimoto RA, et al. Inherited susceptibility to lung cancer may be associated with the T790M drug resistance mutation in EGFR. Nat Genet. 2005;37:1315–6. doi: 10.1038/ng1671. [DOI] [PubMed] [Google Scholar]
- 5.Vikis H, Sato M, James M, et al. EGFR-T790M is a rare lung cancer susceptibility allele with enhanced kinase activity. Cancer Res. 2007;67:4665–70. doi: 10.1158/0008-5472.CAN-07-0217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Prudkin L, Tang X, Wistuba II. Germ-line and somatic presentations of the EGFR T790M mutation in lung cancer. J Thorac Oncol. 2009;4:139–41. doi: 10.1097/JTO.0b013e3181915f92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Girard N, Lou E, Azzoli CG, et al. Analysis of genetic variants in never-smokers with lung cancer facilitated by an Internet-based blood collection protocol: a preliminary report. Clin Cancer Res. 2010;16:755–63. doi: 10.1158/1078-0432.CCR-09-2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tibaldi C, Giovannetti E, Vasile E, et al. Inherited germline T790M mutation and somatic epidermal growth factor receptor mutations in non-small cell lung cancer patients. J Thorac Oncol. 2011;6:395–6. doi: 10.1097/JTO.0b013e3182059a6f. [DOI] [PubMed] [Google Scholar]
- 9.Oxnard GR, Miller VA, Robson ME, et al. Screening for germline EGFR T790M mutations through lung cancer genotyping. J Thorac Oncol. 2012;7:1049–52. doi: 10.1097/JTO.0b013e318250ed9d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kobayashi S, Boggon TJ, Dayaram T, et al. EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N Engl J Med. 2005;352:786–92. doi: 10.1056/NEJMoa044238. [DOI] [PubMed] [Google Scholar]
- 11.Pao W, Miller VA, Politi KA, et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2005;2:225–35. doi: 10.1371/journal.pmed.0020073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Inukai M, Toyooka S, Ito S, et al. Presence of epidermal growth factor receptor gene T790M mutation as a minor clone in non-small cell lung cancer. Cancer Res. 2006;66:7854–8. doi: 10.1158/0008-5472.CAN-06-1951. [DOI] [PubMed] [Google Scholar]
- 13.Regales L, Balak MN, Gong Y, et al. Development of new mouse lung tumor models expressing EGFR T790M mutants associated with clinical resistance to kinase inhibitors. PLoS ONE. 2007;2:e810. doi: 10.1371/journal.pone.0000810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Skaggs BJ, Gorre ME, Ryvkin A, et al. Phosphorylation of the ATP-binding loop directs oncogenicity of drug-resistant bcr-abl mutants. Proc Natl Acad Sci U S A. 2006;103:19466–71. doi: 10.1073/pnas.0609239103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Suda K, Onozato R, Yatabe Y, et al. EGFR T790M mutation: a double role in lung cancer cell survival? J Thorac Oncol. 2009;4:1–4. doi: 10.1097/JTO.0b013e3181913c9f. [DOI] [PubMed] [Google Scholar]