Abstract
Social interaction can have a profound effect on individual behavior, perhaps most salient in interactions between sick suffering children and their parents. Chronic pain is a difficult condition that can produce considerable changes in behaviors in children that can secondarily have profound effects on their parents. It may create a functionally disabling negative feedback loop. Research supports the notion of alterations in the brain of individuals who observe and empathize with loved ones in acute pain. However, neural activity in relation to empathic responses in the context of chronic pain has not been examined. Ongoing suffering with chronic pain in a child can result in child's brain circuit alterations. However, prolonged suffering jointly experienced by the parent may putatively also produce maladaptive changes in their neural networks and consequently in parental behaviors. Here we put forth the conceptual framework for ‘Chronic pain contagion’ (CPC). We review the underlying processes in CPC and we discuss implications for devising and implementing treatments for children in chronic pain and their parents.
Keywords: empathy, chronic pain, pediatric, parent-child, salience network, stress, emotional contagion
Introduction
All mammals are thought to be capable of felt affective experiences (Panksepp and Panksepp, 2013). Observing one's own child suffering from pain is perhaps one of the cruelest aspects of life. Many children afflicted by chronic pain travel down a route that makes it difficult to adhere to normal life processes and experiences. All complex behaviors are brain-derived and chronic pain may be ‘brain modulating’ for both the child and parent; for the child through direct effects of chronic pain (Linnman et al., 2013)and parental responses to their condition, and for the parents whose behaviors may be modified by observing their child's suffering. The children who suffer and the putative manner in which their parent's physiology, behavior, and emotional state are altered as a consequence is the focus of this review. It is a theme generally overlooked clinically, and yet it is seemingly quite obvious that such ‘altered parental state’ may contribute significantly to maladaptive parental behavioral responses and ultimately influence the treatment outcomes (i.e., improvement or deterioration) of their child. Addressing the unique needs of a parent whose child is suffering with chronic pain is emerging as an important area of inquiry and critical point of intervention (Palermo et al., 2014; Vervoort et al., 2014).
Bio-social distress induces abnormal behaviors amongst animals and humans (Decety et al., 2016). Social trauma can affect neural systems and alter normally adaptive behaviors (Feldman, 2015). Dramatic alterations in behavior are exemplified in the animal kingdom amongst elephants who in the last century have experienced significant social trauma from poaching, culls, and habitat loss (Bradshaw et al., 2005) and in human populations exposed to war or mass disasters (Feldman and Vengrober, 2011) that can result in long-lasting effects on brain function and behaviors. This reciprocal pattern has been observed in the behaviors of children of holocaust survivors affected through the transmission of trauma their parents were exposed to (Wiseman et al., 2006). The opposite has also been observed when parents are dealing with chronic health conditions in their children, wherein parenting a child with a chronic condition has a significant impact on parent well-being (Hauser-Cram et al., 2001) (Riva et al., 2014).
In this review we focus on child pain. It is a prototypical example of suffering (often a symptom of many illnesses) and evidence indicates that facing another in pain constitutes a signal of threat and profoundly impacts observers (Goubert et al., 2005; Goubert et al., 2013). Furthermore, it is particularly relevant among parents and children due to the inherent interdependence of this relationship. The behavior of children, when affected by chronic pain, can be significantly altered because of their dependence on their primary caregivers; most often their parents (Palermo et al., 2010; Sieberg et al., 2011). The nature of parent-child interactions is complex and may, in the context of chronic pain, result in profound perturbations of psychobiological processes in both the child and the parent. The complexity of such parent-child interactions are now beginning to be understood in the context of brain neurobiology and pain behavior (for example through the lens of empathy research (Lamm et al., 2016)).
Chronic pain in children may produce a stressor in parent-child interactions that results in short term (in the early course of the condition) and long term (viz., in later life) perturbations in brain processes that manifest as alterations in physiology, behavior, and emotional state in the caregiver. The present manuscript is divided into five main sections. In section (1) Parenting a child with pain – a stressful and lonely journey, we describe the problem parents are faced with; in section (2) Empathy – The Silent Communicator of Another's Internal State, we describe the potentially brain modulating impact of a child's chronic pain on a parent with empathy as a core concept; section (3) High emotional contagion– A driver of heightened distress and pain control behaviors describes how emotionally converging with another's felt experience can lead to empathic distress and maladaptive responses (e.g., miscarried helping behaviors) in parents; in section (4) The Maladaptive Feedback Loop leading to altered brain state, constitutes a fourth section where we argue that, as a consequence of these abnormal parent-child interactions relating to chronic pain, a feedback loop is triggered that in turn resonates with and impacts the child. To end, in section (5) Where do we go from here? Disengaging Negative Circles of Interaction, we provide potential insights to unravel these intricate and sometimes detrimental interaction patterns through presentation of recent innovative research to stimulate novel treatment approaches and provide suggestions for further research on this topic. Although pain and the parent-child dyad are particularly well suited to describe the alterations that unfold, these changes likely apply to other dyads (e.g., partners) and go beyond pain to encompass other chronic medical conditions
1. Parenting a child with pain – a stressful and lonely journey
There is an associated loneliness experienced by parents as it relates to their child's chronic pain. Pain is invisible; it has no objective marker and is thus hard to understand. Further, treatments for chronic pain do not have the same level of efficacy and sensitivity as treatments for most infections. As a result, parents suffer fatigue in the face of their child's chronic pain (Gaughan et al., 2014; Jordan et al., 2007), become socially isolated (Oliveira et al., 2015), and experience increased stress, anxiety, anger, or depressive symptoms (Jordan et al., 2008). A child's chronic pain can have a devastating impact across a parent's various life domains, including the financial strain associated with frequent physician visits. Although most research has focused on the maternal experience of parenting a child with chronic pain, the impact reverberates across both parents, with fathers reporting feeling helpless and ‘stretched’ and having to adopt multiple roles to help manage the day-to-day activities of the family unit (Jordan et al., 2015).
Such struggles are particularly germane when a child's pain condition is ill-defined and not easily treated. A parent's life can become consumed by the search for a diagnosis and cure for the pain condition (Noel et al., 2016). This struggle to deal with child pain is further amplified when the child is younger, if the child is struggling with depressive symptoms, and the pain has persisted for a longer duration (Palermo et al., 2014). A diminished resilience may contribute to the unfolding of maladaptive responses including self-isolation, stopping work, and engaging in (miscarried) helping behaviors. It is a feed-forward-failing system that may contribute to increased allostatic load (McEwen, 2013) ultimately promoting immune dysregulation and inflammation (Jaremka et al., 2013).
2. Empathy – The Silent Communicator of Another's Internal State
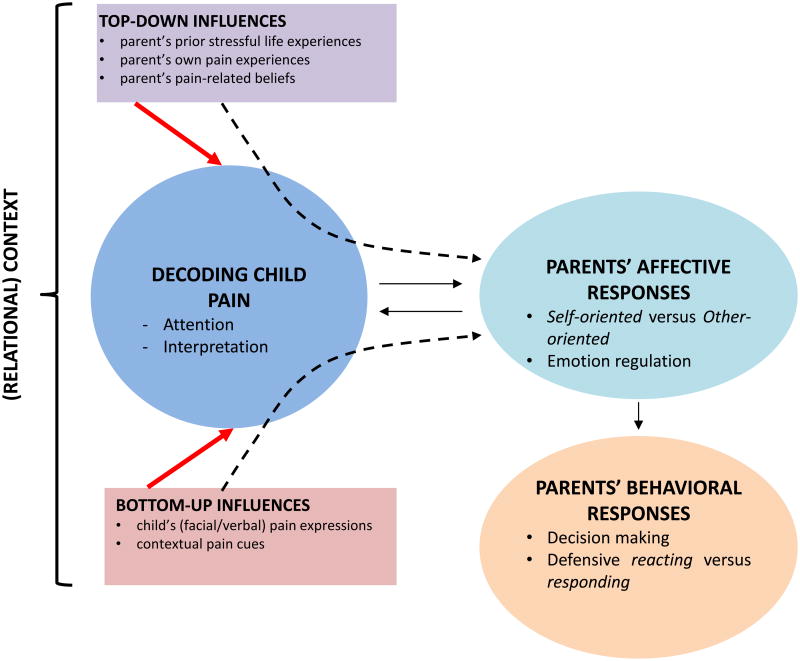
To gain insight into the potentially brain modulating impact of a child's chronic pain on a parent, empathy is a core concept. We can garner a great deal from evidence showing heightened distress and empathic responding when faced with another in pain (Lamm et al., 2016) and the emerging literature on the neurobiological bases of empathy (Shamay-Tsoory, 2011). Empathy in the context of pain is broadly defined as “a sense of knowing the experience of another person with cognitive, affective and behavioural components” (Goubert et al., 2005; Goubert et al., 2013) (Figure 1). Empathy has been conceptualized as encompassing multiple elements including: (i) emotional recognition (recognizing the felt experience of another); (ii) perspective-taking (the capacity to understand another's perspective or mental state; other-oriented); and (iii) emotional contagion (emotionally converging with another's felt experience; self-oriented (Decety and Jackson, 2004; Lamm et al., 2016). Empathizing with another is a two-way street - it involves implicit and explicit processing of another's emotional experiences and also the production of a parallel emotion state in oneself. Empathy in the context of pain is a core process by which the observation of pain may impact others and is an essential process to understanding the plight of a parent whose child is suffering.
Figure 1. Empathy in the Context of Pain (adapted from Goubert et al., 2005).
, Empathy can be defined as “a sense of knowing the experience of another person with cognitive, affective and behavioral components” (Goubert et al., 2005; Goubert et al., 2013). In the context of child pain, parents' empathic responses may be influenced by three types of variables: the child's pain-related (non)verbal expressions (“bottom-up”; e.g., facial expression of pain), the parent's own characteristics and learning history (“top-down”; e.g., previous distressing experiences, parents' history of pain) and contextual variables (eg, parent-child attachment, family climate). The cognitive component of parental empathy refers to processes such as attention to and interpretation of the child's expression of pain. The emotional component refers to parents' self-oriented (distress, physiological resonance) versus other-oriented emotions (sympathy) and the regulation of those emotions. The behavioral component refers to processes of decision making with regard to the child's pain (treatment), and may range from defensive reacting (e.g., avoidance, protective parenting behaviors, miscarried helping) to adequate responding, where the parent is able to keep a self-other distinction and balances the child's (short-term) needs (e.g., rest) with longer-term goals for adaptive child development (e.g., persisting in valued activities in the presence of pain).
2.1 Neural bases for producing empathy
Empathy seems to be a phenomenon shared by both animals (Langford et al., 2006) (Martin et al., 2014) and humans (Goubert et al., 2005; Goubert et al., 2013; Preston and de Waal, 2002). Recent pre-clinical work in mice has demonstrated that familiarity is an important element that can enhance the empathic response (Gonzalez-Liencres et al., 2014). Two neural networks have been implicated for producing empathy: (i) the inferior frontal gyrus and the inferior parietal lobule which are necessary for emotion recognition and emotional contagion; and (ii) the ventromedial prefrontal cortex, temporoparietal region, and the medial temporal lobe, which are involved in self-reflection and autobiographical memory that may drive perspective-taking (Bernhardt and Singer, 2012). These two systems may be differentially involved in how a parent responds to a child's chronic pain. In the case of emotional contagion, dysfunction may be rooted in the temporoparietal junction where the self-other distinction arises. Self-other distinction is likely, as we will outline below, key to the ability to provide adequate helping behavior, while self-other confusion may promote parents' empathic distress and miscarried helping behavior.
2.2 Neurobiologically driven partnership between pain and empathy
Why the close association of pain and empathy? Pain is a critical element in survival and thus a salient issue for the individual experiencing pain and for others to observe and interpret for preservation of self and others (Goubert et al., 2009). Recent reports suggest that empathy for and observation of another in pain can result in a ‘vicarious pain’ experience that has an underlying neural circuitry partially similar to directly experiencing pain (Derbyshire et al., 2013; Fitzgibbon et al., 2012; Fitzgibbon et al., 2010; Lamm et al., 2011; Vachon-Presseau et al., 2011). A few studies have noted the theme of parallel brain regions involved in emotional and sensory processes being recruited when “I feel your pain” (Avenanti et al., 2005) (Hein and Singer, 2008); (Engen and Singer, 2013). Mirror neurons may provide one explanation of how we can experience “second-hand” pain or emotion (Gallese, 2007); (Rizzolatti and Craighero, 2004). Several other mechanisms have been put forward to explain the experience of vicarious pain (Fitzgibbon et al., 2010). Vicarious pain experiences have been primarily reported in individuals with an amputated limb (Fitzgibbon et al., 2010), suggesting that trauma may play a role. In a recent study higher levels of empathic concern (i.e., sympathy and compassion for unfortunate others) were observed among individuals who report vicariously responding to pain in everyday life (Vandenbroucke et al., 2014). Vicarious pain experiences are biologically adaptive and may provide important insights into the parent-child shared experience in the context of pain.
2.3 Shared circuits: Insula and cingulate
Additional core networks activated in the context of empathy have included the anterior insular cortex and medial/anterior cingulate cortex and overlap with activation observed during directly experienced pain (Lamm et al., 2011). In an article by Danziger and colleagues they evaluated empathy to pain in a unique population - individuals with congenital insensitivity to pain (Danziger et al., 2009). These patients showed normal fMRI responses to observed pain in this ‘shared circuit’ (anterior mid-cingulate cortex and anterior insula). As such, these findings provide insights into the brain's ability to evaluate other's feeling to observed pain without having a specific sensory experience of pain itself (i.e., as in a parent whose child is suffering in pain, but does not directly experience the pain themselves; Figure 2). In the context of parent-child interactions, seeing your child suffer from chronic pain may constitute a condition which may trigger vicarious pain experiences in parents, reflecting self-other contagion; and may involve changes in parents at a brain-systems level, with the tempoparietal junction, inferior frontal gyrus, inferior parietal lobule, insula, and cingulate key targets to examine for alterated connectivity and function.
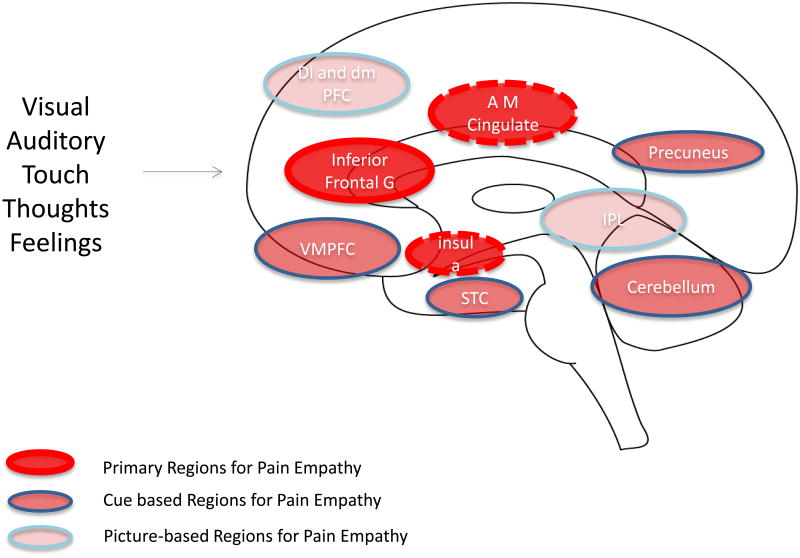
Figure 2. Key Brain Regions for Empathy.
The figure (adapted from (Lamm et al., 2011) summarizes data implicating complex neural networks that overlap for empathy: These include (1) Primary Regions for Pain Empathy which themselves have a large overlap with pain activations in experimental pain; (2) Cue based Regions for Pain Empathy that encompass complex visual, social and other signals; and (3) Picture-based Regions for Pain Empathy that aside from the specific salience of pictures used in the experimental setting, clearly visual inputs related to pictures may contribute to an aversive process. Although only one side of the brain is depicted in the figure, these results are not lateralized.
3. High emotional contagion – A driver of heightened distress and pain control behaviors
The topography of parent empathic behavior in the context of a child's pain provides important clues to its neurobiological roots. Based on the extant literature, high emotional contagion is a front-runner for explaining empathic distress and maladaptive responses (e.g., miscarried helping behaviors) in parents. As parents often are a child's primary care taker, their level of adaptation to stress is an important consideration on how they may manage their behaviors in relation to their suffering child. In resilient parents, adaptive processes handle the condition appropriately by providing heartfelt, calm emotional support during a child's distress. Moreover, positive affect and dispositional optimism can provide enormous beneficial responses to treatments (Bruce et al., 2014). As noted in an examination of interactions between parents and children with cancer, parent adaptive affective empathic responses (empathic concern/other-oriented minus state anxiety/self-oriented) predicted their child's positive disposition and less observed pain/distress during cancer treatment (Harper et al., 2012).
Conversely, empathic (personal) distress is associated with increased pain in children (Penner et al., 2008) and a parent's behavioral response that is likely aimed at regulating their own heightened distress may inadvertently contribute to the maintenance of their child's pain complaints (Dolgin and Phipps, 1989). High levels of emotional contagion (self-orientation) likely drives the empathetic distress exhibited in parents. This is best exemplified through accumulating evidence that supports the influence of parent pain catastrophizing and protective behavior on maladaptive outcomes in children with chronic pain. Parents who engage in catastrophic thinking prioritize pain control, potentially in an effort to decrease their own aversive emotional state (Caes et al., 2012) and have children who tend to engage in greater levels of pain catastrophizing (Lynch-Jordan et al., 2013). Moreover, catastrophic thinking about pain in the parent predicts an increased engagement in protective behaviors (e.g., keeping the child home from school or other activities to avoid pain exacerbation), which is in turn associated with increased child functional disability (Sieberg et al., 2011)and poor school functioning (Logan et al., 2012). Although it seems paradoxical that protective parenting behavior can lead to negative outcomes, this can be understood in the context of the underlying emotional/empathic state (self-oriented) that motivates the behavioral response. In this case, parents' drive to self-care (decrease their distress) dominates, with fewer cognitive resources for other-care (be attuned to the needs of the other independent of their own level of distress (Caes et al., 2011)).
3.1. Brain Systems Recruited in Parent Pain Empathy
Although the candidate regions recruited in parent pain empathy have been identified (i.e., tempoparietal junction, inferior frontal gyrus, inferior parietal lobule, insula, cingulate), these have been based on neuroimaging studies of empathy executed in the context of acute pain (Lamm et al., 2011). In these situations empathy is context-specific and has a defined end. In chronic pain the issue is more complex as it involves parents watching a loved one suffer and child responses towards their caregivers. Here empathy is confounded by the unknown or uncertain duration of pain, the fluctuation in pain intensity and associated child distress. Indeed, with pain chronification, so-called centralization of pain (where maladaptive processes lead to alterations that confer changes in cognition, altered reward and altered saliency amongst others (Borsook et al., 2013b) takes place and may result in an ongoing state, that the longer it lasts, the more difficult it is to treat.
Thus, recruited brain systems likely include the broader salience network (inclusive of cingulate, anterior insula, and medial prefrontal cortex, cerebellum, pulvinar) tasked with encoding sensory and affective stimuli importance and would seem to be critical in the interoceptive processing of others' suffering (Borsook et al., 2013a). Like any condition where there is an ongoing perturbation or stressor, depending on the resilience of the system, chronic activations may lead to circuit dysfunction. Such changes may be enhanced in parents who are more vulnerable due to prior events or circumstances (see section 4.1 Premorbid stressors below). Since emotional and cognitive changes may alter bodily state through the autonomic nervous system, these interoceptive processes may alter the brain through initial bombardment of emotional brain regulatory regions (Critchley, 2009) that affect fear and other neural systems (Panksepp, 2005). Advances in our understanding of the neurocircuitry of empathy have occurred through observation of changes secondary to lesions in specific brain regions that modulate gray matter volume, chemistry and other processes. Brain lesions in areas such as the frontal lobe (the prefrontal cortex, anterior cingulate (ACC), the anterior insula (AI)), the temporal lobe (the temporo-parietal junction (TP), temporal pole (TL)), and the amygdala (Leigh et al., 2013) have been associated with changes in empathy, providing further evidence for these areas as candidate regions for indexing alterations in parent brain state secondary to their child's pain.
4. The Maladaptive Feedback Loop leading to altered brain states
Here we discuss the concept of a maladaptive ‘Feed-Forward-Failure’ process in the interactions between the pain affected child and his/her parents and its potential impact on brain states. When a parent sees his/her child suffering with persistent pain the feedback loop is activated (Figure 3). What remains most salient for a parent when observing their child in pain (forward feed) is filtered through their prior experiences (constitutional makeup; e.g., parent history of pain or illness). This, in turn, leads to empathetic emotional responses that may vary between more self-oriented (distress) responses to more other-oriented emotions (sympathy). This fuels into parental behaviour, with as key element decision-making with regard to handling the child's pain, which may be impaired (e.g., driven by high levels of empathic distress) or cogent (e.g., driven by a resilient state of calmness and other-oriented emotions). Impaired decision making may give rise to defensive reacting (e.g., avoidance, protective parenting behaviors, miscarried helping) while cogent decision making may set the stage for adequate responding, in which the parent balances the child's short-term needs (e.g., rest) ànd longer-term goals for adaptive child development (e.g., persisting in valued activities in the presence of pain).
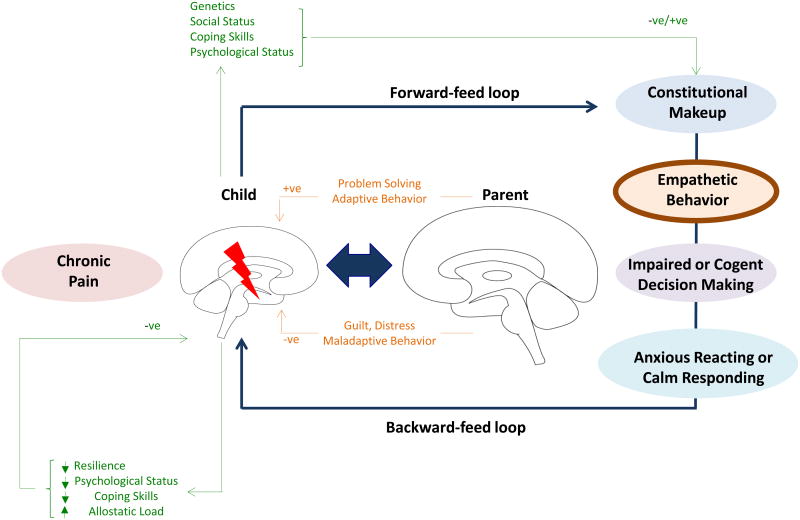
Figure 3. Parent-Child Feedback Loop.
A child in chronic pain sends verbal and nonverbal cues to his/her parent through the forward-feed pathway that is interpreted through the parent's constitutional make-up, comprised of prior stressful life experiences and ongoing daily stressors, among other elements. This sets the stage for parent empathic behavior that can go in one of two directions: cogent decision-making with calm responding or impaired decision-making with anxious reacting. The behavior of the parent is observed by the child (observational learning) and is the backward feed of the loop.
Not only may observing a child's (chronic) pain impact parents' responses, a child may be affected as well by observing his/her parents' reactions to his/her own pain and to the child's pain, a process called observational learning (backward feedback; (Goubert and Simons, 2013; Goubert et al., 2011). In the maladaptive feedback loop, when observing a parent's fearful/stress reactions to (child) pain, the child may learn to be (more) fearful of pain itself. Additionally, by observing parents' attempts at escaping or controlling pain, he/she may learn that avoidance is the most appropriate response to pain (Goubert and Simons, 2013). Extensive evidence is, however, available, that prolonged avoidance may fuel disability and distress. In turn, heightened child disability and distress may fuel distress and avoidance in parents, resulting in a vicious circle (Goubert and Simons, 2013; Simons et al., 2015). It remains to be determined to what extent these maladaptive parent-child processes impact physiological and brain mechanisms in both child and parent.
The parent-child maladaptive feedback loop can be triggered by ongoing and premorbid stressors. The salient issues include (1) the presence of premorbid stressors that render parents more vulnerable (i.e., parent own chronic pain state; (Higgins et al., 2015), (2) empathetic stress related to the child's pain condition (i.e., chronic pain contagion) and parents' caregiving burnout, and (3) feelings of failure or guilt by parents unable to eliminate their child's pain. These ongoing and preexisting stressors can alter major neural networks including the hypothalamic, amygdalar, and hippocampal axes (Dedovic et al., 2009). These changes may secondarily affect cognitive processes further adding to alterations in the parent's brain and consequent ability to be responsive, adapt to stress and not be engulfed in a failing process.
4.1 Premorbid stressors
The presence of premorbid stressors in the parent can serve as the diathesis to an altered brain state and maladaptive behavioral response to a child's pain. The neurobiological impact of stress on the body has been extensively studied through the lens of cortisol dysfunction (Mora et al., 2012). Premorbid vulnerability, such as early life stress, can alter receptor sensitivities within the HPA axis and blunt cortisol reactivity (Carpenter et al., 2007). Furthermore, prolonged early life stress such as growing up in poverty reduces hippocampal-prefrontal connections that alter hippocampal cortisol receptor sensitivities that shape adult cortisol levels and learning processes involving the hippocampus (Sripada et al., 2014). Several fMRI studies have shown that stressful stimuli can modulate neurocircuit function; for example, a study investigating the functioning of certain brain regions after inducing social stress found a decrease in amygdala-hippocampal connectivity that was associated with elevated salivary cortisol (Vaisvaser et al., 2013; Veer et al., 2012). Under normal conditions, the hippocampus inhibits the amygdala, reducing hyperactivity of the HPA axis, while under conditions of chronic stress hippocampal atrophy may disinhibit the amygdala, leading to dysregulation of the HPA axis (Boyle, 2013; Dedovic et al., 2009). A recent study even showed that individuals, particularly women, may be at risk for elevated inflammation when reacting with negative affect to minor daily life stressors (Sin et al., 2015).
Previous studies have also provided evidence that stress appears to induce a shift from cognitively demanding processes in the hippocampus to simultaneously enhance less demanding processing, such as amygdala-dependent processing (Cousijn et al., 2012; Pruessner et al., 2008; Schwabe et al., 2013; van Marle et al., 2009). Evidence suggests that this shift in neural pathways is mediated by cortisol via the mineralocorticoid receptor (MR) – which has also been associated with proinflammatory effects in response to increased cortisol levels (Schwabe et al., 2013; Vogel et al., 2015). A recent study discovered that high MR availability and therefore, sensitivity, may lead to increased binding of available cortisol to MR receptors and result in a dominance of less-demanding limbic responding (distress), rather than cognitively demanding responses (adaptive coping; (Vogel et al., 2015). Thus parents who are already experiencing significant stress in their daily lives are more vulnerable to self-oriented maladaptive limbic reacting (empathic distress) rather than other-oriented cognitively demanding adaptive responding (resilient, calm responding). For example, when a parent who is enduring the stress of a demanding job or caring for an elderly parent is confronted with their child in the midst of a pain-flare, they may be primed to choose the path of least resistance (e.g., keep the child home from school) to alleviate their own distress rather than navigate the cognitively and emotionally demanding path of encouraging their child to function in the face of pain which may be met with resistance. Cortisol and inflammatory markers are likely key neurobiological indices of these behaviors that directly impact child function.
4.2 Empathic stress
In addition to premorbid stress, empathetic stress related to the child's pain condition (i.e., chronic pain contagion) can trigger a cascade of neurobiological changes. Neurons particulary vulnerable to these changes are the Von Economo Neurons (VEN's) in the anterior insula and cingulate cortex that are responsible for salience detection and self-regulation (Hakeem et al., 2009) (Butti et al., 2009); (Allman et al., 2011) (Butti et al., 2013). VEN's are involved in empathy, social awareness, and self-control and in autonomic function. Some of these areas seem to be affected by emotional stressors where awareness of feelings may be altered with a condition that may induce the equivalent of a post-traumatic stress disorder. Relatedly, increased activation in anterior insula and amygdala are reported in patients prone to anxiety (Stein et al., 2007) and there are decreases in gray matter volume in the anterior cingulate and insula among sufferers of PTSD (Chao et al., 2013).
Stress may produce changes that have been considered an allostatic load that contribute to alterations in empathy. Dysregulation of neural networks through mechanisms that may include the autonomic system (Critchley et al., 2013) may be a secondary consequence of the prolonged stress on neural networks that may alter not only empathy, but also other behaviors (see below). In a paper entitled “The empathic, physiological resonance of stress” Buchanan and colleagues discuss how physiological resonance between individuals results in parallel alterations of physiological activity (e.g., cortisol levels) when observing the stress of another (Buchanan et al., 2012). An interesting study in this regard is a prospective study by Slatcher and colleagues (Slatcher et al., 2015), which demonstrated longitudinal associations between partners' perceived responsiveness (i.e., the extent to which people feel understood and cared for by their romantic partner) and diurnal cortisol profiles, suggesting that another's other-oriented empathic responses may positively impact long-term health. An interesting avenue for future research may involve the bidirectional impact of parent-child physiological resonance and pain-related interactions on parents' and children's physical health.
4.3 Empathic burnout –when stress diminishes empathetic interactions
After considering the premorbid stress that a parent brings to the context of their child's pain and the emotional and physiological resonance a parent may experience with their child; it is important to consider that continued resonance can ultimately lead to emotional burnout. Chronic ‘sharing of an emotional state’ with an offspring can lead to maladaptive changes in brain processing and emotional burnout, as high levels of empathy not only provides an advantage for recognizing others emotions, but is also associated with a bias toward negative emotions with a higher risk for empathic distress (Chikovani et al., 2015). Emotional burnout has been linked to decreased activation in the temporoparietal junction and the anterior insula coupled with a decreased ability to identify an internal emotional state (alexithymia) (Tei et al., 2014). These and other influences may alter the empathetic response in parents (Preston, 2013; Shirtcliff et al., 2009; Soria Bauser et al., 2012; Thakkar et al., 2009) including stress reactivity (Obradovic et al., 2010; Rodrigues et al., 2009; Shirtcliff et al., 2009). Such amplifying interactions may be further complicated by the emergence of adolescence among patients and the associated internal emotional reactivity that the patient themselves may be experiencing (Pfeifer et al., 2011). Taken together, the interplay of pain, stress and affective interactions are clearly important and complex (Davis et al., 2008; Ehrmantrout et al., 2011). How parental affect influences children with chronic pain may continue to drive a failing social ecosystem (McDevitt et al., 1992) of which chronic daily stress may be a major contributor (Hay and Diehl, 2010). Individuals exposed to ‘chronic empathy stress’ may thus have a sensitized brain.
4.4 Guilt and Failure
In addition to the premorbid and ongoing stress that a parent may be experiencing, many parents must grapple with the possibility that regardless of the treatment selected, their child may have persistent pain for an unknown duration. This can trigger considerable feelings of guilt and failure (Jordan et al., 2007) in the parent, particularly in circumstances when a parent is heavily involved in the child's life (e.g., helicopter parent; (Padilla-Walker and Nelson, 2012) or exerts a significant amount of parental control (van der Bruggen et al., 2008). This is coupled with an often-held belief that the pain should be eliminated, rather than managed, which in turn amplifies the suffering in the parent and child. Thus, parents seek a multitude of physician consultations and treatments, looking for a rapid fix to eliminate their (and their child's) distress and suffering. Unfortunately this cycle can be perpetuated when clinicians do not provide parents with a rational and honest explanation of treatment outcomes. The reason for this is largely because there are few effective treatments and many treatments have not received close scrutiny (i.e., Randomized Clinical Trials; see (Borsook and Kalso, 2013) and (Gilligan and Borsook, 2015). How the experience of perceived failure and guilt exerts an impact on parent brain systems remains elusive, but may undoubtedly be profound. Altogether, pre-existing life stress, ongoing empathic stress and burnout, and the guilt associated with feeling helpless in the face of a child's pain and suffering deserves further interrogation from a physiological and brain systems perspective.
5. Where do we go from here? Disengaging Negative Circles of Interaction
The interactions between the child with pain and parents can constitute a maladaptive ecosystem. Correction of such an aberrant process may necessarily require a ‘whole biome’ approach rooted in a greater understanding of the neurobiological drivers of dyfunction that inform education, acceptance, perseverance, appropriate medical and psychological treatments, in order to define progressive and realistic outcomes (Simons and Basch, 2015). In some ways part of the challenge is to provide a process that both parent and child embrace. To further the research agenda in this realm the following four concepts seem the most pertinent:
5.1 Understanding the ‘Circle of Engagement’ that leads to a worse outcome for child and parent
Parents are now recognized as integral to successful child chronic pain treatment, not only as consultants and collaborators, but also as co-clients (Simons and Basch, 2015). Moreover, in some cases the brain modulating impact of parenting a child with chronic pain may necessitate the parent to be the primary client. In both instances reversing the negative circle of interaction necessitates parents to: 1) Take stock of their prior and ongoing stressful life experiences that may calibrate their empathic responding, 2) Recognize and regulate their distress in order to avoid reacting automatically to decrease their distress levels (self-oriented) by engaging in (over)protective/pain-controlling behaviors, and 3) Respond to their child's short-term and long-term needs (other-oriented), which in the context of chronic pain requires encouraging scaffolded persistence with activities in the presence of pain to achieve a rewarding and meaningful life experience.
5.2 Garnering a better understanding of the potential changes in the child's and parent's behavior through objective measures of brain and behavioral maladaptation
Although data from self-report and behavioral observation provides evidence to support hypotheses regarding changes in brain metrics in parents, there are currently no known brain imaging studies examining parents of children with chronic pain or of the child-parent dyad to attempt to capture ‘chronic pain contagion’. Not only does current evidence suggest potential brain circuit alterations, appropriate treatments directed toward the parent may ameliorate these compromised connections. With brain imaging results demonstrating increased top-down cognitive control (i.e., enhanced pain-evoked prefrontal cortex activation) after acceptance-based treatment (Jensen et al., 2012), it is likely that these same adaptive changes may be observed in affected parents of children with chronic pain after intervention is directed toward the parent.
5.3 Restorative Pharmacological Treatments – ameliorating the stress: The Oxytocin Story
In considering potential pharmacological treatments, oxytocin may be a novel remedy for the chronic pain contagion dilemma. Oxytocin has been identified as a robust predictor of aspects of empathy including mentalizing (Feeser et al., 2015) and increasing empathic concern among individuals low in emotional empathy (Radke and de Bruijn, 2015). Moreover, oxytocin is considered a stress-buffering hormone, with pre-clinical research indicating that oxytocin administration can reverse the effects of stress on behavior, with these results more pronounced among males (Steinman et al., 2015). In a recent double-blinded, placebo-controlled, cross-over design study oxytocin attenuated pain perception in response to laser evoked potential heat pain (Paloyelis et al., 2015). Most salient to parents of children with chronic pain, a recent study demonstrated robust activation in the neural circuitry of pain (insula and sensorimotor regions) when participants observed pain in others with reductions in empathy-related brain activation (i.e., left insula) observed after intranasal oxytocin administration (Bos et al., 2015). Although the investigators acknowledge that these results are preliminary and need to be cross-validated with behavioral measures, it does stimulate an exciting line of inquiry and potential intervention that has already gained some traction in the field of psychiatry (Shamay-Tsoory and Young, 2016).
5.4 Focusing on eliminating or modulating parental maladaptive behaviors in the context of measured behavioral interventions and specific outcome evaluations
Mindfulness and acceptance-based approaches (Veehof et al., 2011) may be considered for parents who struggle with self-oriented emotional responding. Individuals who are prone to engage in self-oriented emotional responding, avoid negative thoughts, feelings, memories, physical sensations, and other internal experience in the short-term (for the momentary relief) – even when doing so can create harm in the long-run (Hayes and Wilson, 1994), due to the power of negative reinforcement. Importantly, it is not the negative thoughts, emotions, or sensations that are problematic, but how one responds that can cause life impairments. Persistent self-oriented emotional responding is considered to be the source of a wide range of problems; thus treatment approaches that encourage exposure to a broad repertoire of unpleasant internal and external stimuli, such as mindfulness and acceptance and commitment therapy can be useful. Acceptance-based approaches are grounded in solid empirical evidence for their effectiveness among children (Wicksell et al., 2009) and adults (Vowles et al., 2011; Wicksell et al., 2013).
Conclusions
The response to socio-emotional stimuli varies greatly across individuals and in the situation of how parent's behaviors are modified over time when faced with a child that continues to experience pain. Based on numerous studies of empathy, this interaction should activate neural networks of the empathizer, who is not suffering from pain but whose felt experience is profound when observing pain in their child. Moreover, a parents'responses to a child's pain may trigger an important feedback loop that can potentially be mutually brain modulating.
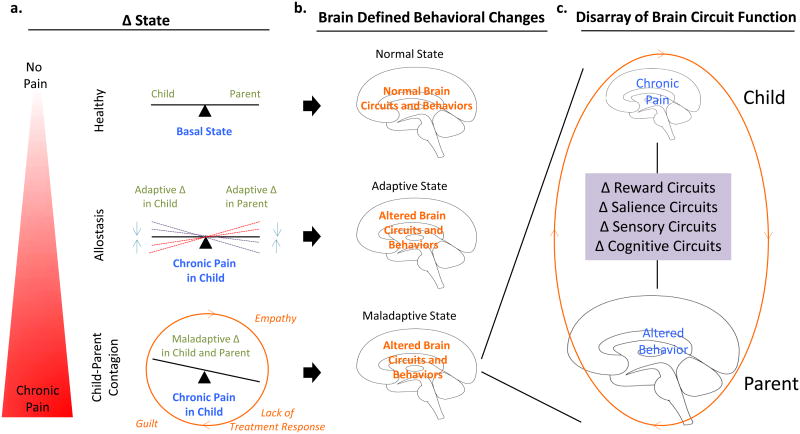
Figure 4. Failing Systems.
Conceptual model of the cumulative impact of ongoing child pain on parent brain circuit function. (a) Pain poses a change and strain to the child's basal state of equilibrium that requires adaptation to achieve allostasis. (b) When a parent's empathy, distress, and guilt overrides the system and triggers a maladaptive behavioral response pattern. (c) Multiple brain systems are altered that are perpetuated in a vicious cycle that can not only lead to further alterations in parent behavior but also contribute to child pain persistence. Modified from Borsook et al., Reward and Anti-Reward Systems in Pain Chronificaiton. Under Review.
Acknowledgments
Supported by a K23 career development award (HD067202) and R01HD08327 to LS, a K24 Mentoring Grant (NINDS NS064050) to DB, and by a Ghent University Special Research Fund grant (BOF15/24j/017) to LG and LS.
References
- Allman JM, Tetreault NA, Hakeem AY, Manaye KF, Semendeferi K, Erwin JM, Park S, Goubert V, Hof PR. The von Economo neurons in the frontoinsular and anterior cingulate cortex. Ann N Y Acad Sci. 2011;1225:59–71. doi: 10.1111/j.1749-6632.2011.06011.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avenanti A, Bueti D, Galati G, Aglioti SM. Transcranial magnetic stimulation highlights the sensorimotor side of empathy for pain. Nat Neurosci. 2005;8:955–960. doi: 10.1038/nn1481. [DOI] [PubMed] [Google Scholar]
- Bernhardt BC, Singer T. The neural basis of empathy. Annu Rev Neurosci. 2012;35:1–23. doi: 10.1146/annurev-neuro-062111-150536. [DOI] [PubMed] [Google Scholar]
- Borsook D, Edwards R, Elman I, Becerra L, Levine J. Pain and analgesia: the value of salience circuits. Prog Neurobiol. 2013a;104:93–105. doi: 10.1016/j.pneurobio.2013.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsook D, Kalso E. Transforming pain medicine: adapting to science and society. European journal of pain. 2013;17:1109–1125. doi: 10.1002/j.1532-2149.2013.00297.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsook D, Kussman BD, George E, Becerra LR, Burke DW. Surgically induced neuropathic pain: understanding the perioperative process. Ann Surg. 2013b;257:403–412. doi: 10.1097/SLA.0b013e3182701a7b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bos PA, Montoya ER, Hermans EJ, Keysers C, van Honk J. Oxytocin reduces neural activity in the pain circuitry when seeing pain in others. NeuroImage. 2015;113:217–224. doi: 10.1016/j.neuroimage.2015.03.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyle LM. A neuroplasticity hypothesis of chronic stress in the basolateral amygdala. Yale J Biol Med. 2013;86:117–125. [PMC free article] [PubMed] [Google Scholar]
- Bradshaw GA, Schore AN, Brown JL, Poole JH, Moss CJ. Elephant breakdown. Nature. 2005;433:807. doi: 10.1038/433807a. [DOI] [PubMed] [Google Scholar]
- Bruce J, Thornton AJ, Powell R, Johnston M, Wells M, Heys SD, Thompson AM, Cairns Smith W, Chambers WA, Scott NW Recovery Study, G. Psychological, surgical, and sociodemographic predictors of pain outcomes after breast cancer surgery: a population-based cohort study. Pain. 2014;155:232–243. doi: 10.1016/j.pain.2013.09.028. [DOI] [PubMed] [Google Scholar]
- Buchanan TW, Bagley SL, Stansfield RB, Preston SD. The empathic, physiological resonance of stress. Soc Neurosci. 2012;7:191–201. doi: 10.1080/17470919.2011.588723. [DOI] [PubMed] [Google Scholar]
- Butti C, Santos M, Uppal N, Hof PR. Von Economo neurons: clinical and evolutionary perspectives. Cortex. 2013;49:312–326. doi: 10.1016/j.cortex.2011.10.004. [DOI] [PubMed] [Google Scholar]
- Butti C, Sherwood CC, Hakeem AY, Allman JM, Hof PR. Total number and volume of Von Economo neurons in the cerebral cortex of cetaceans. J Comp Neurol. 2009;515:243–259. doi: 10.1002/cne.22055. [DOI] [PubMed] [Google Scholar]
- Caes L, Vervoort T, Eccleston C, Goubert L. Parents who catastrophize about their child's pain prioritize attempts to control pain. Pain. 2012;153:1695–1701. doi: 10.1016/j.pain.2012.04.028. [DOI] [PubMed] [Google Scholar]
- Caes L, Vervoort T, Eccleston C, Vandenhende M, Goubert L. Parental catastrophizing about child's pain and its relationship with activity restriction: the mediating role of parental distress. Pain. 2011;152:212–222. doi: 10.1016/j.pain.2010.10.037. [DOI] [PubMed] [Google Scholar]
- Carpenter LL, Carvalho JP, Tyrka AR, Wier LM, Mello AF, Mello MF, Anderson GM, Wilkinson CW, Price LH. Decreased adrenocorticotropic hormone and cortisol responses to stress in healthy adults reporting significant childhood maltreatment. Biological psychiatry. 2007;62:1080–1087. doi: 10.1016/j.biopsych.2007.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao L, Weiner M, Neylan T. Regional cerebral volumes in veterans with current versus remitted posttraumatic stress disorder. Psychiatry Res. 2013;213:193–201. doi: 10.1016/j.pscychresns.2013.03.002. [DOI] [PubMed] [Google Scholar]
- Chikovani G, Babuadze L, Iashvili N, Gvalia T, Surguladze S. Empathy costs: Negative emotional bias in high empathisers. Psychiatry Res. 2015;229:340–346. doi: 10.1016/j.psychres.2015.07.001. [DOI] [PubMed] [Google Scholar]
- Cousijn H, Rijpkema M, Qin S, van Wingen GA, Fernandez G. Phasic deactivation of the medial temporal lobe enables working memory processing under stress. NeuroImage. 2012;59:1161–1167. doi: 10.1016/j.neuroimage.2011.09.027. [DOI] [PubMed] [Google Scholar]
- Critchley HD. Psychophysiology of neural, cognitive and affective integration: fMRI and autonomic indicants. Int J Psychophysiol. 2009;73:88–94. doi: 10.1016/j.ijpsycho.2009.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Critchley HD, Eccles J, Garfinkel SN. Interaction between cognition, emotion, and the autonomic nervous system. Handb Clin Neurol. 2013;117:59–77. doi: 10.1016/B978-0-444-53491-0.00006-7. [DOI] [PubMed] [Google Scholar]
- Danziger N, Faillenot I, Peyron R. Can we share a pain we never felt? Neural correlates of empathy in patients with congenital insensitivity to pain. Neuron. 2009;61:203–212. doi: 10.1016/j.neuron.2008.11.023. [DOI] [PubMed] [Google Scholar]
- Davis MC, Zautra AJ, Younger J, Motivala SJ, Attrep J, Irwin MR. Chronic stress and regulation of cellular markers of inflammation in rheumatoid arthritis: implications for fatigue. Brain, behavior, and immunity. 2008;22:24–32. doi: 10.1016/j.bbi.2007.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decety J, Bartal IB, Uzefovsky F, Knafo-Noam A. Empathy as a driver of prosocial behaviour: highly conserved neurobehavioural mechanisms across species. Philos Trans R Soc Lond B Biol Sci. 2016;371 doi: 10.1098/rstb.2015.0077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decety J, Jackson PL. The functional architecture of human empathy. Behav Cogn Neurosci Rev. 2004;3:71–100. doi: 10.1177/1534582304267187. [DOI] [PubMed] [Google Scholar]
- Dedovic K, Duchesne A, Andrews J, Engert V, Pruessner JC. The brain and the stress axis: the neural correlates of cortisol regulation in response to stress. NeuroImage. 2009;47:864–871. doi: 10.1016/j.neuroimage.2009.05.074. [DOI] [PubMed] [Google Scholar]
- Derbyshire SW, Osborn J, Brown S. Feeling the pain of others is associated with self-other confusion and prior pain experience. Front Hum Neurosci. 2013;7:470. doi: 10.3389/fnhum.2013.00470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolgin MJ, Phipps S. Pediatric pain: the parents' role. Pediatrician. 1989;16:103–109. [PubMed] [Google Scholar]
- Ehrmantrout N, Allen NB, Leve C, Davis B, Sheeber L. Adolescent recognition of parental affect: influence of depressive symptoms. Journal of abnormal psychology. 2011;120:628–634. doi: 10.1037/a0022500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engen HG, Singer T. Empathy circuits. Curr Opin Neurobiol. 2013;23:275–282. doi: 10.1016/j.conb.2012.11.003. [DOI] [PubMed] [Google Scholar]
- Feeser M, Fan Y, Weigand A, Hahn A, Gartner M, Boker H, Grimm S, Bajbouj M. Oxytocin improves mentalizing - pronounced effects for individuals with attenuated ability to empathize. Psychoneuroendocrinology. 2015;53:223–232. doi: 10.1016/j.psyneuen.2014.12.015. [DOI] [PubMed] [Google Scholar]
- Feldman R. Sensitive periods in human social development: New insights from research on oxytocin, synchrony, and high-risk parenting. Dev Psychopathol. 2015;27:369–395. doi: 10.1017/S0954579415000048. [DOI] [PubMed] [Google Scholar]
- Feldman R, Vengrober A. Posttraumatic stress disorder in infants and young children exposed to war-related trauma. J Am Acad Child Adolesc Psychiatry. 2011;50:645–658. doi: 10.1016/j.jaac.2011.03.001. [DOI] [PubMed] [Google Scholar]
- Fitzgibbon BM, Enticott PG, Rich AN, Giummarra MJ, Georgiou-Karistianis N, Bradshaw JL. Mirror-sensory synaesthesia: exploring ‘shared’ sensory experiences as synaesthesia. Neurosci Biobehav Rev. 2012;36:645–657. doi: 10.1016/j.neubiorev.2011.09.006. [DOI] [PubMed] [Google Scholar]
- Fitzgibbon BM, Giummarra MJ, Georgiou-Karistianis N, Enticott PG, Bradshaw JL. Shared pain: from empathy to synaesthesia. Neurosci Biobehav Rev. 2010;34:500–512. doi: 10.1016/j.neubiorev.2009.10.007. [DOI] [PubMed] [Google Scholar]
- Gallese V. Embodied simulation: from mirror neuron systems to interpersonal relations. Novartis Found Symp. 2007;278:3–12. discussion 12-19, 89-96, 216-221. [PubMed] [Google Scholar]
- Gaughan V, Logan D, Sethna N, Mott S. Parents' perspective of their journey caring for a child with chronic neuropathic pain. Pain Manag Nurs. 2014;15:246–257. doi: 10.1016/j.pmn.2012.09.002. [DOI] [PubMed] [Google Scholar]
- Gilligan CJ, Borsook D. The Promise of Effective Pain Treatment Outcomes: Rallying Academic Centers to Lead the Charge. Pain medicine. 2015;16:1457–1466. doi: 10.1111/pme.12772. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Liencres C, Juckel G, Tas C, Friebe A, Brune M. Emotional contagion in mice: the role of familiarity. Behav Brain Res. 2014;263:16–21. doi: 10.1016/j.bbr.2014.01.020. [DOI] [PubMed] [Google Scholar]
- Goubert L, Craig KD, Vervoort T, Morley S, Sullivan MJ, de C W AC, Cano A, Crombez G. Facing others in pain: the effects of empathy. Pain. 2005;118:285–288. doi: 10.1016/j.pain.2005.10.025. [DOI] [PubMed] [Google Scholar]
- Goubert L, Simons LE. Cognitive styles and processes in paediatric pain. In: P M, B S, S W, W Z, editors. Oxford Textbook of Pediatric Pain. Oxford University Press; Oxford: 2013. pp. 95–101. [Google Scholar]
- Goubert L, Vervoort T, Craig KD. Empathy and pain. In: Gebhart RFSGF, editor. Encycopedia of Pain. Second. Springer-Verlag; Heidelberg, Germany: 2013. pp. 1128–1134. [Google Scholar]
- Goubert L, Vervoort T, Crombez G. Pain demands attention from others: the approach/avoidance paradox. Pain. 2009;143:5–6. doi: 10.1016/j.pain.2009.02.023. [DOI] [PubMed] [Google Scholar]
- Goubert L, Vlaeyen JW, Crombez G, Craig KD. Learning about pain from others: an observational learning account. The journal of pain : official journal of the American Pain Society. 2011;12:167–174. doi: 10.1016/j.jpain.2010.10.001. [DOI] [PubMed] [Google Scholar]
- Hakeem AY, Sherwood CC, Bonar CJ, Butti C, Hof PR, Allman JM. Von Economo neurons in the elephant brain. Anat Rec (Hoboken) 2009;292:242–248. doi: 10.1002/ar.20829. [DOI] [PubMed] [Google Scholar]
- Harper FW, Penner LA, Peterson A, Albrecht TL, Taub J. Children's positive dispositional attributes, parents' empathic responses, and children's responses to painful pediatric oncology treatment procedures. J Psychosoc Oncol. 2012;30:593–613. doi: 10.1080/07347332.2012.703771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauser-Cram P, Warfield ME, Shonkoff JP, Krauss MW, Sayer A, Upshur CC. Children with disabilities: a longitudinal study of child development and parent well-being. Monogr Soc Res Child Dev. 2001;66:i–viii. 1–114. discussion 115-126. [PubMed] [Google Scholar]
- Hay EL, Diehl M. Reactivity to daily stressors in adulthood: the importance of stressor type in characterizing risk factors. Psychology and aging. 2010;25:118–131. doi: 10.1037/a0018747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayes SC, Wilson KG. Acceptance and commitment therapy: Altering the verbal support for experiential avoidance. The Behavior analyst / MABA. 1994;17:289–303. doi: 10.1007/BF03392677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hein G, Singer T. I feel how you feel but not always: the empathic brain and its modulation. Curr Opin Neurobiol. 2008;18:153–158. doi: 10.1016/j.conb.2008.07.012. [DOI] [PubMed] [Google Scholar]
- Higgins KS, Birnie KA, Chambers CT, Wilson AC, Caes L, Clark AJ, Lynch M, Stinson J, Campbell-Yeo M. Offspring of parents with chronic pain: a systematic review and meta-analysis of pain, health, psychological, and family outcomes. Pain. 2015;156:2256–2266. doi: 10.1097/j.pain.0000000000000293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaremka LM, Fagundes CP, Peng J, Bennett JM, Glaser R, Malarkey WB, Kiecolt-Glaser JK. Loneliness promotes inflammation during acute stress. Psychol Sci. 2013;24:1089–1097. doi: 10.1177/0956797612464059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen KB, Kosek E, Wicksell R, Kemani M, Olsson G, Merle JV, Kadetoff D, Ingvar M. Cognitive Behavioral Therapy increases pain-evoked activation of the prefrontal cortex in patients with fibromyalgia. Pain. 2012;153:1495–1503. doi: 10.1016/j.pain.2012.04.010. [DOI] [PubMed] [Google Scholar]
- Jordan A, Crabtree A, Eccleston C. ‘You have to be a jack of all trades’: Fathers parenting their adolescent with chronic pain. J Health Psychol. 2015 doi: 10.1177/1359105315580461. [DOI] [PubMed] [Google Scholar]
- Jordan A, Eccleston C, McCracken LM, Connell H, Clinch J. The Bath Adolescent Pain--Parental Impact Questionnaire (BAP-PIQ): development and preliminary psychometric evaluation of an instrument to assess the impact of parenting an adolescent with chronic pain. Pain. 2008;137:478–487. doi: 10.1016/j.pain.2007.10.007. [DOI] [PubMed] [Google Scholar]
- Jordan AL, Eccleston C, Osborn M. Being a parent of the adolescent with complex chronic pain: an interpretative phenomenological analysis. European journal of pain. 2007;11:49–56. doi: 10.1016/j.ejpain.2005.12.012. [DOI] [PubMed] [Google Scholar]
- Lamm C, Bukowski H, Silani G. From shared to distinct self-other representations in empathy: evidence from neurotypical function and socio-cognitive disorders. Philos Trans R Soc Lond B Biol Sci. 2016;371 doi: 10.1098/rstb.2015.0083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamm C, Decety J, Singer T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. NeuroImage. 2011;54:2492–2502. doi: 10.1016/j.neuroimage.2010.10.014. [DOI] [PubMed] [Google Scholar]
- Langford DJ, Crager SE, Shehzad Z, Smith SB, Sotocinal SG, Levenstadt JS, Chanda ML, Levitin DJ, Mogil JS. Social modulation of pain as evidence for empathy in mice. Science. 2006;312:1967–1970. doi: 10.1126/science.1128322. [DOI] [PubMed] [Google Scholar]
- Leigh R, Oishi K, Hsu J, Lindquist M, Gottesman RF, Jarso S, Crainiceanu C, Mori S, Hillis AE. Acute lesions that impair affective empathy. Brain. 2013;136:2539–2549. doi: 10.1093/brain/awt177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linnman C, Becerra L, Lebel A, Berde C, Grant PE, Borsook D. Transient and persistent pain induced connectivity alterations in pediatric complex regional pain syndrome. PloS one. 2013;8:e57205. doi: 10.1371/journal.pone.0057205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logan DE, Simons LE, Carpino EA. Too sick for school? Parent influences on school functioning among children with chronic pain. Pain. 2012;153:437–443. doi: 10.1016/j.pain.2011.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch-Jordan AM, Kashikar-Zuck S, Szabova A, Goldschneider KR. The interplay of parent and adolescent catastrophizing and its impact on adolescents' pain, functioning, and pain behavior. Clin J Pain. 2013;29:681–688. doi: 10.1097/AJP.0b013e3182757720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin LJ, Tuttle AH, Mogil JS. The interaction between pain and social behavior in humans and rodents. Curr Top Behav Neurosci. 2014;20:233–250. doi: 10.1007/7854_2014_287. [DOI] [PubMed] [Google Scholar]
- McDevitt D, Vaudaux P, Foster TJ. Genetic evidence that bound coagulase of Staphylococcus aureus is not clumping factor. Infection and immunity. 1992;60:1514–1523. doi: 10.1128/iai.60.4.1514-1523.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwen BS. The Brain on Stress: Toward an Integrative Approach to Brain, Body, and Behavior. Perspect Psychol Sci. 2013;8:673–675. doi: 10.1177/1745691613506907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mora F, Segovia G, Del Arco A, de Blas M, Garrido P. Stress, neurotransmitters, corticosterone and body-brain integration. Brain Res. 2012;1476:71–85. doi: 10.1016/j.brainres.2011.12.049. [DOI] [PubMed] [Google Scholar]
- Noel M, Beals-Erickson SE, Law EF, Alberts N, Palermo TM. Characterizing the Pain Narratives of Parents of Youth with Chronic Pain. Clin J Pain. 2016 doi: 10.1097/AJP.0000000000000346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obradovic J, Bush NR, Stamperdahl J, Adler NE, Boyce WT. Biological sensitivity to context: the interactive effects of stress reactivity and family adversity on socioemotional behavior and school readiness. Child development. 2010;81:270–289. doi: 10.1111/j.1467-8624.2009.01394.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira VC, Ferreira ML, Morso L, Albert HB, Refshauge KM, Ferreira PH. Patients' perceived level of social isolation affects the prognosis of low back pain. European journal of pain. 2015;19:538–545. doi: 10.1002/ejp.578. [DOI] [PubMed] [Google Scholar]
- Padilla-Walker LM, Nelson LJ. Black Hawk down? Establishing helicopter parenting as a distinct construct from other forms of parental control during emerging adulthood. Journal of adolescence. 2012;35:1177–1190. doi: 10.1016/j.adolescence.2012.03.007. [DOI] [PubMed] [Google Scholar]
- Palermo TM, Eccleston C, Lewandowski AS, Williams AC, Morley S. Randomized controlled trials of psychological therapies for management of chronic pain in children and adolescents: an updated meta-analytic review. Pain. 2010;148:387–397. doi: 10.1016/j.pain.2009.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palermo TM, Valrie CR, Karlson CW. Family and parent influences on pediatric chronic pain: A developmental perspective. The American psychologist. 2014;69:142–152. doi: 10.1037/a0035216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paloyelis Y, Krahe C, Maltezos S, Williams SC, Howard MA, Fotopoulou A. The Analgesic Effect Of Oxytocin In Humans: A Double-Blinded Placebo Controlled Cross-Over Study Using Laser-Evoked Potentials. Journal of neuroendocrinology. 2015 doi: 10.1111/jne.12347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panksepp J. Affective consciousness: Core emotional feelings in animals and humans. Conscious Cogn. 2005;14:30–80. doi: 10.1016/j.concog.2004.10.004. [DOI] [PubMed] [Google Scholar]
- Panksepp J, Panksepp JB. Toward a cross-species understanding of empathy. Trends Neurosci. 2013;36:489–496. doi: 10.1016/j.tins.2013.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penner LA, Cline RJ, Albrecht TL, Harper FW, Peterson AM, Taub JM, Ruckdeschel JC. Parents' Empathic Responses and Pain and Distress in Pediatric Patients. Basic Appl Soc Psych. 2008;30:102–113. doi: 10.1080/01973530802208824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeifer JH, Masten CL, Moore WE, 3rd, Oswald TM, Mazziotta JC, Iacoboni M, Dapretto M. Entering adolescence: resistance to peer influence, risky behavior, and neural changes in emotion reactivity. Neuron. 2011;69:1029–1036. doi: 10.1016/j.neuron.2011.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston SD. The origins of altruism in offspring care. Psychological bulletin. 2013;139:1305–1341. doi: 10.1037/a0031755. [DOI] [PubMed] [Google Scholar]
- Preston SD, de Waal FB. Empathy: Its ultimate and proximate bases. Behav Brain Sci. 2002;25:1–20. doi: 10.1017/s0140525x02000018. discussion 20-71. [DOI] [PubMed] [Google Scholar]
- Pruessner JC, Dedovic K, Khalili-Mahani N, Engert V, Pruessner M, Buss C, Renwick R, Dagher A, Meaney MJ, Lupien S. Deactivation of the limbic system during acute psychosocial stress: evidence from positron emission tomography and functional magnetic resonance imaging studies. Biological psychiatry. 2008;63:234–240. doi: 10.1016/j.biopsych.2007.04.041. [DOI] [PubMed] [Google Scholar]
- Radke S, de Bruijn ER. Does oxytocin affect mind-reading? A replication study. Psychoneuroendocrinology. 2015;60:75–81. doi: 10.1016/j.psyneuen.2015.06.006. [DOI] [PubMed] [Google Scholar]
- Riva R, Forinder U, Arvidson J, Mellgren K, Toporski J, Winiarski J, Norberg AL. Patterns of psychological responses in parents of children that underwent stem cell transplantation. Psychooncology. 2014;23:1307–1313. doi: 10.1002/pon.3567. [DOI] [PubMed] [Google Scholar]
- Rizzolatti G, Craighero L. The mirror-neuron system. Annu Rev Neurosci. 2004;27:169–192. doi: 10.1146/annurev.neuro.27.070203.144230. [DOI] [PubMed] [Google Scholar]
- Rodrigues SM, Saslow LR, Garcia N, John OP, Keltner D. Oxytocin receptor genetic variation relates to empathy and stress reactivity in humans. Proceedings of the National Academy of Sciences of the United States of America. 2009;106:21437–21441. doi: 10.1073/pnas.0909579106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwabe L, Tegenthoff M, Hoffken O, Wolf OT. Mineralocorticoid receptor blockade prevents stress-induced modulation of multiple memory systems in the human brain. Biological psychiatry. 2013;74:801–808. doi: 10.1016/j.biopsych.2013.06.001. [DOI] [PubMed] [Google Scholar]
- Shamay-Tsoory S, Young LJ. Understanding the Oxytocin System and Its Relevance to Psychiatry. Biological psychiatry. 2016;79:150–152. doi: 10.1016/j.biopsych.2015.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamay-Tsoory SG. The neural bases for empathy. The Neuroscientist : a review journal bringing neurobiology, neurology and psychiatry. 2011;17:18–24. doi: 10.1177/1073858410379268. [DOI] [PubMed] [Google Scholar]
- Shirtcliff EA, Vitacco MJ, Graf AR, Gostisha AJ, Merz JL, Zahn-Waxler C. Neurobiology of empathy and callousness: implications for the development of antisocial behavior. Behavioral sciences & the law. 2009;27:137–171. doi: 10.1002/bsl.862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieberg CB, Williams S, Simons LE. Do parent protective responses mediate the relation between parent distress and child functional disability among children with chronic pain? J Pediatr Psychol. 2011;36:1043–1051. doi: 10.1093/jpepsy/jsr043. [DOI] [PubMed] [Google Scholar]
- Simons LE, Basch MC. State of the art in biobehavioral approaches to the management of chronic pain in childhood. Pain management. 2015 doi: 10.2217/pmt.15.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons LE, Smith A, Kaczynski K, Basch M. Living in fear of your child's pain: the Parent Fear of Pain Questionnaire. Pain. 2015;156:694–702. doi: 10.1097/j.pain.0000000000000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sin NL, Graham-Engeland JE, Ong AD, Almeida DM. Affective reactivity to daily stressors is associated with elevated inflammation. Health Psychol. 2015;34:1154–1165. doi: 10.1037/hea0000240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slatcher RB, Selcuk E, Ong AD. Perceived Partner Responsiveness Predicts Diurnal Cortisol Profiles 10 Years Later. Psychol Sci. 2015;26:972–982. doi: 10.1177/0956797615575022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soria Bauser D, Thoma P, Suchan B. Turn to me: electrophysiological correlates of frontal vs. averted view face and body processing are associated with trait empathy. Frontiers in integrative neuroscience. 2012;6:106. doi: 10.3389/fnint.2012.00106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sripada RK, Swain JE, Evans GW, Welsh RC, Liberzon I. Childhood poverty and stress reactivity are associated with aberrant functional connectivity in default mode network. Neuropsychopharmacology. 2014;39:2244–2251. doi: 10.1038/npp.2014.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein MB, Simmons AN, Feinstein JS, Paulus MP. Increased amygdala and insula activation during emotion processing in anxiety-prone subjects. Am J Psychiatry. 2007;164:318–327. doi: 10.1176/ajp.2007.164.2.318. [DOI] [PubMed] [Google Scholar]
- Steinman MQ, Duque-Wilckens N, Greenberg GD, Hao R, Campi KL, Laredo SA, Laman-Maharg A, Manning CE, Doig IE, Lopez EM, Walch K, Bales KL, Trainor BC. Sex-Specific Effects of Stress on Oxytocin Neurons Correspond With Responses to Intranasal Oxytocin. Biological psychiatry. 2015 doi: 10.1016/j.biopsych.2015.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tei S, Becker C, Kawada R, Fujino J, Jankowski KF, Sugihara G, Murai T, Takahashi H. Can we predict burnout severity from empathy-related brain activity? Translational psychiatry. 2014;4:e393. doi: 10.1038/tp.2014.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thakkar KN, Brugger P, Park S. Exploring empathic space: correlates of perspective transformation ability and biases in spatial attention. PloS one. 2009;4:e5864. doi: 10.1371/journal.pone.0005864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vachon-Presseau E, Martel MO, Roy M, Caron E, Jackson PL, Rainville P. The multilevel organization of vicarious pain responses: effects of pain cues and empathy traits on spinal nociception and acute pain. Pain. 2011;152:1525–1531. doi: 10.1016/j.pain.2011.02.039. [DOI] [PubMed] [Google Scholar]
- Vaisvaser S, Lin T, Admon R, Podlipsky I, Greenman Y, Stern N, Fruchter E, Wald I, Pine DS, Tarrasch R, Bar-Haim Y, Hendler T. Neural traces of stress: cortisol related sustained enhancement of amygdala-hippocampal functional connectivity. Front Hum Neurosci. 2013;7:313. doi: 10.3389/fnhum.2013.00313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Bruggen CO, Stams GJ, Bogels SM. Research review: the relation between child and parent anxiety and parental control: a meta-analytic review. Journal of child psychology and psychiatry, and allied disciplines. 2008;49:1257–1269. doi: 10.1111/j.1469-7610.2008.01898.x. [DOI] [PubMed] [Google Scholar]
- van Marle HJ, Hermans EJ, Qin S, Fernandez G. From specificity to sensitivity: how acute stress affects amygdala processing of biologically salient stimuli. Biological psychiatry. 2009;66:649–655. doi: 10.1016/j.biopsych.2009.05.014. [DOI] [PubMed] [Google Scholar]
- Vandenbroucke S, Crombez G, Loeys T, Goubert L. Observing another in pain facilitates vicarious experiences and modulates somatosensory experiences. Front Hum Neurosci. 2014;8:631. doi: 10.3389/fnhum.2014.00631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veehof MM, Oskam MJ, Schreurs KM, Bohlmeijer ET. Acceptance-based interventions for the treatment of chronic pain: a systematic review and meta-analysis. Pain. 2011;152:533–542. doi: 10.1016/j.pain.2010.11.002. [DOI] [PubMed] [Google Scholar]
- Veer IM, Oei NY, Spinhoven P, van Buchem MA, Elzinga BM, Rombouts SA. Endogenous cortisol is associated with functional connectivity between the amygdala and medial prefrontal cortex. Psychoneuroendocrinology. 2012;37:1039–1047. doi: 10.1016/j.psyneuen.2011.12.001. [DOI] [PubMed] [Google Scholar]
- Vervoort T, Trost Z, Sutterlin S, Caes L, Moors A. Emotion regulatory function of parent attention to child pain and associated implications for parental pain control behaviour. Pain. 2014;155:1453–1463. doi: 10.1016/j.pain.2014.04.015. [DOI] [PubMed] [Google Scholar]
- Vogel S, Klumpers F, Krugers HJ, Fang Z, Oplaat KT, Oitzl MS, Joels M, Fernandez G. Blocking the mineralocorticoid receptor in humans prevents the stress-induced enhancement of centromedial amygdala connectivity with the dorsal striatum. Neuropsychopharmacology. 2015;40:947–956. doi: 10.1038/npp.2014.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vowles KE, McCracken LM, O'Brien JZ. Acceptance and values-based action in chronic pain: a three-year follow-up analysis of treatment effectiveness and process. Behaviour research and therapy. 2011;49:748–755. doi: 10.1016/j.brat.2011.08.002. [DOI] [PubMed] [Google Scholar]
- Wicksell RK, Kemani M, Jensen K, Kosek E, Kadetoff D, Sorjonen K, Ingvar M, Olsson GL. Acceptance and commitment therapy for fibromyalgia: a randomized controlled trial. European journal of pain. 2013;17:599–611. doi: 10.1002/j.1532-2149.2012.00224.x. [DOI] [PubMed] [Google Scholar]
- Wicksell RK, Melin L, Lekander M, Olsson GL. Evaluating the effectiveness of exposure and acceptance strategies to improve functioning and quality of life in longstanding pediatric pain--a randomized controlled trial. Pain. 2009;141:248–257. doi: 10.1016/j.pain.2008.11.006. [DOI] [PubMed] [Google Scholar]
- Wiseman H, Metzl E, Barber JP. Anger, guilt, and intergenerational communication of trauma in the interpersonal narratives of second generation Holocaust survivors. Am J Orthopsychiatry. 2006;76:176–184. doi: 10.1037/0002-9432.76.2.176. [DOI] [PubMed] [Google Scholar]