Abstract
This work investigated the microbial toxicity of soluble species that can potentially be leached from the II–VI semiconductor materials, cadmium telluride and cadmium selenide. The soluble ions tested included: cadmium, selenite, selenate, tellurite, and tellurate. Their toxicity towards the acetoclastic and hydrogen-consuming trophic groups in a methanogenic consortium as well as towards a bioluminescent marine bacterium, Aliivibrio fischeri (Microtox® test), was assessed. The acetoclastic methanogenic activity was the most affected as evidenced by the low 50% inhibiting concentrations (IC50) values obtained of 8.6 mg L−1 for both cadmium and tellurite, 10.2 mg L−1 for tellurate, and 24.1 mg L−1 for selenite. Both tellurium oxyanions caused a strong inhibition of acetoclastic methanogenesis at low concentrations, each additional increment in concentration provided progressively less inhibition increase. In the case of the hydrogenotrophic methanogenesis, cadmium followed by selenite caused the greatest inhibition with IC50 values of 2.9 and 18.0 mg L−1, respectively. Tellurite caused a moderate effect as evidenced by a 36.8% inhibition of the methanogenic activity at the highest concentration tested, and a very mild effect of tellurate was observed. Microtox® analyses showed a noteworthy inhibition of cadmium, selenite, and tellurite with 50% loss in bioluminescence after 30 min of exposure of 5.5, 171.1, and 458.6 mg L−1, respectively. These results suggest that the leaching of cadmium, tellurium and selenium ions from semiconductor materials can potentially cause microbial toxicity.
Keywords: II–VI semiconductors, chalcogens, methanogenic toxicity, Microtox®, microbial toxicity, Aliivibrio fischeri
Graphical Abstract
1. Introduction
Cadmium telluride (CdTe) and cadmium selenide (CdSe) are two semiconductors of the II–VI group. Due to their remarkable optical and electrical properties, both compounds are extensively used in the manufacture of electronic devices. CdSe is a good light absorber which possesses excellent photoelectrical characteristics (Feng et al., 2010); therefore, it is used in the production of light emitting diodes (LEDs), photo-electronics, and transistors (Chate et al., 2013). The use of CdSe in the production of high efficiency hybrid solar cells has been investigated (Feng et al., 2010;Chate et al., 2013;Huynh et al., 2002). Likewise, CdTe is used in the production of optoelectronic devices, gamma ray detectors (Bicknell et al., 1987), and laser windows (Punitha et al., 2015). Most importantly, CdTe is used in the photovoltaic industry in the production of thin film solar cells. CdTe photovoltaic devices were ranked as the third most common type of photovoltaic solar panels commercially available in 2013 (Esterly, 2013).
Increasing concerns have arisen due to the implications of the potential release of hazardous substances from CdTe and CdSe containing devices. Firstly, it is expected that the manufacture of electronics based on these semiconductors will grow over time as technology moves forward which implies an increasing release of these compounds in the environment. Specifically, the production of solar panels is expected to increase since solar energy is one the fastest growing market shares of renewable energy (Century, 2015). Despite the remarkable efforts that are being made to recycle electronic waste, it is possible that an important fraction of the decommissioned solar panels in the future will end up discarded in municipal mixed landfills as a result of a lack of regulations related to the disposal of electronic waste in multiple countries. Secondly, there is evidence that toxic compounds might be leached from electronics based on leaching experiments using deionized water as the extraction fluid (Lithner et al., 2012). More importantly, recent works have demonstrated that soluble ions of Cd, Se, and Te, such as divalent cadmium (CdII), selenite (SeO32− (SeIV)), selenate (SeO42−, (SeVI)), tellurite (TeO32− (TeIV)), and tellurate (TeO42− (TeVI)) can leach out from CdSe and CdTe under conditions similar to those commonly found in landfills (Zeng et al., 2015). Thirdly, Cd and selenium (Se) are highly toxic elements which are included in the United States Environmental Protection Agency (USEPA) list of regulated drinking water contaminants, and the disposal of Se and Cd containing waste in municipal solid waste landfills is also regulated. The maximum contaminant levels (MCL) established for Cd and Se are 0.005 mg L−1 and 0.05 mg L−1, respectively, and the toxicity characteristic leaching procedure (TCLP) limit established for Se and Cd is 1 mg L−1. Furthermore, Cd, Se and Te soluble fractions are highly toxic to some microorganisms (Trevors et al., 1986; Macken et al., 2009; Yu et al., 1997; Taylor, 1999). Given these considerations, the presence of soluble ions derived from CdTe and CdSe in the environment might negatively impact several important processes, such as, the anaerobic biodegradation of organic matter and the activity of aquatic organisms.
The aim of this work was to investigate the toxicity of the soluble species potentially released from CdSe and CdTe (CdII, SeIV, SeVI, TeIV and TeVI) towards an anaerobic methanogenic consortium to assess microorganisms involved in anaerobic digestion processes and the Microtox® tests that measure the bioluminescence of the marine bacterium, Aliivibrio fischeri, which is commonly used to assess aquatic toxicity.
2. Materials and methods
2.1. Chemicals
CdII as Cd(OH)2 (≥ 99.99% purity), TeIV as Na2TeO3, and TeVI as K2TeO4•xH2O (x = 0.5) were purchased from Sigma-Aldrich (St. Louis, MO, USA). The stoichiometry of the molecular water of K2TeO4·xH2O was determined by comparing the Te concentration of a TeO42− salt solution against a Te standard (described below). SeIV as Na2SeO3 was acquired from MP Biomedicals (Santa Ana, CA, USA), SeVI as Na2SeO4 (≥ 98% purity) was obtained from ACROS Organics (Geel, Belgium), and the Te standard (1000 mg L−1) was purchased from RICCA Chemical Company (Arlington, TX, USA). All the Microtox® reagents were obtained from ModernWater (Cambridge, UK).
2.2. Inoculum source
An anaerobic granular sludge was obtained and used as the source of inoculum from a full-scale upflow anaerobic sludge blanket reactor treating effluent at Mahou beer brewery (in Guadalajara, Spain). This granular sludge contained 0.0792 g volatile suspended solids (VSS) g−1 wet wt. The maximum methanogenic activities of the sludge were 566.8±64 and 571±26 mg chemical oxygen demand-methane (COD-CH4) g VSS−1 day−1for the assays utilizing acetate and hydrogen as substrate, respectively. The granular sludge was stored at 4°C prior to use. The marine bacterium Aliivibrio fischeri (lyophilized culture of A. fischeri NRRL-B-11177), was obtained from ModernWater (Cambridge, UK).
2.3. Methanogenic toxicity bioassays
2.3.1. Mineral media
The basal medium (M1) used in the experiments assessing the toxicity of the chemical compounds towards acetate-consuming microorganisms contained (in mg L−1 in final medium): CH3COONa (2563), K2HPO4 (250), CaCl2•2H2O (10), MgSO4•7H2O (100), MgCl2•6H2O (100), NH4Cl (280), yeast extract (100), and 1 mL L−1 of a trace elements stock solution described below. The pH was subsequently adjusted to 7.0–7.2, and the medium was finally amended with NaHCO3 to a concentration of 4 g L−1. The basal medium (M2) used in the assays designed to study the toxicity of the chemical compounds to hydrogen-consuming microorganisms consisted of (in mg L−1 in final medium): NH4Cl (280), K2HPO4 (250), CaCl2•2H2O (10), MgCl2•6H2O (183), yeast extract (100) and 1 mL L−1 of a trace elements stock solution. After adjusting the pH to 7.0–7.2, NaHCO3 was added to a final concentration of 3 g L−1. H2 was added via the gas phase (as described below). The trace elements stock solution for both media consisted of (in mg L−1): H3BO3 (50), FeCl2•4H2O (2000), ZnCl2 (50), (NH4)6Mo7O24•4H2O (50), AlCl3•6H2O (90), CoCl2•6H2O (2000), NiCl2•6H2O (50), CuCl2•2H2O (30), NaSeO3•5H2O (100), EDTA (1000), resazurin (200), and 1 mL of HCl (37% wt). CdII and TeVI stock solutions were prepared in demineralized (DI) and acidified water (10 mM HCl), the rest of the salts were diluted in DI water.
2.3.2. Batch bioassays
Methanogenic inhibition assays were carried out in 160 mL glass serum flasks (Wheaton, Milville, NJ, USA). The experimental set up consisted of 25 mL of liquid and 135 mL of gas (headspace) in the bottles. Firstly, 20 mL of 1.25× concentrated mineral medium M1 or M2 (according to assay purpose) along with 1.5g of VSS L−1 of sludge inoculum were added to the serum flasks. After closing the bottles with rubber septa and aluminum crimp seals, the headspaces of the bottles were flushed with N2:CO2 (80:20, v/v) for four minutes to eliminate oxygen. Either sodium acetate (originally amended in the 20 mL of the 1.25× concentrated mineral medium M1 to give a final concentration of 2.5 g COD L−1 in the 25 mL of liquid) or hydrogen (H2) were used as substrates depending on the experiment. H2 was supplied afterwards with a H2/CO2 gas mixture (80:20, v/v) to reach an overpressure of 0.5 atm to the corresponding bottles (final concentration of 1.83 g COD as H2 in headspace L−1liq in 20 mL of 1.25× medium M2, and 140 mL of headspace). The flasks were preincubated overnight in an orbital shaker at 100 rpm in a climate controlled room at 30°C for the adaptation of the sludge to the media.
After the pre incubation period, the bottles were amended with 5 mL of different amounts of the correspondent stock solutions of test chemicals and DI water to reach the desired initial concentration of the compound of interest, and controls were set up using 5 mL of DI water lacking additions of the inhibitory test chemicals. Table S1 summarizes the experimental concentration ranges utilized in the different toxicity bioassays. Subsequently, all bottles were flushed again with the mixture N2/CO2 (80:20, v/v), and H2 was provided to the appropriate (with H2 as the substrate) bottles using H2/CO2 gas mixture (80:20, v/v). The final added substrate concentrations were 2.0 g COD L−1 as acetate or 1.41 g COD as H2 gas L−1liq after completing the dilution corresponding to 25 mL medium and 135 mL of headspace. The controls were carried out in triplicate and the treatments were performed in duplicate.
All the assays were incubated at 30±2°C in an orbital shaker at 100 rpm. In order to monitor the production of methane, gas samples of 100 μL were collected from the headspace of the bottles and analyzed (as described below) every two hours during the first eight to ten hours of incubation, and after that, two or three times per day until the maximum theoretical methane production was reached. The maximum specific methanogenic activity for each concentration tested was obtained from the slope of the cumulative methane produced as a function of time. These values were normalized with respect to the maximum specific activity of the corresponding uninhibited control. The time period used to calculate the maximum activity for each initial concentration was a discrete interval shared by all treatments in each experiment. The time interval used in each case is shown in Table S1 in supplementary information (SI). The normalized methanogenic activities (NMA) were calculated as follows:
The initial concentrations of the compounds that caused a 20%, 50% and 80% decrease in the methanogenic activity compared to the uninhibited control (IC20, IC50 and IC80) were estimated as described elsewhere (Tapia-Rodriguez et al., 2012).
2.4. Microtox®
The acute toxic effect of CdII, TeIV, TeVI, SeIV and SeVI on the bioluminescent marine bacteria A. fischeri was assessed using a Microtox® Model 500 analyzer (Strategic Diagnostics, Inc. SDIX, Newark, DE, USA). Microbial inhibition was measured at 25°C in triplicate experiments. Table S1 shows the range of concentration used for each chemical compound. The concentrations causing 50% decrease in the bacterial luminescence (IC50), compared to the toxicant-free control, after 5, 15, and 30 min of exposure were obtained as previously described (Bulich and Isenberg, 1981).
2.5. Analytical methods
2.5.1. Methane determination
Methane content in the gas phase of the serum flasks was analyzed by gas chromatography using a HP 5890 Series II system (Agilent Technologies, Palo Alto, CA, USA) equipped with a flame ionization detector and a Stabilwax-DA fused silica capillary column (30 m length x 0.53 mm ID, Restek Corporation, Bellefonte, PA, USA). Gas samples of 100 μL were injected using Helium as the carrier gas at a flow rate of 85 mL min−1. The temperatures of the oven, injection port and detector were 140, 180 and 250°C, respectively.
3. Results
3.1. Methanogenic inhibition
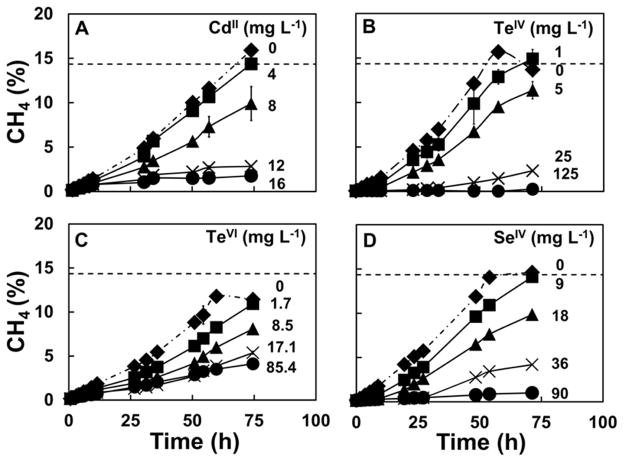
Figure 1 shows the time course of methane production in the assays evaluating acetoclastic methanogenesis in response to being exposed to a gradient of CdII, TeIV, TeVI and SeIV concentrations. The control assays rapidly produced methane reaching the theoretically maximum expected concentration of 14.4% CH4 in the headspace within approximately 60 h. As the concentrations of the potential II/VI semiconductor ions, CdII, TeIV, TeVI and SeIV, were increased, the slopes of the methane production curves clearly decreased as a function of the increasing ion concentration. This relationship was consistent up to a point when the toxicity was too severe. At concentrations of 12 to 16 mg L−1 CdII and 36 mg L−1 SeIV (Figures 1A and D), there was initially some methane production, but the production subsequently slowed down and stopped during the assay. Higher concentrations (90 mg L−1 SeIV and 125 mg L−1 TeIV in Figures 1B and D) caused almost complete inhibition from the very start of the assays. TeVI was also found to be inhibitory at low concentrations; however, only partial inhibition was observed even at the highest concentration used in the assays (Figure 1C). The methane production rate even recovered with an extended incubation time (Figure S1) in the SI section.
Fig 1.
Time course of methane production, expressed as cumulative percentage of methane (% CH4), in the headspace of acetoclastic methanogenic bioassays in the presence of increasing initial concentrations of Cd, Te and Se ions. Legends: (- - -) Theoretical methane production. Panel A, CdII (in mg L−1): 0 (◆), 4 (■), 8 (▲), 12 (×), and 16 (●). Panel B, TeIV (in mg L−1): 0 (◆), 1 (■), 5 (▲), 25 (×), 125 (●). Panel C, TeVI (in mg L−1): 0 (◆), 1.7 (■), 8.5 (▲), 17.1 (×), 85.4 (●). Panel D, SeIV (in mg L−1): 0 (◆), 9 (■), 18 (▲), 36 (×), 90 (●).
The toxicity of SeVI was also tested, but it was not effective in causing inhibition to acetoclastic methanogenic activity (data not shown). Partial inhibition was only observed at environmentally irrelevant concentrations of 4.2 g L−1 SeVI (data not shown).
Figure S2 in SI shows the time course of methane production assays evaluating hydrogenotrophic methanogensis in response to being exposed to a gradient of CdII and SeIV concentrations. The control assays rapidly produced methane reaching the theoretically maximum expected concentration of 10.2% CH4 in the headspace after approximately 60 h. As the concentrations of the potential II/VI semiconductor ions, CdII, and SeIV, were increased, the slopes of the methane production curves decreased as a function of the increasing ion concentration (similar to the acetoclastic methanogenic assay). The inhibitory response was particularly severe between 2 and 5 mg L−1 (Figure S2A in SI). As the concentration reached 12 mg L−1 CdII or 40 mg L−1 SeIV (Figure S2B in SI), the inhibition impact was so severe that the methane production which occurred in the beginning of the assay came to a total standstill before the incubation was complete. TeVI was tested and had no significant toxic effect at the highest concentration used in this experiment (500 mg TeVI L−1, see Figure S3 in SI),. High concentrations of SeVI (4.2 g SeVI L−1, data not shown) caused only partial inhibiton. For TeIV, the inhibitory response was particularly important at 12.5 mg TeIV L−1 however, above that concentration, the toxicity response was maximally 37% and further increases in TeIV concentration did not increase the toxicity further.
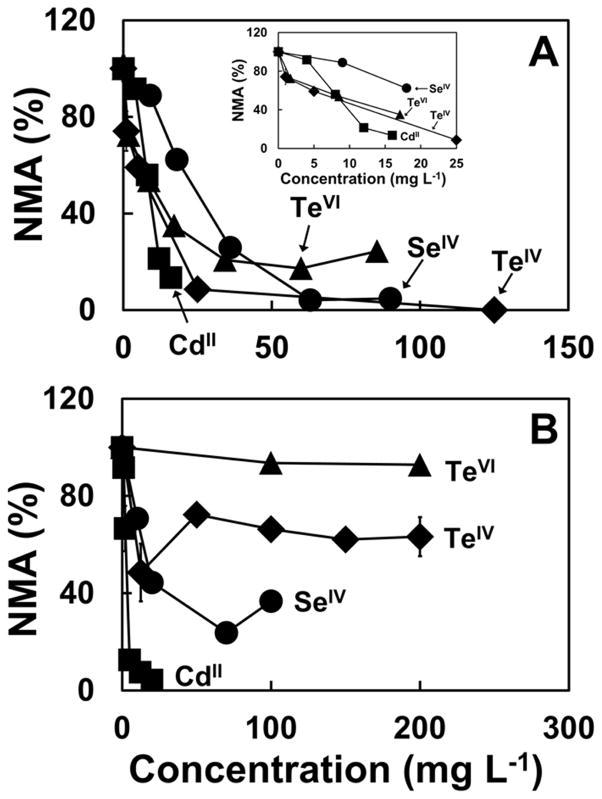
Figure 2 shows the normalized maximum activity (NMA) of the methanogenic anaerobic sludge as a function of the initial concentrations of the compounds utilized in this study, relative to the maximum activity of the control without added toxic compounds. Figure 2A presents the most relevant results for the assays where the acetoclastic activity was studied. CdII, TeIV, TeVI and SeIV all caused concentration related toxicity responses to acetoclastic methanogenic activity in the granular sludge. In all cases, severe inhibitions (> 80%) were evident at concentrations less than 50 mg L−1. There were however noticeable differences in the response pattern.
Fig 2.
Normalized specific methanogenic activity (NMA) as a percent of control activity in acetoclastic (A) and hydrogenotrophic (B) methanogenic experiments as a function of different initial concentrations of Cd, Te or Se ions. Legends: CdII (■), TeIV (◆), TeVI (▲), SeIV (●).
The toxicity response curve to CdII concentrations was the steepest of all the ions tested. CdII concentrations as low as 12 mg L−1 caused around 80% inhibition. However, the inhibition response to CdII was less steep from 0 to 4 mg L−1 most likely due to precipitation of CdII with biogenic sulfide formed from the 39.0 mg L−1 of sulfate (SO42−) in the medium, M1. The Te oxyanions provided a sharp response to the methanogenic acetoclastic activity at low concentrations causing greater than 25% inhibition at the lowest concentration tested of 1 mg L−1 and 1.7 mg L−1 for TeIV and TeVI, respectively. However, each additional increment in concentration provided progressively less toxicity response (pattern of “diminishing returns”). In order to achieve 80% or more inhibition, relatively high concentrations of the Te oxyanions (20.5 and 39.7 mg L−1 for TeIV and TeVI, respectively) were required. This concentration was 3.3 × higher than that needed by CdII for a similar level of inhibition. The response pattern for SeIV was intermediate between that of CdII and the Te oxyanions.
Figure 2B presents the most relevant results for the hydrogenotrophic methanogenic experiments. As was observed with the acetoclastic methanogenic activity, the hydrogenotrophic microorganisms were inhibited by CdII in a steep linear fashion with incrementing concentrations of CdII such hat already at 5 mg L−1 greater than 80% inhibition was observed. The medium in the hydrogenotrophic methanogenic assay did not contain SO42− (M2), and thus the steep inhibition response to CdII started with the lowest concentration tested of 1 mg L−1 because the medium was free of biogenic sulfide that could have precipitated CdII. However, the other ions did not show a strict concentration related response over the full range of concentrations tested. In the case of both TeIV and SeIV, the hydrogenotrophic methanogens had an inhibitory response that became saturated at a given concentration and additional increases in concentration did not result in higher toxicity responses. For TeIV, the response became saturated at concentrations of 50 mg L−1 and higher at an inhibition of 27.7%, For SeIV, the response became saturated at 70 mg L−1 or higher at a maximum inhibition of 76.2%. TeVI caused no noteworthy toxic response up to 200 mg L−1.
The concentrations of the compounds tested in this work which inhibited the acetoclastic and hydrogenotrophic methanogenic activity by 20%, 50% and 80% are summarized in Table 1. CdII and both Te oxyanions were highly inhibitory for the acetoclastic community with IC50 values of approximately 10 or less mg L−1. SeIV was also highly toxic but 2 to 3 fold less toxic compared to CdII and Te oxyanions. SeVI was found to be completely non-toxic even at concentrations as high as 6 g L−1. Only CdII and SeIV were highly toxic to hydrogenotrophic methanogens with IC50 values ranging from approximately 3 to 18 mg L−1.
Table 1.
Summary of the IC20, IC50, IC80 values determined for the II-VI ionic species toward the acetoclastic and hydrogenotrophic methanogenic activities of anaerobic sludge.
| A) Acetoclastic methanogenesis
| ||||||
|---|---|---|---|---|---|---|
| Species | IC20 | Std dev | IC50 | Std dev | IC80 | Std dev |
|
| ||||||
| --------------------------------mg L−1-------------------------------- | ||||||
| CdII | 5.3 | 0.2 | 8.6 | 1.0 | 12.7 | 0.3 |
| TeIV | 1.3 | 0.8 | 8.6 | 0.9 | 20.5 | 0.3 |
| TeVI | 2.4 | 1.8 | 10.2 | 0.8 | 38.1 | 6.7 |
| SeIV | 11.8 | 1.5 | 24.1 | 0.1 | 43.3 | 0.7 |
| SeVI | 3595 | 273 | 5514 | 146 | ≥ 6000.0 | NA |
| B) Hydrogenotrophic methanogesis
| ||||||
|---|---|---|---|---|---|---|
| Species | IC20 | Std dev | IC50 | Std dev | IC80 | Std dev |
|
| ||||||
| --------------------------------mg L−1-------------------------------- | ||||||
| CdII | 1.5 | 0.2 | 2.9 | 0.4 | 4.6 | 0.1 |
| TeIV | 5.0 | 1.2 | ≥ 200 | NA | ≥ 200 | NA |
| TeVI | ≥ 500 | NA | ≥ 500 | NA | ≥ 500 | NA |
| SeIV | 6.8 | 0.0 | 18.0 | 1.1 | ≥ 100 | NA |
| SeVI | ≥ 6000 | NA | ≥ 6000 | NA | ≥ 6000 | NA |
NA = Not available
3.2. Acute toxicity towards Aliivibrio fischeri
Important toxic effects were observed when the bioluminescent marine bacterium A. fischeri was exposed to CdII, SeIV and TeIV. Toxicity was evident as the bioluminescence of the bacteria decreased with time (5 to 30 min of exposure) and with increasing toxic compound concentrations. Figure S4 shows the inhibitory effect on the bioluminescent activity of A. fischeri after being exposed to various concentrations of the most toxic compounds, CdII (Panel A) and SeIV (Panel B); while Table 2 reports the IC50 values for the five compounds assessed in this work for all time intervals. The high toxicity of CdII is evident from the IC50 values of only a few mg L−1 while moderate toxicity was observed for SeIV and TeIV with IC50 values of several hundred mg L−1. No effect was evident when TeVI was tested in the whole range of concentrations, and SeVI caused partial inhibition when the marine bacteria were exposed to high concentrations of approximately 32.5 g L−1 (not environmentally relevant, data not shown) of this oxyanion.
Table 2.
Summary of the 50% inhibitory concentrations (IC50) determined for the various II-VI ionic species toward A. fischeri after different times of exposure.
| Species | IC50
|
|||||
|---|---|---|---|---|---|---|
| 5 min | Std dev | 15 min | Std dev | 30 min | Std dev | |
|
| ||||||
| --------------------------------mg L−1-------------------------------- | ||||||
| CdII | 64.2 | 6.8 | 17 | 0.5 | 5.5 | 0.2 |
| TeIV | ≥ 655 | NA | ≥ 655 | NA | 459 | 30 |
| TeVI | ≥1082 | NA | ≥1082 | NA | ≥1082 | NA |
| SeIV | 776 | 127 | 253 | 23.8 | 171 | 13 |
| SeVI | ≥32491 | NA | ≥32491 | NA | ≥32491 | NA |
NA = Not available
4. Discussion
4.1. Main findings
The toxic impact of CdII, TeIV, TeVI, SeIV and SeVI on the acetatoclastic and hydrogenotrophic microorganisms in the methanogenic community of an anaerobic granular sludge was investigated. Our results indicate that both trophic groups were highly inhibited by the presence of CdII and SeIV. TeIV and TeVI caused important inhibition of the acetoclastic trophic group, while they caused medium and mild inhibitory effects on the hydrogenotrophic methanogens, respectively. Both trophic groups tolerated exposure to extremely high non-environmentally relevant concentrations of SeVI.
The inhibitory response to CdII was the most dramatic of all the ions tested. There was a less steep inhibitory response to this ion at concentrations below 4 mg CdII L−1 in the case of the acetoclastic compared to that of the hydrogenotrophic trophic group which could be attributed to the sequestration of soluble CdII by the biogenic sulfide (Mori et al., 2000) formed from the sulfate in the M1 medium.
One of the chalcogen elements studied, Se, is a well-known trace element required by microorganisms (Heider and Bock, 1993). Selenium is involved in the prosthetic group of key enzymes such as hydrogenases in Clostridia, Escherichia coli and methanogens (Ljungdahl, 2009; Yamamoto et al., 1983; Sorgenfrei et al., 1997). Selenium is also part of a common amino acid, selenocysteine in microorganisms (Peters et al., 2004). Se is added in small amounts to wastewaters to stimulate the production of methane during anaerobic digestion (Lenz et al., 2008; Munk and Lebuhn, 2014). SeVI was for all practical purposes completely non-toxic, and this may be related to the preferred form of selenium as a nutrient. While SeIV was toxic, it was not as toxic to methanogens at low concentrations compared to CdII and TeIV as evidenced by higher IC20 values.
To the best of our knowledge, the methanogenic toxicity of the closely related chalcogen, Te, has never been reported before. Te oxyanions caused an unusual response towards the methanogenic consortium. The acetoclastic methanogens were very sensitive to inhibition by these oxyanions. In contrast, the hydrogenotrophic-methanogens were only mildly inhibited by TeIV and were highly tolerant to TeVI. Both Te species caused a sharp toxic response at the lowest concentrations tested; however, a response of diminishing returns at higher concentrations was observed. This may be due to a capacity of methanogens to express an alternative enzyme system, in response to an inhibition. Induction of such a system would require higher concentrations of tellurium oxyanions. In a previous work, we found that both Te oxyanions might be enzymatically reduced to insoluble Te0 (Ramos-Ruiz et al., 2016). Batch experiments were conducted to study the biological reduction of the oxyanions using an anaerobic granular sludge like the one of this work. The systems were supplied with 20 mg L−1 of TeIV or TeIV, and acetate or H2 as the external source of electrons. The concentration of soluble Te in the media decreased over time until no Te was detected in the liquid phase. At the same time, a black precipitate was found to be formed associated to the granular sludge. In contrast, the concentration of soluble Te remained unchanged in the controls lacking anaerobic granular sludge during the whole experiment. A decrease in the concentration of soluble Te in the medium caused by the biologically-mediated reduction of TeIV and TeVI to insoluble Te0 might have enhanced the tolerance to Te.
The toxic effects of CdII, TeIV, TeVI, SeIV and SeVI on the marine bacteria A. fischeri, was also assessed. Of the six compounds tested in this work, CdII was the most toxic to A. fischeri, SeIV caused the second most important effect; while, TeIV caused only a partial inhibitory effect.
4.2. Comparison to literature data and possible mechanisms
Previous studies have evaluated the inhibitory impact of CdII to methanogens in anaerobic granular sludge utilizing different assays substrates such as glucose (Altas, 2009) and volatile fatty acid mixtures (Lin and Chen, 1999). In these studies, the IC50 values of CdII to methanogenic activity ranged from 36 to 450 mg L−1 which are 1 to 2 orders of magnitude less inhibitory than that observed in this study. The difference with our results might be explained by sulfate amendment in the medium in one of the studies (Altas, 2009). This level of sulfate was approximately 4 times higher than the concentration used in our acetoclastic assay and that might have been reduced to sulfide by sulfate reducing bacteria forming CdS(s). The formation of insoluble cadmium sulfide (CdS(s)) is expected in a sulfide containing medium as can be inferred from its low Ksp value (stability constant) of 10−28.85 (Benjamin, 2002). The formation of sulfide under the same experimental conditions has been demonstrated in a previous study from our research group (Gonzalez-Estrella et al., 2015). The potential yield of biogenic sulfide was in a 1.4 fold stoichiometric excess compared to the maximum amount of CdII (128 mg L−1) used in their methanogenic toxicity assays which might have potentially limited the availability of the soluble Cd to inhibit the methanogenic consortium. However, this hypothesis cannot explain the large differences in CdII in our study with the second study (Lin and Chen, 1999) because the maximum biogenic S production (based on sulfur added to media) could only have precipitated at most 18.5% of the CdII at the highest concentration tested (1000 mg L−1). The large differences in the inhibitory concentrations reported might be potentially explained by several differences in the sludge characteristics such as, the content of extracellular polymeric substances (EPS) or the distribution of the microorganisms in the granules. A pure methanogen culture Methanobacterium thermoautotrophicum KHT-2 (Mori et al., 2000) was moderately tolerant of CdII compared to the methanogens of our anaerobic sludge. No inhibition of methane production was observed when a pure culture of the methanogen Methanobacterium thermoautotrophicum KHT-2 was exposed to 11.2 mg CdII L−1 while the methanogenic activity in the acetoclastic- and hydrogenotrophic-trophic groups of our anaerobic sludge were completely inhibited when exposed to concentrations of 12 mg CdII L−1 (Mori et al., 2000). Nonetheless, it should be noted that strong inhibition of M. thermoautotrophicum KHT-2 was observed at a concentration of 56.2 mg L−1 (Mori et al., 2000). These results might be explained by a high tolerance of M. thermoautotrophicum KHT-2 towards CdII since no sulfide was present in the medium used in the bioassays, or alternatively CdII complexation with organic constituents in the medium may have occurred, resulting in total/partial removal of free CdII from the medium (yeast extract and peptone collectively accounted for 4 g L−1).
The toxic effects of CdII on the marine bacterium, A. fischeri, have been previously reported (Villaescusa et al., 1996). In the case of CdII, similar IC50 values to those obtained in this work were found. For the three commonly used exposure time periods (5, 15 and 30 min) the IC50 values ranged from 10.1 to 33.5 mg Cd L−1. Due to the tendency of Cd to form stable complexes with chloride ions, the toxicity of Cd has been associated to the presence of the cadmium complexes formed with the NaCl in the Microtox® osmotic adjusting solution and not to the presence of CdII alone, which might also be true for the methanogens in the anaerobic sludge since our basal medium was amended with a considerable amount of chloride (Cl−) of ~ 200 mg L−1. The species of Cd present in the Microtox® test have been determined from thermodynamic data at the Microtox® experimental conditions and are reported to be CdCl+, CdCl2 and CdCl3− (Villaescusa et al., 1996).
The results observed in this work correlate well with studies on the toxicity of SeIV and SeVI towards the acetoclastic and hydrogen- consuming microorganisms in anaerobic granular sludge (Lenz et al., 2008). The reported IC50 values of 73.0 mg L−1 and 55.5 mg L−1 for the acetoclastic and hydrogenotrophic activities for SeIV, respectively, are in the same order of magnitude of those reported in this work. The IC50 values for SeVI were 1283 mg L−1 and 3518 mg L−1 for the acetate- and hydrogen-consuming groups, respectively (Lenz et al., 2008). Our results also demonstrated a high tolerance of the methanogenic communities to SeVI. Likewise Te, both Se oxyanions can be reduced to insoluble Se0 by microbial means (Astratinei et al., 2006; Borghese et al., 2014). The lower bioavailability of soluble Se resulting from the formation of insoluble Se0 might have improved the tolerance to Se.
The toxic effects of both Se oxyanions on the marine bacteria A. fischeri have also been assessed previously (Yu et al., 1997). SeIV caused higher inhibitory effects compared to SeVI (Yu et al., 1997), which is in agreement with the results from this work. However, the IC50 values estimated in the present study for each Se oxyanion were higher than those previously reported.
This study reports for the first time on the methanogenic toxicity of Te oxyanions. Previously methanogens were implicated in the formation of Te methylated species (Meyer et al., 2008). Also previously, the toxicity of the Te oxyanions to bacteria was studied (Turner et al., 2012). The valence of Te oxyanions, as well as, the localization of the bacteria (planktonic cells or inside biofilms) had important impacts on the observed toxicity. In the case of planktonic cells of E. coli and Staphylococcus aureus, TeVI was 10× and 3.7× less toxic than TeIV, respectively (based on the minimal inhibitory concentration (MIC) ratios MICTeVI/MICTeIV) (Turner et al., 2012). In the case of bacteria in biofilms, TeVI was 3× and 1.8× less toxic than TeIV, respectively; based on the MIC required to prevent regrowth of the bacteria from a treated biofilm (Turner et al., 2012). The latter results are in agreement with our results, the acetoclastic activity of the methanogenic consortium studied here was 1.2× less affected by TeVI based on the IC50 values than by TeIV (Table 1A). Due to the lower toxicity of both oxyanions to the hydrogen consuming over the acetoclastic trophic group, it was not possible to estimate the IC50 values for TeIV and TeVI in the ranges of concentrations used in this work for the hydrogenotrophic methanogens (Table 1B); however, the methane production was more affected by TeIV than by TeVI (Figure 2B). These findings showed that the toxic effects of the Te oxyanions are also dependent of the trophic group in the methanogenic consortium.
The difference in the toxicity of the Te oxyanions towards both methanogenic trophic groups might be explained by some of the different mechanisms of toxicity resistance that have been widely discussed in the literature, such as the reduction of Te soluble species by enzymatic or non-enzymatic means, Te volatilization or the presence of Te resistance determinant genes (Te1R) (Chasteen et al., 2009). Even though the formation of methylated Te species by methanogens has been previously reported (Meyer et al., 2008), the results of this work indicated that volatilization of Te was not significant in these systems since less than 0.00002% of the total Te amended to the systems was found in the gas phase of the bottles used to determine Te volatilization at the end of the experiment (see SI).
The remarkably higher toxicity levels caused by both oxyanions to the acetate consuming group compared to those caused to the hydrogen consuming group might be attributed to several factors. First of all, evidence points out that the microbial reduction of the Te oxyanions to insoluble Te0 is used as a mechanism to decrease their toxic effects (Turner et al., 2012; Chasteen et al., 2009; Moore and Kaplan, 1992) by lowering its bioavailability. H2 is a superior electron donor for Te reduction according to the lower redox potential of the 2H+/H2 pair (E0′ = −0.414 V (Madigan, 2009)) compared to that of CO2/acetate (E0′ = −0.28 V (Madigan, 2009)). Since the redox potential E0′ (pH 7) of the pairs HTeO4−/HTeO3− and HTeO3−/Te0 is 0.399 V and 0.196 V (Bouroushian, 2010), respectively, this suggests a more thermodynamically favorable formation of insoluble Te0 species in the systems amended with H2 than in those supplied with acetate as substrate. Furthermore, in recent experimental work, H2 was shown to stimulate the microbial reduction of both Te oxyanions in methanogenic granular sludge; whereas acetate did not have a stimulatory effect (Ramos-Ruiz et al., 2016). Therefore, the decreased availability of soluble Te species might potentially lead to an apparent increased tolerance of the hydrogenotrophic methanogens to both oxyanions. The formation of a black precipitate, which is characteristic of the formation of Te0 (Baesman et al., 2007; Ramos-Ruiz et al., 2016), was observed in the bioassays.
The higher inhibition of methane production in the systems amended with acetate might also be explained by a potential competition between the soluble tellurium species and acetate to enter the cells. Evidence points out that some microorganisms, such as the facultative phototroph Rhodobacter capsulatus transport TeIV into the cells through an acetate permease (ActP) which is also responsible for acetate uptake (Borghese and Zannoni, 2010). Therefore, the presence of acetate might have induced the expression of the ActP which provided a mechanism of Te entry which was probably not the case with H2 as substrate. The results of this work are in agreement with the general understanding that TeIV is more toxic than TeVI since higher inhibition of methane production was observed in both methanogenic trophic groups after being exposed to TeIV.
To date, there have also not been any reports on the toxicity of either Te oxyanions towards A. fischeri; however, our results indicate that TeIV is more toxic than TeVI which is consistent with the general conclusion for most microorganisms (Chasteen et al., 2009; Turner et al., 2001; Turner et al., 2012).
5. Conclusion
Of all the potential ions that can potentially be leached from II-VI semiconductor materials, CdII caused the most important toxic effect towards the methanogens in the anaerobic granular sludge and to the marine bacterium A. fischeri. The observed inhibition of the acetoclastic methanogens in the granular sludge agrees with the basic understanding that the toxicity level of the chalcogens (Se and Te) to most microorganisms increases in the following order: SeVI, SeIV, TeVI, and TeIV (Zannoni et al., 2008). In the case of the hydrogenotrophic methanogens, the inhibitory effect increased in the following sequence: SeVI, TeVI, TeIV and SeIV. The toxic effects of TeIV and SeIV to the marine bacterium, A. fischeri, were also observed to be important. This work demonstrates for the first time the toxic effects of the Te oxyanions towards a consortium of methanogens
From the findings of this work, it can be concluded that the presence of soluble ions leaching from II-VI semiconductor materials can potentially negatively affect anaerobic treatment processes such as the methanogenic phase in a landfill. Thus caution should be taken if decommissioned II-V semiconductors materials are disposed of in municipal landfills. Likewise anaerobic treatment processes may be impacted in wastewater treatment systems handling effluents from industrial facilities processing II-V semiconductor materials.
Supplementary Material
Highlights.
Leached ionic species from II-VI semiconductor materials toxic to microorganisms
Toxicity of tellurium oxyanions to methanogens reported for the first time
Toxicity to acetoclastic methanogens increased as follows SeIV < TeVI < TeIV ≈ CdII
Only CdII followed by SeIV caused strong toxicity to hydrogenotrophic methanogens
Aliivibrio fischeri was strongly inhibited by CdII and less by SeIV and TeIV
Acknowledgments
This work was funded in part by a grant of the National Institute of Environment and Health Sciences-supported Superfund Research Program (NIH ES-04940). ARR was partly funded by CONACyT and PROMEP. LHT was partly funded by CAPES.
Footnotes
Additional figures and analytical methods are described in the Supplementary Information section. Supplementary data associated with this article can be found in the online version, at @@@@
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Altas L. Inhibitory effect of heavy metals on methane-producing anaerobic granular sludge. J Hazard Mater. 2009;162:1551–1556. doi: 10.1016/j.jhazmat.2008.06.048. [DOI] [PubMed] [Google Scholar]
- Astratinei V, van Hullebusch E, Lens P. Bioconversion of selenate in methanogenic anaerobic granular sludge. J Environ Qual. 2006;35:1873–1883. doi: 10.2134/jeq2005.0443. [DOI] [PubMed] [Google Scholar]
- Baesman SA, Bullen TD, Dewald J, Zhang DH, Curran S, Islam FS, Beveridge TJ, Oremland RS. Formation of tellurium nanocrystals during anaerobic growth of bacteria that use Te oxyanions as respiratory electron acceptors. Appl Environ Microbiol. 2007;73:2135–2143. doi: 10.1128/AEM.02558-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benjamin MM. Water Chemistry. Waveland Press, Inc; Long Grove, IL: 2002. [Google Scholar]
- Bicknell RN, Giles NC, Schetzina JF. Controlled substitutional doping for CdTe-films. J Vac Sci Technol B. 1987;5:701–704. [Google Scholar]
- Borghese R, Baccolini C, Francia F, Sabatino P, Turner RJ, Zannoni D. Reduction of chalcogen oxyanions and generation of nanoprecipitates by the photosynthetic bacterium Rhodobacter capsulatus. J Hazard Mater. 2014;269:24–30. doi: 10.1016/j.jhazmat.2013.12.028. [DOI] [PubMed] [Google Scholar]
- Borghese R, Zannoni D. Acetate permease (ActP) is responsible for tellurite (TeO32−) uptake and resistance in cells of the facultative phototroph Rhodobacter capsulatus. Appl Environ Microbiol. 2010;76:942–944. doi: 10.1128/AEM.02765-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouroushian M. Electrochemistry of metal chalcogenides. Springer-Verlag; Berlin: 2010. [Google Scholar]
- Bulich AA, Isenberg DL. Use of the luminescent bacterial system for the rapid assessment of aquatic toxicity. Isa T. 1981;20:29–33. [PubMed] [Google Scholar]
- United Nations Environment Programme. Renewables 2015 global status report; key findings. Nairoby, KE: 2015. http://www.ren21.net/wp-content/uploads/2015/07/GSR2015_KeyFindings_lowres.pdf. [Google Scholar]
- Chasteen TG, Fuentes DE, Tantalean JC, Vasquez CC. Tellurite: history, oxidative stress, and molecular mechanisms of resistance. FEMS Microbiol Rev. 2009;33:820–832. doi: 10.1111/j.1574-6976.2009.00177.x. [DOI] [PubMed] [Google Scholar]
- Chate PA, Sathe DJ, Hankare PP, Lakade SD, Bhabad VD. Synthesis and characterization of cubic cadmium selenide by chemical route. J Alloys Compd. 2013;552:40–43. [Google Scholar]
- U.S. Department of Energy’s National Renewable Energy Laboratory. Department of Energy; Washington: 2013. http://www.nrel.gov/docs/fy15osti/62580.pdf. [Google Scholar]
- Feng Z, Zhang Q, Lin L, Quo H, Zhou J, Lin Z. < 0001 >-Preferential growth of cdse nanowires on conducting glass: Template-free electrodeposition and application in photovoltaics. Chem Mater. 2010;22:2705–2710. [Google Scholar]
- Gonzalez-Estrella J, Puyol D, Sierra-Alvarez R, Field JA. Role of biogenic sulfide in attenuating zinc oxide and copper nanoparticle toxicity to acetoclastic methanogenesis. J Hazard Mater. 2015;283:755–763. doi: 10.1016/j.jhazmat.2014.10.030. [DOI] [PubMed] [Google Scholar]
- Heider J, Bock A. Selenium metabolism in microorganisms. Adv Microb Physiol. 1993;35:71–109. doi: 10.1016/s0065-2911(08)60097-1. [DOI] [PubMed] [Google Scholar]
- Huynh WU, Dittmer JJ, Alivisatos AP. Hybrid nanorod-polymer solar cells. Science. 2002;295:2425–2427. doi: 10.1126/science.1069156. [DOI] [PubMed] [Google Scholar]
- Lenz M, Janzen N, Lens PNL. Selenium oxyanion inhibition of hydrogenotrophic and acetoclastic methanogenesis. Chemosphere. 2008;73:383–388. doi: 10.1016/j.chemosphere.2008.05.059. [DOI] [PubMed] [Google Scholar]
- Lin CY, Chen CC. Effect of heavy metals on the methanogenic UASB granule. Water Res. 1999;33:409–416. [Google Scholar]
- Lithner D, Halling M, Dave G. Toxicity of electronic waste leachates to Daphnia magna: screening and toxicity identification evaluation of different products, components, and materials. Arch Environ Contam Toxicol. 2012;62:579–588. doi: 10.1007/s00244-011-9729-0. [DOI] [PubMed] [Google Scholar]
- Ljungdahl LG. A life with acetogens, thermophiles, and cellulolytic anaerobes. Annu Rev of Microbiol. 2009;63:1–25. doi: 10.1146/annurev.micro.091208.073617. [DOI] [PubMed] [Google Scholar]
- Macken A, Giltrap M, Ryall K, Foley B, McGovern E, McHugh B, Davoren M. A test battery approach to the ecotoxicological evaluation of cadmium and copper employing a battery of marine bioassays. Ecotoxicology. 2009;18:470–480. doi: 10.1007/s10646-009-0305-6. [DOI] [PubMed] [Google Scholar]
- Madigan MT, Martinko JM, Dunlap PV, Clark DP. Brock Biology of Microorganisms. 12. Pearson Education, Inc; London: 2009. [Google Scholar]
- Meyer J, Michalke K, Kouril T, Hensel R. Volatilisation of metals and metalloids: An inherent feature of methanoarchaea? Syst Appl Microbiol. 2008;31:81–87. doi: 10.1016/j.syapm.2008.02.001. [DOI] [PubMed] [Google Scholar]
- Moore MD, Kaplan S. Identification of intrinsic high-level resistance to rare-earth-oxides and oxyanions in members of the class proteobacteria - Characterization of tellurite, selenite, and rhodium sesquioxide reduction in Rhodobacter sphaeroides. J Bacteriol. 1992;174:1505–1514. doi: 10.1128/jb.174.5.1505-1514.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori K, Hatsu M, Kimura R, Takamizawa K. Effect of heavy metals on the growth of a methanogen in pure culture and coculture with a sulfate-reducing bacterium. J Biosci Bioeng. 2000;90:260–265. doi: 10.1016/s1389-1723(00)80079-1. [DOI] [PubMed] [Google Scholar]
- Munk B, Lebuhn M. Process diagnosis using methanogenic Archaea in maize-fed, trace element depleted fermenters. Anaerobe. 2014;29:22–28. doi: 10.1016/j.anaerobe.2014.04.002. [DOI] [PubMed] [Google Scholar]
- Peters F, Rother M, Boll M. Selenocysteine-containing proteins in anaerobic benzoate metabolism of Desulfococcus multivorans. J Bacteriol. 2004;186:2156–2163. doi: 10.1128/JB.186.7.2156-2163.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Punitha K, Sivakumar R, Sanjeeviraja C, Ganesan V. Influence of post-deposition heat treatment on optical properties derived from UV-vis of cadmium telluride (CdTe) thin films deposited on amorphous substrate. Appl Surf Sci. 2015;344:89–100. [Google Scholar]
- Ramos-Ruiz A, Field JA, Wilkening JV, Sierra-Alvarez R. Recovery of elemental tellurium nanoparticles by the reduction of tellurium oxyanions in a methanogenic microbial consortium. Environ Sci Technol. 2016;50:1492–1500. doi: 10.1021/acs.est.5b04074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorgenfrei O, Duin EC, Klein A, Albracht SPJ. Changes in the electronic structure around Ni in oxidized and reduced selenium-containing hydrogenases from Methanococcus voltae. Eur J Biochem. 1997;247:681–687. doi: 10.1111/j.1432-1033.1997.00681.x. [DOI] [PubMed] [Google Scholar]
- Tapia-Rodriguez A, Luna-Velasco A, Field JA, Sierra-Alvarez R. Toxicity of uranium to microbial communities in anaerobic biofilms. Water Air Soil Poll. 2012;223:3859–3868. [Google Scholar]
- Taylor DE. Bacterial tellurite resistance. Trends Microbiol. 1999;7:111–115. doi: 10.1016/s0966-842x(99)01454-7. [DOI] [PubMed] [Google Scholar]
- Trevors JT, Stratton GW, Gadd GM. Cadmium transport, resistance, and toxicity in bacteria, algae, and fungi. Can J Microbiol. 1986;32:447–464. doi: 10.1139/m86-085. [DOI] [PubMed] [Google Scholar]
- Turner RJ, Aharonowitz Y, Weiner JH, Taylor DE. Glutathione is a target in tellurite toxicity and is protected by tellurite resistance determinants in Escherichia coli. Can J Microbiol. 2001;47:33–40. [PubMed] [Google Scholar]
- Turner RJ, Borghese R, Zannoni D. Microbial processing of tellurium as a tool in biotechnology. Biotechnol Adv. 2012;30:954–963. doi: 10.1016/j.biotechadv.2011.08.018. [DOI] [PubMed] [Google Scholar]
- Villaescusa I, Martinez M, Pilar M, Murat JC, Hosta C. Toxicity of cadmium species on luminescent bacteria. Fresenius J Anal Chem. 1996;354:566–570. doi: 10.1007/s0021663540566. [DOI] [PubMed] [Google Scholar]
- Yamamoto I, Saiki T, Liu SM, Ljungdahl LG. Purification and properties of NADP-dependent formate dehydrogenase from clostridium-thermoaceticum, a tungsten-selenium-iron protein. J Biol Chem. 1983;258:1826–1832. [PubMed] [Google Scholar]
- Yu R, Coffman JP, VanFleetStalder V, Chasteen TG. Toxicity of oxyanions of selenium and of a proposed bioremediation intermediate, dimethyl selenone. Environ Toxicol Chem. 1997;16:140–145. [Google Scholar]
- Zannoni D, Borsetti F, Harrison JJ, Turner RJ. The bacterial response to the chalcogen metalloids Se and Te. In: Poole RK, editor. Advances in Microbial Physiology. Vol. 53. Elsevier Academic Press, Inc; 2008. pp. 1–72. [DOI] [PubMed] [Google Scholar]
- Zeng C, Ramos-Ruiz A, Field JA, Sierra-Alvarez R. Cadmium telluride (CdTe) and cadmium selenide (CdSe) leaching behavior and surface chemistry in response to pH and O2. J. 2015 doi: 10.1016/j.jenvman.2015.02.033. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.