Abstract
Regulatory T cells (Tregs) are capable of dampening immune-mediated inflammation and avert the destructive effects of uncontrolled inflammation. Distinct molecules and pathways, including various transcription factors, phosphatases and kinases, impact the ability of Tregs to function as negative regulators of the immune response, and are presumably amenable to therapeutic manipulation. Here, we discuss recently identified molecular networks and the therapeutic potential for treating autoimmune diseases.
Cellular Requirements of Tregs
As early as the late 1960’s it was recognized that neonatal thymectomy of mice led to autoimmunity [1]. In the 1970’s interleukin-2 (IL-2) was discovered as an important driver of the immune response [2] yet, in the early 1990’s it was shown that IL-2 and IL-2 receptor deficiency in mice led to autoimmune pathology [3, 4] pointing to a paradox to be resolved later. In 1995 Sakaguchi proved that CD4+ CD25high T-cells were functionally regulatory T-cells (Tregs)(Box 1)f [5]. From then, Tregs gained intense attention both in terms of their ability to control inflammation, autoimmune responses, as well as to hamper the ability of the immune system to control the growth of tumor cells [6].
Box 1. Thymically-derived and Peripherally-induced Tregs.
Tregs can develop in the thymus and in peripheral tissues. Peripheral Tregs (iTregs) differentiate from naïve CD4+ T cells in peripheral tissues following activation from antigen encounter in the presence of TGF-β, and in the absence of inflammatory cytokines such as IL-6, IFN-γ, and IL-4 [1]. Thymically derived Tregs (nTregs) require T cell receptor (TCR) signaling via self-peptide and MHC class II recognition [2]. Both of these distinct populations require stable expression of Foxp3, as germline deletion of Foxp3 leads to Treg deficiency and lethal autoimmunity [3] [4] [5]. It appears that Foxp3 controls Treg lineage development through its expression in thymic epithelial cells, but does not shape T cell repertoire within the thymus [6] [7]. Tregs also require IL-2 and TGF-β, which act to stabilize the expression of Foxp3 [8].
Recent work has uncovered many key molecular networks required to develop and sustain Treg function. Here, we discuss critical molecules including the kinases/phosphatases mTOR, PP2A, and PTEN, the transcription factors Foxo1, Bach2 and Helios, the essential cellular processes of autophagy and ubiquitination and finally review signaling pathways which either promote or repress the Treg program (Box 3). Together, the diverse cellular requirements of Tregs present new therapeutic targets to treat autoimmune diseases and cancer.
Box 3. Autophagy.
Autophagy is a process targeting intracellular components for lysosomal degradation (e.g. proteins, etc.) and recycling them while maintaining homeostasis during metabolic or infectious stress. The process has emerged as a critical homeostatic mechanism in T cells, directing proliferation and differentiation. In Tregs, autophagy is more active compared to Tconv cells and the process supports their survival. When autophagy genes have been selectively deleted, mice have developed inflammatory disorders with a contracted Treg compartment [15] [16]. Autophagy deficient Tregs have demonstrated increased apoptosis, and decreased Foxp3 expression, as well as increased levels of mTORC1 activity and glycolysis [15] [16]. One possibility is that autophagy degrades lipids to fatty acids, which can then fuel fatty acid oxidation, required for Treg metabolism.
Tregs and Immunosuppression
Interest in Tregs was intensified after forkhead box P3 (FoxP3) was defined as a master transcription factor linked to their function along with high expression of surface CD25. Neither CD25 nor FoxP3 are perfect markers identifying Tregs because CD25 can be readily expressed in antigen or mitogen-stimulated T cells and effector T cells can express FoxP3 transiently in humans and mice [7, 8]. In 1959, a spontaneous systemic auto-inflammatory phenotype in mice was described and coined the scurfy mouse[9]. Subsequently, the Rudensky [10] and Sakaguchi [11] labs reported that the scurfy mutation in male mice resided within the foxp3 gene, and furthermore, that this phenotype was linked to Foxp3 deficiency. In humans, an X-linked familial autoimmune syndrome of undetermined origin was also linked to FoxP3 deficiency [12], The syndrome is characterized by immune dysregulation, polyendocrinopathy, enteropathy, and defined by the acronym IPEX [13] [12]. Sequencing of the foxp3 gene in many IPEX cases revealed functionally detrimental mutations within the gene itself at the 3’UTR region [14]. These studies established the critical role of FoxP3 in the disease.
Observations in scurfy mice and IPEX demonstrated that Foxp3-expressing Tregs had the capacity to negatively regulate the immune response. In their absence, an exaggerated immune response ensued that eventually led to the development of autoimmune disease. Since the initial observations in scurfy mice and IPEX syndrome in humans, aberrant Treg function has been linked to many autoimmune conditions such as connective tissue diseases (e.g. SLE), endocrinopathies (e.g. type 1 diabetes), inflammatory bowel disease (e.g. Crohn’s disease), and central nervous system autoimmune diseases (e.g. multiple sclerosis). In summary, the crucial role of Tregs in the regulation of the immune response has been clearly established.
Differentiation of Tregs Requires TCR Engagement and Foxp3 Expression in the Presence of IL-2 and TGF-β
Tregs express high levels of IL-2 receptor α (CD25), and their proper development and survival requires the presence of IL-2 [15] [16]. Yet, Tregs do not produce IL-2 [16] and therefore depend on other sources for this cytokine. IL-2 receptor-driven by the proximal lck promoter has been shown to restore the thymic generation of Tregs and to rescue IL-2 receptor deficient mice from lethal autoimmunity [17]. IL-2 has been demonstrated to upregulate Foxp3 expression in thymic Treg cell precursors. Additionally, over expression of STAT5 (IL-2-induced transcription factor) using STAT5 transgenic mice dramatically increased the number of Tregs compared to wild type mice [18]. Therefore, it is reasonable to propose that increasing STAT5 by exogenous IL-2 represents a means to empower Tregs. Indeed, Tregs utilize the IL-2>IL-2R>Jak1/3>STAT5 pathway more efficiently than conventional T-cells, and low dose IL-2 treatment has met clinical success for the treatment of graft versus host disease, hepatitis C related vasculitis and SLE (see below) [19] [20].
Treg development also depends on TGF-β signaling, as TGF-β receptor deficiency has been shown to impede the generation of Tregs [21].This was evident in mouse studies in which conditional deletion of TGF-β interrupted the development of nTregs at postnatal days 3–5 but not at day 7 (by which time nTregs had become highly sensitive to the presence of IL-2) [21]. As Tregs develop in the thymus, they undergo positive and negative selection following exposure to self-antigen and affinity of the TCR to these antigens will determine T-cell fate (reviewed in ref [22]) In this regard TGF-β also facilitates nTreg development by inhibiting apoptosis during negative selection in the thymus. Mechanistically this is due to TGF-β elevating the expression of the anti-apoptotic gene BCL-2. Indeed, mice deficient in IL-2 receptor and TGF-β receptor fail to develop Tregs in the thymus [23].
The conserved non-coding DNA sequence (CNS) elements at the foxp3 locus play a role in stability of Tregs. Following T cell activation (e.g. after signals 1 and 2) the CNS3 is accessed and associated with c-Rel, and this binding is thought to enable the opening of foxp3 locus [24]. Thymic and to a lesser extent peripheral CD4+Foxp3+ cells were significantly lost in mice lacking CNS3 but without affecting the intensity of Foxp3 per cell [25]. Later it was shown that CNS3 might increase the probability of Foxp3 induction in response to TCR stimulation and to expand the TCR repertoire of Tregs [26].
Role of foxp3 in Maintaining Treg Lineage Stability
Foxp3 in Tregs
Mouse studies have shown us that sustained expression of Foxp3 is an important factor in Treg lineage stability. Foxp3 preserves Treg suppressive function and opposes the transition of Tregs into an effector phenotype. In cell fate mapping experiments, most Tregs that assumed a proinflammatory effector phenotype had lost Foxp3 expression [27–29]. Foxp3 negative “ex-Tregs” expressed low levels of CD25, GITR, and CTLA4, produced by IFN-γ, IL-17 and thus, could not suppress conventional T cells in vitro [7, 30]. These studies clearly demonstrate that without Foxp3, Tregs are more likely to lose suppressive function and possibly assume an inflammatory phenotype.
Phenotypic plasticity allows Tregs to adapt to diverse and changing environmental cues of various tissues. In the setting of inflammation or lymphopenia, finding that these “ex-Tregs” promote inflammation has provoked controversy and raised concerns regarding the validity and safety of ongoing clinical trials which transfer Tregs adoptively to treat autoimmune diseases [31].
TCR and IL-2 Signaling Stabilize Foxp3 Expression
There are several ways by which Tregs maintain Foxp3 expression and function. The intronic enhancer at the Foxp3 locus termed the CNS 2 has binding sites for the transcription factors NFAT and STAT5. Binding of NFAT and STA5 make CNS2 the downstream target of both TCR and IL-2 signaling (Figure 1) [32]. In fact, studies have shown that CNS2 is crucial for the maintenance of Foxp3 expression in the presence of pro-inflammatory cytokines or limited levels of IL-2. This highlights an important mechanism by which Tregs stability is maintained in the setting of inflammation when there is increased IL-2 signaling [33, 34].
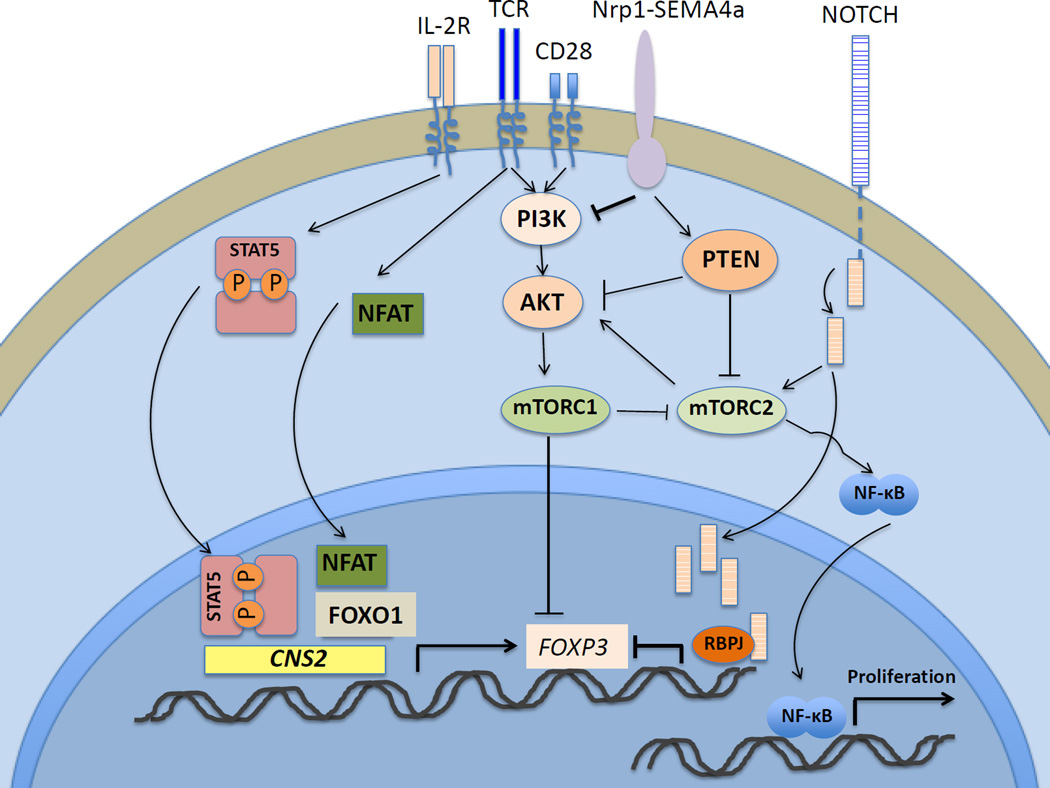
Figure 1. Treg-specific Pathways Affecting Foxp3 Expression.
Murine studies have revealed that TCR and IL-2 lead to NFAT and STAT5b binding to the CNS2 locus, stabilizing Foxp3 expression. TCR engagement and IL-2 trigger PI3K-Akt-mTOR signaling which is negatively regulated by PTEN. Negative regulation of the PI3K-Akt-mTOR axis stabilizes Foxp3 expression. Nrp-1-SEMA4a inhibits PI3K and promotes PTEN, stabilizing Foxp3 expression. Depicted are two pathways of Notch signaling; the non-canonical promotes mTORC2 signaling tagging FOXO1 preventing nuclear translocation thereby destabilizing Foxp3 expression; the canonical pathway facilitates NF-κB nuclear translocation resulting in cellular proliferation.
Epigenetic Traits that Maintain Foxp3 Expression
Foxp3 expression is maintained by multiple epigenetic traits including DNA methylation and histone modifications. The CNS2 site is hypomethylated in Tregs compared to conventional T cells, where this region is fully methylated (closed conformation, transcriptionally inactive) [35, 36]. Importantly, one way IL-2 signaling enhances transcription of Foxp3 is by maintaining hypomethlylation at CNS2 [35]. The hypomethylation at CNS2 physically depends on DNA methlytranserase 1 (Dnmt1) and Tet methylcytosine dioxygenase 2 (Tet2) [37, 38].
Conditional deletion of Dnmt1 has been shown to alter expression of several hundred pro-inflammatory and other genes in Tregs, exemplifying the importance of Dnmt1 for maintenance of the core gene program underlying Treg development and function [37, 38]. IL-2, along with epigenetic changes in foxp3 locus were found to be connected through Tet2, since down-regulation of Tet2 destabilized the expression of Foxp3 in IL-2-deficient Treg cells. In fact, Tet2 led to hypomethylation and stability of Foxp3 regardless of the presence of IL-2. These studies have stressed how methylation of the Foxp3 locus can determine cell fate. [37, 38]
Foxp3 binds to many histone-modifying proteins that promote epigenetic stability. This hetergenous group includes the histone deacetylases (HDACs), p300, TIP 60 and Enhancer of zeste homolog 2 (Ezh2). Although some of these proteins interact directly with Foxp3, many of them function to repress effector molecules upregulated during inflammation and capable of destabilizing Tregs [39] [40].
Moreover, Tregs have been shown to express elevated levels of the deubiquitinase USP7 which is attached to Foxp3 in the nucleus. In a murine adoptive transfer-induced colitis model it was shown that the USP7-Foxp3 association resulted in a decrease in proteasome-mediated degradation of Foxp3 [41]. In contrast, in a proinflammatory milieu, Foxp3 was reported to be polyubiquitinated and degraded, thus resulting in elimination of Tregs. This process was shown to occur in an Hsp70-dependent manner, following the interaction of Foxp3 with the E3 ubiquitin ligase Stub1 [42]. Cbl-b is another E3 ubiquitin ligase that may also affect the development Tregs, and together with Stub1, to lead to Foxp3 ubiquitination and degradation in the proteasome in mice lacking CD28 [43]. Interestingly, treatment with a proteasome inhibitor could overcome Foxp3 degradation in these mice and enable the proper thymic development of Tregs [43].
The lineage stability and function of Tregs is enforced by sustained Foxp3 expression. Stable Foxp3 expression is accomplished by multiple epigenetic and non-epigenetic mechanisms, which together represent the coordinated genetic landscape required to sustain Treg function.
Known Molecular Requirements for Treg Sustenance and Function
Transcription Factors
Foxo1 (another member of the Forkhead box family) has been shown to be essential in maintaining the suppressive function of mature Tregs. When Foxo1 is deleted specifically in Tregs, mice develop a systemic inflammatory condition resembling the scurfy mouse. Foxo1 acts as a transcriptional enhancer during Treg development [44]. However, it can also prohibit the production of IFN-γ. In wild type Tregs, Foxo1 binds directly to the Ifng locus inhibiting IFN-γ production. Deletion of Foxo1 in Tregs allows production of aberrantly high levels of IFN-γ, which in turn, impair Treg suppressor function [45]. Under inflammatory conditions, TCR signaling in all T-cells activates Akt which exports Foxo1 from the nucleus [45]. Wild type Tregs may have diminished IFN-γ production in an inflammation setting because they express relatively greater amounts of Foxo1 than conventional T-cells [45]. Therefore, Foxo1 expression in Tregs may protect these cells from assuming an inflammatory phenotype in a pro-inflammatory environment.
Bach2 is a transcription factor that regulates immune activation. In the context of CD4+ T cell activation in the periphery, Bach2 has been shown to repress genes associated with polarization and differentiation of helper T cells, and simultaneously promote the peripheral induction of Tregs [46]. Mice lacking Bach2 develop a wasting disease that is due to both a severe reduction in Tregs and to non-repressed effector CD4+T cell differentiation [[46]. Although the stability of Foxp3 in existing Tregs and suppression of T-cell proliferation were not shown to be affected in the absence of Bach2, these Tregs were unable to suppress colitis in a lymphopenic host upon Treg adoptive transfer [46]. In another study, mice harboring Bach2-deficient T cells presented preferentially reduced survival of Helios-negative Tregs, and associated with reduced Bcl-2 and Mcl-1 levels, but elevated Bim/Bcl-2 ratios [47]. Due to these imbalances, these mice presented a fatal Th2-type chronic inflammatory lung disease.
Helios, a member of the Ikaros transcription factor family, was characterized as a marker of thymically derived Tregs [48]. Recently, Helios was specifically deleted in Tregs by generation of Foxp3cre/Heliosfl/fl mice [49] [50]. These mice developed systemic autoimmune pathology characterized by increased germinal center formation, lymphocytic infiltration into non-lymphoid organs and glomerulonephritis [49] [50]. Helios deficient Tregs lost their capacity to suppress, and instead, gained effector function. They exhibited diminished Foxp3 expression and increased IFN-γ and IL-17 production. Mechanistically, aberrant Treg function was linked to decreased activation of the STAT5 signaling pathway [49]. Other groups have also reported that the expression of helios serves to enhance Treg functions [51]. Taken together, these studies are indicative the vital role of Helios in maintaining Treg stability and function.
Kinases and Phosphatases
The mechanistic or mammalian target of rapamycin (mTOR) is an intracellular serine threonine kinase, central to T cell function. In T cells mTOR integrates cell signaling pathways and metabolism, which enables cell-type specific responses. Upon T cell activation TCR, CD28 and IL-2 activate PI3K via receptor tyrosine kinases. PI3K, via AKT, promotes mTOR signaling. T effector cells and Tregs depend on mTOR signaling. In T effector cells increased mTOR signaling has been shown to enhance the inflammatory environment [52]. By contrast, Tregs must restrain mTOR tin order to preserve their suppressive function [52].
Increased mTOR activity is associated with pro-inflammatory effector function by skewing T cell development towards the Th1 and Th17 lineages, while simultaneously contracting the Treg population [53]. Indeed, when mTOR was deleted in CD4+T-cells, all conventional CD4+T-cells assumed a Treg phenotype [54]. These phenotypic changes occurred even in the presence of Th1, Th2 and Th17 polarizing environments in vitro. Despite these polarizing conditions T cells lacking mTOR expressed Foxp3 and TGF-β.
T cell metabolism is also regulated by mTOR in a cell lineage specific manner. For instance, when T effector cells are activated they depend on glycolysis to support their nutrient needs. When stimulated, mTOR upregulates genes associated with the pentose phosphate pathway and the Glut 1 transporter thereby facilitating glucose entry into cells, and, glycolysis [55] [56]. In contrast, in order to maintain functional capacity in Tregs, mTOR appears to be required not for glycolysis but rather, for lipogenesis [55] [56].
Recently it was established that the distinct multi-protein complex of mTOR mTORC1 was essential to Treg function. When mTORC1 was conditionally knocked out in Tregs, the mice developed a severe lymphoproliferative autoimmune condition [57]. This was a perplexing and unexpected result because the Akt-mTORC1 axis was known to antagonize de novo Treg differentiation. Another unexpected result was that wild type Tregs had higher basal levels of mTORC1 activity compared to conventional T cells. Mechanistically, TCR and IL-2 signaling increased mTORC1 activity in wild type Tregs via the PI3k-Akt axis. Furthermore, this increase in mTORC1 was necessary for Tregs to meet their metabolic demands, by promoting cholesterol and lipid metabolism, specifically the mevalonate pathway [57]. These findings illustrate how mTORC1 integrates TCR, IL-2 and lipogenic pathways that are essential for Treg function. Although complete deletion of mTORC1 resulted in Treg dysfunction and autoimmunity, unrestrained and highly active mTORC1 disrupted Treg homeostatic function [57].
PTEN is a protein phosphatase that negatively regulates PI3K by blocking the conversion of phosphatidylinositol to PIP3 [59]. It is highly expressed in Tregs regulating their differentiation [60]. It has been reported that the Neuropillin-1-semaphorin-4a axis stabilizes Treg function by restraining PI3K and potentiating PTEN activity (Figure 1) [61]. In 2015, two studies determined that Tregs absolutely required the phosphatase PTEN to maintain their suppressive function. When PTEN was conditionally knocked out in Tregs, mice developed a systemic lymphoprolieferative autoimmunity resembling SLE with high titer antinuclear antibodies (ANAs) and immune complex glomerular nephritis [62, 63]. These mice presented increased Th1 and T-follicular helper (Tfh) cell responses, as well as overactive B-cells with increased germinal center formation. Functionally, PTEN deficient Tregs lost CD25 and Foxp3 expression and their metabolism was skewed towards glycolysis. Some of the PTEN deficient Tregs even gained an effector phenotype in the presence of high circulating levels of IFN-γ [62, 63].
Mechanistically, all of these changes were attributed to increased mTORC2 and Akt activity. Normally PTEN is the principal negative regulator of the PI3K-AKT axis [59]. These findings demonstrated how PTEN negatively can regulate mTORC2, as well as maintain a commitment to Treg cell lineage and a suppressive function. Furthermore, previous studies showed that mTORC2-driven pathways led to a Th2 phenotype [62, 63], but that increased mTORC2 activity also led to Th1 and Tfh, primarily by IFN-γ [62, 63].
Novel Treg-specific mechanisms relying on environmental and cytosolic cues to restrain mTORC1 have recently been described, and one example is protein phosphatase 2A (PP2A) [58]. Indeed, PP2A signaling was recently shown to be crucial for Treg function. The catalytic activity of PP2A in Tregs from mice was reported to be increased at levels significantly higher than in conventional Tconv cells [58]. In this study, mice deficient in PP2Ac Tregs developed spontaneously severe, progressive, multi-organ autoimmunity characterized by wasting, dermatitis, scaly tails and ears, eyelid crusting and in some occasions overt skin rash and ulcerations. Animals also suffered a lymphoproliferative syndrome with secondary lymphoid organ enlargement. T cells were activated in all lymphoid organs and produced IL-17, while multiple autoantibodies were detected in the serum. Activation of PP2A required the presence of ceramide, and Foxp3 expression led to PP2A accumulation by inhibiting the gene sgms1, whose product is ceramide degrading enzyme sphingomyelinase synthase 1 [58]. Of note, functional Tregs contain high amounts of ceramides, and PP2Ac was demonstrated to inactivate mTORC1 without affecting mTORC2 and PI(3)K/Akt pathways (Figure2). Rapamycin (mTROC1 inhibitor) treatment in vitro of PP2Adeficient Treg cells reduced the production of IL-2 and IL17 and normalized their increased glycolytic and oxidative phosphorylation rates to the levels of PP2Asuficient Treg cells. Finally, in vivo administration of rapamycin to mice lacking PP2A in Tregs reduced leukocyte infiltration in the affected peripheral tissues [58]. Again, this study highlights how intracellular components such as PP2A provide differential signaling to promote Treg functional homeostasis.
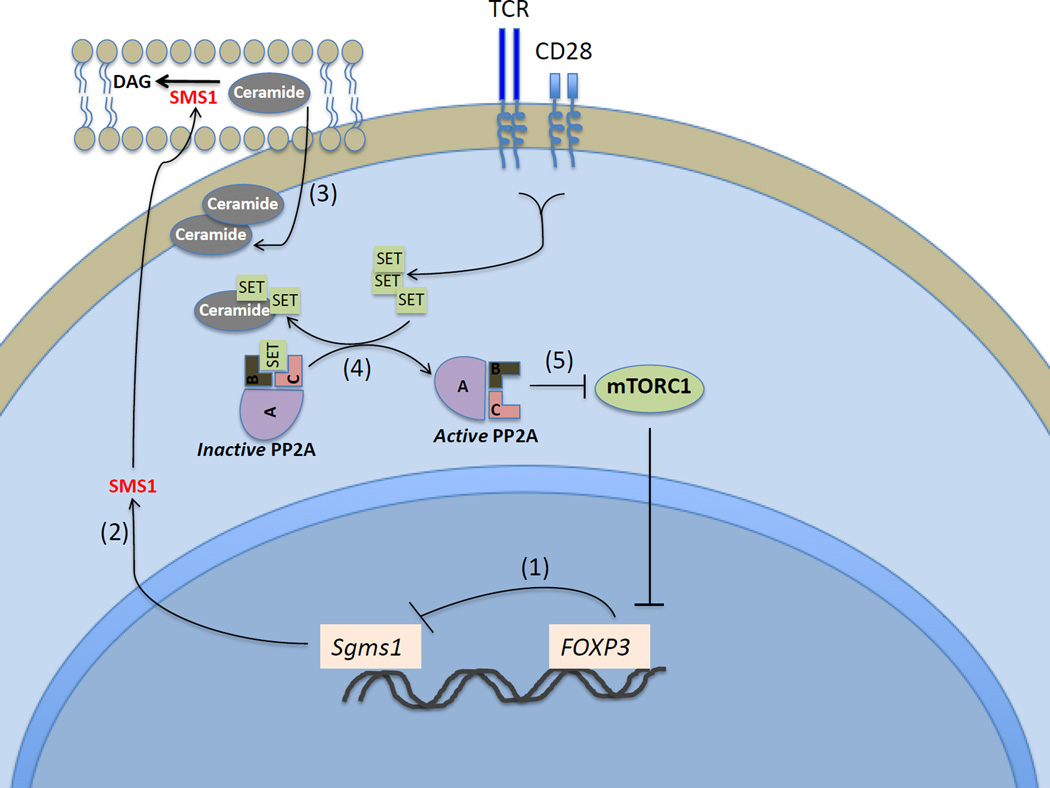
Figure 2. [A2]PP2A Is Required for Treg Function.
(1) Foxp3 inhibits sgms1; (2) SMS1 protein is not produced, therefore ceramide is not degraded, accumulating in the cytoplasm; (3) SET is upregulated upon T cell activation and ceramide binds SET, making it less available to inhibit PP2A. (4) PP2A is activated when unbound from SET (5) Active PP2A inhibits mTORC1 activity, thereby promoting Foxp3 expression.
Repressors of Treg Function
Notch family receptors have been well characterized, directing the differentiation fates of different T cell subsets. Notch signals through two different pathways. In the canonical pathway Notch receptors are cleaved, traffic the nucleus and interact with the recombination signal binding protein-Jk (RBPJ), ultimately leading to expression of target genes. A second (noncanonical) pathway involves mTORC2 and its associated adaptor protein Rictor. In contrast to PTEN, Notch destabilizes Treg function by enhancing mTORC2. [A1]Notch overexpression in Tregs has been reported to cause overactive canonical and non-canonical signals, resulting in systemic autoimmunity in mice [64]. In this study, increased mTORC2 activity ultimately decreased Foxp3 expression, as Foxo1 activity was prevented and could not provide the epigenetic stability of Foxp3. Tregs overexpressing Notch also demonstrated plasticity because they acquired a Th1 phenotype. Conversely, in a separate mouse strain where Notch was deleted in Tregs, animals displayed overall decreased Th1 activity, and Tregs were less prone to undergo apoptosis. Together these results demonstrate that restraining mTORC2 can promote the functional homeostasis of Tregs [64].
From another perspective, Special AT-rich Sequence Binding Protein (SATB1) is a genome organizer expressed in T cells which regulates chromatin structure and gene expression[65]. The acquisition of a functional Treg phenotype was reported to require the repression of SATB1 [65]. Specifically, Foxp3 was shown to suppress the SATB1 locus and a failure to do so resulted in the loss of suppressive function, the establishment of transcriptional effector T cell programs and the induction of effector T cell cytokines. Another set of experiments showed that conditional knockout of SATB1 in the T cell compartment (accomplished through generation of lck-CRE / SATB1(flox/flox) mice), resulted in a significant reduction in both CD4+CD8+ T cells in the thymus and in Tregs, leading to the development of autoimmune disease within 16 weeks after birth [66].
Tregs in Human Autoimmune Diseases
Monogenic Diseases Associated with Loss of Treg Function
Tregs represent the fundamental T-cell capable of promoting self-tolerance and balancing excessive inflammation. Several monogenic conditions illustrate the importance of functioning Tregs to human immune homeostasis. A prototype example is Foxp3 deficiency resulting in IPEX syndrome in males. It is fatal within the first several years of life, unless rescued with bone marrow transplantation. IPEX patients have elevated autoantibodies levels, suggesting significant adaptive immune dysregulation [67] [68] [69].
Other monogenic conditions associated with Treg dysfunction include CD25 deficiency. Humans with this deficiency have both autoimmunity and immunodeficiency, which clinically resembles an IPEX like phenotype. CD25 deficiency does not affect the number of Foxp3+ cells in the circulation, but rather, impairs their suppressive function by diminishing IL-10 production, altering the metabolic fitness of Tregs and losing the “IL-2” sink [70, 71].
STAT5b deficiency also is implicated in the development of autoimmunity as STAT5b has multiple effects essential to Treg function. It stabilizes the expression of FoxP3 by binding the locus; it is required for signal transduction of common gamma-chain cytokines such as IL-2. Clinically, STAT5b deficiency manifests in chronic diarrhea, eczema and lymphoid interstitial pneumonitis [72, 73].
CTLA4 deficiency in mice leads to massive lymphoproliferartion and multiorgan inflammation[74]. Humans with CTLA4 mutations display autosomal dominant immune dysregulation syndrome characterized by hypogammaglobulinemia, recurrent infections and multiple autoimmune clinical features. CTLA4 expression has been show to be decreased in Tregs, compromising their function, although without decreasing their number [75]. Severe Treg cell deficiency, with depressed FoxP3 expression, has been noted in patients who harbor a nonsense mutation in the gene encoding LPS-responsive beige-like anchor (LRBA), known to present common variable immunodeficiency and autoimmunity [76]. LRBA colocalizes with CTLA4 in endosomal vesicles, and LRBA deficiency or knockdown has been reported to lead to increased CTLA4 turnover, resulting in reduced levels of CTLA4 protein in FoxP3+ Tregs and activated conventional T cells [77]. Interestingly, CTLA4-Ig administration corrected cell abnormalities and led to clinical improvement [77].
Tregs in Systemic Autoimmune Diseases
Tregs have been implicated in the pathophysiology of systemic rheumatic diseases, although their role is less defined than in monogenic traits. In SLE there have been several subsets of Tregs identified in the peripheral blood. However, Treg numbers and function have varied considerably [78–81]. This likely reflects the heterogenicity of the disease. In human studies, critical controls have been missed, such as documentation of time of disease onset, therapeutic interventions and state of health (i.e. flares vs. remission). Taking systemic lupus erythematosus (SLE) as an example, other factors have made the study of Tregs in SLE problematic. This has been due in part to differences in experimental protocols, including the isolation of Tregs, flow cytometry settings, and either the presence or absence of antigen presenting cells (APCs) in ex vivo functional assays [78]. Many discrepancies have also stemmed from a lack of unique Treg marker. Taking all of this into account, the majority of reports on human SLE demonstrate aberrant Treg function and abnormal total Treg numbers [78–81]. Furthermore, there are histological changes in the inflamed kidney. In kidney biopsy samples from patients with active proliferative lupus nephritis and anti-neutrophil cytoplasmic antibody associated vasculitis, a study reported a decrease in the number of Foxp3+ to CD3+ cells, relative to patients with hypertensive nephropathy, and pure membranous lupus nephritis class 5 LN [82]. Changes in such numbers might be due in part, to the overall decrease in levels of circulating IL-2, which is required for maintenance of Treg function, but further studies are required to fully address the nature of these differences (82].
Targeting Tregs to Control Autoimmune Diseases
A general therapeutic strategy to control autoimmune diseases might include an effort to maintain the functional integrity of Tregs in the periphery (Table 1). It is reasonable to suppose that this approach might protect the host from aberrant Tregs that react to self-antigen with inflammatory signals, and also, potentially maintain Tregs induced in the peripheral tissue from the pool of naïve CD4+ T cells.
Table 1.
Treg Associated Therapeutic Targets for Autoimmune Diseases
| Therapeutic Target | Drug | Mechanism | Reported Application/ Potential Use |
Reference |
|---|---|---|---|---|
| Cytokine Signaling | Aldesleukin (low-dose IL-2) | STAT5 activator | SLE | [104] |
| HCV related vasculitis | [105] | |||
| Graft versus host disease | [106] | |||
| Kinase/Phosphatase | Rapamycin, | mTORC1 inhibitor | SLE, RA, JIA, SS | [107] |
| Everolimus | mTORC1 inhibitor | RA, SLE | [108] [109] | |
| KN-93 | CaMK IV inhibitor | SLE | [110] | |
| Fingolomid (FTY720) | PP2A activator | SLE, RA, MS | [111] [112] [113] |
|
|
Epigenetic Foxp3 Stabilizers |
5-aza—2’-deoxycytidine | Dnmt1 inhibitor | NR | |
| ACY-738 | HDAC 6 inhibitor | SLE | [114] | |
| Tubastatin A | HDAC 6 inhibitor | RA | [115] | |
| Pyrazolopyrimdine derivatives | FOXO1 Stabilizer | NR | ||
Legend: Systemic lupus erythematosus SLE, Rheumatoid arthritis RA, Juvenile idiopathic arthritis JIA, Sjögren’s syndrome SS, Multiple sclerosis MS, Not Reported NR, Hepatitis C HCV
Pharmacologic blockade of the PI3K-AKT-mTOR axis is also being regarded as having considerable therapeutic potential. Studies have demonstrated the positive effects of restraining mTOR signaling to promote Treg survival and stability. [83, 84]. Although first approved as an immunosuppressant in 1999, rapamycin was later found to expand the Treg population [84], and might potentially provide therapeutic value in patients lacking Foxp3 expression or function.
As outlined earlier, Foxp3 expression is compromised when mTORC2 is activated. Presumably this occurs because mTORC2 prevents Foxo1 from entering the nucleus and stabilizing Foxp3 expression. Therefore, drugs promoting nuclear transloaction of Foxo1 represent another potential target, as in the case of pyrazolopyrimidine derivatives. These compounds not only promote Foxo1 nuclear translocation but simultaneously inhibit P13K, thus targeting the upstream and downstream effects of mTORC2 signaling [85].
Other molecules limiting mTOR activity work by diverse mechanisms of action but include S1P receptor blockers [86] [87], antioxidants such as metformin and N-Acetyl Cysteine [88] [89], or Calmodulin kinase II/IV inhibitors [90–94]. There are also ATP competitive inhibitors of the kinase domain of mTOR such as Torin 2 (reviewed in [53, 95]. Rationale to treat autoimmune conditions with mTOR inhibitors has been further supported by an ever increasing number of positive reports from studies in patients and animal models for SLE, RA and Sjögren’s syndrome [53]. For instance, a prospective clinical trial using rapamycin for the treatment of SLE was completed in 2015, and once collected, the results might provide further insight into the therapeutic efficacy of mTOR inhibitors [53].
While targeting mTORC2 provides epigenetic stability of Foxp3 expression via Foxo1 nuclear translocation, many other epigenetic targets exist. These include but again are not limited to forced CpG demethylization with Dnmt1 inhibitor 5—aza-2-deoxycitidine (5-Aza) which have been administered to subjects in conjunction with rapamycin inhibitors [96, 97]. Other modalities include targeting histone acetalization. With specific HDAC inhibitors (not pan-inhibitors) for example, HDAC 6 inhibitors have selectively enhanced Treg function in models of murine lupus, protecting the mice from developing lupus nephritis. [98, 99].
IL 2 as outlined earlier is essential to the differentiation and function of Tregs. Low dose IL-2 treatment of lupus prone mice ([90] [100]) as well as in a few SLE patients [20] [101] has been found to significantly modulate the severity of the disease. Interestingly, although not involving autoimmune conditions, there have been two promising clinical trials where GVHD and HCV have been successfully treated upon IL-2 treatment. In both of these studies the clinical amelioration was associated with augmented numbers of peripheral Tregs and enhanced suppressive function [102, 103].
From the studies reviewed above there are many potential targets to be exploited, which could enhance Treg function in a cell specific manner. These include augmenting the intracellular concentration of ceramides to diminish mTORC1 signaling. Other potential targets could include interfering with autophagy in cells, but this type of approach should be considered with caution as hydroxychloroquine, (an autophagy inhibitor), is central to the treatment of SLE.
Concluding Remarks
Tregs are essential negative regulators of the immune response. Lack of functional Tregs can lead to the development of systemic autoimmune diseases. Recently, a number of molecules which enforce Treg function have been identified. These may represent novel therapeutic targets to potentially treat certain autoimmune conditions. (Box 4). Although low-dose IL-2 and rapamycin have been used with significantly positive clinical outcomes for certain patients, further IL-2 based trials should be conducted. In addition, many other potential therapeutic targets should be further exploited. One example includes the activation of PP2A in order to increase the intracellular levels of ceramide and restrain mTORC1 signaling. (see Outstanding Questions). These molecules could be potentiallu used not only to stabilize Tregs but also to enhance their suppressive function in conditions such as Juvenile Idiopathic Arthritis and Multiple Sclerosis, where T effector cells are resistant to Treg suppression. It is clear that many studies targeting pathways in Tregs to alleviate autoimmune diseases warrant further validation and study. Nevertheless, molecules that might empower Treg function are very likely to provide exciting new treatment modalities for various autoimmune conditions.
Box 4. The Clinician’s Corner.
Tregs are indispensable in controlling immune responses. Their absence is linked to autoimmunity and inflammation and their function is continuously being assessed in a number of systemic autoimmune conditions such as SLE.
Administration of ex vivo expanded Tregs has been considered for the treatment of autoimmune diseases, but this is a controversial, and not yet established therapy
A number of molecules including Foxo1, mTOR, PTEN, Notch and PP2A are involved in the maintenance of Treg function. Drugs that deliberately increase or decrease the expression of molecules controlling Treg function such as rapamycin, might potentially empower Treg function.
Rare patients who lack functioning molecules such as Foxp3, IL-2 receptor, STAT5B or CTLA harbor defective Tregs and develop severe autoimmune diseases or immunodeficiencies.
References for BOXES
- 1.Chen W, et al. Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J. Exp. Med. 2003;198:1875–1886. doi: 10.1084/jem.20030152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Josefowicz SZ, et al. Regulatory T cells: mechanisms of differentiation and function. Annu. Rev. Immunol. 2012;30:531–564. doi: 10.1146/annurev.immunol.25.022106.141623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hori S, et al. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003;299:1057–1061. [PubMed] [Google Scholar]
- 4.Fontenot JD, et al. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 2003;4:330–336. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 5.Khattri R, et al. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat. Immunol. 2003;4:337–342. [PubMed] [Google Scholar]
- 6.Hsieh CS, et al. An intersection between the self-reactive regulatory and nonregulatory T cell receptor repertoires. Nat. Immunol. 2006;7:401–410. doi: 10.1038/ni1318. [DOI] [PubMed] [Google Scholar]
- 7.Feng Y, et al. A mechanism for expansion of regulatory T-cell repertoire and its role in self-tolerance. Nature. 2015;528:132–136. doi: 10.1038/nature16141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li X, Zheng Y. Regulatory T cell identity: formation and maintenance. Trends Immunol. 2015;36:344–353. doi: 10.1016/j.it.2015.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chaudhry A, et al. CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science. 2009;326:986–991. doi: 10.1126/science.1172702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zheng Y, et al. Regulatory T-cell suppressor program co-opts transcription factor IRF4 to control T(H)2 responses. Nature. 2009;458:351–356. doi: 10.1038/nature07674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koch MA, et al. The transcription factor T-bet controls regulatory T cell homeostasis and function during type 1 inflammation. Nat. Immunol. 2009;10:595–602. doi: 10.1038/ni.1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Y, et al. An essential role of the transcription factor GATA-3 for the function of regulatory T cells. Immunity. 2011;35:337–348. doi: 10.1016/j.immuni.2011.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cipolletta D. Adipose tissue-resident regulatory T cells: phenotypic specialization, functions and therapeutic potential. Immunology. 2014;142:517–525. doi: 10.1111/imm.12262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Arpaia N, et al. A Distinct Function of Regulatory T Cells in Tissue Protection. Cell. 2015;162:1078–1089. doi: 10.1016/j.cell.2015.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wei J, et al. Autophagy enforces functional integrity of regulatory T cells by coupling environmental cues and metabolic homeostasis. Nat. Immunol. 2016;17:277–285. doi: 10.1038/ni.3365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kabat AM, et al. The autophagy gene Atg16l1 differentially regulates Treg and TH2 cells to control intestinal inflammation. Elife. 2016;5 doi: 10.7554/eLife.12444. [DOI] [PMC free article] [PubMed] [Google Scholar]
Outstanding Questions.
Is there a role for ceramides in preventing autoimmunity, given the fact that their concentration is increased in Tregs?
Can we further identify molecules that can regulate cellular signaling and directly impact the epigenetic landscape and function of Tregs?
What other epigenetic strategies can be contemplated to stabilize Foxp3 expression and therefore putatively prevent autoimmunity? Are these feasible? Will they require combination therapies?
Other than rapamycin, could alternative therapies be envisaged and used in combination to synergistically enhance Treg function? And can these be clinically applicable to treat autoimmune diseases?
Box 2. Treg Heterogeneity and Non-suppressive Treg Functions.
Treg heterogeneity
Tregs are a heterogeneous group of cells and in addition to FoxP3, they express transcription factors associated with different T helper (Th) subsets which they specifically suppress. For instance, Tregs expressing Tbet, IRF4 and Stat3 dampen the Th1, Th2 and Th17 responses, respectively [9] [10] [11]. Interestingly, lack of GATA-3, which is a central transcription factor in Th2 cells, has been shown to reduce the expression of Foxp3 [12]. These GATA-3 deficient Tregs were found to acquire a Th17 phenotype, ultimately leading to the development of a spontaneous inflammatory disease in mice [12].
Non-suppressive Treg Functions
There are also well defined populations of tissue resident Tregs vital to tissue homeostasis; Tregs that express peroxisome proliferator activated receptor gamma (PPARg) have been implicated in metabolic homeostasis in visceral adipose tissue [13]. Another study has indicated that Tregs express amphiregulin and have been reported to contribute to preventing severe lung injury after Influenza viral infection in mice. Notably, absence of amphiregulin was not found to affect the suppressor function of these Tregs [14].
Trends.
After differentiation in either the thymus or at peripheral sites Tregs are capable of losing Foxp3 expression and producing pro-inflammatory cytokines potentially contributing to autoimmunity and inflammation.
There are many epigenetic and transcriptional programs driven by the T cell receptor and interleukin-2 to protect the expression of Foxp3 in an inflammatory setting. Many new potential therapies to treat autoimmune or inflammatory conditions are aimed at maintaining epigenetic stability.
The PI3K-Akt-mTOR pathway is differentially modulated in Tregs compared to effector T-cells and represents a promising therapeutic target to functionally empower Tregs to suppress inflammation.
New and important insights have been obtained regarding the requirement of phosphatases and kinases in integrating cellular and metabolic signals within Tregs during autoimmune diseases.
Acknowledgments
Funding was provided by NIH grant RO1 AI068787 (GCT), and by NIH T32AI074549 training grant (IRK).
Glossary
- Scurfy Mice
Mouse strain developing spontaneous fatal lymphoproliferative autoimmune disease due to a missense mutation within the foxp3 gene, resulting in a lack of CD4+Foxp3+ Tregs.
- IPEX
An X-linked familial autoimmune syndrome caused by mutations in the FOXP3 locus; clinically characterized by immune dysregulation, polyendocrinopathy and enteropathy.
- Conventional T Cells
T lymphocytes that express a TCR as well as a CD4 or CD8 co-receptor.
- ex-Tregs
Tregs that have lost Foxp3 expression, suppressive function and which produce proinflammatory cytokines.
- Pyrazolopyrimidine derivatives
Compounds that promote Foxo1 nuclear translocation and simultaneously inhibit P13K, thus diminishing the upstream and downstream effects of mTORC2 signaling.
- CNS (2&3)
Conserved non-coding DNA sequences at the foxp3 locus that stabilize Tregs by either inducing Foxp3 expression (CNS 3) or maintaining Foxp3 expression (CNS 2).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Nishizuka Y, Sakakura T. Thymus and reproduction: sex-linked dysgenesia of the gonad after neonatal thymectomy in mice. Science. 1969;166:753–755. doi: 10.1126/science.166.3906.753. [DOI] [PubMed] [Google Scholar]
- 2.Malek TR. The biology of interleukin-2. Annu. Rev. Immunol. 2008;26:453–479. doi: 10.1146/annurev.immunol.26.021607.090357. [DOI] [PubMed] [Google Scholar]
- 3.Suzuki H, et al. Deregulated T cell activation and autoimmunity in mice lacking interleukin-2 receptor beta. Science. 1995;268:1472–1476. doi: 10.1126/science.7770771. [DOI] [PubMed] [Google Scholar]
- 4.Willerford DM, et al. Interleukin-2 receptor alpha chain regulates the size and content of the peripheral lymphoid compartment. Immunity. 1995;3:521–530. doi: 10.1016/1074-7613(95)90180-9. [DOI] [PubMed] [Google Scholar]
- 5.Sakaguchi S, et al. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. 1995;155:1151–1164. [PubMed] [Google Scholar]
- 6.Josefowicz SZ, et al. Regulatory T cells: mechanisms of differentiation and function. Annu. Rev. Immunol. 2012;30:531–564. doi: 10.1146/annurev.immunol.25.022106.141623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Miyao T, et al. Plasticity of Foxp3(+) T cells reflects promiscuous Foxp3 expression in conventional T cells but not reprogramming of regulatory T cells. Immunity. 2012;36:262–275. doi: 10.1016/j.immuni.2011.12.012. [DOI] [PubMed] [Google Scholar]
- 8.Allan SE, et al. Activation-induced FOXP3 in human T effector cells does not suppress proliferation or cytokine production. Int. Immunol. 2007;19:345–354. doi: 10.1093/intimm/dxm014. [DOI] [PubMed] [Google Scholar]
- 9.Russell WL, et al. Exceptional Inheritance of a Sex-Linked Gene in the Mouse Explained on the Basis That the X/O Sex-Chromosome Constitution Is Female. Proc. Natl. Acad. Sci. U. S. A. 1959;45:554–560. doi: 10.1073/pnas.45.4.554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fontenot JD, et al. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 2003;4:330–336. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 11.Hori S, et al. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003;299:1057–1061. [PubMed] [Google Scholar]
- 12.Bennett CL, et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 2001;27:20–21. doi: 10.1038/83713. [DOI] [PubMed] [Google Scholar]
- 13.Powell BR, et al. An X-linked syndrome of diarrhea, polyendocrinopathy, and fatal infection in infancy. J. Pediatr. 1982;100:731–737. doi: 10.1016/s0022-3476(82)80573-8. [DOI] [PubMed] [Google Scholar]
- 14.Wildin RS, et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 2001;27:18–20. doi: 10.1038/83707. [DOI] [PubMed] [Google Scholar]
- 15.Fontenot JD, et al. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat. Immunol. 2005;6:1142–1151. doi: 10.1038/ni1263. [DOI] [PubMed] [Google Scholar]
- 16.Barron L, et al. Cutting edge: mechanisms of IL-2-dependent maintenance of functional regulatory T cells. J. Immunol. 2010;185:6426–6430. doi: 10.4049/jimmunol.0903940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Malek TR, et al. CD4 regulatory T cells prevent lethal autoimmunity in IL-2Rbeta-deficient mice. Implications for the nonredundant function of IL-2. Immunity. 2002;17:167–178. doi: 10.1016/s1074-7613(02)00367-9. [DOI] [PubMed] [Google Scholar]
- 18.Burchill MA, et al. Distinct effects of STAT5 activation on CD4+ and CD8+ T cell homeostasis: development of CD4+CD25+ regulatory T cells versus CD8+ memory T cells. J. Immunol. 2003;171:5853–5864. doi: 10.4049/jimmunol.171.11.5853. [DOI] [PubMed] [Google Scholar]
- 19.Klatzmann D, Abbas AK. The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat. Rev. Immunol. 2015;15:283–294. doi: 10.1038/nri3823. [DOI] [PubMed] [Google Scholar]
- 20.von Spee-Mayer C, et al. Low-dose interleukin-2 selectively corrects regulatory T cell defects in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 2015 doi: 10.1136/annrheumdis-2015-207776. [DOI] [PubMed] [Google Scholar]
- 21.Liu Y, et al. A critical function for TGF-[beta] signaling in the development of natural CD4+CD25+Foxp3+ regulatory T cells. Nat. Immunol. 2008;9:632–640. doi: 10.1038/ni.1607. [DOI] [PubMed] [Google Scholar]
- 22.Li MO, Rudensky AY. T cell receptor signalling in the control of regulatory T cell differentiation and function. Nat. Rev. Immunol. 2016;16:220–233. doi: 10.1038/nri.2016.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ouyang W, et al. Transforming Growth Factor-β Signaling Curbs Thymic Negative Selection Promoting Regulatory T Cell Development. Immunity. 2010;32:642–653. doi: 10.1016/j.immuni.2010.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rao S, et al. c-Rel is required for chromatin remodeling across the IL-2 gene promoter. J. Immunol. 2003;170:3724–3731. doi: 10.4049/jimmunol.170.7.3724. [DOI] [PubMed] [Google Scholar]
- 25.Zheng Y, et al. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature. 2010;463:808–812. doi: 10.1038/nature08750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Feng Y, et al. A mechanism for expansion of regulatory T-cell repertoire and its role in self-tolerance. Nature. 2015;528:132–136. doi: 10.1038/nature16141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yurchenko E, et al. Inflammation-driven reprogramming of CD4+ Foxp3+ regulatory T cells into pathogenic Th1/Th17 T effectors is abrogated by mTOR inhibition in vivo. PLoS One. 2012;7:e35572. doi: 10.1371/journal.pone.0035572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duarte JH, et al. Natural Treg cells spontaneously differentiate into pathogenic helper cells in lymphopenic conditions. Eur. J. Immunol. 2009;39:948–955. doi: 10.1002/eji.200839196. [DOI] [PubMed] [Google Scholar]
- 29.Komatsu N, et al. Heterogeneity of natural Foxp3+ T cells: a committed regulatory T-cell lineage and an uncommitted minor population retaining plasticity. Proc. Natl. Acad. Sci. U. S. A. 2009;106:1903–1908. doi: 10.1073/pnas.0811556106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhou X, et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nat. Immunol. 2009;10:1000–1007. doi: 10.1038/ni.1774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hori S. Lineage stability and phenotypic plasticity of Foxp3(+) regulatory T cells. Immunol. Rev. 2014;259:159–172. doi: 10.1111/imr.12175. [DOI] [PubMed] [Google Scholar]
- 32.Burchill MA, et al. Linked T Cell Receptor and Cytokine Signaling Govern the Development of the Regulatory T Cell Repertoire. Immunity. 2008;28:112–121. doi: 10.1016/j.immuni.2007.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Feng Y, et al. Control of the inheritance of regulatory T cell identity by a cis element in the Foxp3 locus. Cell. 2014;158:749–763. doi: 10.1016/j.cell.2014.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Li X, et al. Function of a Foxp3 cis-element in protecting regulatory T cell identity. Cell. 2014;158:734–748. doi: 10.1016/j.cell.2014.07.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rubtsov YP, et al. Stability of the regulatory T cell lineage in vivo. Science. 2010;329:1667–1671. doi: 10.1126/science.1191996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Floess S, et al. Epigenetic control of the foxp3 locus in regulatory T cells. PLoS Biol. 2007;5:e38. doi: 10.1371/journal.pbio.0050038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang L, et al. Foxp3+ T-regulatory cells require DNA methyltransferase 1 expression to prevent development of lethal autoimmunity. Blood. 2013;121:3631–3639. doi: 10.1182/blood-2012-08-451765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nair VS, Oh KI. Down-regulation of Tet2 prevents TSDR demethylation in IL2 deficient regulatory T cells. Biochem. Biophys. Res. Commun. 2014;450:918–924. doi: 10.1016/j.bbrc.2014.06.110. [DOI] [PubMed] [Google Scholar]
- 39.DuPage M, et al. The chromatin-modifying enzyme Ezh2 is critical for the maintenance of regulatory T cell identity after activation. Immunity. 2015;42:227–238. doi: 10.1016/j.immuni.2015.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Morikawa H, et al. Differential roles of epigenetic changes and Foxp3 expression in regulatory T cell-specific transcriptional regulation. Proc. Natl. Acad. Sci. U. S. A. 2014;111:5289–5294. doi: 10.1073/pnas.1312717110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van Loosdregt J, et al. Stabilization of the transcription factor Foxp3 by the deubiquitinase USP7 increases Treg-cell-suppressive capacity. Immunity. 2013;39:259–271. doi: 10.1016/j.immuni.2013.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen Z, et al. The ubiquitin ligase Stub1 negatively modulates regulatory T cell suppressive activity by promoting degradation of the transcription factor Foxp3. Immunity. 2013;39:272–285. doi: 10.1016/j.immuni.2013.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhao Y, et al. E3 Ubiquitin Ligase Cbl-b Regulates Thymic-Derived CD4+CD25+ Regulatory T Cell Development by Targeting Foxp3 for Ubiquitination. J. Immunol. 2015;194:1639–1645. doi: 10.4049/jimmunol.1402434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Samstein RM, et al. Foxp3 exploits a pre-existent enhancer landscape for regulatory T cell lineage specification. Cell. 2012;151:153–166. doi: 10.1016/j.cell.2012.06.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ouyang W, et al. Novel Foxo1-dependent transcriptional programs control T(reg) cell function. Nature. 2012;491:554–559. doi: 10.1038/nature11581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Roychoudhuri R, et al. BACH2 represses effector programs to stabilize T(reg)-mediated immune homeostasis. Nature. 2013;498:506–510. doi: 10.1038/nature12199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kim EH, et al. Bach2 regulates homeostasis of Foxp3+ regulatory T cells and protects against fatal lung disease in mice. J. Immunol. 2014;192:985–995. doi: 10.4049/jimmunol.1302378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Thornton AM, et al. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J. Immunol. 2010;184:3433–3441. doi: 10.4049/jimmunol.0904028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kim HJ, et al. Stable inhibitory activity of regulatory T cells requires the transcription factor Helios. Science. 2015;350:334–339. doi: 10.1126/science.aad0616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nakagawa H, et al. Instability of Helios-deficient Tregs is associated with conversion to a T-effector phenotype and enhanced antitumor immunity. Proc. Natl. Acad. Sci. U. S. A. 2016;113:6248–6253. doi: 10.1073/pnas.1604765113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Takatori H, et al. Helios Enhances Treg Cell Function in Cooperation With FoxP3. Arthritis Rheumatol. 2015;67:1491–1502. doi: 10.1002/art.39091. [DOI] [PubMed] [Google Scholar]
- 52.Spence A, et al. Targeting Treg signaling for the treatment of autoimmune diseases. Curr. Opin. Immunol. 2015;37:11–20. doi: 10.1016/j.coi.2015.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Perl A. Activation of mTOR (mechanistic target of rapamycin) in rheumatic diseases. Nat Rev Rheumatol. 2016;12:169–182. doi: 10.1038/nrrheum.2015.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Delgoffe GM, et al. The kinase mTOR regulates the differentiation of helper T cells through the selective activation of signaling by mTORC1 and mTORC2. Nat. Immunol. 2011;12:295–303. doi: 10.1038/ni.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Coe DJ, et al. Metabolic regulation of regulatory T cell development and function. Front. Immunol. 2014;5:590. doi: 10.3389/fimmu.2014.00590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Michalek RD, et al. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 2011;186:3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zeng H, et al. mTORC1 couples immune signals and metabolic programming to establish T(reg)-cell function. Nature. 2013;499:485–490. doi: 10.1038/nature12297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Apostolidis SA, et al. Phosphatase PP2A is requisite for the function of regulatory T cells. Nat. Immunol. 2016;17:556–564. doi: 10.1038/ni.3390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Powell JD, et al. Regulation of immune responses by mTOR. Annu. Rev. Immunol. 2012;30:39–68. doi: 10.1146/annurev-immunol-020711-075024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Walsh PT, et al. PTEN inhibits IL-2 receptor-mediated expansion of CD4+ CD25+ Tregs. J. Clin. Invest. 2006;116:2521–2531. doi: 10.1172/JCI28057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Delgoffe GM, et al. Stability and function of regulatory T cells is maintained by a neuropilin-1-semaphorin-4a axis. Nature. 2013;501:252–256. doi: 10.1038/nature12428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Shrestha S, et al. Treg cells require the phosphatase PTEN to restrain TH1 and TFH cell responses. Nat. Immunol. 2015;16:178–187. doi: 10.1038/ni.3076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Huynh A, et al. Control of PI(3) kinase in Treg cells maintains homeostasis and lineage stability. Nat. Immunol. 2015;16:188–196. doi: 10.1038/ni.3077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Charbonnier LM, et al. Control of peripheral tolerance by regulatory T cell-intrinsic Notch signaling. Nat. Immunol. 2015;16:1162–1173. doi: 10.1038/ni.3288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Beyer M, et al. Repression of the genome organizer SATB1 in regulatory T cells is required for suppressive function and inhibition of effector differentiation. Nat. Immunol. 2011;12:898–907. doi: 10.1038/ni.2084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kondo M, et al. SATB1 Plays a Critical Role in Establishment of Immune Tolerance. J. Immunol. 2016;196:563–572. doi: 10.4049/jimmunol.1501429. [DOI] [PubMed] [Google Scholar]
- 67.Torgerson TR, Ochs HD. Immune dysregulation, polyendocrinopathy, enteropathy, X-linked: forkhead box protein 3 mutations and lack of regulatory T cells. J. Allergy Clin. Immunol. 2007;120:744–750. doi: 10.1016/j.jaci.2007.08.044. quiz 751-742. [DOI] [PubMed] [Google Scholar]
- 68.Gambineri E, et al. Clinical and molecular profile of a new series of patients with immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome: inconsistent correlation between forkhead box protein 3 expression and disease severity. J. Allergy Clin. Immunol. 2008;122:1105–1112. e1101. doi: 10.1016/j.jaci.2008.09.027. [DOI] [PubMed] [Google Scholar]
- 69.Kucuk ZY, et al. A challenging undertaking: Stem cell transplantation for immune dysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome. J. Allergy Clin. Immunol. 2016;137:953–955. e954. doi: 10.1016/j.jaci.2015.09.030. [DOI] [PubMed] [Google Scholar]
- 70.Sharfe N, et al. Human immune disorder arising from mutation of the alpha chain of the interleukin-2 receptor. Proc. Natl. Acad. Sci. U. S. A. 1997;94:3168–3171. doi: 10.1073/pnas.94.7.3168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Caudy AA, et al. CD25 deficiency causes an immune dysregulation, polyendocrinopathy, enteropathy, X-linked-like syndrome, and defective IL-10 expression from CD4 lymphocytes. J. Allergy Clin. Immunol. 2007;119:482–487. doi: 10.1016/j.jaci.2006.10.007. [DOI] [PubMed] [Google Scholar]
- 72.Bernasconi A, et al. Characterization of immunodeficiency in a patient with growth hormone insensitivity secondary to a novel STAT5b gene mutation. Pediatrics. 2006;118:e1584–e1592. doi: 10.1542/peds.2005-2882. [DOI] [PubMed] [Google Scholar]
- 73.Nadeau K, et al. STAT5b deficiency: an unsuspected cause of growth failure, immunodeficiency, and severe pulmonary disease. J. Pediatr. 2011;158:701–708. doi: 10.1016/j.jpeds.2010.12.042. [DOI] [PubMed] [Google Scholar]
- 74.Tivol EA, et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995;3:541–547. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 75.Schubert D, et al. Autosomal dominant immune dysregulation syndrome in humans with CTLA4 mutations. Nat. Med. 2014;20:1410–1416. doi: 10.1038/nm.3746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Charbonnier LM, et al. Regulatory T-cell deficiency and immune dysregulation, polyendocrinopathy, enteropathy, X-linked-like disorder caused by loss-of-function mutations in LRBA. J. Allergy Clin. Immunol. 2015;135:217–227. doi: 10.1016/j.jaci.2014.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lo B, et al. AUTOIMMUNE DISEASE. Patients with LRBA deficiency show CTLA4 loss and immune dysregulation responsive to abatacept therapy. Science. 2015;349:436–440. doi: 10.1126/science.aaa1663. [DOI] [PubMed] [Google Scholar]
- 78.Ohl K, Tenbrock K. Regulatory T cells in systemic lupus erythematosus. Eur. J. Immunol. 2015;45:344–355. doi: 10.1002/eji.201344280. [DOI] [PubMed] [Google Scholar]
- 79.Lyssuk EY, et al. Reduced number and function of CD4+CD25highFoxP3+ regulatory T cells in patients with systemic lupus erythematosus. Adv. Exp. Med. Biol. 2007;601:113–119. [PubMed] [Google Scholar]
- 80.Bonelli M, et al. Quantitative and qualitative deficiencies of regulatory T cells in patients with systemic lupus erythematosus (SLE) Int. Immunol. 2008;20:861–868. doi: 10.1093/intimm/dxn044. [DOI] [PubMed] [Google Scholar]
- 81.Chowdary Venigalla RK, et al. Reduced CD4+,CD25− T cell sensitivity to the suppressive function of CD4+,CD25high,CD127−/low regulatory T cells in patients with active systemic lupus erythematosus. Arthritis Rheum. 2008;58:2120–2130. doi: 10.1002/art.23556. [DOI] [PubMed] [Google Scholar]
- 82.Afeltra A, et al. The involvement of T regulatory lymphocytes in a cohort of lupus nephritis patients: a pilot study. Intern. Emerg. Med. 2015;10:677–683. doi: 10.1007/s11739-015-1212-x. [DOI] [PubMed] [Google Scholar]
- 83.Daniel C, et al. Enhancement of antigen-specific Treg vaccination in vivo. Proc. Natl. Acad. Sci. U. S. A. 2010;107:16246–16251. doi: 10.1073/pnas.1007422107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Battaglia M, et al. Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood. 2005;105:4743–4748. doi: 10.1182/blood-2004-10-3932. [DOI] [PubMed] [Google Scholar]
- 85.Link W, et al. Chemical interrogation of FOXO3a nuclear translocation identifies potent and selective inhibitors of phosphoinositide 3-kinases. J. Biol. Chem. 2009;284:28392–28400. doi: 10.1074/jbc.M109.038984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Liu G, et al. The S1P1-mTOR axis directs the reciprocal differentiation of TH1 and Treg cells. Nat. Immunol. 2010;11:1047–1056. doi: 10.1038/ni.1939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Liu G, et al. The receptor S1P1 overrides regulatory T cell-mediated immune suppression through Akt-mTOR. Nat. Immunol. 2009;10:769–777. doi: 10.1038/ni.1743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lai ZW, et al. N-acetylcysteine reduces disease activity by blocking mammalian target of rapamycin in T cells from systemic lupus erythematosus patients: a randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2012;64:2937–2946. doi: 10.1002/art.34502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yin Y, et al. Normalization of CD4+ T cell metabolism reverses lupus. Sci. Transl. Med. 2015;7:274ra218. doi: 10.1126/scitranslmed.aaa0835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Koga T, et al. KN-93, an inhibitor of calcium/calmodulin-dependent protein kinase IV, promotes generation and function of Foxp3(+) regulatory T cells in MRL/lpr mice. Autoimmunity. 2014;47:445–450. doi: 10.3109/08916934.2014.915954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Ichinose K, et al. Suppression of autoimmunity and organ pathology in lupus-prone mice upon inhibition of calcium/calmodulin-dependent protein kinase type IV. Arthritis Rheum. 2011;63:523–529. doi: 10.1002/art.30085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Koga T, et al. CaMK4-dependent activation of AKT/mTOR and CREM-α underlies autoimmunity-associated Th17 imbalance. The Journal of Clinical Investigation. 124:2234–2245. doi: 10.1172/JCI73411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fujikawa K, et al. Calcium/calmodulin-dependent protein kinase II (CaMKII) regulates tumour necrosis factor-related apoptosis inducing ligand (TRAIL)-mediated apoptosis of fibroblast-like synovial cells (FLS) by phosphorylation of Akt. Clin. Exp. Rheumatol. 2009;27:952–957. [PubMed] [Google Scholar]
- 94.Westra J, et al. Expression and regulation of HIF-1alpha in macrophages under inflammatory conditions; significant reduction of VEGF by CaMKII inhibitor. BMC Musculoskelet. Disord. 2010;11:1–11. doi: 10.1186/1471-2474-11-61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Benjamin D, et al. Rapamycin passes the torch: a new generation of mTOR inhibitors. Nat Rev Drug Discov. 2011;10:868–880. doi: 10.1038/nrd3531. [DOI] [PubMed] [Google Scholar]
- 96.Polansky JK, et al. DNA methylation controls Foxp3 gene expression. Eur. J. Immunol. 2008;38:1654–1663. doi: 10.1002/eji.200838105. [DOI] [PubMed] [Google Scholar]
- 97.Josefowicz SZ, et al. Cutting edge: TCR stimulation is sufficient for induction of Foxp3 expression in the absence of DNA methyltransferase 1. J. Immunol. 2009;182:6648–6652. doi: 10.4049/jimmunol.0803320. [DOI] [PubMed] [Google Scholar]
- 98.Hancock WW, et al. HDAC inhibitor therapy in autoimmunity and transplantation. Ann. Rheum. Dis. 2012;71(Suppl 2):i46–i54. doi: 10.1136/annrheumdis-2011-200593. [DOI] [PubMed] [Google Scholar]
- 99.Regna NL, et al. Specific HDAC6 inhibition by ACY-738 reduces SLE pathogenesis in NZB/W mice. Clin. Immunol. 2016;162:58–73. doi: 10.1016/j.clim.2015.11.007. [DOI] [PubMed] [Google Scholar]
- 100.Mizui M, et al. IL-2 protects lupus-prone mice from multiple end-organ damage by limiting CD4-CD8- IL-17-producing T cells. J. Immunol. 2014;193:2168–2177. doi: 10.4049/jimmunol.1400977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Humrich JY, et al. Rapid induction of clinical remission by low-dose interleukin-2 in a patient with refractory SLE. Ann. Rheum. Dis. 2015;74:791–792. doi: 10.1136/annrheumdis-2014-206506. [DOI] [PubMed] [Google Scholar]
- 102.Saadoun D, et al. Regulatory T-cell responses to low-dose interleukin-2 in HCV-induced vasculitis. N. Engl. J. Med. 2011;365:2067–2077. doi: 10.1056/NEJMoa1105143. [DOI] [PubMed] [Google Scholar]
- 103.Koreth J, et al. Interleukin-2 and regulatory T cells in graft-versus-host disease. N. Engl. J. Med. 2011;365:2055–2066. doi: 10.1056/NEJMoa1108188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.von Spee-Mayer C, et al. Low-dose interleukin-2 selectively corrects regulatory T cell defects in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 2015 doi: 10.1136/annrheumdis-2015-207776. [DOI] [PubMed] [Google Scholar]
- 105.Saadoun D, et al. Regulatory T-cell responses to low-dose interleukin-2 in HCV-induced vasculitis. N. Engl. J. Med. 2011;365:2067–2077. doi: 10.1056/NEJMoa1105143. [DOI] [PubMed] [Google Scholar]
- 106.Koreth J, et al. Interleukin-2 and regulatory T cells in graft-versus-host disease. N. Engl. J. Med. 2011;365:2055–2066. doi: 10.1056/NEJMoa1108188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Perl A. Activation of mTOR (mechanistic target of rapamycin) in rheumatic diseases. Nat Rev Rheumatol. 2016;12:169–182. doi: 10.1038/nrrheum.2015.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Bruyn GA, et al. Everolimus in patients with rheumatoid arthritis receiving concomitant methotrexate: a 3-month, double-blind, randomised, placebo-controlled, parallel-group, proof-of-concept study. Ann. Rheum. Dis. 2008;67:1090–1095. doi: 10.1136/ard.2007.078808. [DOI] [PubMed] [Google Scholar]
- 109.Yap DY, et al. Proliferation signal inhibitors in the treatment of lupus nephritis: preliminary experience. Nephrology (Carlton) 2012;17:676–680. doi: 10.1111/j.1440-1797.2012.01646.x. [DOI] [PubMed] [Google Scholar]
- 110.Koga T, et al. KN-93, an inhibitor of calcium/calmodulin-dependent protein kinase IV, promotes generation and function of Foxp3(+) regulatory T cells in MRL/lpr mice. Autoimmunity. 2014;47:445–450. doi: 10.3109/08916934.2014.915954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ando S, et al. FTY720 exerts a survival advantage through the prevention of end-stage glomerular inflammation in lupus-prone BXSB mice. Biochem. Biophys. Res. Commun. 2010;394:804–810. doi: 10.1016/j.bbrc.2010.03.078. [DOI] [PubMed] [Google Scholar]
- 112.Han Y, et al. FTY720 Abrogates Collagen-Induced Arthritis by Hindering Dendritic Cell Migration to Local Lymph Nodes. J. Immunol. 2015;195:4126–4135. doi: 10.4049/jimmunol.1401842. [DOI] [PubMed] [Google Scholar]
- 113.La Mantia L, et al. Fingolimod for relapsing-remitting multiple sclerosis. Cochrane Database Syst Rev. 2016;4:CD009371. doi: 10.1002/14651858.CD009371.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Regna NL, et al. Specific HDAC6 inhibition by ACY-738 reduces SLE pathogenesis in NZB/W mice. Clin. Immunol. 2016;162:58–73. doi: 10.1016/j.clim.2015.11.007. [DOI] [PubMed] [Google Scholar]
- 115.Lee J, et al. A novel histone deacetylase 6-selective inhibitor suppresses synovial inflammation and joint destruction in a collagen antibody-induced arthritis mouse model. Int. J. Rheum. Dis. 2015;18:514–523. doi: 10.1111/1756-185X.12501. [DOI] [PubMed] [Google Scholar]