Abstract
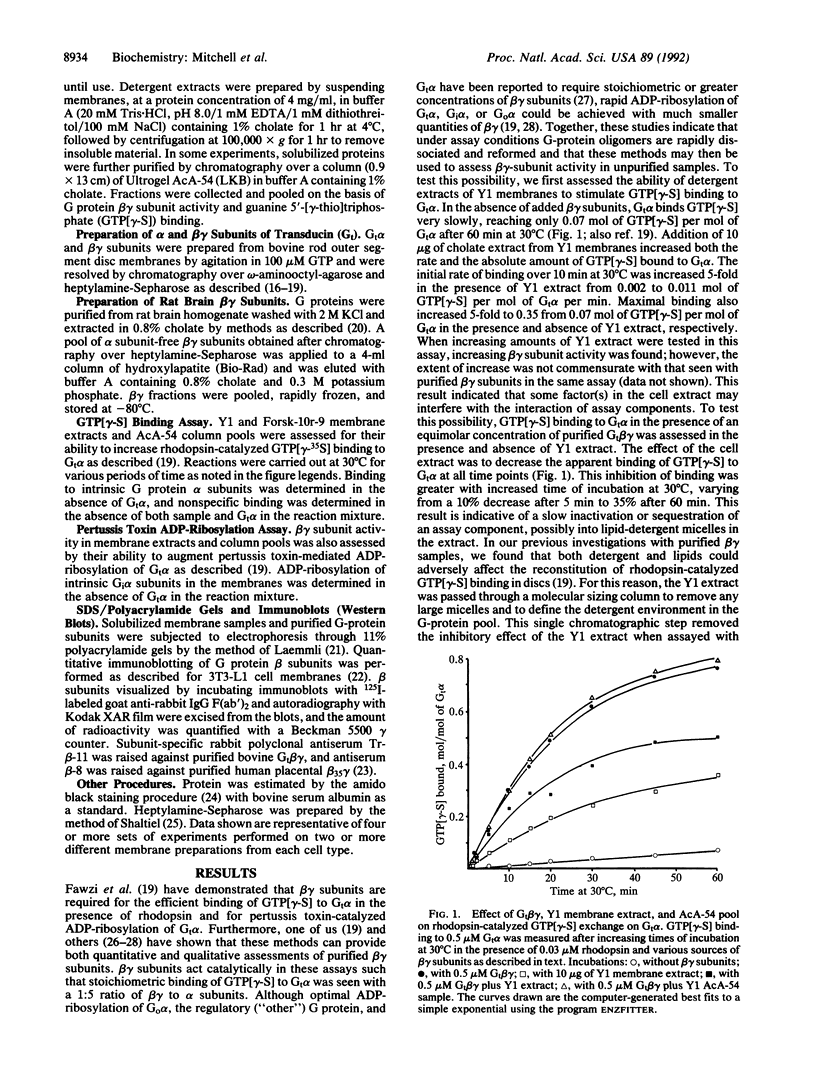
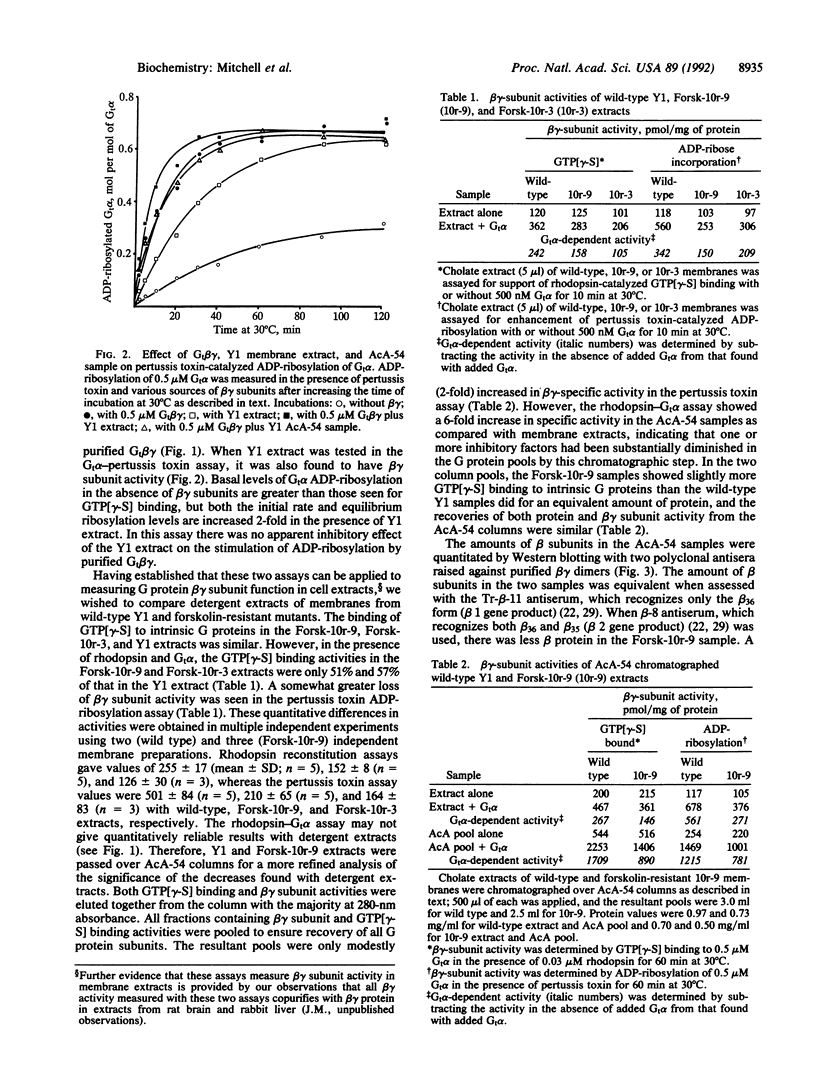
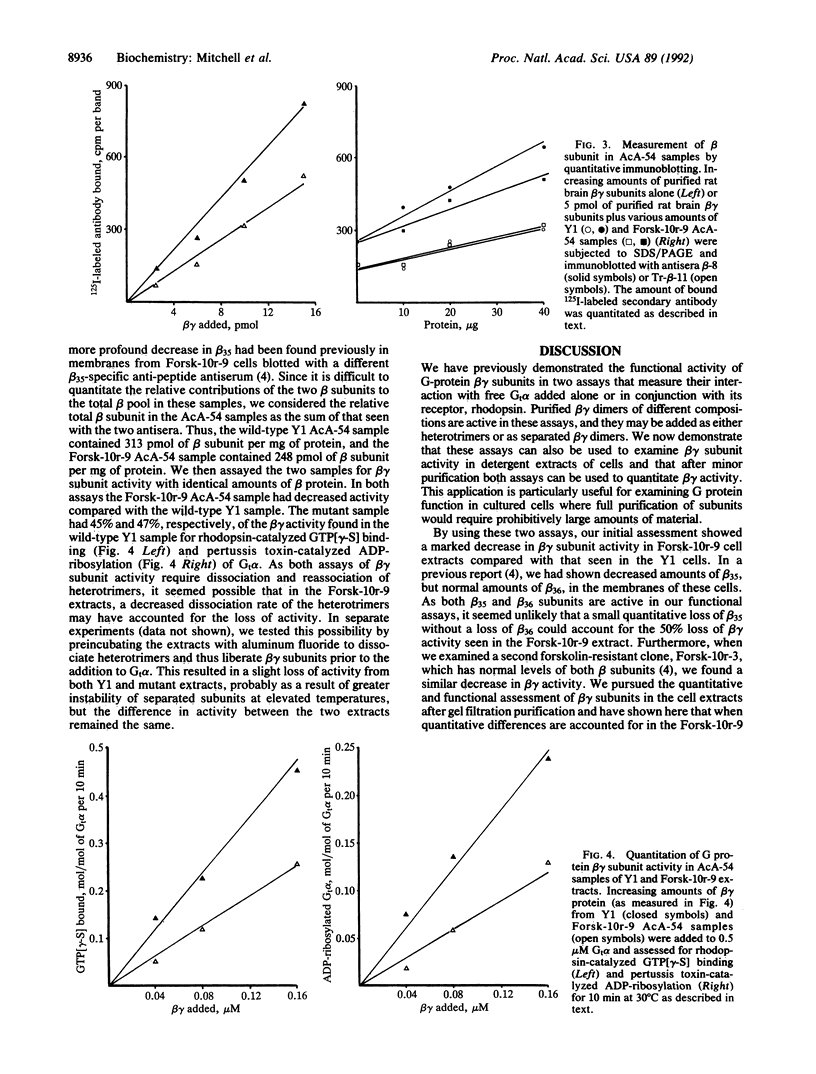
Forskolin-resistant mutants derived from Y1 adrenocortical cells display decreased responsiveness both to receptor and postreceptor stimulators of adenylyl cyclase and decreased amounts of the alpha subunits of the GTP-binding proteins (G proteins) that mediate stimulation (Gs) and inhibition (Gi) of adenylyl cyclase--namely, Gs alpha and Gi alpha-2. This phenotype is suggestive of a mutation that affects the processing or plasma membrane incorporation of G protein alpha subunits. Since the membrane attachment of heterotrimeric G proteins has been ascribed in part to the beta gamma subunits, we examined the quantity and functional activity of beta gamma subunits in wild-type Y1 and forskolin-resistant Forsk-10r-9 and Forsk-10r-3 cells. We now show that two assays previously used to examine the activity of purified beta gamma subunits--namely, to support either rhodopsin-catalyzed guanyl nucleotide exchange on Gt alpha or pertussis toxin-catalyzed ADP-ribosylation of Gt alpha--can be used with detergent extracts of cells. In both assays the beta gamma activity in Forsk-10r-9 and Forsk-10r-3 extracts was decreased by 53-76% compared with wild-type Y1 extracts. When normalized for immunoreactive beta subunit, the beta gamma activity in the Forsk-10r-9 samples was decreased by 55-57% compared with the wild-type Y1 samples. These results suggest that a mutation of one of the G protein beta or gamma subunits may result in the multiple defects of adenylyl cyclase activity and apparent loss of G protein alpha subunits seen in the forskolin-resistant mutant cells. The frequency with which these spontaneous mutations arise in the Y1 cell line suggests that they may contribute more generally to genetic abnormalities in signal transduction.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Buss J. E., Mumby S. M., Casey P. J., Gilman A. G., Sefton B. M. Myristoylated alpha subunits of guanine nucleotide-binding regulatory proteins. Proc Natl Acad Sci U S A. 1987 Nov;84(21):7493–7497. doi: 10.1073/pnas.84.21.7493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey P. J., Graziano M. P., Gilman A. G. G protein beta gamma subunits from bovine brain and retina: equivalent catalytic support of ADP-ribosylation of alpha subunits by pertussis toxin but differential interactions with Gs alpha. Biochemistry. 1989 Jan 24;28(2):611–616. doi: 10.1021/bi00428a029. [DOI] [PubMed] [Google Scholar]
- Evans T., Fawzi A., Fraser E. D., Brown M. L., Northup J. K. Purification of a beta 35 form of the beta gamma complex common to G-proteins from human placental membranes. J Biol Chem. 1987 Jan 5;262(1):176–181. [PubMed] [Google Scholar]
- Fawzi A. B., Fay D. S., Murphy E. A., Tamir H., Erdos J. J., Northup J. K. Rhodopsin and the retinal G-protein distinguish among G-protein beta gamma subunit forms. J Biol Chem. 1991 Jul 5;266(19):12194–12200. [PubMed] [Google Scholar]
- Fawzi A. B., Northup J. K. Guanine nucleotide binding characteristics of transducin: essential role of rhodopsin for rapid exchange of guanine nucleotides. Biochemistry. 1990 Apr 17;29(15):3804–3812. doi: 10.1021/bi00467a030. [DOI] [PubMed] [Google Scholar]
- Florio V. A., Sternweis P. C. Mechanisms of muscarinic receptor action on Go in reconstituted phospholipid vesicles. J Biol Chem. 1989 Mar 5;264(7):3909–3915. [PubMed] [Google Scholar]
- Fung B. K. Characterization of transducin from bovine retinal rod outer segments. I. Separation and reconstitution of the subunits. J Biol Chem. 1983 Sep 10;258(17):10495–10502. [PubMed] [Google Scholar]
- Gautam N., Northup J., Tamir H., Simon M. I. G protein diversity is increased by associations with a variety of gamma subunits. Proc Natl Acad Sci U S A. 1990 Oct;87(20):7973–7977. doi: 10.1073/pnas.87.20.7973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gierschik P., Codina J., Simons C., Birnbaumer L., Spiegel A. Antisera against a guanine nucleotide binding protein from retina cross-react with the beta subunit of the adenylyl cyclase-associated guanine nucleotide binding proteins, Ns and Ni. Proc Natl Acad Sci U S A. 1985 Feb;82(3):727–731. doi: 10.1073/pnas.82.3.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilman A. G. G proteins: transducers of receptor-generated signals. Annu Rev Biochem. 1987;56:615–649. doi: 10.1146/annurev.bi.56.070187.003151. [DOI] [PubMed] [Google Scholar]
- Graziano M. P., Freissmuth M., Gilman A. G. Expression of Gs alpha in Escherichia coli. Purification and properties of two forms of the protein. J Biol Chem. 1989 Jan 5;264(1):409–418. [PubMed] [Google Scholar]
- Hekman M., Holzhöfer A., Gierschik P., Im M. J., Jakobs K. H., Pfeuffer T., Helmreich E. J. Regulation of signal transfer from beta 1-adrenoceptor to adenylate cyclase by beta gamma subunits in a reconstituted system. Eur J Biochem. 1987 Dec 1;169(2):431–439. doi: 10.1111/j.1432-1033.1987.tb13630.x. [DOI] [PubMed] [Google Scholar]
- Hildebrandt J. D., Codina J., Rosenthal W., Birnbaumer L., Neer E. J., Yamazaki A., Bitensky M. W. Characterization by two-dimensional peptide mapping of the gamma subunits of Ns and Ni, the regulatory proteins of adenylyl cyclase, and of transducin, the guanine nucleotide-binding protein of rod outer segments of the eye. J Biol Chem. 1985 Nov 25;260(27):14867–14872. [PubMed] [Google Scholar]
- Huff R. M., Neer E. J. Subunit interactions of native and ADP-ribosylated alpha 39 and alpha 41, two guanine nucleotide-binding proteins from bovine cerebral cortex. J Biol Chem. 1986 Jan 25;261(3):1105–1110. [PubMed] [Google Scholar]
- Jones T. L., Simonds W. F., Merendino J. J., Jr, Brann M. R., Spiegel A. M. Myristoylation of an inhibitory GTP-binding protein alpha subunit is essential for its membrane attachment. Proc Natl Acad Sci U S A. 1990 Jan;87(2):568–572. doi: 10.1073/pnas.87.2.568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kühn H. Light- and GTP-regulated interaction of GTPase and other proteins with bovine photoreceptor membranes. Nature. 1980 Feb 7;283(5747):587–589. doi: 10.1038/283587a0. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Mumby S. M., Heukeroth R. O., Gordon J. I., Gilman A. G. G-protein alpha-subunit expression, myristoylation, and membrane association in COS cells. Proc Natl Acad Sci U S A. 1990 Jan;87(2):728–732. doi: 10.1073/pnas.87.2.728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neer E. J., Lok J. M., Wolf L. G. Purification and properties of the inhibitory guanine nucleotide regulatory unit of brain adenylate cyclase. J Biol Chem. 1984 Nov 25;259(22):14222–14229. [PubMed] [Google Scholar]
- Parker E. M., Kameyama K., Higashijima T., Ross E. M. Reconstitutively active G protein-coupled receptors purified from baculovirus-infected insect cells. J Biol Chem. 1991 Jan 5;266(1):519–527. [PubMed] [Google Scholar]
- Patten J. L., Levine M. A. Immunochemical analysis of the alpha-subunit of the stimulatory G-protein of adenylyl cyclase in patients with Albright's hereditary osteodystrophy. J Clin Endocrinol Metab. 1990 Nov;71(5):1208–1214. doi: 10.1210/jcem-71-5-1208. [DOI] [PubMed] [Google Scholar]
- Robishaw J. D., Kalman V. K., Moomaw C. R., Slaughter C. A. Existence of two gamma subunits of the G proteins in brain. J Biol Chem. 1989 Sep 25;264(27):15758–15761. [PubMed] [Google Scholar]
- Schaffner W., Weissmann C. A rapid, sensitive, and specific method for the determination of protein in dilute solution. Anal Biochem. 1973 Dec;56(2):502–514. doi: 10.1016/0003-2697(73)90217-0. [DOI] [PubMed] [Google Scholar]
- Schimmer B. P. Adenylate cyclase activity in Y1 mouse adrenocortical tumor cells: some properties of the enzyme associated with purified plasma membrane fractions. Can J Biochem Cell Biol. 1983 Jul;61(7):547–552. doi: 10.1139/o83-070. [DOI] [PubMed] [Google Scholar]
- Schimmer B. P. Adrenocortical Y1 cells. Methods Enzymol. 1979;58:570–574. doi: 10.1016/s0076-6879(79)58173-7. [DOI] [PubMed] [Google Scholar]
- Schimmer B. P., Tsao J., Borenstein R., Endrenyi L. Forskolin-resistant Y1 mutants harbor defects associated with the guanyl nucleotide-binding regulatory protein, Gs. J Biol Chem. 1987 Nov 15;262(32):15521–15526. [PubMed] [Google Scholar]
- Schimmer B. P., Tsao J., Collie G., Wong M., Schulz P. Analysis of the mutation to forskolin-resistance in Y1 adrenocortical tumor cells. Endocr Res. 1984;10(3-4):365–386. doi: 10.1080/07435808409036507. [DOI] [PubMed] [Google Scholar]
- Schimmer B. P., Tsao J. Decreased levels of guanyl nucleotide-binding regulatory protein alpha-subunits in Y1 adrenocortical tumor cell mutants resistant to forskolin. Mol Endocrinol. 1990 Nov;4(11):1698–1703. doi: 10.1210/mend-4-11-1698. [DOI] [PubMed] [Google Scholar]
- Schimmer B. P., Tsao J. Isolation of forskolin-resistant adrenal cells defective in the adenylate cyclase system. J Biol Chem. 1984 May 10;259(9):5376–5379. [PubMed] [Google Scholar]
- Schultz A. M., Tsai S. C., Kung H. F., Oroszlan S., Moss J., Vaughan M. Hydroxylamine-stable covalent linkage of myristic acid in G0 alpha, a guanine nucleotide-binding protein of bovine brain. Biochem Biophys Res Commun. 1987 Aug 14;146(3):1234–1239. doi: 10.1016/0006-291x(87)90780-7. [DOI] [PubMed] [Google Scholar]
- Shaltiel S. Hydrophobic chromatography. Methods Enzymol. 1974;34:126–140. doi: 10.1016/s0076-6879(74)34012-8. [DOI] [PubMed] [Google Scholar]
- Smigel M. D., Northup J. K., Gilman A. G. Characteristics of the guanine nucleotide-binding regulatory component of adenylate cyclase. Recent Prog Horm Res. 1982;38:601–624. doi: 10.1016/b978-0-12-571138-8.50019-6. [DOI] [PubMed] [Google Scholar]
- Sternweis P. C., Northup J. K., Smigel M. D., Gilman A. G. The regulatory component of adenylate cyclase. Purification and properties. J Biol Chem. 1981 Nov 25;256(22):11517–11526. [PubMed] [Google Scholar]
- Sternweis P. C., Robishaw J. D. Isolation of two proteins with high affinity for guanine nucleotides from membranes of bovine brain. J Biol Chem. 1984 Nov 25;259(22):13806–13813. [PubMed] [Google Scholar]
- Sternweis P. C. The purified alpha subunits of Go and Gi from bovine brain require beta gamma for association with phospholipid vesicles. J Biol Chem. 1986 Jan 15;261(2):631–637. [PubMed] [Google Scholar]
- Tamir H., Fawzi A. B., Tamir A., Evans T., Northup J. K. G-protein beta gamma forms: identity of beta and diversity of gamma subunits. Biochemistry. 1991 Apr 23;30(16):3929–3936. doi: 10.1021/bi00230a018. [DOI] [PubMed] [Google Scholar]
- Watkins D. C., Northup J. K., Malbon C. C. Regulation of G-proteins in differentiation. Altered ratio of alpha- to beta-subunits in 3T3-L1 cells. J Biol Chem. 1987 Aug 5;262(22):10651–10657. [PubMed] [Google Scholar]
- Yasumura Y., Buonassisi V., Sato G. Clonal analysis of differentiated function in animal cell cultures. I. Possible correlated maintenance of differentiated function and the diploid karyotype. Cancer Res. 1966 Mar;26(3):529–535. [PubMed] [Google Scholar]



