Abstract
While being in the center of attention and exposed to other’s evaluations humans are prone to experience embarrassment. To characterize the neural underpinnings of such aversive moments, we induced genuine experiences of embarrassment during person-group interactions in a functional neuroimaging study. Using a mock-up scenario with three confederates, we examined how the presence of an audience affected physiological and neural responses and the reported emotional experiences of failures and achievements. The results indicated that publicity induced activations in mentalizing areas and failures led to activations in arousal processing systems. Mentalizing activity as well as attention towards the audience were increased in socially anxious participants. The converging integration of information from mentalizing areas and arousal processing systems within the ventral anterior insula and amygdala form the neural pathways of embarrassment. Targeting these neural markers of embarrassment in the (para-)limbic system provides new perspectives for developing treatment strategies for social anxiety disorders.
Keywords: embarrassment, limbic system, mentalizing, publicity, social anxiety, social immersion
Introduction
Since the time of the ancient philosophers (Aristotle; 384–322 B.C.), the distinction between a “public” and a “private” realm has been a central tenet of political theory (Arendt, 1958; Sennett, 1974), jurisprudence (Warren and Brandeis, 1890), and the social sciences (Weintraub, 1997). The presence of others in the public space deeply affects human psychology and the emotional consequences of one’s actions (Gilovich et al., 2000). One of humankind’s most common fears centers around failing to uphold one’s public image within social encounters (Leary and Kowalski, 1995). The expected negative evaluation ‘in the eyes of others’ (Tangney et al., 2007) during ‘public deficiencies’ is the main cause of embarrassment (Miller, 1996). Mental-state attribution is therefore the lynchpin of the emotion of embarrassment, which regulates so many aspects of interpersonal behavior (Tangney et al., 2007) whenever others might potentially act as an audience, be it at school, work or during leisure time (Miller, 1996).
Excessive and persistent concerns about the evaluations of others are a hallmark of social anxiety. While everybody experiences mild forms of social anxiety occasionally (Leary and Kowalski, 1995), social anxiety disorders are a major burden for society (Kessler et al., 2005), and in affected individuals, the fear of embarrassment can even lead to social withdrawal and depression (Schneier, 1992). The neurobiology of how humans process situations that trigger their embarrassment, and how this contributes to social anxiety disorders, remains largely unknown, but would provide akey to understanding the neurobiological mechanisms of social anxieties.
The traditional ‘spectator approach’ in social neuroscience involves measuring the brain activity of participants in isolation while they are viewing photographs or movies of actors, or vignettes of fictional social situations. In these paradigms, the participant cannot interact with the targets of his/her social cognition, and his/her social reputation is not at stake. In typical social interactions, however, we not only perceive what others do, but we also need to (a) react in ways which are appropriate to others’ actions, and (b) maintain our social reputation while we are the focus of other people’s evaluation. The traditional ‘spectator approach’ fails to capture the motor involvement and emotional significance associated with these two aspects of our social world (Hasson et al., 2012; Schilbach et al., 2013). For instance, single cell recordings in monkeys have revealed that a spectator paradigm, in which a monkey watches movies of actions, greatly underestimates premotor mirror responses compared to when the same monkey witnesses a human act ‘live’, in a shared peripersonal space where direct interactions are possible (Caggiano et al., 2011, 2009).
The limits of the traditional ‘spectator approach’ are a considerable hindrance in the endeavor to gain a mechanistic understanding of embarrassment (Krach et al., 2013), as embarrassment is defined by the social context: failing in front of a judging audience. To this day, neuroimaging studies have induced social stress or rejection and were able to overcome this hindrance by implementing interaction paradigms using social or performance feedback (Cooper et al., 2014; Muscatell et al., 2014; Somerville et al., 2006; Wager et al., 2009). However, all we know about the neural substrates of the emotion of embarrassment originates from ‘spectator approaches’, in which the participant did not fail him- or herself, and was not monitored by an audience, but merely read brief fictional stories (e.g. “I was not dressed properly for the occasion”; Finger et al., 2006; Takahashi et al., 2004). We have all, on occasion, confidently said something blatantly incorrect in front of an audience we wished to impress, and the feelings accompanying this can be overwhelmingly intense: blushing, pounding heart, feeling terrible, and a vivid image of how others are mocking us for our failings in their heads. While imagining fictional situations such as “I was not dressed properly for the occasion” might capture some of the rational cognitions that are triggered by real embarrassing situations, the hot emotional rush that is the hallmark of embarrassment (Buss, 1980), and the paralyzing claws of social anxieties, have not been addressed by previous studies.
With this caveat in mind, it is perhaps unsurprising that past experiments using fictional scenarios (Finger et al., 2006; Takahashi et al., 2004) have emphasized the role of the medial prefrontal cortex (mPFC) and the precuneus, so-called “mentalizing areas” which are involved in putting oneself in the mental world of others (Frith and Frith, 1999; Tangney et al., 2007). However, the mPFC and precuneus not only are engaged when we are thinking about another person’s mind set, but also when we reflect about ourselves (in the context of others; Müller-Pinzler et al., in revision), we think about future events or just spend time mind-wandering or daydreaming (Critcher and Gilovich, 2010; Schooler et al., 2011). To frame these regions as mentalizing areas is thus simplified and suggests a specificity of processing in these structures that might not generally uphold given more recent evidence. Within the context of the present studies and also the current undertaking in decomposing embarrassment, we will nonetheless keep this term and aim to provide empirical evidence that might be helpful in understanding the ongoing psychological processes.
By contrast to the previous evidence for activity in these mentalizing areas, the limbic system, which is involved in all facets of emotional experiences (Adolphs et al., 1995; Morris et al., 1998; Phan et al., 2002), and the dorsal anterior insula processing the corresponding affective arousal (Critchley, 2005), were not activated in these studies. The lack of evidence for involvement of these regions is surprising but might be due to the previous experimental paradigms that trigger rational cognitions to a greater degree than the hot emotional rush of embarrassing situations. To provide a mechanistic understanding of embarrassment, and to examine its relevance for social anxieties, we therefore need to devise a new paradigm which enables us to capture the emotional dimension of failing in public within a neuroimaging set-up.
According to current models of embarrassment, two factors need to converge to evoke embarrassment. The first factor is a deviation from personal standards, the failure to show appropriate behavior, such as physical pratfalls, loss of control over the body, or cognitive shortcomings (Miller, 1996). The second factor is the publicity of one’s behavior, which motivates individuals to think about others’ evaluations when they are the center of attention. The interaction of both factors, namely the ‘public failure’, is at the core of the “aversive state of abashment, flusterment and chagrin” of embarrassment (Miller, 1996). For this reason, and in line with a ‘psychological constructivist’ approach to emotions (Lindquist et al., 2012), embarrassment should not be localized in one single brain region, but should manifest itself in the interaction of distinct brain systems which integrate the components of failure and publicity.
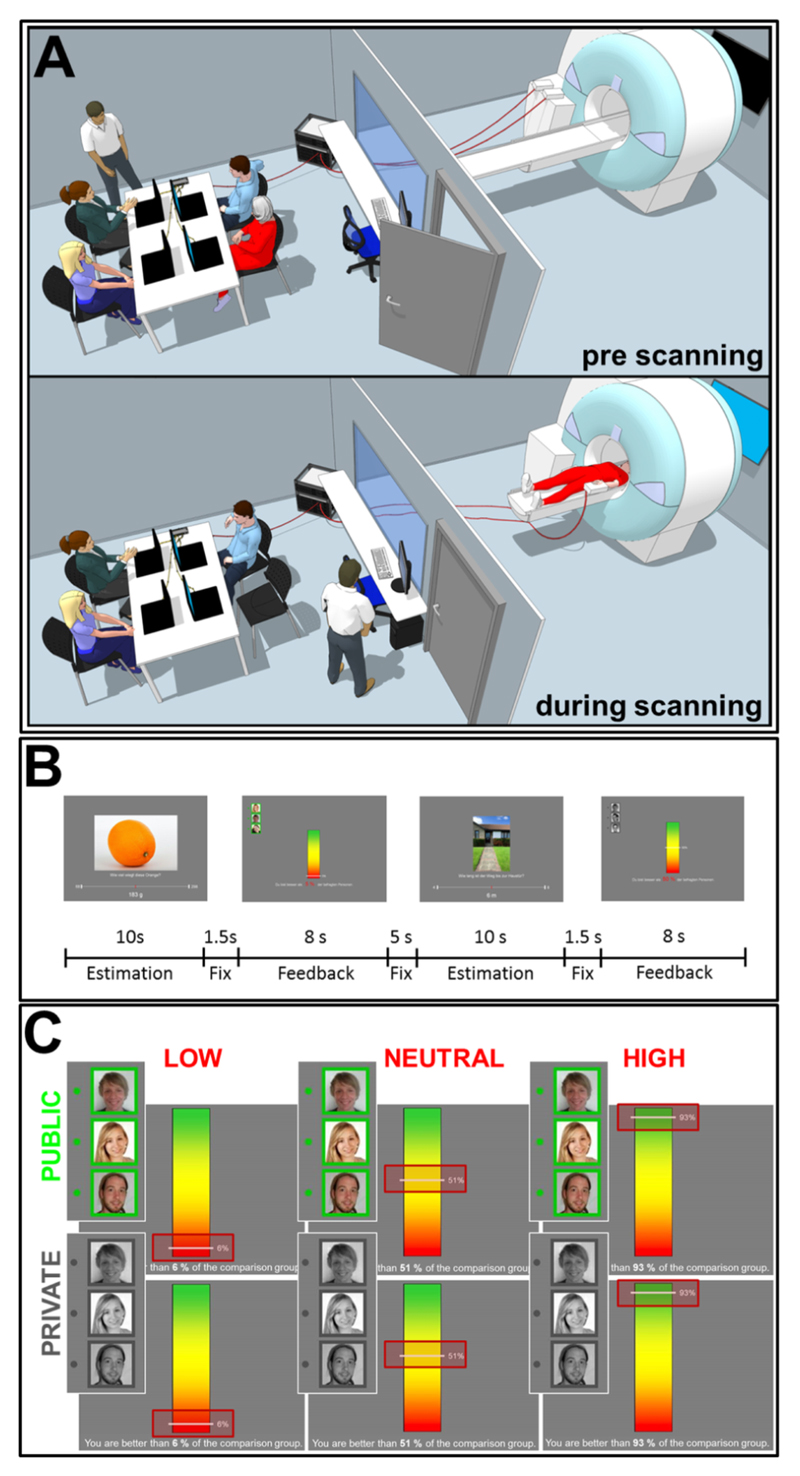
To explore the interaction between failure and publicity, we set up a staged person-group interaction in which a participant was made to fail in front of three confederates pretending to be fellow research participants (see Fig. 1a, Material and Methods, and Movie A.1). We combined brain imaging with measures of pupillometry and eye-gaze behavior to capture both the neural and physiological correlates of embarrassment and to explore how these variables are related to social anxiety. Participants were required to estimate the properties of objects, i.e. sizes, amounts, or weights, during a restricted period of time. We selected the domain of cognitive abilities to induce embarrassment because they are highly relevant for the human self-concept (Marsh, 1990) and social image, meaning that public cognitive shortcomings are very effective triggers of embarrassment. Participants then received manipulated feedback on their estimation accuracy: a bar chart in the center of the screen displaying the exact percentile of the participant’s performance, and photographs of the three confederates’ faces on the upper left-hand side on the screen. The level of feedback (i.e. PERFORMANCE) induced failure or achievement through either low (LOW; 1-15%) or high (HIGH; 85-99%) alleged percentiles of accuracy. As a control condition, mediocre feedback was provided (NEUT; 40-60%). Independent of PERFORMANCE, we manipulated the publicity of the feedback (PUBLICITY) by informing the participant whether the feedback on his/her performance was also being presented to the three confederates outside the scanner (PUB; green frame around the photographs, 50% of trials) or was only visible to him/her (PRIV; gray frame around the photographs).
Figure 1.
Experimental set-up and design. A Set-up of the fMRI experiment. During the pre-scanning phase (upper picture), the participant (red clothes) and the three confederates practice the cognitive estimation task while sitting in front of their notebooks in the preparation room adjacent to the scanner room. During scanning (lower picture), the participant lies in the MRI believing that the three confederates are completing the same task in the preparation room and are able to follow his/her performance on their notebook screens via cable connections. B Timing of the fMRI paradigm. Estimation questions are presented for 10 s followed by a fixation cross presented for 1.5 s and the feedback presented for 8 s. After an intertrial interval of 5 s, the next trial starts. C Design of the fMRI paradigm. There are six different conditions resulting from the PERFORMANCE (3) x PUBLICITY (2) levels. PERFORMANCE is either LOW, mediocre (NEUTRAL) or HIGH and is indicated by a line marking the exact percent value of the relative estimation performance (red frames). Half of the feedback is made PUBLIC and visible to the audience (green frames) and the other half is PRIVATE and only visible to the participant him- or herself (gray frames). For further details see also Movie A. 1.
According to the considerations above, we hypothesized that mentalizing areas (mPFC and precuneus) would be involved whenever participants receive public feedback. In addition, we hypothesized that the dorsal aspect of the anterior insula (dAI), which is implicated in the processing of arousal, would be active whenever the participant’s performance deviated from his/her expectations; thus especially during perceived failures or achievements (Critchley, 2005; Seeley et al., 2007). There is accumulating evidence demonstrating amygdala involvement in various negative and positive emotions (Adolphs et al., 1995; Morris et al., 1998; Phan et al., 2002), and the amygdala is particularly active in a socially evaluative context (Guyer et al., 2008; Lorberbaum et al., 2004). Additionally, meta-analyses of neuroimaging data consistently show that ventral aspects of the anterior insula (vAI), which are densely connected to the amygdala (Mesulam and Mufson, 1982), are central in human affect (Chang et al., 2013; Deen et al., 2011; Kelly et al., 2012). Thus, we finally expected (para-)limbic regions (vAI and amygdala) to play a critical role in the specific integration of both aspects: the publicity of one’s failures.
Based on these assumptions our core hypothesis is that embarrassment should manifest in a unique functional integration in core affect regions of systems involved in mentalizing about the thoughts of the audience and systems involved in the arousal associated with unexpectedly low performance. This hypothesis can be broken down in three specific hypotheses. First, that mentalizing brain regions will show a main effect of publicity, being more active in public than private conditions. Second, that the dAI will show a main effect of performance, in which unusually high or low performance will show stronger activity than neutral performance. Finally, in line with the constructivist understanding of how the brain processes emotions (Lindquist et al., 2012), that the integration of signals from these two systems onto core affect regions (vAI and amygdala) would be particularly high during the failures that trigger the “chagrin of embarrassment” (Miller, 1996).
Materials and Methods
Participants
Twenty-seven healthy naive participants took part in the functional magnetic resonance imaging (fMRI) study (17 females and 10 males; aged 18-28 years; M = 23.11; SD = 2.58). All participants had normal or corrected-to-normal vision, no past neurological or psychiatric history, and were not taking any medication. On average, participants had spent 16.39 years in education (range 12-22; SD = 2.51). All participants received 25 € compensation for approximately 2.5 h involvement in the study. The study was approved by the local ethics committee (AZ 08/10) and written informed consent was obtained from all participants involved in the study.
Pre-scanning procedure and set-up of the cover story
We used a cover story to create a socially immersive environment in which genuine experiences of embarrassment were induced. Together with three confederates, the participant was invited to take part in a study on the neural basis of cognitive estimation. The participant and the confederates arrived at the same time in the preparation room. The room was adjacent to the scanner, which was equipped with four laptop computers that were wired via a local area network. Computers in the preparation room looked like they were also connected to the MRI system (see Fig. 1a for the experimental setting). After signing informed consent forms, the participant and the confederates solved a German translation of the Wonderlic Personnel Test (Wonderlic, 1996) in a shortened period of time (6 min). Based on a faked rank order in the IQ test, providing the participant with the highest score, the participant was selected to enter the MRI and to complete the estimation task in the scanner. The experimenter justified the selection procedure based on the alleged positive correlation of cognitive estimation performance with IQ. The confederates were instructed to complete the same task outside the scanner in the preparation room.
Contenders had to estimate sizes, weights or quantities of pictured objects or living beings in a restricted time period of 10 s (e.g. “How long is this screw?”; see also Fig. 1; see FMRI paradigm and experimental design section for a detailed description of the experimental design). After each trial, the participant then received manipulated feedback on his/her accuracy in the form of a short sentence and a colored bar with a line marking the exact percentile ranging from 0% to 100%. This information indicated how well the participant had performed compared to an alleged reference group of 350 university students who, according to the cover story, had been tested beforehand. In addition, the participant was informed that the three contenders in the adjacent room would be informed about his/her performance during 50% of the trials as a frame of reference to the person with the highest IQ. In these trials, the performance of the participant was projected onto the three screens of the confederates, who thus formed the audience. After careful instructions, the participant and the confederates practiced the estimation task outside the scanner with five example situations. The participant was able to see that his/her performance was displayed on the confederates’ screens in the public feedback situations, while in the private feedback situations all contenders received only their own performance feedback. After the instruction period, which lasted approximately 45 min, the participant was then led into the MRI.
FMRI paradigm and experimental design
Each trial consisted of an estimation period, which lasted 10 s and a consecutive feedback period which was presented for 8 s. Estimation and feedback were separated by a fixation cross for 1.5 s, and a low-level baseline period showing a fixation cross for 5 s was interleaved between feedback and the following trial (see Fig. 1B for the timing of the paradigm). During the estimation period, continuous response scales below the pictures determined a range of possible answers, and participants indicated their responses by navigating a pointer on the response scale with button presses of the right and left hands. The upper and lower ends of the response scales were designed such that all answers in between were plausible for the specific object. In this way, it was not possible for participants to know how well they had performed and every kind of feedback they received for their estimation was plausible. Stimuli were presented on an LCD screen with the Presentation 11.0 software package (Neurobehavioral Systems, Albany, CA, USA, http://www.neurobs.com/).
Failures and achievements (i.e. PERFORMANCE) as well as the influence of the audience (i.e. PUBLICITY) were manipulated in a 2 × 3 factorial within-subject design. Participants received either faked low performance feedback (LOW; e.g. "You are better than 5% of the reference participants"; percentiles ranging from 1 to 15%), high performance feedback (HIGH; percentiles ranging from 85 to 99%), or mediocre performance feedback (NEUT; percentiles ranging from 40 to 60%) on their estimation performance. Feedback was given either publicly (PUB; performance is exposed to the confederates outside the MRI) or privately (PRIV; performance is visible only to the participant inside the MRI). The feedback screens contained photographs of the three confederates’ faces in the upper left corner as a cue for the publicity of an event. Photographs were displayed in black and white, with a gray frame, during PRIV trials, and were colored, with a green frame, during PUB trials (see Fig. 1b for the stimuli). The use of the photographs constituted part of the cover story, as participants had also been asked to send in a photograph of themselves to be used during the experiment. Each performance feedback (LOW, HIGH, NEUT) was either public or private in equal measure.
Trials were presented in a fixed pseudo-randomized order. The two HIGH and LOW performance conditions included 17 trials for each PUB and PRIV, respectively. The NEUT condition included nine trials each in the PUB and PRIV conditions, resulting in a total of 86 trials which were presented in two consecutive fMRI runs. The duration of the total fMRI experiment was 35.12 min.
Post-fMRI examination
After the fMRI data acquisition, the socially immersive environment was re-established, with the confederates playing their roles until they were guided to adjacent rooms for a post-experimental examination. During the post-experimental examination, the participant provided self-reports of experienced emotions in the MRI for three estimation trials from each condition. Embarrassment and pride (as well as anxiety, anger, sadness, and happiness) were rated on a 9-point unipolar scale (1 = not at all, 9 = very strong) using descriptive adjectives within a set of emotions. Participants subsequently completed the German version of the social interaction anxiety scale (SIAS; Mattick and Clarke, 1998) and, after verifying that they had not detected the true intention of the study with two suggestive questions (“Did you recognize a systematic in the performance feedback? If you did, what was it?” and “Did you think the performance feedback was related to your performance?”), which none of them had, they were debriefed. Notably, none of the participants indicated having looked through the cover story and revealed that the other “participants” in fact were confederates throughout the post-fMRI examination and after debriefing.
Analyses of behavioral data
All non-imaging data were analyzed with PASW Statistics 18 (Chicago: SPSS Inc.). Post-fMRI self-reports of each emotion were averaged within conditions and analyzed for each emotion separately using analyses of variance (ANOVAs) with PERFORMANCE (LOW, HIGH, NEUT) and PUBLICITY (PUB, PRIV) as within-subject factors. To test the effects on the experience of embarrassment and pride in the MRI, a priori contrasts comparing embarrassment ratings in LOW vs. NEUT and pride ratings in HIGH vs. NEUT were calculated. The specific influence of PUBLICITY on emotions was tested with the PUB vs. PRIV × HIGH vs. LOW interaction with paired t-tests for all self-reports of emotion as obtained in the post-fMRI examination. To compare self-reports of embarrassment, anxiety, anger, sadness, pride and happiness with each other an ANOVA with PERFORMANCE, PUBLICITY and emotion self-reports as additional factor EMOTION was implemented. A priori contrasts were chosen contrasting LOW vs. NEUT and comparing embarrassment ratings versus all other emotion ratings. Additionally self-reports for all emotions in the LOW condition were compared using paired t-tests.
Analyses of pupil dilation
Eye-tracking data were assessed during the fMRI paradigm. Pupil diameter and gaze behavior were recorded non-invasively in one eye at 500 Hz using an MRI-compatible Eyelink-1000 device (SR Research, Kanata, ON, Canada) with manufacturer-recommended settings for calibration and blink detection. Periods of blinks were cut out and values in this gap were interpolated by piecewise cubic interpolation. The pupil trace was subsequently z-normalized over the whole session. To characterize the pupil dilation for each trial by a single value, we subtracted the baseline pupil size during the first 200 ms of each trial from the average value during the last second of each trial.The condition averaged value for the pupil dilation was then entered into a repeated measures ANOVA. A priori contrasts were implemented to compare LOW and HIGH to NEUT, and paired t-tests were implemented in order to test the PUB vs. PRIV × HIGH vs. LOW interaction.
Neuroimaging data
Image acquisition
Participants were scanned at 3T (Siemens Trio, Erlangen) with 36 near-axial slices and a distance factor of 10% providing whole-brain coverage. An echo planar imaging (EPI) sequence was used for acquisition of 503 functional volumes during each of the two sessions of the experiment, resulting in a total of 1,006 functional volumes (TR= 2.2 s, TE = 30 ms, flip angle= 90°, slice thickness = 3 mm, FoV= 192).
Analysis of functional imaging data
FMRI data were analyzed using SPM8 (www.fil.ion.ucl.ac.uk/spm). The first three functional volumes of each of the two sessions were discarded from further analyses, leaving 500 EPI volumes per session. These were corrected for timing differences of the slice acquisition, motion-corrected and spatially normalized to the standard template of the Montreal Neurological Institute (MNI). The normalized volumes were resliced with a voxel size of 2 × 2 × 2 mm and smoothed with an 8 mm full-width-at-half-maximum isotropic Gaussian kernel. To remove low-frequency drifts, functional images were high-pass filtered at 1/256.
Statistical analyses were performed in a two-level, mixed-effects procedure. The fixed-effects GLM on the first level included eight epoch regressors modeling the hemodynamic responses to the different performance feedback conditions (6; PERFORMANCE: LOW, HIGH, NEUT × PUBLICITY: PUB, PRIV), the estimation periods as one regressor (1), and the instruction phase (1) for each of the two sessions. Additionally, performance feedback percent values were entered as parametric modulators for the six conditions to explain additional within-subject variance in neural activation within each condition. To account for noise due to head movement, six additional regressors modeling head movement parameters were introduced.
To analyze the effects on the second level, we always kept one of the factors constant and implemented an ANOVA design with the remaining factor. This was done to correctly account for the dependencies of the two repeated factors. To test the effects of PERFORMANCE the ANOVA model on the second level included the three levels LOW, HIGH and NEUT while the contrasts on the first level were computed as the average across both levels of PUBLICITY. To test the effects for PUBLICITY, the ANOVA model on the second level included the two levels PUB and PRIV, while the contrasts on the first level were computed as the average across the three levels of PERFORMANCE. The interaction of PERFORMANCE and PUBLICITY was tested by contrasting the LOW_PUB-LOW_PRIV and HIGH_PUB-HIGH_PRIV on the first level and implementing an ANOVA design on the second level with one factor and two-levels.
For all analyses we conducted planned comparisons in the respective ANOVA design to test the hypothesized effects. First, arousal-related brain regions were identified through a conjunction analysis of both positive and negative deviations from neutral in the PERFORMANCE (LOW-NEUT∩HIGH-NEUT). Effects of the audience were examined by contrasting public with private feedback in the PUBLICITY model (PUB-PRIV). The differential influence of publicity on low compared to high performance feedback was identified by computing a contrast for the respective model for the interaction ((LOW_PUB-LOW_PRIV)-(HIGH_PUB-HIGH_PRIV)).
Correspondence of pupil dilation, gaze behavior, self-reported embarrassment and trait social anxiety with neuroimaging parameters
To investigate the trial-by-trial association of pupil dilation and neural activation, pupil dilation values were introduced as a parametric modulator in a first-level GLM (see Paulus et al., 2015). The first-level GLM included the hemodynamic responses during feedback (1), the cognitive estimation periods (1), and the instruction phase (1). The parametric modulators of hemodynamic responses during feedback contained first, the exact percent values of PERFORMANCE, second, the deviation from 50% to control for any within-subject variance in neural activation due to PERFORMANCE, and third, the trial-by-trial pupil dilation value. To account for noise due to head movement, six additional regressors modeling head movement parameters were introduced. On the second level, β-maps of the pupil dilation as parametric modulator were analyzed using a one-sample t-test.
Gaze dwell time was calculated as the sum of the time for which the gaze was oriented towards the photographs of the confederates’ faces during the feedback in one trial. The association between dwell time on the confederates’ faces and neural activation was investigated within a GLM with trial-by-trial dwell time on the confederates’ faces as a parametric modulator. The parametric modulators of the hemodynamic responses during feedback were first, the publicity of the performance and second, the trial-by-trial dwell time on the confederates’ faces. On the second level, β-maps of the dwell time as parametric modulator were analyzed using a one-sample t-test.
We further explored the association of trait-level social anxiety and self-reported embarrassment during LOW_PUB with neural activation during PUB-PRIV. To account for the influence of potential outliers, robust regressions using M-estimators with Huber weights were calculated. Difference scores for the dwell time on the confederates’ faces were then calculated for PUB-PRIV. To assess whether the relationship between social anxiety and differences in neural activation was mediated by differential gaze behavior, Sobel tests were computed, with the dwell time difference scores as mediator. Another mediation analysis was performed for the relationship of dwell time difference scores and self-reports of embarrassment implementing FFA activation as a mediator.
Functional connectivity analyses
Emotion-specific functional connectivity patterns were examined with "psychophysiological interaction" (PPI) analyses as implemented in SPM8. Two seed regions were selected due to their involvement in processing affective arousal, and two seed regions were chosen due to their involvement in mentalizing in the presence of an audience. The left and right dAI peaks were defined based on the conjunction of LOW-NEUT∩HIGH-NEUT. The precuneus and an mPFC peak voxel were defined by the contrast PUB-PRIV. For all regions, time series were extracted as the first eigenvariate from all voxels that showed a positive effect within a sphere of 6 mm radius around the peak voxel (see Table 1). Variance explained by the movement regressors and the hemodynamic responses induced by the estimation period and the instruction were removed with an effects-of-interest correction. To examine embarrassment-specific connectivity patterns, PPI regressors contrasted LOW vs. HIGH performance separately for PUB and PRIV for each time series. For each of the examined regions, a separate fixed-effects GLM was implemented on the first level, including (i) the original time series, (ii) the psychological regressor contrasting the hemodynamic response of LOW and HIGH performances, (iii) the PPI regressor coding the dynamics in the connectivity with the seed region, and (iv) the regressors of the original design matrix. Resulting β-maps of the PPI regressors were analyzed on the second level within an ANOVA design, each including the PUB and PRIV conditions for one of the seed regions.
Table 1.
Increased Functional Connectivity During Low Compared to High Performance.
| Seed Region | Side | MNI Coordinates |
T | p | |||
|---|---|---|---|---|---|---|---|
| Target Region | x | y | z | ||||
| right dAI (40, 24, -10 mm) | |||||||
| amygdala | R | 34 | 0 | -24 | 4.49 | .002 | |
| amygdala | L | -32 | 0 | -26 | 4.40 | .002 | |
| vAI | R | 42 | 6 | -10 | 3.38 | .036 | |
| vAI | L | -40 | 0 | -14 | 4.19 | .004 | |
| left dAI (-32, 20, -10 mm) | |||||||
| amygdala | R | 36 | 0 | -20 | 4.10 | .005 | |
| amygdala | L | -32 | 0 | -26 | 3.67 | .015 | |
| vAI | R | 44 | 2 | -8 | 4.35 | .003 | |
| vAI | L | -40 | -2 | -14 | 4.27 | .003 | |
| precuneus (-4, -52, 38 mm) | |||||||
| amygdala | R | 38 | -2 | -24 | 3.33 | .041 | |
| vAI | R | 40 | -4 | -10 | 3.62 | .022 | |
| vAI | L | -38 | -6 | -14 | 3.88 | .009 | |
| mPFC (0, 60, 18 mm) | |||||||
| vAI | L | -40 | -4 | -16 | 3.30 | .036 | |
Note. Increased functional connectivity between our seed regions (the precuneus and mPFC as well as the left and right dAIs) within our a priori search volumes (amygdala and vAI) during low vs high performance (LOW-HIGH). P-values are FWE-corrected within the search volumes. Abbreviations: vAI = ventral anterior insula; mPFC = medial prefrontal cortex; dAI = dorsal anterior insula.
Thresholding procedures
Both, the effects of neural activation, as well as the dynamics in connectivity were first examined in the whole brain. To do so, we applied a family-wise error (FWE)-correction at p<.05, at the voxel level as implemented in SPM8 for all contrasts. In a second step, we restricted our search volume to the predefined brain systems involved in emotional processing, the amygdala and the vAI, to increase our statistical power in these a priori regions. These regions within the (para-)limbic system were defined anatomically based on the automated anatomical labeling atlas (AAL; Tzourio-Mazoyer et al., 2002) for the amygdala and based on the parcellation study by Kelly and colleagues (2012) for the vAI. Analyses within this restricted search volume were again thresholded at p<.05, FWE-corrected for this search volume.
Results
Behavioral data and pupil dilation
The feedback given to participants, independent of PUBLICITY, significantly induced the expected emotions. Self-reports after scanning indicated stronger embarrassment experiences in cases of low performance (LOW vs NEUT, F(1,26) = 88.15, p < .001) and stronger pride experiences in cases of high performance (HIGH vs NEUT, F(1,26) = 335.32, p < .001). Comparisons between self-reports of all emotion assessed after scanning (embarrassment, anxiety, anger, sadness, pride, and happiness) showed that embarrassment was the most prominent emotion during low performance (for means and standard deviations of emotion self-reports for all conditions see Table 2). There was a significant interaction of PERFORMANCE and EMOTION (F(12,312) = 124.07, p < .001). In the LOW compared to the NEUT condition self-reports of embarrassment were higher than for anxiety, sadness, anger, happiness, and pride (all ps < .001). Further, in the LOW performance condition the embarrassment experience exceeded all other emotions (anxiety, sadness, anger, happiness, pride; all ps < .005). This suggests that embarrassment was the most prominent emotion that was induced by the low performance feedback in the current task.
Table 2.
Self-reports of emotions for the performance conditions.
| PRIV |
PUB |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Performance condition | LOW |
NEUT |
HIGH |
LOW |
NEUT |
HIGH |
||||||
| M | SD | M | SD | M | SD | M | SD | M | SD | M | SD | |
| Emotion | ||||||||||||
| Embarrassment | 3.26 | 1.84 | 1.56 | 1.22 | 0.40 | 0.43 | 4.04 | 2.00 | 1.57 | 1.16 | 0.45 | 0.48 |
| Anger | 1.79 | 1.77 | 1.31 | 1.40 | 0.27 | 0.46 | 2.04 | 1.95 | 1.02 | 1.10 | 0.32 | 0.43 |
| Sadness | 2.39 | 1.80 | 1.35 | 1.24 | 0.40 | 0.48 | 2.50 | 1.85 | 1.24 | 1.15 | 0.33 | 0.39 |
| Fear | 0.74 | 1.28 | 0.45 | 0.78 | 0.33 | 0.56 | 0.70 | 1.26 | 0.49 | 0.83 | 0.27 | 0.43 |
| Pride | 0.39 | 0.53 | 1.54 | 1.28 | 5.13 | 1.76 | 0.43 | 0.56 | 1.83 | 1.26 | 5.40 | 1.71 |
| Happiness | 1.21 | 1.20 | 1.99 | 1.37 | 4.87 | 1.62 | 0.98 | 1.17 | 2.33 | 1.44 | 5.34 | 1.43 |
Note. M = mean, SD = standard deviation. LOW = low performance feedback, HIGH = high performance feedback, NEUT = mediocre performance feedback, PUB = public condition, PRIV = private condition.
The publicity of one’s performance affected the experience of embarrassment (F(1,52) = 7.75, p = .010) but did not affect pride (F(1,52) = 0.98, p = .330). The effect of PUBLICITY depended on PERFORMANCE. Exposing one’s performances to the public increased the self-reported experience of embarrassment (t(26) = 2.40, p = .012) more during LOW than HIGH performance. There was also a significant triple interaction of PUBLICITY, PERFORMANCE and EMOTION (F(1,26) = 2.841, p = .001). This was mainly driven by differential effects of the PUB and PRIV conditions during HIGH and LOW performances on embarrassment and happiness as indicated by planned comparisons. Whereas embarrassment showed a significantly greater increase in the LOW condition compared to the HIGH condition when the performance was public, happiness showed the opposite effect with greater increase in happiness in response to PUB compared to PRIV performance in the HIGH condition (t(26) = -2.24, p = .017). For all other emotions, the interaction of PERFORMANCE and PUBLICITY was not significant (ts(26) < 1.65, p > .56). These findings are in line with the idea that the audience is specifically relevant for embarrassment during failures (Miller, 1996), but is less relevant for the experience of pride during achievements (Seidner et al., 1988).
Pupil dilation, indicating sympathetic arousal (see e.g. Paulus et al., 2015), was also augmented in low and high performances (LOW vs NEUT, F(1,26) = 14.84, p = .001; HIGH vs NEUT, F(1,26) = 5.89, p = .022). As for the behavioral data, the effect of PUBLICITY depended on PERFORMANCE. Exposing one’s performances to the public increased the pupil dilation (t(26) = 1.94, p = .032) more during LOW than HIGH performance.
FMRI data
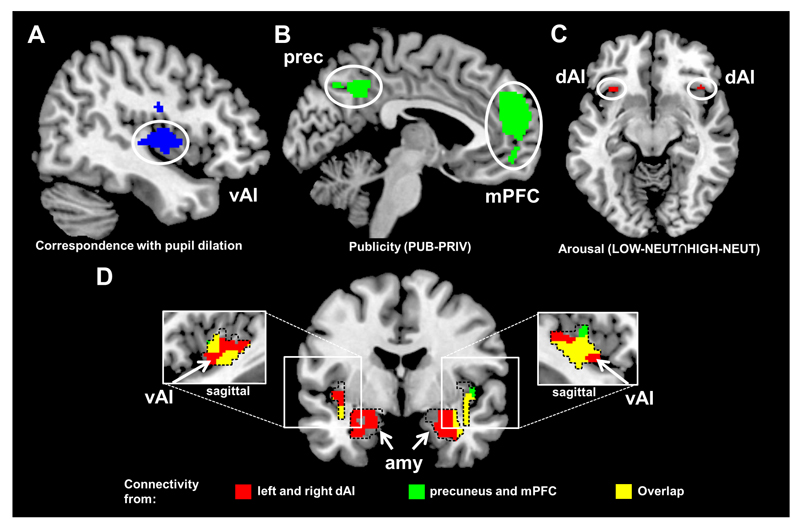
Both low and high performances (LOW-NEUT∩HIGH-NEUT) yielded significant increases in bilateral dorsal anterior insula (dAI) activation (right dAI: 40, 24, -10 mm, t(78) = 5.17, p = .020, corrected; left dAI: -32, 20, -10 mm, t(78) = 5.56, p = .005, corrected; see Fig. 2c). Trial-by-trial variability in pupil dilation was positively linked to neural activation of the right insula (t(26) = 8.57, p < .001; see Fig. 2a and Table A. 4) and somatosensory cortex areas (t(26) = 7.80, p = .001), areas associated with the processing of sympathetic arousal. Compared to low performance, high performance yielded strong and distributed activations including striatal areas (HIGH-LOW, t(52) = 13.22, p < .001; see Fig. A. 1 and Table A. 1), which is in line with the strong reward potential of positive feedback on cognitive performances (Bandura, 1982; Hattie and Timperley, 2007; Weiner, 1985). However, compared to high performance, low performance showed no specific activation.
Figure 2.
FMRI results. A Pupil dilation as indicator of arousal. Increased activation of the ventral anterior insula (vAI) associated with increases in pupil dilation during individual feedback trials across all conditions (p < .05, whole-brain corrected). For further details see also Table A.4. B Audience effect. Increased activation of the medial prefrontal cortex (mPFC) and precuneus during public (PUB) compared to private (PRIV) performance averaged across PERFORMANCE levels was associated with mentalizing processes when thinking about others’ evaluations (p < .05, whole-brain corrected, see also Table A. 2) C Performance effect. Increased activation of the dorsal anterior insula (dAI) during LOW and HIGH compared to mediocre (NEUT) performance indicating increased processing of affective arousal (conjunction analysis LOW-NEUT∩HIGH-NEUT, p < .05, whole-brain corrected). For further results see also Fig. A.1 and Table A.1. D Increased functional connectivity during embarrassment. Psychophysiological interaction analyses revealed increased functional connectivity during LOW compared to HIGH performance (p < .05 uncorrected for display purposes, results survive correction within our amygdala and vAI search volumes). Seed regions are based on the arousal (left and right dAIs; results presented in red, for seed regions see Fig. 2c) and mentalizing (precuneus and mPFC; results presented in green, for seed regions see Fig. 2b) networks. Overlap is presented in yellow. Dashed lines illustrate the anatomically defined search volumes including the amygdalae (AAL; Tzourio-Mazoyer et al., 2002) and the functional vAI (Kelly et al., 2012).
PUBLICITY (PUB-PRIV) was associated with a characteristic pattern of activation in the mentalizing network, comprising the mPFC (t(52) = 7.26, p < .001, corrected) and the precuneus (t(52) = 6.35, p = .001, corrected; see Fig. 2b and Table A. 2), which might be an indicator of increased thinking about others’ evaluations (Frith and Frith, 1999). In line with this, participants’ gaze dwelled longer on the faces of the confederates (F(1,52) = 18.02, p < .001; for means and standard deviations of dwell times see Table A. 3), and the fusiform face area (FFA) was activated more strongly during PUB than during PRIV (t(52) = 6.16, p = .002, corrected). Variability in the gaze dwell times on the faces on the within-subject level was also linked to activation of the fusiform gyrus (t(26) = 9.92, p < .001, corrected; see Fig. A. 1 and Table A. 4).
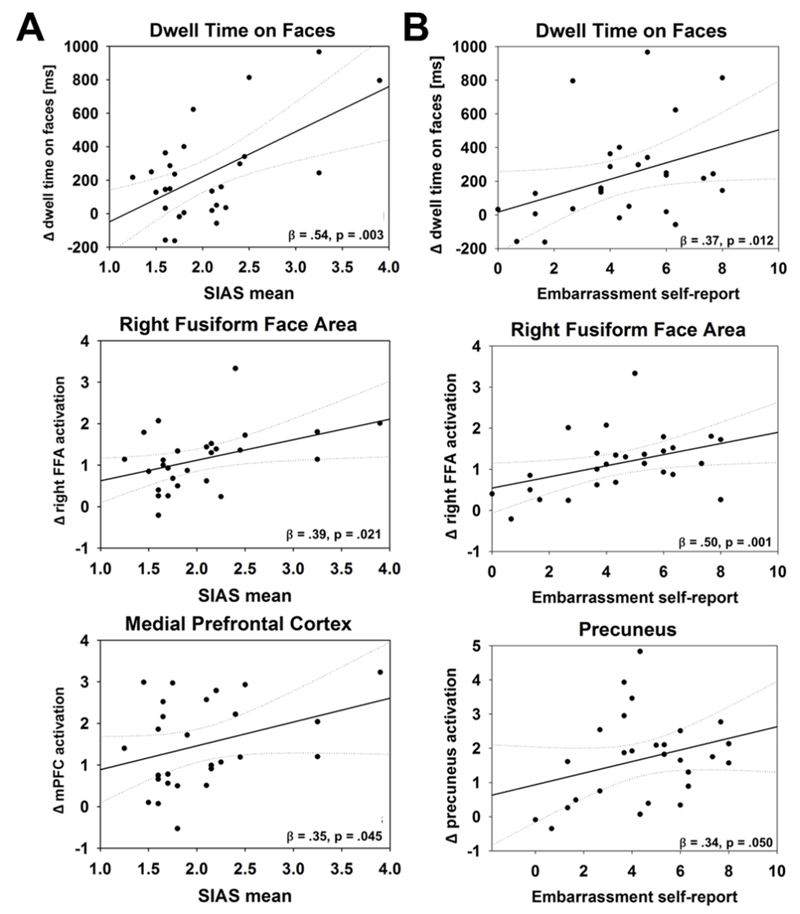
Neural activation of the mPFC (robust regression: β = .35, p = .045), the FFA (robust regression: β = .39, p = .021), and trend-wise of the precuneus (β = .26, p = .078) during PUB compared to PRIV was positively associated with social anxiety scores (see Fig. 3a). These associations were mediated by dwell time of gaze on the pictures of the confederates’ faces during PUB compared to PRIV (Sobel tests: mPFC: p = .04, FFA: p = .04). This finding corroborates the clinical characterization of social anxiety as being related to excessive and maladaptive concerns about being evaluated by others and attentional shifts towards potentially threatening social cues in the environment (Morrison and Heimberg, 2013). Activation of the FFA (robust regression: β = .50, p = .001) and precuneus (robust regression: β = .34, p = .050) in the contrast PUB-PRIV were positively associated with self-reports of embarrassment (as reported during LOW_PUB see Fig. 3b). The robust regression of mPFC activity and self-reports of embarrassment was not significant (β = .25, p = .114). Additionally, participants who reported stronger embarrassment during LOW_PUB feedback also showed increased dwell time of gaze on the confederates’ faces (robust regression: β = .37, p = .012). This association of dwell time and self-report of embarrassment was trend-wise mediated by FFA activation in the PUB-PRIV contrast (Sobel tests: FFA: p = .05). These findings suggest that paying attention to the audience and engaging in mentalizing about the other’s evaluations of one’s own situation might increase the own experience of embarrassment during public situations.
Figure 3.
Modulatory effects of social anxiety and self-reported embarrassment. β-values result from robust regression analyses using Huber’s M-estimators presented with one-tailed p-values. A Scatter plots of trait social anxiety and PUBLICITY effects for gaze dwell times and activation data. There was a positive correlation between social anxiety (mean SIAS scores) and differences in dwell time on the confederates’ faces between the public and private conditions (top). Participants with higher social anxiety scores also showed increased activation within the right fusiform face area (FFA, middle) and the mPFC (bottom) during PUB-PRIV (averaged parameter estimates within the FFA and mPFC clusters from the PUBLICITY effect, Fig. 2b). B Scatter plots of self-reports of embarrassment and PUBLICITY effects for gaze and activation data. The upper part shows a positive correlation between self-reports of embarrassment during LOW_PUB feedback and differences in dwell time of gaze on the confederates’ faces between the public and private conditions. Participants who reported experiencing stronger embarrassment during LOW_PUB feedback also showed increased activation within the right fusiform face area (FFA, middle) and the precuneus (bottom) during PUB-PRIV feedback.
The interaction of PERFORMANCE × PUBLICITY did not survive correction for multiple comparison (FWE) at the whole brain level.
We used PPI analyses to explore our hypothesis that embarrassment manifests in a unique pattern of functional connectivity between brain regions processing performance and brain regions processing publicity. To do so we explored whether the precuneus and the mPFC, brain regions involved in publicity, and the arousal related activations in the dAIs are also more strongly integrated with regions involved in processing core affect (vAI and amygdala) during low compared to high performances (see Fig. 2d). We indeed found that the mentalizing network revealed increased coupling with the amygdala and ventral aspects of the anterior insula during LOW compared to HIGH performances. The arousal-related left and right dAI showed greater connectivity with the same regions during LOW performance (see Table 1). Accordingly, the concept of embarrassment, as the interaction of failure and publicity, seems to map onto a similar construct in the brain: an increased functional connectivity between regions involved in mentalizing and (para-)limbic structures involved in affect.
Discussion
A core goal of our study was to characterize the neurobiological mechanisms of embarrassment while immersing individuals in an authentic social situation that really matters for their social integrity. Through manipulated feedback of failure, we induced a genuine affective experience of embarrassment (as verified by self-report), and triggered physiological and neural responses associated with increased arousal (pupil dilation and stronger activation of the dAI). When participants were informed that their failure was public, they experienced greater embarrassment and showed signs of increased sympathetic activation in terms of pupil dilation. This modulation was not evident for pride in response to feedback of achievements, which yielded distributed activations and strong responses of the brain’s reward circuits in the striatum.
According to psychological concepts, and in line with the constructivist understanding of how the brain processes emotions (Lindquist et al., 2012), embarrassment has been suggested to be a unique combination of (a) one’s failure and related arousal on the one hand, and (b) mentalizing about how this failure will damage the opinion that others have of oneself on the other hand (Krach et al., 2011; Miller, 1996; Paulus et al., 2013; 2014; Tangney et al., 2007). Our neuroimaging data suggest that this functional description of embarrassment as arousal in the context of unfavorable mentalizing maps well onto the brain.
Notably, activations in the mPFC are far from being specific to processes of mentalizing, and have been reported in a multitude of ‘tasks’ which do not involve embarrassment (Buckner et al., 2008; Critcher and Gilovich, 2010). The role of the mPFC has also been extensively debated in the context of the default mode of the brain where people engage in mind-wandering, self-reflection, or daydreaming (Schooler et al., 2011; Smallwood and Schooler, 2006). Nonetheless, the positive association of mPFC activation with the fixation on the audience’s faces supports the notion that the mPFC activity in the present experiment might indeed relate to thinking about the evaluations in-the-eyes-of-others and not mind-wandering or daydreaming.
The significance of this public-private distinction also manifests in the context of social anxiety and thereby contributes to the understanding of its etiology, where excessive and persistent concerns about the evaluations of others are central to the symptomatology (Morrison and Heimberg, 2013; Schneier, 1992). Our correlational data with sub-clinical social anxiety support the notion that in public contexts, persons with greater social anxiety pay increased attention to others which might be related to the clinically well-described negative bias in terms of what the audience might think of them (Morrison and Heimberg, 2013). We found a positive association between social anxiety and increases of gaze dwell time on the social cues in the public relative to the private condition. The co-occurring increase in mentalizing activation was mediated by this attentional bias to the audience. While there is initial evidence for increased mentalizing activity in patients with social anxiety in the context of face perception (Blair et al., 2011a) or negative self-evaluations (Blair et al., 2011b), our integrated perspective on how attentional shifts to the audience induce greater mentalizing activation goes beyond what previous studies on the neural foundations of social anxiety have described. The present paradigm could thus offer a novel perspective to characterize the altered processing of publicity in the pathology of these disorders in a dynamic and interactive setting.
Whenever performances deviated from neutral, we found robust activation of the dAI, a region strongly associated with salience and arousal (Critchley, 2005; Seeley et al., 2007). Given that embarrassment was experienced much more during low than high performances, this dAI activation is not a specific neural marker for the embarrassment experience. Both, the effects for the publicity of an event, as well as this deviation from an anticipated standard of an event, refer to the main effects in our two-factorial design. We further found an interaction of publicity and performance at the behavioral and physiological level: public failure, i.e. the combination of publicity and low performance, to trigger elevated levels of self-reported embarrassment and pupil dilation as an indicator for sympathetic arousal (Bradley et al., 2008; Paulus et al., 2015). Surprisingly, we did not find a matching interaction between both factors, the public failure, on the level of single voxel BOLD activity. More recent conceptualizations of how the brain processes emotions however depart from a traditional localist approach in which particular voxels are associated with particular emotions. Instead, discrete emotional categories such as embarrassment, shame, guilt, or pride, are thought to be constructed by the concerted dynamics of brain networks, with each network not being specific for one emotion (Lindquist et al., 2012), but the interaction between the brain regions creating a unique configuration. In line with these conceptualizations, we found the conditions of embarrassment to be associated with a unique pattern of functional connectivity. In particular, when we contrast low with high performances, the vAI and amygdala showed increased coupling with distinct brain networks, the bilateral dAI and the mPFC as well as precuneus. The integrated activity across arousal processing and mentalizing networks with these (para-)limbic structures might create a neural ensemble activity that supports the experience we call embarrassment. The dense anatomical connections between the ventral and dorsal aspects of the anterior insula and the amygdala (Cerliani et al., 2012) might constitute the structural underpinning of this integration, in line with accumulated evidence on the integrative role of the vAI in emotion processing (Chang et al., 2013; Kelly et al., 2012). Importantly, activations of the vAI and the amygdala, too, are themselves by no means specific for the experience of embarrassment (Adolphs, 2008; Chang et al., 2013; Kelly et al., 2012), making it highly improbable that the activity in these regions alone would trigger embarrassment.
The present findings provide neurophysiological support for psychological models of embarrassment which state that humans are usually motivated to maintain a positive social image whenever they act in public (Leary and Kowalski, 1990) and feel extreme discomfort if their social integrity is threatened (Miller, 1996). Cognitive shortcomings are among the most common and salient threats to one’s social integrity, and might be especially emphasized in a student setting and under circumstances in which the expectations on one’s cognitive performance are high (Miller, 1996). In the current experiment, this was induced by allegedly selecting participants based on their general mental ability. To understand and reflect on one’s own social image, one needs to model others’ evaluations and their view of oneself, which is relevant for identifying threats to social integrity (Eisenberger et al., 2003). This information processed in the mentalizing network seems to gain particular importance in the context of failure, as we observe a dynamic increase of connectivity of the mentalizing system with (para-)limbic regions for failure as compared to achievement. Together with the concurrent increase in the connectivity with neural systems associated with the processing of arousal, this specific integration of information across both brain systems might explain the publicity effect we found on the behavioral level, which was evident only for the embarrassment in response to public failures. During achievements, there is less integration of mentalizing and arousal-related information in (para-)limbic regions, and hence, publicity does not influence the experience of pride accordingly (Seidner et al., 1988). In conclusion, the present paradigm offers a broader perspective to investigate the processing of publicity and its impact on social emotions with the potential to characterize the altered neural pathways in social anxiety disorders in context of a dynamic and socially interactive setting.
Supplementary Material
Supplementary data to this article can be found online at http://dx.doi.org/10.1016/j.neuroimage.2015.06.036.
Movie A.1. Experimental procedure. The movie illustrates the experimental procedure with the pre-scanning phase, the scanning phase and the post-experimental examination in the original setting of the study.
Acknowledgments
Research leading to this manuscript has been funded by the German Research Foundation (DFG; KR3803/2-1, KR3803/7-1, EI 852/3), the Research Foundation of the Philipps-University Marburg (PhD scholarship for SF and grant), the von Behring-Röntgen-Stiftung (KR 60-0023), and by The Netherlands Organization for Scientific Research (NWO; VENI Grant 451-09-006 to VG).
Footnotes
The authors declare no competing financial interests.
References
- Adolphs R. Fear, faces, and the human amygdala. Curr Opin Neurobiol. 2008;18:166–72. doi: 10.1016/j.conb.2008.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adolphs R, Tranel D, Damasio H, Damasio aR. Fear and the human amygdala. J Neurosci. 1995;15:5879–91. doi: 10.1523/JNEUROSCI.15-09-05879.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arendt H. The Human Condition. University of Chicago Press; Chicago: 1958. The public and the private realm. [Google Scholar]
- Bandura A. Self-efficacy mechanism in human agency. Am Psychol. 1982;37:122–147. [Google Scholar]
- Blair KS, Geraci M, Korelitz K, Otero M, Towbin K, Ernst M, Leibenluft E, Blair RJR, Pine DS. The pathology of social phobia is independent of developmental changes in face processing. Am J Psychiatry. 2011a;168:1202–1209. doi: 10.1176/appi.ajp.2011.10121740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blair KS, Geraci M, Otero M, Majestic C, Odenheimer S, Jacobs M, Blair RJR, Pine DS. Atypical modulation of medial prefrontal cortex to self-referential comments in generalized social phobia. Psychiatry Res - Neuroimaging. 2011b;193:38–45. doi: 10.1016/j.pscychresns.2010.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley MM, Miccoli L, Escrig MA, Lang PJ. The pupil as a measure of emotional arousal and autonomic activation. Psychophysiology. 2008;45:602–7. doi: 10.1111/j.1469-8986.2008.00654.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckner RL, Andrews-Hanna JR, Schacter DL. The brain’s default network: anatomy, function, and relevance to disease. Ann N Y Acad Sci. 2008;1124:1–38. doi: 10.1196/annals.1440.011. [DOI] [PubMed] [Google Scholar]
- Buss AH. Self-consciousness and social anxiety. Freeman; San Francisco: 1980. [Google Scholar]
- Caggiano V, Fogassi L, Rizzolatti G, Pomper JK, Thier P, Giese Ma, Casile A. View-based encoding of actions in mirror neurons of area f5 in macaque premotor cortex. Curr Biol. 2011;21:144–8. doi: 10.1016/j.cub.2010.12.022. [DOI] [PubMed] [Google Scholar]
- Caggiano V, Fogassi L, Rizzolatti G, Thier P, Casile A. Mirror neurons differentially encode the peripersonal and extrapersonal space of monkeys. Science (80 -.) 2009;324:403–6. doi: 10.1126/science.1166818. [DOI] [PubMed] [Google Scholar]
- Cerliani L, Thomas RM, Jbabdi S, Siero JCW, Nanetti L, Crippa A, Gazzola V, D’Arceuil H, Keysers C. Probabilistic tractography recovers a rostrocaudal trajectory of connectivity variability in the human insular cortex. Hum Brain Mapp. 2012;33:2005–34. doi: 10.1002/hbm.21338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang LJ, Yarkoni T, Khaw MW, Sanfey AG. Decoding the role of the insula in human cognition: functional parcellation and large-scale reverse inference. Cereb Cortex. 2013;23:739–49. doi: 10.1093/cercor/bhs065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper JC, Dunne S, Furey T, O’Doherty JP. The role of the posterior temporal and medial prefrontal cortices in mediating learning from romantic interest and rejection. Cereb Cortex. 2014;24:2502–11. doi: 10.1093/cercor/bht102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Critcher CR, Gilovich T. Inferring attitudes from mindwandering. Personal Soc Psychol Bull. 2010;36:1255–1266. doi: 10.1177/0146167210375434. [DOI] [PubMed] [Google Scholar]
- Critchley HD. Neural mechanisms of autonomic, affective, and cognitive integration. J Comp Neurol. 2005;493:154–66. doi: 10.1002/cne.20749. [DOI] [PubMed] [Google Scholar]
- Deen B, Pitskel NB, Pelphrey KA. Three systems of insular functional connectivity identified with cluster analysis. Cereb Cortex. 2011;21:1498–506. doi: 10.1093/cercor/bhq186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberger NI, Lieberman MD, Williams KD. Does rejection hurt? An FMRI study of social exclusion. Science. 2003;302:290–2. doi: 10.1126/science.1089134. [DOI] [PubMed] [Google Scholar]
- Finger EC, Marsh Aa, Kamel N, Mitchell DGV, Blair JR. Caught in the act: the impact of audience on the neural response to morally and socially inappropriate behavior. Neuroimage. 2006;33:414–21. doi: 10.1016/j.neuroimage.2006.06.011. [DOI] [PubMed] [Google Scholar]
- Frith CD, Frith U. Interacting minds-a biological basis. Science. 1999;286:1692–5. doi: 10.1126/science.286.5445.1692. [DOI] [PubMed] [Google Scholar]
- Gilovich T, Medvec VH, Savitsky K. The spotlight effect in social judgment: An egocentric bias in estimates of the salience of one’s own actions and appearance. J Pers Soc Psychol. 2000;78:211–222. doi: 10.1037//0022-3514.78.2.211. [DOI] [PubMed] [Google Scholar]
- Guyer AE, Lau JYF, McClure-Tone EB, Parrish J, Shiffrin ND, Reynolds RC, Chen G, Blair RJR, Leibenluft E, Fox Na, Ernst M, et al. Amygdala and ventrolateral prefrontal cortex function during anticipated peer evaluation in pediatric social anxiety. Arch Gen Psychiatry. 2008;65:1303–12. doi: 10.1001/archpsyc.65.11.1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasson U, Ghazanfar Aa, Galantucci B, Garrod S, Keysers C. Brain-to-brain coupling: a mechanism for creating and sharing a social world. Trends Cogn Sci. 2012;16:114–21. doi: 10.1016/j.tics.2011.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hattie J, Timperley H. The Power of Feedback. Rev Educ Res. 2007;77:81–112. [Google Scholar]
- Kelly C, Toro R, Di Martino A, Cox CL, Bellec P, Castellanos FX, Milham MP. A convergent functional architecture of the insula emerges across imaging modalities. Neuroimage. 2012;61:1129–42. doi: 10.1016/j.neuroimage.2012.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593–602. doi: 10.1001/archpsyc.62.6.593. [DOI] [PubMed] [Google Scholar]
- Krach S, Cohrs JC, de Echeverría Loebell NC, Kircher T, Sommer J, Jansen A, Paulus FM. Your flaws are my pain: linking empathy to vicarious embarrassment. PLoS One. 2011;6:e18675. doi: 10.1371/journal.pone.0018675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krach S, Müller-Pinzler L, Westermann S, Paulus FM. Advancing the neuroscience of social emotions with social immersion. Behav Brain Sci. 2013;36:427–8. doi: 10.1017/S0140525X12001951. [DOI] [PubMed] [Google Scholar]
- Leary MR, Kowalski RM. Social Anxiety. The Guilford Press; New York: 1995. [Google Scholar]
- Leary MR, Kowalski RM. Impression Management: A Literature Review and Two-Component Model. Psychol Bull. 1990;107:34–47. [Google Scholar]
- Lindquist KA, Wager TD, Kober H, Bliss-Moreau E, Barrett LF. The brain basis of emotion: A meta-analytic review. Behav Brain Sci. 2012;35:121–43. doi: 10.1017/S0140525X11000446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorberbaum JP, Kose S, Johnson MR, Arana GW, Sullivan LK, Hamner MB, Ballenger JC, Lydiard RB, Brodrick PS, Bohning DE, George MS. Neural correlates of speech anticipatory anxiety in generalized social phobia. Neuroreport. 2004;15:2701–5. [PubMed] [Google Scholar]
- Marsh HW. A multidimensional, hierarchical model of self-concept: Theoretical and empirical justification. Educ Psychol Rev. 1990;2:77–172. [Google Scholar]
- Mattick RP, Clarke JC. Development and validation of measures of social phobia scrutiny fear and social interaction anxiety. Behav Res Ther. 1998;36:455–70. doi: 10.1016/s0005-7967(97)10031-6. [DOI] [PubMed] [Google Scholar]
- Mesulam MM, Mufson EJ. Insula of the old world monkey. III: Efferent cortical output and comments on function. J Comp Neurol. 1982;212:38–52. doi: 10.1002/cne.902120104. [DOI] [PubMed] [Google Scholar]
- Miller RS. Embarrassment: Poise and Peril in Everyday Life. The Guilford Press; New York: 1996. [Google Scholar]
- Morris JS, Friston KJ, Büchel C, Frith CD, Young AW, Calder AJ, Dolan RJ. A neuromodulatory role for the human amygdala in processing emotional facial expressions. Brain. 1998;121:47–57. doi: 10.1093/brain/121.1.47. [DOI] [PubMed] [Google Scholar]
- Morrison AS, Heimberg RG. Social anxiety and social anxiety disorder. Annu Rev Clin Psychol. 2013;9:249–74. doi: 10.1146/annurev-clinpsy-050212-185631. [DOI] [PubMed] [Google Scholar]
- Müller-Pinzler L, Rademacher L, Paulus FM, Krach S. When your friends make you cringe: social closeness modulates vicarious embarrassment related neural activity. in revision doi: 10.1093/scan/nsv130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muscatell Ka, Dedovic K, Slavich GM, Jarcho MR, Breen EC, Bower JE, Irwin MR, Eisenberger NI. Greater amygdala activity and dorsomedial prefrontal-amygdala coupling are associated with enhanced inflammatory responses to stress. Brain Behav Immun. 2014;43:46–53. doi: 10.1016/j.bbi.2014.06.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulus FM, Müller-Pinzler L, Westermann S, Krach S. On the distinction of empathic and vicarious emotions. Front Hum Neurosci. 2013;7:196. doi: 10.3389/fnhum.2013.00196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulus FM, Krach S, Blanke M, Roth C, Belke M, Sommer J, Müller-Pinzler L, Menzler K, Jansen A, Rosenow F, Bremmer F, et al. Fronto-insula network activity explains emotional dysfunctions in juvenile myoclonic epilepsy: Combined evidence from pupillometry and fMRI. Cortex. 2015;65:219–231. doi: 10.1016/j.cortex.2015.01.018. [DOI] [PubMed] [Google Scholar]
- Paulus FM, Müller-Pinzler L, Jansen A, Gazzola V, Krach S. Mentalizing and the role of the posterior superior temporal sulcus in sharing others’ embarrassment. Cereb Cortex. 2014 doi: 10.1093/cercor/bhu011. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- Phan KL, Wager T, Taylor SF, Liberzon I. Functional neuroanatomy of emotion: a meta-analysis of emotion activation studies in PET and fMRI. Neuroimage. 2002;16:331–48. doi: 10.1006/nimg.2002.1087. [DOI] [PubMed] [Google Scholar]
- Schilbach L, Timmermans B, Reddy V, Costall A, Bente G, Schlicht T, Vogeley K. Toward a second-person neuroscience. Behav Brain Sci. 2013;36:393–414. doi: 10.1017/S0140525X12000660. [DOI] [PubMed] [Google Scholar]
- Schneier FR. Social Phobia: Comorbidity and Morbidity in an Epidemiologic Sample. Arch Gen Psychiatry. 1992;49:282. doi: 10.1001/archpsyc.1992.01820040034004. [DOI] [PubMed] [Google Scholar]
- Schooler JW, Smallwood J, Christoff K, Handy TC, Reichle ED, Sayette Ma. Meta-awareness, perceptual decoupling and the wandering mind. Trends Cogn Sci. 2011;15:319–326. doi: 10.1016/j.tics.2011.05.006. [DOI] [PubMed] [Google Scholar]
- Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, Reiss AL, Greicius MD. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci. 2007;27:2349–56. doi: 10.1523/JNEUROSCI.5587-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidner LB, Stipek DJ, Feshbach ND. A developmental analysis of elementary school-aged children’s concepts of pride and embarrassment. Child Dev. 1988;59:367–77. doi: 10.1111/j.1467-8624.1988.tb01472.x. [DOI] [PubMed] [Google Scholar]
- Sennett R. The fall of public man. Cambridge University Press; Cambridge: 1974. [Google Scholar]
- Smallwood J, Schooler JW. The restless mind. Psychol Bull. 2006;132:946–958. doi: 10.1037/0033-2909.132.6.946. [DOI] [PubMed] [Google Scholar]
- Somerville LH, Heatherton TF, Kelley WM. Anterior cingulate cortex responds differentially to expectancy violation and social rejection. Nat Neurosci. 2006;9:1007–8. doi: 10.1038/nn1728. [DOI] [PubMed] [Google Scholar]
- Takahashi H, Yahata N, Koeda M, Matsuda T, Asai K, Okubo Y. Brain activation associated with evaluative processes of guilt and embarrassment: an fMRI study. Neuroimage. 2004;23:967–74. doi: 10.1016/j.neuroimage.2004.07.054. [DOI] [PubMed] [Google Scholar]
- Tangney JP, Stuewig J, Mashek DJ. Moral emotions and moral behavior. Annu Rev Psychol. 2007;58:345–72. doi: 10.1146/annurev.psych.56.091103.070145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, Mazoyer B, Joliot M. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15:273–89. doi: 10.1006/nimg.2001.0978. [DOI] [PubMed] [Google Scholar]
- Wager TD, Waugh CE, Lindquist M, Noll DC, Fredrickson BL, Taylor SF. Brain mediators of cardiovascular responses to social threat, Part I: Reciprocal dorsal and ventral sub-regions of the medial prefrontal cortex and heart-rate reactivity. Neuroimage. 2009;47:821–35. doi: 10.1016/j.neuroimage.2009.05.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren SD, Brandeis LD. The Right to Privacy. Harv Law Rev. 1890;4:193–220. [Google Scholar]
- Weiner B. An attributional theory of achievement motivation and emotion. Psychol Rev. 1985;92:548–73. [PubMed] [Google Scholar]
- Weintraub J. The theory and politics of the public/private distinction. In: Weintraub J, Kumar K, editors. Public and Private in Thought and Practice: Perspectives on a Grand Dichotomy. University of Chicago Press; Chicago: 1997. pp. 1–42. [Google Scholar]
- Wonderlic. Wonderlic Personnel Test. Wonderlic Inc.; Libertyville, IL: 1996. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.