Abstract
A rediscovery of three-dimensional culture has led to the development of organ biogenesis, homeostasis and disease models applicable to human tissues. The so-called organoids that have recently flourished serve as valuable models bridging between cell lines or primary cells grown on the bottom of culture plates and experiments performed in vivo. Though not recapitulating all aspects of organ physiology the miniature organs generated in a dish are useful models emerging for the pancreas, starting from embryonic progenitors, adult cells, tumor cells and stem cells. This review focusses on the currently available systems and their relevance to the study of the pancreas, of β-cells and of several pancreatic diseases including diabetes. We discuss the expected future developments for studying human pancreas development and function, for developing diabetes models and for producing therapeutic cells.
Keywords: bioengineering, diabetes, modelling, disease, stem cells, therapy, organoids, spheroids, spheres
Why three-dimensional (3D) culture models?
Cells, their function and their dysfunction can be studied in the body in their natural niche. This environment is chemically and structurally complex; it provides local as well as distant cues via hormones and circulating metabolites and can vary with time. The in vivo approaches are essential to study β-cell physiology, including the inputs β-cells receive from numerous metabolic tissues. They are also essential to decipher the development of the pancreas, which is coordinated with the development of other organs. In such a complex environment, it is however tedious and difficult to disentangle the fine mechanisms that govern function. For example, studies based on gene knock-outs often identify secondary consequences rather than the primary effect of a gene. Growing the cells of interest in controlled synthetic environments has been very informative to dissect their function in a simpler context. However growing primary cells freshly extracted from the body for long periods of time is not an easy task and requires identification of culture conditions that suit the cells. The primary culture of adult pancreatic cells has been reported for the three main cell types of the adult organ: acini, ducts and islets, but the differentiated exocrine and endocrine cells plated in the bottom of culture plates rapidly lose their differentiated characteristics, while suspension culture, particularly for islets, enables maintenance for a few weeks but without expansion [1–3].
Acinar, α-, β-and ductal cell lines have been derived and selected for their ease of culture. However, not all pancreatic tissues have a cell line equivalent. There is notably no cell line that has the characteristics of a pancreas progenitor, though the 266-6 line expresses some markers of progenitors but not the ability to differentiate into endocrine cells [4, 5]. Moreover the existing cell lines do not faithfully recapitulate all the features of the cell one wants to model. For example, many β-cell lines poorly secrete insulin. Moreover, while endocrine cells have a very limited and controlled ability to proliferate, cell lines have been selected to proliferate efficiently and are inadequate models for the study of endocrine cell proliferation and the cell cycle.
With regards to in vitro models of pancreas development, the culture of primary cells in 2D has been challenging, and isolated pancreatic progenitors were maintained at best for a few days in small numbers [6, 7]. However, researchers have been able to grow entire pancreatic buds, in general with their associated mesenchyme, from multiple stages of development in diverse media and culture systems, including growth on the bottom of a dish, at the interface between air and liquid or in collagen [8–11]. A possible reason is that pancreatic cells are epithelial cells which thrive in contact with other epithelial cells and in conditions that enable their polarization. Hybrid systems where progenitor cells are cultured in 3 dimensions (3D) have recently emerged combining the simplicity and controllability of in vitro culture with the possibility to reconstitute niches more similar to the natural niche. Following the example of the intestine, stomach, liver, optic cup, and brain [12–17], where 3D culture of stem or progenitor cells has enabled the generation of mini-organs with structural and functional similarities to parental organs, pancreas organoids have also emerged to model the pancreas.
A brief history of organoids
Organoids can be defined as a group of primary cells, embryonic stem cells (ESCs) or induced pluripotent stem cells (iPSCs) grown in vitro owing to their self-renewal capacities and differentiating into a 3D structure that assumes a similar organization and functionality as an organ. As an organ, they contain multiple cell types. In practice the functionality of organoids generated so far remains partial but their use has become in a few years part of the tool box of many laboratories in academia or in industry [18]. The leading work of the Clevers laboratory in developing intestinal organoids from intestinal stem cells in 2009 [19] was followed by an engineering and conceptual breakthrough, the development of organized eye cups from ESCs in 2011 by the Sasai laboratory [14]. This was continued in other organs such as the stomach, liver, lungs, prostate, kidney, pancreas, brain and many others [13, 15, 16, 20–24]. Some of their limitations are that they often lack cell types that are relevant for the complete organ function such as mesenchymal/stromal cells, blood vessels and nerves, but this can also be an asset by reducing the complexity of organ function. They usually rely on artificial extracellular matrices, often Matrigel, to promote their self-organization.
Organoids are tools to decipher the potency of stem cells, the nature of their niches and the development of the organ structure in a self-organizing process [17, 25]. They can also be powerful means to generate 3D models of human organs and of their dysfunctions, especially when initiated from human stem cells. This review focuses on the organoids developed from pancreatic cells and their potential use for understanding pancreas development, homeostasis and regeneration as well as disease modeling or therapy. Organoids modelling the pancreas remain underdeveloped as compared to the widespread use of intestinal organoids and their current limitations will be discussed.
Pancreatospheres: pancreatic progenitors, ductal cells or acinar cells expanding into hollow spheres
Frequently, epithelial cells grown in 3D develop into spheres which can be either filled or hollow but exhibit limited organization. A typical early example is the neurosphere system [26]. Though not strictly speaking organoids because of their limited organization as compared to the source organ, these 3D models are useful to expand cell types and study them. For example, spheres composed of distal lung epithelial cells or cholangiocytes have been used to study the mechanisms of cystic fibrosis and to test therapeutic drugs [27, 28]. Pancreatospheres have also been generated from pancreas progenitors, ductal cells and acinar cells.
Pancreatospheres from embryonic pancreas
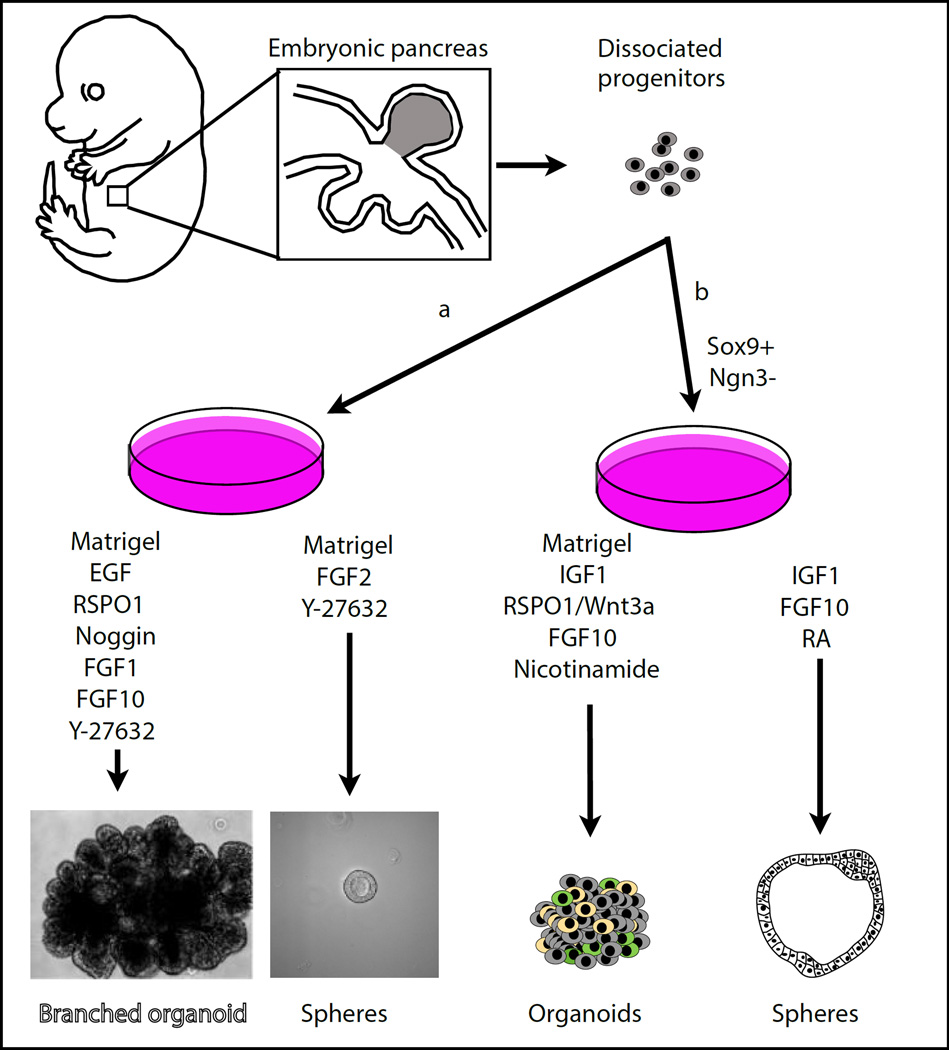
Sugiyama at al. isolated SOX9-positive and NEUROG3-negative multipotent embryonic progenitors from the E11.5 mouse pancreas [29] and showed that they could grow at clonal density in vitro in Matrigel. They formed spheres of polarized epithelial cells, most expressing Mucin1 apically and SOX9, with a subpopulation of SOX9-negative cells expressing C-peptide, glucagon and NEUROG3. This expansion protocol required the co-culture with mesenchymal cells and a defined culture medium, containing B27, FGF10, RA, IGF1, insulin and several other components (Fig. 1a). The spheres could be passaged from partially dissociated clusters of SOX9-expressing cells but after 3 passages, a decrease in multipotency is documented. Genetic manipulations of these cells showed that they recapitulate several aspects of pancreas development. An interesting feature of this system is that culturing progenitors in lower oxygen levels than common culture conditions, closer to physiological levels (5%), induced an increase of several endocrine markers, at the expense of cell proliferation. As a result, this culture system allowed the generation of glucose-responsive Insulin+ cells, which accounted for 20% of the total epithelial cells in culture. The surprising aspect of this observation is that ex vivo pancreatic explant vascularization and oxygenation has rather been shown to promote endocrine differentiation [30, 31]; thus, it is unclear whether low oxygen has an effect on differentiation or rather on selection in the pancreatosphere model.
Figure 1. Spheres and organoids from embryonic tissues.
Embryonic pancreatic progenitors from E10.5 (a) [32]) or E11.5 (b) [29]) mouse embryos are expanded in Matrigel and in presence of the medium components indicated. The expanding cells organize into spheres or organoids. Modified from [74].
Culturing pancreas progenitors in spheres is robust and can be done in diverse media. Greggio et al. [32] expanded similar pancreatospheres from E10.5 progenitors cultured in Matrigel with B27, FGF2 and the ROCK-inhibitor Y-27632 (Fig. 1b). The resulting spheres could be repeatedly passaged and resembled the structures obtained by Sugiyama et al., with homogeneous expression of progenitor markers such as SOX9, PDX1 and HNF1b and rare endocrine and acinar cells. These experiments showed that although single progenitors can initiate pancreatospheres with a low efficiency (2%), they grow better when in contact with other cells, reaching 90% chances of forming spheres for groups of more than 4 cells. Spheres were also grown in Matrigel from pancreata of human fetuses at 8–11 wpc in Matrigel using a medium containing EGF, FGF10, R-spondin, and the ROCK inhibitor, Y-27362[33]. Retrieval of EGF led the spheres filling with cells, some of which expressed endocrine markers.
Recently, spheres were also obtained from human pancreas progenitors derived from hESCs [34]. The progenitors were produced using 2D protocols until they expressed PDX1 and NKX6.1 [35], and seeded on Matrigel in a medium containing B27, ascorbic acid, insulin, hydrocortisone, FGF2, retinoic acid and the rock inhibitor Y267632. They formed spheres that expanded for 12 days, could be serially passaged and maintained progenitor markers SOX9/PDX1/NKX6.1 in most cells. Addition of a TGFb-inhibitor (A8301), the Notch inhibitor DBZ and subsequently FGF7 and EGF promoted differentiation into 10–15% ductal cells expressing carbonic anhydrase II and up to 1% cells expressing carboxypeptidase 1, likely acinar cells though this is also a progenitor marker in the mouse pancreas [36].
Such systems enable the easy maintenance of pancreas progenitors over long periods of time and their expansion to large numbers, and thus already enabled multiplexed screening for conditions that promote endocrine differentiation or other aspects of development [29]. Though still at a small scale, with improved targeting for genetic screens and penetration for drug screening, this could be scaled up.
Clonal culture from adult pancreatic ducts
Similar culture systems were developed from adult ductal cells, a cell population that naturally proliferates, though with a slow turnover [37]. All these systems also rely on the use of Matrigel as a 3D scaffold and though using different media and additives they share a few soluble factors, notably R-spondin1 [38] [39] [40].
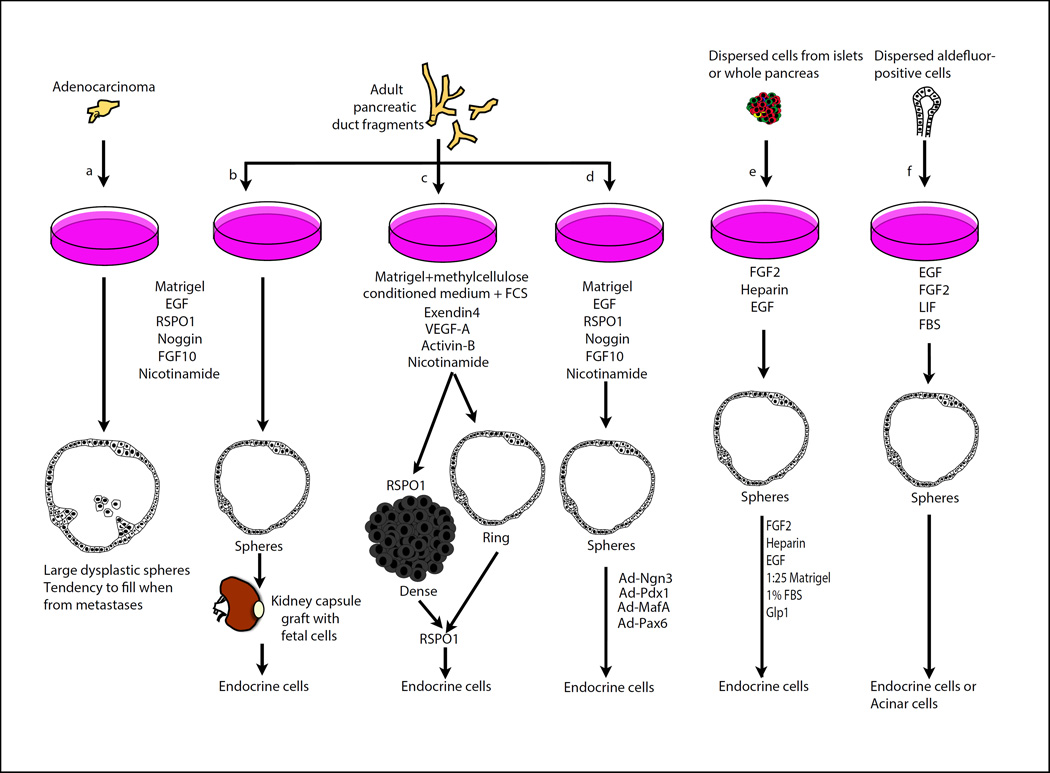
A first method is based on the FACS isolation of CD133/SOX9+ cells from adult mice, likely ductal cells or a large subpopulation of them (Fig. 2c) [38]. The growth system is complex, employing a soft gel made of methylcellulose/5% Matrigel, ESC-derived pancreatic-like cell conditioned medium, fetal calf serum, nicotinamide, activin B, exendin-4 and VEGF-A. The spheres in culture were mainly composed of ductal-like cells, but some cells expressing very low levels of acinar and/or endocrine markers were detected. R-spondin1 is instrumental in this system to promote the growth of colonies over weeks and it induces at the same time as spheres, the generation of compact high-cellularity colonies with higher endocrine progenitor markers. Transferring colonies into PolyEthyleneGlycol (PEG)-laminin promoted endocrine and acinar differentiation although it is unclear whether this promotes differentiation or selection.
Figure 2. Adult spheres and further endocrine differentiation.
Spheres forming from mouse and human adenocarcinoma (a) [57] [34], adult murine ducts (b) [39] (c) [38], or human pancreatic ducts (d) [40]), islets (e) [47, 48] or aldefluor-labelled exocrine/terminal duct cells (f) [51]. They are expanded in semisolid matrixes (Matrigel) (a-d) or in suspension on non-adhesive plates (e, f) and generate dense (c) or hollow (a-f) spheres in presence of the soluble factors indicated. Different approaches have been used to generate endocrine cells from these cultures, as indicated. Modified from [74].
Starting from duct fragments, the groups of H. Clevers and H. Heimberg could clonally expand ductal cells in Matrigel in the presence of EGF, R-spondin1 and FGF10 (Fig. 2b) [39]. Additional factors such as Noggin and Nicotinamide further improved culture over months. In this culture system, a small population of LGR5+ cells emerged. This marker is a stem cell marker of the intestine as well as several other stem cell populations in other organs but is not expressed in the pancreas unless it is injured by partial duct ligation. The spheres grown in vitro maintained an adult duct signature. A latent endocrine differentiation potential was revealed after transplantation in vivo under the kidney capsule following aggregation with embryonic pancreatic cells. How similar these cells are to endogenous endocrine cells remains to be addressed.
The expansion of adult ductal populations in spheres in vitro was also achieved in human (Fig. 2d) [40]. The culture system also requires Matrigel, as well as the soluble factors EGF, R-spondin1, FGF10, Noggin and nicotinamide. These cells could be passaged up to 3 months. Although no endocrine cells were found to spontaneously differentiate in vitro, adenoviral overexpression of NEUROG3, MAFA, PDX1 and PAX6 converted the ductal cells into endocrine progeny with many hallmarks of β-cells, including genes necessary for the synthesis, processing, storage and secretion of insulin in response to glucose or depolarization stimuli.
These systems are not strictly speaking pancreas organoids as they are essentially composed of ductal cells and they grow in spherical shapes rather than forming elongating ducts. There is a lot of debate regarding whether the ducts contribute to the formation of endocrine cells after birth in homeostasis, under physiological adaptations such as pregnancy or in response to endocrine cell loss [41–46]. The experiments done in ductal spheres add an element to the controversy. They suggest that adult pancreatic populations expanded in vitro and are amenable to endocrine differentiation, at least after overexpression of transcription factors. The degree of differentiation without transcriptional reprogramming needs to be further explored, conducting thorough validation of the cells expressing endocrine markers, including functional exploration.
Clonal suspension culture from non-ductal sources
Although the existence of multipotent stem cell populations in the pancreas is still debated, tissue culture may uncover rare populations or latent properties. Starting from dispersed adult islet or ductal populations in suspension cultures, Seaberg et al. identified cells that could form colonies leading to differentiation of insulin-expressing cells and neurons, both in mouse and human [47], in the presence of FGF2 and EGF (Fig. 2e). Cells that have the ability to expand and differentiate into both endocrine cells and neurons have not been identified in vivo in the ducts and in islets and a characterization of the cells exhibiting these properties, if of biological relevance, would be extremely interesting. Though the experiment suggests that these cells reside both in ducts and islets it is possible that this feature is due to imperfect isolation of ducts from islets. Subsequent work showed that the colonies can start from insulin-expressing cells that reduce several differentiation markers as they expand. However, it is still unclear whether a specific subpopulation of insulin-expressing can expand under these conditions or if all β-cells do so at low frequency [48].
Lastly, spheres were also initiated from centroacinar/distal ducts cells. These cells were first described as domains of active Notch/HES1 signaling in the adult pancreas. They are typically located close to the center of acini and can rapidly proliferate following pancreatic injury [49, 50]. Rovira et al. demonstrated that this population is characterized by the expression of ALDH1a1/7 and E-cadherin, a molecular signature that is used for their isolation (Fig. 2f) [51]. When seeded in suspension in the presence of EGF, FGF2, LIF and serum these rare cells (1% of pancreas) form expanding spheres. These spheres are compact, full of cells. Observations over multiple systems and organs suggest that cells grown in Matrigel tend to form monolayers whereas they form cell-filled spheres in suspension. A few cells maintained ALDH1 and SOX9 and many spheres exhibited either endocrine or acinar markers. The endocrine cells could secrete insulin in response to glucose, though not efficiently. These culture conditions may uncover the ability of exocrine or centroacinar cells to become endocrine under exceptional circumstances not found in homeostatic conditions in vivo [52–55]. The fact that spheres generate either endocrine or acinar cells rather than both suggests that this population may not be homogeneous and may contain distinct acinar progenitors and ductal cells/progenitors.
Pancreas organoids self-organizing from embryonic progenitors
We have recently identified conditions that enable the expansion, differentiation and self-organization of dissociated pancreas progenitors into a miniature organ in a dish [32]. This includes the initial formation of microlumen, as seen in the E11.5 pancreas, their coalescence into a ductal network, branching [56], the maintenance of expanding HNF1b/SOX9/PDX1-positive cells in the center, the differentiation and segregation of acinar cells to the tips of the in vitro-formed tubes as well as the formation of endocrine progenitors and endocrine cells in the center. We observed that these formations are initiated from early pancreas progenitors before the secondary transition but not later (unpublished). The formation of organoids depends on specific culture conditions in Matrigel, with a medium whose important components are knockout serum replacement, high levels of FGF10 potentiated by heparin, the ROCK-inhibitor Y-27632 and other factors that increase the efficiency of progenitor expansion (R-spondin1, EGF). The same progenitors grown in Matrigel in other media (see above and [32]) form spheres that maintain pancreas progenitors but fail to differentiate and do not self-organize. Expansion was found to require FGF and Notch signaling, as required in vivo for pancreas development. No organoid was found to form from single cells and their formation required clusters of at least 4 cells, leading to 100% organoid formation from clusters of more than 12 starting cells. Interestingly, another publication reported some level of self-organization though seemingly less structured, smaller and studied too superficially to clarify how similar the two systems are. The medium used in this system is different, with high Wnt levels in the absence of retinoic acid.
These systems recapitulate many aspects of pancreas development but the degree of maturity and functionality of differentiated exocrine and endocrine cells remains to be evaluated.
Pancreas tumor models
Organoids/spheroids have been developed as models to study mouse and human pancreatic tumor progression. Based on the protocol established to grow ductal spheres [39], spheres were developed from mouse tumor models or by transduction of oncogenes into normal spheres (Fig. 2a) [34, 57]. Moreover, small modifications to the system, including the use of TGFβ-pathway inhibitors (A83-01 and Noggin) together with R-Spondin1, Wnt3a-conditioned media, EGF and PGE2 enabled to grow spheres derived from patients with adenocarcinoma after tumor resection or from material collected by endoscopy. These spheres exhibited a different histological aspect, with a more dysplastic epithelium than the normal spheres, resembling the Pancreatic Intraepithelial Neoplasias (PanINs) and EGF- and noggin- independent proliferation unlike normal ducts. These cultures are of great interest as they enable the amplification of tumor cells without harming the patient to study the type of mutations this patient has and the associated RNA and protein profile of tumors. This could enable drug testing, guide personalized therapeutic choices, and could help in the surveillance during treatment upon re-sampling. The mutations were found to be similar in the original tumor and the amplified material, though with limiting information regarding the endoscopic biopsies, which suggest that biases linked to selection in culture are not a major problem. Orthotopic transplantation into Nude mice enabled to further study the patient material, including its progression over time and the stromal reaction it elicited. Although this system is not in the strict sense an organoid system as it does recapitulate the histology but not the shape of the ducts and does not contain other pancreatic cell types, it is expected to become very useful to help in the study and treatment of pancreatic cancer and is a first example of the utility of 3D culture system in pancreatic disease modelling.
Pancreatic tumor spheres were also grown in an independent study [34] in medium containing B27, ascorbic acid, insulin, hydrocortisone, FGF2, retinoic acid and the rock inhibitor Y267632. Interestingly, only pancreatic ductal adenocarcinoma grew in these conditions but not intraductal papillary mucinous cystic neoplasms (IPMNs). In this study, treatment with the drug most commonly used to treat PDAC, Gemcitabin, showed no effect on cell survival or expansion but a small scale screen for other drugs targeting epigenetic regulators revealed promising new therapeutic options. The authors also proposed hESC-derived pancreas progenitor spheres transduced with oncogenes as a model of cancer. It would be interesting to broaden the spectrum of tumors to represent the diversity of mutations in this cancer, as recently done as a bio banking effort with colon cancer [58].
Future directions: how can 3D in vitro culture advance our approach to pancreatic diseases and therapy?
Models of human pancreas development and developmental diseases
An exciting perspective of organoids would be to have a 3D model of human pancreas development to complement the knowledge from model organisms. Pancreatic cells from human embryos can be used to initiate such organoids. Initial efforts in this direction show the maintenance of pancreas progenitors in spheres and some signs of endocrine differentiation [33]. These achievements should be developed for a more faithful morphogenesis recapitulating duct and acinar cell formation as well as for further characterization of the endocrine cells. Considering the scarcity of human embryos, an alternative consists in using pancreas progenitors produced from human ES and induced pluripotent stem cells as an initial source of cells (iPSCs), as done for other organs such as the intestine, eye or brain [14–16]. The tools developed for other organoids may be applied to inactivate genes, modify their expression, localization or to introduce disease-alleles [59, 60]. Pioneered in the pancreas with the use of viruses to target organoid cells [29], the recent gene editing technologies should enable the modification either in the starting ES/iPSCs or directly in organoids as done in organoids from other organs [61, 62]. Such disease models would be particularly useful to study genetic diseases that affect pancreas development, such as neonatal diabetes, certain Maturity Onset Diabetes of the Young and possibly other forms of diabetes where developmental defects may contribute. Biopsied cells from patients could also potentially be developed into pancreas organoids. At the moment, such biopsies can be performed in the pancreatic ducts which develop into spheres in culture [57]. An alternative would consist in collecting more accessible biopsies and converting the patient´s cells into iPSCs. Reconstitution of organoids from such cells would enable to better understand the mechanisms of the disease and to screen for compounds for treatment. For drug testing, the thickness of organoids can be a limitation and methods to ameliorate small molecule penetration will need further development. Moreover, the cost of Matrigel, its complexity and batch variability are limitations for scaling up and standardizing screening that should be overcome by designing new 3D niches, possibly using more defined polymers.
Modelling diseases of the adult
As pioneered for pancreatic cancer, pancreas organoids would be very useful to model adult diseases as well. In the cancer field, models with multiple cell types, particularly acinar cells, and more relevant morphogenesis would extend the current possibilities to study other types of pancreatic cancers such as pancreatoblastoma, acinar cell carcinoma, cystic and mucinous cancers, insulinoma and other endocrine tumors. The conversion of acini into ducts during the early steps of adenocarcinoma would be worth studying in such a system as well as the intra papillary mucinous neoplasia. The process of metastasis could also possibly be scrutinized and also the importance of epithelial cell interactions in cancer progression. Moreover, the models open the avenue to drug efficacy tests [34] that can be performed on different tumor types organized in banks [58] or made more personal on the patient´s tumor.
Another class of diseases, the monogenic genetic diseases which develop postnatally such as cystic fibrosis or the less frequent Shwachman Diamond or Johansson Blizzard syndrome may also benefit from pancreas organoid models. For cystic fibrosis, intestinal and cholangiocyte organoids have been used for this purpose and have enabled to make new discoveries on cystic fibrosis in these organs but there is no model yet for the pancreas [61, 63, 64]. Again, model cells could either be extracted from patients, providing the relevant genetic background and modifier alleles that may affect the penetrance and expressivity, or engineered in ESCs.
For all these models, if the organoid is produced by recapitulating developmental steps of organogenesis, it will be essential to assess whether the cells of these organoids have adult features. If the cells are biopsied from adult organs, they must also be grown in conditions maintaining their adult functional characteristics.
Some diseases such as pancreatitis and diabetes have important contributions from inflammation or immunity and the fact that organoids often lack stromal cells, blood vessels, lymphatics and nerves makes the modelling of diseases in which interactions between multiple tissues matter difficult. Organoids can nevertheless be used to test the effect of specific diffusible molecules produced by inflammatory or immune cells [65]. It is also possible to include the relevant blood vessels [66], inflammatory or immune cells [67] in organoids, as initiated with liver and intestinal organoids. For type 1 and type 2 diabetes, which are diseases that develop over long periods of time, the ability to grow organoids on the long term is an important feature. It may not be generally crucial to include the exocrine components of the pancreas to model diabetes and the ability to grow islets in 3D for longer periods of time than currently possible would already be very valuable. For this purpose, culturing islets in Matrigel or other 3D hydrogels would deserve more investigations as compared to the current suspension culture. Including non-islet cells may in some cases be relevant. For example, there are cross-talks between islets and the exocrine system, notably the reduction in exocrine mass in type 1 diabetes, and this could be investigated in organoids containing all cell types.
Finally intestinal organoids have been developed to study organ infection by pathogens, which could be valuable for the pancreas too [68].
Organoids as production systems for β-cells or islets
In the last 10 years, there has been fast progress in our ability to generate β-cells in vitro starting from ESCs/iPSCs, with the purpose of transplanting these cells into patients whose cells have been destroyed (T1D) or have lost functionality (some T2D). Generating functional cells has been challenging and the Insulin+ cells produced in vitro were initially not responsive to glucose [69]. Protocols optimized based on the understanding of normal β-cell development and on testing more and more complex cytokine cocktails have led to the production of cells that are closer to our β-cells though not fully functional [70, 71]. Interestingly, these protocols have incorporated steps of 3D culture though it is not clear how the 3D environment contributes to production and maturation. Since 3D cues such as apico-basal and planar polarity complex activities are needed for appropriate endocrine cell differentiation [72, 73], it is possible that 3D culture systems improve the production of functional β-cells from ESCs/iPSCs in vitro. However, limitations due to the signals exchanged by cells in 3D culture, leading to self-organization and differentiation, may complicate the control of maturation by the medium.
The development of new matrices that are cheaper than Matrigel, less complex, more controllable and safe for transplantation will be a crucial step in this direction.
Although the use of organoids and other 3D cultures is not yet as generally prevalent as it has become to study the intestine, one can expect that their use will become widespread in the coming years, complementing in vivo investigations, experiments in cell lines and in cultured islets and that they will greatly facilitate investigation on human tissues.
Callouts.
-
-
Organoids are a group of primary cells, embryonic stem cells or induced pluripotent stem cells grown in vitro owing to their self-renewal capacities and differentiating into a 3D structure that assumes a similar organization and functionality as an organ.
-
-
As compared to 2D culture systems, organoids mix several organ cell types and respect their natural spatial arrangement. The spatial architecture of cells is often important for their function.
-
-
Pancreas organoids can be used to model mouse and human pancreas development and emerging evidence show that they can be used to model disease.
Acknowledgments
This work was supported by the Novo Nordisk Foundation, a Juvenile Diabetes Research Foundation grant [41-2009-775], National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) [1U01DK089570-01], as part of the Beta Cell Biology Consortium, and grant [12-126875] from Det Frie Forskningsråd/Sundhed og Sygdom.
Footnotes
Conflict of interest statement
The authors have no conflict of interest to disclose.
References
- 1.Blauer M, Nordback I, Sand J, Laukkarinen J. A novel explant outgrowth culture model for mouse pancreatic acinar cells with long-term maintenance of secretory phenotype. Eur J Cell Biol. 2011;90:1052–1060. doi: 10.1016/j.ejcb.2011.07.004. [DOI] [PubMed] [Google Scholar]
- 2.Houbracken I, de Waele E, Lardon J, et al. Lineage tracing evidence for transdifferentiation of acinar to duct cells and plasticity of human pancreas. Gastroenterology. 2011;141:731–741. doi: 10.1053/j.gastro.2011.04.050. 741 e731-734. [DOI] [PubMed] [Google Scholar]
- 3.Paraskevas S, Maysinger D, Wang R, Duguid TP, Rosenberg L. Cell loss in isolated human islets occurs by apoptosis. Pancreas. 2000;20:270–276. doi: 10.1097/00006676-200004000-00008. [DOI] [PubMed] [Google Scholar]
- 4.Thompson N, Gesina E, Scheinert P, Bucher P, Grapin-Botton A. Rna profiling and chromatin immunoprecipitation-sequencing reveal that ptf1a stabilizes pancreas progenitor identity via the control of mnx1/hlxb9 and a network of other transcription factors. Mol Cell Biol. 2012;32:1189–1199. doi: 10.1128/MCB.06318-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ornitz DM, Palmiter RD, Messing A, et al. Elastase i promoter directs expression of human growth hormone and sv40 t antigen genes to pancreatic acinar cells in transgenic mice. Cold Spring Harb Symp Quant Biol. 1985;50:399–409. doi: 10.1101/sqb.1985.050.01.050. [DOI] [PubMed] [Google Scholar]
- 6.Sugiyama T, Rodriguez RT, McLean GW, Kim SK. Conserved markers of fetal pancreatic epithelium permit prospective isolation of islet progenitor cells by facs. Proc Natl Acad Sci U S A. 2007;104:175–180. doi: 10.1073/pnas.0609490104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Furuyama K, Kawaguchi Y, Akiyama H, et al. Continuous cell supply from a sox9-expressing progenitor zone in adult liver, exocrine pancreas and intestine. Nat Genet. 2011;43:34–41. doi: 10.1038/ng.722. [DOI] [PubMed] [Google Scholar]
- 8.Miralles F, Czernichow P, Scharfmann R. Follistatin regulates the relative proportions of endocrine versus exocrine tissue during pancreatic development. Development. 1998;125:1017–1024. doi: 10.1242/dev.125.6.1017. [DOI] [PubMed] [Google Scholar]
- 9.Duvillie B, Attali M, Bounacer A, et al. The mesenchyme controls the timing of pancreatic beta-cell differentiation. Diabetes. 2006;55:582–589. doi: 10.2337/diabetes.55.03.06.db05-0839. [DOI] [PubMed] [Google Scholar]
- 10.Burke ZD, Li WC, Slack JM, Tosh D. Isolation and culture of embryonic pancreas and liver. Methods Mol Biol. 2010;633:91–99. doi: 10.1007/978-1-59745-019-5_7. [DOI] [PubMed] [Google Scholar]
- 11.Johansson KA, Dursun U, Jordan N, et al. Temporal control of neurogenin3 activity in pancreas progenitors reveals competence windows for the generation of different endocrine cell types. Dev Cell. 2007;12:457–465. doi: 10.1016/j.devcel.2007.02.010. [DOI] [PubMed] [Google Scholar]
- 12.Barker N, Huch M, Kujala P, et al. Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell. 2010;6:25–36. doi: 10.1016/j.stem.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 13.Huch M, Dorrell C, Boj SF, et al. In vitro expansion of single lgr5+ liver stem cells induced by wnt-driven regeneration. Nature. 2013;494:247–250. doi: 10.1038/nature11826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Eiraku M, Takata N, Ishibashi H, et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature. 2011;472:51–56. doi: 10.1038/nature09941. [DOI] [PubMed] [Google Scholar]
- 15.Lancaster MA, Renner M, Martin CA, et al. Cerebral organoids model human brain development and microcephaly. Nature. 2013;501:373–379. doi: 10.1038/nature12517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Spence JR, Mayhew CN, Rankin SA, et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011;470:105–109. doi: 10.1038/nature09691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sasai Y, Eiraku M, Suga H. In vitro organogenesis in three dimensions: Self-organising stem cells. Development. 2012;139:4111–4121. doi: 10.1242/dev.079590. [DOI] [PubMed] [Google Scholar]
- 18.Fatehullah A, Tan SH, Barker N. Organoids as an in vitro model of human development and disease. Nat Cell Biol. 2016;18:246–254. doi: 10.1038/ncb3312. [DOI] [PubMed] [Google Scholar]
- 19.Sato T, Vries RG, Snippert HJ, et al. Single lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–265. doi: 10.1038/nature07935. [DOI] [PubMed] [Google Scholar]
- 20.McCracken KW, Cata EM, Crawford CM, et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 2014;516:400–404. doi: 10.1038/nature13863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chua CW, Shibata M, Lei M, et al. Single luminal epithelial progenitors can generate prostate organoids in culture. Nat Cell Biol. 2014;16:951–961. doi: 10.1038/ncb3047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dye BR, Hill DR, Ferguson MA, et al. In vitro generation of human pluripotent stem cell derived lung organoids. eLife. doi: 10.7554/eLife.05098. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Takasato M, Er PX, Chiu HS, et al. Kidney organoids from human ips cells contain multiple lineages and model human nephrogenesis. Nature. 2015;526:564–568. doi: 10.1038/nature15695. [DOI] [PubMed] [Google Scholar]
- 24.Rock JR, Onaitis MW, Rawlins EL, et al. Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc Natl Acad Sci U S A. 2009;106:12771–12775. doi: 10.1073/pnas.0906850106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sasai Y. Cytosystems dynamics in self-organization of tissue architecture. Nature. 2013;493:318–326. doi: 10.1038/nature11859. [DOI] [PubMed] [Google Scholar]
- 26.Reynolds BA, Weiss S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science. 1992;255:1707–1710. doi: 10.1126/science.1553558. [DOI] [PubMed] [Google Scholar]
- 27.Sampaziotis F, Cardoso de Brito M, Madrigal P, et al. Cholangiocytes derived from human induced pluripotent stem cells for disease modeling and drug validation. Nat Biotechnol. 2015;33:845–852. doi: 10.1038/nbt.3275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hannan NR, Sampaziotis F, Segeritz CP, Hanley NA, Vallier L. Generation of distal airway epithelium from multipotent human foregut stem cells. Stem Cells Dev. 2015;24:1680–1690. doi: 10.1089/scd.2014.0512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sugiyama T, Benitez CM, Ghodasara A, et al. Reconstituting pancreas development from purified progenitor cells reveals genes essential for islet differentiation. Proc Natl Acad Sci U S A. 2013;110:12691–12696. doi: 10.1073/pnas.1304507110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Heinis M, Simon MT, Ilc K, et al. Oxygen tension regulates pancreatic beta-cell differentiation through hypoxia-inducible factor 1alpha. Diabetes. 2010;59:662–669. doi: 10.2337/db09-0891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shah SR, Esni F, Jakub A, et al. Embryonic mouse blood flow and oxygen correlate with early pancreatic differentiation. Dev Biol. 2011;349:342–349. doi: 10.1016/j.ydbio.2010.10.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Greggio C, De Franceschi F, Figueiredo-Larsen M, et al. Artificial three-dimensional niches deconstruct pancreas development in vitro. Development. 2013;140:4452–4462. doi: 10.1242/dev.096628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bonfanti P, Nobecourt E, Oshima M, et al. Ex vivo expansion and differentiation of human and mouse fetal pancreatic progenitors are modulated by epidermal growth factor. Stem Cells Dev. 2015;24:1766–1778. doi: 10.1089/scd.2014.0550. [DOI] [PubMed] [Google Scholar]
- 34.Huang L, Holtzinger A, Jagan I, et al. Ductal pancreatic cancer modeling and drug screening using human pluripotent stem cell- and patient-derived tumor organoids. Nat Med. 2015;21:1364–1371. doi: 10.1038/nm.3973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nostro MC, Sarangi F, Yang C, et al. Efficient generation of nkx6-1+ pancreatic progenitors from multiple human pluripotent stem cell lines. Stem Cell Reports. 2015;4:591–604. doi: 10.1016/j.stemcr.2015.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhou Q, Law AC, Rajagopal J, et al. A multipotent progenitor domain guides pancreatic organogenesis. Dev Cell. 2007;13:103–114. doi: 10.1016/j.devcel.2007.06.001. [DOI] [PubMed] [Google Scholar]
- 37.Magami Y, Azuma T, Inokuchi H, et al. Heterogeneous cell renewal of pancreas in mice: [(3)h]-thymidine autoradiographic investigation. Pancreas. 2002;24:153–160. doi: 10.1097/00006676-200203000-00006. [DOI] [PubMed] [Google Scholar]
- 38.Jin L, Feng T, Shih HP, et al. Colony-forming cells in the adult mouse pancreas are expandable in matrigel and form endocrine/acinar colonies in laminin hydrogel. Proc Natl Acad Sci U S A. 2013;110:3907–3912. doi: 10.1073/pnas.1301889110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Huch M, Bonfanti P, Boj SF, et al. Unlimited in vitro expansion of adult bi-potent pancreas progenitors through the lgr5/r-spondin axis. EMBO J. 2013;32:2708–2721. doi: 10.1038/emboj.2013.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lee J, Sugiyama T, Liu Y, et al. Expansion and conversion of human pancreatic ductal cells into insulin-secreting endocrine cells. eLife. 2013;2:e00940. doi: 10.7554/eLife.00940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Inada A, Nienaber C, Katsuta H, et al. Carbonic anhydrase ii-positive pancreatic cells are progenitors for both endocrine and exocrine pancreas after birth. Proc Natl Acad Sci U S A. 2008;105:19915–19919. doi: 10.1073/pnas.0805803105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xu X, D'Hoker J, Stange G, et al. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell. 2008;132:197–207. doi: 10.1016/j.cell.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 43.Courtney M, Gjernes E, Druelle N, et al. The inactivation of arx in pancreatic alpha-cells triggers their neogenesis and conversion into functional beta-like cells. PLoS Genet. 2013;9:e1003934. doi: 10.1371/journal.pgen.1003934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Solar M, Cardalda C, Houbracken I, et al. Pancreatic exocrine duct cells give rise to insulin-producing beta cells during embryogenesis but not after birth. Dev Cell. 2009;17:849–860. doi: 10.1016/j.devcel.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 45.Kopp JL, Dubois CL, Schaffer AE, et al. Sox9+ ductal cells are multipotent progenitors throughout development but do not produce new endocrine cells in the normal or injured adult pancreas. Development. 2011;138:653–665. doi: 10.1242/dev.056499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dor Y, Brown J, Martinez OI, Melton DA. Adult pancreatic beta-cells are formed by self-duplication rather than stem-cell differentiation. Nature. 2004;429:41–46. doi: 10.1038/nature02520. [DOI] [PubMed] [Google Scholar]
- 47.Seaberg RM, Smukler SR, Kieffer TJ, et al. Clonal identification of multipotent precursors from adult mouse pancreas that generate neural and pancreatic lineages. Nat Biotechnol. 2004;22:1115–1124. doi: 10.1038/nbt1004. [DOI] [PubMed] [Google Scholar]
- 48.Smukler SR, Arntfield ME, Razavi R, et al. The adult mouse and human pancreas contain rare multipotent stem cells that express insulin. Cell Stem Cell. 2011;8:281–293. doi: 10.1016/j.stem.2011.01.015. [DOI] [PubMed] [Google Scholar]
- 49.Hayashi KY, Tamaki H, Handa K, et al. Differentiation and proliferation of endocrine cells in the regenerating rat pancreas after 90% pancreatectomy. Arch Histol Cytol. 2003;66:163–174. doi: 10.1679/aohc.66.163. [DOI] [PubMed] [Google Scholar]
- 50.Stanger BZ, Stiles B, Lauwers GY, et al. Pten constrains centroacinar cell expansion and malignant transformation in the pancreas. Cancer Cell. 2005;8:185–195. doi: 10.1016/j.ccr.2005.07.015. [DOI] [PubMed] [Google Scholar]
- 51.Rovira M, Scott SG, Liss AS, et al. Isolation and characterization of centroacinar/terminal ductal progenitor cells in adult mouse pancreas. Proc Natl Acad Sci U S A. 2010;107:75–80. doi: 10.1073/pnas.0912589107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Desai BM, Oliver-Krasinski J, De Leon DD, et al. Preexisting pancreatic acinar cells contribute to acinar cell, but not islet beta cell, regeneration. J Clin Invest. 2007;117:971–977. doi: 10.1172/JCI29988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kopinke D, Brailsford M, Shea JE, et al. Lineage tracing reveals the dynamic contribution of hes1+ cells to the developing and adult pancreas. Development. 2011;138:431–441. doi: 10.1242/dev.053843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature. 2008;455:627–632. doi: 10.1038/nature07314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Minami K, Okuno M, Miyawaki K, et al. Lineage tracing and characterization of insulin-secreting cells generated from adult pancreatic acinar cells. Proc Natl Acad Sci U S A. 2005;102:15116–15121. doi: 10.1073/pnas.0507567102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dahl-Jensen SB, Figueiredo-Larsen M, Grapin-Botton A, Sneppen K. Short-range growth inhibitory signals from the epithelium can drive non-stereotypic branching in the pancreas. Phys Biol. 2016;13:016007. doi: 10.1088/1478-3975/13/1/016007. [DOI] [PubMed] [Google Scholar]
- 57.Boj SF, Hwang CI, Baker LA, et al. Organoid models of human and mouse ductal pancreatic cancer. Cell. 2015;160:324–338. doi: 10.1016/j.cell.2014.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.van de Wetering M, Francies HE, Francis JM, et al. Prospective derivation of a living organoid biobank of colorectal cancer patients. Cell. 2015;161:933–945. doi: 10.1016/j.cell.2015.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Koo BK, Sasselli V, Clevers H. Retroviral gene expression control in primary organoid cultures. Curr Protoc Stem Cell Biol. 2013;27 doi: 10.1002/9780470151808.sc05a06s27. Unit 5A.6. [DOI] [PubMed] [Google Scholar]
- 60.Koo BK, Stange DE, Sato T, et al. Controlled gene expression in primary lgr5 organoid cultures. Nat Methods. 2012;9:81–83. doi: 10.1038/nmeth.1802. [DOI] [PubMed] [Google Scholar]
- 61.Schwank G, Koo BK, Sasselli V, et al. Functional repair of cftr by crispr/cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell. 2013;13:653–658. doi: 10.1016/j.stem.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 62.Matano M, Date S, Shimokawa M, et al. Modeling colorectal cancer using crispr-cas9-mediated engineering of human intestinal organoids. Nat Med. 2015;21:256–262. doi: 10.1038/nm.3802. [DOI] [PubMed] [Google Scholar]
- 63.Dekkers JF, Wiegerinck CL, de Jonge HR, et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat Med. 2013;19:939–945. doi: 10.1038/nm.3201. [DOI] [PubMed] [Google Scholar]
- 64.Liu J, Walker NM, Ootani A, Strubberg AM, Clarke LL. Defective goblet cell exocytosis contributes to murine cystic fibrosis-associated intestinal disease. J Clin Invest. 2015;125:1056–1068. doi: 10.1172/JCI73193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Farin HF, Karthaus WR, Kujala P, et al. Paneth cell extrusion and release of antimicrobial products is directly controlled by immune cell-derived ifn-gamma. J Exp Med. 2014;211:1393–1405. doi: 10.1084/jem.20130753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Takebe T, Sekine K, Enomura M, et al. Vascularized and functional human liver from an ipsc-derived organ bud transplant. Nature. 2013;499:481–484. doi: 10.1038/nature12271. [DOI] [PubMed] [Google Scholar]
- 67.Nozaki K, Mochizuki W, Matsumoto Y, et al. Co-culture with intestinal epithelial organoids allows efficient expansion and motility analysis of intraepithelial lymphocytes. J Gastroenterol. 2016;51:206–213. doi: 10.1007/s00535-016-1170-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Bartfeld S, Bayram T, van de Wetering M, et al. In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection. Gastroenterology. 2015;148:126–136.e6. doi: 10.1053/j.gastro.2014.09.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Schiesser JV, Wells JM. Generation of beta cells from human pluripotent stem cells: Are we there yet? Ann N Y Acad Sci. 2014;1311:124–137. doi: 10.1111/nyas.12369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rezania A, Bruin JE, Arora P, et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat Biotechnol. 2014;32:1121–1133. doi: 10.1038/nbt.3033. [DOI] [PubMed] [Google Scholar]
- 71.Pagliuca FW, Millman JR, Gurtler M, et al. Generation of functional human pancreatic beta cells in vitro. Cell. 2014;159:428–439. doi: 10.1016/j.cell.2014.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cortijo C, Gouzi M, Tissir F, Grapin-Botton A. Planar cell polarity controls pancreatic beta cell differentiation and glucose homeostasis. Cell Rep. 2012;2:1593–1606. doi: 10.1016/j.celrep.2012.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kesavan G, Sand FW, Greiner TU, et al. Cdc42-mediated tubulogenesis controls cell specification. Cell. 2009;139:791–801. doi: 10.1016/j.cell.2009.08.049. [DOI] [PubMed] [Google Scholar]
- 74.Greggio C, De Franceschi F, Grapin-Botton A. In vitro-produced pancreas organogenesis models in three dimensions: Self-organization from few stem cells or progenitors. Stem Cells. 2015;43:8–15. doi: 10.1002/stem.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]