Abstract
Kindler syndrome1, 2 is a genetic disorder mainly characterized by increased skin fragility and photosensitivity,3, 4 making the use of treatments based on radiation difficult or even prohibited. Thus, cases reporting Kindler syndrome patients treated with radiotherapy are rare. In this study, we report clinical outcomes and care provided for a rare case of a Kindler syndrome patient submitted to radiotherapy. Diagnosed with squamous cell carcinoma involving the buccal mucosa, the patient was exclusively treated with radiotherapy, with 70 Gy delivered on the PTV with the Volumetric Modulated Arc technique. The patient's reaction regarding control of the lesion is relevant compared to patients not affected by the syndrome. We noticed acute reactions of the skin and buccal mucosa after few radiotherapy sessions, followed by a fast reduction in the tumor volume. The efficacy of radiotherapy along with multidisciplinary actions allowed treatment continuity, leading to a complete control of the lesion and life quality improvement and showed that the use of radiotherapy on Kindler syndrome patients is possible.
Abbreviations: KS, Kindler syndrome; GTV, gross tumor volume; CTV, clinical tumor volume; PTV, planning tumor volume; HDMLC, high definition multileaf collimator; OAR, organs at risk; PRV, planning risk volume; QUANTEC, quantitative analysis of normal tissue effects in the clinic; NTO, normal tissue optimization; RTOG, Therapy Oncology Group
Keywords: Kindler syndrome, Head and neck cancer, Radiotherapy, Skin fragility and photosensitivity
1. Case representation
A female patient, previously diagnosed with Kindler syndrome, presented with an initial lesion on the right cheek, believed to be due to the syndrome. The lesion was, however, diagnosed as squamous cell carcinoma involving buccal mucosa. Six months after the initial complaint, the disease had already extended to the skin of upper and lower right lips and jugal mucosa. Due to the condition of the lesion, radiotherapy was ruled the best treatment option. However, radiotherapy represents a challenge to head and neck cancer patients with Kindler syndrome due to their characteristic photosensitivity and to unknown consequences of high energy exposure. By the end of the treatment, the external visible tumor mass resolved completely with controlled toxicity of epithelitis and mucositis.
2. Introduction
Radiotherapy applied to head and neck pathologies yields good responses regarding local control of the disease and toxicity on surrounding tissues. It was shown efficient, both exclusively and combined with chemotherapy or surgery, in several oral cavity tumor studies in patients with favorable conditions.5, 6, 7 However, there are few results described from patients affected by syndromes that hinder the application of radiotherapy, such as Kindler syndrome (KS).8 These patients present increased skin and organ toxicity in response to radiation due to photosensitivity, preventing the use of radiotherapy. However, although usually not indicated, radiotherapy can sometimes be the only treatment option for KS patients with head and neck tumors. In this work, we describe the use of radiotherapy to treat an oral cavity tumor on a KS patient. We also discuss its evolution and care provided to allow the treatment.
The patient was diagnosed with a primary lesion on the oral cavity, on the jugal mucosa, with oro-cutaneous fistula, and bone invasion detected on tomography. The lesion was previously assessed by the surgical team and deemed unresectable and at high risk of complications with chemotherapy, which would have only a palliative role if exclusive. Therefore, in a multidisciplinary assessment, exclusive radiotherapy was chosen as a potentially curative option. Radiation was applied in 33 fractions, with a total dose of 70 Gy on the lesion primary volume and 54 Gy on lymph drainage. The Volumetric Modulated Arc Therapy was used to deliver conformational doses to the tumor volume with high gradient on the boundaries between the lesion and healthy tissues to reduce skin toxicity.9, 10, 11
The treatment definition demanded a multidisciplinary team effort, including weekly reviews with the radiation oncologist, monitoring by the dental team with 560 nm laser applications, and adequate nutrition by tube feeding due to the esophageal stenosis caused by the syndrome.
During treatment, we observed a reduction in the tumor mass and acute toxicity, with epithelitis and mucositis, reaching Grade I according to the RTOG12 study on week two, evolving to Grade II throughout treatment, affecting mainly the primary lesion surroundings and lips. Local response is observed on week one, with a tumor regression of 10%, followed by relief of neoplasia-related pain. Fistula improved continuously until full response by the end of week seven, showing that radiotherapy is a viable prescription, even in KS patients.
2.1. Reported case
A 55-year-old female patient, with no history of smoking, previous or current, in good general condition (ECOG performance status 1) and tumor staging cT4cN0M0. Before treatment, the lesion was ulcerated, with oro-cutaneous fistula, affecting the skin of upper and lower right lips, labial commissure, and right jugal mucosa. The patient also presented skin alterations such as fibrosis, cutaneous atrophy and bullous lesions on the oral mucosa and lips, as shown in Fig. 1. The patient also has a history of esophageal stenosis previously treated with endoscopic dilation.
Fig. 1.

Status of the lesion before treatment. Fissure on right cheek showing the tumor extending to the lips, and blisters on the lips caused by Kindler syndrome.
The patient was diagnosed with KS, a type of autossomic recessive epidermolisis bullosa, characterized by blisters on the skin, photosensitivity, progressive poikiloderma, and extensive skin atrophy.1, 2, 3, 4, 13 The oral cavity is frequently affected and common aspects include: microstomia, tongue mobility abnormalities, ulcerations, and occurrence of malignant tumors from the metaplasia of ulcerations. In addition to cutaneous involvement, the illness can also affect the mucosa, worsening progressively with age, and can cause dental alterations such as gingivitis and periodontitis. Extra-cutaneous affections such as esophageal stenosis, colitis and other gastrointestinal affections are also present. However, the main reason for concern is patient's photosensitivity and skin fragility upon high energy radiation exposure whose consequences are still unknown.
The patient was submitted to computerized tomography for the reconstruction of risk and treatment volumes. GTV1 delimitation corresponds to the primary tumor volume boundaries (43.1 cm3). Clinical CTV1 (138.8 cm3) and planning volume PTV1 (192.3 cm3) are estimated from GTV1 expansion, with margins of 1 cm and 1.4 cm, respectively.14 Lymph node chains are associated to CTV2 and PTV2 relative to the elective treatment chains, at Level IA/B, IIA/B, III, IV. Level V and retrofaringeal chain were, thus, excluded because they are considered a low risk of commitment by the radiation oncologists.
2.2. Physical planning
The patient was treated with a 6 MV beam on a Varian Trilogy linear accelerator equipped with 120 leaves HDMLC (with spatial resolution of 0.25 at the isocenter) and planned using Eclipse treatment planning system (version 11.0 Varian Medical System). Considering the skin acute toxicity and aiming to minimize the effects of radiation on tissues, we used the volumetric arc therapy (fast arc) of modulated intensity [1112,15]. For head and neck tumor treatment, fast arc has advantages over 3D and modulated intensity (IMRT) techniques15, 16 because it is able to deliver an appropriate dose distribution on the PTVs and lymph drainage, sparing the surrounding tissues with shorter treatment time. Physical treatment was optimized to deliver two dose levels, with the main total dose of 70 Gy at the primary lesion and 54 Gy at the lymph node volume delimited to levels I to VA. The treatment was applied in 33 fractions during 6.6 weeks.
The organs at risk (OAR) drawn were parotid glands, spinal cord plus a 5 mm margin to create PRV, oral cavity, pharynx and trachea region, which were 3 mm away from any PTV. All described OARs received the upper dose limit according to the TDs prescribed on The Quantitative Analysis of Normal Tissue Effects in the Clinic (QUANTEC).17 To avoid hot spots on healthy tissues and an abrupt dose drop on the surroundings of PTVs, we used the Normal Tissue Optimization (NTO) tool with the following parameters: 0.5 cm of distance from the PTV, 100% initial dose, 40% final dose, and 0.08 fall off. Two co-planar arcs with rotation of 358o and difference of 40° between collimator angles form the radiation fields setup and create a dose distribution that keeps the hot spot within the GTV volume and 1.5 cm away from the patient's skin boundaries, as shown in Fig. 2. Total GTV1 and PTV1 coverage reached 100% and 95% of the prescribed dose, respectively. The gradient of dose reached 8%, obtained from the ratio between dose values of 98% and 2%. Homogeneity and conformity indices were 0.16 and 1.03, respectively. Values obtained from optimization are consistent with the results obtained on the report planning.18, 19, 20
Fig. 2.
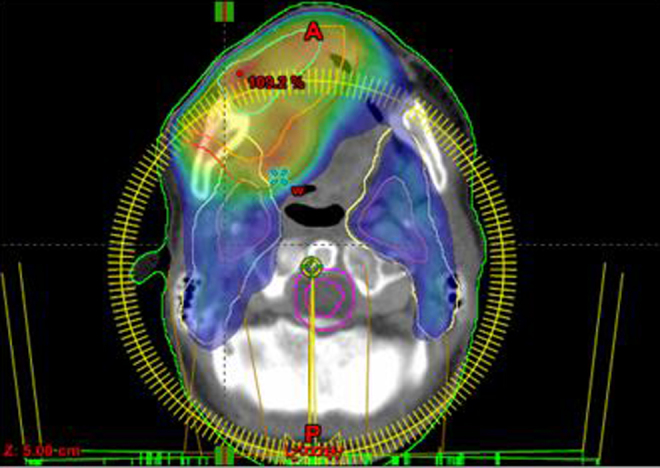
Axial section representing treatment volumes GTV1, CTV1, PTV1, CTV2 and PTV2. Gradient of dose on treatment volumes of 8%, with 73.3% coverage at the PTV2.
3. Results
The assessment of response to radiotherapy is performed with lesion monitoring using the RECIST guideline criterion21 and the skin toxicity degrees throughout treatment. To check the lesion clinical reduction, outer dimensions were measured from daily photographs. For skin toxicity, we used the concepts established on the RTOG reported studies.17 The classification was determined by the radiation oncologist on weekly examinations.
Fig. 1 shows the lesion after the first radiotherapy application. The oro-cutaneous lesion outer area on the right cheek was estimated as 4 cm2. The patient also presented lesions due to KS as facial skin fibrosis, and bullous lesions on the oral mucosa and lips. Table 1 presents tumor evolution and toxicity for each week of treatment.
Table 1.
Description of tumor reduction and toxicity grade for mucositis and epithelitis.
| Period (week) | Tumor | Mucositis | Epithelitis |
|---|---|---|---|
| 1st (Five fractions) | A mild reduction of the superficial lesion area | Grade III, bullous lesions and moderate pain | Grade I, Bright and moderate erythema, |
| 2st (Ten fractions) | Reduction of around 50% from initial lesion area | Grade III, unchanged | Grade I, pronounced edema |
| 3st (Fifteen fractions) | Continuous reduction of lesion area and relief of neoplasia-related pain | Grade III, moderate fibrosis | Grade II, patchy moist desquamation |
| 4th (Twenty fractions) | Reduction of nearly 90% of the initial lesion area and reduction of pain | Grade III, unchanged | Grade II, unchanged |
| 5th (Twenty five fractions) | Complete reduction of lesion | Grade III, unchanged | Grade II, unchanged |
| 6th (Thirty three fractions) | Complete reduction of lesion, good quality of life and no neoplasia-related pain | Grade III, unchanged | Grade II, unchanged |
By the end of the first week, the skin reaction was more aggressive than that usually reported for the same treatment period.22, 23 This is likely due to the increased skin sensitivity to radiation caused by KS. Facial toxicity reached Grade III for mucositis and Grade I for epithelitis following the 5th fraction of radiotherapy. For the neck region, the toxicity for epithelitis was Grade I. As an attempt to reduce skin damage and prevent treatment discontinuation, laser therapy with 560 nm laser and Mometasone ointment medicine were applied daily. The lesion had a mild improvement, with an estimated reduction of the superficial lesion area of 10%.
On week two, following ten fractions of radiotherapy, the toxicity of skin and mucosa remained unchanged, both for the neck and cheek. However, a surprising response was observed in the lesion area. The patient also reported relief of neoplasia-related pain on week three. During treatment, we observed a slight worsening of mucositis but still classified as grade III until 6.6 weeks. However, epithelitis began as grade I but evolved to grade II by the end of week three. The radiotherapy produces a continuous improvement of lesion local control evolving to a complete resolution of the fistula on the fifth week of treatment, as shown in Fig. 3.
Fig. 3.

Progressive reduction of the lesion, toxicity Grade III for mucositis and Grade II for epithelitis.
4. Discussion
KS is associated with gene KIND1, located on 20p12.3, that codes for protein kindlin_1. This protein plays an important role on keratinocytes focal contact in epidermis, and the lack of its expression causes skin fragility and abnormalities. However, the relationship between kindlin_1 protein and photosensitivity, a unique characteristic of KS, is still unknown. Furthermore, the relationship between the response to ionizing radiation, both in healthy and tumor tissues, and the genes related to the syndrome raises even more questions.
To our knowledge, literature shows only one report of radiotherapy on a KS patient.24 In that case, authors used a total dose of 72 Gy, fractionated in 8 weeks with 1.8 Gy/fr, on a 34-year-old patient with non-resectable squamous cell carcinoma (SCC) located on the hard palate and in stage I. Authors also reported a complete tumor regression two weeks after the last application. This common feature suggests a more intense response of tumor cells to ionizing radiation, possibly related to KS.
Results presented here suggest that KS affects both normal and tumor cells, and that the mechanisms of ionizing radiation interaction with the cells produce more damage on the tumor cell, leading to a faster response. Using the criteria stated on RECIST guideline,21 the tumor reached full response with complete remission of the lesion in all directions (Fig. 3) at the external cheek. However, the extension of the lesion reduction in the oral cavity was not measurable, due to necrosis and fibrosis in the area. One month after the end of the treatment, the local control started to show signs of a new tumor growth. This lesion was assessed as resectable and the patient was submitted to surgery at the fifth month.
At the beginning of treatment, there was a great concern about the increase of toxicity of the skin and lips, particularly due to the rapid acute effect in the first week. However, the toxicity remained almost constant throughout the treatment, except for an increase in Epithelitis toxicity on the neck and cheek, reaching a Grade II at the end of the treatment. While the effects of ionizing radiation on the skin and mucosa of KS patients are unknown, the toxicity was found below our expectations, either because it reached a threshold or due to the effects of laser and medicine used. The Grade II of toxicity observed with this patient is slightly above those obtained in our routine service or reported in literature and is very similar to that reported for a KS case.24
5. Conclusion
Here, we present the treatment evolution for a patient diagnosed with squamous cell carcinoma associated to Kindler syndrome. Results show a rapid tumor response and pain relief. Toxicity due to photosensitivity was successfully managed, allowing treatment continuity. This work suggests that the use of advanced techniques in treatment planning, associated to toxicity control measures enables the use of radiotherapy in Kindler syndrome patients.
Consent
Written informed consent was obtained from the patient for publication of this Case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Authors’ contribution
AC (correspondent author), WC and TP are medical physicists and contributed substantially to the treatment, radiation planning, acquisition of data, analysis and interpretation of data. AB, CS and FM are the physicians responsible for the patient treatment since the patient's admission to the Clinic. They were also involved in drafting the manuscript. KL is the dentist responsible for the daily laser sections and contributed to the manuscript. CN is the research manager and have supervised the manuscript preparation. MS is a nurse responsible for taking daily pictures and monitoring the treatment evolution and also participated in the manuscript. All authors read and approved the final manuscript.
Conflict of interest
None declared.
Financial disclosure
None declared.
Contributor Information
Ademar Caldeira, Email: adcafi@yahoo.com.br.
William Correia Trinca, Email: william.trinca@maededeus.com.br.
Thais Pires Flores, Email: thais.pires.flores@hotmail.com.
Andrea Barleze Costa, Email: andrea.barleze@gmail.com.
Claudio de Sá Brito, Email: claudiodesabrito@gmail.com.
Karen Loureiro Weigert, Email: karenwodonto@gmail.com.
Maryana Schwartzhaupt Matos, Email: maryana.matos@gmail.com.
Carmela Nicolini, Email: carmela.pesquisa@gmail.com.
Fernando Mariano Obst, Email: fernando.obst@terra.com.
References
- 1.Kindler T. Congenital poikiloderma with traumatic bulla formation and progressive cutaneous atrophy. Br J Dermatol. 1954;66(3):104–111. doi: 10.1111/j.1365-2133.1954.tb12598.x. [DOI] [PubMed] [Google Scholar]
- 2.Sharon J.W., Irwin M.W.H. Kindler surprise: mutations in a novel actin-associated protein cause Kindler syndrome. J Dermatol Sci. 2005;38(3):169–175. doi: 10.1016/j.jdermsci.2004.12.026. [DOI] [PubMed] [Google Scholar]
- 3.Siege D.H., Ashotn G.H., Penagos S.H.G. Loss of Kindlin-1, a human homolog of the Caenorhabditis elegans actin – extracellular-matrix linker protein UNC-112, causes Kindler syndrome. Am J Hum Genet. 2003;73(1):174–187. doi: 10.1086/376609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Has C., Burger B., Volz A., Kohlhase J., Bruckner L., Itin P. Mild clinical phenotype of kindler syndrome associated with late diagnosis and skin cancer. Dermatology. 2010;5(221):309–312. doi: 10.1159/000320235. [DOI] [PubMed] [Google Scholar]
- 5.Justin W., Robert J.A., Christopher G.M., Werning J., William M. Mendenhall efficacy of elective nodal irradiation in skin squamous cell carcinoma of the face, ears, and scalp. Radiat Oncol. 2015;10:199. doi: 10.1186/s13014-015-0509-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen A.M., Grekin R.C., Garcia J., Bucci M.K., Margolis L.W. Radiation therapy for cutaneous squamous cell carcinoma involving the parotid area lynphnodes: dose and volume considerations. Int J Radiat Oncol Biol Phys. 2007;69(5):77–80. doi: 10.1016/j.ijrobp.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 7.National Comprehensive Cancer Network; Fort Washington: 2014. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Squamous Cell Skin Cancer, Version 1.2015. Available from: http://www.nccn.org/professionals/physician_gls/pdf/squamous.pdf [updated October 24, 2014; cited 06.04.15] [Google Scholar]
- 8.Kenneth T., Steeves A., Richard J.S. Marcia: radiation therapy for squamous cell carcinoma in dystrophic epidermolysis bullosa: case reports and literature review. Am J Clin Oncol. 1997;20(1):55–58. doi: 10.1097/00000421-199702000-00012. [DOI] [PubMed] [Google Scholar]
- 9.Teoh M., Clark C.H., Wood K., Whitaker S., Nisbet: A. Volumetric modulated arc therapy: a review of current literature and clinical use in practice. Br J Radiol. 2011;84(1007):967–996. doi: 10.1259/bjr/22373346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nutting C.M., Morden J.P., Harrington K.J. Trial management group Parotid-sparing intensity modulated versus conventional radiotherapy in head and neck cancer (PARSPORT): a phase 3 multicentre randomised controlled trial. Lancet Oncol. 2011;12(10):127–136. doi: 10.1016/S1470-2045(10)70290-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu Q., Mohan R., Morris M., Lauve A., Schmidt-Ullrich R. Simultaneous integrated boost intensity-modulated radiotherapy for locally advanced head-and-neck squamous cell carcinomas, dosimetric results. Int J Radiat Oncol Biol Phys. 2003;56(105):573–583. doi: 10.1016/s0360-3016(02)04617-5. [DOI] [PubMed] [Google Scholar]
- 12.Cox J.D., Stetz J., Pajak T.F. Toxicity criteria of the Radiation Therapy Oncology Group (RTOG) and the European Organization for Research and Treatment of Cancer (EORTC) Int J Radiat Oncol Biol Phys. 1995;31(5):1341–1346. doi: 10.1016/0360-3016(95)00060-C. [DOI] [PubMed] [Google Scholar]
- 13.Fine J.D., Bauer E.A., Tuderman L.B. The Classification of inherited epidermolysis bullosa (EP): report of the Third International Consensus Meeting on Diagnosis and Classification of EP. J Am Acad Dermatol. 2008;58(6):931–946. doi: 10.1016/j.jaad.2008.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Vanetti E., Clivio A., Nicolini G. Volumetric modulated arc radiotherapy for carcinomas of the oro-pharynx, hypo-pharynx and larynx: a treatment planning comparison with fixed field IMRT. Radiother Oncol. 2009;92(1) doi: 10.1016/j.radonc.2008.12.008. [DOI] [PubMed] [Google Scholar]
- 15.Johnston M., Clifford S., Bromley R., Back M., Oliver L., Eade T. Volumetric-modulated arc therapy in head and neck radiotherapy: a planning comparison using simultaneous integrated boost for nasopharynx and oropharynx carcinoma. Clin Oncol (R Coll Radiol) 2011;23(8):503–511. doi: 10.1016/j.clon.2011.02.002. [DOI] [PubMed] [Google Scholar]
- 16.Guckenberger M., Richter A., Krieger T., Wilbert J., Baier K., Flentje M. Is a single arc sufficient in volumetric-modulated arc therapy (VMAT) for complex-shaped target volumes. Radiother Oncol. 2009;93(2):259–265. doi: 10.1016/j.radonc.2009.08.015. [DOI] [PubMed] [Google Scholar]
- 17.Bentzen S.M., Constine L.S., Deasy J.O. Quantitative analyses of normal tissue effects in the clinic (QUANTEC) Int J Radiat Oncol Biol Phys. 2010;76(Suppl. 3):S3–S9. doi: 10.1016/j.ijrobp.2009.09.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lomax N.J., Scheib G.S. Quantifying the degree of conformity in radiosurgery treatment planning. Int J Radiat Oncol Biol Phys. 2003;55(5):1409–1419. doi: 10.1016/s0360-3016(02)04599-6. [DOI] [PubMed] [Google Scholar]
- 19.Pesznyák C., Béla D., Takácsi N.Z., Major T., Polgár C. Dosimetry analysis of intensity-modulated and conformal radiation therapy for head and neck tumors. Magy Onkol. 2015;59(2):95–101. [PubMed] [Google Scholar]
- 20.ICRU-prescribing, recording, and reporting photon-bean intensity modulated radiation therapy (IMRT). The International Comission on Radiation Units and Measurements. ICRU Report 83. J ICRU. 2010;10(1):1473–6691. [Google Scholar]
- 21.Eisenhauer E.A., Therasse P., Bogaerts J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur J Can. 2009;(45):228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 22.Andrä C., Rauch J., Li M. Excellent local control and survival after postoperative or definitive radiation therapy for sarcomas of the head and neck. Radiat Oncol. 2015;10(10):1186–13014. doi: 10.1186/s13014-015-0449-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee N., Chuang C., Quivey J.M. Skin toxicity due to intensity-modulated radiotherapy for head-and-neck carcinoma. Int J Radiat Oncol Biol Phys. 2002;53(3):630–637. doi: 10.1016/s0360-3016(02)02756-6. [DOI] [PubMed] [Google Scholar]
- 24.Lotem M., Raben M., Zeltser R. Kindler syndrome complicated by squamous cell carcinoma of the hard palate: successful treatment with high-dose radiation therapy and granulocyte–macrophage colony-stimulating factor. 2001;144(6):1284–1286. doi: 10.1046/j.1365-2133.2001.04262.x. [DOI] [PubMed] [Google Scholar]


