Abstract
We report on a 20-year-old patient with a 6-month history of recurrent abdominal pain and a 3-day history of vomiting, hypertension, seizures, and encephalopathy. The brain MRI showed posterior reversible encephalopathy syndrome, and continuous EEG (cEEG) monitoring showed lateralized periodic discharges plus fast activity. Comprehensive CSF studies were negative. Because of severe abdominal pain without a definite etiology, we requested urine porphobilinogen and serum and fecal porphyrins, which suggested acute intermittent porphyria (AIP). The patient had a complete resolution of her symptoms with carbohydrate loading and high caloric diet. Acute intermittent porphyria is potentially life-threatening without proper management and prevention of triggers if it is not recognized.
Keywords: Porphyria, LPDs, PRES, Seizures
1. Introduction
Acute intermittent porphyria (AIP) is an autosomal dominant disorder, which results from the partial deficiency of porphobilinogen (PBG) deaminase, an enzyme of the heme biosynthesis pathway [1]. Symptoms of AIP include abdominal pain, nausea, vomiting, behavioral changes, seizures, tachycardia, and hypertension [1]. There are previous reports of AIP associated with posterior reversible encephalopathy syndrome (PRES), which is thought to result from peaks in hypertension [2].
We report a case of a patient with severe recurrent abdominal pain of unclear etiology, hypertension, tachycardia, and seizures, associated with PRES on brain MRI and lateralized periodic discharges plus fast activity (LPDs + F) [3] on continuous EEG monitoring (cEEG), who was diagnosed with AIP.
2. Case presentation
This is a 20-year-old female with a 6-month history of recurrent abdominal pain. She underwent cholecystectomy 3 months prior to admission but had no relief of her symptoms. She was admitted with acute pyelonephritis, nausea, vomiting, worsening of abdominal pain, and constipation for three days. Her vital signs on admission showed uncontrolled hypertension (BP = 148/118 mm Hg) and tachycardia (HR = 120 bpm). She was unable to eat during the last few days. On the second day of her admission, she had two generalized tonic–clonic seizures. She received lorazepam and levetiracetam, and her clinical seizures resolved; however, she continued to be lethargic, and a stat EEG was requested, that showed LPDs + F over the right temporoparietal region. The patient was transferred to the ICU for blood pressure control and was started on treatment with acyclovir, antihypertensive medications, and antibiotics. She became profoundly lethargic and assumed a fetal position in bed. On neurological examination, there were left lower extremity clonus and a left Babinski sign. Head CT was normal, cerebral spinal fluid (CSF) was unremarkable, and comprehensive CSF profiles were negative, including PCR for Herpes Simplex virus, Varicella Zoster virus, Lyme disease, and Cryptococcus. The brain MRI showed PRES (Fig. 1). Because the stat EEG showed LPDs + F, a highly epileptogenic pattern, cEEG was initiated to rule out the possibility of recurrent electrographic seizures or nonconvulsive status epilepticus (Fig. 2). There was one electrographic seizure arising from the right temporoparietal region lasting less than 1 min seen during the first 12 h of recording but none afterwards during the next two days of recording. Therefore, clinical and electrographic seizures completely subsided with treatment. Acyclovir was discontinued, and high dose steroids were started, considering the possibility of immunological and inflammatory etiologies. The patient showed clinical improvement, but her abdominal pain persisted, and the diagnosis of an acute porphyria was suspected. Urine, then serum and fecal porphyrins were requested. The results were as follows: urine PBG level was 177.4 μmol/L (reference: 0 to 0.88 μmol/L), serum porphyrin level was 58 nmol/L (reference: 0 to 15 nmol/L), fecal coproporphyrin was 18 nmol/g (reference: 0 to 45 nmol/g), and fecal protoporphyrin was 10 nmol/g (reference: 0–100 nmol/g). Taken together, the highly increased urine PBG levels, slightly increased plasma porphyrin levels, and normal fecal porphyrin levels, were suggestive of AIP. The patient's abdominal pain resolved in less than 24 h with carbohydrate (glucose) loading and highly caloric diet. Therefore, intravenous hemin was not necessary at that time. She was discharged home in stable conditions and offered to follow up at a porphyria center of her choice for further confirmatory genetic testing, management, and counseling. The patient was seen in our epilepsy outpatient clinic one month later. She was not taking any medications. She was asymptomatic, and her neurological exam was normal. A follow-up brain MRI showed remarkable improvement.
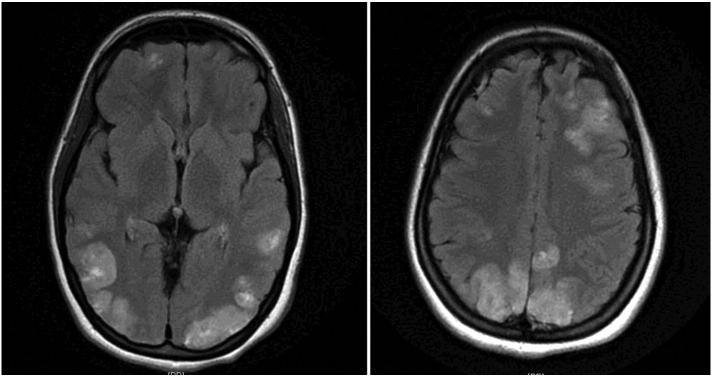
Fig. 1.
Brain MRI scan, axial T2 FLAIR, shows faint multifocal and bilateral hyperintensities in the frontal lobes and parietooccipital lobes, extending to the left greater than right posterior temporal regions.
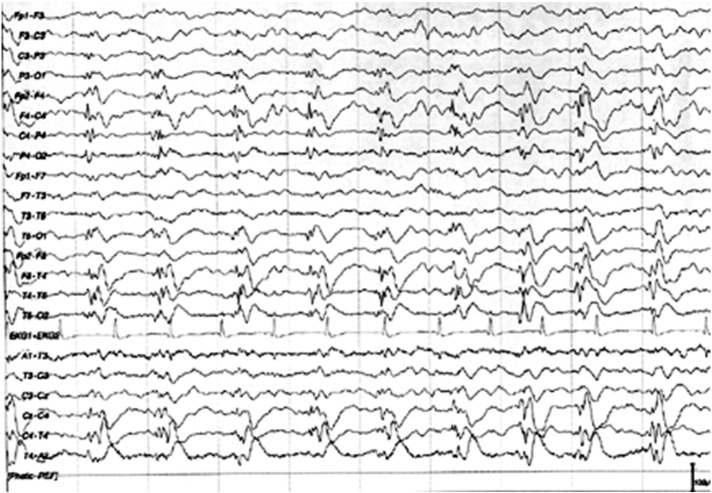
Fig. 2.
EEG, bipolar longitudinal montage, shows lateralized periodic discharges plus fast activity (LPDs + F) over the right temporoparietal–occipital region.
3. Discussion
The neurological manifestations of AIP are potentially life-threatening without proper diagnosis, management, and elimination of triggers. We report an unusual case of AIP presenting with PRES, seizures, and LPDs + F, which led to additional complexity in the differential diagnosis and management of this patient.
The clinical presentation of our patient fits the diagnosis of PRES, which usually leads to altered consciousness and seizures. Neuroimaging studies, including head CT and brain MRI, confirm the diagnosis of PRES and typically show bilateral symmetrical brain edema more prominent but not limited to the parietal and occipital regions. The cEEG monitoring showed LPDs + F, which are usually seen as a result of acute or subacute brain lesions, including stroke, infections, particularly Herpes Simplex encephalitis, and rapidly growing brain tumors, but can also be found in chronic epilepsy. More recently, LPDs have been described in patients with PRES [4]. Skiba et al. emphasized the importance of cEEG monitoring to diagnose LPDs and recurrent seizures in patients who present with PRES, once these patients may need treatment with antiseizure drugs during the acute period [4].
Our patient improved clinically with supportive care and treatment that target tight blood pressure control. The only remaining symptom that raised the diagnostic possibility of acute porphyria a few days after her admission was the residual abdominal pain of unknown etiology. Taken together, her symptoms of recurrent abdominal pain, nausea and vomiting, tachycardia, and hypertension, which were likely exacerbated by fasting and infection, leading to encephalopathy and seizures, were suggestive of an acute porphyria attack [1]. Exacerbation of acute porphyrias is usually secondary to multiple environmental and physiological factors, including drugs, infection, hormonal changes, or fasting [1].
When acute porphyria is suspected, screening for urinary PBG is tested first because these intermediates are increased in the urine of all three most common acute porphyrias, including AIP, hereditary coproporphyria (HCP), and variegate porphyria (VP), [1]. The patient's urinary PBG was highly elevated and suggestive of an acute porphyria. In an attempt to further differentiate the acute porphyrias, we requested serum and fecal porphyrin levels. Our findings of markedly increased urine PBG levels, slightly increased plasma porphyrin levels, and normal fecal porphyrin levels were suggestive of AIP. Acute intermittent porphyria is the commonest acute porphyria.
It is likely that PRES is a contributing factor to the occurrence of seizures in patients with AIP [5]. Seizures are not uncommon and occur in up to 10–20% of patients at any time during the attacks of AIP [6]. Of note is the fact that several antiseizure drugs, including phenytoin, carbamazepine, primidone, and valproic acid, are considered unsafe and may provoke or worsen an attack of porphyria [1]. Fortunately, our patient, who had no definite diagnosis at presentation, received levetiracetam in the emergent setting, which was previously used with success for the management of status epilepticus in a patient with AIP [7].
4. Conclusions
This case illustrates that the diagnosis of AIP may be challenging. Her initial symptoms including peaks of hypertension, encephalopathy, and seizures could be explained by PRES. The finding of LPDs + F over the temporoparietal region on cEEG was likely secondary to PRES. The initial question asked was what caused PRES in this young patient. It was the presence of persistent abdominal pain without a definite etiology that led to the diagnosis of an acute intermittent porphyria.
Conflict of interest
None of the authors have any conflict of interest to disclose with respect to this manuscript.
Contributor Information
Diosely C. Silveira, Email: dsilveira@jfkhealth.org.
Michelle H. Lucena, Email: michelle_lucena@hotmail.com.
References
- 1.Anderson K.E., Bloomer J.R., Bonkonsky J.P., Pierach C.A., Pimstone N.R., Desnick R.J. Recommendations for diagnosis and treatment of acute porphyrias. Ann Intern Med. 2005;142:439–450. doi: 10.7326/0003-4819-142-6-200503150-00010. [DOI] [PubMed] [Google Scholar]
- 2.Zhao B., Wei Q., Wang Y., Chen Y., Shang H. Posterior reversible encephalopathy syndrome in acute intermittent porphyria. Pediatr Neurol. 2014;51:457–460. doi: 10.1016/j.pediatrneurol.2014.05.016. [DOI] [PubMed] [Google Scholar]
- 3.Hirsch L.J., LaRoche S.M., Gaspard N., Gerard E., Svoronos A., Herman S.T. American Clinical Neurophysiology Society's standardized critical care EEG terminology: 2012 version. J Clin Neurophysiol. 2013;30:1–27. doi: 10.1097/WNP.0b013e3182784729. [DOI] [PubMed] [Google Scholar]
- 4.Skiba V., Etienne M., Miller J.A. Development of chronic epilepsy after recurrent episodes of posterior reversible encephalopathy syndrome associated with periodic lateralized epileptiform discharges. Seizure. 2011;20:93–95. doi: 10.1016/j.seizure.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 5.Roth C., Ferbert A. The posterior reversible encephalopathy syndrome: what is certain? What is new? Pract Neurol. 2011;11:136–144. doi: 10.1136/practneurol-2011-000010. [DOI] [PubMed] [Google Scholar]
- 6.Thadani H., Deacon A., Peters T. Diagnosis and management of porphyria. BMJ. 2000;320:1647–1651. doi: 10.1136/bmj.320.7250.1647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bhatia R., Vibha D., Srivastava M.V.P., Prasad K., Manjari T., Singh M.B. Epilepsia. 2008;49(5):934–936. doi: 10.1111/j.1528-1167.2007.01518_2.x. [DOI] [PubMed] [Google Scholar]