Abstract
Background
The health implications of weight gain after antiretroviral therapy (ART) for HIV infection are not well characterized and may differ from weight gain among uninfected individuals. We use data from the Veterans Aging Cohort Study (VACS) to determine whether weight gain after ART has a similar association with incident type-2 diabetes mellitus (DM) as weight gained among HIV-uninfected (uninfected) individuals.
Methods
We explored associations of weight gain and incident diabetes (A1c ≥ 6.5 %), in VACS, a national observational study of HIV infected (HIV+) individuals demographically matched 1:2 to uninfected controls. From 2000 to 2011, weight change was assessed in the year following ART initiation for HIV+ individuals and date of first available BMI for uninfected individuals. We estimated hazard ratios (HR) and 95% CI adjusted for baseline BMI using Cox regression.
Results
HIV+ individuals had lower prevalence of DM at baseline (12%, HIV+, 23% uninfected) and lower incident diabetes (5% HIV+, 11% uninfected). The association of weight gain with risk of DM was linear for HIV+ and uninfected but the slope of the association was steeper for HIV+. For each 5 pounds of weight gained, HIV+ had 14% increased risk of DM (HR, 1.14; 95% CI, 1.10–1.17) and uninfected individuals had 8% increased risk (HR: 1.08; 95% CI, 1.07–1.10) (p<0.01 for interaction).
Conclusion
Weight gained in the first year after ART initiation is associated with greater risk of DM than that among uninfected individuals. HIV+ individuals initiating ART who are not underweight should avoid substantial weight gain.
Keywords: Diabetes, HIV, weight gain, obesity, Veterans, Inflammation
After initiating antiretroviral therapy (ART), most HIV infected individuals (HIV+) gain weight, many becoming overweight or obese,1 and the clinical implications of this weight gain are not well understood. While weight gain of 10–20 pounds after ART initiation is associated with a survival advantage among normal weight individuals2, weight gain may exacerbate risk of comorbid disease. Among uninfected individuals, obesity dramatically increases risk of type 2 diabetes mellitus (DM), hypertension, hyperlipidemia and associated cardiovascular disease3 and obesity increases risk for hepatic cirrhosis and several forms of cancer4. Further, in uninfected individuals, obesity is associated with inflammation and hypercoagulability5. Concentrations of circulating IL-6, tumor necrosis factor (TNF)-α, and CRP are elevated in obesity and predict incidence of diabetes6. Given the increased risk for metabolic syndrome, cardiovascular disease, liver disease, cancer, and the putative harmful role of chronic inflammation among those with HIV7,8, minimizing weight gain after ART would seem advisable (except in those who are underweight)9.
However, weight gain after ART may not be the same as weight gain in middle aged individuals free of HIV infection. In earlier studies, higher BMI has been associated with slower HIV progression10 and mild to moderate obesity has been associated with better HIV survival compared to non-obese HIV+ individuals11. Overweight BMI (kg/m2) categories are associated with lower declines in CD4 count and greater gains in CD4 count compared to normal BMI1. In more recent studies, weight gain after ART is associated with HIV-1 RNA suppression and CD4 count response2,12,13.
Complicating the picture further, ART has been found to be associated with incident DM14. This finding may be explained by direct effects of medications, particularly the promotion of hyperglycemia via resistance to or decreased production of insulin, an effect primarily observed for protease inhibitors15. Thus, HIV infected individuals on ART may be at higher risk of developing DM as they age.
A characterization of weight gain and DM incidence among HIV+ initiating ART compared to that observed among uninfected individuals may offer important insight regarding competing effects of HIV-1 RNA suppression, weight gain, and ART toxicity. Using data from the Veterans Aging Cohort Study (VACS), which includes a large sample of HIV+ and demographically and behaviorally similar uninfected individuals, we explore this association.
Methods
Study Population
The Veterans Aging Cohort study (VACS) has been described previously16. Briefly, VACS is an ongoing observational cohort of HIV+ Veterans, matched 1:2 with uninfected Veterans by age-, sex-, race-, and site-of-care, who received medical care at a Department of Veterans Affairs (VA) healthcare facility. Patients were identified from VA national electronic health records (EHR). Clinical information includes International Classification of Diseases, Ninth Edition diagnosis codes (ICD-9 codes) from inpatient and outpatient visits, laboratory results and all medications dispensed from VA pharmacies. Height and weight from clinic visits as of 2000 is available. VACS was approved by the Human Investigation Committee at the Yale School of Medicine and the VA Connecticut Human Subjects Subcommittee.
Eligible patients entered VACS between January 1, 1999 and September 30, 2011. Patients who died in the year after cohort entry or had no VA visit were excluded. We defined baseline as date of ART initiation for HIV+ and date of first available BMI for uninfected. To ensure that all patients had a year of history before starting follow-up, the earliest baseline date was January 1, 2000. Eligible HIV+ were ART naïve, defined as no record of any prior antiretroviral (ARV) prescription, plus HIV-1 RNA >500 copies/ mL in the 180 days prior to ART initiation. ART was defined as the combination of at least three antiretroviral drugs. Patients were required to have a BMI value (mass in kg/height2 in meters) recorded at baseline and at least one value recorded in the subsequent 18 months. HIV+ usually have 3–4 clinic visits per year and uninfected have clinic visits as warranted by their individual conditions. Patients with prevalent diabetes at baseline were excluded. We used a previously validated algorithm (4) based on diagnosis of diabetes, blood glucose levels and prescription for oral anti-hyperglycemic agent or insulin; or hemoglobin A1c (A1c) ≥ 6.5 % to identify existing diabetes. This broad definition was used to maximize sensitivity to identify those who already had diabetes. Eligible patients were required to have a recorded blood glucose level in order to restrict analyses to subjects with VA laboratory data. One pregnant woman was excluded.
Weight change and BMI
The exposure of interest was weight change over a one-year period. Weight was obtained at baseline and one year later. For HIV+, we used weight closest to but no more than 90 days prior to ART initiation, and first available BMI for uninfected. We used weight closest to the target date of one year if a value was available within ±30 days. Otherwise we used simple linear interpolation from two weights within ±180 days of the one-year date, to estimate the one-year weight. We considered weight change as a gain or loss of more than 5 pounds, believing ±5 pounds to be an appropriate threshold for exceeding random variation in weight. Patients were categorized by baseline BMI as underweight (<18.5), normal weight 18.5 to <25), overweight (25 to <30), and obese (≥30)17.
Outcome
The primary outcome was incident diabetes. Patients were followed up to 5 years until last VA visit on or before September 30, 2012, diagnosis of diabetes, or death, whichever occurred first. We defined incident diabetes as A1c ≥ 6.5 % in accordance with 2010 American Diabetes Association (ADA) guidelines18. This very specific definition unifies severity of disease and date of onset. Accuracy is better than other criteria because A1c is not impacted by fasting status, has less day-to-day variability than blood glucose measurements and only slightly attenuates the association between HIV and diabetes19.
Covariates
Patient characteristics and potential confounders included demographics (age, race, sex), smoking status (current, former, never)20, hepatitis C infection (HCV) and calendar year at baseline. Race/ethnicity was categorized as white, black, Hispanic, or other/unknown. Demographic data, pharmacy records and ICD-9 codes (from VA outpatient and inpatient visits) were from data any time prior to baseline. Baseline CD4+ cell count/mm3, HIV-1 RNA copies/mL were those closest to but within 180 days before the index date.
Statistical Analyses
We summarized and compared characteristics of HIV+ and uninfected, using Chi-square or Wilcoxon rank-sum as appropriate. We summarized weight change in the first year by HIV status, overall and stratified by baseline BMI. We then compared rates of incident diabetes associated with weight change (lost >5, ±5 pounds, >5 to 10, >10 to 15, >15 to 20, >20 to 30, ≥30 pounds), by HIV status, stratified by baseline BMI. To account for potential confounding we analyzed risk of incident DM as a function of weight change using multivariable Cox models. We first used seven weight change categories (as above) to check for linearity and thresholds, stratified by HIV status. We then restricted our multivariable regression analysis to those with baseline BMI >18.5 because we found a very low rate of incident diabetes (21 events) and because weight gain is beneficial in underweight HIV+.21–23 Formal testing for interactions (likelihood ratio test) showed significant interaction between HIV status and weight gain for risk of incident DM. To allow for directly interpretable hazard ratios (HR) from a single model, we created two composite variables, one for weight change in uninfected and one for weight change in HIV+, both scaled per 5-pound change. All analyses were conducted with SAS, version 9.4; p values <0.05 were considered significant.
We also conducted several sensitivity analyses. We adjusted for number of blood glucose levels recorded, excluded patients with cancer diagnosis up to 12 months after baseline, HIV+ who did not achieve viral suppression (<500 copies/mL), and those with HCV. We also restricted analysis to those with any A1c before baseline, and those with any A1c during follow-up. In further analyses restricted to HIV+, we adjusted for protease inhibitor (PI) use, defined as receipt of at least one PI during the first year of ART because some PIs have been found to be associated with increased risk of metabolic complications including insulin resistance and incident diabetes15,24–26. In a separate model we adjusted for initial regimens containing zidovudine or stavudine, as these two drugs have been associated with development of diabetes14,27.
Results
Among 23,175 HIV+ entering VACS after January 1, 1999 and under follow-up for at least one year, we excluded 2,226 who initiated ART before January 1, 2000 and 5,310 who had not initiated ART by September 30, 2011. We also excluded those without documented HIV-RNA >500 copies/ml who were assumed to have initiated ART before entering VA care and were excluded, leaving 9,423 potential patients (Figure 1). Initial and subsequent BMI was available for 8,325 (89%). Among 47,340 uninfected comparators entering the cohort after January 1, 1999 and under follow-up a year after entry, 45,429 had baseline BMI, and subsequent BMI was available for 32,706 (72%).
Figure 1.
Patient flow diagram.
The prevalence of DM at baseline was 960/8325 (11.5%) among HIV+ and 7490/32706 (22.9%) among uninfected. Within strata by HIV status, DM was more common in those with HCV infection. Prevalence of DM increased with increasing BMI across all subgroups by HIV and HCV status. Patients with existing DM were excluded from subsequent analyses.
Our final sample, after excluding 751 patients without blood glucose during follow-up, had 7,177 HIV+ and 24,621 uninfected at risk of DM. The 31,798 Veterans were primarily male, and approximately half were black (Table 1). Compared to uninfected patients, HIV+ were older, less likely to be overweight or obese, and more likely to be current smokers or have HCV infection. Median baseline year was 2006 for HIV+ and 2004 for uninfected individuals, and median length of follow-up was 4.9 and 5.0 years, respectively. About half of patients ended follow-up at 5 years, and 1/3 at last VA visit. HIV+ patients were less likely to develop DM and more likely to die than uninfected patients. Among HIV+, 60% initiated ART with a non-nucleoside reverse transcriptase inhibitor (NNRTI) based regimen, 35% with a PI based regimen, and 5% with a triple nucleoside reverse transcriptase inhibitor regimen (Table 1). Forty percent of regimens included zidovudine or stavudine.
Table 1.
Baseline characteristics of 31,798 Veterans at risk of incident diabetes, 2000–2011.
| HIV + | HIV − | |||||
|---|---|---|---|---|---|---|
| N = 7177 | N = 24621 | p | ||||
| Age, years, Median (IQR) | 50 | (43,56) | 48 | (41,54) | <0.001 | |
| Male, N (%) | 6944 | (96.8) | 23813 | (96.7) | 0.88 | |
| Race, N (%) | White | 2592 | (36.1) | 10782 | (43.8) | <0.001 |
| Black | 3764 | (52.4) | 10692 | (43.4) | ||
| Hispanic | 453 | (6.3) | 1544 | (6.3) | ||
| Other/unknown | 368 | (5.1) | 1603 | (6.5) | ||
| BMI, kg/m2 | Underweight <18.5 | 332 | (4.6) | 276 | (1.1) | <0.001 |
| N(%) | Normal 18.5 to <25 | 3557 | (49.6) | 5378 | (21.8) | |
| Overweight 25 to <30 | 2322 | (32.4) | 9464 | (38.4) | ||
| Obese >= 30 | 966 | (13.5) | 9503 | (38.6) | ||
| Median (IQR) | 25 | (22,28) | 28 | (25,32) | <0.001 | |
| Smoking, N (%) | Never | 2006 | (28.0) | 7534 | (30.6) | <0.001 |
| Current | 4149 | (57.8) | 11854 | (48.1) | ||
| Former | 843 | (11.7) | 4456 | (18.1) | ||
| Unknown | 179 | (2.5) | 777 | (3.2) | ||
| Hepatitis C Infection, N (%) | 2303 | (32.1) | 3513 | (14.3) | <0.001 | |
| Start of follow-up, year |
2000–2002 | 1660 | (23.1) | 8336 | (33.9) | <0.001 |
| 2003–2005 | 1805 | (25.1) | 6610 | (26.8) | ||
| 2006–2008 | 1778 | (24.8) | 5166 | (21.0) | ||
| 2009–2011 | 1934 | (26.9) | 4509 | (18.3) | ||
| Median (IQR) | 2006 | ('03,'09) | 2004 | ('02,'08) | <0.001 | |
| Followed until | 5 years | 3533 | (49.2) | 13731 | (55.8) | |
| Last VA visit | 2763 | (38.5) | 7291 | (29.6) | <0.001 | |
| Incident diabetes | 357 | (5.0) | 2656 | (10.8) | ||
| Death | 524 | (7.3) | 943 | (3.8) | ||
| Total years, Median (IQR) | 4.9 | (2.6,5.0) | 5.0 | (3.0,5.0) | <0.001 | |
| CD4, cells/mm3, Median (IQR) | 218 | (85, 338) | NA | |||
| HIV-1 RNA, log copies/mL, Median (IQR) | 4.8 | (4.3, 5.3) | NA | |||
| ART regimen | NNRTI | 4321 | (60.2) | |||
| PI | 2503 | (34.9) | ||||
| Triple nucleoside | 353 | (4.9) | ||||
| AZT or D4T | 2886 | (40.2) | ||||
ART = Antiretroviral regimen, NNRTI = Non-nucleoside reverse transcriptase inhibitor, PI = Protease inhibitor
Weight change in one year
Weight gain exceeding 5 pounds was common (Table 2). More HIV+ gained weight (48%) than uninfected (31%) and median weight gain was greater in HIV+, 4 pounds (IQR −4,15) compared to 1 pound (IQR: −5,10) in uninfected (p<0.001). [Among HIV+ not on ART, not included in our primary analysis, median weight gain over 12 months was 0.7 (IQR −6.4, 7.8) pounds.] Among HIV+, lower baseline BMI was associated with higher probability of weight gain. Two thirds of those with baseline BMI <18.5 (underweight) gained weight in the 12 months after ART initiation, as did half of the normal weight and more than 40% of those who were overweight or obese at baseline. At every level of BMI, weight gain was more common and the amount of weight gained was greater in HIV+ compared to uninfected. Among uninfected patients one third gained weight across all levels of baseline BMI. Weight loss was more frequent in uninfected (24%) than HIV+ (21%; p<0.001). Among patients who lost weight, 60% of HIV+ and 82% of uninfected were overweight or obese at baseline.
Table 2.
Weight change after 1 year by baseline BMI.
| HIV + | HIV − | ||||
|---|---|---|---|---|---|
| Baseline BMI | Weight change | N | (%) | N | (%) |
| Overall | N = 7177 | N = 24621 | |||
| Lost >5 lbs | 1495 | (20.8) | 5824 | (23.7) | |
| Remained within +/− 5 lbs | 2249 | (31.3) | 11062 | (44.9) | |
| Gained >5 lbs | 3433 | (47.8) | 7735 | (31.4) | |
| Median (IQR) | 4.3 | (−3.5,14.9) | 1.0 | (−4.9,7) | |
| Underweight <18.5 | N = 332 | N = 276 | |||
| Lost >5 lbs | 25 | (7.5) | 31 | (11.2) | |
| Remained within +/− 5 lbs | 83 | (25.0) | 154 | (55.8) | |
| Gained >5 lbs | 224 | (67.5) | 91 | (33.0) | |
| Median (IQR) | 13 | (2.8,26.3) | 2.0 | (−2.9,7.6) | |
| Normal 18.5 to <25 | N = 3557 | N = 5378 | |||
| Lost >5 lbs | 575 | (16.2) | 1024 | (19.0) | |
| Remained within +/− 5 lbs | 1160 | (32.6) | 2576 | (47.9) | |
| Gained >5 lbs | 1822 | (51.2) | 1778 | (33.1) | |
| Median (IQR) | 5.6 | (−1.7,16.1) | 1.7 | (−3.5,7.3) | |
| Overweight 25 to <30 | N = 2322 | N = 9464 | |||
| Lost >5 lbs | 599 | (25.8) | 2017 | (21.3) | |
| Remained within +/− 5 lbs | 731 | (31.5) | 4554 | (48.1) | |
| Gained >5 lbs | 992 | (42.7) | 2893 | (30.6) | |
| Median (IQR) | 3.0 | (−5.4,12) | 1.0 | (−4.1,7) | |
| Obese >= 30 | N = 966 | N = 9503 | |||
| Lost >5 lbs | 296 | (30.6) | 2752 | (29.0) | |
| Remained within +/− 5 lbs | 275 | (28.5) | 3778 | (39.8) | |
| Gained >5 lbs | 395 | (40.9) | 2973 | (31.3) | |
| Median (IQR) | 1.7 | (−7,11.2) | 1.0 | (−4.1,7) | |
Rates of Incident Diabetes
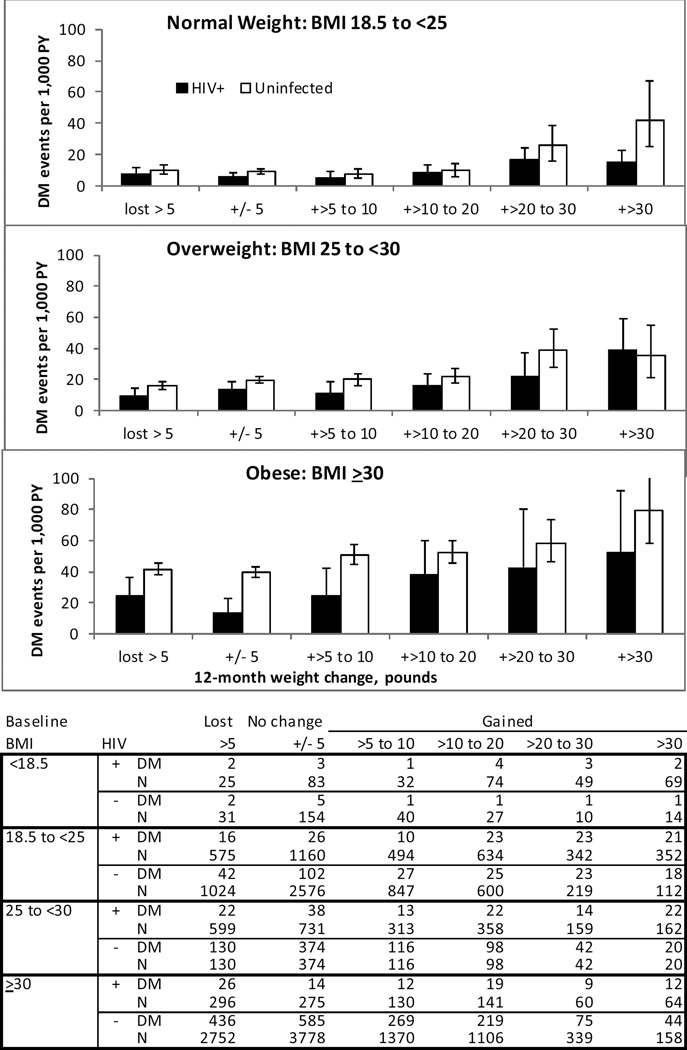
During follow-up, 24% of patients developed incident DM; 357 HIV+ in 17,780 person-years (PY), (13 /1000 PY) and 2,656 uninfected in 126,761 PY, (27/1000 PY) (p<0.001). In unadjusted analyses, rates of incident DM were higher with increasing baseline BMI and with increasing weight gain (Figure 2). Among those with baseline BMI < 18.5 incidence of DM per 1000 PY was 12 and 10 for HIV+ and uninfected respectively, 8 and 11 for BMI 18.5 to <25, 15 and 20 for BMI 25 to <30 and 26 and 44 for BMI 30 and higher.
Figure 2.
Rate of incident diabetes mellitus during median follow-up of 5.0 years, by baseline BMI, according to amount of weight gained in 12 months, in HIV+ and Uninfected Veterans, 2000–2011. (BMI <18.5 not shown due to sparse data).
There were only 21 incident DM events among those underweight at baseline. HIV+ had lower rates of diabetes than uninfected across all weight change strata, except those with baseline BMI 25 to <30, who gained more than 30 pounds in one year. An association between incident diabetes and weight gain was evident in both HIV+ and uninfected across all levels of baseline BMI. An exception to this pattern was seen in those with baseline BMI ≥30 who lost weight; incidence rates were higher than in those without weight change.
Independent Risk Factors for Incident Diabetes
In multivariable analysis restricted to those with baseline BMI >18.5, stratified by HIV status, risk of DM increased significantly with weight gain of 10 pounds or more (Figure 3), for both HIV+ and uninfected. Models were adjusted for age, race, sex, baseline BMI, smoking, calendar year at baseline and HCV infection. The association appeared to be linear and stronger in HIV+. Based on these results, we used weight gain as a continuous variable, restricted to those with baseline BMI ≥18, and those without weight loss. We found significantly increased risk of DM with increasing age, black or Hispanic race/ethnicity, and being overweight or obese at baseline. Smoking history and later calendar year were associated with increased risk (Table 3). HCV infection was associated with increased risk in HIV+. These findings persisted when adding a continuous BMI term to account for subtle differences by HIV status or stratifying by baseline BMI (Supplemental Table 2).
Figure 3.
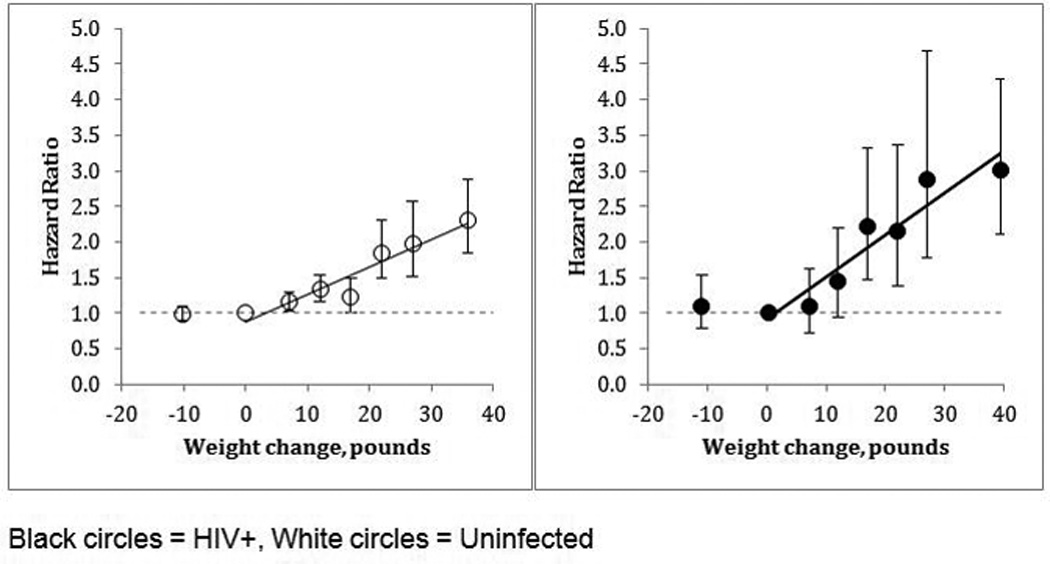
Relative risk of diabetes in HIV+(N = 6,845) and uninfected (N = 24,345) Veterans, by weight change in a one-year interval compared to those without weight change, adjusted for age, race, sex, baseline BMI, smoking, HCV infection and calendar year at baseline. (Excludes those with baseline BMI <18.5 kg/m2)
Table 3.
Multivariable Cox model for risk of incident diabetes mellitus in 5,375 HIV+ veterans and 18,552 uninfected veterans, based on 2,315 events (278 HIV+, 2037 uninfected) during a median of 5.0 years of follow-up. (Excludes those with baseline BMI <18.5 and those who lost weight during follow-up).
| HIV+ | Uninfected | Overall | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Predictor variable | HR | (95% CI) | p value | HR | (95% CI) | p value | HR | (95% CI) | p value | |
| Age, per 5 years | 1.16 | (1.09, 1.23) | <0.001 | 1.16 | (1.13, 1.19) | <0.001 | 1.16 | (1.14, 1.18) | <0.001 | |
| Race | White | Ref | Ref | Ref | ||||||
| Black | 1.69 | (1.30, 2.20) | <0.001 | 1.56 | (1.42, 1.72) | <0.001 | 1.57 | (1.44, 1.72) | <0.001 | |
| Hispanic | 1.67 | (1.03, 2.71) | 0.04 | 1.86 | (1.58, 2.19) | <0.001 | 1.84 | (1.57, 2.14) | <0.001 | |
| Baseline BMI, 2 |
Normal, 18.5 to 24.9 | Ref | Ref | Ref | ||||||
| kg/m | Overweight, 25to 29.9 | 2.11 | (1.61, 2.77) | <0.001 | 2.08 | (1.77, 2.45) | <0.001 | 2.05 | (1.79, 2.35) | <0.001 |
| Obese, >30 | 3.40 | (2.48, 4.65) | <0.001 | 4.57 | (3.92, 5.32) | <0.001 | 4.39 | (3.85, 5.01) | <0.001 | |
| Smoking | Ever vs never | 0.99 | (0.76, 1.28) | 0.930 | 1.14 | (1.04, 1.26) | 0.006 | 1.12 | (1.03, 1.23) | 0.011 |
| Calendar year | 1.02 | (0.98, 1.06) | 0.33 | 1.03 | (1.02, 1.05) | <0.001 | 1.03 | (1.02, 1.04) | <0.001 | |
| HCV infected vs not | 1.33 | (1.04, 1.71) | 0.02 | 1.04 | (0.91, 1.18) | 0.61 | 1.09 | (0.97, 1.22) | 0.13 | |
| HIV infected vs not | 0.56 | (0.47, 0.66) | <0.001 | |||||||
| Weight gain, per | HIV + | 1.13 | (1.10, 1.17) | <0.001 | 1.14 | (1.10, 1.17) | <0.001 | |||
| 5pounds | Uninfected | 1.08 | (1.07, 1.10) | <0.001 | 1.08 | (1.07, 1.10) | <0.001 | |||
Interaction of weight change and HIV p<0.01
In a final combined model, formal tests of interaction were significant for HIV and weight gain, but not for HIV and smoking, calendar year or HCV infection. After adjusting for age, race, baseline BMI, smoking history, and calendar year at baseline (Table 3); HCV infection was associated with higher risk of incident DM compared to uninfected individuals (HR = 1.09, 95% CI 1.00, 1.18) and HIV infection was associated with lower risk (HR = 0.56, 95% CI 0.47, 0.66) However, risk of DM associated with weight gain was greater among HIV+ individuals. Each 5-pound increment was associated with 14% increased risk of incident DM in HIV+ and 8% increased risk in uninfected (HR = 1.14 (95% CI: 1.10, 1.17) vs. 1.08 (95% CI: 1.07, 1.10), p for interaction <0.01).
Sensitivity Analyses
We performed several sensitivity analyses (data not otherwise shown). The associations with weight gain and incident DM were similar when we adjusted for number of blood glucose levels recorded; excluded those with cancer, those with HIV+ not achieving viral suppression, and those with HCV. When we restricted to those with A1c measured before baseline, the number of events declined to 390, but the association with weight gain remained significant and differential. There was some suggestion of increased DM risk with PI use (HR = 1.14 (95% CI: 0.90, 1.45) and zidovidine or stavudine containing regimens (HR = 1.30 (95% CI: 0.92, 1.82) but these did not reach statistical significance.
Discussion
In VACS, HIV+ had lower prevalence and incidence of DM compared with uninfected individuals. Incidence among HIV+ remained lower compared with uninfected after adjustment for established risk factors including age and BMI. However, incident DM increased substantially with weight gain of 10 or more pounds in both groups and this increased risk was greater for HIV+ compared to uninfected. For every 5 pounds of weight gained, HIV+ showed greater risk of DM compared to demographically similar uninfected individuals.
Other investigators have reported the association of HIV status and/or baseline BMI with incidence of DM28–33. Prior work from VACS has documented that HIV+ individuals are at increased risk of weight gain in the first year following initiation of ART compared to demographically matched uninfected comparators and that weight gain after ART confers no survival advantage among overweight or obese individuals2. The current study extends these observations to show that weight gain after ART is associated with excess risk of DM compared with weight gain among uninfected individuals34.
Weight gain after ART initiation is likely a double-edged sword with respect to inflammation. On one hand, successful suppression of HIV-1 RNA with ART is logically accompanied by a rapid decrease in the metabolic demand associated with HIV infection35. If established eating habits remain unchanged, decreased metabolic demand inevitably results in increased weight. Thus, all else equal, weight gain after ART may indicate a strong response to treatment and a decreased inflammatory burden. Evidence supporting this assumption includes the observation that weight gain after ART is highly correlated with HIV-1 RNA suppression and CD4 count recovery, independent of weight status at ART initiation2,13. Consistent with this hypothesis, median weight gain over 12 months among HIV+ not on ART was much less (0.7 IQR −6.4, 7.8 pounds--data not otherwise shown). On the other hand, among middle-aged and older uninfected individuals, weight gain contributes to inflammatory burden and risk of incident DM36,37. Because chronic inflammation may be more pronounced among HIV+ than uninfected individuals even after ART38, excess risk of diabetes associated with weight gain among HIV+ supports the contention that DM is a disease of inflammation36,37.
An alternative explanation for our findings is the possibility that ART, rather than HIV itself, interacts with risk of incident DM associated with weight gain. No consistent picture has emerged for a drug class effect7,14,39–41. Potential drug interactions, lengths of exposure, and likely weight change confound the ability to isolate individual drug effects. Our study, which was focused on comparing risk of diabetes by HIV status, is not well suited to characterizing treatment effects. Future work is needed to explore this question.
Our findings concerning DM prevalence and incidence among HIV+ are consistent with prior studies. Incidence rates of diabetes among HIV+ in our study, 13 per 1000 PY in HIV+, are higher than those reported by D:A:D (2.9/1000 PY)28 but similar to rates reported by the French Hospital Database on HIV (11.7/1000 PY)33. The Multicenter AIDS Cohort Study reported even higher rates (47/1000 PY)40. These differences are likely explained by differences in age and the proportion of overweight and obese individuals in the sample. VACS had a median age at the time of this analysis of 50 years compared to a mean age of 36 years in D:A:D. Our HIV+ patients were more likely to overweight or obese (45.9%) versus D:A:D (29.2%).Median baseline BMI was 24 (IQR 21–26) in VACS, slightly lower than MACS (26, IQR 24–28)39. Prior studies have also found that HIV+ status is associated with reduced risk of DM after adjustment for established risk factors7,4241,43.
Our analysis has important strengths. We included uninfected, demographically matched and behaviorally similar comparators, without which we could not have determined how DM risk differs by HIV status. Additionally, risk of DM is strongly associated with advanced age. The age of our cohort more accurately estimated risk of DM in the modern era in which half those living with HIV in the US are 50 years of age or older. Finally, after excluding prevalent cases, we used a standard biomarker for incident DM thereby eliminating possible bias introduced if general medical providers’ assignment of ICD-9 diagnostic codes differed from infectious disease providers.
Our study has limitations. Most importantly, our focus was on comparing effects of weight gain after ART among HIV infected individuals to effects of weight gain more generally among uninfected individuals. As a result, our analyses is not well suited to determining the role of particular ART regimens in incident DM and cannot differentiate whether the differences in risk of DM associated with weight gain is due to HIV itself or to ART. It is not a simple matter to tease effects of chronic HIV from those of chronic ART exposure. Those initiating ART with a protease inhibitor based regimen differ in important ways from those who received an NNRTI based regimen. In our study they were more likely to be HCV co infected (33% vs. 28%), to have initiated ART in earlier years, and to be exposed to zidovudine, stavudine, or didanosine (data not otherwise shown). A careful analysis of the role of ART regimen in risk of diabetes incidence would require time updated data on ART exposure and time updated adjustment to account for confounding. Such an analysis is beyond the scope of this paper.
Other limitations include that our cohort is primarily male and gender differences may be important to consider when evaluating risk of DM associated with weight change1,39,44. We also focused on weight change during the first 12 months of ART because that is the period in which the maximum immunologic and virologic response to ART occurs45. It would be interesting to see whether risk for DM remains differential by HIV status for weight gained after this 12 month window. Additionally, we did not have information on diet and exercise for this sample. A subset of our cohort was surveyed about exercise habits and, in this restricted sample, HIV+ were equally likely to exercise as uninfected individuals. While we have no data on diet, we have no reason to think that HIV+ have a worse diet than uninfected veterans. Finally, patients receiving more frequent healthcare screening may be more likely to be diagnosed with DM. HIV+ individuals received fewer A1c tests than uninfected in VACS and in sensitivity analyses, adjusted for number of glucose measurements, results were similar to our main analysis.
Whether due to increased levels of inflammation, adverse effects of antiretrovirals, or some as yet unappreciated cause, for those aging with HIV, weight gain is associated with an even greater risk of DM than among uninfected individuals. Because it is easier to avoid weight gain than to lose weight once gained, ART initiation provides an important opportunity for clinicians to counsel patients. In conjunction with prior work, this study supports recommendations for HIV+ patients initiating ART; namely, 1) patients initiating ART typically gain weight in the first year beyond that observed among uninfected individuals, 2) this weight gain is associated with excess risk of diabetes compared with uninfected individuals, 3) while gaining 10–20 pounds is associated with a survival advantage among those not overweight, there is no advantage to weight gain among overweight or obese individuals at ART initiation. To minimize DM risk after ART initiation, normal weight patients should avoid gaining more than 10 pounds; overweight or obese patients should avoid weight gain altogether.
Supplementary Material
Acknowledgments
The authors thank the US Department of Veterans Affairs and the VACS staff and participants for their extraordinary cooperation and involvement.
Funding: This study was supported by National Institutes of Health: AHRQ (R01-HS018372), National Institute on Alcohol Abuse and Alcoholism (U24-AA020794, U01-AA020790, U01-AA020795, U01-AA020799, U24-AA022001, U24 AA022007, U10 AA013566-completed), National Heart and Lung Blood Institute (R01-HL095136; R01-HL090342) National Institute of Allergy and Infectious Disease (U01-A1069918),, National Institute on Drug Abuse (R01DA035616), and the Veterans Health Administration Office of Research and Development (VA REA 08-266, VA IRR Merit Award; VISN1 CDA (V1CDA2012-20)).
Footnotes
Conflict of Interest: No potential conflicts of interest relevant to this article were reported.
Author Contributions: A.J., J.T. and M.H. jointly conceived the study, supervised the statistical analyses, and wrote the manuscript. J.T. conducted statistical analyses and reviewed and edited the manuscript. K.A., A.B., K.C., M.F., C.G., D.L., D.R., M.R., C.R., and K.H. contributed to study design, data interpretation, and discussion and reviewed and edited the manuscript. A.J, is the guarantor of this work and, as such, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Meetings at which parts of this data was presented:
1) Conference on Retroviruses and Opportunistic Infections, Atlanta, GA, March 4, 2013
Disclaimer: The views expressed in this manuscript represent those of the authors and do not necessarily represent those of the Department of Veterans Affairs.
Contributor Information
Melissa Herrin, VA Connecticut Healthcare System, West Haven, and Yale University School of Medicine, New Haven, Connecticut
Janet P. Tate, VA Connecticut Healthcare System, West Haven, and Yale University School of Medicine and Public Health, New Haven, Connecticut
Kathleen M. Akgün, Department of Internal Medicine and General Internal Medicine, VA Connecticut Healthcare System, West Haven, CT, USA; and Department of Internal Medicine, Yale University School of Medicine, New Haven, CT, USA
Adeel A. Butt, University of Pittsburgh School of Medicine, VA Pittsburgh Healthcare System, Hamad Medical Corporation, Doha, Qatar
Kristina Crothers, University of Washington School of Medicine, Seattle, WA
Matthew S. Freiberg, Vanderbilt University School of Medicine and VA Pittsburgh Healthcare System
Cynthia L. Gibert, School of Medicine and Health Sciences, The George Washington University School of Medicine and Health Sciences; Medical Service/Infectious Diseases, Veterans Affairs Medical Center
David A. Leaf, VA Greater Los Angeles Healthcare System, and David Geffen School of Medicine at UCLA Los Angeles, California
David Rimland, VA Medical Center and Emory University School of Medicine, Atlanta, Georgia
Maria C. Rodriguez-Barradas, Infectious Diseases Section, Michael E. DeBakey Veterans Affairs (VA) Medical Center and Department of Medicine, Baylor College of Medicine, Houston, Texas
Chris B. Ruser, VA Connecticut Healthcare System, West Haven, and Yale University School of Medicine
Kevan C. Herold, Yale Diabetes Center, Yale University School of Medicine
Amy C. Justice, VA Connecticut Healthcare System, West Haven, and Yale University School of Medicine and Public Health, New Haven, Connecticut
REFERENCES
- 1.Crum-Cianflone N, Roediger MP, Eberly L, et al. Increasing Rates of Obesity among HIV-Infected Persons during the HIV Epidemic. Plos One. 2010;5(4) doi: 10.1371/journal.pone.0010106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yuh B, Tate J, Butt AA, et al. Weight change after antiretroviral therapy and mortality. Clin. Infect. Dis. 2015;60(12):1852–1859. doi: 10.1093/cid/civ192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zalesin KC, Franklin BA, Miller WM, Peterson ED, McCullough PA. Impact of obesity on cardiovascular disease. The Medical clinics of North America. 2011;95(5):919–937. doi: 10.1016/j.mcna.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 4.Ramos-Nino ME. The role of chronic inflammation in obesity-associated cancers. ISRN oncology. 2013;2013:697521. doi: 10.1155/2013/697521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Deeks SG, Tracy R, Douek DC. Systemic effects of inflammation on health during chronic HIV infection. Immunity. 2013;39(4):633–645. doi: 10.1016/j.immuni.2013.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11(2):98–107. doi: 10.1038/nri2925. [DOI] [PubMed] [Google Scholar]
- 7.Butt AA, McGinnis K, Rodriguez-Barradas MC, et al. HIV infection and the risk of diabetes mellitus. Aids. 2009;23(10):1227–1234. doi: 10.1097/QAD.0b013e32832bd7af. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Phillips AN, Neaton J, Lundgren JD. The role of HIV in serious diseases other than AIDS. Aids. 2008;22(18):2409–2418. doi: 10.1097/QAD.0b013e3283174636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim DJ, Westfall AO, Chamot E, et al. Multimorbidity Patterns in HIV-Infected Patients: The Role of Obesity in Chronic Disease Clustering. J Acq Immun Def Synd. 2012;61(5):600–605. doi: 10.1097/QAI.0b013e31827303d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Palenicek JP, Graham NMH, He YD, et al. Weight-Loss Prior to Clinical Aids as a Predictor of Survival. Journal of Acquired Immune Deficiency Syndromes and Human Retrovirology. 1995;10(3):366–373. [PubMed] [Google Scholar]
- 11.Shor-Posner G, Campa A, Zhang GY, et al. When obesity is desirable: A longitudinal study of the Miami HIV-1-infected drug abusers (MIDAS) cohort. J Acq Immun Def Synd. 2000;23(1):81–88. doi: 10.1097/00126334-200001010-00011. [DOI] [PubMed] [Google Scholar]
- 12.Lakey W, Yang LY, Yancy W, Chow SC, Hicks C. Short Communication From Wasting to Obesity: Initial Antiretroviral Therapy and Weight Gain in HIV-Infected Persons. AIDS Res Hum Retroviruses. 2013;29(3):435–440. doi: 10.1089/aid.2012.0234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shikuma CM, Zackin R, Sattler F, et al. Changes in weight and lean body mass during highly active antiretroviral therapy. Clin Infect Dis. 2004;39(8):1223–1230. doi: 10.1086/424665. [DOI] [PubMed] [Google Scholar]
- 14.De Wit S, Sabin CA, Weber R, et al. Incidence and risk factors for new-onset diabetes, in HIV-infected patients - The data collection on adverse events of Anti-HIV drugs (D : A : D) study. Diabetes Care. 2008;31(6):1224–1229. doi: 10.2337/dc07-2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Samaras K. The Burden of Diabetes and Hyperlipidemia in Treated HIV Infection and Approaches for Cardiometabolic Care. Curr HIV/AIDS Rep. 2012;9(3):206–217. doi: 10.1007/s11904-012-0124-x. [DOI] [PubMed] [Google Scholar]
- 16.Fultz SL, Skanderson M, Mole LA, et al. Development and verification of a "virtual" cohort using the national VA health information system. Med Care. 2006;44(8):S25–S30. doi: 10.1097/01.mlr.0000223670.00890.74. [DOI] [PubMed] [Google Scholar]
- 17.WHO Global Database on Body Mass Index (BMI): an interactive surveillance tool for monitoring nutrition transition. Public Health Nutr. 2006;9(5):658–658. [Google Scholar]
- 18.Diagnosis and classification of diabetes mellitus. Diabetes Care. 2010;33(Suppl 1):S62–S69. doi: 10.2337/dc10-S062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tien PC, Schneider MF, Cox C, et al. Association of HIV Infection With Incident Diabetes Mellitus: Impact of Using Hemoglobin A1C as a Criterion for Diabetes. Jaids-J Acq Imm Def. 2012;61(3):334–340. doi: 10.1097/QAI.0b013e31826bfc32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McGinnis KA, Brandt CA, Skanderson M, et al. Validating smoking data from the Veteran's Affairs Health Factors dataset, an electronic data source. Nicotine & tobacco research : official journal of the Society for Research on Nicotine and Tobacco. 2011;13(12):1233–1239. doi: 10.1093/ntr/ntr206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Crum-Cianflone NF, Roediger M, Eberly LE, et al. Obesity among HIV-infected persons: impact of weight on CD4 cell count. Aids. 2010;24(7):1069–1072. doi: 10.1097/QAD.0b013e328337fe01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Madec Y, Szumilin E, Genevier C, et al. Weight gain at 3 months of antiretroviral therapy is strongly associated with survival: evidence from two developing countries. Aids. 2009;23(7):853–861. doi: 10.1097/QAD.0b013e32832913ee. [DOI] [PubMed] [Google Scholar]
- 23.Palermo B, Bosch RJ, Bennett K, Jacobson JM. Body mass index and CD4+ T-lymphocyte recovery in HIV-infected men with viral suppression on antiretroviral therapy. HIV Clin Trials. 2011;12(4):222–227. doi: 10.1310/HCT1204-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carr A, Samaras K, Burton S, et al. A syndrome of peripheral lipodystrophy, hyperlipidaemia and insulin resistance in patients receiving HIV protease inhibitors. Aids. 1998;12(7):F51–F58. doi: 10.1097/00002030-199807000-00003. [DOI] [PubMed] [Google Scholar]
- 25.Hadigan C, Meigs JB, Corcoran C, et al. Metabolic abnormalities and cardiovascular disease risk factors in adults with human immunodeficiency virus infection and lipodystrophy. Clin Infect Dis. 2001;32(1):130–139. doi: 10.1086/317541. [DOI] [PubMed] [Google Scholar]
- 26.Vigouroux C, Gharakhanian S, Salhi Y, et al. Diabetes, insulin resistance and dyslipidaemia in lipodystrophic HIV-infected patients on highly active antiretroviral therapy (HAART) Diabetes Metab. 1999;25(3):225–232. [PubMed] [Google Scholar]
- 27.Jerico C, Knobel H, Montero M, et al. Metabolic syndrome among HIV-infected patients - Prevalence, characteristics, and related factors. Diabetes Care. 2005;28(1):132–137. doi: 10.2337/diacare.28.1.132. [DOI] [PubMed] [Google Scholar]
- 28.Achhra AC, Mocroft A, Reiss P, et al. Short-term weight gain after antiretroviral therapy initiation and subsequent risk of cardiovascular disease and diabetes: the D:A:D study. Hiv Med. 2015 doi: 10.1111/hiv.12294. [DOI] [PubMed] [Google Scholar]
- 29.Araujo S, Banon S, Machuca I, Moreno A, Perez-Elias MJ, Casado JL. Prevalence of insulin resistance and risk of diabetes mellitus in HIV-infected patients receiving current antiretroviral drugs. Eur. J. Endocrinol. 2014;171(5):545–554. doi: 10.1530/EJE-14-0337. [DOI] [PubMed] [Google Scholar]
- 30.Butt AA, Fultz SL, Kwoh CK, Kelley D, Skanderson M, Justice AC. Risk of diabetes in HIV infected veterans pre- and post-HAART and the role of HCV coinfection. Hepatology. 2004;40(1):115–119. doi: 10.1002/hep.20289. [DOI] [PubMed] [Google Scholar]
- 31.Butt AA, McGinnis K, Rodriguez-Barradas MC, et al. HIV infection and the risk of diabetes mellitus. AIDS. 2009;23(10):1227–1234. doi: 10.1097/QAD.0b013e32832bd7af. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Butt AA, McGinnis KA, Skanderson M, et al. Diabetes Mellitus in HIV Infected Persons - Role of CD4 Counts. 13th Conference on Retroviruses and Opportunistic Infections (CROI 2006); February 5–9, 2006; Denver CO. 2006. [Google Scholar]
- 33.Capeau J, Bouteloup V, Katlama C, et al. Ten-year diabetes incidence in 1046 HIV-infected patients started on a combination antiretroviral treatment. Aids. 2012;26(3):303–314. doi: 10.1097/QAD.0b013e32834e8776. [DOI] [PubMed] [Google Scholar]
- 34.Achhra ACMA, Reiss P, Sabin C, Ryom L, de Wit S, Smith C, Monforte AD, Phillips A, Weber R, Lundgren J, Law MG. Impact of short-term change in body mass index (BMI) after antiretroviral therapy (ART) initiation on subsequent risk of cardiovascular disease (CVD) and diabetes in HIV-positive individuals: the D:A:D study; 20th International AIDS Conference; 2014. Abstract WEAB0103(Melbourne, AUS) [Google Scholar]
- 35.Batterham MJ. Investigating heterogeneity in studies of resting energy expenditure in persons with HIV/AIDS: a meta-analysis. Am. J. Clin. Nutr. 2005;81(3):702–713. doi: 10.1093/ajcn/81.3.702. [DOI] [PubMed] [Google Scholar]
- 36.Esser NL-PS, Piette J, Scheen AJ, Paquot N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract. 2014 Apr; doi: 10.1016/j.diabres.2014.04.006. [DOI] [PubMed] [Google Scholar]
- 37.Aroor AR, McKarns S, Demarco VG, Jia G, Sowers JR. Maladaptive immune and inflammatory pathways lead to cardiovascular insulin resistance. Metabolism. 2013;62(11):1543–1552. doi: 10.1016/j.metabol.2013.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wada NI, Jacobson LP, Margolick JB, et al. The effect of HAART-induced HIV suppression on circulating markers of inflammation and immune activation. AIDS. 2015;29(4):463–471. doi: 10.1097/QAD.0000000000000545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Amorosa V, Synnestvedt M, Gross R, et al. A tale of 2 epidemics: the intersection between obesity and HIV infection in Philadelphia. J Acq Immun Def Synd. 2005;39(5):557–561. [PubMed] [Google Scholar]
- 40.Brown TT, Cole SR, Li XH, et al. Antiretroviral therapy and the prevalence and incidence of diabetes mellitus in the Multicenter AIDS Cohort Study. Arch Intern Med. 2005;165(10):1179–1184. doi: 10.1001/archinte.165.10.1179. [DOI] [PubMed] [Google Scholar]
- 41.El-Sadr WM, Mullin CM, Carr A, et al. Effects of HIV disease on lipid, glucose and insulin levels: results from a large antiretroviral-naive cohort. Hiv Med. 2005;6(2):114–121. doi: 10.1111/j.1468-1293.2005.00273.x. [DOI] [PubMed] [Google Scholar]
- 42.Rasmussen LD, Mathiesen ER, Kronborg G, Pedersen C, Gerstoft J, Obel N. Risk of diabetes mellitus in persons with and without HIV: a Danish nationwide population-based cohort study. PLoS One. 2012;7(9):e44575. doi: 10.1371/journal.pone.0044575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kilby JM, Tabereaux PB. Severe hyperglycemia in an HIV clinic: preexisting versus drug-associated diabetes mellitus. J Acquir Immune Defic Syndr Hum Retrovirol. 1998;17(1):46–50. doi: 10.1097/00042560-199801010-00007. [DOI] [PubMed] [Google Scholar]
- 44.Justman JE, Benning L, Danoff A, et al. Protease inhibitor use and the incidence of diabetes mellitus in a large cohort of HIV-infected women. Jaids-J Acq Imm Def. 2003;32(3):298–302. doi: 10.1097/00126334-200303010-00009. [DOI] [PubMed] [Google Scholar]
- 45.Althoff KN, Justice AC, Gange SJ, et al. Virologic and immunologic response to HAART, by age and regimen class. Aids. 2010;24(16):2469–2479. doi: 10.1097/QAD.0b013e32833e6d14. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.