Abstract
Manool, a diterpene isolated from Salvia officinalis, was evaluated by the XTT colorimetric assay for cytotoxicity and selectivity against different cancer cell lines: B16F10 (murine melanoma), MCF-7 (human breast adenocarcinoma), HeLa (human cervical adenocarcinoma), HepG2 (human hepatocellular carcinoma), and MO59J, U343 and U251 (human glioblastoma). A normal cell line (V79, Chinese hamster lung fibroblasts) was used to compare the selectivity of the test substance. Manool exhibited higher cytotoxic activity against HeLa (IC50 = 6.7 ± 1.1 µg/mL) and U343 (IC50 = 6.7 ± 1.2 µg/mL) cells. In addition, in the used experimental protocols, the treatment with manool was significantly more cytotoxic for different tumor cell lines than for the normal cell line V79 (IC50 = 49.3 ± 3.3 µg/mL), and showed high selectivity. These results suggest that manool may be used to treat cancer without affecting normal cells.
Keywords: Manool, Salvia officinalis, Cancer cells, Cytotoxicity, Selectivity
Introduction
The success of cancer chemotherapy depends on the development of drugs that selectively destroy tumor cells, or at least limit their proliferation without causing severe side effects (Nussbaumer et al. 2011). The main difficulty in preventing cancer progression is the increased resistance, low specificity and high toxicity of commercially available drugs (Riedl et al. 2011). Plants, especially their secondary metabolites, have long been used for the treatment of cancer and several effective anticancer drugs exerting direct or indirect effects have been discovered (Gragg et al. 2009).
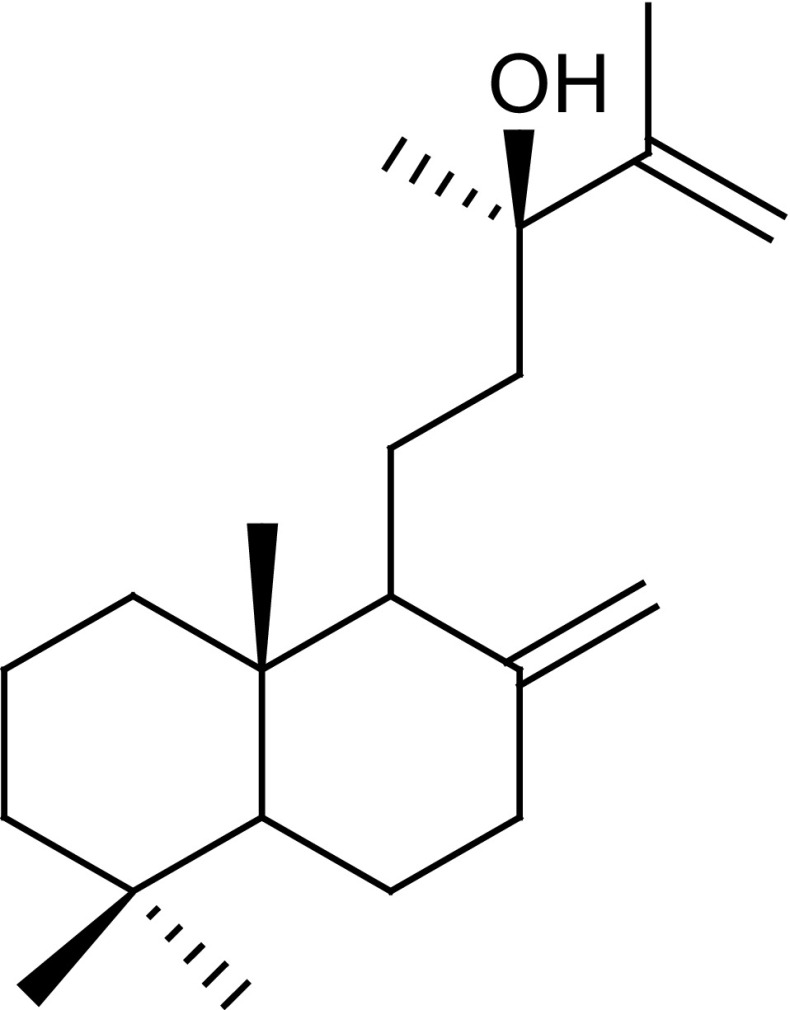
Salvia officinalis L., a medicinal plant of the family Lamiaceae popularly known as salvia or sage, is an aromatic herb which is widely distributed in the world (Badiee et al. 2012). The chemical composition of sage is very complex and the diterpene manool (Fig. 1) is one of the most abundant components found in its essential oils and extracts (Farhat et al. 2009). Among other biological properties, manool has shown activity against Staphylococcus aureus (Ulubelen et al. 1994) and exhibited promising minimal inhibitory concentration (MIC) values against several bacteria associated with periodontitis (Souza et al. 2011).
Fig. 1.
Chemical structure of the diterpene manool
In view of the biological relevance and easy availability of manool, the present study investigated its cytotoxic activity against seven different cancer cell lines. In order to better understand the cytotoxic activity of manool, a normal cell line (V79, Chinese hamster lung fibroblasts) was used to compare its selectivity. The choice of V79 cells for comparison is due the fact it is a well characterized cell line and commonly used in cytotoxicity studies (Bradley et al. 1981; Munari et al. 2014).
Materials and methods
Isolation of the diterpene manool
For the isolation of manool, dried leaves of certified Salvia officinalis (1.0 kg) were purchased from Nutri Comércio de Ervas Ltda. (São Paulo, Brazil). The identification was carried out by Prof. Milton Groppo and a voucher specimen (SPFR 15178) was deposited in the herbarium of the Departamento de Biologia, Universidade de São Paulo, Ribeirão Preto, SP, Brazil. The plant material was then pulverized, and exhaustively extracted with dichloromethane (5 L), yielding 45.5 g of the crude extract. The extract was resuspended in 300 mL methanol/water (9:1) and filtered. The soluble fraction was partitioned with n-hexane (300 mL, three times), yielding 10.6 g of a hexane-soluble fraction after solvent evaporation under reduced pressure. This fraction was submitted to vacuum chromatography over silicagel 60H (500 g; Merck; art. 7736; Darmstadt, Germany) (Pelletier et al. 1986) using n-hexane and increasing amounts of ethyl acetate as eluents (1500 mL each fraction). The second fraction (n-hexane/ethyl acetate 8:2; 2.89 g) was then partitioned by column chromatography over silicagel 60 (100 g; Merck; art. 7734) using n-hexane/ethyl acetate 9:1) as eluent. Fifty fractions were collected and combined after TLC analysis (Silicagel; Whatman; art. 4420222; St. Louis, MO, USA) and also using n-hexane/ethyl acetate 9:1 as mobile phase. The second combined fraction (200 mg) was identified as the diterpene manool.
The identification of manool was performed by 1H-and 13C-NMR analysis using Bruker DPX 400 spectrometer (400 MHz for 1H and 100 MHz for 13C). The sample was dissolved in CDCl3, and the spectra were calibrated with the solvent signals at d 7.26 (1H) and d 77.0 (13C). The experimental values were then compared with literature data (Bastard et al. 1984; Ulubelen et al. 1997) in order to confirm the structural identification.
Cell lines and culture conditions
Different cell lines were used in evaluating the cytotoxic activity: non-tumor cell line, V79 (Chinese hamster lung fibroblasts), courtesy of Laboratório de Mutagênese da Universidade Estadual de Londrina (Paraná, Brazil); and seven tumor cell lines, B16F10 (murine melanoma), courtesy of Departamento de Bioquímica da Faculdade de Medicina da Universidade de São Paulo (Campus de Ribeirão Preto, São Paulo, Brazil); MCF-7 (human breast adenocarcinoma) and HepG2 (hepatocellular carcinoma), courtesy of Laboratório de Mutagênese do Departamento de Ciências Biológicas da Universidade Estadual Paulista (Campus de Araraquara, São Paulo, Brazil); HeLa (human cervical adenocarcinoma) and MO59 J (human glioblastoma), purchased from the Cell Bank of Universidade Federal do Rio de Janeiro (Brazil); U343 and U251 (human glioblastoma), courtesy of Laboratório de Oncologia Pediátrica do Hospital das Clínicas de Ribeirão Preto (Universidade de São Paulo, Brazil).
Cells from the 4th through to the 12th passage were used. The different cell lines were maintained as monolayers in plastic culture flasks (25 cm2) containing HAM-F10 plus DMEM (1:1; Sigma-Aldrich; St. Louis, MO, USA) or only DMEM depending on the cell line, supplemented with 10 % fetal bovine serum (Nutricell; Campinas, São Paulo, Brazil) and 2.38 mg/mL Hepes (Sigma-Aldrich) at 37 °C in a humidified 5 % CO2 atmosphere. Antibiotics (0.01 mg/mL streptomycin and 0.005 mg/mL penicillin; Sigma-Aldrich) were added to the medium to prevent bacterial growth.
Cytotoxicity testing
Manool was directly diluted in DMSO (final concentration of 0.1 %) and the medium to obtain concentrations of 0.49–1000 µg/mL. The final concentration of DMSO was less than 1 % and had no negative effects on the cell lines. Doxorubicin (DXR, Zodiac; São Paulo, Brazil), (S)-(+)-camptothecin (CPT, Sigma-Aldrich) and etoposide (VP16, Sigma-Aldrich) were used as positive controls. For the experiments, 104 cells were plated onto 96-well microplates. Each well received 100 µL HAM-F10/DMEM or DMEM medium containing the different concentrations of manool and the cells were cultured in a 5 % CO2 atmosphere for 24 h at 37 °C. After incubation with manool and the chemotherapeutic drugs (DXR, CPT and VP16), the culture medium was removed and the cells were washed with 100 µL phosphate-buffered saline and exposed to 100 µL HAM-F10 culture medium without phenol red. Next, 25 µL XTT (Roche Diagnostics; Mannheim, Germany) was added to each well and the microplates were incubated for 17 h at 37 °C. Absorbance of the samples was read in a microplate reader (ELISA, Asys UVM 340/Microwin 2000) at a wavelength of 450 nm and a reference length of 620 nm.
Calculation of the selectivity index
The selectivity index was calculated by dividing the IC50 value of manool on V79 cells by the IC50 value determined for cancer cells.
Statistical analysis
Cytotoxicity was assessed using the IC50 response parameter (50 % cell growth inhibition) calculated with the GraphPad Prism program, plotting cell survival against the respective concentrations of manool. One-way ANOVA was used for the comparison of means (P < 0.05).
Results
Table 1 shows the IC50 values and the selectivity index for the cell lines treated with manool and the positive controls DXR, CPT and VP16. The IC50 values for tumor cell lines treated with manool ranged from 6.7 ± 1.1 to 49.3 ± 3.3 µg/mL. The highest IC50 value of manool was 28.5 ± 0.3 µg/mL, while the lowest IC50 was 6.7 ± 1.2 µg/mL.
Table 1.
IC50 values and selectivity index (SI) of the different cell lines after treatment with manool and the positive controls
| Cell line | Treatment (µg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|
| Manool | DXR | CPT | VP16 | |||||
| IC50 | SI | IC50 | SI | IC50 | SI | IC50 | SI | |
| V79 | 49.3 ± 3.3 | – | 7.8 ± 0.9 | – | 19.6 ± 4.6 | – | 45.9 ± 7.5 | – |
| B16F10 | 15.6 ± 0.1a | 3.2 | 3.8 ± 0.2 | 2.0 | 20.2 ± 1.9 | 1.0 | 48.9 ± 8.5 | 0.9 |
| MCF-7 | 17.1 ± 0.8a | 2.9 | 5.4 ± 1.3 | 1.4 | 36.1 ± 12.5 | 0.5 | 82.7 ± 15.6 | 0.6 |
| HeLa | 6.7 ± 1.1a | 7.4 | 21.9 ± 9.1 | 0.4 | 19.4 ± 0.8 | 1.0 | 225.5 ± 31.8 | 0.2 |
| HepG2 | 28.5 ± 0.3a | 1.7 | 62.1 ± 2.0 | 0.1 | 11.9 ± 1.9 | 1.6 | 235.4 ± 6.5 | 0.2 |
| MO59 J | 9.6 ± 0.8a | 5.1 | 6.9 ± 2.1 | 1.1 | 15.5 ± 0.6 | 1.3 | 58.9 ± 3.1 | 0.8 |
| U343 | 6.7 ± 1.2a | 7.4 | 0.7 ± 0.3 | 11.1 | 5.7 ± 1.0 | 3.4 | 2.2 ± 0.1 | 20.9 |
| U251 | 13.1 ± 1.6a | 3.8 | 6.3 ± 2.5 | 1.2 | 11.1 ± 2.8 | 1.8 | 42.9 ± 0.4 | 1.1 |
V79, Chinese hamster lung fibroblasts; B16F10, murine melanoma; MCF-7, human breast adenocarcinoma; HeLa, human cervical adenocarcinoma, HepG2, human hepatocellular carcinoma; MO59 J, U343, and U251, human glioblastoma; DXR, doxorubicin; CPT, (S)-(+)-camptothecin; VP16, etoposide. The values are the mean ± SD (n = 3)
aSignificantly different compared to the normal cell line (V79) (P < 0.05). The selectivity index is the ratio of IC50 values of manool on V79 cells to those found in the cancer cell lines
In general, the tumor cell lines treated with manool displayed the lowest IC50 values. The most pronounced cytotoxic effects were observed in HeLa and U343 cells, with an IC50 value of 6.7 ± 1.1 µg/mL for both cell lines. The IC50 values obtained for all cell lines tested were significantly lower than that observed for normal V79 cells (49.3 ± 3.3 µg/mL) (Table 1).
The IC50 values found for manool are lower than those observed in the treatments with known chemotherapeutic drugs, particularly VP16, in some of the tumor cell lines tested. Doxorubicin, CPT and VP16 are currently used for routine cancer treatment. It was therefore interesting to compare the effects of these drugs with manool. The selective index (SI) found for the manool for some cell lines tested were greater than those observed for the positive controls. The highest selectivity indices were observed for the HeLa and U343 cell lines (7.4) (Table 1).
Discussion
Manool exerted cytotoxic effects on all cell lines tested and was significantly more cytotoxic against the cancer cell lines than normal cells, as well as the positive controls tested, exhibiting high selectivity indices. According to the American National Center Institute, substances presenting IC50 values <30 µg/mL in tumor cell lines are considered promising candidates for anticancer drug development (Suffness and Pezzuto 1990) and manool may therefore be a promising anticancer drug.
In a study conducted by Pratsinis et al. (2010) investigating diterpenes and flavonoids isolated from Greek propolis, including manool, the latter was active against a human colorectal adenocarcinoma (HT29) (IC50 = 6.5 ± 0.3 µg/mL) and fibrosarcoma (HT1080) (IC50 = 19.3 ± 2.2 µg/mL) cell line in the MTT assay. These data demonstrate the selectivity manool since the IC50 values for the normal human skin fibroblasts (AG01523c; DSF9 and DSF60) was >29 μg/mL. These data agree with the present findings showing that manool was cytotoxic against tumor cells at low concentrations, but did not directly affect normal cells at the same concentrations.
The literature showed manool as an important cytotoxic agent. In a cell cycle analysis following incubation of exponentially growing HT-29 cells with manool at a concentration of 50 μM (14.5 μg/mL) for 24–48 h, Pratsinis et al. (2010) suggested that manool exerts its cytostatic activity after the first 24 h and reduces the percentage of malignant cells in the S phase. Furthermore, manool can block cells in the G2/M phase of the cell cycle, increasing the percentage of cells at G2/M by approximately 2.5 fold compared to control (Pratsinis et al. 2010).
Manool is a hydroxylated labdane-type diterpene and one of the major compounds found in sage essential oil (Farhat et al. 2009). Labdane-type diterpenoids have been suggested to possess the potential for anticancer drug development. Two labdane-type diterpenoids, yunnancoronarin A and B, showed interesting IC50 values (0.27 and 0.69 µg/mL) against lung adenocarcinoma (A549) and leukemia (K562) cell lines, respectively. Kumrit et al. (2010) evaluated the cytotoxic activity of villosin and ellipticine, labdane-type diterpenoids, against human small cell lung cancer (NCI-H187) and non-cancerous (Vero cells) cell lines. The results showed that villosin exhibited potent cytotoxic activity (IC50 of 0.12 μg/mL) against NCI-H187, while the IC50 for ellipticine was 0.44 μg/mL. Moreover, ellipticine was very toxic to Vero cells (IC50 of 1.81 μg/mL), whereas toxicity of villosin was undetectable at a concentration lower than 49.93 μg/mL.
In conclusion, the treatment with manool was significantly more cytotoxic for different tumor cell lines than for the normal cell line V79, and showed high selectivity. Therefore, manool may be a promising anticancer drug.
Acknowledgments
The authors would like to thank the São Paulo Research Foundation (FAPESP, Grant # 2011/21310-2), Brazil.
Compliance with ethical standards
Conflict of interest
The authors declare no conflicts of interest.
References
- Badiee P, Nasirzadeh AR, Motaffaf M. Comparison of Salvia officinalis L. essential oil and antifungal agents against Candida species. J Pharm Technol Drug Res. 2012;1:7. doi: 10.7243/2050-120X-1-7. [DOI] [Google Scholar]
- Bastard J, Duc DK, Fetizon M, Francis MJ, Grant PK, Weavers RT, Kaneko C, Baddeley GV, Bernassau JM, Burfitt IR, Wovkulich PM, Wenkert E. C-13 nuclear magnetic-resonance spectroscopy of naturally-occurring substances.81. Cmr spectroscopy of labdane diterpenes and related substances. J Nat Prod. 1984;47:592–599. doi: 10.1021/np50034a004. [DOI] [Google Scholar]
- Bradley MO, Bhuyan B, Francis MC, Langenbach R, Peterson A, Huberman E. Mutagenesis by chemical agents in V79 Chinese hamster cells: a review and analysis of the literature. A report of the Gene-Tox program. Mutat Res. 1981;87:81–142. doi: 10.1016/0165-1110(81)90029-4. [DOI] [PubMed] [Google Scholar]
- Farhat MB, Jordán MJ, Chaouech-Hamada R, Landoulsi A, Sotomayor JA. Variations in essential oil, phenolic compounds, and antioxidant activity of Tunisian cultivated Salvia officinalis L. J Agric Food Chem. 2009;57:10349–10356. doi: 10.1021/jf901877x. [DOI] [PubMed] [Google Scholar]
- Gragg GM, Grothaus PG, Newman DJ. Impact of natural products on developing new anti-cancer agents. Chem Rev. 2009;109:3012–3043. doi: 10.1021/cr900019j. [DOI] [PubMed] [Google Scholar]
- Kumrit I, Suksamrarn A, Meepawpan P, Songsri S, Nuntawong N. Labdane-type diterpenes from Hedychium gardnerianum with potent cytotoxicity against human small cell lung cancer cells. Phytother Res. 2010;24:1009–1013. doi: 10.1002/ptr.3057. [DOI] [PubMed] [Google Scholar]
- Munari CC, Oliveira PF, Campos JC, Martins SP, Costa JC, Bastos JK, Tavares DC. Antiproliferative activity of Solanum lycocarpum alkaloidic extract and their constituents, solamargine and solasonine, in tumor cell lines. J Nat Med. 2014;68:236–241. doi: 10.1007/s11418-013-0757-0. [DOI] [PubMed] [Google Scholar]
- Nussbaumer S, Bonnabry P, Veuthey JL, Fleury-Souverain S. Analysis of anticancer drugs: a review. Talanta. 2011;85:2265–2289. doi: 10.1016/j.talanta.2011.08.034. [DOI] [PubMed] [Google Scholar]
- Pelletier SW, Chokshi HP, Desai HK. Separation of diterpenoid alkaloid mixtures using vacuum liquid chromatography. J Nat Prod. 1986;49:892–900. doi: 10.1021/np50047a021. [DOI] [Google Scholar]
- Pratsinis H, Kletsas D, Melliou E, Chinou I. Antiproliferative activity of Greek propolis. J Med Food. 2010;13:286–290. doi: 10.1089/jmf.2009.0071. [DOI] [PubMed] [Google Scholar]
- Riedl S, Zweytick D, Lohner K. Membrane-active host defense peptides—Challenges and perspectives for the development of novel anticancer drugs. Chem Phys Lipids. 2011;164:766–781. doi: 10.1016/j.chemphyslip.2011.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Souza AB, de Souza MGM, Moreira MA, Moreira MR, Furtado NAJC, Martins CHG, Bastos JK, dos Santos RA, Heleno VCG, Ambrosio SR, Veneziani RCS. Antimicrobial evaluation of diterpenes from Copaifera langsdorffii oleoresin against periodontal anaerobic bacteria. Molecules. 2011;16:9611–9619. doi: 10.3390/molecules16119611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suffness M, Pezzuto JM. Assays related to cancer drug discovery. In: Hostettmann K, editor. Methods in plant biochemistry: assay for bioactivity. London: Academic Press; 1990. pp. 71–133. [Google Scholar]
- Ulubelen A, Topcu G, Eri C, Sonmez U, Kartal M, Kurucu, Bozok-Johansson C. Terpenoids from Salvia sclarea. Phytochemistry. 1994;36:971–974. doi: 10.1016/S0031-9422(00)90474-6. [DOI] [PubMed] [Google Scholar]
- Ulubelen A, Sonmez U, Topcu G. Diterpenoids from the roots of Salvia sclarea. Phytochemistry. 1997;44:1297–1299. doi: 10.1016/S0031-9422(96)00709-1. [DOI] [Google Scholar]