Abstract
Background
The aim of the study was to explore the effectiveness of electrochemotherapy (ECT) during the treatment of melanoma patients with BRAF inhibitors. Its effectiveness was tested on BRAF mutated and non-mutated melanoma cells in vitro and in combination with BRAF inhibitors.
Materials and methods
ECT with bleomycin was performed on two human melanoma cell lines, with (SK-MEL-28) or without (CHL-1) BRAF V600E mutation. Cell survival was determined using clonogenic assay to determine the effectiveness of ECT in melanoma cells of different mutation status. Furthermore, the effectiveness of ECT in concomitant treatment with BRAF inhibitor vemurafenib was also determined in BRAF mutated cells SK-MEL-28 with clonogenic assay.
Results
The survival of BRAF V600E mutated melanoma cells was even lower than non-mutated cells, indicating that ECT is effective regardless of the mutational status of melanoma cells. Furthermore, the synergistic interaction between vemurafenib and ECT with bleomycin was demonstrated in the BRAF V600E mutated melanoma cells.
Conclusions
The effectiveness of ECT in BRAF mutated melanoma cells as well as potentiation of its effectiveness during the treatment with vemurafenib in vitro implies on clinical applicability of ECT in melanoma patients with BRAF mutation and/or during the treatment with BRAF inhibitors.
Key words: electrochemotherapy, BRAF inhibitors, vemurafenib, melanoma
Introduction
Recently, there have been great advances in the treatment of metastatic melanoma, including targeted therapy with BRAF and MEK inhibitors, and immunomodulation either with anti-CTLA-4 or anti-PD-1 or anti-PD-L1 antibodies.1-5
BRAF inhibitors are based on the fact that 50% of melanoma tumors harbor BRAF V600 mutations, which cause increased activation of MAP kinase signaling pathway that results in melanoma cell proliferation.6 Inhibitors of BRAF V600 mutated melanoma, such as vemurafenib and dabrafenib, increase progression-free survival and overall survival1,7,8, but unfortunately the resistance mechanisms usually appear to re-establish the signaling pathway and the disease progresses in the matter of months after the start of treatment.9
For the progressive disease after treatment with BRAF inhibitors, other locoregional treatments are usually needed to control tumor relapses. Electrochemotherapy (ECT) provides this approach. It is a combined local treatment in which locally-applied high voltage electric pulses are used to facilitate the uptake of non-permeant chemotherapeutic drugs.10 It is used mainly for the treatment of cutaneous and subcutaneous metastases of different tumor histology, with a complete and objective response rate of 59.4% and 84.1%, respectively.11 Specifically for melanoma, the complete and objective response rates of 56.8% and 80.6% were obtained.11
The unexplored question is whether ECT is effective in BRAF mutated melanoma cells, and whether ECT is effective as concomitant treatment to therapy with BRAF inhibitors. In the first reported case regarding treatment of a patient undergoing dabrafenib treatment, ECT proved to be effective on progressing tumor nodules.12 Based on this report, studies on effectiveness of ECT with bleomycin in BRAF V600 mutated, compared to nonmutated melanoma cells are warranted. Besides this aim, we also investigated whether the concomitant vemurafenib treatment of cells can affect the effectiveness of ECT in melanoma cells in vitro.
Materials and methods
Cell lines and culturing
Human melanoma cell line SK-MEL-28 (American Type Culture Collection (ATCC), Manassas, VA, USA) with BRAF V600E mutation was cultivated in an Advanced MEM medium (Gibco, Thermo Fisher Scientific, Waltham, MA, USA), supplemented with 5% fetal bovine serum (FBS, Gibco), 10 mM L-glutamine (GlutaMAX, Gibco), 100 U/ml penicillin (Grünenthal, Aachen, Germany) and 50 pg/μl gentamicin (Krka, Novo mesto, Slovenia) in a 5% CO2 humidified incubator at 37°C.
Human melanoma cell line CHL-1 (ATCC) without BRAF V600 mutations was cultured in an Advanced RPMI 1640 medium (Gibco), supplemented with 5% FBS, 10 mM L-glutamine, 100 U/ml penicillin and 50 pg/ml gentamicin in a 5% CO 2humidified incubator at 37°C.
Electrochemotherapy (ECT)
Melanoma cells were grown as a monolayer until they reached at least 80% confluence. The medium was removed and the cells were washed with phosphate-buffered saline (PBS, Merck Millipore, Darmstadt, Germany). After that, cells were detached from the surface with 0.25% trypsin/EDTA in Hank’s buffer (Gibco). After detachment, trypsin was inactivated with an equal amount of cell culture medium with FBS and cells were collected and centrifuged. For electroporation, 88 μl of cell suspension (25 × 106 cells/ml) was prepared in electroporation buffer (125 mM sucrose, 10 mM K2HPO4, 2.5 mM KH2PO4, 2 mM MgCl2 × 6H20) at 4°C. Cell suspension was mixed with 22 pl of different stock concentrations of bleomycin (Bleomycin medac, Medac, Wedel, Germany) to reach a final concentration of 1.4 × 10−12 M, 1.4 × 10−11 M, 1.4 × 10−10 M, 1.4 × 10−9 M, 1.4 × 10−8 M, 1.4 × 10−7 M, 1.4 × 10−6 M. Out of 110 pl of mixture, 50 pl served as a control for bleomycin treatment and other 50 pl was pipetted between two stainless steel parallel plate electrodes (2 mm apart) and 8 square-wave electric pulses (amplitude over distance ratio of 1300 V/ cm, duration of 100 ps and frequency of 1 Hz) were applied. Electric pulses were generated with the electric pulse generator GT-01 (Faculty of Electrical Engineering, University of Ljubljana, Ljubljana, Slovenia). The cells were incubated 5 min after electroporation at room temperature, and then cell culture medium was added. Afterwards, clonogenic assay was performed. Experimental groups were denoted: BLM (different concentrations of bleomycin); BLM + EP (ECT with bleomycin of different concentrations). Each group was normalized to the control group of same treatment regimen with 0 M bleomycin (Ctrl, EP).
Clonogenic assay
After the treatment, cells were plated in 6 cm petri dishes with 4 ml of culture medium for clonogenic assay. The number of plated cells was in a range of 300-4000 cells, based on predicted cytotoxicity of the treatment. Colonies were formed after 12 and 8 days for SK-MEL-28 and CHL-1, respectively. After the colonies were formed, they were fixed and stained with crystal violet solution (Sigma-Aldrich, St. Louis, MO, USA) and counted. The colonies containing less than 50 cells were disregarded. Plating efficiency was calculated for each experimental group as the ratio between counted colonies and the number of plated cells. Plating efficiency of each treatment group was normalized to the untreated cells group, representing cell survival. IC90 value was determined (drug concentration required to reduce cell survival for 90%).
Vemurafenib sensitivity
Cell lines were tested for their sensitivity to vemurafenib treatment in order to confirm the selectivity of vemurafenib on BRAF V600E mutated cells and to determine the optimal concentration for the combination treatment. Cells were detached from cell culture dishes and prepared for clonogenic assay. Vemurafenib (MedChem Express, Monmouth Junction, NJ, USA) was obtained in 10 mM DMSO solution and was added to 6 cm culture dishes with 4 ml of culture medium to reach a final concentration of 0.5 pM, 2.5 pM, 5 pM and 10 pM. The final amount of DMSO in the cell culture medium represented 20000x — 1000x dilution and was not toxic to the cells. Afterwards, cells were cultured in a 5% CO2 humidified incubator at 37°C until the formation of colonies, which were counted and analyzed as described. Each group was normalized to the control group with 0 pM vemurafenib.
Combination of ECT and vemurafenib
ECT was performed as described. After that, cells were plated in 6 cm petri dishes for clonogenic assay and vemurafenib was added in each dish to reach a final concentration of 0.5 pM. Afterwards, cells were cultured in a 5% CO2 humidified incubator at 37°C until the formation of colonies, which were counted and analyzed as described. Experimental groups were denoted: BLM (different concentrations of bleomycin); BLM + EP (ECT with bleomycin of different concentrations); BLM + VMF (bleomycin of different concentrations and addition of 0.5 pM vemurafenib), BLM + EP + VMF (ECT with bleomycin of different concentrations and addition of 0.5 pM vemurafenib). Each group was normalized to the control group of same treatment regimen with 0 M bleomycin (Ctrl, EP, Ctrl + VMF, EP + VMF).
Statistical analysis
The mode of interaction between the treatments with independent mode of action was calculated at the level of IC90 by the method developed by Spector.13 For the analysis and graphical representation, SigmaPlot Software (version 12.0, Systat Software, London, UK) was used.
Results
Sensitivity of BRAF V600E mutated melanoma cells to vemurafenib
Vemurafenib treatment was tested for selective cytotoxicity to BRAF V600E mutated cells. Selectivity of BRAF inhibitor vemurafenib was confirmed on SK-MEL-28 BRAF V600E mutated melanoma cells. Cell survival was reduced with increasing vemurafenib concentration with IC90 5 pM (Figure 1A). In contrast, vemurafenib treatment did not reduce the survival of BRAF V600E non-mutated cell line CHL-1. The cell survival was higher than 94%, also with the highest concentration of vemurafenib used (Figure 1B). These results also provided bases for the combined vemurafenib and ECT treatment of cells, the concentration of 0.5 pM was selected, which reduced cell survival to 50%.
Figure 1.
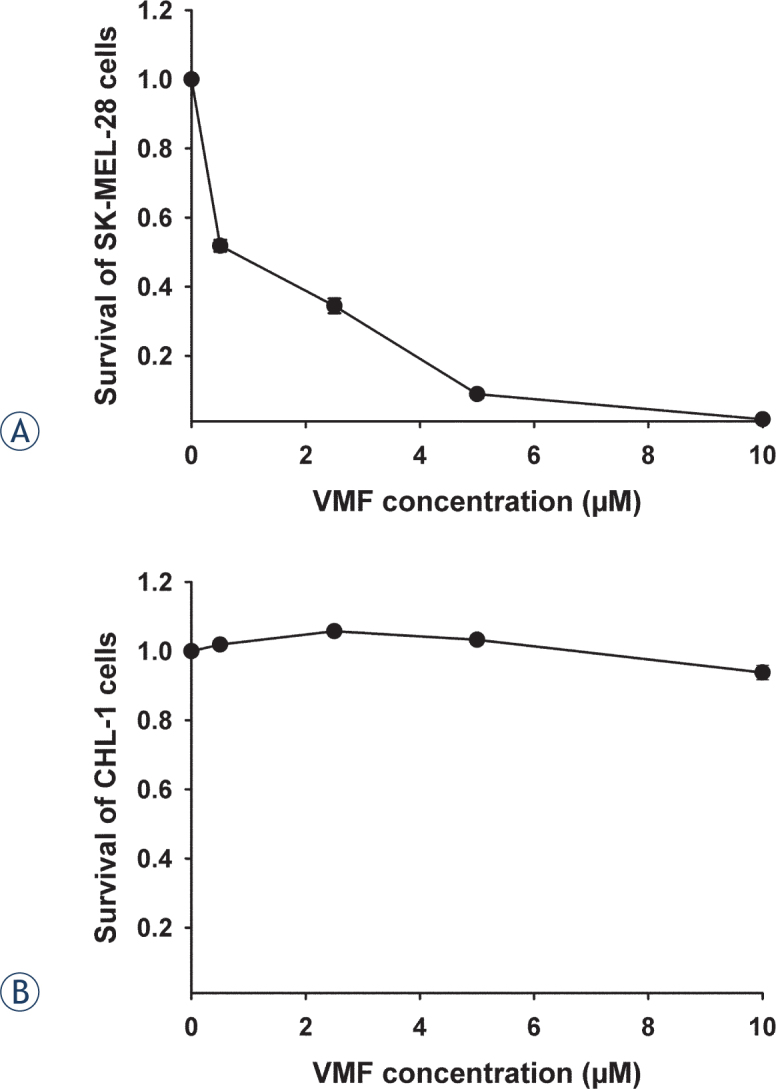
Survival of melanoma cells after vemurafenib (VMF) treatment. Survival of (A) BRAF V600E mutated melanoma cells SK-MEL-28 and (B) non-mutated melanoma cells CHL-1.
BRAF V600E mutated melanoma cells are more sensitive to ECT than non-mutated melanoma cells
The effectiveness of ECT was tested on BRAF V600E mutated melanoma cells (SK-MEL-28) and non-mutated melanoma cells (CHL-1). ECT effectively reduced survival of both cell lines with IC90 3.8 × 10−10 M and 7.7 × 10−10 M for SK-MEL-28 and CHL-1, respectively. ECT was even more effective on BRAF V600E mutated SK-MEL-28 cells that required 2 times lower concentration of BLM at IC90, compared to non-mutated cells CHL-1 (Figure 2), confirming that ECT is an effective method for treatment of also BRAF V600E mutated melanoma cells.
Figure 2.
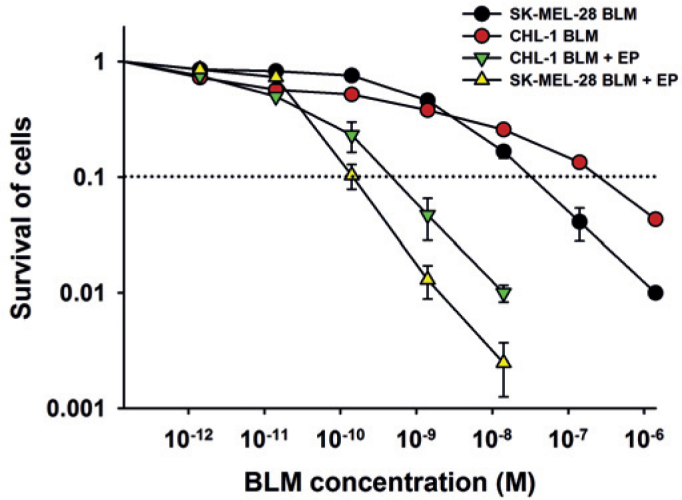
Survival of BRAF V600E mutated SK-MEL-28 melanoma cells and non-mutated CHL-1 cells after ECT with bleomycin. BLM (bleomycin); BLM + EP (ECT with bleomycin). Dotted line represents the IC90 value.
Concomitant vemurafenib treatment increased ECT effectiveness
Concomitant treatment with ECT and vemurafenib was simulated in vitro on cells in the way, that ECT treated cells were seeded into dishes containing 0.5 μM vemurafenib. The vemurafenib treatment decreased survival of SK-MEL-28 cells for 50%. If the effect of the vemurafenib was eliminated (normalized to control groups with added vemurafenib for groups BLM + VMF and BLM + EP + VMF), as shown in Figure 3, then an increased effectiveness of ECT was observed on BRAF mutated SK-MEL-28 cells. A 4.5 times lower concentration of BLM was needed at the IC90 for cells treated with vemurafenib (BLM + EP + VMF; IC90 value 8.5 × 10−11 M) compared to cells without vemurafenib treatment (BLM + EP; IC90 value 3.8 × 10−10 M). The potentiation was more than additive (Figure 3), in fact, according to the method developed by Spector et al. was synergistic.13
Figure 3.
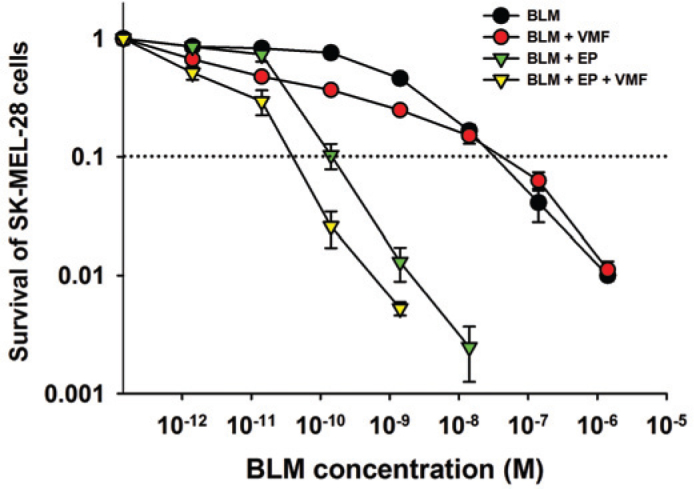
Survival of BRAF V600E mutated SK-MEL-28 melanoma cells after concomitant treatment with ECT and vemurafenib. BLM (bleomycin); BLM + VMF (bleomycin and 0.5 pM vemurafenib); BLM + EP (ECT with bleomycin); BLM + EP + VMF (ECT with bleomycin and 0.5 pM vemurafenib). Dotted line represents the IC90 value.
Discussion
This study demonstrates the effectiveness of ECT with bleomycin on BRAF V600E mutated melanoma cells. The effectiveness was higher to that on BRAF non-mutated cells. Furthermore, an interaction of ECT and vemurafenib treatment was observed in BRAF mutated melanoma cells, indicating on more than additive or synergistic effectiveness.
BRAF inhibitors provide a clear benefit to patients with disseminated disease. The effect is mediated by inhibition of cell proliferation through the inhibition of the MAPK pathway.14 Patients often present with multiple metastases, that may not all respond to the treatment either due to their bigger size or due to the development of the resistance to BRAF inhibitors. In such cases additional therapy is needed, that has proven effectiveness also in BRAF mutated melanoma cells. Our study demonstrates that ECT with BLM is as effective, or even more effective on BRAF mutated, compared to non-mutated melanoma cells, although the exact biological mechanism still needs to be explored. The data support the recent observation on melanoma patient undergoing dabrafenib treatment where some nodules were treated with ECT. ECT proved effective even on tumor nodules, which were in progression during the dabrafenib treatment12, which indicates that it could be effective also on dabrafenib resistant tumor clones.
The other aspect that was clarified is that ECT can be successfully implemented also during the treatment with BRAF inhibitors. The in vitro results demonstrated that vemurafenib and ECT treatment has synergistic effectiveness. This is of clinical importance, since the in vitro data indicate that there is no need to wait for the discontinuation of treatment with BRAF inhibitors and can be given concomitantly. Based on the observation of the Valpione et al, the tolerability of the combined treatment is of great importance, as was observed in the described case.12 The possibility of the enhanced effectiveness further supports the approach to use ECT concomitantly during the treatment with BRAF inhibitors; however, further clinical studies with larger number of patients are needed to fully support the fact that the combined treatment does not cause additional undesired side effects. If the interaction of the treatments will be observed also in vivo, this may lead also to reduction of BLM dosage in ECT.
The interaction of BRAF inhibitors with radiotherapy has also been demonstrated, both on tumor and normal tissue. In vitro study demonstrated radiosensitization of BRAF mutated melanoma cells with BRAF inhibitor PLX-4032 by clonogenic and invasion assay and was associated with enhancement of G1 cell cycle arrest.15Furthermore, the combination of BRAF inhibitor and irradiation was proven to be effective also in high-grade gliomas, harboring BRAF V600E mutation. Radiosensitization was observed by PLX-4720 BRAF inhibitor in vitro, whereas in BRAF non-mutated glioma cells the radiosensitizing effect was not observed.16 The clinical studies, however, demonstrated the radiosensitization of normal tissue as well. There is still not consensus whether the normal tissue damage is acceptable17, or that they are so severe that this requires profound investigations in the future.18
Eventually ECT will most probably find its place in combination with systemic treatments, with targeted therapies as well as with recently emerging immune checkpoint inhibitors. ECT was recently evaluated also combined with the immunotherapeutic approaches, immune checkpoint inhibitors. The first clinical study of Mozillo et al. reported on safety of the combined ipilimumab and ECT with good therapeutic responsiveness.19 The second report also demonstrated effectiveness and safety of a sequential treatment with ECT plus ipilimumab, which induced a durable complete response of multiple cutaneous metastases with vitiligo-like lesions indicating on involvement of immune response.20 Obviously ECT is progressing into concomitant treatment with melanoma targeted therapies and immunotherapies, therefore its safety and effectiveness needs to be established. Specifically, due to induction of the immunogenic cell death induced in tumors21, ECT of tumors may serve as in situ vaccination that could be boosted by concomitant immunotherapeutic approach.22 The consequences may be both, the potentiated local response and also increased side effects.
In conclusion, the effectiveness of ECT in BRAF mutated cells implies on clinical applicability of ECT in BRAF mutated melanoma tumors. Furthermore, its effectiveness also during the treatment with BRAF inhibitors was demonstrated, with synergistic effectiveness. These results are encouraging, but need to be extended to more cell lines, and in vivo studies on experimental tumors, evaluating both the tumor and normal tissue response. The study should also be extended to patient-derived melanoma cell lines and also on clones which develop resistance to the therapy with BRAF inhibitors to verify if ECT maintains its effectiveness on the resistant clones. Such studies will then predict the tumor response, and possible side effects.
Acknowledgments
The authors acknowledge the financial support from the state budget by the Slovenian Research Agency (program no. P3-0003). The research was conducted in the scope of LEA EBAM (French-Slovenian European Associated Laboratory: Pulsed Electric Fields Applications in Biology and Medicine). The research is a result of the networking efforts of the COST Action TD1104. We wish to thank Mira Lavric (Institute of Oncology Ljubljana, Ljubljana, Slovenia) for the help with cell culturing.
Disclosure: No potential conflicts of interest were disclosed.
References
- 1.Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507–2516. doi: 10.1056/NEJMoa1103782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hauschild A, Grob JJ, Demidov LV, Jouary T, Gutzmer R, Millward M. et al. Dabrafenib in BRAF-mutated metastatic melanoma: a multicentre, open-label, phase 3 randomised controlled trial. Lancet. 2012;380:358–65. doi: 10.1016/S0140-6736(12)60868-X. [DOI] [PubMed] [Google Scholar]
- 3.Hodi FS, O’day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–2454. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ugurel S, Rohmel J, Ascierto PA, Flaherty KT, Grob JJ, Hauschild A. et al. Survival of patients with advanced metastatic melanoma: the impact of novel therapies. Eur J Cancer. 2016;53:125–134. doi: 10.1016/j.ejca.2015.09.013. [DOI] [PubMed] [Google Scholar]
- 6.Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S. et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–954. doi: 10.1038/nature00766. [DOI] [PubMed] [Google Scholar]
- 7.Puzanov I, Amaravadi RK, McArthur GA, Flaherty KT, Chapman PB, Sosman JA. et al. Long-term outcome in BRAF(V600E) melanoma patients treated with vemurafenib: patterns of disease progression and clinical management of limited progression. Eur J Cancer. 2015;51:1435–1443. doi: 10.1016/j.ejca.2015.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Scholtens A, Geukes foppen MH, Blank CU, van Thienen JV, van Tinteren H, Haanen JB. Vemurafenib for BRAF V600 mutated advanced melanoma: results of treatment beyond progression. J Cancer. 2015;51:642–652. doi: 10.1016/j.ejca.2015.01.009. [DOI] [PubMed] [Google Scholar]
- 9.Trunzer K, Pavlick AC, Schuchter L, Gonzalez R, McArthur GA, Hutson TE. et al. Pharmacodynamic effects and mechanisms of resistance to vemurafenib in patients with metastatic melanoma. J Clin Oncol. 2013;31:1767–1774. doi: 10.1200/JCO.2012.44.7888. [DOI] [PubMed] [Google Scholar]
- 10.Sersa G, Miklavcic D, Cemazar M, Rudolf Z, Pucihar G, Snoj M. Electrochemotherapy in treatment of tumours. Eur J Surg Oncol. 2008;34:232–240. doi: 10.1016/j.ejso.2007.05.016. [DOI] [PubMed] [Google Scholar]
- 11.Mali B, Jarm T, Snoj M, Sersa G, Miklavcic D. Antitumor effectiveness of electrochemotherapy: a systematic review and meta-analysis. Eur J Surg Oncol. 2013;39:4–16. doi: 10.1016/j.ejso.2012.08.016. [DOI] [PubMed] [Google Scholar]
- 12.Valpione S, Campana LG, Pigozzo J, Chiarion-sileni V. Consolidation electrochemotherapy with bleomycin in metastatic melanoma during treatment with dabrafenib. Radiol Oncol. 2015;49:71–74. doi: 10.2478/raon-2014-0035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Spector SA, Tyndall M, Kelley E. Effects of acyclovir combined with other antiviral agents on human cytomegalovirus. Am J Med. 1982;73:36–39. doi: 10.1016/0002-9343(82)90060-2. [DOI] [PubMed] [Google Scholar]
- 14.Hertzman johansson C, Egyhazi brage S. BRAF inhibitors in cancer therapy. Pharmacol Ther. 2014;142:176–182. doi: 10.1016/j.pharmthera.2013.11.011. [DOI] [PubMed] [Google Scholar]
- 15.Sambade MJ, Peters EC, Thomas NE, Kaufmann WK, Kimple RJ, Shields JM. Melanoma cells show a heterogeneous range of sensitivity to ionizing radiation and are radiosensitized by inhibition of B-RAF with PLX-4032. Radiother Oncol. 2011;98:394–399. doi: 10.1016/j.radonc.2010.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dasgupta T, Olow AK, Yang X, Hashizume R, Nicolaides TP, Tom M. et al. Survival advantage combining a BRAF inhibitor and radiation in BRAF V600E-mutant glioma. J Neurooncol. 2016;126:385–393. doi: 10.1007/s11060-015-1939-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hecht M, Zimmer L, Loquai C, Weishaupt C, Gutzmer R, Schuster B. et al. Radiosensitization by BRAF inhibitor therapy-mechanism and frequency of toxicity in melanoma patients. Ann Oncol. 2015;26:1238–1244. doi: 10.1093/annonc/mdv139. [DOI] [PubMed] [Google Scholar]
- 18.Zahnreich S, Mayer A, Loquai C, Grabbe S, Schmidberger H. Radiotherapy with BRAF inhibitor therapy for melanoma: progress and possibilities. Future Oncol. 2016;12:95–106. doi: 10.2217/fon.15.297. [DOI] [PubMed] [Google Scholar]
- 19.Mozzillo N, Simeone E, Benedetto L, Curvietto M, Giannarelli D, Gentilcore G. et al. Assessing a novel immuno-oncology-based combination therapy: Ipilimumab plus electrochemotherapy. Oncoimmunol. 2015;4:e1008842. doi: 10.1080/2162402X.2015.1008842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brizio M, Fava P, Astrua C, Cavaliere G, Savoia P. Complete regression of melanoma skin metastases after electrochemotherapy plus ipilimumab treatment: an unusual clinical presentation. Eur J Dermatol. 2015;25:271–272. doi: 10.1684/ejd.2015.2522. [DOI] [PubMed] [Google Scholar]
- 21.Calvet CY, Famin D, Andre FM, Mir LM. Electrochemotherapy with bleomycin induces hallmarks of immunogenic cell death in murine colon cancer cells. Oncoimmunol. 2014;3:e28131. doi: 10.4161/onci.28131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sersa G, Teissie J, Cemazar M, Signori E, Kamensek U, Marshall G. et al. Electrochemotherapy of tumors as in situ vaccination boosted by immunogene electrotransfer. Cancer Immunol Immunother. 2015;64:1315–1327. doi: 10.1007/s00262-015-1724-2. [DOI] [PMC free article] [PubMed] [Google Scholar]


