Abstract
Objective
To explore the effect of right dorsolateral prefrontal cortex (DLPFC) repetitive transcranial magnetic stimulation (rTMS) on memory, and its correlation with levels of hippocampal brain metabolites detected by proton magnetic resonance spectroscopy (1H-MRS) in recently detoxified alcohol-dependent patients.
Materials and methods
In this randomized, double-blind sham-controlled trial, alcohol-dependent patients were enrolled and randomized into two groups: the experimental group (rTMS, 10 Hz, on right DLPFC, 20 sessions) and the control group (sham stimulation). Memory function was assessed using Hopkins Verbal Learning Test-Revised (HVLT-R) and Brief Visuospatial Memory Test-Revised (BVMT-R) before and after treatment. 1H-MRS was used to detect the levels of N-acetyl aspartic acid (NAA), choline (Cho), and creatine (Cr) in bilateral hippocampi before and after treatment.
Results
Thirty-eight patients (18 in the experimental group and 20 in the control group) were included in the analyses. The experimental group showed significantly greater changes in HVLT-R, BVMT-R, NAA/Cr, and Cho/Cr after rTMS from baseline than the control group. The percentage change in BVMT-R and HVLT-R correlated with the percentage change in NAA/Cr and Cho/Cr in the right brain.
Conclusion
High-frequency right DLPFC rTMS was associated with improvement in memory dysfunction, which is correlated with levels of hippocampal brain metabolites detected by 1H-MRS in recently detoxified alcohol-dependent patients.
Keywords: alcohol dependence, memory, repetitive transcranial magnetic stimulation, MR spectroscopy
Introduction
Long-term excessive alcohol use can lead to numerous neurological complications including damage to the cerebral cortex,1 and impaired cognitive2 and neuropsychological functioning.3 Although these effects may be partially reversed after alcohol use ceases,4 a treatment that can augment brain recovery during the early stage of alcohol abstinence would be beneficial.
Repetitive transcranial magnetic stimulation (rTMS), a noninvasive neuromodulation technique, has been used as treatment for various neuropsychiatric disorders, including depression,5 obsessive–compulsive disorder,6 schizophrenia,7 and substance addiction.8 Modulation of the release of dopamine and glutamate in the mesolimbic dopaminergic system, a major pathway associated with substance-seeking behaviors, is a plausible mechanism underlying the effect of rTMS on addiction.9,10 The dorsolateral prefrontal cortex (DLPFC) has been shown to be associated with regulation of craving through the mesolimbic pathway.11 In randomized controlled trials, high-frequency rTMS over the right DLPFC was found to significantly reduce craving and stabilize cognitive performance in alcohol-dependent patients.12,13 DLPFC has also been shown to play a key role in memory function.14 Although studies have demonstrated that DLPFC rTMS is associated with enhancement of memory,15,16 few studies have evaluated the effect of DLPFC rTMS on memory in alcohol-dependent patients. We hypothesize that DLPFC rTMS could improve memory in alcohol-dependent patients who have recently undergone detoxification treatment.
Changes in levels of brain metabolites often precede abnormalities in brain structure, and proton magnetic resonance spectroscopy (1H-MRS) is a recognized method for quantifying such changes. 1H-MRS has been used to evaluate hippocampal brain metabolites in various populations, including patients with mild cognitive impairment,17 patients with Alzheimer’s disease,18 cognitively normal older adults,19 and patients receiving whole brain radiotherapy.20 Results of these studies show hippocampal brain metabolites to be reliable preclinical markers of cognitive and memory function.
In this clinical trial, we aimed to investigate the effect of high-frequency DLPFC rTMS on memory in recently detoxified alcohol-dependent patients and evaluate the relationship between the effect and changes in levels of hippocampal brain metabolites as detected by 1H-MRS.
Materials and methods
Subjects
This study was a single-center, randomized, double-blind, sham-controlled trial. Alcohol-dependent patients who were admitted to the Mental Health Institute of the First Hospital of Hebei Medical University from January 2011 to January 2012 were eligible for enrollment in this study. The inclusion criteria were as follows: 1) alcohol dependence in accordance with the diagnostic criteria of the International Classification of Diseases, 10th Revision, 2) Alcohol Dependence Scale score ≥14,21 3) Clinical Institute Withdrawal Assessment of Alcohol Revised Scale22 (CIWA-Ar) score <7, 4) right-handedness, and 5) ability to cooperate for cognitive function testing and magnetic resonance imaging (MRI). Patients were excluded if any of the following conditions were present: 1) Wernicke’s encephalopathy, 2) Korsakoff syndrome, 3) schizophrenia, 4) intellectual disability, 5) history of cranial trauma or severe neurological diseases, 6) dependence on psychoactive substances (excluding tobacco), and 7) contraindication to rTMS. All patients provided written informed consent, and the study was approved by the Institution Review Board of the First Hospital of Hebei Medical University.
Patients were administered detoxification treatment consisting of oral diazepam 20–50 mg daily for 1–3 weeks. After the second week of alcohol detoxification, the patients were evaluated by two physicians and only those with a CIWA-Ar score <7 were enrolled and included in the analyses. A total of 40 patients were enrolled and randomized to the experimental group or the control group using a random number table; patients with odd numbers were allocated to the experimental group and those with even numbers to the control group.
Intervention
A Magstim RAPID2 stimulator (Magstim, Carmarthenshire, UK) equipped with a figure-8-shaped coil was used to deliver rTMS. Stimulation was administered over the right DLPFC. All patients received four treatment sessions. Duration of each treatment session was 5 days with a 2-day interval between sessions. During each session, patients received eight trains daily (10 Hz, 80% of resting motor threshold, 10 seconds on, 5 seconds off; 800 pulses). The control group was administered sham stimulation with the same parameters but with the coil placed perpendicular to the scalp.
Evaluation
Evaluations were performed by two physicians, and the results were reviewed by the senior chief physician. Evaluators and the patients were unaware of group assignment.
Verbal and visuospatial episodic memory (immediate, delayed, and recognition for both) were assessed using the Hopkins Verbal Learning Test-Revised (HVLT-R)23 and Brief Visuospatial Memory Test-Revised (BVMT-R),24 respectively. 1H-MRS was used to detect bilateral hippocampal brain metabolites.
HVLT-R is a tool used to assess verbal learning and memory that requires recall of a list of 12 words (from three semantic categories) over three learning trials, free recall after a delay, and a recognition trial. BVMT-R is a common assessment for visual learning that requires patients to reproduce six figures at the correct location after a 10-second presentation. Each figure is scored a maximum of two points (one point for correct form, one point for correct location), and the total score over three trials is calculated.
For measurement of brain metabolites, MRI was performed on a GE Signa TwinSpeed 1.5T MRI (GE Healthcare, Buckinghamshire, UK), using three-dimensional spoiled gradient echo. Hippocampal volume was measured using coronal and sagittal planes. Point-resolved echo spin spectroscopy was utilized to collect single-voxel magnetic resonance spectra from the left and right sides. Shimming and water inhibition were automatically completed by the scanner, with a water suppression rate >98%. Scanning was performed with the following parameters: repetition time =1,500 ms, echo time =35 ms, number of acquisitions =128, voxel size =20×20×20 mm, and acquisition time =3 minutes 48 seconds for each site. 1H-MRS was applied on bilateral hippocampi to detect N-acetyl aspartic acid (NAA), choline (Cho), and creatine (Cr). Image processing was performed at a workstation using vendor-provided software (GE-Funtool; GE Healthcare Bio-Sciences Corp.) to measure the levels of NAA, Cho, and Cr. As the Cr concentration remains relatively constant, its concentration was used as an internal reference for calculating the metabolite ratio (NAA/Cr and Cho/Cr).25
Statistical analysis
Continuous variables are presented as median with inter-quartile range, and categorical variables are presented as count with percentage. To compare the characteristics and outcomes between the two groups, Mann–Whitney U-test and chi-square tests were used for continuous and categorical variables, respectively. Cohen’s d was calculated to compare the changes in HVLT-R and BVMT-R scores between the groups. Correlation between the percentage changes in memory function and measures of brain metabolites was tested by Spearman’s rank correlation coefficient. A two-tailed P<0.05 indicated statistical significance. All statistical analyses were performed using IBM SPSS Statistics for Windows, version 19.0 (IBM Corporation, Armonk, NY, USA).
Results
A total of 40 patients were enrolled in this study. Two patients in the experimental group were excluded due to being lost to follow-up; therefore, data from 18 patients in the experimental group and 20 patients in the control group were included in the analyses. There was no significant difference between the experimental and the control groups regarding median age (49 years vs 48 years, P=0.536), sex (male percentage: 66.7% vs 65.0%, P=0.914), or median duration of alcohol use (21.0 years vs 18.5 years, P=0.438).
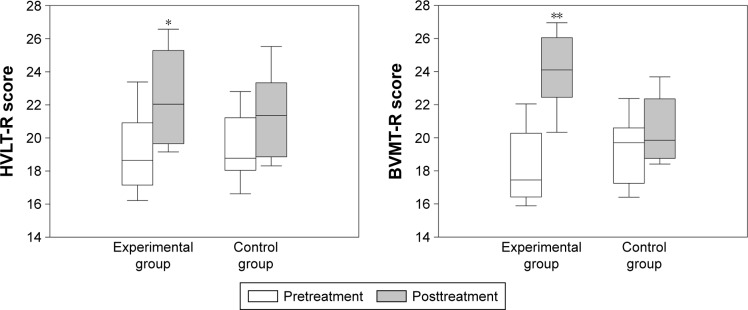
Change in memory function after rTMS is presented in Figure 1. Both groups showed improvement in HVLT-R and BVMT-R scores from baseline. Improvement was significantly greater in the experimental group than the control group for both HVLT-R (change: 3.1 vs 1.7, P=0.003, Cohen’s d=1.15) and BVMT-R (change: 6.0 vs 1.2, P<0.001, Cohen’s d=3.56).
Figure 1.
Change in memory function assessed by HVLT-R and BVMT-R after high-frequency right dorsolateral prefrontal cortex rTMS in recently detoxified alcohol-dependent patients.
Notes: Both groups showed improved memory after rTMS from baseline. The experimental group had significantly greater improvement than the control group in HVLT-R (*P=0.003, Cohen’s d=1.15) and BVMT-R (**P<0.001, Cohen’s d=3.56) scores.
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; HVLT-R, Hopkins Verbal Learning Test-Revised; BVMT-R, Brief Visuospatial Memory Test-Revised.
Regarding changes in measurements of hippocampal brain metabolites, both groups exhibited bilateral increases in NAA/Cr and Cho/Cr after rTMS from baseline. The experimental group had significantly greater percentage change from baseline than the control group in NAA/Cr (left: 16.3% vs 5.7%, P=0.023; right: 28.7% vs 6.5%, P=0.001) and Cho/Cr (left: 26.7% vs 6.1%, P=0.005; right: 29.3% vs 6.8%, P<0.001; Table 1).
Table 1.
The changes of brain metabolites after treatment
| Variables | Experimental group (n=18) | Control group (n=20) | P-value |
|---|---|---|---|
| Left brain | |||
| NAA | |||
| Pretreatment | 55 (51–61) | 50 (46–56) | 0.032 |
| Posttreatment | 57 (49–62) | 52 (49–55) | 0.065 |
| Change | 3 (−3 to 6) | 2 (−1 to 3) | 0.527 |
| Change (%) | 5.5 (−5.8 to 9.7) | 3.8 (−1.9 to 6.8) | 0.804 |
| Cho | |||
| Pretreatment | 52 (42–55) | 47 (44–51) | 0.285 |
| Posttreatment | 54 (50–63) | 49 (44–53) | 0.005 |
| Change | 7 (3–12) | 2 (−3 to 4) | 0.004 |
| Change (%) | 12.5 (5.5–25.9) | 3.3 (−5.7 to 8.0) | 0.004 |
| Cr | |||
| Pretreatment | 45 (43–48) | 43 (41–46) | 0.090 |
| Posttreatment | 41 (39–43) | 43 (39–44) | 0.371 |
| Change | −5 (−6 to −1) | −3 (−3 to 1) | 0.017 |
| Change (%) | −8.8 (−13.9 to −2.4) | −5.3 (−7.5 to 1.2) | 0.021 |
| NAA/Cr | |||
| Pretreatment | 1.2 (1.2–1.3) | 1.2 (1.1–1.3) | 0.292 |
| Posttreatment | 1.4 (1.1–1.6) | 1.3 (1.2–1.3) | 0.077 |
| Change | 0.2 (0.1–0.3) | 0.1 (0–0.1) | 0.017 |
| Change (%) | 16.3 (8.3–26.7) | 5.7 (1.4–12.1) | 0.023 |
| Cho/Cr | |||
| Pretreatment | 1.1 (0.9–1.3) | 1.1 (1.0–1.1) | 0.930 |
| Posttreatment | 1.4 (1.2–1.5) | 1.1 (1.1–1.2) | 0.001 |
| Change | 0.3 (0.1–0.4) | 0.1 (0–0.1) | 0.003 |
| Change (%) | 26.7 (4.6–40.6) | 6.1 (−1.4 to 12.8) | 0.005 |
| Right brain | |||
| NAA | |||
| Pretreatment | 54 (49–56) | 52 (47–55) | 0.333 |
| Posttreatment | 60 (57–65) | 54 (47–58) | 0.000 |
| Change | 7 (4–13) | 3 (−2 to 4) | 0.001 |
| Change (%) | 11.8 (7.8–27.7) | 4.5 (−3.2 to 8.3) | 0.002 |
| Cho | |||
| Pretreatment | 49 (42–52) | 49 (42–53) | 0.736 |
| Posttreatment | 58 (53–64) | 48 (46–55) | 0.004 |
| Change | 10 (6–13) | 4 (1.0 to 6) | 0.001 |
| Change (%) | 22.9 (9.5–30.8) | 7.8 (0.7–13.0) | 0.003 |
| Cr | |||
| Pretreatment | 46 (43–49) | 45 (42–48) | 0.340 |
| Posttreatment | 43 (40–45) | 44 (43–46) | 0.268 |
| Change | −4 (−6 to 1) | 0 (−3 to 2) | 0.077 |
| Change (%) | −8.7 (−10.7 to 2.4) | −1.1 (−6.6 to 3.3) | 0.047 |
| NAA/Cr | |||
| Pretreatment | 1.2 (1.0–1.2) | 1.1 (1.0–1.2) | 0.988 |
| Posttreatment | 1.4 (1.2–1.6) | 1.2 (1.1–1.3) | 0.002 |
| Change | 0.3 (0.1–0.4) | 0.1 (0–0.1) | 0.001 |
| Change (%) | 28.7 (6.4–40.1) | 6.5 (−1.5 to 10.1) | 0.001 |
| Cho/Cr | |||
| Pretreatment | 1.0 (1.0–1.1) | 1.1 (0.9–1.2) | 0.599 |
| Posttreatment | 1.4 (1.2–1.5) | 1.1 (1.0–1.2) | 0.001 |
| Change | 0.3 (0.2–0.5) | 0.1 (0–0.2) | <0.001 |
| Change (%) | 29.3 (14.6–45.6) | 6.8 (−0.2 to 14.5) | <0.001 |
Note: Data are presented as median (interquartile range).
Abbreviations: NAA, N-acetyl aspartic acid; Cho, choline compound; Cr, creatine compound.
Correlations between the percentage change in memory function and the percentage change in brain metabolites are presented in Table 2. In the left brain, the percentage change in BVMT-R significantly and positively correlated with the percentage change in NAA/Cr (r=0.324) and Cho/Cr (r=0.340). No significant correlation between HVLT-R and brain metabolites was noted in the left brain. In the right brain, the percentage change in both HVLT-R and BVMT-R showed significant positive correlation with the percentage change in both NAA/Cr and Cho/Cr (Table 2).
Table 2.
Correlations between the percentage changes of memory function and the percentage change of brain metabolites by Spearman’s rank correlation coefficient
| Variables | HVLT-R | BVMT-R |
|---|---|---|
| Left brain | ||
| NAA | 0.045 | 0.138 |
| Cho | −0.027 | 0.354* |
| Cr | −0.127 | −0.254 |
| NAA/Cr | 0.274 | 0.324* |
| Cho/Cr | 0.062 | 0.340* |
| Right brain | ||
| NAA | 0.345* | 0.561* |
| Cho | 0.254 | 0.352* |
| Cr | −0.311 | −0.352* |
| NAA/Cr | 0.392* | 0.532* |
| Cho/Cr | 0.405* | 0.461* |
Note:
P<0.05.
Abbreviations: HVLT-R, Hopkins Verbal Learning Test-Revised; BVMT-R, Brief Visuospatial Memory Test-Revised; NAA, N-acetyl aspartic acid; Cho, choline compound; Cr, creatine compound.
No major adverse events were reported. Minor adverse events were reported in eight patients: five patients complained of headache (three in experimental group and two in control group) and three patients experienced dizziness (two in experimental group and one in control group). All adverse events resolved spontaneously.
Discussion
In this study, treatment with high-frequency DLPFC rTMS was associated with improved memory in recently detoxified alcohol-dependent patients. Furthermore, improvement in memory positively correlated with increase in levels of brain metabolites, an indication of brain recovery. To the best of our knowledge, this is the first study to assess the effect of high-frequency DLPFC rTMS on memory and evaluate the correlation between the effect on memory and change in levels of hippocampal brain metabolites in this population.
The effects of DLPFC rTMS on craving reduction, mood modulation, and cognitive improvement in alcohol-dependent patients have been investigated in several studies. Mishra et al reported a significant reduction in alcohol craving in alcohol-detoxified patients following ten sessions of 10 Hz right DLPFC rTMS.12 In a follow-up study, the authors found similar significant reductions in craving when rTMS was administered over either the right or left DLPFC.26 Herremans et al found no effect on craving after one session of 20 Hz right DLPFC rTMS in detoxified alcohol-dependent patients. The author commented that the lack of effect may have resulted from inadequate stimulation.27 In a subsequent study, however, the authors found that a single session of 20 Hz right DLPFC rTMS was sufficient to significantly increase the stability of cognitive performance even without a reduction in craving.13 Furthermore, a study by Girardi et al reported significant reductions in both craving and depressive symptoms with 20 sessions of 20 Hz bilateral DLPFC rTMS as adjunctive therapy to standard drug treatment in concurrent alcohol-dependent patients with dysthymic disorder.28 In a study of bilateral DLPFC rTMS in patients with major depressive disorder with or without alcohol dependence, significant improvement in depressive symptoms was noted regardless of alcohol dependence.29 In our study, right DLPFC rTMS was associated with significant improvement in memory as assessed by HVLT-R and BVMT-R. The results provide further evidence for DLPFC rTMS as a potential treatment for improving memory and stabilizing cognitive performance in this population.
In this study, change in levels of bilateral hippocampal NAA, Cho, and Cr after DLPFC rTMS was evaluated using 1H-MRS. Previous studies have shown that hippocampal brain metabolites are useful preclinical predictors for cognitive decline and memory dysfunction.19 In a cross-sectional study, lower values of left hippocampal NAA/Cr were found to be significantly associated with poorer verbal learning (r=0.44) and memory retention (r=0.41) in older people suffering from long-term depression.30 In a prospective study, a significant decrease in hippocampal NAA level was noted after whole brain radiation, and left hippocampal NAA level was found to correlate with declined memory after radiotherapy.20 In the current study, degree of improvement in memory after high-frequency DLPFC rTMS was positively and significantly correlated with increase in hippocampal NAA/Cr and Cho/Cr. The results strongly support the positive effect of DLPFC rTMS on memory recovery in recently detoxified alcohol-dependent patients.
There were several limitations in this study. First is the limited number of patients enrolled in this study. Second, we did not control for some potential confounding factors, including body mass index, education level, and tobacco dependence. Third, as most participants reported that their drinking habits varied over time, amount of alcohol use could not be precisely assessed. Additionally, duration of alcohol use was self-reported, and recall bias is an inherent limitation of self-reported data. Finally, we did not evaluate craving; therefore, we could not evaluate the relationship between improvement in memory and learning and improvement in alcohol dependence.
Conclusion
High-frequency DLPFC rTMS was associated with improvement in memory in recently detoxified alcohol-dependent patients, and the improvement was positively correlated with increase in levels of hippocampal brain metabolites. Further investigations in a greater number of patients are necessary to better explore the anticraving and memory-improving effects of this potentially beneficial treatment.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Kril JJ, Halliday GM, Svoboda MD, Cartwright H. The cerebral cortex is damaged in chronic alcoholics. Neuroscience. 1997;79(4):983–998. doi: 10.1016/s0306-4522(97)00083-3. [DOI] [PubMed] [Google Scholar]
- 2.Stavro K, Pelletier J, Potvin S. Widespread and sustained cognitive deficits in alcoholism: a meta-analysis. Addict Biol. 2013;18(2):203–213. doi: 10.1111/j.1369-1600.2011.00418.x. [DOI] [PubMed] [Google Scholar]
- 3.Beatty WW, Tivis R, Stott HD, Nixon SJ, Parsons OA. Neuropsychological deficits in sober alcoholics: influences of chronicity and recent alcohol consumption. Alcohol Clin Exp Res. 2000;24(2):149–154. [PubMed] [Google Scholar]
- 4.Gazdzinski S, Durazzo TC, Meyerhoff DJ. Temporal dynamics and determinants of whole brain tissue volume changes during recovery from alcohol dependence. Drug Alcohol Depend. 2005;78(3):263–273. doi: 10.1016/j.drugalcdep.2004.11.004. [DOI] [PubMed] [Google Scholar]
- 5.Fitzgerald PB, Sritharan A, Daskalakis ZJ, de Castella AR, Kulkarni J, Egan G. A functional magnetic resonance imaging study of the effects of low frequency right prefrontal transcranial magnetic stimulation in depression. J Clin Psychopharmacol. 2007;27(5):488–492. doi: 10.1097/jcp.0b013e318151521c. [DOI] [PubMed] [Google Scholar]
- 6.Greenberg BD, Ziemann U, Corá-Locatelli G, et al. Altered cortical excitability in obsessive-compulsive disorder. Neurology. 2000;54(1):142–147. doi: 10.1212/wnl.54.1.142. [DOI] [PubMed] [Google Scholar]
- 7.Lee SH, Kim W, Chung YC, et al. A double blind study showing that two weeks of daily repetitive TMS over the left or right temporoparietal cortex reduces symptoms in patients with schizophrenia who are having treatment-refractory auditory hallucinations. Neurosci Lett. 2005;376(3):177–181. doi: 10.1016/j.neulet.2004.11.048. [DOI] [PubMed] [Google Scholar]
- 8.Eichhammer P, Johann M, Kharraz A, et al. High-frequency repetitive transcranial magnetic stimulation decreases cigarette smoking. J Clin Psychiatry. 2003;64(8):951–953. doi: 10.4088/jcp.v64n0815. [DOI] [PubMed] [Google Scholar]
- 9.Feil J, Zangen A. Brain stimulation in the study and treatment of addiction. Neurosci Biobehav Rev. 2010;34(4):559–574. doi: 10.1016/j.neubiorev.2009.11.006. [DOI] [PubMed] [Google Scholar]
- 10.Everitt BJ, Belin D, Economidou D, Pelloux Y, Dalley JW, Robbins TW. Review. Neural mechanisms underlying the vulnerability to develop compulsive drug-seeking habits and addiction. Philos Trans R Soc Lond B Biol Sci. 2008;363(1507):3125–3135. doi: 10.1098/rstb.2008.0089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Boggio PS, Sultani N, Fecteau S, et al. Prefrontal cortex modulation using transcranial DC stimulation reduces alcohol craving: a double-blind, sham-controlled study. Drug Alcohol Depend. 2008;92(1–3):55–60. doi: 10.1016/j.drugalcdep.2007.06.011. [DOI] [PubMed] [Google Scholar]
- 12.Mishra BR, Nizamie SH, Das B, Praharaj SK. Efficacy of repetitive transcranial magnetic stimulation in alcohol dependence: a sham-controlled study. Addiction. 2010;105(1):49–55. doi: 10.1111/j.1360-0443.2009.02777.x. [DOI] [PubMed] [Google Scholar]
- 13.Herremans SC, Vanderhasselt M-A, De Raedt R, Baeken C. Reduced intra-individual reaction time variability during a Go-NoGo task in detoxified alcohol-dependent patients after one right-sided dorsolateral prefrontal HF-rTMS session. Alcohol Alcohol. 2013;48(5):552–557. doi: 10.1093/alcalc/agt054. [DOI] [PubMed] [Google Scholar]
- 14.Curtis CE, D’Esposito M. Persistent activity in the prefrontal cortex during working memory. Trends Cogn Sci. 2003;7(9):415–423. doi: 10.1016/s1364-6613(03)00197-9. [DOI] [PubMed] [Google Scholar]
- 15.Gagnon G, Schneider C, Grondin S, Blanchet S. Enhancement of episodic memory in young and healthy adults: a paired-pulse TMS study on encoding and retrieval performance. Neurosci Lett. 2011;488(2):138–142. doi: 10.1016/j.neulet.2010.11.016. [DOI] [PubMed] [Google Scholar]
- 16.Balconi M. Dorsolateral prefrontal cortex, working memory and episodic memory processes: insight through transcranial magnetic stimulation techniques. Neurosci Bull. 2013;29(3):381–389. doi: 10.1007/s12264-013-1309-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ackl N, Ising M, Schreiber YA, Atiya M, Sonntag A, Auer DP. Hippocampal metabolic abnormalities in mild cognitive impairment and Alzheimer’s disease. Neurosci Lett. 2005;384(1–2):23–28. doi: 10.1016/j.neulet.2005.04.035. [DOI] [PubMed] [Google Scholar]
- 18.Schuff N, Amend D, Ezekiel F, et al. Changes of hippocampal N-acetyl aspartate and volume in Alzheimer’s disease. A proton MR spectroscopic imaging and MRI study. Neurology. 1997;49(6):1513–1521. doi: 10.1212/wnl.49.6.1513. [DOI] [PubMed] [Google Scholar]
- 19.Kantarci K, Weigand SD, Przybelski SA, et al. MRI and MRS predictors of mild cognitive impairment in a population-based sample. Neurology. 2013;81(2):126–133. doi: 10.1212/WNL.0b013e31829a3329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pospisil P, Kazda T, Bulik M, et al. Hippocampal proton MR spectroscopy as a novel approach in the assessment of radiation injury and the correlation to neurocognitive function impairment: initial experiences. Radiat Oncol. 2015;10:211. doi: 10.1186/s13014-015-0518-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ross HE, Gavin DR, Skinner HA. Diagnostic validity of the MAST and the alcohol dependence scale in the assessment of DSM-III alcohol disorders. J Stud Alcohol. 1990;51(6):506–513. doi: 10.15288/jsa.1990.51.506. [DOI] [PubMed] [Google Scholar]
- 22.Sullivan JT, Sykora K, Schneiderman J, Naranjo CA, Sellers EM. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar) Br J Addict. 1989;84:1353–1357. doi: 10.1111/j.1360-0443.1989.tb00737.x. [DOI] [PubMed] [Google Scholar]
- 23.Benedict RHB, Schretlen D, Groninger L, Brandt J. Hopkins Verbal Learning Test – Revised: normative data and analysis of inter-form and test–retest reliability. Clin Neuropsychol. 1998;12(1):43–55. [Google Scholar]
- 24.Benedict RHB, Schretlen D, Groninger L, Dobraski M, Shpritz B. Revision of the Brief Visuospatial Memory Test: studies of normal performance, reliability, and validity. Psychol Assess. 1996;8(2):145–153. [Google Scholar]
- 25.Bendszus M, Weijers HG, Wiesbeck G, et al. Sequential MR imaging and proton MR spectroscopy in patients who underwent recent detoxification for chronic alcoholism: correlation with clinical and neuropsychological data. AJNR Am J Neuroradiol. 2001;22(10):1926–1932. [PMC free article] [PubMed] [Google Scholar]
- 26.Mishra BR, Praharaj SK, Katshu MZUH, Sarkar S, Nizamie SH. Comparison of anticraving efficacy of right and left repetitive transcranial magnetic stimulation in alcohol dependence: a randomized double-blind study. J Neuropsychiatry Clin Neurosci. 2015;27(1):e54–e59. doi: 10.1176/appi.neuropsych.13010013. [DOI] [PubMed] [Google Scholar]
- 27.Herremans SC, Baeken C, Vanderbruggen N, et al. No influence of one right-sided prefrontal HF-rTMS session on alcohol craving in recently detoxified alcohol-dependent patients: results of a naturalistic study. Drug Alcohol Depend. 2012;120(1–3):209–213. doi: 10.1016/j.drugalcdep.2011.07.021. [DOI] [PubMed] [Google Scholar]
- 28.Girardi P, Rapinesi C, Chiarotti F, et al. Add-on deep transcranial magnetic stimulation (dTMS) in patients with dysthymic disorder comorbid with alcohol use disorder: a comparison with standard treatment. World J Biol Psychiatry. 2015;16(1):66–73. doi: 10.3109/15622975.2014.925583. [DOI] [PubMed] [Google Scholar]
- 29.Rapinesi C, Curto M, Kotzalidis GD, et al. Antidepressant effectiveness of deep transcranial magnetic stimulation (dTMS) in patients with major depressive disorder (MDD) with or without alcohol use disorders (AUDs): a 6-month, open label, follow-up study. J Affect Disord. 2014;174:57–63. doi: 10.1016/j.jad.2014.11.015. [DOI] [PubMed] [Google Scholar]
- 30.Jagannathan NR, Desai NG, Raghunathan P. Brain metabolite changes in alcoholism: an in vivo proton magnetic resonance spectroscopy (MRS) study. Magn Reson Imaging. 1996;14(5):553–557. doi: 10.1016/0730-725x(96)00048-3. [DOI] [PubMed] [Google Scholar]