Abstract
Introduction
It is unknown whether the magnitude of rib fracture (RF) displacement predicts pain medication requirements in blunt chest trauma patients.
Methods
Adult blunt RF patients undergoing chest computed tomography (CT) admitted to an urban Level 1 trauma center (2007–2012) were retrospectively reviewed. Pain management in those with displaced RF (DRF), non-displaced RF (NDRF), or combined DRF and NDRF (CRF) was compared by univariate analysis. Linear regression models were developed to determine whether total opioids requirements [expressed as log morphine equianalgesic dose (MED)] could be predicted by the magnitude of RF displacement (expressed as the sum of the Euclidean distance of all displaced RF) or number of RF, after adjusting for patient and injury characteristics.
Results
There were 245 patients, of whom 39 (16%) had DRF only, 77 (31%) had NDRF only, and 129 (53%) had CRF. Opioids were given to 224 (91%) patients. Compared to DRF (mean1.7 RF/patient) and NDRF patients (2.4 RF/patient), those with CRF (6.8 RF/patient) were older and had more RF per patient and a higher ISS and MED (251 vs. 53 and 105 mg, respectively, p<0.0001 and p=0.0045). They also more frequently received patient controlled analgesia. DRF patients had a lower mean ISS and MED and received more epidural analgesia compared with patients with NDRF. Total MED was associated with both the magnitude of RF displacement (p<0.0001) and the number of RF (p<0.0001). Every 5 mm increase in total displacement predicted a 6.3% increase in mean MED (p=0.0035) while every additional RF predicted an 11.2% increase in MED (p=0.0001). These associations included adjustment for age, ISS, and presence of chest tubes.
Conclusion
The magnitude of RF displacement and the number of RF predicted opioids requirements. This information may assist in anticipating patients with blunt RF who might have higher analgesic requirements.
Level of Evidence
Prognostic study, level III.
Keywords: Rib fractures, displacement, pain
INTRODUCTION
Rib fractures (RF) from blunt force are one of the most common injuries in trauma patients. Their main clinical manifestation is chest wall pain, which is the principal cause of morbidity as well as the main treatment target in these patients. Adequate pain control allows patients with RF to better expand their chest wall and deepen their cough, and thus, diminishes the risk for atelectasis and pneumonia. In contrast, poorly controlled chest wall pain increases the risks of complications, mortality, and long-term disabilities (1–4).
Multiple modalities have been proposed for managing RF pain including opioid and non-opioid pain medications, delivered either by enteral, parenteral or regional means (5–10). More recently, surgical rib fixation of RF is gaining popularity as a pain control paradigm in selected groups of patients (11–12). In these groups, the number and location of RF commonly serve as selection criteria. Nevertheless, the impact of the magnitude of RF displacement on selecting patients to operative vs. non-operative management has not yet been investigated. Furthermore, the association between the presence and magnitude of RF displacement with pain medication requirements has never been directly evaluated. As a first step in addressing these unknowns, we very recently described the association between RF anatomy and magnitude with intra-thoracic injuries and clinical outcomes (13). Specifically, we found a direct positive correlation between the presence and magnitude of RF displacement with thoracic co-injuries, days on mechanical ventilation, and length of stay.
The present study is a subsequent analysis of our retrospective data, which aimed to investigate the association between the presence and magnitude of RF displacement with opioid requirements as well as the use of epidural and patient controlled analgesia (PCA) in patients with RF. We hypothesized that RF displacement is associated with increased opioids requirements and use of PCA and epidural analgesia.
METHODS
The Institutional Review Board of Tufts Medical Center, an urban Level I trauma center verified by the American College of Surgeons, approved the study.
Study population
The trauma registry of Tufts Medical Center was probed to retrospectively review all adult (≥16 year-old) patients admitted with blunt RF and who underwent a chest-computed tomography (CT) between 2007 and 2012. Retrieved parameters included demographics (age, sex), mechanism of injury, pre-existing comorbidities, thoracic co-injuries, Injury Severity Score (ISS), and inpatient use of pain medications and paradigms. Opioid analgesic doses were calculated as the total dose for the entire admission.
It should be noted that the same dataset was used in our recent study, which focused on the anatomy of RF displacement and its association with thoracic co-injuries, days on mechanical ventilation, and hospital length of stay (13).
At Tufts Medical Center, the admitting trauma surgeon, who is aware of all patient’s injuries, determines initial pain management for each patient with RF. Later, the covering attending, as needed, may modify that original strategy. The same attending may consult the Anesthesia Pain Management Team for epidural analgesia if pain cannot be adequately controlled with high doses of narcotics or other pain-relief medications. It should be noted that during the study period RF pain management by both the Trauma and Anesthesia teams has not changed, and that paravertebral blocks were not utilized in any of the studied patients.
Study Design
Three different data analyses were performed. First, the characteristics of the DRF, NDRF and CRF groups were compared. Second, univariate analysis was performed to evaluate the associations between intravenous morphine equianalgesic dose (MED, 14) and the magnitude of RF displacement and their number. Third, multivariable linear regression models were developed to determine whether MED was predicted by the magnitude of RF displacement or the number of RF. The details of the statistical analyses are provided below.
Measurement of RF displacement
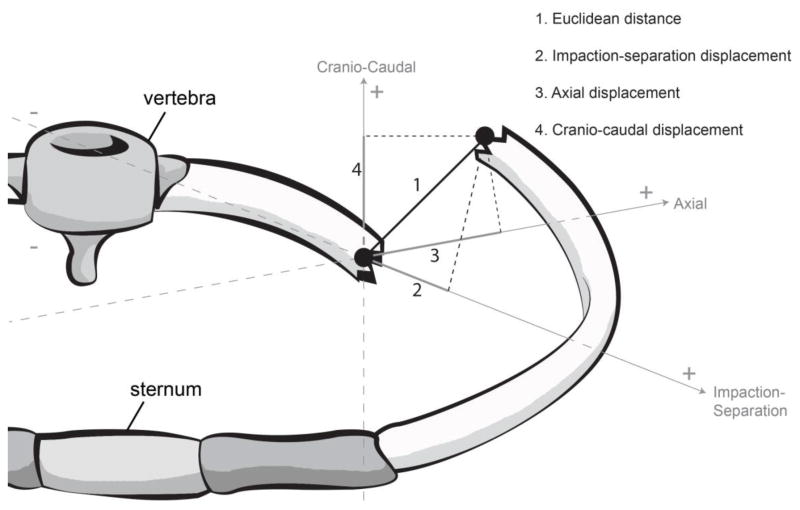
The methodology used in the present study for measuring RF displacement was recently described in detail (13). In brief, RF displacement was defined as any visible misalignment of cortexes of a rib fracture edge. It was measured in mm using axial (axial displacement), sagittal [cranio-caudal (CC) displacement] and coronal [impaction-separation (IS) displacement] CT images. The measurements were done using the PACS system tool. The 3D (Euclidean) distance (EDist) was calculated for each RF using the Pythagorean formula: EDist = (axial2+CC2+IS2)1/2 (Figure 1). Total RF displacement was defined as the sum of all Euclidian distances for each patient.
Figure 1.
Measurement of rib fracture displacement (see text for details).
A single team member (trauma surgeon), blinded to the clinical outcomes, performed all measurements. Repeating the measurements of twenty randomly selected displaced RF from twenty different patients assessed intra-rater reliability, and interclass correlation coefficients (ICC) and their 95% confidence intervals (CI) were computed separately for each axis. Intra-rater reliability was excellent, with the following ICCs (95% CI) for each axis: axial: 0.97 (0.93, 0.99); sagittal: 0.90 (0.78, 0.96); and coronal: 0.93 (0.84, 0.97).
Statistical analysis
Group comparisons
Statistical analysis was performed using SAS 9.4 (SAS Institute, Cary, NC). Differences in demographic and clinical characteristics between the DRF, NDRF and CRF groups were determined by χ2 tests for categorical variables and either t-tests or Mann-Whitney U tests for continuous variables, depending on the skewness of their distributions. All statistical testing was two sided with α=0.05 unless otherwise noted.
Univariate analyses
Two simple linear regression models were tested to examine the associations between MED and the magnitude of rib fracture displacement (continuous variable) or the number of rib fractures (whether displaced or not). As these two variables were strongly collinear, they were not included in a single regression model. Due to skewness, MED was transformed to the logarithmic scale (base 10) for the linear regression analyses.
Multivariable analyses
Two multivariable linear regression models were developed to determine whether MED was predicted by the total magnitude of RF displacement or the number of RF, after adjusting for patient and other injury characteristics. A multivariable linear regression model was built using a stepwise selection process using an a priori set of variables identified based on clinical judgment. These variables included age, ISS, presence of chest tube, hemothorax, pneumothorax, bilateral versus unilateral fractures, and pulmonary contusion. Only patients who received narcotics, whether alone or in combination with other pain medications, were included in the regression analyses.
RESULTS
Patient characteristics and outcomes
The characteristics and outcome of the studied patients are presented in Table 1. There were 245 patients, of whom 16% had DRF only, 31% had NDRF only, and 53% had CRF. Compared to DRF and NDRF patients, those with CRF were older and had more RF per patient and a higher ISS. DRF only patients had a lower ISS compared with patients with NDRF only. There was no difference among the groups in the median number of preexisting comorbidities and complications, and mortality. Comparison between patients who had at least one DRF (n=168) and patients with NDRF only (n=77) indicated a higher use of opioids (94.6 vs. 87%, p=0.041), patient controlled analgesia (27.1 vs. 10.4, p=0.003) and epidural analgesia (9.00 vs. 0.00%, p=0.006%) in the DRF group. Contraindications for epidural analgesia (spine fractures or coagulopathy) were distributed evenly between the three groups (Table 2).
Table 1.
Characteristics and outcome of patients with rib fractures.
| DRF (n=39) | NDRF (n=77) | CRF (n=129) | P value DRF vs NDRF | P value DRF vs CRF | P Value NDRF vs CRF | |
|---|---|---|---|---|---|---|
| Number of rib fractures (Mean ± SD) | 1.7 ± 1.2 | 2.4 ± 2.3 | 6.8 ± 3.6 | 0.0634 | <0.0001 | <0.0001 |
| Age (Mean years ± SD) | 60.3 ± 22.1 | 53.7 ± 21.9 | 61.3 ± 17.9 | 0.1295 | 0.7826 | 0.0077 |
| Female, n (%) | 11 (28.2) | 25 (32.5) | 40 (31.0) | na | na | na |
| ISS, Median (IQR range) | 9 (5, 13) | 13 (9, 17) | 17 (12, 25) | 0.0311 | <0.0001 | 0.0003 |
| Number of comorbidities Median (IQR range) |
1 (0, 2) | 1 (0, 1) | 1 (0, 2) | na | na | na |
| Number of complications, n (%) | 1 (2.6) | 7 (9.1) | 7 (5.4) | na | na | na |
| Mortality, n (%) | 0 (0) | 1 (1.3) | 6 (4.7) | na | na | na |
DRF, displaced rib fracture; NDRF, non-displace rib fracture; CRF – combined displaced and nondisplaced rib fracture; ISS, Injury Severity Score; IQR, interquartile range; na, not applicable, as ANOVA was not significant (>0.05).
Table 2.
Usage of pain medications and paradigms in patients with rib fractures.
| Total (n=245) | DRF (n=39) | NDRF (n=77) | CRF (n=129) | P value DRF vs NDRF | P value DRF vs CRF | P Value NDRF vs CRF | |
|---|---|---|---|---|---|---|---|
| Any PO/IV opioid, n (%) | 224 (92.2) | 37 (97.4) | 67 (87.0) | 120 (93.8) | 0.0757 | 0.3871 | 0.0988 |
| Morphine equianalgesic dose (mg), median (IQR)* | 135.8 (50.83–440.0) | 53.3 (27.5, 178) | 105.0 (370.7, 5965) | 251.2 (80.00, 600.7) | 0.0416 | <0.0001 | 0.0045 |
| Patient-Controlled Analgesia, n (%) | 53 (21.8) | 5 (13.2) | 8 (10.4) | 40 (31.3) | 0.6592 | 0.0276 | 0.0006 |
| Epidural analgesia, n (%) | 15 (6.2) | 3 (7.9) | 0 (0) | 12 (9.4) | 0.0125 | 0.7799 | 0.0056 |
| Contraindications for epidural analgesia (spine fracture/ coagulopathy), n (%) | 93 (38.0) | 11 (28.2) | 27 (35.1) | 55 (42.6) | 0.4571 | 0.1059 | 0.2828 |
DRF, displaced rib fracture; NDRF, non-displace rib fracture; CRF – combined displaced and nondisplaced rib fracture; PO, per os; IV, intravenous; IQR, interquartile range;
among patients receiving opioids.
Pain management
Opioids were given to 92% of RF patients with no percent difference among the studied groups (Table 2). Multiple medications were used, with intravenous hydromorphine most commonly given (Figure 2). Among patients prescribed opioids, those with CRF had the highest MED. In addition, patients in the latter group also more commonly used PCA while patients with DRF only more frequently received epidural analgesia (Table 2). Univariate linear regression analysis showed a significant association between opioid dose and RF displacement (R2 =0.124; p<0.0001, Figure 3A) and the number of RF (R2 =0.198, p<0.0001, Figure 3B).
Figure 2.
Pain medication usage in patients with RF (n=245).
Figure 3.
Linear regression showing correlation between opioid usage and total rib fracture displacement (A) or number of rib fractures (B).
For the multivariable models, stepwise linear regression found that decreased age, increased ISS, and the presence of chest tube were significant predictors of MED and were retained in both models (Table 3). The adjusted models showed that a 5 mm increase in total displacement predicted 6.3% increase in mean total opioid dose (p=0.0035), and that every additional RF predicted an 11.2% increase in a mean total opioid dose (p=0.0001, Table 3). Predictors of epidural and PCA analgesia were not modeled due to the small number of patients in who were treated with these modalities.
Table 3.
Predictors of pain medication usage expressed as log morphine equianalgesic dose.
| Variable | Parameter Estimate | Standard Error | p-value |
|---|---|---|---|
| Model A | |||
| Intercept | 1.93060 | 0.14670 | <.0001 |
| Total displacement (mm) | 0.00531 | 0.00180 | 0.0035 |
| Age | −0.00610 | 0.00198 | 0.0023 |
| Chest tube | 0.28492 | 0.11041 | 0.0105 |
| Injury Severity Score | 0.02974 | 0.00399 | <.0001 |
| Model B | |||
| Intercept | 1.91546 | 0.14468 | <.0001 |
| Number of rib fractures | 0.04599 | 0.01185 | 0.0001 |
| Age | −0.00723 | 0.01986 | 0.0003 |
| Chest tube | 0.21412 | 0.11258 | 0.0585 |
| Injury Severity Score | 0.02673 | 0.00409 | <.0001 |
DISCUSSION
The present study identified two novel clinically relevant findings. First, it shows that the magnitude of RF displacement is associated and is a predictor of opioid requirement in patients with blunt RF. Specifically, it demonstrates that every 5mm displacement increases mean opioid dose by 6.3%. Second, this work quantifies for the first time the already established relationship between the number of fractured ribs and chest wall pain, so that each RF increases opioid use by 11.2%. The discussion below addresses these new findings.
It is conceivable that RF displacement is a manifestation of a more severe chest wall injury, and therefore, an association between RF displacement and clinical outcome does exist. Indeed, multiple studies emphasized this notion and some went further to suggest that RF displacement could be used as an indication for surgical fixation. For example, a recent review of the surgical approach to RF emphasized displacement, defined as overlapping of the fracture ends, as an indication for rib plating (15). Another study, which surveyed trauma, thoracic and orthopedic surgeons, showed that 27% of responders view displacement (one or more rib width) as an indication for surgical repair of RF (16). Also, a survey of patients who underwent rib plating mentioned RF displacement as an indication for surgical intervention (17). Furthermore, a case-control study to assess the effect of surgical treatment of RF also viewed “significantly displaced fractures” as an indication for surgery (18). Lastly, a retrospective study of 46 patients with surgical rib fixation used rib displacement as an indication for surgery (19). It should be noted, however, that none of the above cited studies investigated the association between RF displacement and pain management and outcome. This lack of information served as the nidus for the present study.
The results presented herein indicate that the degree of RF displacement incrementally increases pain medication requirement. There are several possible explanations for this finding. First, RF displacement may reflect a higher amount of energy absorbed by the chest wall during the initial impact, which inevitably leads to increased tissue damage and inflammation. Second, displaced RF inherently lead to enhanced movements of the RF edges with subsequent augmented inflammation and slower healing.
The present study agrees and confirms previous reports indicating that the number of RF predicts the level of chest wall pain. The Eastern Association for the Surgery of Trauma (EAST) recommends in its practice guidelines to use the number of RF as an indication for epidural analgesia (5). Moreover, a recently published secondary analysis of a randomized trial directly showed that each additional RF was associated with worse SF-36 Physical Functioning and Bodily Pain scores (20). However, none of the reports published so far quantified the relationship between the number of fractured ribs and pain management. Thus, our finding that each RF increases opioid use by 11.2% is novel. It is also clinically relevant, as it allows the physician caring for RF patients to anticipate and plan their pain medication requirements.
It should be noted that the total RF displacement was collinear with the number of RF, and therefore, could not have been included in the same multivariable regression model. Both parameters were identified as significant predictors of opioid requirements, with the number of RF being a stronger predictor than displacement. What does this mean for the clinician? While counting RF is much simpler than measuring RF displacement, in two patients with the same number of RF, but with or without RF displacement, the patient with displacement will likely require more pain medication, and this requirement will be associated with the total magnitude of RF displacement.
We also examined possible associations between opioid requirements and the level and position of RF. However, this analysis was limited since almost all patients had RF at multiple locations and levels − 86% of patients had mid level (ribs 3–9) and 74% had both mid and upper level RF. In addition, 50% of patients had both anterolateral and posterior RF. A preliminary analysis in patients with only posterior vs. only anterolateral RF found no difference in opioid requirements between these two groups.
Our findings support the consideration of DRF as an indication for more aggressive pain management such as PCA or epidural analgesia. This is because patients with at least one RF had a higher utilization of PCA, and only patients in this group were treated with epidural analgesia. In addition, our data show that patients with CRF require higher doses of analgesics compared with patients who have only DRF or NDRF, conceivably due to the higher number of fractures per patient in that group (6.8 vs. 2.4 and 1.7). As none of our patients underwent RF plating, we cannot comment on associations among the study outcomes or RF displacement and RF fixation.
As described in the manuscript, both regression models were adjusted for ISS, presence of chest tube, and age. As ISS reflects the presence of RF as well as associated intra- and extra-thoracic injuries, it is not surprising that it was found to correlate with MED. Another expected association was the correlation between MED and chest tube placement, which is a painful procedure. The observed association between decreased MED and increasing age is likely derived from the reluctance to use opioids in the elderly to minimize delirium in this group.
Interestingly, DRF patients required less pain medications than NDRF patients. It is possible that this observation emanates from the fewer fractures in the DRF group (1.7 compared to 2.4). In that respect, it should be noted that the likelihood of having both displaced and non-displaced fractures and a higher opioid requirement increased with the number of RF.
Our study has few limitations. First, it is a retrospective review of a relatively small cohort of patients. Second, a single person measured all RF displacements. Although the intra-rater reliability was high, there is still a small risk for an operator bias. Third, the efficacy of pain management could not be assessed, as pain scores were not recorded in the database. Fourth, the relative small number of patients in the present study did not allow us to study the effect of compromised mental status (traumatic brain injury), additional injuries (long bone and/or pelvic fractures) or surgeries (laparotomy, thoracotomy) on the quantity and selection of pain management. Fifth, we had only scattered information regarding prior history of narcotics use of the studied patients, and thus, were unable to correlate it with inpatient narcotic requirements.
CONCLUSION
The magnitude of RF displacement and the number of RF were identified as predictors of opioid requirements, and the relationship between these parameters was quantified. Therefore, knowing the degree of RF displacement as well as the number of fractured ribs may assist the trauma surgeon in anticipating patients with blunt RF who might have higher analgesic requirements.
Acknowledgments
This manuscript was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, Award UL1TR001064.
This research paper was made possible through the help of Frank Massaro, PharmD, Assistant Director of Pharmacy, Tufts Medical Center, Boston, MA.
Footnotes
There are no conflicts of interest to declare
The manuscript was presented as a poster at the 74th annual meeting of American Association for the Surgery of Trauma, September 9–12, 2015, in Las Vegas, Nevada
AUTHOR CONTRIBUTION
N.B., M.A., H.A. conducted the literature search. N.B., J.B., S.A., and R.R. designed this study. N.B., M.A., H.A., S.A., and S.H. performed data collection. N.B. and J.B. analyzed the data. N.B., J.B., and R.R. contributed to data interpretation. N.B. wrote the manuscript, which J.B., S.A., and R.R. critically revised.
Contributor Information
Nikolay Bugaev, Email: nbugaev@tuftsmedicalcenter.org, Division of Trauma & Acute Care Surgery, Tufts Medical Center, Tufts University School of Medicine. Boston, MA.
Janis L. Breeze, Email: jbreeze@tuftsmedicalcenter.org, Tufts Clinical and Translational Science Institute, Tufts University, and Institute for Clinical Research and Health Policy Studies, Tufts Medical Center, Boston MA.
Majid Alhazmi, Email: dr.majed.m.h@hotmail.comis, Division of Trauma & Acute Care Surgery, Tufts Medical Center, Tufts University School of Medicine, Boston, MA.
Hassan S. Anbari, Email: hassan_suheil@yahoo.com, Division of Trauma & Acute Care Surgery, Tufts Medical Center, Tufts University School of Medicine, Boston, MA.
Sandra S. Arabian, Email: sarabian@tuftsmedicalcenter.org, Division of Trauma & Acute Care Surgery, Tufts Medical Center, Tufts University School of Medicine. Boston, MA.
Sharon Holewinski, Email: sholewinski@tuftsmedicalcenter.org, Division of Trauma & Acute Care Surgery, Tufts Medical Center, Tufts University School of Medicine. Boston, MA.
Reuven Rabinovici, Email: rrabinovici@tuftsmedicalcenter.org, Division of Trauma & Acute Care Surgery, Tufts Medical Center, Tufts University School of Medicine. Boston, MA.
References
- 1.Battle CE, Hutchings H, Evans PA. Risk factors that predict mortality in patients with blunt chest wall trauma: a systematic review and meta-analysis. Injury. 2012;43(1):8–17. doi: 10.1016/j.injury.2011.01.004. [DOI] [PubMed] [Google Scholar]
- 2.Jones KM, Reed RL, 2nd, Luchette FA. The ribs or not the ribs: which influences mortality? Am J Surg. 2011;202(5):598–604. doi: 10.1016/j.amjsurg.2010.09.029. [DOI] [PubMed] [Google Scholar]
- 3.Battle CE, Hutchings H, James K, Evans PA. The risk factors for the development of complications during the recovery phase following blunt chest wall trauma: a retrospective study. Injury. 2013;44(9):1171–6. doi: 10.1016/j.injury.2012.05.019. [DOI] [PubMed] [Google Scholar]
- 4.Gordy S1, Fabricant L1, Ham B1, Mullins R1, Mayberry J2. The contribution of rib fractures to chronic pain and disability. Am J Surg. 2014;207(5):659–63. doi: 10.1016/j.amjsurg.2013.12.012. [DOI] [PubMed] [Google Scholar]
- 5.Simon BJ, Cushman J, Barraco R, Lane V, Luchette FA, Miglietta M, Roccaforte DJ, Spector R EAST Practice Management Guidelines Work Group. Pain management guidelines for blunt thoracic trauma. J Trauma. 2005;59(5):1256–67. doi: 10.1097/01.ta.0000178063.77946.f5. [DOI] [PubMed] [Google Scholar]
- 6.Unsworth A, Curtis K, Asha SE. Treatments for blunt chest trauma and their impact on patient outcomes and health service delivery. Scand J Trauma Resusc Emerg Med. 2015;23:17. doi: 10.1186/s13049-015-0091-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bayouth L, Safcsak K, Cheatham ML, Smith CP, Birrer KL, Promes JT. Early intravenous ibuprofen decreases narcotic requirement and length of stay after traumatic rib fracture. Am Surg. 2013;79(11):1207–12. [PubMed] [Google Scholar]
- 8.Yang Y, Young JB, Schermer CR, Utter GH. Use of ketorolac is associated with decreased pneumonia following rib fractures. Am J Surg. 2014;207(4):566–72. doi: 10.1016/j.amjsurg.2013.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Truitt MS, Murry J, Amos J, Lorenzo M, Mangram A, Dunn E, Moore EE. Continuous intercostal nerve blockade for rib fractures: ready for primetime? J Trauma. 2011;71(6):1548–52. doi: 10.1097/TA.0b013e31823c96e0. [DOI] [PubMed] [Google Scholar]
- 10.Menditto VG, Gabrielli B, Marcosignori M, Screpante F, Pupita G, Polonara S, Salvi A, Raggetti GM, Pomponio G. A management of blunt thoracic trauma in an emergency department observation unit: pre-post observational study. J Trauma Acute Care Surg. 2012;72(1):222–8. doi: 10.1097/TA.0b013e3182140cad. [DOI] [PubMed] [Google Scholar]
- 11.de Moya M, Bramos T, Agarwal S, Fikry K, Janjua S, King DR, Alam HB, Velmahos GC, Burke P, Tobler W. Pain as an indication for rib fixation: a bi-institutional pilot study. J Trauma. 2011;71(6):1750–4. doi: 10.1097/TA.0b013e31823c85e9. [DOI] [PubMed] [Google Scholar]
- 12.Khandelwal G, Mathur RK, Shukla S, Maheshwari A. A prospective single center study to assess the impact of surgical stabilization in patients with rib fracture. Int J Surg. 2011;9(6):478–81. doi: 10.1016/j.ijsu.2011.06.003. [DOI] [PubMed] [Google Scholar]
- 13.Bugaev N, Breeze JL, Alhazmi M, Anbari HS, Arabian SS, Rabinovici R. Displacement Patterns of Blunt Rib Fractures and Their Relationship to Thoracic Co-Injuries: Minimal Displacements Count. Am Surg. 2016;82(3):199–206. [PubMed] [Google Scholar]
- 14.American Pain Society (APS) Principles of Analgesic Use in the Treatment of Acute Pain and Cancer Pain. 6. Glenview, IL 60025: 2008. [Google Scholar]
- 15.Nirula R, Mayberry JC. Rib fracture fixation: controversies and technical challenges. Am Surg. 2010;76(8):793–802. [PubMed] [Google Scholar]
- 16.Mayberry JC, Ham LB, Schipper PH, Ellis TJ, Mullilns RJ. Surveyed opinion of American trauma, orthopedic, and thoracic surgeons on rib and sternal fracture repair. J Trauma. 2009;66(3):875–9. doi: 10.1097/TA.0b013e318190c3d3. [DOI] [PubMed] [Google Scholar]
- 17.Majercik S, Cannon Q, Granger SR, Vanboerum DH, White TW. Long-term patient outcomes after surgical stabilization of rib fractures. Am J Surg. 2014;208(1):88–92. doi: 10.1016/j.amjsurg.2013.08.051. [DOI] [PubMed] [Google Scholar]
- 18.Nirula R, Allen B, Layman R, Falimirski ME, Somberg LB. Rib fracture stabilization in patients sustaining blunt chest injury. Am Surg. 2006;72(4):307–9. doi: 10.1177/000313480607200405. [DOI] [PubMed] [Google Scholar]
- 19.Mayberry JC1, Kroeker AD, Ham LB, Mullins RJ, Trunkey DD. Long-term morbidity, pain, and disability after repair of severe chest wall injuries. Am Surg. 2009 May;75(5):389–94. [PubMed] [Google Scholar]
- 20.Dhillon TS1, Galante JM, Salcedo ES, Utter GH. Characteristics of chest wall injuries that predict postrecovery pulmonary symptoms: A secondary analysis of data from a randomized trial. J Trauma Acute Care Surg. 2015;79(2):179–87. doi: 10.1097/TA.0000000000000718. [DOI] [PubMed] [Google Scholar]