Abstract
Objective
To evaluate the efficacy of behavioral counseling combined with technology-based self-monitoring for sodium restriction in hemodialysis (HD) patients.
Design
Randomized clinical trial.
Setting
Outpatient HD centers.
Subjects
English literate adults undergoing in-center intermittent HD for at least 3 months.
Interventions
Over a 16-week period, both the Intervention and the Attention Control groups were shown six educational modules on the HD diet. The Intervention group also received Social Cognitive Theory-based behavioral counseling and monitored their diets daily using hand-held computers.
Main Outcome Measures
Average daily interdialytic weight gain (IDWGA) was calculated for every HD treatment over the observation period by subtracting the post-dialysis weight at the previous treatment time (t-1) from the pre-dialysis weight at the current treatment time (t), dividing by the number of days between treatments. Three 24-hour dietary recalls were obtained at baseline, 8 weeks, and 16 weeks, and evaluated using the Nutrient Data System for Research.
Results
179 participants were randomized and 160 (89.4%) completed final measurements. IDWGA did not differ significantly by treatment group at any time point considered (p>0.79 for each). A significant differential change in dietary sodium intake observed at 8 weeks (−372 mg/day; p=0.05) was not sustained at 16 weeks (−191 mg/day; p=0.32).
Conclusion
The BalanceWise Study intervention appeared to be feasible and acceptable to HD patients, although IDWGA was unchanged and the desired behavioral changes observed at 8 weeks were not sustained. Unmeasured factors may have contributed to the mixed findings, and further research is needed to identify the appropriate patients for such interventions.
Keywords: behavioral research, controlled clinical trials, computers, handheld, salt
INTRODUCTION
Mortality among hemodialysis (HD) patients remains high in the United States, with cardiovascular causes accounting for more than one-third of mortality in incident and prevalent end-stage renal disease (ESRD) patients.1 One factor contributing to poor cardiovascular outcomes is chronic volume overload2,3, which is a function of excess fluid intake in relation to residual renal function and fluid removal with dialysis treatments. Interdialytic weight gain (IDWG) is commonly used as an indirect measure of fluid intake in HD patients. Differences in IDWG as small as 0.51 kg (less than 2¼ cups of fluid) have been shown to be independently associated with uncontrolled hypertension in HD patients4, which contributes to left ventricular hypertrophy.5 According to the United States Renal Data System (USRDS), all-cause mortality is highest on the first treatment day of the week after the 2-day weekend hiatus from HD (when fluid volume for the week is likely at its highest).6 Because rapid fluid removal is associated with cardiovascular morbidity and mortality7, the focus must be on longer HD treatments (often not well received by patients) and on decreasing IDWGs.
Dietary sodium intake is a modifiable behavior that is associated with higher IDWG8 and all-cause mortality in HD patients. Although sodium restriction is recommended routinely for HD patients10, the pervasive use of sodium chloride (as salt) by food manufacturers and foodservice operations and increased reliance on these foods makes sodium restriction difficult.11–14 Despite long-standing recommendations to limit sodium intakes in the U.S., average dietary sodium intake in the general population has remained stable in amounts approximately 1.5 times the Tolerable Upper Intake Level (UL).15,16 Adopting a lower sodium diet may be even more difficult for HD patients, as they may experience greater barriers to dietary change than does the general population (e.g., ill health, a time consuming treatment regimen, numerous other dietary restrictions). The purpose of the BalanceWise Study was to evaluate the efficacy of a technology-supported behavioral intervention for reducing IDWGs and dietary sodium intake in patients undergoing intermittent HD. Our hypotheses were that, compared to the Attention Control group, the Intervention group would demonstrate a lower average daily IDWG (IDWGA) and lower dietary sodium intake at 8 weeks and that these changes would be sustained at 16 weeks.
METHODS
Design
The BalanceWise Study was a randomized clinical trial of maintenance HD patients recruited from 3 dialysis chains comprising 17 dialysis units in Western Pennsylvania (DaVita HealthCare Partners Inc., Dialysis Clinic Inc., and Fresenius Medical Care North America). Within dialysis units, participants were randomized to either an Intervention of 16 weeks duration or an Attention Control. The intervention consisted of behavioral counseling paired with dietary self-monitoring using a hand held computer, i.e., a personal digital assistant (PDA). Study dietitians delivered the intervention or attention control materials during regularly-scheduled HD treatments. This study was approved by the Institutional Review Board of the University of Pittsburgh.
Study population
To be eligible, participants had to be community-dwelling adults over 18 years of age who had been undergoing intermittent in-center HD for at least 3 months (to permit initial nutritional stabilization). Excluded from the study were individuals who could not read, write, or speak English (because the PDA software was available only in English); could not see the PDA or use a stylus to make selections from the PDA screen; had overt dementia; planned to move out of the area or change dialysis centers within the next 16 weeks; were scheduled for a living donor transplant within the study period; were deemed by dialysis center staff to have a life expectancy of less than 12 months (to avoid over-burdening such participants); or were institutionalized (e.g., in a nursing home, personal care facility, or incarcerated, as such participants would have limited control over their dietary intake). We also excluded those who were unwilling to speak 1–2 times/week with a study dietitian or record their food consumption during the 16-week study period.
The target sample size of 200 (100 in the Intervention and 100 in the Attention Control group) was based on pilot data showing an estimated average reduction of 0.06 kg in IDWGA at 16 weeks for Intervention participants and an increase of 0.18 kg for Attention Control participants, with corresponding within-participant standard deviations of 0.53 kg and 0.57 kg. Based on these estimates, the study provided 80% power to detect a differential change from baseline of 0.24 kg at 16 weeks in IDWGA between participants in the Intervention and Attention Control groups based on a two-sided 0.05-level t-test, and 80% power to detect treatment differences of approximately 0.5 standard deviations in other outcomes. Sample size estimates were computed in Stata. Our target sample size allowed for an expected 17% loss of participants to death, transplant, relocation, switch to peritoneal dialysis, or study withdrawal.
Recruitment and randomization procedures
Two different participant recruitment strategies were deemed acceptable by the corporate dialysis chains cooperating in this study. In two of the three chains, a dialysis center nurse, social worker, or dietitian performed a pre-screening evaluation of all patients and, from those who remained eligible, secured a Health Insurance Portability and Accountability Act (HIPAA) release. In the third dialysis chain, patients interested in discussing the study referred themselves to the study staff person at the conclusion of their HD treatment, at which time pre-screening was performed and a HIPAA waiver obtained. After obtaining signed informed consent, study staff verified eligibility, ascertained that participants could use the PDA, and collected baseline measurements. Within each dialysis center, participants were then randomized using a permuted block algorithm developed by the study statistician. To assure consistency in protocol implementation between participating dialysis units, dialysis unit staff were not involved in any intervention, attention control, or measurement activities. The intervention and attention control, activities were conducted by study staff as an addition to, but not as a replacement for, standard care.
Intervention and Attention Control Group Activities
Both Groups
During scheduled HD appointments, a study dietitian delivered six Dietary Educational Modules to participants in both groups using Microsoft PowerPoint®, shown on a laptop computer. Participants were invited to ask questions or obtain further clarification at this time. These modules included: (1) Overview of the HD diet, (2) Sodium and fluid restrictions, (3) Strategies for maintaining adequate calorie intake, (4) Strategies for maintaining adequate protein intake, (5) Phosphorus restrictions, and (6) Potassium restrictions. The purpose of these educational modules was to assure comparability regarding participant knowledge of different aspects of the standard HD dietary regimen in both study groups, provide some (albeit not equivalent) attention to the Attention Control group, and serve as a retention incentive.
Intervention Group
Social cognitive theory (SCT)-based behavioral counseling was delivered to Intervention group participants via 1-to-1, face-to-face meetings with a study dietitian during regularly-scheduled HD treatments.17,18 Counseling was delivered twice/week during the first 8 weeks, weekly in weeks 9–12, and every other week during weeks 13–16. Counseling was guided by the 16 SCT-based modules described in Table 1. The counseling was focused on building a sense of self-efficacy (confidence) regarding adherence to the HD diet, particularly adherence to dietary sodium and fluid intake recommendations.
Table 1.
Social Cognitive Theory (SCT) modules used to guide BalanceWise intervention counseling sessions
| SCT Modules | Content |
|---|---|
| Goals and Values | Participant’s goals for life, their treatment regimen and the study. Participant’s readiness and confidence in their ability to make dietary changes. |
| Goals for Health and Diet | Elicit participant’s perspectives on the importance of their diet for maintaining health while on hemodialysis. |
| Sodium | Review of PDA entries to identify dietary patterns/discrepancies from ideal behavior. |
| Monitoring | Elicit participant statements regarding what they have discovered from self-monitoring, and any discrepancies noted from the recommended dietary regimen. Discussion of the importance of self-monitoring to nutritional self-awareness, vigilance. |
| Self Assessment and Setting Goals | Guide participant in reviewing PDA targets and graphs for identifiable patterns of nonadherence to dietary sodium targets. Assist the participant in setting incremental, easily achievable goals that are specific and measureable. |
| Calories | Counsel participants on the importance of adequate calorie intake. Guide participant in reviewing PDA targets and graphs to assure that they are consuming sufficient calories. |
| Self Reward | Introduce the notion of self-reward as useful for reinforcing newly acquired behaviors. Elicit participant statements about successes they have experienced as a result of the study (e.g. greater knowledge, control, confidence regarding their diet; reduced IDWGs; reduced post-dialytic symptoms). Emphasize the importance of the participant taking credit for their successes. Discussion of “slips” and what can be learned from them. |
| Problem Solving 1 | Elicit from participants high risk situations that could increase the likelihood of nonadherence (e.g. holidays, social events, cravings, dialysis treatments, family or friends). Explore how these high risk situations contribute to nonadherence. |
| Problem Solving 2 | Explore possible solutions to high risk situations and assist the participant in setting anticipatory goals. |
| Stimulus Control | Introduce the notion of cues to habitual behaviors, and minimizing or removing cues to break the chain of events leading to habitual behaviors. Explore the participant’s cues and assist them in setting goals to break habitual behaviors. |
| Social Support | Elicit from participants their perspectives on the extent to which family and friends are aware and supportive of their dietary regimen. Explore with the participant actions they can take to enhance support from family and friends. |
| Lapses and Relapses | Elicit from participants their perspectives on situations that could result in a lapse or relapse (e.g. illness, hospitalization, major life changes). Discuss the importance of making new goals. Engage the participant in anticipatory problem solving. |
| Protein | Counsel participants on the importance of adequate protein intake. Guide participant in reviewing PDA targets and graphs to assure that they are consuming sufficient protein. |
| Putting it all together | Review the major components of the intervention: determining values and goals, self-monitoring, identifying problems, setting goals, self-reward, and problem solving (including attention to high risk situations, cues, social support, lapses and relapses). |
| Potassium (optional) | When monthly lab values indicate hyperkalemia, guide participant in reviewing the PDA record to identify high potassium foods and problem solve dietary solutions. |
| Phosphorus (optional) | When monthly lab values indicate hyperphosphatemia, guide participant in reviewing the PDA record to identify high phosphorus foods and problem solve dietary solutions. Refer to renal dietitian to discuss adherence to phosphorus binders. |
A sense of self-efficacy was promoted through use of mastery experiences, verbal persuasion, and physiological states. Mastery experiences included emphasizing past successes; setting incremental, easily achievable goals; providing positive feedback on goal achievement; identifying modifiable barriers to healthy behavior; and developing participant expertise in both the regimen and in problem solving around barriers to adherence. Verbal persuasion included providing examples of the participant’s previous successes to demonstrate their capability (e.g., “You stayed under your sodium ceiling and your IDWGs were lower this week. You can do it again.”). The sessions included discussion of the physiologic benefits participants experienced as a result of reduced IDWGs (e.g., reduced malaise, nausea, diaphoresis, chest pain, cramping, and hypotension during dialysis; reduced fatigue and thirst between dialysis sessions).
In addition to behavioral counseling, participants in the Intervention group were engaged in technology-based self-monitoring. Participants recorded their meals into the BalanceLog® (Microlife; Golden, CO) software program on their PDAs. Each participant received 1:1 training on using the PDA from a study dietitian, and retrained as needed. The PDA record was reviewed by the study dietitian with each intervention contact. BalanceLog® was used by the participants to track the sodium content of their meals and by the study dietitians to provide targeted counseling. PDAs were used to enhance mastery by: (1) minimizing the information burden of vigilance to dietary sodium intake, (2) providing graphical, meal-by-meal feedback regarding achievement of dietary goals, (3) permitting straightforward integration of nutritional information, and (4) allowing participants to make real-time connections between sodium, IDWGs, and symptoms. Because HD patients are at risk of malnutrition, the PDA records also were used by participants and study dietitians to assure that sodium restrictions did not result inadvertently in inadequate calorie or protein intake.
BalanceLog® was individually programmed for the nutritional requirements of each participant, per the National Kidney Foundation guidelines for calories and protein. The calorie goal was 30 kcals/kg of body weight/day for those >60 years of age, and 35 kcal/kg/day for those <60 years.19 The protein goal was 1.2 g/kg of body weight/day.20 We initially programmed BalanceLog® for a sodium ceiling of 2,400 mg/day, and reduced this to 2,000 mg/day once the participant was able to achieve 2,400 mg/day on most days of the week. BalanceLog® was programmed to permit the entry of 3 meals and 1 snack each day. This software is described in detail elsewhere.21 Intervention participants were permitted to keep their PDAs at the conclusion of the trial. A tracking system was used to record the frequency of, content of, and time devoted to intervention activities.
Attention Control Group
To prevent a differential loss of participants, at the conclusion of the trial Attention Control group participants were provided PDAs programmed with their nutritional requirements and offered training in the use of the PDAs.
Study measures
Participants were weighed prior to and following each hemodialysis treatment. Pre-and post-dialysis weights were abstracted from the participant’s dialysis flow sheet for the HD treatments for 17 weeks (1 week pre-randomization through 16 weeks post-randomization). Because the number of days between HD treatments varied, we calculated IDWGA by subtracting the post-dialysis weight at the previous treatment time (t-1) from the predialysis weight at the current treatment time (t), and divided by the number of days between treatments.
Dietary sodium intake was assessed via 24-hour dietary recalls at baseline (i.e., pre-intervention), 8 weeks (i.e., at the conclusion of the intensive phase of the intervention), and 16 weeks.1 Preliminary data suggested that dietary intake of HD patients varied by day of the week22, so three 24-hour recalls were collected on each participant including 1-dialysis weekday, 1 non-dialysis weekday, and 1 non-dialysis weekend day. The first two recalls were obtained face-to-face (with guidance in portion size provided). Given the time-consuming nature of inpatient dialysis treatments, to minimize burden associated with extra travel to the dialysis unit (or other central data collection site) on nondialysis days, the third recall was obtained via telephone. The potential for desirability response set bias was mitigated by collecting unscheduled dietary recalls during a 2-week period surrounding each measurement time point. Data were entered into the Nutrient Data System for Research.23
Socio-demographic and physical characteristics of participants likely to be associated with weight gains between HD treatments and/or dietary intake included age, income, gender, weight, body mass index, Kt/V, serum albumin, and etiology of ESRD. These data were collected at baseline.
Statistical Analyses
For descriptive purposes, IDWGA values at baseline, 8, 12, and 16 weeks were estimated using the mean of the IDWGAs for the 1-week period prior to the specified measurement time point (e.g., baseline IDWGA was the mean of the IDWGAs from the 3 dialysis treatments performed during the 1-week period prior to randomization; Week 8 IDWGA was the mean of the IDWGAs during Week 7). We modeled the time-specific IDWGA values across the 17 weeks using a random intercept linear model with continuous time, a dummy variable for treatment, and a treatment by time interaction; this interaction term is the treatment effect of interest. We assessed linearity within treatment groups over time using linear splines, with knots defined at the time points when the intensity of the intervention contact changed (i.e., 8 and 12 weeks). The primary treatment comparison was an unadjusted intent-to-treat analysis.
Adherence of Intervention group participants to the self-monitoring protocol was evaluated in terms of the number of meal or snack entries made into the PDA each day. Dietary sodium was averaged across the three recalls at each measurement time point. We modeled time-specific mean dietary sodium intake values using a random intercept linear model with dummy variables for treatment group, time, and the treatment by time interaction. To account for missing sodium intake data, we adjusted for predictors of missing dietary recalls identified in preliminary analyses; the resultant random effects model provides unbiased estimates of intervention effects assuming a “missing at random” mechanism. We used appropriate linear combinations of the parameters to compare time-specific means and changes from baseline between the Intervention and Attention Control groups. Because preliminary analyses indicated an extremely high baseline dietary sodium intake for one Intervention group participant (20,056 mg/day) with no follow-up values, the modeling of dietary sodium was repeated excluding this participant as part of a sensitivity analysis.
RESULTS
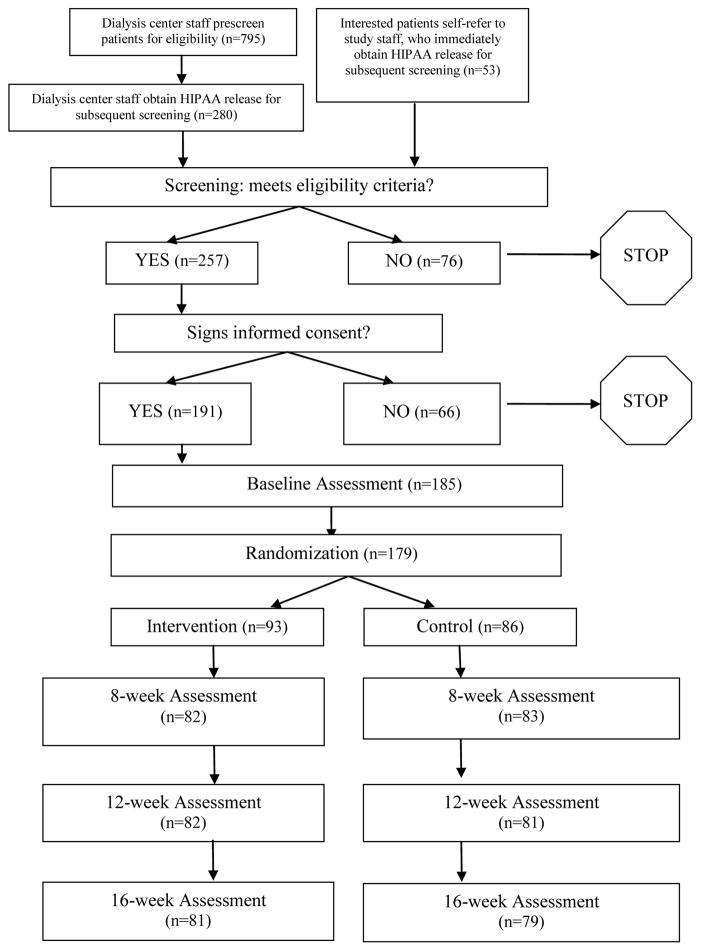
Participants were recruited during September 2009 through September 2012. Of the 848 HD patients available, 257 were eligible, 191 consented, 185 provided baseline data, 179 were randomized, and 160 (89.4%) completed the final 16-week measurement assessment. The CONSORT diagram is shown in Figure 1. The primary reasons for study exclusion for those patients who signed the HIPAA release and participated in baseline screening were: unwillingness to record diet, difficulty reading a newspaper, initiation of dialysis within the past 3 months, and unwillingness to speak with a study dietitian 1–2 times per week (Table 2).
Figure 1.
CONSORT diagram for the BalanceWise Study (September 2009 – April 2013)
Table 2.
Reasons for BalanceWise Study ineligibility identified among potential participants at the baseline assessment
| Overall (n=76)* | ||
|---|---|---|
| n | % | |
| Unwilling to record diet | 23 | 30.3 |
| Difficulty reading a newspaper (visual or literacy problems) | 22 | 28.9 |
| Dialysis < 3 months | 14 | 18.4 |
| Unwilling to speak with study dietitian 1–2 times per week | 10 | 13.2 |
| Planning to relocate to a nonparticipating dialysis center | 3 | 3.9 |
| Scheduled to receive a living donor transplant | 1 | 1.3 |
| Unable to read or write in English | 1 | 1.3 |
| Lives in an institutional setting | 1 | 1.3 |
| Unable to turn-on or follow directions for using the PDA | 1 | 1.3 |
(Some participants met multiple exclusion criteria)
Eligible patients who enrolled in the study were middle-aged, African-American, male, unpartnered, and unemployed (Table 3). Most had at least a high school education. About one-fourth did not report annual household income; among those that did, more than half reported an annual household income of <$40,000. The median duration of ESRD was 33 months (interquartile range 16–68 months). The distribution of ESRD etiology was consistent with that reported in USRDS, although somewhat fewer BalanceWise Study participants had an etiology of diabetes (39.9%, compared to 44.2% nationally).24 Median Kt/V differed somewhat between the groups with Attention Control group participants being better dialyzed. Otherwise, no significant differences were observed in baseline characteristics of the Intervention and Attention Control group participants.
Table 3.
Baseline characteristics of BalanceWise Study participants
| Variable | Overall (n=178)* | Technology-Supported Behavioral Intervention (n=93) | Attention Control (n=85)* | p-value | |||
|---|---|---|---|---|---|---|---|
|
| |||||||
| n | % | n | % | n | % | ||
| Race | |||||||
| African Am | 110 | 61.8 | 59 | 63.4 | 51 | 60.0 | 0.82 |
| White | 66 | 37.1 | 33 | 35.5 | 33 | 38.8 | |
| Unknown | 2 | 1.2 | 1 | 1.1 | 1 | 1.2 | |
|
| |||||||
| Latino or Hispanic | 4 | 2.3 | 2 | 2.2 | 1 | 2.4 | >0.99 |
|
| |||||||
| Male gender | 101 | 56.7 | 57 | 61.3 | 44 | 51.8 | 0.20 |
|
| |||||||
| Married or partnered | 74 | 41.6 | 43 | 46.2 | 31 | 36.5 | 0.19 |
|
| |||||||
| Income | |||||||
| <$10,000 | 20 | 11.2 | 10 | 10.8 | 10 | 11.8 | 0.78 |
| $10,000–$19,999 | 33 | 18.5 | 18 | 19.4 | 15 | 17.6 | |
| $20,000–$29,999 | 23 | 12.9 | 11 | 11.8 | 12 | 14.1 | |
| $30,000–$39,999 | 18 | 10.1 | 8 | 8.6 | 10 | 11.8 | |
| $40,000–$49,999 | 11 | 6.2 | 6 | 6.5 | 5 | 5.9 | |
| $50,000–$74,999 | 17 | 9.6 | 6 | 6.5 | 11 | 12.9 | |
| >$75,000 | 3 | 1.7 | 2 | 2.2 | 1 | 1.2 | |
| Prefer not answer | 45 | 25.3 | 28 | 20.1 | 17 | 20.0 | |
| Don’t know | 8 | 4.5 | 4 | 4.3 | 4 | 4.7 | |
|
| |||||||
| Income meets needs | |||||||
| Yes | 55 | 31.1 | 26 | 28.3 | 29 | 34.1 | 0.44 |
| No | 114 | 64.4 | 63 | 68.5 | 51 | 60.0 | |
| Don’t know | 8 | 4.5 | 3 | 3.3 | 5 | 5.9 | |
|
| |||||||
| Employed | 24 | 13.5 | 12 | 12.9 | 12 | 14.1 | 0.82 |
|
| |||||||
| Etiology of ESRD | |||||||
| Diabetes | 74 | 39.9 | 37 | 39.8 | 34 | 40.0 | 0.07 |
| Hypertension | 49 | 27.5 | 19 | 20.4 | 30 | 35.5 | |
| Glomerular disease | 20 | 11.2 | 14 | 15.1 | 6 | 7.1 | |
| Polycystic disease | 6 | 3.4 | 5 | 5.4 | 1 | 1.2 | |
| Neoplasms/tumors | 13 | 7.3 | 9 | 9.7 | 4 | 4.7 | |
| Others | 19 | 10.7 | 9 | 9.7 | 10 | 11.8 | |
|
| |||||||
| Median | IQR | Median | IQR | Median | IQR | ||
|
| |||||||
| Age (years) | 60 | (51, 70) | 62 | (53, 71) | 60 | (50, 69) | 0.38 |
|
| |||||||
| Duration of ESRD (mo.) | 33 | (16, 68) | 33 | (14, 66) | 33 | (17, 71) | 0.60 |
|
| |||||||
| Education (years) | 12 | (12, 14) | 12 | (12, 14) | 12 | (12, 14) | 0.82 |
|
| |||||||
| BMI (kg/m2) | 28 | (24, 35) | 30 | (24, 36) | 28 | (24, 34) | 0.21 |
|
| |||||||
| Weight (kg) | 82 | (70,101) | 84 | (73,106) | 81 | (69, 93) | 0.16 |
|
| |||||||
| Albumin (g/dl) | 4.0 | (3.7,4.2) | 4.0 | (3.7,4.2) | 4.0 | (3.7,4.2) | 0.76 |
|
| |||||||
| Dialysis adequacy (Kt/V) | 1.5 | (1.4,1.7) | 1.5 | (1.4,1.7) | 1.6 | (1.5,1.8) | <0.001 |
ESRD, end-stage renal disease; IQR, interquartile range; BMI, body mass index
Differences between the Technology-Supported Behavioral Intervention Group and the Attention Control group were assessed using Chi-squared test for categorical variables and rank sum test for continuous variables.
One participant who did not complete the baseline demographic assessment was subsequently randomized to the Attention Control group (data not available in this table)
Of the 191 individuals enrolling, 31 (16.2%) did not complete the study. Three individuals withdrew prior to randomization, and 3 were excluded post hoc (1 for confusion, 1 was a family relative of another participant, and 1 due to residence in an institutional setting). Of those who continued beyond beyond randomization, 14 Intervention participants and 11 Attention Control participants did not complete the study for the following reasons: 10 withdrew voluntarily (6 intervention, 4 control); 5 discontinued the study due to illness; 3 died; 3 relocated to a nonparticipating dialysis center; 2 were admitted to an institutional setting; and 2 were transferred to home hemodialysis.
Of the 25 sessions scheduled over the 16-week intervention period, intervention participants attended an average of 18.8 sessions (SD=4.9). During the study, interventionists met with participants 1,753 times; the average duration of these sessions was 18.3 minutes (SD=10.0).2 Participants missed a total of 173 sessions. Reasons for missed sessions included hospitalization (n=61, 35.3%); skipped treatment (n=43, 24.9%); participant refusal (n=15, 8.7% due to illness or inconvenience); travel (i.e., dialyzed elsewhere; n=7, 4.1%); insufficient time (e.g., scheduling issues; n=4, 2.3%); or other (n=43, 25.9%). Average time devoted to orienting participants to the study and training them in the use of the PDA was 21.4 minutes (SD=9.8). Intervention participants entered an average of 1.6 meals per day (SD=1.1). Nine (9.7%) intervention participants had difficulty with using or were unwilling to use the PDA to record their diet and (consistent with an intent-to-treat approach) were offered the option of using a paper diary. PDA records were available for use in delivering targeted counseling for 1,098 (62.6%) of the intervention sessions. When no electronic or paper meal record was available, counseling targeted dietary behaviors addressed with prior intervention sessions.
Participants who did not complete all three dietary recalls (i.e., at baseline, 8, and 16 weeks) did not differ from those who did with respect to socioeconomic characteristics or duration of ESRD (p>0.20 for each; data not shown). There was some evidence that etiology of ESRD differed between those who did and did not complete all three dietary recalls (p=0.06); diabetes was the etiology in 20% of those who did not compared to 42% of those who did complete these assessments (data not shown).
Table 4 shows time-specific average IDWGA and dietary sodium intake values at baseline, 8 weeks, and 16 weeks, as well as changes in sodium intake between measurement time points. No treatment differences in time-specific IDWGAs were apparent in the random effects model (p>0.79; Table 4), and the linear spline analysis demonstrated no significant departure from linearity (p=0.18). However, compared to the Attention Control group, the change in reported dietary sodium intake was significantly lower in the Intervention group at 8 weeks (−371 mg/day, p =0.05), but not 16 weeks (−191 mg/day, p = 0.32), adjusted for diabetes status. The adjusted differential changes in sodium intake observed at 8 and 16 weeks were attenuated and no longer statistically significant when the Intervention participant who reported a very high baseline sodium intake was excluded (−317 mg/day; p=0.08 at 8 weeks and −136 mg/day; p=0.46 at 16 weeks).
Table 4.
Time-specific IDWGA and dietary sodium intake at baseline and weeks 8, 12, and 16, and time-specific change from baseline at 8 and 16 weeks.
| Outcome | Time | Technology-Supported Behavioral Intervention (n=93) | Attention Control (n=86) | ||||
|---|---|---|---|---|---|---|---|
| n* | Mean | SD | n* | Mean | SD | ||
| Time-specific IDWGA (kg/day) | Baseline | 88 | 1.2 | 0.6 | 80 | 1.2 | 0.5 |
| Week 8 | 81 | 1.1 | 0.6 | 80 | 1.2 | 0.5 | |
| Week 12 | 80 | 1.2 | 0.5 | 79 | 1.2 | 0.5 | |
| Week 16 | 78 | 1.1 | 0.6 | 79 | 1.2 | 0.5 | |
|
| |||||||
| Time-specific dietary sodium intake (mg/day) | Baseline | 92 | 2555 | 2090 | 85 | 2298 | 957 |
| Week 8 | 82 | 2316 | 931 | 80 | 2573 | 1139 | |
| Week 16 | 78 | 2371 | 1483 | 78 | 2447 | 1067 | |
|
| |||||||
| Time-specific change from baseline in sodium intake (mg/day) | Baseline to Week 8 | 82 | −71 | 1027 | 79 | 237 | 991 |
| Baseline to Week 16 | 78 | 59 | 1483 | 77 | 131 | 993 | |
IDWGA, average daily interdialytic weight gain
Dietary sodium time-specific sample means and changes from baseline were estimated from three 24-hour dietary recalls at baseline, week 8, and week 16.
Some participants (16 intervention and 11 Attention Control) have missing sodium data due to missing dietary recalls, and some have missing IDGWA data due to missing hemodialysis sessions.
DISCUSSION
In this report, we have presented the primary outcomes of the BalanceWise study. No differences were seen in IDWGAs, either between groups at each follow-up time or over time in within either group. However, the differential change in reported dietary sodium intake at 8 weeks provides some encouragement that the technology-supported behavioral intervention may impact behavior, at least in the short-term; this effect was not sustained at 16 weeks.
Despite recommendations to limit sodium intake, few intervention studies have evaluated the efficacy of dietary sodium restriction alone in patients on HD. Two abstracts8,25 and one commentary26 published in the late 1990s to early 2000s reported significant reductions in IDWG associated with a short-term (2 to 14 days) low-sodium diet in relatively small cohorts of HD patients (n = 30 to 35). More recently, Telini et al. (2013)27 randomized HD patients to a restricted salt diet or control groups, and despite significant differences in salt intake at 8 and 16 weeks, reported no within or between group differences in IDWG. Of interest, the group that reduced their salt intake showed significant reductions in several inflammatory markers at 8 and 16 weeks.
Several factors may have contributed to the null findings of the BalanceWise intervention on IDWGAs. The magnitude and variability of IDWGAs in BalanceWise were much lower than expected based on our pilot data. We initially considered restricting the sample to those with high IDWGs, but preliminary data suggested large within-subject variability and limited ability to identify those patients who could benefit most from such an intervention. IDWGs are influenced by other potential confounding factors, such as the dialysate sodium concentration (which we did not measure). Also, according to a consensus statement by Hecking et al. (2013)28, the majority of intermittent HD patients remain fluid overloaded following HD treatments (i.e., >15% above “normal” extracellular fluid volume, or carrying ~ 2.5 liters of extra fluid despite HD treatment). Volume-overloaded patients consume less fluid, and volume-depleted patients consume more fluid. The impact of the intervention may be confounded by the dialysis prescription, a factor that was not controlled in the present study. While control group participants were somewhat better dialyzed, both groups were well-dialyzed according to US guidelines for 3x/week dialysis.
Adding to these issues, IDWGAs may not have differed between treatment groups due to factors that led to a limited efficacy of the BalanceWise intervention on reducing dietary sodium intake. Socioeconomically disadvantaged individuals (who were over-represented in BalanceWise) have less exposure to computer technology, which could serve as a barrier to self-monitoring. However, an average recording of 1.6 meals per day over 16 weeks is better than what has been observed in other dietary intervention studies in healthier populations that employ technology-based self-monitoring.29–32 It is important to note that the PDA was programmed to permit entry of 4 meals. Participants selected the meal occasion (breakfast, lunch, dinner, or snack) prior to logging the foods they consumed. If participants forgot to select a new meal, they may have logged foods from multiple meals as a single meal. It is also possible that they consumed more than 4 meals during the day and, so, were forced to log foods from more than 1 dining occasion as a single meal. Thus, participants may have consumed more than the reported average of 1.6 meals per day.
With regard to the intervention, counseling participants about dietary behavior was done after-the-fact, during encounters in dialysis units. Interventions delivered in settings where the target behavior does not occur may not translate to the real-life situations experienced by participants. Also, efforts to engage participants in dietary behavior change are less likely to be effective if delivery of the intervention is not temporally consistent with the occurrence of the behavior to be changed. Smart phone technology and mobile applications that could be used to intervene in the time and place dietary behaviors occur were not used in our investigation, as they were just coming onto the market at the time the BalanceWise Study was implemented. Newer technologies may improve intervention efficacy.33
We are unable to report on the characteristics of patients from whom HIPAA releases were not obtained and, so, we are unable to describe the extent to which those enrolling in the study differed from the population of HD patients receiving care in the participating dialysis centers. It is possible that the study population was less than optimal for this intervention. BalanceWise participants had a median HD vintage of almost 3 years and reported relatively low dietary sodium intakes at baseline. Participants also were willing to meet frequently with a study dietitian and record their dietary intake on a daily basis, perhaps representing the most adherent patients among whom it would be difficult to make improvements.
It is also possible that beneficial outcomes went undetected due to measurement error. Dietary sodium intake is known to be measured with considerable error. Indeed, a recently published study showed the accuracy of dietary sodium intake estimates to be limited (based on an R2 value of 0.09 compared to urinary sodium).34 In HD patients, urinary sodium (if any) is not indicative of dietary sodium intake, so we had to rely on dietary assessment. The two-pass 24-hour recall method likely underestimated dietary sodium intake, and possessed more instrumental bias than the aforementioned study34, which used the USDA Automated Multiple-Pass Method. Not surprisingly, given the low baseline dietary sodium intake, the BalanceWise intervention did not result in large reductions. Such an intervention may be more efficacious in patients as they initiate HD, a time of transition when many difficult adjustments must be made by patients and mortality is particularly high.
PRACTICAL APPLICATION
The use of technology-based self-monitoring to support a behavioral intervention targeting dietary sodium restriction appears to be feasible and acceptable to HD patients. Although the intervention did not affect IDWGAs, it was somewhat efficacious in reducing dietary sodium intake at 8 weeks but not at 16 weeks. Additional research is needed to examine the extent to which such an intervention is efficacious for those who are initiating HD, demonstrating consistently excessive IDWGs, and/or consuming excessive amounts of sodium, controlling for dialysate sodium concentration and dialysis prescription. Researchers also may want to consider alternative measurement methods that better capture dietary sodium intake.
Table 5.
Parameter estimates from the random effects linear models for IDWGA over time and mean dietary sodium at baseline, 8, and 16 weeks.
| Outcome | Time | Technology-Supported Behavioral Intervention | Attention Control | Difference (Intervention – Control)† | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||||
| Mean | 95% CI | Mean | 95% CI | Coef. | 95% CI | p-value | |||||
| Time-specific mean IDWGA (kg/day) | Baseline | 1.14 | 1.05 | 1.24 | 1.16 | 1.06 | 1.26 | −0.017 | −0.16 | 0.12 | 0.80 |
| Week 8 | 1.16 | 1.07 | 1.25 | 1.17 | 1.07 | 1.26 | −0.007 | −0.14 | 0.13 | 0.92 | |
| Week 12 | 1.17 | 1.08 | 1.26 | 1.17 | 1.08 | 1.27 | −0.001 | −0.14 | 0.13 | 0.99 | |
| Week 16 | 1.18 | 1.08 | 1.28 | 1.18 | 1.08 | 1.27 | 0.004 | −0.13 | 0.14 | 0.95 | |
|
| |||||||||||
| Time-specific change in dietary sodium intake (mg/day)* | Baseline to Week 8 | −125.3 | −386.8 | 136.2 | 245.8 | −20.7 | 512.2 | −371.5 | −742.7 | −0.3 | 0.05 |
| Baseline to Week 16 | −49.8 | −316.1 | 216.5 | 141.2 | −127.7 | 410.1 | −191.3 | −567.5 | 184.9 | 0.32 | |
IDWGA, average daily interdialytic weight gain
Estimated time-specific differences (Intervention-Control) are summarized. The model for dietary sodium adjusts for diabetes status.
Adjusted for diabetes status,
For dietary sodium, this difference compares Intervention and Control participants with the same diabetes status.
Acknowledgments
The authors thank the management and staff of the participating dialysis units from DaVita HealthCare Partners Inc., Dialysis Clinic Inc., and Fresenius Medical Care North America; research study dietitians Beth Hall, BA, RD, LDN, and Susan Stark, MS, RD, CSR, LDN, for conducting the interventions; Deborah Klinvex, BA, for conducting the dietary recall interviews, D. Scott Obrosky, MS, for developing the data tracking system, and Tienna Luster for data management.
Support
The work of this paper was supported by the following grants: NIH/NINR/R01-NR010135, NIH/NINR/NIDDK/NHLBI/NIA- K24-NR012226, NIH/NIA/R01-AG027017, NIH/NIA/P30-AG024827, NIH/NIA/K07-AG033174. NIH played no role in study design; collection, analysis, and interpretation of data; writing the report; or the decision to submit the report for publication.
Footnotes
PDA data were not used to evaluate dietary sodium intake because control group participants did not receive their PDAs until after the study measurements had concluded, and because using the PDA record as a measure would confound independent and dependent variables.
Observations with a recorded intervention time of <0 or >120 minutes were considered erroneous and dropped from this analysis (n=19).
Financial Disclosure Declaration. The authors have no financial disclosures.
Trial Registration. Clinical trials.gov identifier: NCT01125202
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.USRDS. Annual Data Report. Volume 2: Atlas ESRD, Chapter 4 – Cardiovascular Disease. [Accessed March 26, 2015];2013 Available at: http://www.usrds.org/
- 2.Kalantar-Zadeh K, Regidor DL, Kovesdy CP, et al. Fluid retention is associated with cardiovascular mortality in patients undergoing long-term hemodialysis. Circulation. 2009;119(5):671–679. doi: 10.1161/CIRCULATIONAHA.108.807362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wizemann V, Wabel P, Chammey P, Zaluska W, Moissl U, Rode C, Malecka-Masalska T, Marcelli D. The mortality risk of overhydration in haemodialysis patients. Nephrol Dial Transplant. 2009;24(5):1574–1579. doi: 10.1093/ndt/gfn707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rahman M, Dixit A, Donley V, et al. Factors associated with inadequate blood pressure control 8in hypertensive hemodialysis patients. Am J Clin Nutri. 1999;33(3):498–506. doi: 10.1016/s0272-6386(99)70187-3. [DOI] [PubMed] [Google Scholar]
- 5.Foley RN, Parfrey PS, Harnett JD, Kent GM, Murray DC, Barre PE. Impact of hypertension on cardiomyopathy, morbidity and mortality in end-stage renal disease. Kidney Int. 1996;49(5):1379–1385. doi: 10.1038/ki.1996.194. [DOI] [PubMed] [Google Scholar]
- 6.USRDS. Annual Data Report. Volume 2: Atlas ESRD, Chapter 5: Mortality. [Accessed March 26, 2015];2012 Available at: http://www.usrds.org/
- 7.Flythe JE, Kimmel SE, Brunelli SM. Rapid fluid removal during dialysis is associated with cardiovascular morbidity and mortality. Kidney Int. 2011;79(2):250–257. doi: 10.1038/ki.2010.383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rigby-Mathews A, Scribner BH, Ahmad S. Control of interdialytic weight gain (IDWG) without water restriction in hemodialysis patients. J Am Soc Nephrol. 1999;10(Abstract):A1346. [Google Scholar]
- 9.McCausland FR, Waikar SS, Brunelli SM. Increased dietary sodium is independently associated with greater mortality among prevalent hemodialysis patients. Kidney Int. 2012;82(2):204–211. doi: 10.1038/ki.2012.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4) suppl 3:S1–153. [PubMed] [Google Scholar]
- 11.Carrigan A, Klinger A, Choquette SS, et al. Contribution of food additives to sodium and phosphorus content of diets rich in processed foods. J Ren Nutr. 2014;24(1):13–19. doi: 10.1053/j.jrn.2013.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Arquette C. Are there fast-food choices for end-stage renal disease patients? A look at phosphorus and potassium content in common fast foods. J Ren Nutr. 2014;24(3):e19–e21. [Google Scholar]
- 13.Smith LP, Ng SW, Popkin BM. Trends in US home food preparation and consumption: Analyssi of national nutrition surveys and time use studies from 1965–1966 to 2007–2008. Nutr J. 2013;12:45. doi: 10.1186/1475-2891-12-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Moubarac JC, Batal M, Martins AP, et al. Processed and ultra-processed food products: Consumption trends in Canada from 1938 to 2011. Can J Diet Pract Res. 2011;75(1):15–21. doi: 10.3148/75.1.2014.15. [DOI] [PubMed] [Google Scholar]
- 15.Bernstein AM, Willett WC. Trends in 24-h urinary sodium excretion in the United States, 1957–2003: A systematic review. Am J Clin Nutr. 2010;92(5):1172–1180. doi: 10.3945/ajcn.2010.29367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Loria CM, Mussolino ME, Cogswell ME, et al. Usual sodium intakes compared with current dietary guidelines: United States, 2005–2008. MMWR Morb Mortal Wkly Rep. 2011;60(41):1413–1417. [PubMed] [Google Scholar]
- 17.Bandura A. Social Foundations of Thought & Action: A Social Cognitive Theory. Englewood Cliffs, NJ: Prentice Hall; 1986. [Google Scholar]
- 18.Bandura A. Self-Efficacy: The Exercise of Control. New York, NY: Freeman and Co; 1997. [Google Scholar]
- 19.National Kidney Foundation Kidney Disease Outcomes Quality Initiative. Guideline 3: Management of Protein and Energy Intake. Daily Energy Intake for Maintenance Dialysis Patients. Am J Kidney Dis. 2000;35(6):S44. [Google Scholar]
- 20.National Kidney Foundation Kidney Disease Outcomes Quality Initiative. Guideline 3: Management of Protein and Energy Intake. Dietary Protein Intake in Maintenance Hemodialysis. Am J Kidney Dis. 2000;35(6):S40. [Google Scholar]
- 21.Sevick MA, Zickmund S, Korytkowski M, et al. Design, feasibility, and acceptability of an intervention using personal digital assistant-based self-monitoring in managing type 2 diabetes. Contemp Clin Trials. 2008;29(3):396–409. doi: 10.1016/j.cct.2007.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stark S, Snetselaar L, Hall B, et al. Nutritional Intake in Adult Hemodialysis Patients. Top Clin Nutr. 2011;26(1):45–56. doi: 10.1097/TIN.0b013e3181faba4c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gillis BP, Caggiula AW, Jones FL, et al. Features of the nutrient database and analysis system for the modification of diet in Renal Disease Study. Control Clin Trials. 1994;15(1):44–58. doi: 10.1016/0197-2456(94)90027-2. [DOI] [PubMed] [Google Scholar]
- 24.USRDS. Annual Data Report. Volume 2: End-stage Renal Disease (ESRD) in the United States, Chapter 1: Incidence, Prevalence, Patient Characteristics, and Treatment Modality. [Accessed March 26, 2015];2014 Available at: http://www.usrds.org/2014/
- 25.Cohen EP, Salatnik L, Leach N, Ahuja M. Reduction of interdialytic weight gain: Effect of low salt diet. J Am Soc Nephrol. 1999;10(Abstract):A237. [Google Scholar]
- 26.Maduell F, Navarro V. Dietary salt intake and blood pressure in haemodialysis patients. Nephrol Dial Transplant. 2000;15:2063. doi: 10.1093/ndt/15.12.2063. [DOI] [PubMed] [Google Scholar]
- 27.Telini LSR, Beduschi GC, Caramori JCT, Castro JH, Martin LC, Barretti P. Effect of dietary sodium restriction on body water, blood pressure, and inflammation in hemodialysis patients: A prospective randomized controlled study. Int Urol Nephrol. 2014;46:91–97. doi: 10.1007/s11255-013-0382-6. [DOI] [PubMed] [Google Scholar]
- 28.Hecking M, Karaboyas A, Antlanger M, et al. Significance of interdialytic weight gain versus chronic volume overload: consensus opinion. Am J Nephrol. 2013;38(1):78–90. doi: 10.1159/000353104. [DOI] [PubMed] [Google Scholar]
- 29.Sevick MA, Zickmund S, Korytkowski M, et al. Design, feasibility, and acceptability of an intervention using personal digital assistant-based self-monitoring in managing type 2 diabetes. Contemp Clin Trials. 2008;29(3):396–409. doi: 10.1016/j.cct.2007.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Glanz K, Murphy S, Moylan J, Evensen D, Curb JD. Improving dietary self-monitoring and adherence with hand-held computers: A pilot study. Am J Health Promot. 2006;20(3):165–170. doi: 10.4278/0890-1171-20.3.165. [DOI] [PubMed] [Google Scholar]
- 31.Yon BA, Johnson RK, Harvey-Berino J, Gold BC, Howard AB. Personal digital assistants are comparable to traditional diaries for dietary self-monitoring during a weight loss program. J Behav Med. 2007;30(2):165–175. doi: 10.1007/s10865-006-9092-1. [DOI] [PubMed] [Google Scholar]
- 32.Stark S, Snetselaar L, Piraino B, et al. PDA self-monitoring adherence rates in two dialysis dietary intervention pilot studies: BalanceWise-HD and BalanceWise-PD. J Ren Nutr. 2011;21(6):492–498. doi: 10.1053/j.jrn.2010.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shiffman S, Stone AA, Hufford MR. Ecological momentary assessment. Annu Rev Clin Psychol. 2008;4:1–32. doi: 10.1146/annurev.clinpsy.3.022806.091415. [DOI] [PubMed] [Google Scholar]
- 34.Rhodes DG, Murayi T, Clemens JC, et al. The USDA Automated Multiple-Pass Method accurately assesses population sodium intakes. Am J Clin Nutr. 2013;97:958–964. doi: 10.3945/ajcn.112.044982. [DOI] [PubMed] [Google Scholar]