Abstract
Introduction
Immune system activation is involved in Huntington’s disease (HD) pathogenesis and biomarkers for this process could be relevant to study the disease and characterise the therapeutic response to specific interventions. We aimed to study inflammatory cytokines and microglial markers in the CSF of HD patients.
Methods
CSF TNF-α, IL-1β, IL-6, IL-8, YKL-40, chitotriosidase, total tau and neurofilament light chain (NFL) from 23 mutation carriers and 14 healthy controls were assayed.
Results
CSF TNF-α and IL-1β were below the limit of detection. Mutation carriers had higher YKL-40 (p = 0.003), chitotriosidase (p = 0.015) and IL-6 (p = 0.041) than controls. YKL-40 significantly correlated with disease stage (p = 0.007), UHDRS total functional capacity score (r = -0.46, p = 0.016), and UHDRS total motor score (r = 0.59, p = 4.5*10−4) after adjustment for age.
Conclusion
YKL-40 levels in CSF may, after further study, come to have a role as biomarkers for some aspects of HD. Further investigation is needed to support our exploratory findings.
Introduction
Huntington’s disease (HD) is a neurodegenerative condition in which progressive decline in cognitive, motor and behaviour functions are a consequence of neuronal dysfunction and death. The primary cause of HD is known–a polyglutamine expansion in the first exon of the HTT gene[1, 2]–but the mechanisms by which mutant huntingtin (mHTT) protein leads to neuronal cell death still need clarification. The immune system, both peripherally and in the central nervous system (CNS), has been implicated in HD pathogenesis[3, 4]. Numerous post-mortem and in vivo studies have shown that complement activation[5], microglial activation[6],[7] and concentrations of pro-inflammatory and immunomodulatory cytokines IL-1β, IL-6, IL-8, IL-10, CCl2 and TNF-α[4, 8] are increased in peripheral blood in HD patients compared with controls. Animal studies suggest that inhibition of inflammatory pathways could ameliorate HD severity[9, 10]. One compound aimed at modulating inflammatory glial activity, laquinimod, is currently being tested in HD and several other clinical trials are being prepared.[11, 12]
Biomarkers reflecting these peripheral and/or central derangements of neuroinflammation could be useful to better characterise disease progression and the therapeutic response to specific interventions. Cerebrospinal fluid (CSF) is a relatively accessible body fluid, enriched in brain-derived proteins[13], that has proven to be of value as a source of biomarkers in HD[14] and other neurodegenerative diseases[4, 15, 16]. In 2014, Vinther-Jensen and colleagues found a statistically non-significant trend towards increasing CSF levels of the microglial marker YKL-40 with later disease stage, but no association with disease severity or replication has been sought in HD CSF collected under strictly standardised conditions.[17]
In this work, we explored immune-associated substances previously reported to be altered in CSF or plasma in HD, namely proinflammatory cytokines–TNF-α, IL-1β, IL-6 and IL-8 –and microglial markers–YKL-40 and chitotriosidase–in the CSF of well-characterised patients, to determine what markers are capable of predicting clinical severity in HD. By measuring total tau and neurofilament light chain (NFL)–established markers of neuronal cell death[18, 19]–in the same CSF samples, we also examined whether neuroinflammation and neurodegeneration are linked.
Materials and Methods
Ethical approval was given by the joint University College London/ University College London Hospitals ethics committee. All patients gave informed written consent before enrolment. Patient consent, inclusion and exclusion criteria, clinical assessment, CSF collection and storage were as previously published.[14] In brief, samples were collected after an overnight fast at the same time of day and centrifuged and aliquoted rapidly on ice using a standardised protocol and polypropylene plasticware.[14] Healthy controls were contemporaneously recruited, drawn from a population with a similar age to patients, and clinically well, so the risk of incidental neurodegenerative diseases was very low. Relevant aspects of clinical severity were quantified using the total functional capacity (TFC) and total motor score (TMS) components of the Unified Huntington’s Disease Rating Scale (UHDRS).[20] Disease burden score, a function of age and CAG repeat length known to predict many features of HD onset and progression, was calculated. [21, 22]
CSF analyte quantification
CSF TNF-α, IL-1β, IL-6 and IL-8 were measured using a Meso Scale Discovery antibody-based tetra-plex array with electrochemiluminiscence detection according to the manufacturer’s instructions (Meso Scale Discovery, Gaithersburg, MD, USA). CSF TNF-α and IL-1β concentrations were below the lower limits of detection (LODs) of the assay for all samples. CSF IL-6 and IL-8 concentrations were above the LODs (0.32 pg/mL and 0.25 pg/mL, respectively) in all samples, except for one in which IL-6 was <0.32 pg/mL. CSF YKL-40 concentration was measured by enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (R&D Systems Inc. Minneapolis, MN). This assay has an LOD of 6.25 ng/mL and all samples were well above this limit.
CSF chitotriosidase was measured using an in-house enzyme activity assay, as previously described.[23] Two samples in the HD group and eight samples in the control group were below the LOD of the assay (0.2 nkat/L). This distribution of undetectable values was statistically significant between the two groups (Chi-square 10.3569, p = 0.00129), in keeping with a disease-related difference (see results section). CSF total tau was quantified using the INNOTEST enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (Fujirebio, Ghent, Belgium). CSF NFL was measured using the NF-light ELISA according to the manufacturer’s instructions (UmanDiagnostics, Umeå, Sweden). All samples had CSF total tau and NFL concentrations above the LODs for the assays (45.8 and 50 ng/L, respectively). All measurements were performed as single analyses in one round of experiments using one batch of reagents. Based on internal quality control samples intra-assay coefficients of variation were 30% for IL-6, 12% for IL-8, 7% for YKL-40, 10% for chitotriosidase, 10% for total tau and 15% for NFL. Laboratory technicians were board-certified and blinded to clinical data.
Statistical analysis
Statistical analysis was performed with Stata 14 software (StataCorp, TX, USA). We used one-way ANOVA to assess baseline intergroup differences. Potentially confounding demographic variables (age, gender) were examined in preliminary analyses and those found significant were included as covariates for subsequent analyses. Samples below the LOD were assigned the LOD concentration. The distributions of all tested molecules were tested for normality. Two-group comparisons were tested using unpaired two-tailed t-test or Wilcoxon log-rank test. Stepwise forward logistic regression analysis was used to find combinations of molecules with better diagnostic performance, and these combinations’ receiver operating characteristics were compared with the best molecule alone. To test associations with disease progression we calculated Pearson’s and partial correlations coefficients. Where appropriate, bootstrapping with 1,000 repetitions was applied to non-normal variables. Significance level was defined as p<0.05. Where there was concern about a single participant group or outliers unduly influencing the analysis, a sensitivity analysis was conducted, repeating the tests with those datapoints excluded.
Results
Participants’ characteristics
Thirty-seven cross-sectional CSF samples were obtained from 14 healthy controls, 3 pre-manifest gene expansion carriers (HDGECs), and 20 manifest HDGECs. Details are given in Table 1. There was no significant difference in age or gender distribution among the included groups. CAG repeat number did not vary significantly between HDGEC groups.
Table 1. Characteristics of included participants by disease stage.
| n (%) | Age (±SD) | M:F ratio | CAG | TFC | TMS | Disease burden | |
|---|---|---|---|---|---|---|---|
| Total sample | 37 | 47 (±12) | 14:23 | ||||
| Healthy controls | 14 (38) | 44 (±13) | 4:10 | N/A | 13 | N/A | N/A |
| Pre-manifest | 3 (8) | 40 (±10) | 1:2 | 42 | 13 | 1 | 252 |
| Early stage | 15 (41) | 50 (±12) | 6:9 | 44 | 11 | 27 | 407 |
| Moderate stage | 5 (14) | 58 (±2) | 3:2 | 43 | 5 | 50 | 421 |
| Intergroup differences | p = 0.077 | p = 0.716 | p = 0.261 | p = 1*10−8 | p = 0.005 | p = 0.007 |
M:F, male to female ratio; CAG, Cytosine-adenosine-guanine repeats; TFC, total functional score; TMS, total motor score; N/A, not applicable.
CSF concentrations
Medians and interquartile range (IQR) of CSF concentrations of quantified substances are shown in Table 2. Only IL-8 was normally distributed. CSF TNF-α and IL-1β concentrations were below the limit of detection in all cases. No substance varied between genders. YKL-40 and IL-8 were significantly correlated with age in healthy control participants (r = 0.82, p = 2.6*10−12 for YKL-40 and r = 0.50, p = 0.002 for IL-8) so age was used as a covariate for these analyses. No other associations with age were found.
Table 2. CSF biomarkers concentrations by disease stage.
| YKL-40ng/mL(median, IQR) | Chitotriosidasenkat/L(median, IQR) | IL-6pg/mL(median, IQR) | IL-8pg/mL(median, IQR) | NfLpg/mL(median, IQR) | taupg/mL(median, IQR) | |
|---|---|---|---|---|---|---|
| Total | 116.76 (103.43) | 0.47 (0.46) | 0.85 (0.40) | 33.50 (7.50) | 1223 (1625) | 238 (105) |
| Healthy controls | 83.071 (58.133) | 0.20 (0.27) | 0.70 (0.30) | 34.00 (19.00) | 300 (134) | 192 (104) |
| Pre-manifest | 91.420 (54.687) | 0.47 (0.32) | 0.90 (0.40) | 28.00 (11.00) | 842 (365) | 196 (87) |
| Early stage | 124.757 (95.458) | 0.51 (0.34) | 1.00 (0.80) | 33.00 (5.00) | 1969 (1126) | 252 (126) |
| Moderate stage | 194.121 (16.371) | 0.41 (0.82) | 0.95 (0.80) | 38.00 (10.50) | 2231 (994) | 384 (183) |
IQR, interquartile range; IL, interleukin; NfL, neurofilament light chain.
Effects of haemoglobin contamination
The concentration of CSF haemoglobin was not found to be associated with the concentrations of any analytes, and therefore we assume any minimal blood contamination did not interfere with quantification of the analytes.
Comparison between healthy controls versus gene expansion carriers
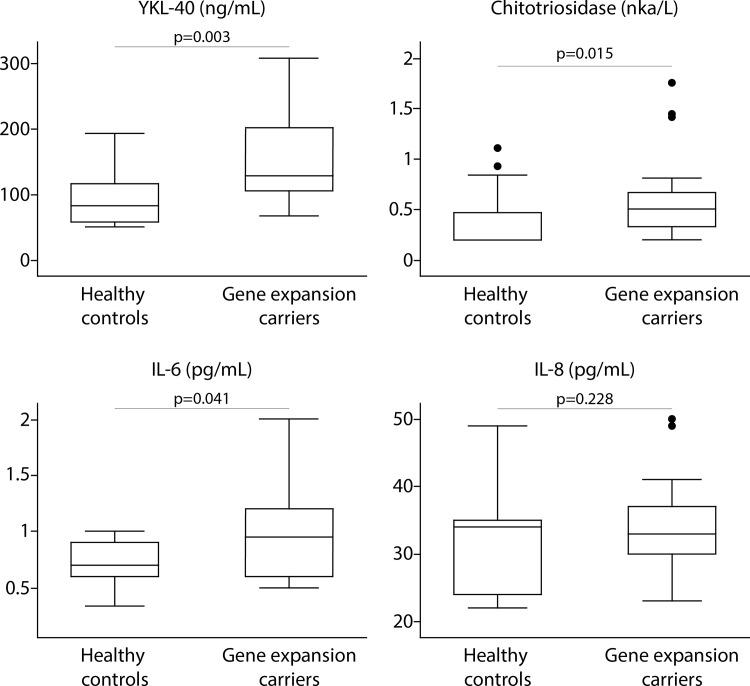
CSF YKL-40, chitotriosidase and IL-6 were significantly elevated in HDGECs compared with controls (Fig 1; p = 0.003, AUC = 0.797 for YKL-40, p = 0.015, AUC = 0.7419 for chitotriosidase; p = 0.041, AUC = 0.7029 for IL-6). IL-8 was not significantly altered between these groups (p = 0.228, AUC = 0.5909). Forward stepwise logistic regression analysis showed that the combination of YKL40 and IL-6 had a higher diagnostic power (AUC = 0.8620) than any of the individual molecules. However, this combination was not statistically significantly better than YKL-40 alone (p = 0.2674).
Fig 1. Comparison in CSF levels of inflammatory molecules between 14 healthy controls versus 23 gene expansion carriers.
The p-values for unpaired two-tailed t-test (Il-8) or Wilcoxon log-rank test (YKL-40, chitotriosidase, IL-6) are shown.
Associations with disease stage
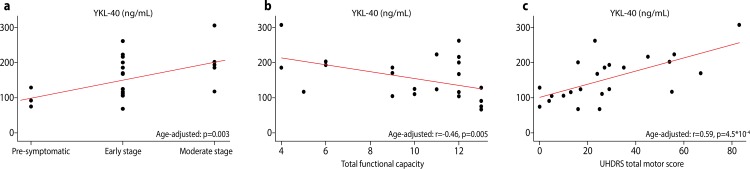
In HDGECs, there was a statistically significant positive association between CSF YKL-40 level and disease stage (p = 0.001, Fig 2A). This association remained significant after controlling for age (p = 0.003). Repeating the analysis excluding premanifest individuals (p = 0.018), outliers (p = 0.005) and both (p = 0.013) supported the statistical significance of this association. CSF IL-8 level, too, was significantly positively associated with disease stage (p = 0.023), but not after controlling for age (p = 0.153). Chitotriosidase and IL-6 were not significantly associated with disease stage (p = 0.891 and p = 0.398, respectively).
Fig 2. Relationship between CSF concentration of YKL-40 and measures of disease progression and clinical severity.
a, disease stage; b, UHDRS total functional score; c, UHDRS total motor score. The p-values for partial Pearson’s correlation coefficient adjusted for age are shown.
Associations with clinical severity
There was a statistically significant negative association between CSF YKL-40 and TFC after controlling for age (r = -0.46, p = 0.005; Fig 2B), but not disease burden score (r = -0.36, p = 0.090). CSF chitotriosidase, IL-6 and IL-8 were not significantly associated with TFC (r = 0.12, p = 0.607; r = -0.16, p = 0. 531 and r = -0.37, p = 0.093, respectively).
CSF YKL-40 was significantly positively associated with TMS (r = 0.65, p = 1.3*10−5, Fig 2C). This association remained significant after controlling for age (r = 0.59, p = 4.5*10−4), and also after controlling for disease burden score (r = 0.49, p = 0.037), indicating that CSF YKL-40 can independently predict severity of motor manifestations beyond the known predictive value of age and CAG repeat length. Chitotriosidase, IL-6 and IL-8 were not statistically significantly associated with TMS (r = -0.15, p = 0.234; r = -0.08, p = 0.666; and r = 0.23, p = 0.3055, respectively).
Associations with markers of neuronal death
Because they had been found to be associated with measures of clinical severity, YKL-40 and IL-6 were further examined for association with CSF tau and NFL levels, each of which has been shown to be associated with neuronal death in neurodegenerative and other conditions.[24–26] There were statistically significant positive associations between YKL-40 and both total tau (r = 0.65,.p = 1.4*10−5) and NFL (r = 0.72,.p = 3.7*10−26), which remained significant after controlling for age (r = 0.69, p = 1.2*10−4; r = 0.72, p = 1.5*10−9, respectively), and disease burden score (r = 0.51, p = 0.006; r = 0.55, p = 3.5*10−6, respectively), again suggesting that YKL-40 level can predict these markers of neuronal death, independent of their mutual association with age and CAG repeat length. IL-6 did not show any such association with tau or NFL (r = 0.17, p = 0.559; r = 0,23, p = 0.300, respectively).
Discussion
In this exploratory cross-sectional study using CSF samples collected and processed under rigorously controlled conditions, we show that CSF concentrations of the microglial markers YKL-40 and chitotriosidase, and the pro-inflammatory cytokine IL-6, were elevated in HDGECs compared with healthy controls.
YKL-40 was most robustly associated with clinical severity: age-adjusted YKL-40 CSF concentrations in gene expansion carriers were associated with disease stage and with clinical measurements of disease progression—UHDRS TFC and UHDRS TMS. These latter associations remained statistically significant after adjustment for disease burden score, indicating that CSF YKL-40 has independent clinical predictive power, beyond its association with age and CAG repeat length. To our knowledge, the only other CSF substance for which such independent predictive power has been reported is the pathological agent itself, mutant huntingtin protein (mHTT).[14]
Furthermore, CSF YKL-40 was associated with the CSF markers of neuronal death tau and NFL; and retained this predictive power after adjustment for the mutually-associated predictors age and CAG repeat length.
YKL-40, also known as chitinase 3-like protein 1 (CHI3L1), is a member of the 18 glycosyl hydrolase family, without enzymatic activity and with poorly understood function.[27] During neuroinflammation YKL-40 is expressed and secreted by microglia[28] and is associated with astrocytosis and astrocytic motility.[29] Neuroinflammation is thought to have an important role on HD pathogenesis and mHTT has a direct effect on the NFκB pathway.[30] This interaction induces expression of pro-inflammatory molecules and may explain the elevation of YKL-40 in HD CSF.
IL-6 in CSF–previously shown to be elevated in HD blood plasma–was found to be associated with disease stage but not with more granular clinical measures of severity or neuronal death. IL-6 is known to cross the blood-brain barrier[31] so it seems most likely that its CSF level reflects systemic elevation or possibly parallel CNS and peripheral immune activation.[4] Nevertheless these results should be interpreted with caution since the coefficient of variation of this assay was relatively high.
To our knowledge, this is the first study describing the potential of CSF YKL-40 as an independent predictor of clinical severity and neuronal death in HD. Our findings differ from those of Vinther-Jensen et al, who did not find a statistically significant association between CSF YKL-40 and disease stage or severity.[17] Differences in sample collection, processing and assay methodology may explain the differences in our findings. Our standardised protocols aim to reduce avoidable inter-subject variability by controlling fasting state, time of day, collection methodology, processing methods and equipment. Still, it would be sensible to further validate these differences in larger cohorts. To our knowledge, our study is the first to explore the relationships between CSF inflammatory markers and clinical variables such as TFC and TMS, and biomarkers of neuronal cell death.[17]
Notwithstanding the striking findings in respect of YKL-40, this exploratory study requires validation in larger studies such as the HDClarity initiative.[32] The longitudinal predictive power of CSF YKL-40 should be examined in studies with repeat CSF collection. Furthermore, evaluation in blood of biomarkers proposed from CSF is an important focus of our work. Blood has hitherto failed to yield meaningful biomarkers of CNS disease activity but this remains an important aim if suitably CNS-specific blood markers can be identified.[33]
Conclusion
We conclude that IL-6, chitotriosidase and YKL-40 show disease-related elevations in CSF in HD, affirming the role of microglial activation and the innate immune system in the disease. CSF YKL-40 in particular can independently predict clinical severity and neuronal death, and may be a useful targeted biomarker for the contribution of microglial dysfunction to disease activity.
Supporting Information
(XLSX)
Acknowledgments
We thank the patient and control volunteers who contributed to this study. FBR and EJW are supported by CHDI Foundation. LMB, SJT and EJW are supported by the Medical Research Council. HZ is supported by the Swedish Research Council, European Research Council, and the Knut and Alice Wallenberg Foundation. SJT is supported by the Wolfson Foundation. EJW received funds from GlaxoSmithKline foundation. This work was supported in part by the National Institute for Health Research University College London Hospitals Biomedical Research Centre and the UCL Leonard Wolfson Experimental Neurology Centre.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This work was supported by the CHDI Foundation to EW and FBR, GlaxoSmithKline foundation to EW, Swedish Research Council to HZ, European Research Council to SJT and HZ, Medical Research Council to EW, SJT and LB, Knut ooch Alice Wallenbergs Stiftelse to HZ and the Wolfson Foundation to SJT. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Gusella JF, Wexler NS, Conneally PM, Naylor SL, Anderson MA, Tanzi RE, et al. A polymorphic DNA marker genetically linked to Huntington's disease. Nature. 1983;306(5940):234–8. Epub 1983/11/17. . [DOI] [PubMed] [Google Scholar]
- 2.Group THsDCS. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. The Huntington's Disease Collaborative Research Group. Cell. 1993;72(6):971–83. Epub 1993/03/26. . [DOI] [PubMed] [Google Scholar]
- 3.Moller T. Neuroinflammation in Huntington's disease. J Neural Transm (Vienna). 2010;117(8):1001–8. 10.1007/s00702-010-0430-7 . [DOI] [PubMed] [Google Scholar]
- 4.Bjorkqvist M, Wild EJ, Thiele J, Silvestroni A, Andre R, Lahiri N, et al. A novel pathogenic pathway of immune activation detectable before clinical onset in Huntington's disease. The Journal of experimental medicine. 2008;205(8):1869–77. Epub 2008/07/16. 10.1084/jem.20080178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Singhrao SK, Neal JW, Morgan BP, Gasque P. Increased complement biosynthesis by microglia and complement activation on neurons in Huntington's disease. Experimental neurology. 1999;159(2):362–76. Epub 1999/10/03. 10.1006/exnr.1999.7170 . [DOI] [PubMed] [Google Scholar]
- 6.Sapp E, Kegel KB, Aronin N, Hashikawa T, Uchiyama Y, Tohyama K, et al. Early and progressive accumulation of reactive microglia in the Huntington disease brain. Journal of neuropathology and experimental neurology. 2001;60(2):161–72. Epub 2001/03/29. . [DOI] [PubMed] [Google Scholar]
- 7.Tai YF, Pavese N, Gerhard A, Tabrizi SJ, Barker RA, Brooks DJ, et al. Microglial activation in presymptomatic Huntington's disease gene carriers. Brain: a journal of neurology. 2007;130(Pt 7):1759–66. Epub 2007/04/03. 10.1093/brain/awm044 . [DOI] [PubMed] [Google Scholar]
- 8.Silvestroni A, Faull RL, Strand AD, Moller T. Distinct neuroinflammatory profile in post-mortem human Huntington's disease. Neuroreport. 2009;20(12):1098–103. Epub 2009/07/11. 10.1097/WNR.0b013e32832e34ee . [DOI] [PubMed] [Google Scholar]
- 9.Zwilling D, Huang SY, Sathyasaikumar KV, Notarangelo FM, Guidetti P, Wu HQ, et al. Kynurenine 3-monooxygenase inhibition in blood ameliorates neurodegeneration. Cell. 2011;145(6):863–74. Epub 2011/06/07. 10.1016/j.cell.2011.05.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kwan W, Magnusson A, Chou A, Adame A, Carson MJ, Kohsaka S, et al. Bone marrow transplantation confers modest benefits in mouse models of Huntington's disease. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2012;32(1):133–42. Epub 2012/01/06. 10.1523/JNEUROSCI.4846-11.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wild EJ, Tabrizi SJ. Targets for future clinical trials in Huntington's disease: what's in the pipeline? Movement disorders: official journal of the Movement Disorder Society. 2014;29(11):1434–45. Epub 2014/08/27. 10.1002/mds.26007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.A Clinical Study in Subjects With Huntington's Disease to Assess the Efficacy and Safety of Three Oral Doses of Laquinimod [Internet]. Bethesda (MD): National Library of Medicine (US) 2014. [cited 2016 Mar 02]. Available from: https://ClinicalTrials.gov/show/NCT02215616. [Google Scholar]
- 13.Reiber H. Proteins in cerebrospinal fluid and blood: barriers, CSF flow rate and source-related dynamics. Restorative neurology and neuroscience. 2003;21(3–4):79–96. Epub 2003/10/08. . [PubMed] [Google Scholar]
- 14.Wild EJ, Boggio R, Langbehn D, Robertson N, Haider S, Miller JR, et al. Quantification of mutant huntingtin protein in cerebrospinal fluid from Huntington's disease patients. The Journal of clinical investigation. 2015;125(5):1979–86. Epub 2015/04/07. 10.1172/JCI80743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hirsch EC, Hunot S. Neuroinflammation in Parkinson's disease: a target for neuroprotection? The Lancet Neurology. 2009;8(4):382–97. Epub 2009/03/20. 10.1016/S1474-4422(09)70062-6 . [DOI] [PubMed] [Google Scholar]
- 16.Fiala M, Veerhuis R. Biomarkers of inflammation and amyloid-beta phagocytosis in patients at risk of Alzheimer disease. Experimental gerontology. 2010;45(1):57–63. Epub 2009/08/25. 10.1016/j.exger.2009.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vinther-Jensen T, Budtz-Jorgensen E, Simonsen AH, Nielsen JE, Hjermind LE. YKL-40 in cerebrospinal fluid in Huntington's disease—a role in pathology or a nonspecific response to inflammation? Parkinsonism & related disorders. 2014;20(11):1301–3. Epub 2014/09/16. 10.1016/j.parkreldis.2014.08.011 . [DOI] [PubMed] [Google Scholar]
- 18.Blennow K, Hampel H, Weiner M, Zetterberg H. Cerebrospinal fluid and plasma biomarkers in Alzheimer disease. Nature reviews Neurology. 2010;6(3):131–44. Epub 2010/02/17. 10.1038/nrneurol.2010.4 . [DOI] [PubMed] [Google Scholar]
- 19.Norgren N, Rosengren L, Stigbrand T. Elevated neurofilament levels in neurological diseases. Brain research. 2003;987(1):25–31. Epub 2003/09/23. . [DOI] [PubMed] [Google Scholar]
- 20.Huntington's disease study group. Unified Huntington's Disease Rating Scale: reliability and consistency. Huntington Study Group. Movement disorders: official journal of the Movement Disorder Society. 1996;11(2):136–42. Epub 1996/03/01. 10.1002/mds.870110204 . [DOI] [PubMed] [Google Scholar]
- 21.Penney JB Jr., Vonsattel JP, MacDonald ME, Gusella JF, Myers RH. CAG repeat number governs the development rate of pathology in Huntington's disease. Annals of neurology. 1997;41(5):689–92. Epub 1997/05/01. 10.1002/ana.410410521 . [DOI] [PubMed] [Google Scholar]
- 22.Tabrizi SJ, Scahill RI, Owen G, Durr A, Leavitt BR, Roos RA, et al. Predictors of phenotypic progression and disease onset in premanifest and early-stage Huntington's disease in the TRACK-HD study: analysis of 36-month observational data. The Lancet Neurology. 2013;12(7):637–49. Epub 2013/05/15. 10.1016/S1474-4422(13)70088-7 . [DOI] [PubMed] [Google Scholar]
- 23.Mattsson N, Tabatabaei S, Johansson P, Hansson O, Andreasson U, Mansson JE, et al. Cerebrospinal fluid microglial markers in Alzheimer's disease: elevated chitotriosidase activity but lack of diagnostic utility. Neuromolecular medicine. 2011;13(2):151–9. Epub 2011/05/14. 10.1007/s12017-011-8147-9 . [DOI] [PubMed] [Google Scholar]
- 24.Rodrigues FB, Byrne L, McColgan P, Robertson N, Tabrizi SJ, Leavitt BR, et al. Cerebrospinal fluid total tau concentration predicts clinical phenotype in Huntington's disease. Journal of Neurochemistry. 2016:n/a-n/a. 10.1111/jnc.13719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Samgard K, Zetterberg H, Blennow K, Hansson O, Minthon L, Londos E. Cerebrospinal fluid total tau as a marker of Alzheimer's disease intensity. International journal of geriatric psychiatry. 2010;25(4):403–10. Epub 2009/08/04. 10.1002/gps.2353 . [DOI] [PubMed] [Google Scholar]
- 26.Lu CH, Macdonald-Wallis C, Gray E, Pearce N, Petzold A, Norgren N, et al. Neurofilament light chain: A prognostic biomarker in amyotrophic lateral sclerosis. Neurology. 2015;84(22):2247–57. Epub 2015/05/03. 10.1212/WNL.0000000000001642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Aronson NN, Blanchard CJ, Madura JD. Homology modeling of glycosyl hydrolase family 18 enzymes and proteins. J Chem Inf Comput Sci. 1997;37(6):999–1005. 10.1021/ci970236v . [DOI] [PubMed] [Google Scholar]
- 28.Bonneh-Barkay D, Bissel SJ, Wang G, Fish KN, Nicholl GC, Darko SW, et al. YKL-40, a marker of simian immunodeficiency virus encephalitis, modulates the biological activity of basic fibroblast growth factor. The American journal of pathology. 2008;173(1):130–43. Epub 2008/06/17. 10.2353/ajpath.2008.080045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bonneh-Barkay D, Bissel SJ, Kofler J, Starkey A, Wang G, Wiley CA. Astrocyte and macrophage regulation of YKL-40 expression and cellular response in neuroinflammation. Brain Pathol. 2012;22(4):530–46. 10.1111/j.1750-3639.2011.00550.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Trager U, Andre R, Lahiri N, Magnusson-Lind A, Weiss A, Grueninger S, et al. HTT-lowering reverses Huntington's disease immune dysfunction caused by NFkappaB pathway dysregulation. Brain: a journal of neurology. 2014;137(Pt 3):819–33. Epub 2014/01/25. 10.1093/brain/awt355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Banks WA, Plotkin SR, Kastin AJ. Permeability of the blood-brain barrier to soluble cytokine receptors. Neuroimmunomodulation. 1995;2(3):161–5. Epub 1995/05/01. . [DOI] [PubMed] [Google Scholar]
- 32.Wild EJ, Borowsky B, HDClarity investigators, editors. HDClarity: A new multi-site cerebrospinal fluid collection initiative to facilitate therapeutic development for Huntington’s disease. 11th Annual CHDI HD Therapeutics Conference 2016; 2016; Palm Springs, CA.
- 33.Byrne LM, Wild EJ. Cerebrospinal Fluid Biomarkers for Huntington's Disease. Journal of Huntington's disease. 2016;5(1):1–13. Epub 2016/04/01. 10.3233/JHD-160196 . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.