Abstract
Background
Antifungal prophylaxis is currently regarded as the gold standard in situations with allo-genetic hematopoietic stem cell transplantation (allo-HSCT). However, the epidemiological information regarding prophylaxis of invasive fungal diseases (IFDs) is not clear in China.
Methods
We report the first large-scale (1053 patients) observational study of the prophylaxis and management of IFDs among patients with allo-HSCT in China.
Results
The incidence rates of IFD after primary antifungal prophylaxis (PAP), secondary antifungal prophylaxis (SAP), and non-prophylaxis were 22.7 vs. 38.6 vs. 68.6 %, respectively (P = 0.0000). The median time from transplantation to IFD was 45 days in PAP patients, 18 days in SAP patients, and 12 days in non-prophylaxis patients. Aspergillus spp. represents the most common type of fungal infection. Independent risk factors for IFD in allo-HSCT patients with PAP were age, having human leukocyte antigen (HLA)-haploidentical or matched unrelated donor, decreased albumin levels, and the use of itraconazole as the prophylactic antifungal agent. Among SAP transplant recipients, there was no significant risk factor for IFDs. The incidence rates of overall survival (OS) in the PAP, SAP, and no prophylaxis groups were 85.07, 78.80, and 74.82, respectively (P = 0.01).
Conclusions
This observational study indicates that prophylaxis of IFD is helpful to reduce the incidence of IFDs and improve the OS of patients after allo-HSCT.
Keywords: Invasive fungal diseases, Allogenetic, Stem cell, Transplantation, Prophylaxis, China
Key points
This first large-scale observational study of invasive fungal disease (IFD) in China indicated that prophylaxis of IFD is helpful to improve the OS of patients after allo-genetic hematopoietic stem cell transplantation (allo-HSCT). For patients with a history of IFD, effective prevention of IFD is particularly important because they have higher incidence of IFD.
Background
Although the control of bacterial infections in patients with hematological malignancies has been significantly improved with broad-spectrum antibiotics in the past decades, treating invasive fungal diseases (IFDs) is still a major problem in these patients, especially in patients undergoing allogenetic hematopoietic stem cell transplantation (allo-HSCT) [1–3]. Epidemiological data from the USA, China, and parts of Europe have shown that the incidence of IFDs in patients after allo-HSCT has increased dramatically in the past several years, substantially increasing the morbidity and mortality rates [4–9].
The diagnosis and treatment guidelines for IFDs, including some that are specific to HSCT patients, have been developed by academic societies in different world regions [10–12]. Notwithstanding the guidance and new forms of antifungal agents, treatment is often delayed because of nonspecific disease presentation and a lack of reliable diagnostic techniques, leading to poor clinical outcomes. Prophylaxis of IFDs is currently regarded as the gold standard in situations with allo-HSCT. Over the years, different scientific societies have established a series of recommendations for antifungal prophylaxis based on prospective studies performed with different drugs [13–17].
However, until recently, data on the prophylaxis of IFDs and real-world management of IFDs in patients with allo-HSCT have been gleaned primarily from single-center and retrospective studies in China. Here, we report the first large-scale observational study of the prophylaxis and management of IFDs among adults and children who have undergone allo-HSCT in China. Data came from the China Assessment of Antifungal Therapy in Hematological Disease (CAESAR) study.
Methods
Study design
The CAESAR study was a multicenter, prospective, observational study performed in 35 hematology centers across China, including two children’s hospitals. Subjects were consecutive patients of any age with a hematological malignancy who were hospitalized during the study period either after allogeneic or autologous HSCT or to receive intravenous chemotherapy. The overall study methods have been described previously [7, 18]. This observational study was conducted as a part of the CAESAR study and focused on the prophylaxis of IFDs in 1053 patients who underwent allo-HSCT in 31 HSCT centers.
All patients in each study center who were hospitalized and had undergone allo-HSCT between January 1, 2011 and October 30, 2011 were enrolled in this study. Data were collected by chart reviews and included the following parameters: demographic characteristics, antecedent hematologic disease, type of HSCT, conditioning regimen, IFD risk factors, clinical features suggestive of IFD, laboratory findings such as imaging, histopathology, and fungal cultures, treatment outcome of IFD, and mortality. Each IFD was categorized as proven, probable, or possible, according to the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) 2008 criteria [19]. Patients were diagnosed as having suspected IFD if they had IFD risk factors; were observed to have symptoms, radiological abnormalities, or indirect microbiological evidence of fungal infection; and were treated empirically with antifungal agents but could not be diagnosed with proven, probable, or possible IFD according to the EORTC/MSG 2008 criteria [20]. Patients were followed for 6 months after the date of transplantation; the follow-up was completed on April 30, 2012. In all, 18 patients were lost to follow-up.
Statistical analysis
In accordance with common practice and to maximize diagnostic accuracy, the incidence of IFD was mainly calculated based on proven and probable cases combined. The cumulative incidence was calculated as the incidence of proven plus probable IFD for the first 187 days after transplantation, divided by the number of cases at risk. Data were grouped according to primary antifungal prophylaxis (PAP) or secondary antifungal prophylaxis (SAP). PAP refers to patients without a history of fungal infection and need to be protected to avoid fungal infection after HSCT [21]. SAP is a rational strategy for patients with proven or probable IFD within 6 months before transplantation [22]. In particular, all allo-HSCT patients (n = 1045) in the CAESAR study were included in the analysis of overall survival (OS) for comparison of the OS differences in the PAP, SAP, and no prophylaxis groups. The characteristics of all of the allo-HSCT patients were described in the CAESAR study [7].
Statistics were primarily descriptive and were compared using analysis of variance, the Wilcoxon rank-sum test, or the chi-squared test, as appropriate. Risk factors for IFD were analyzed using univariate analysis followed by multivariate logistic regression. Kaplan-Meier analysis and the log-rank test were used to compare OS between different groups of patients. A two-sided P value of less than 0.05 was denoted as statistically significant. All statistical analyses were performed with SAS software version 9.1 (SAS, Cary, NC, USA).
Results
Patient characteristics and antifungal prophylaxis regimen
A total of 1053 patients who underwent allo-HSCT were enrolled from 31 HSCT centers across China. Their baseline demographic and clinical characteristics at the time of admission for transplantation are shown in Table 1. Among them, 906 patients received prophylactic antifungal treatment before or after transplantation. Previous IFDs were noted in 88 patients (8.4 %, 88/1053), including 7 with proven IFDs and 81 with probable IFDs.
Table 1.
Patient characteristics and antifungal prophylaxis regimen
| Characteristic | PAP (N = 818) | SAP (N = 88) | Non-antifungal prophylaxis (n = 147) |
|---|---|---|---|
| Age, mean (range/%)/case | 28.5 (1–63) | 32.2 2–58) | 29.6 (4–61) |
| 0–6 | 33 (4 %) | 3 (3.4 %) | 4 (2.7 %) |
| >6–16 | 129 (15.8 %) | 10 (11.4 %) | 20 (13.6 %) |
| >16–40 | 488 (59.7 %) | 49 (55.7 %) | 85 (57.8 %) |
| >40–65 | 168 (20.5 %) | 26 (29.5 %) | 38 (25.9 %) |
| Gender | |||
| Male | 505 (61.7 %) | 51 (58.0 %) | 87 (59.2 %) |
| Female | 313 (38.3 %) | 37 (42.0 %) | 60 (40.8 %) |
| E-COG | |||
| 0 | 227 (27.8 %) | 18 (20.5 %) | 48 (32.7 %) |
| 1 | 438 (53.5 %) | 54 (61.4 %) | 74 (50.3 %) |
| 2 | 89 (10.9 %) | 11 (12.5 %) | 17 (11.6 %) |
| 3 | 51 (6.2 %) | 5 (5.7 %) | 7 (4.8 %) |
| 4 | 13 (1.6 %) | – | 1 (0.7 %) |
| Underlying disease | |||
| Acute myeloid leukemia | 289 (35.3 %) | 44 (50.0 %) | 43 (29.3 %) |
| Acute lymphocytic leukemia | 241 (29.5 %) | 32 (36.4 %) | 37 (25.2 %) |
| Chronic myeloid leukemia | 100 (12.2 %) | 3 (3.4 %) | 23 (15.6 %) |
| Aplastic anemia | 69 (8.4 %) | – | 13 (8.8 %) |
| Myelodysplastic syndrome | 62 (7.6 %) | 2 (2.3 %) | 16 (10.9 %) |
| Non-Hodgkin’s lymphoma | 23 (2.8 %) | 4 (4.5 %) | 4 (2.7 %) |
| Chronic lymphocytic leukemia | 4 (0.5 %) | – | – |
| Multiple myeloma | 2 (0.2 %) | – | 1 (0.7 %) |
| Hodgkin’s disease | 2 (0.2 %) | – | 3 (2.0 %) |
| Myeloprolififerative neoplasms | 2 (0.2 %) | – | – |
| Solid tumor | 1 (0.1 %) | – | – |
| Hereditary and metabolic disorders | 9 (1.1 %) | – | – |
| Paroxysmal nocturnal hemoglobinuria | 1 (0.1 %) | – | 1 (0.7 %) |
| Othersa | 13 (1.6 %) | 3 (3.4 %) | 6 (4.1 %) |
| Transplantation type | |||
| HLA-matched related (sibling) | 335 (41.0 %) | 35 (39.8 %) | 77 (52.4 %) |
| Haploidentical | 269 (32.9 %) | 26 (29.5 %) | 35 (23.8 %) |
| Unrelated | 213 (26.0 %) | 27 (30.7 %) | 35 (23.8 %) |
| Source of stem cells | |||
| PB | 417 (51.0 %) | 67 (76.1 %) | 105 (71.4 %) |
| BM + PB | 348 (42.5 %) | 19 (21.6 %) | 20 (13.6 %) |
| BM | 33 (4.0 %) | – | 7 (4.8 %) |
| CB | 15 (1.8 %) | 1 (1.1 %) | 3 (2.0 %) |
| BM + CB | 3 (0.4 %) | – | – |
| BM + PB + CB | 2 (0.2 %) | 1 (1.1 %) | 8 (5.4 %) |
| Conditioning regimen | |||
| Myeloablative | |||
| Yes | 729 (89.1 %) | 83 (94.3 %) | 124 (84.4 %) |
| No | 89 (10.9 %) | 5 (5.7 %) | 23 (15.6 %) |
| Total body radiotherapy | |||
| Yes | 122 (14.9 %) | 23 (26.1 %) | 13 (8.8 %) |
| No | 696 (85.1 %) | 65 (73.9 %) | 134 (91.2 %) |
| Antithymocyte globulin | |||
| Yes | 516 (63.1 %) | 52 (59.1 %) | 64 (43.5 %) |
| No | 302 (36.9 %) | 36 (40.9 %) | 83 (56.5 %) |
| Concomitant disease | 137 (16.7 %) | 22 (25.0 %) | 23 (15.6 %) |
| Cardiovascular disease | 24 (2.9 %) | 2 (2.3 %) | 4 (2.7 %) |
| Diabetes | 24 (2.9 %) | 2 (2.3 %) | 6 (4.1 %) |
| Viral hepatitis | 22 (2.7 %) | 9 (10.2 %) | 4 (2.7 %) |
| CMV infection | 1 (0.1 %) | 2 (2.3 %) | 1 (0.7 %) |
| Tuberculosis | 14 (1.7 %) | 3 (3.4 %) | 2 (1.4 %) |
| Autoimmune diseases | 9 (1.1 %) | 1 (1.1 %) | – |
| Othersb | 61 (7.5 %) | 5 (5.7 %) | 8 (5.4 %) |
| aGVHD | |||
| I–II aGVHD | 257 (31.4 %) | 27 (30.7 %) | 40 (27.2 %) |
| III–IV aGVHD | 57 (7.0 %) | 7 (8.0 %) | 15 (10.2 %) |
| cGVHD | |||
| Limited | 57 (7.0 %) | 9 (10.3 %) | 10 (6.85) |
| Extensive | 24 (2.9 %) | 2 (2.3 %) | 4 (2.7 %) |
| Drugs for IFD prophylaxis | |||
| Single drug | 667 (81.5 %) | 69 (78.4 %) | – |
| Fluconazole | 429 (64.3 %) | 8 (11.6 %) | – |
| Itraconazole | 103 (15.4 %) | 17 (24.6 %) | – |
| Voriconazole | 71 (10.6 %) | 32 (46.4 %) | – |
| Caspofungin | 4 (0.6 %) | 6 (8.7 %) | – |
| Amphotericin B | 2 (0.3 %) | 1 (1.4 %) | – |
| Drug combination | 151 (18.5 %) | 19 (21.6 %) | – |
| Route of administration | |||
| Oral | 466 (57.0 %) | 29 (33.0 %) | – |
| Intravenous | 214 (26.2 %) | 37 (42.0 %) | – |
| Oral + intravenous | 138 (16.9 %) | 22 (25.0 %) | – |
| Time of prophylaxis before HSCT mean (SD), days | 10.8 (6.57) | 12.0 (6.43) | – |
E-COG Eastern Cooperative Oncology Group, PAP primary antifungal prophylaxis, SAP secondary antifungal prophylaxis, BM bone marrow, PB peripheral blood, CB cord blood, CMV cytomegalovirus, aGVHD acute graft versus host disease, cGVHD chronic graft versus host disease, IFD invasive fungal diseases, HSCT hematopoietic stem cell transplantation
aOthers include chronic myelomonocytic leukemia (n = 3), T lymphoblastic lymphoma leukemia (n = 2), mixed-lineage leukemia (n = 10), aggressive NK cell leukemia (n = 3), myeloid sarcoma (n = 1), plasmablasticlymphoma (n = 3)
bOthers include infectious disease (n = 18), digestive system diseases (n = 10), cholecystolithiasis (n = 7), mouth disease (n = 8), urological diseases (n = 6), intestinal diseases (n = 7), endocrine diseases (n = 7), surgery (n = 5), cerebrovascular disease (n = 4), tumor (n = 2)
The average duration of antifungal prophylaxis before HSCT was 10.9 ± 6.56 days (10.8 ± 6.57 days in the PAP group and 12.0 ± 6.43 days in the SAP group). Single antifungal agents were the most common prophylaxis regimen (667/818 in the PAP group and 69/88 in the SAP group). Fluconazole (429/818, 64.9 %) and voriconazole (32/88, 46.4 %) were the most widely used agents for PAP and SAP, respectively (Table 1). All antifungal agents were given in accordance with the recommended doses and schedule.
Efficacy of PAP and SAP on the prophylaxis of IFD occurrence
In the 1053 patients, 321 (30.5 %) patients were identified as having an IFD, including 13 (1.2 %) with proven IFD, 81 (7.7 %) with probable IFD, and 227 (21.6 %) with possible IFD. The incidence rates of proven, probable, and possible IFD in the PAP, SAP, and non-prophylaxis groups were 0.7 vs. 0.0 vs. 4.8 %, 7.0 vs. 11.4 vs. 9.5 %, and 15.0 vs. 27.3 vs. 54.4 %, respectively (Table 2). The median time from transplantation to IFD was 45 days in the PAP group (interquartile range (IQR), 16 to 75), 18 days in the SAP group (IQR, 7 to 26), and 12 days in the non-prophylaxis group (IQR, 0 to 93), respectively.
Table 2.
The recurrence rates of IFD in patients from the PAP, SAP, and non-prophylaxis groups
| Characteristic | PAP (N = 818) | SAP (N = 88) | Non-prophylaxis (n = 147) | P value |
|---|---|---|---|---|
| IFD patients | 186 (22.7 %) | 34 (38.6 %) | 101 (68.7 %) | 0.0000 |
| Proven | 6 (0.7 %) | 0 (0.0 %) | 7 (4.8 %) | |
| Probable | 57 (7.0 %) | 10 (11.4 %) | 14 (9.5 %) | |
| Possible | 123 (15.0 %) | 24 (27.3 %) | 80 (54.4 %) | |
| Patients without IFD | 632 (77.3 %) | 54 (61.4 %) | 46 (31.3 %) |
Characteristics of patients with IFD occurrence
Thirteen patients were diagnosed with proven IFD (6 in the PAP group and 7 in the non-prophylaxis group), and 81 patients were diagnosed with probable IFD (57 in the PAP group, 10 in the SAP group, and 14 in the non-prophylaxis group). Among the 94 patients with IFD, 56 were male and 38 were female. The underlying diseases were acute myeloid leukemia (n = 32), acute lymphocytic leukemia (n = 32), myelodysplastic syndrome (n = 10), chronic myeloid leukemia (n = 8), non-Hodgkin’s lymphoma (n = 5), aplastic anemia (n = 5), and others (n = 2). Twenty patients received human leukocyte antigen (HLA)-matched sibling HSCT, 42 patients received HLA-haploidentical HSCT, and 32 patients received HLA-matched unrelated HSCT.
Detailed data regarding the distribution of fungal pathogens and the methods of microbiological diagnoses of IFDs are shown in Table 3. Of the 89 etiological pathogens identified in patients with proven or probable IFDs, 62 (69.7 %) were Aspergillus, including 19 that were exclusively diagnosed with two positive galactomannan tests. Unspecified Aspergillus was the most common mold (31 cases), followed by Aspergillus flavus (5 cases), and Aspergillus fumigatus (2 case) isolates.
Table 3.
Identified etiological pathogens in IFD cases
| Fungal species | Overall population | PAP | SAP | Non-prophylaxis |
|---|---|---|---|---|
| Overall | 89 | 55 | 7 | 27 |
| Candida spp. | 27 | 14 | 3 | 10 |
| Unclassified Candida | 8 | 5 | 0 | 3 |
| Candida tropicalis | 3 | 2 | 0 | 1 |
| Candida albicans | 3 | 2 | 0 | 1 |
| Candida parapsilosis | 3 | 2 | 0 | 1 |
| Candida krusei | 3 | 1 | 1 | 1 |
| Candida glabrata | 1 | 1 | 0 | 0 |
| Candida albicans | 5 | 1 | 2 | 2 |
| Not specifieda | 1 | 0 | 0 | 1 |
| Aspergillus spp. | 62 | 41 | 4 | 17 |
| Positive GM tests only | 19 | 15 | 1 | 3 |
| Aspergillus flavus | 5 | 3 | 0 | 2 |
| Aspergillus versicolor | 1 | 1 | 0 | 0 |
| Aspergillus fumigatus | 2 | 2 | 0 | 0 |
| Aspergillus albicans | 1 | 0 | 0 | 1 |
| Mucor | 1 | 0 | 0 | 1 |
| Cryptococcus | 1 | 1 | 0 | 0 |
| Pityrosporion ovale | 1 | 1 | 0 | 0 |
| Not specifiedb | 31 | 18 | 3 | 10 |
aIncluding cases with yeasts identified in tissue but negative cultures
bIncluding cases with hyphae identified in tissue but negative cultures
The infection sites of all categories of IFD were also analyzed. Of all the 54 infection sites identified in patients with IFDs, the most common location of infection was the lower respiratory tract (64.8 %, 35/54), followed by blood stream infections only (13.0 %, 7/54), central nervous system (5.6 %, 3/54), spleen (1.9 %, 1/54), and three other sites (14.8 %, 8/54). Further analysis showed that Aspergillus is the main pathogen of lower respiratory tract fungal infection (85.7 %, 30/35). In other infection sites, there was no significant difference in the infection rate between Aspergillus and Candida.
Risk factors for proven and probable IFD
The risk factors for proven and probable IFDs among PAP and SAP transplant recipients are presented in Tables 4 and 5. Univariate analyses revealed that HLA-haploidentical HSCT, HLA-matched unrelated HSCT, the use of antithymocyte globulin, prolonged profound neutropenia (>14 days), renal impairment, decreased albumin levels, and Epstein-Barr virus and cytomegalovirus viremia were independent risk factors for proven and probable IFDs in allo-HSCT patients with PAP (P < 0.05). Among the allo-HSCT patients with SAP, the use of antithymocyte globulin was the only obviously significant risk factor (P = 0.043).
Table 4.
Risk factors for proven/probable IFD among allo-HSCT patients in the PAP group
| Factor | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| Patients with proven/probable IFD (n/N) | Incidence of proven/probable IFD (%) | Comparison (P value) | SE | OR (95 % CI) | Comparison (P value) | |
| Age | ||||||
| ≤18 years old | 9/182 | 4.95 | 0.15 | 0.44 | 2.78 (1.18–6.55) | 0.02 |
| >18 years old | 54/636 | 8.49 | ||||
| Transplantation type | ||||||
| HLA-matched related (sibling) | 10/335 | 2.99 | 0.0001 | |||
| Haploidentical | 31/269 | 11.52 | 0.65 | 6.08 (1.69–21.86) | 0.01 | |
| Unrelated | 22/213 | 10.33 | 0.60 | 8.07 (2.50–26.10) | 0.00 | |
| Antithymocyte globulin | ||||||
| Yes | 49/516 | 4.64 | 0.01 | 0.57 | 0.40 (0.13–1.24) | 0.11 |
| No | 14/302 | 9.50 | ||||
| Glucocorticoidsa | ||||||
| Yes | 58/678 | 8.55 | 0.05 | 0.58 | 1.37 (0.44–4.23) | 0.58 |
| No | 5/140 | 3.57 | ||||
| GVHDb | ||||||
| Non-GVHD | 31/428 | 7.24 | 0.15 | 0.40 | 1.63 (0.74–3.58) | 0.22 |
| aGVHD I–II | 16/230 | 6.96 | ||||
| aGVHD III–IV | 8/52 | 15.38 | ||||
| cGVHD local | 2/60 | 3.33 | ||||
| cGVHD extensive | 3/27 | 11.11 | ||||
| Prolonged, profound neutropenia | ||||||
| ANC > 500/mm3 | 1/14 | 7.14 | 0.0006 | |||
| ANC < 500/mm3, <10 days | 6/142 | 4.23 | 1.22 | 0.40 (0.04–4.33) | 0.45 | |
| ANC < 500/mm3, 10–14 days | 4/176 | 2.27 | 1.28 | 0.14 (0.01–1.73) | 0.13 | |
| ANC < 500/mm3, >14 days | 52/486 | 10.70 | 1.16 | 0.99 (0.10–9.60) | 0.99 | |
| EBV viremiac | ||||||
| Yes | 10/62 | 16.13 | 0.03 | 0.44 | 1.35 (0.58–3.17) | 0.49 |
| No | 43/646 | 6.66 | ||||
| Untested | 10/110 | 9.09 | ||||
| CMV viremiac | ||||||
| Yes | 33/267 | 12.36 | 0.004 | 0.33 | 1.18 (0.61–2.26) | 0.63 |
| No | 29/528 | 5.49 | ||||
| Untested | 1/23 | 4.35 | ||||
| Renal impairment | ||||||
| Yes | 10/60 | 16.67 | 0.019 | 0.43 | 1.38 (0.59–3.24) | 0.46 |
| No | 53/758 | 6.99 | ||||
| Decreased albumin | ||||||
| Yes | 39/317 | 12.30 | 0.0001 | 0.31 | 1.98 (1.08–3.62) | 0.03 |
| No | 24/501 | 4.79 | ||||
| Time of IFD prophylaxis | ||||||
| <35 days | 33/396 | 8.33 | 0.60 | 0.30 | 1.33 (0.74–2.39) | 0.35 |
| ≥35 days | 30/416 | 7.21 | ||||
| Drugs for IFD prophylaxis | ||||||
| Fluconazole/fluconazole + itraconazolee | 39/498 | 7.83 | 0.17 | 0.46 | 1.76 (0.72–4.31) | 0.21 |
| Itraconazolee | 14/103 | 13.59 | 0.53 | 3.14 (1.11–8.86) | 0.03 | |
| Voriconazolee | 2/71 | 2.82 | 0.85 | 0.95 (0.18–5.01) | 0.95 | |
| Otherd | 8/144 | 5.56 | ||||
ANC absolute neutrophil count, EBV Epstein-Barr virus
aIncluding dexamethasone, methylprednisolone, prednisone, and hydrocortisone
bBy multivariate analysis, OR is for cGVHD extensive/aGVHD III and IV degree versus cGVHD local/aGVHD I and II degree/non-GVHD
cBy multivariate analysis, OR is for with EBV viremia versus without EBV viremia or untested/CMV viremia versus without CMV viremia or untested
dIncluding caspofungin, micafungin, amphotericin B, fluconazole + caspofungin, fluconazole + micafungin, fluconazole + voriconazole, itraconazole + micafungin, voriconazole + caspofungin, and fluconazole + caspofungin + voriconazole
eCompared with others
The italicized data reflected significant difference
Table 5.
Risk factors for proven/probable IFD among allo-HSCT patients in SAP group
| Factor | Univariate analysis | Muitivariate analysis | ||||
|---|---|---|---|---|---|---|
| Patients with proven/probable IFD (n/N) | Incidence of proven/probable IFD (%) | Comparison (P value) | SE | OR (95 % CI) | Comparison (P value) | |
| Transplantation type | ||||||
| HLA-matched related (sibling) | 1/35 | 2.86 | 0.08 | |||
| Haploidentical | 5/26 | 19.23 | 1.77 | 2.48 (0.08–79.46) | 0.61 | |
| Unrelated | 4/27 | 14.81 | 1.77 | 2.24 (0.07–72.11) | 0.65 | |
| Antithymocyte globulin | ||||||
| Yes | 9/52 | 17.31 | 0.04 | 1.68 | 2.49 (0.09–67.66) | 0.59 |
| No | 1/36 | 2.78 | ||||
| Prolonged, profound neutropenia | ||||||
| ANC > 500/mm3 | 0/1 | 0.00 | 0.23 | 1.13 | 5.90 (0.65–53.89) | 0.12 |
| ANC < 500/mm3, <10 days | 1/18 | 5.56 | ||||
| ANC < 500/mm3, 10–14 days | 0/17 | 0.00 | ||||
| ANC < 500/mm3, >14 days | 9/52 | 17.31 | ||||
| Liver impairment | ||||||
| Yes | 5/24 | 20.83 | 0.12 | 0.74 | 2.07 (0.49–8.79) | 0.33 |
| No | 5/64 | 7.81 | ||||
| Time of IFD prophylaxis | ||||||
| <35 days | 5/40 | 12.50 | 1.00 | 0.76 | 1.10 (0.25–4.86) | 0.90 |
| ≥35 days | 5/48 | 10.42 | ||||
| Drugs for IFD prophylaxis | ||||||
| Fluconazole/itraconazole/ | 3/27 | 11.11 | 1.00 | 0.96 | 0.87 (0.13–5.64) | 0.88 |
| Fluconazole + itraconazoleb | ||||||
| Voriconazoleb | 4/32 | 12.50 | 0.91 | 1.50 (0.25–8.96) | 0.66 | |
| Othersa | 3/29 | 10.34 | ||||
aIncluding voriconazole + caspofungin and fluconazole + caspofungin + voriconazole
bCompared with others
The italicized data reflected significant difference
Multivariate analyses demonstrated that independent risk factors for proven and probable IFDs in allo-HSCT patients with PAP were age >18 years (odds ratio (OR), 2.78; 95 % confidence interval (CI), 1.18–6.55; P < 0.05), having an HLA-matched unrelated donor (OR, 8.07; 95 % CI, 2.50–26.10; P < 0 .01), having an HLA-haploidentical donor (OR, 6.08; 95 % CI, 1.69–21.86; P < 0.01), decreased albumin levels (OR, 1.98; 95 % CI, 1.08–3.62; P < 0.05), and the use of itraconazole as the antifungal prophylactic agent (OR, 3.41; 95 % CI, 1.11–8.86; P < 0 .05) (Table 4). Among the SAP transplant recipients, there was no significant risk factor for proven or probable IFD.
Overall antifungal therapy
A total of 321 IFD patients were provided therapeutic antifungal treatment. A single antifungal agent was used for treatment in 116 (36.1 %) patients; 144 (44.9 %) patients required two agents, and 61 (19.0 %) patients required three or more agents during the course of their treatment. Among the PAP transplant recipients, itraconazole (34.4 %, 64/186 in all-category IFD; 46.1 %, 29/63 in proven or probable IFD) was the most popular agent for the initial therapy. Among the SAP and non-prophylaxis transplant recipients, voriconazole (36.3 %, 49/135 in all-category IFD; 25.8 %, 8/31 in proven or probable IFD) was the most common agent for the initial therapy. Whether the patients in PAP, SAP, or non-prophylaxis groups, voriconazole as a single agent or in combination with other agents (35.8 %, 115/321) was the most popular agent for antifungal treatment.
The median overall treatment duration in patients who started and completed antifungal treatment while hospitalized was 36 days (IQR, 21–65) for PAP transplant recipients, 39 days (IQR, 24–58) for SAP transplant recipients, and 35 days (IQR, 20–67) for non-prophylaxis transplant recipients. A total of 175 patients (54.5 %, 94 in the PAP group, 20 in the SAP group, and 61 in the non-prophylaxis group) continued treatment after leaving the hospital.
Outcomes including mortality
At the end of follow-up, 171 patients had died, resulting in an overall mortality rate of 16.2 % (171/1053). Compared with the overall study population, the mortality rate was markedly higher in patients with proven (5/13; 38.5 %), probable (19/81; 23.5 %), or possible (58/227; 25.6 %) IFDs. Furthermore, the mortality rate among patients treated for suspected IFD despite failing to meet the EORTC/MSG diagnostic criteria [16] (25/166; 15.1 %) was higher than that among patients who were not suspected of having IFD and, therefore, not treated with antifungal agents (64/566; 11.3 %) (Fig. 1, P < 0.001). In addition, we compared the incidence of OS in the PAP, SAP, and no prophylaxis groups. There was a significant difference among the three groups (85.07 vs. 78.80 vs. 74.82 %, respectively, Fig. 2, P = 0.01).
Fig. 1.
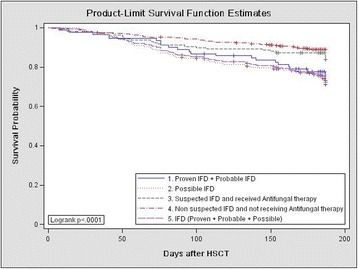
Overall survival of patients with proven/probable IFD, possible IFD, or suspected IFD and who received antifungal therapy; those with no suspected IFD and who did not receive antifungal therapy; and those without any IFD (proven, probable, or possible)
Fig. 2.
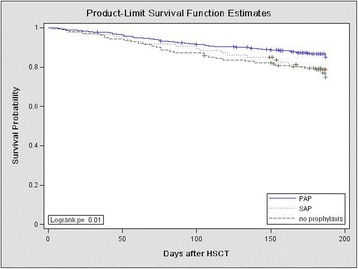
Overall survival of allo-HSCT patients receiving PAP, SAP, or no prophylaxis
Discussion
Invasive infections remain major infectious threats to patients undergoing allo-HSCT and are associated with high fatality rates [23–28]. The diagnosis of IFDs continues to be difficult to establish because they do not manifest with specific clinical or radiographic signs or symptoms [29]. In the recent years, there has been a concerted effort to identify alternative procedures for the future diagnosis of fungus. They include the targeting of fungal antigens by enzyme-linked immunosorbent assay (ELISA) or lateral flow devices (LFDs) [30], detection of siderophores [31], and amplification of fungal nucleic acids from tissue and body fluids [32] as well as application of matrix-assisted laser desorption ionization TOF mass spectrometry (MALDI-TOF MS) [33]. Each of the above methods has advantages and disadvantages, and not all of which have entered clinical practice [34]. The difficulty in obtaining a timely diagnosis as well as the high morbidity and mortality rates associated with IFDs provide a rationale for antifungal prophylaxis in patients undergoing allo-HSCT. Nonetheless, antifungal prophylaxis remains a topic of some controversy, with no clear consensus among different centers [35–37].
The CAESAR study is the first population-based and the largest prospective observational study of the incidences of IFDs in patients receiving HSCT [7]. This observational study was conducted as a part of the CAESAR study and focused on the prophylaxis of IFDs in patients who underwent allo-HSCT. In the present analysis of adults and children at risk for IFD due to allo-HSCT, non-prophylaxis transplant recipients had a higher incidence of proven/probable/possible IFD than PAP and SAP transplant recipients at 6 months (68.6 vs. 22.7 and 38.6 %, P = 0.0000). This result was in accord with the classic randomized clinical trials on PAP in HSCT recipients and patients with hematological malignancies [38–40]. Furthermore, the OS was significantly different among PAP, SAP, and no prophylaxis patients. It was revealed that antifungal prophylaxis was helpful to reduce the incidence of IFD and improve the survival of patients after transplantation.
The most recently published European guidelines pointed out that SAP should be administered to patients with a previous IFD to prevent recurrence of the previous IFD or onset of a new IFD during a new at-risk phase, mainly referring to a prolonged neutropenic period induced by chemotherapy or a phase of severe immunosuppression after allo-HSCT [16]. Several studies have reported success rates for SAP, which was proven to be effective in preventing IFD recurrence [41, 42]. In the study, the cumulative incidence of IFD increased particularly rapidly during the first month after transplantation in the SAP group and non-prophylaxis group, suggesting that this is a high-risk period during which health care providers should pay particularly close attention to signs of emerging IFD in SAP and non-prophylaxis patients. On the contrary, the median time of IFD occurrence in the PAP group was 45 days after HSCT. This finding indicated that patients who had a previous IFD and had no antifungal prophylaxis were more likely to experience a breakthrough fungal infection in the early stage after transplantation.
As 22.7 % of patients treated with PAP and 38.6 % of patients treated with SAP in the study went on to develop proven, probable, or possible IFD, there remains an unmet need for education about the appropriate timing and choice of antifungal agent for prophylaxis in China. Among PAP transplant recipients, the most commonly prescribed prophylactic agents were fluconazole and itraconazole, which may be less effective than posaconazole [43]. The use of itraconazole as the antifungal prophylactic agent also proved the independent risk factor for IFD occurrence in our study. An update to the cost-effectiveness of posaconazole vs. fluconazole or itraconazole in the prevention of IFD among neutropenic patients in the USA has shown that posaconazole is a cost-effective alternative to fluconazole or itraconazole in the prevention of IFD among neutropenic patients [44]. Among SAP transplant recipients, there was no significant risk factor for proven or probable IFD. The characteristics of the observational study and the small sample size of the patients treated with SAP may be the main reasons. Prospective and randomized studies assessing the risk factors for SAP are needed in the future.
The epidemiological characteristics of IFD continue to evolve in transplant patients. A major contributor is the widespread use of azole prophylaxis since the early 1990s, which results in less candidiasis but more frequent mold infections in hematologic malignancies [25, 45]. In most [46–48] but not all [49] cases, Aspergillus spp. represent the most frequent cause of fungal-related morbidity in patients with HSCT. In our study, Aspergillus spp. also predominated in culture-proven or histologically proven pathogens and was more than two times as common as Candida spp. The mortality among patients who developed IFD (24.1 %) was almost double than that in the overall population of HSCT patients (15.0 %), and regression analysis confirmed that the development of IFD is a significant independent risk factor for death, highlighting the grave prognosis of those with IFD and the need for a timely diagnosis and prompt treatment. The incidence rates of invasive aspergillosis, however, need to be interpreted with caution as establishing this diagnosis often requires invasive procedures that are difficult to perform in severely ill patients. The low rate of proven/probable IFD in the present study suggests the need to improve diagnostic techniques to treat IFD as early and accurately as possible. Under the existing conditions, in order to reduce the IFD-related mortality of allo-HSCT patients, preemptive antifungal therapy should be given to patients with possible or suspected IFD.
The current study has several limitations. The main limitation was its observational nature. Confounding factors cannot be controlled effectively in observational studies, frequently due to biased selection of patients or treatment protocols. In the analysis of mortality by antifungal therapy, the uncontrolled study design means that no firm conclusions can be drawn regarding the relative effect of different treatments. Another limitation was the relatively short duration. The fact that the overall treatment duration was not recorded in patients who were still taking antifungal treatment after discharge from the hospital further limited data analysis. A longer follow-up would have allowed a more comprehensive analysis of patients who developed IFD after discharge. Procedures for diagnosis, prophylaxis, and treatment of IFD were not prespecified in the protocol but were performed according to usual practice and local clinical guidelines [19]. Furthermore, in some centers, diagnostic procedures were conducted only among patients with suspected clinical signs or symptoms of IFD. The observational study also meant that the diagnosis of specific infections, although guided by EORTC/MSG 2008 criteria, was limited to the data collected according to local hospitals’ usual practice; there was no mandatory requirement for diagnostic microbiological testing or the use of a centralized laboratory to validate the results.
Conclusions
In conclusion, the results of the present observational study indicate that prophylaxis of IFD among patients receiving allo-HSCT for hematological malignancy in China is broadly in line with the recommended practice and is helpful to reduce the incidence of IFD and improve the OS of patients after transplantation. For patients with a history of IFD, active and effective prevention of fungal infections is particularly important because these patients have a higher incidence of IFD occurrence and a shorter incubation period. Different from the traditional view, for the allo-HSCT patients with PAP, itraconazole was a less effective treatment than other antifungal drugs. Aspergillus spp. represents the most common type of fungal infection in patients with allo-HSCT. Due to the limited diagnostic techniques, it is necessary to give antifungal therapy to patients who do not meet the EORTC/MSG 2008 criteria but show clinical evidence of fungal infection.
Acknowledgements
The authors would like to thank the patients and investigators at each study site, without whom this study would not have been possible. The CAESAR study group investigators not listed as authors were as follows: Jing Chen, Shanghai Children’s Medical Center, Shanghai; Zhixiang Shen, Rui Jin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai; Juan Li and Waiyi Zou, The First Affiliated Hospital of Sun Yat-Sen University, Guangzhou; Chun Wang, Jun Yang, and Yu Cai, The First People’s Hospital of Shanghai, Shanghai; Yang Xiao and Yonghua Li, The General Hospital of Guangzhou Military Command of PLA, Guangzhou; Yongping Song and Yuewen Fu, Henan Tumor Hospital affiliated to Zhengzhou University, Zhengzhou; Kang Yu, Xudong Hu, and Xingzhou Ren, The First Affiliated Hospital of Wenzhou Medical College, Wenzhou; Huisheng Ai and Jianhui Qiao, Chinese PLA 307 Hospital, Beijing; Jianmin Wang and Xianmin Song, Changhai Hospital of the Second Military Medical University, Shanghai; Hai Bai and Chunbang Wang, The General Hospital of Lanzhou Military Area, Lanzhou; Yongmin Tang and Heping Shen, Children’s Hospital of Zhejiang University Medical school, Hangzhou; Jianyong Li and Xiaoyan Zhang, Jiangsu Province Hospital, Nanjing; Xin Du and Chengwei Luo, Guangdong General Hospital, Guangzhou; Jian Ouyang and Yong Xu, Nanjing Drum Tower Hospital, Nanjing; Huo Tan and Runhui Zheng, The First Affiliated Hospital of Guangzhou Medical University, Guangzhou; Ting Liu and Juan Xu, West China Hospital, Sichuan University, Chengdu; Jin Zhou and Liming Li, The First Hospital of Harbin Medical University, Harbin; Jun Ma, Harbin Hematologic Tumor Institution, Harbin; Zhuogang Liu, Shengjing Hospital of China Medical University, Shenyang; Jianda Hu, Fujian Medical University Union Hospital, Fuzhou; Ping Zou and Yong You, Wuhan Union Hospital, Huazhong University of Science and Technology, Wuhan; Lin Liu and Li Wang, The First Affiliated Hospital of Chongqing Medical University, Chongqing; Yan Li and Ran Gao, The First Affiliated Hospital of China Medical University, Shenyang; Zhanxiang Liu, Chinese PLA General Hospital (301 Hospital), Beijing; Mangju Wang, The First Hospital of Peking University, Beijing; Guopan Yu, Nanfang Hospital, Nanfang Medical University, Guangzhou; Ling Wang, Rui Jin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai; Jun Wang, The First Affiliated Hospital of Soochow University, Suzhou; Guixin Zhang, Institute of Hematology and Blood Diseases Hospital, Chinese Academy of Medical Sciences, Tianjin; Xia Qin, Shanghai Children’s Medical Center, Shanghai; Liya Ma and Wangzhuo Xie, The First Affiliated Hospital of Medical School of Zhejiang University, Hangzhou, China.
Funding
This work was supported by Merck Sharp & Dohme (China), who sponsored the study. The support for the manuscript development was provided by Merck & Co., Inc. (Whitehouse Station, NJ).
Availability of data and materials
The datasets supporting the conclusions of this article are included within the article.
Authors’ contributions
XH and XZ contributed to the conception and design of the study. FM, MZ, HH, DW, LY, and HR were involved in the development of the methodology and participated in the analysis. LG and YS interpreted the data and drafted the paper. All authors contributed in the writing, review, and revision of the article and final approval for submission.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
This observational study was conducted in accordance with the Declaration of Helsinki, International Conference on Harmonization Good Clinical Practice, and nationally mandated ethical requirements. The study protocol and informed consent document were reviewed and approved by the ethics committee of Peking University People’s Hospital. All participating institutions obtained ethical approval separately before initiation of the study. All study participants provided informed consent.
Abbreviations
- allo-HSCT
Allo-genetic hematopoietic stem cell transplantation
- CAESAR
China Assessment of Antifungal Therapy in Hematological Disease
- ELISA
Enzyme-linked immunosorbent assay
- EORTC/MSG
European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group
- HLA
Human leukocyte antigen
- IFDs
Invasive fungal diseases
- LFDs
Lateral flow devices
- MALDI-TOF MS
Matrix-assisted laser desorption ionization TOF mass spectrometry
- OS
Overall survival
- PAP
Primary antifungal prophylaxis
- SAP
Secondary antifungal prophylaxis.
References
- 1.Girmenia C, Raiola AM, Piciocchi A, Algarotti A, Stanzani M, Cudillo L, et al. Incidence and outcome of invasive fungal diseases after allogeneic stem cell transplantation: a prospective study of the Gruppo Italiano Trapianto Midollo Osseo (GITMO) Biol Blood Marrow Transplant. 2014;20:872–80. doi: 10.1016/j.bbmt.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 2.Lv M, Huang XJ. Allogeneic hematopoietic stem cell transplantation in China: where we are and where to go. J Hematol Oncol. 2012;5:10. doi: 10.1186/1756-8722-5-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lin R, Liu Q. Diagnosis and treatment of viral diseases in recipients of allogeneic hematopoietic stem cell transplantation. J Hematol Oncol. 2013;6:94. doi: 10.1186/1756-8722-6-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kontoyiannis DP, Marr KA, Park BJ, Alexander BD, Anaissie EJ, Walsh TJ, et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001–2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis. 2010;50:1091–100. doi: 10.1086/651263. [DOI] [PubMed] [Google Scholar]
- 5.Neofytos D, Horn D, Anaissie E, Steinbach W, Olyaei A, Fishman J, et al. Epidemiology and outcome of invasive fungal infection in adult hematopoietic stem cell transplant recipients: analysis of Multicenter Prospective Antifungal Therapy (PATH) Alliance registry. Clin Infect Dis. 2009;48:265–73. doi: 10.1086/595846. [DOI] [PubMed] [Google Scholar]
- 6.Pagano L, Caira M, Nosari A, Van Lint MT, Candoni A, Offidani M, et al. Fungal infections in recipients of hematopoietic stem cell transplants: results of the SEIFEM B-2004 studyeSorveglianza Epidemiologica Infezioni Fungine Nelle Emopatie Maligne. Clin Infect Dis. 2007;45:1161–70. doi: 10.1086/522189. [DOI] [PubMed] [Google Scholar]
- 7.Sun Y, Meng F, Han M, Zhang X, Yu L, Huang H, et al. Epidemiology, management, and outcome of invasive fungal disease in patients undergoing hematopoietic stem cell transplantation in China: a multicenter prospective observational study. Biol Blood Marrow Transplant. 2015;21:1117–26. doi: 10.1016/j.bbmt.2015.03.018. [DOI] [PubMed] [Google Scholar]
- 8.Lai Y, Chen Y, Hu D, Jiang M, Liu Q, Liu L, et al. Multicenter phase ii study of a combination of cyclosporine a, methotrexate and mycophenolate mofetil for GVHD prophylaxis: results of the Chinese Bone Marrow Transplant Cooperative Group (CBMTCG) J Hematol Oncol. 2014;7:59. doi: 10.1186/s13045-014-0059-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chang Y, Zhao X, Xu L, Zhang X, Wang Y, Han W, et al. Donor-specific anti-human leukocyte antigen antibodies were associated with primary graft failure after unmanipulated haploidentical blood and marrow transplantation: a prospective study with randomly assigned training and validation sets. J Hematol Oncol. 2015;8:84. doi: 10.1186/s13045-015-0182-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arendrup MC, Dzajic E, Jensen RH, Johansen HK, Kjaeldgaard P, Knudsen JD, et al. Epidemiological changes with potential implication for antifungal prescription recommendations for fungaemia: data from a nationwide fungaemia surveillance programme. Clin Microbiol Infect. 2013;19:E343–53. doi: 10.1111/1469-0691.12212. [DOI] [PubMed] [Google Scholar]
- 11.Mousset S, Buchheidt D, Heinz W, Ruhnke M, Cornely OA, Egerer G, et al. Treatment of invasive fungal infections in cancer patients-updated recommendations of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Oncology (DGHO) Ann Hematol. 2014;93:13–32. doi: 10.1007/s00277-013-1867-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alothman AF, Al-Musawi T, Al-Abdely HM, Salman JA, Almaslamani M, Yared N, et al. Clinical practice guidelines for the management of invasive Candida infections in adults in the Middle East region: expert panel recommendations. J Infect Public Health. 2014;7:6–19. doi: 10.1016/j.jiph.2013.08.002. [DOI] [PubMed] [Google Scholar]
- 13.Pappas PG, Kauffman CA, Andes D, Benjamin DJ, Jr, Calandra TF, Edwards JE, Jr, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis. 2009;48:503–35. doi: 10.1086/596757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cornely OA, Böhme A, Buchheidt D, Einsele H, Heinz WJ, Karthaus M, et al. Primary prophylaxis of invasive fungal infections in patients with hematologic malignancies. Recommendations of the Infectious Diseases Working Party of the German Society for Haematology and Oncology. Haematologica. 2009;94:113–22. doi: 10.3324/haematol.11665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marr KA, Bow E, Chiller T, Maschmeyer G, Ribaud P, Segal B, et al. Fungal infection prevention after hematopoietic cell transplantation. Bone Marrow Transplant. 2009;44:483–7. doi: 10.1038/bmt.2009.259. [DOI] [PubMed] [Google Scholar]
- 16.Maertens J, Marchetti O, Herbrecht R, Cornely OA, Flückiger U, Frêre P, et al. European guidelines for antifungal management in leucemia and hematopoietic stem cell transplant recipients: summary of the ECIL 3-2009 update. Bone Marrow Transplant. 2011;46:709–18. doi: 10.1038/bmt.2010.175. [DOI] [PubMed] [Google Scholar]
- 17.Baden LR, Bensinger W, Angarone M, Casper C, Dubberke ER, Freifeld AG, et al. Prevention and treatment of cancer-related infections. J Natl Compr Canc Netw. 2012;10:1412–45. doi: 10.6004/jnccn.2012.0146. [DOI] [PubMed] [Google Scholar]
- 18.Sun Y, Huang H, Chen J, Li J, Ma J, Li J, et al. Invasive fungal infection in patients receiving chemotherapy for hematological malignancy: a multicenter, prospective, observational study in China. Tumor Biol. 2015;36:757–67. doi: 10.1007/s13277-014-2649-7. [DOI] [PubMed] [Google Scholar]
- 19.De Pauw B, Walsh TJ, Donnelly JP, Stevens DA, Edwards JE, Calandra T, et al. Revised definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis. 2008;46:1813–21. doi: 10.1086/588660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.The Chinese Invasive Fungal Infection Committee The diagnostic criteria and treatment guideline of invasive fungal infection in patients with Hematological disease/malignances (3rd version) Zhonghua Nei Ke Za Zhi [Chinese Journal of Internal Medicine] 2010;49:451–4. [Google Scholar]
- 21.Girmenia C, Barosi G, Piciocchi A, Arcese W, Aversa F, Bacigalupo A, et al. Primary prophylaxis of invasive fungal diseases in allogeneic stem cell transplantation: revised recommendations from a consensus process by Gruppo Italiano Trapianto Midollo Osseo (GITMO) Biol Blood Marrow Transplant. 2014;20:1080–8. doi: 10.1016/j.bbmt.2014.02.018. [DOI] [PubMed] [Google Scholar]
- 22.Allinson K, Kolve H, Gumbinger HG, Vormoor HJ, Ehlert K, Groll AH. Secondary antifungal prophylaxis in paediatric allogeneic haematopoietic stem cell recipients. J Antimicrob Chemother. 2008;61:734–42. doi: 10.1093/jac/dkm521. [DOI] [PubMed] [Google Scholar]
- 23.Huang XJ. Current status of haploidentical stem cell transplantation for leukemia. J Hematol Oncol. 2008;1:27. doi: 10.1186/1756-8722-1-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wan L, Zhang Y, Lai Y, Jiang M, Song Y, Zhou J, et al. Effect of granulocyte-macrophage colony-stimulating factor on prevention and treatment of invasive fungal disease in recipients of allogeneic stem-cell transplantation: a prospective multicenter randomized phase IV trial. J Clin Oncol. 2015. [Epub ahead of print]. [DOI] [PubMed]
- 25.Montesinos P, Rodríguez-Veiga R, Boluda B, Martínez-Cuadrón D, Cano I, Lancharro A, et al. Incidence and risk factors of post-engraftment invasive fungal disease in adult allogeneic hematopoietic stem cell transplant recipients receiving oral azoles prophylaxis. Bone Marrow Transplant. 2015;50:1465–72. doi: 10.1038/bmt.2015.181. [DOI] [PubMed] [Google Scholar]
- 26.Wang CH, Kan LP, Lin HA, Chang FY, Wang NC, Lin TY, et al. Clinical efficacy and safety of primary antifungal prophylaxis with posaconazole versus fluconazole in allogeneic blood hematopoietic stem cell transplantation recipients—a retrospective analysis of a single medical center in Taiwan. Microbiol Immunol Infect. 2014 doi: 10.1016/j.jmii.2014.07.009. [DOI] [PubMed] [Google Scholar]
- 27.Girmenia C, Ferretti A, Barberi W. Epidemiology and risk factors for invasive fungal diseases in hematopoietic stem cell transplantation. Curr Opin Hematol. 2014;21:459–65. doi: 10.1097/MOH.0000000000000086. [DOI] [PubMed] [Google Scholar]
- 28.Gao L, Zhang C, Gao L, Liu Y, Su Y, Wang S, et al. Favorable outcome of haploidentical hematopoietic stem cell transplantation in Philadelphia chromosome-positive acute lymphoblastic leukemia: a multicenter study in Southwest China. J Hematol Oncol. 2015;8:90. doi: 10.1186/s13045-015-0186-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Aguado JM, Vázquez L, Fernández-Ruiz M, Villaescusa T, Ruiz-Camps I, Barba P, et al. Serum galactomannan versus a combination of galactomannan and polymerase chain reaction-based Aspergillus DNA detection for early therapy of invasive aspergillosis in high-risk hematological patients: a randomized controlled trial. Clin Infect Dis. 2015;60:405–14. doi: 10.1093/cid/ciu833. [DOI] [PubMed] [Google Scholar]
- 30.White PL, Parr C, Thornton C, Barnes RA. Evaluation of real-time PCR, galactomannan enzyme-linked immunosorbent assay (ELISA), and a novel lateral-flow device for diagnosis of invasive aspergillosis. J Clin Microbiol. 2013;51:1510–6. doi: 10.1128/JCM.03189-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Petrik M, Franssen GM, Haas H, Laverman P, Hörtnagl C, Schrettl M, et al. Preclinical evaluation of two 68Ga-siderophores as potential radiopharmaceuticals for Aspergillus fumigatus infection imaging. Eur J Nucl Med Mol Imaging. 2012;39:1175–83. doi: 10.1007/s00259-012-2110-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Johnson G, Nolan T, Bustin SA. Real-time quantitative PCR, pathogen detection and MIQE. Methods Mol Biol. 2013;943:1–16. doi: 10.1007/978-1-60327-353-4_1. [DOI] [PubMed] [Google Scholar]
- 33.Marinach-Patrice C, Fekkar A, Atanasova R, Gomes J, Djamdjian L, Brossas JY, et al. Rapid species diagnosis for invasive candidiasis using mass spectrometry. PLoS ONE. 2010;5:e8862. doi: 10.1371/journal.pone.0008862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Johnson G, Ferrini A, Dolan SK, Nolan T, Agrawal S, Doyle S, et al. Biomarkers for invasive aspergillosis: the challenges continue. Biomark Med. 2014;8:429–51. doi: 10.2217/bmm.13.129. [DOI] [PubMed] [Google Scholar]
- 35.Döring M, Eikemeier M, Cabanillas Stanchi KM, Hartmann U, Ebinger M, Schwarze CP, et al. Antifungal prophylaxis with posaconazole vs. fluconazole or itraconazole in pediatric patients with neutropenia. Eur J Clin Microbiol Infect Dis. 2015;34:1189–200. doi: 10.1007/s10096-015-2340-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fleming S, Yannakou CK, Haeusler GM, Clark J, Grigg A, Heath CH, et al. Consensus guidelines for antifungal prophylaxis in haematological malignancy and haemopoietic stem cell transplantation, 2014. Intern Med J. 2014;44:1283–97. doi: 10.1111/imj.12595. [DOI] [PubMed] [Google Scholar]
- 37.Vehreschild MJ, von Bergwelt-Baildon M, Tran L, Shimabukuro-Vornhagen A, Wisplinghoff H, Bangard C, et al. Feasibility and effectiveness of posaconazole prophylaxis in combination with micafungin bridging for patients undergoing allogeneic stem cell transplantation: a 6-yr analysis from the cologne cohort for neutropenic patients. Eur J Haematol. 2014;93:400–6. doi: 10.1111/ejh.12368. [DOI] [PubMed] [Google Scholar]
- 38.Tragiannidis A, Dokos C, Lehrnbecher T, Groll AH, Groll AH. Antifungal chemoprophylaxis in children and adolescents with haematological malignancies and following allogeneic haematopoietic stem cell transplantation. Drugs. 2012;72:685–704. doi: 10.2165/11599810-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 39.Wang JF, Xue Y, Zhu XB, Fan H. Efficacy and safety of echinocandins versus triazoles for the prophylaxis and treatment of fungal infections: a meta-analysis of RCTs. Eur J Clin Microbiol Infect Dis. 2015;34:651–9. doi: 10.1007/s10096-014-2287-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Takagi S, Araoka H, Uchida N, Uchida Y, Kaji D, Ota H, et al. A prospective feasibility study of primary prophylaxis against invasive fungal disease with voriconazole following umbilical cord blood transplantation with fludarabine-based conditioning. Int J Hematol. 2014;99:652–8. doi: 10.1007/s12185-014-1529-7. [DOI] [PubMed] [Google Scholar]
- 41.Liu Q, Lin R, Sun J, Xiao Y, Nie D, Zhang Y, et al. Antifungal agents for secondary prophylaxis based on response to initial antifungal therapy in allogeneic hematopoietic stem cell transplant recipients with prior pulmonary aspergillosis. Biol Blood Marrow Transplant. 2014;20:1198–203. doi: 10.1016/j.bbmt.2014.04.016. [DOI] [PubMed] [Google Scholar]
- 42.Vehreschilda JJ, Sieniawski M, Reuterb S, Arenza D, Reichertd D, Maertense J, et al. Efficacy of caspofungin and itraconazole as secondary antifungal prophylaxis: analysis of data from a multinational case registry. Int J Antimicrobl Agents. 2009;34:446–50. doi: 10.1016/j.ijantimicag.2009.06.025. [DOI] [PubMed] [Google Scholar]
- 43.Bow EJ, Vanness DJ, Slavin M, Cordonnier C, Cornely OA, Marks DI, et al. Systematic review and mixed treatment comparison meta-analysis of randomized clinical trials of primary oral antifungal prophylaxis in allogeneic hematopoietic cell transplant recipients. BMC Infect Dis. 2015;15:128. doi: 10.1186/s12879-015-0855-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sung AH, Marcella SW, Xie Y. An update to the cost-effectiveness of posaconazole vs fluconazole or itraconazole in the prevention of IFD among neutropenic patients in the United States. J Med Econ. 2015;18:341–8. doi: 10.3111/13696998.2014.1000460. [DOI] [PubMed] [Google Scholar]
- 45.Groll AH, Castagnola E, Cesaro S, Dalle JH, Engelhard D, Hope W, et al. Fourth European Conference on Infections in Leukaemia (ECIL-4): guidelines for diagnosis, prevention, and treatment of invasive fungal diseases in paediatric patients with cancer or allogeneic haemopoietic stem-cell transplantation. Lancet Oncol. 2014;15:e327–40. doi: 10.1016/S1470-2045(14)70017-8. [DOI] [PubMed] [Google Scholar]
- 46.van Hal SJ, Gilroy NM, Morrissey CO, Worth LJ, Szer J, Tam CS, et al. Survey of antifungal prophylaxis and fungal diagnostic tests employed in malignant haematology and haemopoietic stem cell transplantation (HSCT) in Australia and New Zealand. Intern Med J. 2014;44:1277–82. doi: 10.1111/imj.12594. [DOI] [PubMed] [Google Scholar]
- 47.Held J, Schmidt T, Thornton CR, Kotter E, Bertz H. Comparison of a novel Aspergillus lateral-flow device and the Platelia® galactomannan assay for the diagnosis of invasive aspergillosis following haematopoietic stem cell transplantation. Infection. 2013;41:1163–9. doi: 10.1007/s15010-013-0472-5. [DOI] [PubMed] [Google Scholar]
- 48.Ji Y, Xu LP, Liu DH, Chen YH, Han W, Zhang XH, et al. Positive results of serum galactomannan assays and pulmonary computed tomography predict the higher response rate of empirical antifungal therapy in patients undergoing allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2011;17:759–64. doi: 10.1016/j.bbmt.2010.11.002. [DOI] [PubMed] [Google Scholar]
- 49.Omer AK, Ziakas PD, Anagnostou T, Coughlin E, Kourkoumpetis T, McAfee SL, et al. Risk factors for invasive fungal disease after allogeneic hematopoietic stem cell transplantation: a single center experience. Biol Blood Marrow Transplant. 2013;19:190–6. doi: 10.1016/j.bbmt.2013.05.018. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets supporting the conclusions of this article are included within the article.


