Abstract
BACKGROUND/OBJECTIVES
Corn silk (CS) extract contains large amounts of maysin, which is a major flavonoid in CS. However, studies regarding the effect of CS extract on cholesterol metabolism is limited. Therefore, the purpose of this study was to determine the effect of CS extract on cholesterol metabolism in C57BL/6J mouse fed high-fat diets.
MATERIALS/METHODS
Normal-fat group fed 7% fat diet, high-fat (HF) group fed 25% fat diet, and high-fat with corn silk (HFCS) group were orally administered CS extract (100 mg/kg body weight) daily. Serum and hepatic levels of total lipids, triglycerides, and total cholesterol as well as serum free fatty acid, glucose, and insulin levels were determined. The mRNA expression levels of acyl-CoA: cholesterol acyltransferase (ACAT), cholesterol 7-alpha hydroxylase (CYP7A1), farnesoid X receptor (FXR), lecithin cholesterol acyltransferase (LCAT), low-density lipoprotein receptor, 3-hyroxy-3-methylglutaryl-coenzyme A reductase (HMG-CoA reductase), adiponectin, leptin, and tumor necrosis factor α were determined.
RESULTS
Oral administration of CS extract with HF improved serum glucose and insulin levels as well as attenuated HF-induced fatty liver. CS extracts significantly elevated mRNA expression levels of adipocytokines and reduced mRNA expression levels of HMG-CoA reductase, ACAT, and FXR. The mRNA expression levels of CYP7A1 and LCAT between the HF group and HFCS group were not statistically different.
CONCLUSIONS
CS extract supplementation with a high-fat diet improves levels of adipocytokine secretion and glucose homeostasis. CS extract is also effective in decreasing the regulatory pool of hepatic cholesterol, in line with decreased blood and hepatic levels of cholesterol though modulation of mRNA expression levels of HMG-CoA reductase, ACAT, and FXR.
Keywords: Corn silk, maysin, cholesterol, high-fat, adiponectin
INTRODUCTION
Prolonged high-fat diet consumption causes increases in adipose tissue size and body weight. Enlarged adipose tissue secretes more leptin, tumor necrosis factor α (TNF-α), and less adiponectin [1]. Altered adipocytokines due to weight gain play an important role in lipid and cholesterol metabolism, with implications for diabetes and cardiovascular health [1,2].
Corn silk (CS), which is the yellowish thread-like strands from corn herbal hwabongwa (Zey mays L.), has been used as treatments for obesity, kidney-related diseases, neurodegenerative diseases, and diabetes [3,4,5,6,7,8,9,10,11,12,13]. CS extract includes various bio-functional substances such as tartaric acid, isoquercitrin, allantoin, stigmasterol, hordenine, maysin, resin, cryptoxanthin, and anticyanins [14,15]. CS extract also contains large amounts of maysin, which is a major flavonoid in CS [16]. Maysin is a flavone glycoside containing luteolin, a biologically active compound, attached to a carbohydrate [17].
Domestic research on the bio-function of CS or maysin has investigated the relationships between CS and radiation-induced oxidative stress [12], neuroprotective effects [13], flavonoids [15], and adipogenesis [18,19], Until now, only corn germ extract from maize has received individual recognition as having bioactive Class 2 functionality associated with "skin- protection" from the Korea Food and Drug Administration.
In studies regarding maysin or CS and lipid metabolism, CS water extract showed inhibitory effects against adipogenesis and lipids through regulation of CCAAT/enhancer binding protein α (C/EBPα) and Peroxisome proliferator activated receptor γ (PPARγ) expression [18,19]. However, studies on cholesterol metabolism and CS are limited. Therefore, the purpose of this study was to determine the effect of CS extract on cholesterol metabolism in C57BL/6J mice fed high-fat diets. In this study, we used C57BL/6J mice as an animal model.
MATERIALS AND METHODS
Animals and study design
Four-week-old male C57BL/6J mice (DaeHan Bio Link, Korea) were maintained on a conventional diet for 1 week and then divided into three groups using a randomized block design according to weight: normal-fat group (NF group, n = 10), high-fat group (HF group, n = 10), and high-fat CS group (HFCS, n = 10). It is well established that the C57BL/6 J mouse strain is susceptibile to weight gain and blood lipid parameters when fed a high-fat/ high-cholesterol diet, showing profound interactions between dietary and genetic factors influencing atherogenesis [20]. Rats were maintained in conditioned rooms (temperature, 23℃; relative humidity, 55%). All animal studies were conducted in accordance with the Dankook University ethics committee guidelines for the care and use of laboratory animals (Approve No: 15-025).
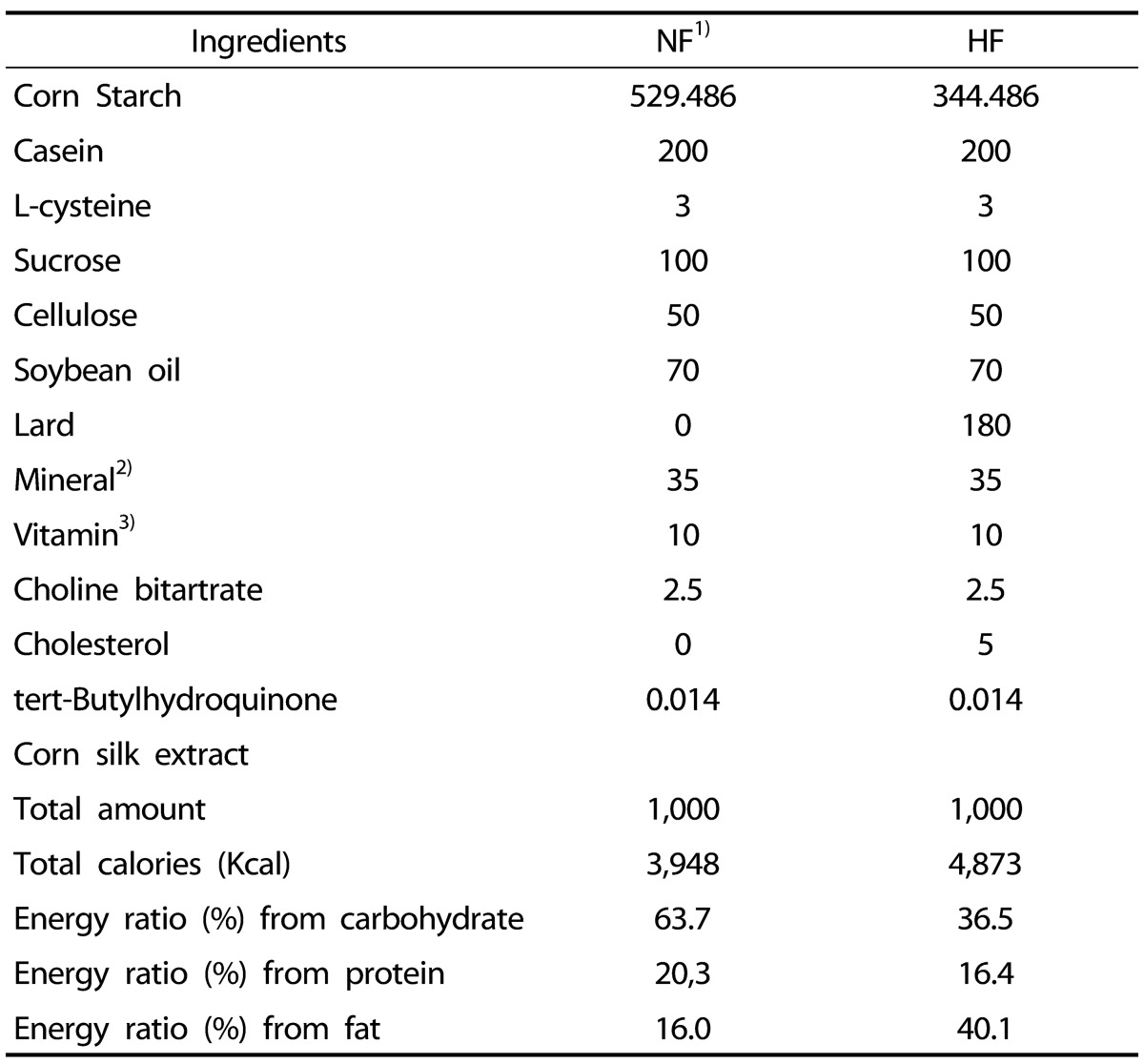
All rats were fed an AIN-93G diet [21] with modified fat contents for 8 weeks (Table 1). The NF group was fed 7% fat diet (as soybean oil, diet weight basis) while the HF group was fed 25% fat diet (7% soybean oil and 18% lard, including 0.5% cholesterol, diet weight basis). The HFCS group was fed 25 % fat diet (HF diet) and orally administered CS extract (100 mg/kg body weight) daily. To ensure the same experimental conditions, the NF group and HF group were provided the same amount of distilled water as the amount of CS extract. The CS extract contained 2,783.54 mg/100 g of maysin, and this was provided from the Rural Development Administration, National Institute of Crop Science.
Table 1. Compositions of experimental diets (g/kg).
1) NF: normal-fat diet, HF: high-fat diet
2) Mineral mixture (per kg): Calcium carbonate anhydrous, 357 g; Potassium phosphate monobasic, 196 g; Potassium citrate tripotassium monohydrate, 70.78 g; Potassium sulfate Sodium chloride, 74 g: Magnesium oxide, 24 g; Ferric citrate, 6.06 g; Zinc carbonate, 1.65 g; Sodium meta-silicate, 1.45 g; Manganous carbonate, 0.63 g; Cupric carbonate, 0.30 g; Chromium potassium sulfate, 0.275 g; Boric acid, 81.5 mg; Sodium fluoride, 63.5 mg; Nickel carbonate, 31.8 mg; Lithium chloride, 17.4 mg; Sodium selenate anhydrous, 10.25 mg; Potassium iodate, 10.0 mg; Ammonium paramolybdate, 6.66 mg; Powdered sucrose, 221.026 g
3) Vitamin mixture (per kg) : Nicotinic acid, 3.0 g; Ca Pantothenate, 1.6 g; Pyridoxine HCl 0.7 g; Thiamin HCl, 0.6 g; Riboflavin 0.6 g; Folic acid, 0.2 g; Biotin, 0.02 g; Vitamin B12, 2.5 g; Vitamin E, 15.0 g; Vitamin A, 0.8g; Vitamin D3, 0.25 g;Vitamin K-1, 0.075 g; Powderedsucrose
Sample preparations
Blood was collected from the heart with a syringe. Serum from the blood was collected by centrifugation at 4,000 × g for 30 min and then stored at - 70℃. The liver and epididymal fat pads were removed, and wet weights were measured. The samples of liver and epididymal fat were stored at - 70℃ for gene mRNA expression analysis.
Liver fat accumulation
Liver fat accumulation was determined by H&E staining. Briefly, freshly cut liver sections from samples were fixed in buffered formalin. Following fixation, the liver was embedded in paraffin and 5-µm thick paraffin sections were stained with H&E solution (Sigma Aldrich, MO, USA). After stained samples were re-hydrated with xylene and a decreasing ethanol series, liver images were obtained using camera.
Serum analysis
Total glucose, total lipids, triglycerides, total cholesterol, and free fatty acids in serum were determined using an auto blood analyzer (Roche, Germany). Serum insulin was determined by an ELISA kit (Molecular device, USA).
Liver lipid analysis
Liver homogenates were extracted with chroloform: methanol (2:1, v/v) using a modified method described by Folch et al. [22]. Total lipids, triglycerides, and cholesterol in liver homogenates were determined by a previously published method [23].
Total RNA isolation, reverse transcription, and real-time PCR
In order to determine mRNA expression of genes related with cholesterol metabolism in the liver as well as adipose tissue (epididymal fat pad), total RNA isolation, reverse transcription, and real-time PCR were conducted using previously published methods [23]. Total liver or adipose tissue RNA was isolated using TRI-reagent (Sigma aldrich, MO, USA) according to the manufacturer's protocol. cDNA was synthesized using 3 µg of total RNA with SuperScript II reverse transcriptase (Invitrogen, CA, USA). Real-time PCR was performed using the modified method recently described in detail [23]. The mRNA expression levels of acyl-CoA: cholesterol acyltransferase (ACAT), cholesterol 7-alpha hydroxylase (CYP7A1), farnesoid X receptor (FXR), lecithin cholesterol acyltransferase (LCAT), low-density lipoprotein receptor (LDL-R), 3-hyroxy-3-methylglutaryl-CoA reductase (HMG-CoA reductase), adiponectin, leptin, and tumor necrosis factor-α (TNF-α) were determined. Forward/reverse primers for genes were described in Table 2. mRNA expression was analyzed with an Applied Biosystems StepOne Plus RT-PCR system (Applied biosystems, CA, USA). Fold difference of gene mRNA expression was calculated using the 2-ΔΔCT method with an endogenous control gene.
Table 2. Primer sequences used for real-time polymerase chain reaction.
ACAT; acyl-CoA: cholesterol acyltransferase, CYP7A1; cholesterol 7-alpha hydroxylase, FXR; farnesoid X receptor, LCAT; lecithin cholesterol acyltransferase, LDL-R; low density lipoprotein receptor, HMG-CoA reductase; 3-hyroxy-3-methylglutarylcoenzyme A reductase, TNF-α; tumor necrosis factor-α, GAPDH: glyceraldehyde 3-phosphate dehydrogenase
Statistical analysis
Statistical analysis was performed using Statistical Analysis System software (SAS Institute, Cary, NC, USA). Data were expressed as means with standard deviation, and statistically significant differences among groups were evaluated using one way-analysis of variance (AVOVA) test. Statistically significant differences among means of groups were tested at α = 0.05 using Duncan's multiple range tests.
RESULTS
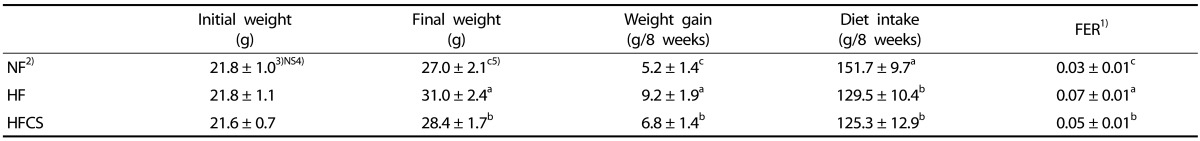
Effects of CS extract on weights and dietary intakes
The initial body weights of the three groups were similar, but final body weight of the HFCS group (28.4 ± 1.7 g) was significantly higher than that of the NF group (27.0 ± 2.1 g, P < 0.05) and significantly lower than that of the HF group (31.0 ± 2.4 g, P < 0.05, Table 3). Mean food intakes in both the HF and HFCS groups were lower, but the food efficiency ratio (FER) was significantly lower in the HFCS group than in the HF group (P < 0.05).
Table 3. Initial body weight, final body weight, weight gain, diet intake, and food efficiency ratio (FER) of mice fed experimental diet for 7 weeks.
1) FER = body weight gain for experimental period/food intake for experimental period.
2) NF; Normal fat diet, HF; High-fat diet, HFCS; High-fat diet + Corn silk extract (100 mg/kg body weight)
3) Mean ± SD
4) NS; Not significant
5) Different letters indicate significant differences among α = 0.05 by Duncan's multiple range test.
Effects of CS extract on serum glucose and insulin levels
As shown in Table 4, serum glucose level in the HFCS group (78.0 ± 21.1 mg /100 mL) was significantly lower than that in the NF group (104.3 ± 10.0 mg /100 mL) or HF group (105.2 ± 5.5 mg /100 mL). No significant difference in serum insulin level was observed between the HFCS group (0.26 ± 0.06 mg/mL) and NF group (0.25 ± 0.06 mg/mL), but a significant difference was observed compared to the HF group (0.54 ± 0.09 mg/mL).
Table 4. Concentration of serum glucose and insulin.
1) NF; Normal-fat diet, HF; High-fat diet, HFCS; High-fat diet + Corn silk extract (100 mg/kg body weight)
2) Mean ± SD
3) Different letters indicate significant differences among α = 0.05 by Duncan's multiple range test.
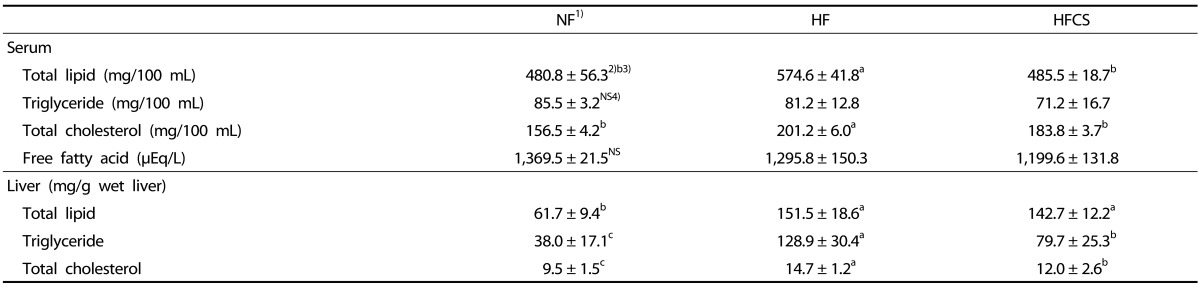
Effects of CS extract on serum and liver lipid profiles
The total serum lipid and total cholesterol levels in the HFCS group were significantly lower than those in the HF group, respectively (485.5 ± 18.7 mg /100 mL vs. 574.6 ± 41.8 mg /100 mL in total lipid, P < 0.05, and 183.8 ± 3.7 mg / 100 mL vs. 201.2 ± 6.0 mg /100 mL in total cholesterol, P < 0.05, Table 5). The serum concentrations of triglycerides and free fatty acids in the HFCS group were lower compared to those in the HF group, but the difference was not significant. The liver triglyceride and total cholesterol concentrations in the HFCS group were significantly lower than those in the HF group, respectively (79.7 ± 25.3 mg/g wet liver vs. 128.9 ± 30.4 mg/g wet liver for triglycerides, P < 0.05, and 12.0 ± 2.6 mg/g wet liver vs. 14.7 ± 1.2 mg/g wet liver in total cholesterol, P < 0.05).
Table 5. Effect of Corn silk extract on serum and liver lipid profiles.
1) NF; Normal-fat diet, HF; High-fat diet, HFCS; High-fat diet + Corn silk extract (100 mg/kg body weight)
2) Mean ± SD
3) Different letters indicate significant differences among α = 0.05 by Duncan's multiple range test.
4) NS; Not significant
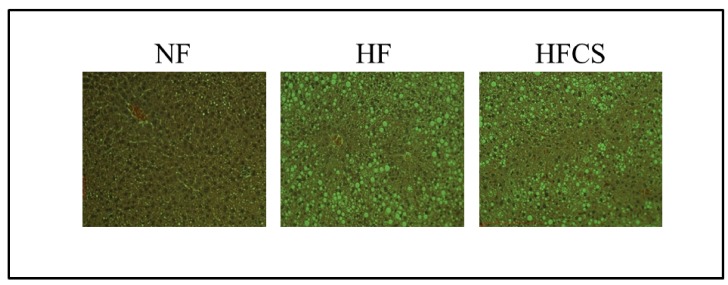
Effects of CS extract on liver fat accumulation in mice livers
H&E staining showed an increase in fat droplets in rat livers after HF diet feeding. Fat droplets decreased in size after CS extract supplementation, in comparison to HF diet only (Fig. 1).
Fig. 1. Effect of Corn silk extract on lipid deposition in the liver.
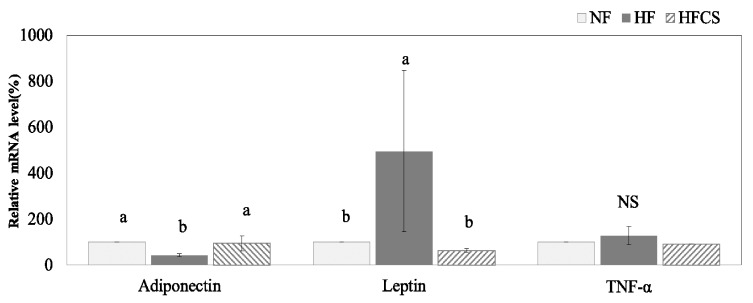
Effects of CS on mRNA expression of adipocytokines
The mRNA expression of adipocytokines in adipose tissue is shown in Fig. 2. The mRNA expression of adiponectin in the HF group decreased significantly (P < 0.05), whereas it increased upon consumption of CS extract up to a level similar to that in the NF group. The mRNA expression of leptin in the HF group significantly increased (P < 0.05), whereas it was reduced upon consumption of CS extract. The mRNA expression of TNF-α was not affected according to experimental treatment.
Fig. 2. Effect of Corn silk extract on mRNA expression of adipocytokines in adipose tissue of mouse fed high-fat diet.
Total RNA was isolated using TRI-reagent and cDNA was synthesized using 3 µg of total RNA with SuperScriptⅡ reverse transcriptase. Real-time PCR was performed using SYBR green and standard procedures to assess mRNA expression of primer in adipose tissue (epididymal fat pad) samples obtained from each group. An Applied Biosystem StepOne softwere v2.1 was used. Each bar represents the mean ± S.D, and different letters above each bar indicate significant differences among groups at α = o.o5 as determined by Duncan's multiple range test. TNF-α; tumor necrosis factor-α.
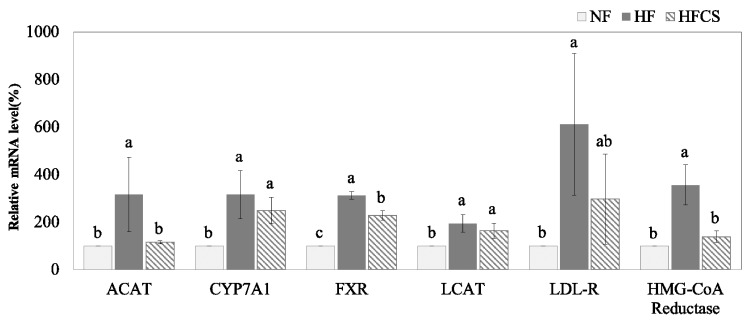
mRNA expression of genes related to cholesterol metabolism
The mRNA expression levels of all genes related to cholesterol metabolism in the HF group were significantly higher compared to those in the NF group (P < 0.05, Fig. 3). The mRNA expression levels of hepatic ACAT, FXR, and HMG-CoA reductase in the HFCS group were significantly reduced compared to those in the HF group (P < 0.05). However, mRNA expression levels of hepatic CYP7A1, LCAT, and LDL-R between the HF group and HFCS group were not statistically different.
Fig. 3. Effect of Corn silk extract on mRNA expression of enzymes related to cholesterol metabolism in livers of mice fed high-fat diet.
Total RNA was isolated using TRI-reagent, and cDNA was synthesized using 3 µg of total RNA with SuperScriptⅡ reverse transcriptase. Real-time PCR was performed using SYBR green and standard procedures to assess mRNA expression of primer in liver samples obtained from each group. An Applied Biosystem StepOne softwere v2.1 was used. Each bar represents the mean ± S.D, and different letters above each bar indicate significant differences among groups at α = o.o5 as determined by Duncan's multiple range test. ACAT; acyl-CoA: cholesterol acyltransferase, CYP7A1; cholesterol 7-alpha hydroxylase, FXR; farnesoid X receptor, LCAT; lecithin cholesterol acyltransferase, LDL-R; low density lipoprotein receptor, HMG-CoA reductase; 3-hyroxy-3-methylglutaryl-coenzyme A reductase.
DISCUSSION
Hepatic cholesterol metabolism, which is directly related to obesity, is an important physiological process in lipid metabolic disorders [24]. At present, the cholesterol-lowering effect of CS extract and its related gene expression have not been studied. Thus, the purpose of this study was to investigate whether or not CS extract has a protective effect against high-fat-induced cholesterol metabolism in rats.
We observed a significant increase in weight gain and fat content of the liver in the HF group, whereas oral administration of CS extract with high-fat diet for 8 weeks caused weight loss and attenuated high-fat-induced fatty liver, in addition to improvement of serum glucose and insulin levels. The reduced mRNA expression of adiponectin and increased mRNA expression of leptin due to prolonged HF feeding were also attenuated by CS extract consumption.
In general, decreasing body weight and fat mass has been reported to improve insulin sensitivity [1]. Several adipocytokines, including leptin and adiponectin secreted from adipocytes, are reported to regulate insulin resistance and lipid metabolism [25,26,27]. Adiponectin was shown to increase fatty acid combustion and energy consumption, in part via PPARα activation [28] or through activation of AMP-activated protein kinase (AMPK) [26], leading to decreased fat content in the liver and thereby direct regulation of glucose metabolism and insulin sensitivity. Based on those studies, it can be suggested that CS extract caused weight loss and improved blood levels of glucose and insulin through increased mRNA expression of adiponectin.
Cholesterol metabolism is controlled by synthesis and excretion in the body. This study showed that CS extract inhibits cholesterol synthesis through modulation of mRNA expression levels of HMG-CoA reductase, ACAT, and FXR in line with changes in cholesterol levels observed in the serum and liver. Both HMG-CoA reductase and ACAT are rate-controlling enzymes of cholesterol synthesis [29]. HMG-CoA reductase catalyzes conversion of HMG-CoA to mevalonate, the precursor of isoprenoid groups incorporated into many end products such as cholesterol [28]. ACAT catalyzes esterification of free cholesterol into cholesterol ester (CE), which is a major source of very low-density lipoprotein (VLDL) and LDL formation [30].
HMG-CoA reductase inhibitors have been shown to lower plasma LDL cholesterol through one or a combination of the following mechanisms: (1) enhanced LDL catabolism by up-regulation of hepatic LDL receptor or (2) reduced production of apoprotein B (apo B), which is the primary component of VLDL and LDL particles [31,32,33]. The HMG-CoA reductase inhibitor, atorvastatin, inhibits cholesterol biosynthesis in vivo, reducing both VLDL and LDL concentrations, primarily by decreasing apo B secretion into plasma [32,33]. The results of these above studies are consistent with this study. Thus, it can be suggested that reduced mRNA expression of HMG-CoA reductase and ACAT in the HFCS group led to blocked cholesterol synthesis in the body and reduction of cholesteryl ester for incorporation into VLDL, with consequent reduction in the availability of hepatic cholesterol for lipoprotein synthesis [34].
This study demonstrated reduced mRNA expression of FXR as well as depleted CYP7A1 in the HFCS group compared to the HF group, although the difference was not statistically significant. FXR is a member of the nuclear receptor superfamily of ligand-regulated transcription factors that regulates bile acid synthesis, a major form of cholesterol excretion, through transcriptional regulation of CYP7A1, the rate-limiting enzyme in the classic bile acid biosynthetic pathway [35,36]. Thus, the result of this study suggest that decreased blood and liver cholesterol levels in the HFCS group are not related with cholesterol excretion since mRNA expression of FXR and CYP7A1 was depleted in the HFCS group compared to the HF group.
In conclusion, CS extract supplementation with high-fat diet improves adipocytokine secretion due to weight loss, glucose homeostasis, as well as reduced blood and hepatic levels of cholesterol. Decreased synthesis of cholesterol through down-regulation of hepatic mRNA expression of HMG-CoA reductase and ACAT as well as mRNA expression of FXR is responsible for the blood cholesterol-lowering effects of CS extract. Thus, the demonstrated effects of CS extracts on cholesterol synthesis are thought to improve serum cholesterol levels and decrease risk of cardiovascular-related diseases.
Footnotes
CONFLICT OF INTEREST: The authors declare no potential conflicts of interests.
References
- 1.Koska J, Stefan N, Permana PA, Weyer C, Sonoda M, Bogardus C, Smith SR, Joanisse DR, Funahashi T, Krakoff J, Bunt JC. Increased fat accumulation in liver may link insulin resistance with subcutaneous abdominal adipocyte enlargement, visceral adiposity, and hypoadiponectinemia in obese individuals. Am J Clin Nutr. 2008;87:295–302. doi: 10.1093/ajcn/87.2.295. [DOI] [PubMed] [Google Scholar]
- 2.Matsuzawa Y. The metabolic syndrome and adipocytokines. FEBS Lett. 2006;580:2917–2921. doi: 10.1016/j.febslet.2006.04.028. [DOI] [PubMed] [Google Scholar]
- 3.Doan DD, Nguyen NH, Doan HK, Nguyen TL, Phan TS, van Dau N, Grabe M, Johansson R, Lindgren G, Stjernström NE. Studies on the individual and combined diuretic effects of four Vietnamese traditional herbal remedies (Zea mays, Imperata cylindrica, Plantago major and Orthosiphon stamineus) J Ethnopharmacol. 1992;36:225–231. doi: 10.1016/0378-8741(92)90048-v. [DOI] [PubMed] [Google Scholar]
- 4.Hasanudin K, Hashim P, Mustafa S. Corn silk (Stigma maydis) in healthcare: a phytochemical and pharmacological review. Molecules. 2012;17:9697–9715. doi: 10.3390/molecules17089697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kan A, Orhan I, Coksari G, Sener B. In-vitro neuroprotective properties of the Maydis stigma extracts from four corn varieties. Int J Food Sci Nutr. 2012;63:1–4. doi: 10.3109/09637486.2011.590797. [DOI] [PubMed] [Google Scholar]
- 6.Velazquez DV, Xavier HS, Batista JE, de Castro-Chaves C. Zea mays L. extracts modify glomerular function and potassium urinary excretion in conscious rats. Phytomedicine. 2005;12:363–369. doi: 10.1016/j.phymed.2003.12.010. [DOI] [PubMed] [Google Scholar]
- 7.Zhao W, Yin Y, Yu Z, Liu J, Chen F. Comparison of anti-diabetic effects of polysaccharides from corn silk on normal and hyperglycemia rats. Int J Biol Macromol. 2012;50:1133–1137. doi: 10.1016/j.ijbiomac.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 8.Farsi DA, Harris CS, Reid L, Bennett SA, Haddad PS, Martineau LC, Arnason JT. Inhibition of non-enzymatic glycation by silk extracts from a Mexican land race and modern inbred lines of maize (Zea mays) Phytother Res. 2008;22:108–112. doi: 10.1002/ptr.2275. [DOI] [PubMed] [Google Scholar]
- 9.Suzuki R, Okada Y, Okuyama T. The favorable effect of style of Zea mays L. on streptozotocin induced diabetic nephropathy. Biol Pharm Bull. 2005;28:919–920. doi: 10.1248/bpb.28.919. [DOI] [PubMed] [Google Scholar]
- 10.Guo J, Liu T, Han L, Liu Y. The effects of corn silk on glycaemic metabolism. Nutr Metab (Lond) 2009;6:47. doi: 10.1186/1743-7075-6-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maksimović Z, Dobrić S, Kovacević N, Milovanović Z. Diuretic activity of Maydis stigma extract in rats. Pharmazie. 2004;59:967–971. [PubMed] [Google Scholar]
- 12.Bai H, Hai C, Xi M, Liang X, Liu R. Protective effect of maize silks (Maydis stigma) ethanol extract on radiation-induced oxidative stress in mice. Plant Foods Hum Nutr. 2010;65:271–276. doi: 10.1007/s11130-010-0172-6. [DOI] [PubMed] [Google Scholar]
- 13.Choi DJ, Kim SL, Choi JW, Park YI. Neuroprotective effects of corn silk maysin via inhibition of H2O2-induced apoptotic cell death in SK-N-MC cells. Life Sci. 2014;109:57–64. doi: 10.1016/j.lfs.2014.05.020. [DOI] [PubMed] [Google Scholar]
- 14.Maksimović Z, Malencić D, Kovacević N. Polyphenol contents and antioxidant activity of Maydis stigma extracts. Bioresour Technol. 2005;96:873–877. doi: 10.1016/j.biortech.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 15.Kim SL, Kim MJ, Lee YY, Jung GH, Son BY, Lee JS, Kwon YU, Park YI. Isolation and identification of flavonoids from corn silk. Korean J Crop Sci. 2014;59:435–444. [Google Scholar]
- 16.Waiss AC, Jr, Chan BG, Elliger CA, Wiseman BR, McMillian WW, Widstrom NW, Zuber MS, Keaster AJ. Maysin, a flavone glycoside from corn silks with antibiotic activity toward corn earworm. J Econ Entomol. 1979;72:256–258. [Google Scholar]
- 17.Lee EA, Byrne PF, McMullen MD, Snook ME, Wiseman BR, Widstrom NW, Coe EH. Genetic mechanisms underlying apimaysin and maysin synthesis and corn earworm antibiosis in maize (Zea mays L.) Genetics. 1998;149:1997–2006. doi: 10.1093/genetics/149.4.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jeong HW, Lee JW, Kim WS, Choe SS, Kim KH, Park HS, Shin HJ, Lee GY, Shin D, Lee H, Lee JH, Choi EB, Lee HK, Chung H, Park SB, Park KS, Kim HS, Ro S, Kim JB. A newly identified CG301269 improves lipid and glucose metabolism without body weight gain through activation of peroxisome proliferator-activated receptor alpha and gamma. Diabetes. 2011;60:496–506. doi: 10.2337/db09-1145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Min OJ, Sharma BR, Park CM, Rhyu DY. Effect of myadis stigma water extract on adipogenesis and blood glucose in 3T3-L1 adipocytes and db/db mice. Korean J Pharmacogn. 2011;42:201–208. [Google Scholar]
- 20.Schreyer SA, Wilson DL, LeBoeuf RC. C57BL/6 mice fed high fat diets as models for diabetes-accelerated atherosclerosis. Atherosclerosis. 1998;136:17–24. doi: 10.1016/s0021-9150(97)00165-2. [DOI] [PubMed] [Google Scholar]
- 21.Reeves PG. Components of the AIN-93 diets as improvements in the AIN-76A diet. J Nutr. 1997;127:838S–841S. doi: 10.1093/jn/127.5.838S. [DOI] [PubMed] [Google Scholar]
- 22.Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957;226:497–509. [PubMed] [Google Scholar]
- 23.Ha AW, Kim WK. The effect of fucoxanthin rich power on the lipid metabolism in rats with a high fat diet. Nutr Res Pract. 2013;7:287–293. doi: 10.4162/nrp.2013.7.4.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dai W, Wang K, Zheng X, Chen X, Zhang W, Zhang Y, Hou J, Liu L. High fat plus high cholesterol diet lead to hepatic steatosis in zebrafish larvae: a novel model for screening anti-hepatic steatosis drugs. Nutr Metab (Lond) 2015;12:42. doi: 10.1186/s12986-015-0036-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kadowaki T, Yamauchi T, Kubota N, Hara K, Ueki K, Tobe K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J Clin Invest. 2006;116:1784–1792. doi: 10.1172/JCI29126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li Y, Qin G, Liu J, Mao L, Zhang Z, Shang J. Adipose tissue regulates hepatic cholesterol metabolism via adiponectin. Life Sci. 2014;118:27–33. doi: 10.1016/j.lfs.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 27.Liu Q, Yuan B, Lo KA, Patterson HC, Sun Y, Lodish HF. Adiponectin regulates expression of hepatic genes critical for glucose and lipid metabolism. Proc Natl Acad Sci U S A. 2012;109:14568–14573. doi: 10.1073/pnas.1211611109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yamauchi T, Kamon J, Waki H, Imai Y, Shimozawa N, Hioki K, Uchida S, Ito Y, Takakuwa K, Matsui J, Takata M, Eto K, Terauchi Y, Komeda K, Tsunoda M, Murakami K, Ohnishi Y, Naitoh T, Yamamura K, Ueyama Y, Froguel P, Kimura S, Nagai R, Kadowaki T. Globular adiponectin protected ob/ob mice from diabetes and ApoEdeficient mice from atherosclerosis. J Biol Chem. 2003;278:2461–2468. doi: 10.1074/jbc.M209033200. [DOI] [PubMed] [Google Scholar]
- 29.DeBose-Boyd RA. Feedback regulation of cholesterol synthesis: sterol-accelerated ubiquitination and degradation of HMG CoA reductase. Cell Res. 2008;18:609–621. doi: 10.1038/cr.2008.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Krause BR, Pape ME, Kieft K, Auerbach B, Bisgaier CL, Homan R, Newton RS. ACAT inhibition decreases LDL cholesterol in rabbits fed a cholesterol-free diet. Marked changes in LDL cholesterol without changes in LDL receptor mRNA abundance. Arterioscler Thromb. 1994;14:598–604. doi: 10.1161/01.atv.14.4.598. [DOI] [PubMed] [Google Scholar]
- 31.Davignon J, Montigny M, Dufour R. HMG-CoA reductase inhibitors: a look back and a look ahead. Can J Cardiol. 1992;8:843–864. [PubMed] [Google Scholar]
- 32.Burnett JR, Wilcox LJ, Telford DE, Kleinstiver SJ, Barrett PH, Newton RS, Huff MW. Inhibition of HMG-CoA reductase by atorvastatin decreases both VLDL and LDL apolipoprotein B production in miniature pigs. Arterioscler Thromb Vasc Biol. 1997;17:2589–2600. doi: 10.1161/01.atv.17.11.2589. [DOI] [PubMed] [Google Scholar]
- 33.Burnett JR, Wilcox LJ, Telford DE, Kleinstiver SJ, Barrett PH, Newton RS, Huff MW. Inhibition of ACAT by avasimibe decreases both VLDL and LDL apolipoprotein B production in miniature pigs. J Lipid Res. 1999;40:1317–1327. [PubMed] [Google Scholar]
- 34.Llaverías G, Laguna JC, Alegret M. Pharmacology of the ACAT inhibitor avasimibe (CI-1011) Cardiovasc Drug Rev. 2003;21:33–50. [PubMed] [Google Scholar]
- 35.Evans MJ, Mahaney PE, Borges-Marcucci L, Lai K, Wang S, Krueger JA, Gardell SJ, Huard C, Martinez R, Vlasuk GP, Harnish DC. A synthetic farnesoid X receptor (FXR) agonist promotes cholesterol lowering in models of dyslipidemia. Am J Physiol Gastrointest Liver Physiol. 2009;296:G543–G552. doi: 10.1152/ajpgi.90585.2008. [DOI] [PubMed] [Google Scholar]
- 36.Mencarelli A, Fiorucci S. FXR an emerging therapeutic target for the treatment of atherosclerosis. J Cell Mol Med. 2010;14:79–92. doi: 10.1111/j.1582-4934.2009.00997.x. [DOI] [PMC free article] [PubMed] [Google Scholar]