Abstract
It has been proposed that cancer stem cells (CSCs) are involved in tumor resistance to chemotherapy and tumor relapse. The goal of the present study was to determine the effect of low-dose 5-fluorouracil (5-Fu) on enriched hepatocellular CSC-like cells. Increased cell motility and epithelial-mesenchymal transition were observed by migration assay in human hepatoblastoma PLC/RAF/5 cells following 5-Fu treatment, as well as a significant enhancement in their sphere-forming abilities. CSC-like cells were identified by side population cell analysis. The percentage of CSC-like cells in the surviving cells was greatly increased in response to 5-Fu. These findings indicate that low-dose 5-Fu treatment may efficiently enrich the CSC-like cell population in PLC/RAF/5 cells.
Keywords: tumor, hepatocellular carcinoma, cancer stem cell-like cells, chemotherapy, 5-fluorouracil
Introduction
Hepatocellular carcinoma is one of the most common cancers, with high mortality rates worldwide (1,2). The fluorinated pyrimidine 5-fluorouracil (5-Fu) is an antimetabolite that is frequently used in the treatment of patients with liver, breast and stomach cancer (3–5). However, 32.5–61.5% of hepatocellular carcinoma patients experience incurable recurrent cancer within 5 years of chemotherapy, which is a consequence of a small percentage of chemotherapy-resistant residual tumor cells that facilitate the development of recurrent progressive disease (6).
There is accumulating evidence that chemoresistance is associated with the acquisition of epithelial-mesenchymal transition (EMT) and cancer stem cell (CSC)-like phenotypes in cancer (7,8). In addition, CSCs have been demonstrated to be responsible for initiating tumor formation, maintaining tumor growth, generating distant metastases and causing relapse following treatment (9). The identification and characterization of liver CSC-like cells may aid to develop effective and targeted therapies (10). It has been reported that CSCs are resistant to chemotherapy, thus suggesting that these CSCs control self-renewal, metastasis and chemoresistance (11).
Side population (SP) cell sorting was previously applied for the identification and isolation of CSCs in various solid tumors (12). It has been reported that SP cells exhibit distinct projection pattern by actively effluxing the Hoechst 33342 fluorescence dye from the cytoplasm through verapamil-sensitive ATP-binding cassette (ABC) transporters (13). That approach specifically characterizes the tumorigenic liver CSC population.
In the present study, it was demonstrated that the exposure to chemotherapy (5-Fu) treatment induced a CSC-like profile in surviving hepatocellular carcinoma cells, thus offering a novel approach to study the tumorigenesis of hepatocellular carcinoma cells.
Materials and methods
Reagents
Dulbecco's modified Eagle's medium (DMEM), penicillin and streptomycin were purchased from HyClone (GE Healthcare Life Sciences, Logan, UT, USA). Fetal bovine serum (FBS) and 0.25% trypsin were purchased from Gibco (Thermo Fisher Scientific, Inc., Waltham, MA, USA). 5-Fu was purchased from NanJing KeyGen Biotech Co., Ltd. (Nanjing, Jiangsu, China). The anti-human ABC sub-family G member 2 (ABCG2) antibody (catalog no. 3765-1) was purchased from Epitomics (Burlingame, CA, USA). The anti-human B-cell lymphoma (BCL)-2 (catalog no. 15071) and anti-human BCL-2-associated X protein (catalog no. 2774) antibodies were purchased from CST Biological Reagents Company Limited (Shanghai, China). The anti-human forkhead box protein M1 (FOXM1; catalog no. ab55006) antibody was purchased from Abcam (Cambridge, MA, USA). The anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH; catalog no. G8795) antibody was purchased from Sigma-Aldrich (Merck Millipore, Darmstadt, Germany). Hoechst 33342 fluorescent dye and verapamil were purchased from Invitrogen (Thermo Fisher Scientific, Inc.). Matrigel invasion chambers were purchased from BD Biosciences (Franklin Lakes, NJ, USA). Crystal violet was purchased from Sigma-Aldrich (Merck Millipore).
Cell culture
The PLC/RAF/5 human hepatocellular carcinoma cell line was a gift from the First Affiliated Hospital of Sun Yat-sen University (Guangzhou, China). Cells were cultured in high-glucose DMEM containing 10% FBS, 100 U/ml penicillin and 100 µg/ml streptomycin in 5% CO2 at 37°C. The resistant sub-line (PLC/RAF/5/5-Fu) was generated by treating the PLC/RAF/5 cells for several passages with a low concentration of 5-Fu. Cells in the logarithmic growth phase were treated intermittently with a low concentration of 5-Fu (1 µg/ml). The medium was changed every 2 days to remove the dead cells. 5-Fu (1 µg/ml) was added when the logarithmic growth phase was reached. The resulting resistant phenotype was confirmed to be stable over ≥6 passages without 5-Fu.
Cell migration assay
Cell migration was determined with a wound-healing assay. Briefly, cells were seeded in 12-well plates at equal density and grown to 80% confluency. Artificial gaps were generated with an sterile pipette tip. The areas of the wound were marked and photographed with a digital camera system. Cell migration distance (in mm) was calculated with ImageJ software (v2.1.4.7; National Institutes of Health, Bethesda, MD, USA). Each experiment was repeated three times.
Matrigel invasion assay
The invasive ability of PLC/RAF/5 and PLC/RAF/5/5-Fu cells was determined using 24-well Matrigel invasion chambers. Cells were seeded into the upper inserts at a density of 1×105 cells/insert in serum-free DMEM. The outer wells were filled with DMEM containing 10% FBS as chemoattractant. Cells were incubated for 24 h, and then non-invading cells were removed by swabbing the top Matrigel with a cotton swab. Membranes containing invading cells were stained with crystal violet. The number of invading cells on the entire membrane was counted under a light microscope.
Detection of SP cells
Detection of SP cells was performed as described previously with certain modifications (13). Briefly, cells were incubated with Hoechst 33342 dye at 10 µg/ml, either alone or in combination with the ABC-transporter inhibitor verapamil (50 mM), for 90 min at 37°C. Upon staining, the cells were centrifuged and resuspended in phosphate-buffered saline (PBS) containing 1 mg/ml propidium iodide (PI) for flow cytometry analysis.
Sphere-forming assay
The sphere-forming ability of untreated and chemotherapy-treated PLC/RAF/5 cells was determined as described previously (14). The sphere-forming ability of the cells was photographed over 21 days using a phase-contrast microscope. Cellular aggregates with a diameter >50 µm were classified as ‘spheres’.
Cell cycle analysis
Cells were harvested, washed twice with ice-cold PBS and fixed with 70% ethanol at −20°C overnight. Cells were then treated with PI (20 µg/ml) in the presence of RNase (100 µg/ml) for 1 h at 37°C. A BD FACSCalibur™ flow cytometer (BD Biosciences) was used to perform the flow cytometry assay.
Western blotting
The protein extracts of PLC/RAF/5 and PLC/RAF/5/5-Fu cells (20 µg protein each) were subjected to 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a polyvinylidene fluoride membrane via a semi-dry transfer apparatus. Once the membrane had been blocked in 5% non-fat milk for 1 h at room temperature, it was incubated with the aforementioned primary antibodies against BAX, BCL-2, FOXM1, ABCG2 and GAPDH (dilution, 1:500, 1:500, 1:1,000, 1:1,000 and 1:1,000, respectively) overnight at 4°C, followed by incubation with the horseradish peroxidase (HRP)-linked secondary antibody (dilution, 1:5,000; catalog no. SA00001-1/SA00001-2; ProteinTech Group Inc., Chicago, IL, USA). A Chemiluminescence HRP Substrate kit (catalog no. WBKLS0500; Merck Millipore). was used for detection.
Statistical analyses
Data were expressed as the mean ± standard error. Significance tests were performed using SPSS version 17.0 software (SPSS, Inc., Chicago, IL, USA). Statistical significance was determined using the Student's t-test. P<0.05 was considered to indicate a statistically significant difference.
Results
Cell cycle analysis
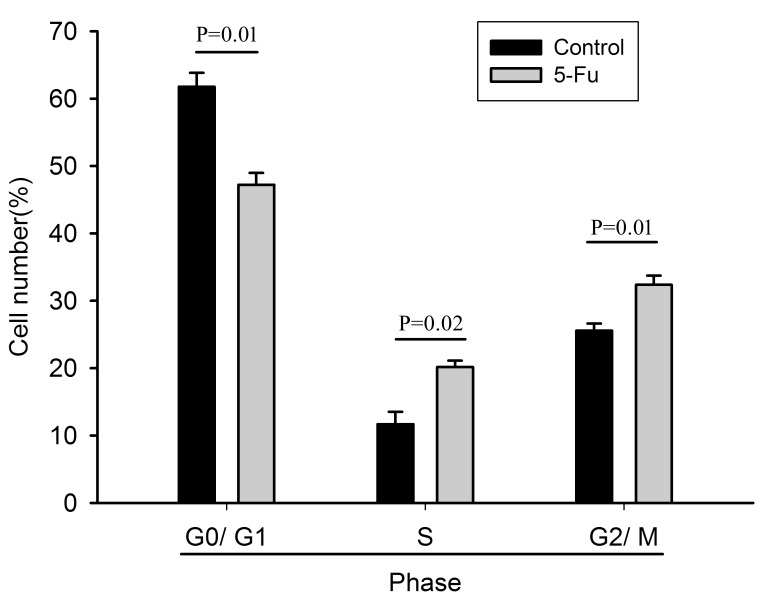
PI cell cycle assay demonstrated that the ratio of G0/G1-phase cells in the 5-Fu-treated cells was lower than that in the untreated cells, but the ratio of S- and G2/M-phase cells was significantly increased (Fig. 1).
Figure 1.
Cell cycle analysis of PLC/RAF/5 and PLC/RAF/5/5-Fu cells. Quantification of cell proliferation using propidium iodide staining by flow cytometry (n=3). Data were expressed as the percentage of cells in each phase. Control, PLC/RAF/5 cells; LC/RAF/5/5-Fu cells, 5-Fu-treated cells. 5-Fu, 5-fluorouracil.
EMT and migration
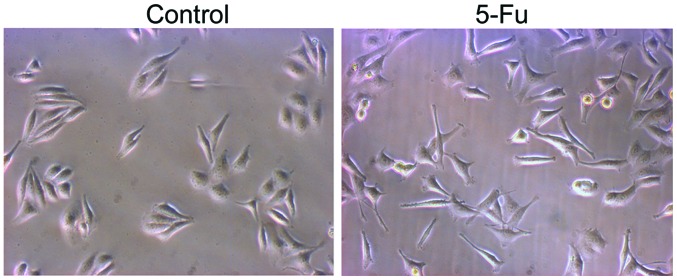
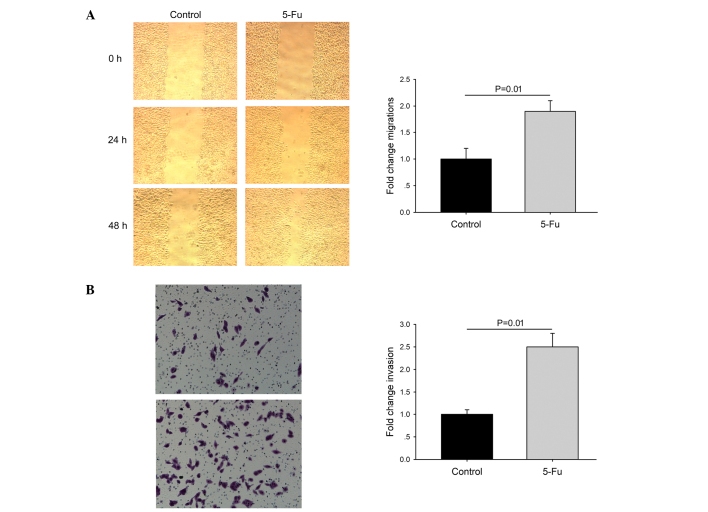
PLC/RAF/5 cells were subjected to repeated long-term treatment with 5-Fu, as described above. Treatment with 5-Fu resulted in the appearance of epithelioid cells in the PLC/RAF/5 cell line (Fig. 2). Compared with the untreated cells, a number of 5-Fu-treated PLC/RAF/5 cells underwent a remarkable cellular enlargement (≤3-fold), which may be due to the formation of multinucleated cells by inhibition of the mitotic cycle. Cell migration was determined by wound-healing and Matrigel invasion assays. Compared with control cells, 5-Fu-treated cells exhibited increased motility (Fig. 3).
Figure 2.
Morphological features of PLC/RAF/5 cells under normal culture conditions (control) and upon treatment with 5-Fu. Magnification, ×200. 5-Fu, 5-fluorouracil.
Figure 3.
Migration and invasion of PLC/RAF/5 and PLC/RAF/5/5-Fu cells. (A) Cell migration was measured by the scratch assay. Columns represent the fold-change over the migration distance of control cells. (B) Dark staining corresponding to invasive cells (crystal violet stain; magnification, ×100). Invasion assay was quantified and represented as bar charts, where the columns correspond to the fold-change over the number of control cells that invaded. Data are expressed as the mean ± standard error. Each experiment was performed in triplicate in five independent occasions. Each error bar indicates the variation between the means of these five independent experiments. Control, PLC/RAF/5 cells; PLC/RAF/5/5-Fu cells, PLC/RAF/5 cells treated with 5-Fu. 5-Fu, 5-fluorouracil.
Chemotherapy increases SP cells
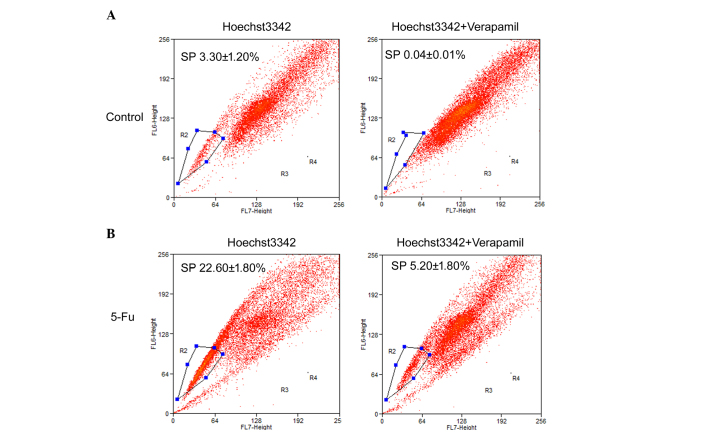
SP cells exhibit a distinct projection pattern by actively effluxing the Hoechst 33342 dye from the cytoplasm through verapamil-sensitive ABC transporters. In the present study, the fluorescent dye, Hoechst 33342, was used alongside fluorescence-activated cell sorting analysis to identify SP cells from the PLC/RAF/5 cell line.
The results of the flow cytometric analysis of PLC/RAF/5 cells (with or without 5-Fu treatment) stained with Hoechst 33342 are presented in Fig. 4. For the untreated PLC/RAF/5 cells, 3.3±1.2% of the total population was comprised of SP cells (Fig. 4A), whereas SP cells represented 22.6±1.8% of the total population of 5-Fu-treated cells (Fig. 4B). The level of SP cell population was substantially diminished in the presence of verapamil, an inhibitor of ABC transporters, in both 5-Fu-treated and untreated cells (Fig. 4). The present results suggest that a significantly higher population of SP cells exists within the 5-Fu-treated cells in comparison with the untreated cells.
Figure 4.
Analysis of SP cells in (A) PLC/RAF/5 (control) and (B) 5-Fu-treated PLC/RAF/5 cells by flow cytometry analysis. Cells were stained with Hoechst 33342 in the absence or presence of verapamil. The SP cells were gated and represented as a percentage of the total population. Flow cytometry analyses were performed in triplicate. 5-Fu, 5-fluorouracil; SP, side population.
Chemotherapy increases sphere formation
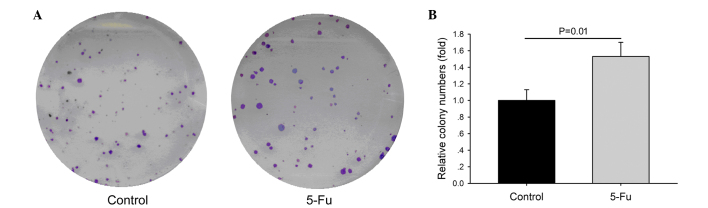
It has been reported that sphere formation is an important feature for the survival of liver CSCs. The present study evaluated the sphere-forming abilities of 5-Fu-treated and untreated cells (Fig. 5). In long-term cultures, 5-Fu treated cells demonstrated the ability to form spheres on low-attachment plates. Within 20 days, the aggregates formed by 5-Fu-treated cells acquired the shape of spheres, which were significantly greater in number and bigger in size than those observed in the control cells.
Figure 5.
Effects of chemotherapy on the sphere-forming ability of PLC/RAF/5 cells. (A) Images are representative of a section of a 24-well plate. Crystal violet stain; magnification, ×40. (B) Columns represent the fold-change over the number of control cells that formed spheres. Control, PLC/RAF/5 cells; PLC/RAF/5/5-Fu, PLC/RAF/5 cells treated with 5-Fu. 5-Fu, 5-fluorouracil.
Western blot detection
The expression levels of BAX, BCL-2, ABCG2 and FOXM1 proteins in PLC/RAF/5 and PLC/RAF/5/5-Fu cells were detected by western blotting (Fig. 6). The expression levels of BCL-2, ABCG2 and FOXM1 proteins increased in the PLC/RAF/5/5-Fu cells, while the expression levels of BAX protein were markedly lower than those displayed by the untreated cells (Fig. 6).
Figure 6.
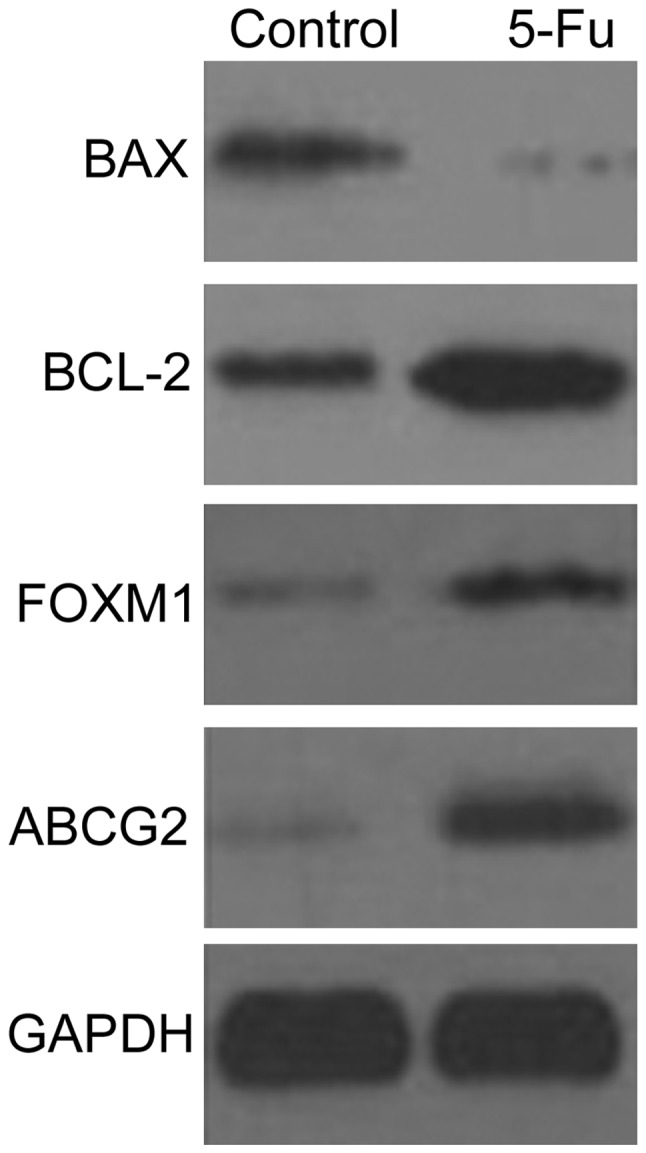
The expression levels of BAX, BCL-2, ABCG2 and FOXM1 proteins in PLC/RAF/5 (control) and PLC/RAF/5/5-Fu (5-Fu) cells were detected by western blot analysis. BCL-2, B-cell lymphoma-2; BAX, BCL-2-associated X protein; ABCG2, ATP-binding cassette sub-family G member 2; FOXM1, forkhead box protein M1; GAPDH, 3-phosphate dehydrogenase. 5-Fu, 5-fluorouracil.
Discussion
Previous researchers hypothesized that tumors are complex tissues where abnormal growth is driven by CSCs (15). CSCs exhibit tumor-like features such as uncontrolled growth, but they also maintain their innate capacity to self-renew and differentiate into a phenotypically heterogeneous progeny (16). Accumulating evidence reveals that CSCs are closely correlated with the outcome of chemotherapy due to their role in chemoresistance (17). It was suggested that tumor recurrence and metastasis may result from the resistance of residual CSCs following chemotherapy (18). Therefore, separation and enrichment of CSCs by chemotherapeutic drugs may provide a novel and convenient way to understand the characteristics of CSCs.
In multiple tumor cell lines, SP cell purification is employed to separate CSCs (12,13). The study conducted by Chiba et al indicated that the SP cells purified from four hepatocellular carcinoma cell lines exhibited stem cell-like properties in culture and in xenotransplant assay (12). In the present study, the percentage of SP cells increased from 3.3±1.2 to 22.6±1.8% following 5-Fu treatment. Therefore, low-dose repeated 5-Fu treatment may be used to induce the enrichment of liver CSCs. ABCG2 is one of the ABC transporter protein family members that is capable of pumping intracellular drugs outside the cell (19). Enhanced expression of ABCG2 in CSCs of hepatocellular carcinoma tissues has been observed, which is considered as a potential molecular marker for CSCs (20,21). In the present study, the expression of ABCG2 protein was increased in PLC/RAF/5/5-Fu cells, alongside an increased percentage of SP cells. Furthermore, the PLC/RAF/5/5-Fu cells had increased sphere-forming abilities. Thus, it is possible that tumor recurrence and drug resistance following hepatocellular carcinoma chemotherapy may result from residual SP cells.
A previous study indicated that, in normal stem/progenitor cells present in various tissues and organs, high levels of anti-apoptotic proteins were expressed, including BCL-2 (22). It has also been reported that 5-Fu treatment may increase the protein and messenger RNA expression levels of BAX, which is one of the BCL-2 family members with anti-apoptotic characteristics (23). The present study indicated that the expression of BCL-2 protein was increased, while the expression of BAX protein was decreased, in PLC/RAF/5/5-Fu cells. It is possible that the upregulation of BCL-2 and the downregulation of BAX resulted in resistance against chemotherapeutic drugs.
The FOXM1 transcription factor belongs to the forkhead family (24). It has been reported that abnormal expression of FOXM1 causes the proliferation of stem cell components, thus leading to tumorigenesis (25). FOXM1 can methylate and recombine DNA to resist cell differentiation and induce the proliferation of stem/progenitor cells (26). A previous in vivo study also demonstrated that FOXM1 is important in the self-renewal of hepatocellular carcinoma stem cells (27). Therefore, it is possible that the upregulation of FOXM1 in PLC/RAF/5/5-Fu cells led to the increased number of SP cells observed in the current study. Thus, FOXM1 appears to be a novel target for developing anti-hepatocellular carcinoma drugs.
In summary, the present study indicated that, compared with untreated cells, PLC/RAF/5/5-Fu cells had increased anti-apoptotic and sphere-forming abilities, and exhibited an increase in SP cell number and drug-resistance proteins, and a decrease in sensitivity against 5-Fu. Thus, the present study established a simple and effective strategy to enrich liver CSC-like cells using a low dose of 5-Fu.
Acknowledgements
The present study was supported by grants from the National Natural Science Foundation of China (Beijing, China; grant number 81200465), the Guangdong Natural Science Foundation (Guangzhou, China; grant number 2014A030313785), the Shenzhen Foundation of Science and Technology (Shenzhen, China; grant numbers GJHZ20140414170821192 and JCYJ20140414170821337), and the Health and Family Planning Commission of Shenzhen Municipality (Shenzhen, China; grant number 201303046).
References
- 1.Segura-Lopez FK, Güitrón-Cantu A, Torres J. Association between spp. infections and hepatobiliary malignancies: A review. World J Gastroenterol. 2015;21:1414–1423. doi: 10.3748/wjg.v21.i5.1414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fujimoto A, Totoki Y, Abe T, Boroevich KA, Hosoda F, Nguyen HH, Aoki M, Hosono N, Kubo M, Miya F, et al. Whole-genome sequencing of liver cancers identifies etiological influences on mutation patterns and recurrent mutations in chromatin regulators. Nat Genet. 2012;44:760–764. doi: 10.1038/ng.2291. [DOI] [PubMed] [Google Scholar]
- 3.Gustavsson B, Carlsson G, Machover D, Petrelli N, Roth A, Schmoll HJ, Tveit KM, Gibson F. A review of the evolution of systemic chemotherapy in the management of colorectal cancer. Clin Colorectal Cancer. 2015;14:1–10. doi: 10.1016/j.clcc.2014.11.002. [DOI] [PubMed] [Google Scholar]
- 4.Wang C, Guo Y, Wang J, Min Z. Annexin A2 knockdown inhibits hepatoma cell growth and sensitizes hepatoma cells to 5-fluorouracil by regulating β-catenin and cyclin D1 expression. Mol Med Rep. 2015;11:2147–2152. doi: 10.3892/mmr.2014.2906. [DOI] [PubMed] [Google Scholar]
- 5.Focaccetti C, Bruno A, Magnani E, Bartolini D, Principi E, Dallaglio K, Bucci EO, Finzi G, Sessa F, Noonan DM, Albini A. Effects of 5-fluorouracil on morphology, cell cycle, proliferation, apoptosis, autophagy and ROS production in endothelial cells and cardiomyocytes. PLoS One. 2015;10:e0115686. doi: 10.1371/journal.pone.0115686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sun HC, Tang ZY. Preventive treatments for recurrence after curative resection of hepatocellular carcinoma-a literature review of randomized control trials. World J Gastroenterol. 2003;9:635–640. doi: 10.3748/wjg.v9.i4.635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kumar D, Gupta D, Shankar S, Srivastava RK. Biomolecular characterization of exosomes released from cancer stem cells: Possible implications for biomarker and treatment of cancer. Oncotarget. 2015;6:3280–3291. doi: 10.18632/oncotarget.2462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Abubaker K, Latifi A, Luwor R, Nazaretian S, Zhu H, Quinn MA, Thompson EW, Findlay JK, Ahmed N. Short-term single treatment of chemotherapy results in the enrichment of ovarian cancer stem cell-like cells leading to an increased tumor burden. Mol Cancer. 2013;12:24. doi: 10.1186/1476-4598-12-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ma S, Chan KW, Lee TK, Tang KH, Wo JY, Zheng BJ, Guan XY. Aldehyde dehydrogenase discriminates the CD133 liver cancer stem cell populations. Mol Cancer Res. 2008;6:1146–1153. doi: 10.1158/1541-7786.MCR-08-0035. [DOI] [PubMed] [Google Scholar]
- 10.Jing F, Kim HJ, Kim CH, Kim YJ, Lee JH, Kim HR. Colon cancer stem cell markers CD44 and CD133 in patients with colorectal cancer and synchronous hepatic metastases. Int J Oncol. 2015;46:1582–1588. doi: 10.3892/ijo.2015.2844. [DOI] [PubMed] [Google Scholar]
- 11.Oliver TG, Mercer KL, Sayles LC, Burke JR, Mendus D, Lovejoy KS, Cheng MH, Subramanian A, Mu D, Powers S, et al. Chronic cisplatin treatment promotes enhanced damage repair and tumor progression in a mouse model of lung cancer. Genes Dev. 2010;24:837–852. doi: 10.1101/gad.1897010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chiba T, Kita K, Zheng YW, Yokosuka O, Saisho H, Iwama A, Nakauchi H, Taniguchi H. Side population purified from hepatocellular carcinoma cells harbors cancer stem cell-like properties. Hepatology. 2006;44:240–251. doi: 10.1002/hep.21227. [DOI] [PubMed] [Google Scholar]
- 13.Song J, Chang I, Chen Z, Kang M, Wang CY. Characterization of side populations in HNSCC: Highly invasive, chemoresistant and abnormal Wnt signaling. PLoS One. 2010;5:e11456. doi: 10.1371/journal.pone.0011456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Latifi A, Abubaker K, Castrechini N, Ward AC, Liongue C, Dobill F, Kumar J, Thompson EW, Quinn MA, Findlay JK, Ahmed N. Cisplatin treatment of primary and metastatic epithelial ovarian carcinomas generates residual cells with mesenchymal stem cell-like profile. J Cell Biochem. 2011;112:2850–2864. doi: 10.1002/jcb.23199. [DOI] [PubMed] [Google Scholar]
- 15.Mannelli G, Magnelli L, Deganello A, Busoni M, Meccariello G, Parrinello G, Gallo O. Detection of putative stem cells markers, CD44/CD133, in primary and lymph node metastases in head and neck squamous cell carcinomas. A preliminary immunohistochemical and in vitro study. Clin Otolaryngol. 2015;40:312–320. doi: 10.1111/coa.12368. [DOI] [PubMed] [Google Scholar]
- 16.Arnold KM, Opdenaker LM, Flynn D, Sims-Mourtada J. Wound healing and cancer stem cells: Inflammation as a driver of treatment resistance in breast cancer. Cancer Growth Metastasis. 2015;8:1–13. doi: 10.4137/CGM.S11286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Xu ZY, Tang JN, Xie HX, Du YA, Huang L, Yu PF, Cheng XD. 5-Fluorouracil chemotherapy of gastric cancer generates residual cells with properties of cancer stem cells. Int J Biol Sci. 2015;11:284–294. doi: 10.7150/ijbs.10248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Alama A, Gangemi R, Ferrini S, Barisione G, Orengo AM, Truini M, Bello MG, Grossi F. CD133-positive cells from non-small cell lung cancer show distinct sensitivity to cisplatin and afatinib. Arch Immunol Ther Exp (Warsz) 2015;63:207–214. doi: 10.1007/s00005-015-0330-5. [DOI] [PubMed] [Google Scholar]
- 19.Wang M, Wang Y, Zhong J. Side population cells and drug resistance in breast cancer. Mol Med Rep. 2015;11:4297–4302. doi: 10.3892/mmr.2015.3291. [DOI] [PubMed] [Google Scholar]
- 20.Leccia F, Del Vecchio L, Mariotti E, Di Noto R, Morel AP, Puisieux A, Salvatore F, Ansieau S. ABCG2, a novel antigen to sort luminal progenitors of BRCA1-breast cancer cells. Mol Cancer. 2014;13:213. doi: 10.1186/1476-4598-13-213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Guzel E, Karatas OF, Duz MB, Solak M, Ittmann M, Ozen M. Differential expression of stem cell markers and ABCG2 in recurrent prostate cancer. Prostate. 2014;74:1498–1505. doi: 10.1002/pros.22867. [DOI] [PubMed] [Google Scholar]
- 22.Feuerhake F, Sigg W, Höfter EA, Dimpfl T, Welsch U. Immunohistochemical analysis of Bcl-2 and Bax expression in relation to cell turnover and epithelial differentiation markers in the non-lactating human mammary gland epithelium. Cell Tissue Res. 2000;299:47–58. doi: 10.1007/s004410050005. [DOI] [PubMed] [Google Scholar]
- 23.Lv XG, Ji MY, Dong WG, Lei XF, Liu M, Guo XF, Wang J, Fang C. EBP50 gene transfection promotes 5-fluorouracil-induced apoptosis in gastric cancer cells through Bax- and Bcl-2-triggered mitochondrial pathways. Mol Med Rep. 2012;5:1220–1226. doi: 10.3892/mmr.2012.805. [DOI] [PubMed] [Google Scholar]
- 24.Quan M, Wang P, Cui J, Gao Y, Xie K. The roles of FOXM1 in pancreatic stem cells and carcinogenesis. Mol Cancer. 2013;12:159. doi: 10.1186/1476-4598-12-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bergamaschi A, Madak-Erdogan Z, Kim YJ, Choi YL, Lu H, Katzenellenbogen BS. The forkhead transcription factor FOXM1 promotes endocrine resistance and invasiveness in estrogen receptor-positive breast cancer by expansion of stem-like cancer cells. Breast Cancer Res. 2014;16:436. doi: 10.1186/s13058-014-0436-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Teh MT, Gemenetzidis E, Patel D, Tariq R, Nadir A, Bahta AW, Waseem A, Hutchison IL. FOXM1 induces a global methylation signature that mimics the cancer epigenome in head and neck squamous cell carcinoma. PLoS One. 2012;7:e34329. doi: 10.1371/journal.pone.0034329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yamashita T, Kitao A, Matsui O, Hayashi T, Nio K, Kondo M, Ohno N, Miyati T, Okada H, Yamashita T, et al. Gd-EOB-DTPA-enhanced magnetic resonance imaging and alpha-fetoprotein predict prognosis of early-stage hepatocellular carcinoma. Hepatology. 2014;60:1674–1685. doi: 10.1002/hep.27093. [DOI] [PMC free article] [PubMed] [Google Scholar]