Abstract
Background
Lactobacilli are a group of beneficial bacteria whose anti cancer effects have been evaluated in different cancer cell lines as well as animal models and human subjects. Such anti cancer effects can be exerted via different mechanisms such as modulation of immune response as well as inhibition of pathogens colonization. In addition, lactobacilli have direct cytotoxic effects against cancer cells which may be exerted through modulation of expression cancer related pathways.
Objectives
The aim of this study is to find the mechanism of anti cancer effects of two lactobacilli strains, Lactobacillus. crispatus (LC) and Lactobacillus. rhamnosus (LR).
Materials and Methods
We analyzed expression of some mTOR and Wnt/ β-catenin pathways genes in three cancer cell lines (HeLa, MDA-MB-231 and HT-29) following treatment with LC and LR culture supernatants.
Results
Of note, the expression of CCND1 as a marker of cell proliferation, survival, and angiogenesis, has been decreased following LR treatment in all cell lines. In addition, the expression of SFRP2, an antagonist of Wnt pathway, has been increased in HT-29 following LR treatment and in HeLa cells following LR and LC treatments. Furthermore, we have demonstrated the downregulation of S6K1 expression, a marker of poor prognosis, following LR treatment in HT-29 and following LR and LC treatments in MDA-MB-231 cell line.
Conclusions
Consequently, lactobacilli can modulate expression of mTOR and Wnt/ β-catenin pathways genes in cancer cell lines in a strain specific as well as cell type specific manner.
Keywords: Cancer, mTOR, Wnt/ β-Catenin, Lactobacillus crispatus, Lactobacillus rhamnosus
1. Background
Lactobacilli are a group of probiotic bacteria which colonize the human gastrointestinal tract as well as female urogenital tract (1). Several studies have shown tumor-suppressing properties for certain lactobacillus strains (2, 3). Such anti tumor effects have been postulated to be exerted via different mechanisms such as inhibition of pathogens colonization (4), induction of immune system (5, 6), direct cytotoxic effects on cancer cells (2, 3), antimutagenic effects (7) as well as modulation of carcinogens metabolism and prevention of DNA from oxidative damage (8).
MDA-MB-231 is an aggressive and highly metastatic cell line originated from a high grade tumor (9, 10). HeLa is a cervical cancer cell line in which integration of the human papilloma virus type 18 (HPV-18) genome is proposed as a initiator event in the tumorigenesis (11). Previously we have demonstrated that treatment with lactobacilli culture supernatants decreases the expression of HPV E6 oncogene so it may be of therapeutic value (12). HT-29 is a colorectal adenocarcinoma cell line in which the apoptotic effects of different lactobacilli strains have been evaluated (13).
The phosphoinositide 3 kinase (PI3K)/Akt/mammalian (or mechanistic) target of rapamycin (mTOR) pathway is an essential pathway leading to cell growth and tumor proliferation. This pathway is involved in resistance to endocrine therapy, HER2-directed therapy and cytotoxic therapy in breast cancer (14). In addition, this pathway has been shown to be frequently deregulated in cervical cancers (15) as well as colorectal cancers (16). Activation of mTOR complex 1 leads to phosphorylation of some factors and results in selective overexpression of cyclin D1, Bcl-2, Bcl-xL and vascular endothelial growth factor (VEGF) as well as the nucleocytoplasmic transport of selected mRNA such as cyclin D1. Consequently, it increases cell proliferation, survival, and angiogenesis. S6K1 is a critical regulator of cell growth, which phosphorylates ribosomal protein S6 and other important targets. Both eIF4E and S6K1 are involved in cellular transformation, and their overexpression has associated with poor cancer prognosis. mTOR and RICTOR are implicated in AKT phosphorylation and activation and have role in AKT interaction with the apoptosis regulator BAD. mTOR complex 2 has been involved in the posphorylation of PRKCA (17) which is in turn implicated in various cellular processes such as cell adhesion, cell transformation and cell cycle checkpoint. PRKCA has been associated with metastatic potential of breast cancer through the activation of matrix metaloproteinases and has been regarded as poor prognostic marker as well as a therapeutic target in cancer patients (18, 19).
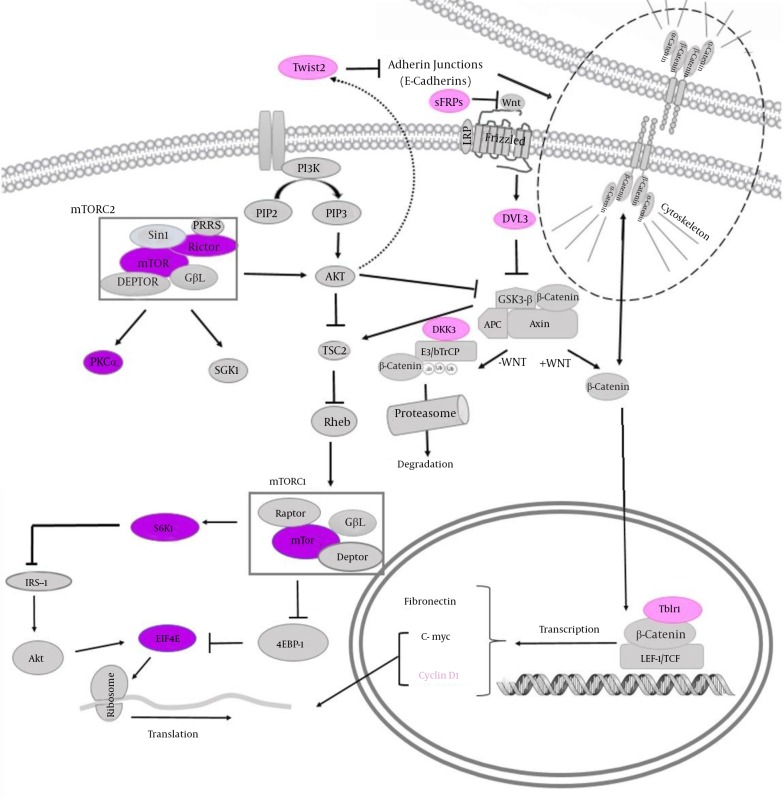
Deregulation of the canonical Wnt/β-catenin signaling pathway has been implicated in several cancers including breast, cervical and colorectal cancers (20). SFRP2 encodes a soluble modulator of Wnt signaling. Methylation of this gene is a potential marker for the presence of colorectal cancer (21), cervical cancer (22) as well as breast cancer (23). Similar to other secreted Frizzled related proteins, SFRP2 acts as antagonist of Wnt pathway by squelching Wnt ligands (24). DKK3 codes for a Dickkopf (Dkk) protein which effectively inhibits Wnt signaling by preventing Wnt interaction with LRPs (24). DVL3 codes one of Disheveled (Dvl) proteins which becomes phosphorylated upon Wnt stimulation thereby stabilizes β-catenin (24). TWIST genes code for an essential factor for epithelial-mesenchymal transition (EMT). The key TWIST isoform which couples aberrant signals from EMT to senescence has been shown to be TWIST2. This isoform has been suggested as an important candidate biomarker for cervical cancer prognosis (25). TBLR1 has a critical role in nuclear β-catenin function. In addition, the depletion of TBL1-TBLR1 has considerably hindered Wnt-beta-catenin-induced gene expression and oncogenic growth in vitro and in vivo (26). Finally, CCND1 codes for cyclin D1, an oncogene and an important positive regulator of the G1/S phase which is a downstream target of β-catenin (27). Figure 1 shows the position of selected proteins in the mTOR and Wnt pathways as well as the interaction of these pathways.
Figure 1. The Position of Selected Genes in mTOR (in Purple) and Wnt/ β-Catenin (in Pink) Pathways and Interactions of These Pathways.
2. Objectives
In this study, we aimed at determination of cellular pathways involved in the cytotoxic effects of two lactobacilli strains namely, L. rhamnosus and L. crispatus against MDA-MB-231, HeLa and HT-29 cancer cell lines.
3. Materials and Methods
3.1. Selection of Genes From mTOR and Wnt/β-Catenin Pathways
Based on previous expression studies, 5 genes were selected from mTOR pathway to evaluate expression of different parts of this pathway: RICTOR, S6K1, EIF4E, PRKCA and MTOR. Furthermore, SFRP2, TBLR1, DVL3, CCND1, DKK3 and TWIST2 genes were chosen from Wnt/β-catenin pathway to evaluate their expression following treatment with lactobacilli supernatants. Selection of genes from this pathway was based on previous reports regarding epigenetic deregulation in various cancer types. Fold changes in the expression of these genes have been analyzed after certain treatments.
3.2. Cell culture
This study has been approved by the ethical committee of Shahid Beheshti University of Medical Sciences. Human cervical cancers (HeLa), breast cancer (MDA-MB-231), colorectal cancer (HT-29) as well as human lung fibroblst (MRC5) cell lines cell lines were purchased from the Pasteur Institute, National Cell Bank of Iran and cultured according to previous studies (28).
3.3. Preparation of Supernatants From Lactobacillus Cultures
Microaerophilic conditions were applied for culture of L. crispatus strain SJ-3C-US and L. rhamnosus strain GG in de Man Rogosa Sharpe (MRS) broth (Merck; pH 6.5) as described in previous publications (28). To check the probable effect of lactobacilli-produced lactic acid on cell cultures, the pH of MRS controls have been adjusted by lactic acid based on the corresponding lactobacilli supernatant pH. In brief, the experiments comprised L. crispatus supernatant, pH 4.3 (LCS); L. rhamnosus supernatant, pH 4.05 (LRS); MRS, pH 6.5 and MRS adjusted with lactate (MRL) pH 4.05 or 4.3.
3.4. MTT Assay
Cell growth inhibition was computed by MTT [3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide] assay kit (Sigma, St. Louis , MO) according to previous studies (29). Overnight treatments with lactobacilli culture supernatants cells performed with different concentrations from 10% to 100% (v/v). Plates were incubated at 37°C under 5% (v/v) CO2. Cell viability was computed using the following equation (Equation 1):
| Equation 1. |
3.5. RNA Isolation, cDNA Synthesis and Quantitative RT-PCR (qRT-PCR)
The AccuZolTM total RNA extraction solution (Bioneer, Korea) was used to isolate total RNA from cultured cells. Nanodrop 2000 c spectrophotometer (Thermo Scientific) was used for determination of RNA concentration. Changes in mRNA expression of desired genes were analyzed by quantitative PCR (qPCR) after reverse transcription of 1 μg RNA from each sample with the PrimeScript RT reagent kit (Takara Bio, Ohtsu, Japan). mRNA quantification of genes was implemented in a rotor gene 3000 corbette detection system using SYBR Premix Ex Taq (Takara Bio, Ohtsu, Japan). Primer sequences are listed in Table 1. PCR condition was as follows: an initial denaturation at 95°C for 1 minute, and 40 cycles at 95°C for 15 seconds and 65°C for 1 minute. The final PCR reaction consisted 10 mL SYBR Green master mix, 2 mL cDNA, 0.5 mL each forward and reverse primer (10 pmol) and 7 mL nuclease-free water. Experiments were performed in duplicate for each data point. B2ACTIN mRNA was amplified as a normalizer, and fold changes in each target mRNA expression relative to B2ACTIN were calculated. Melting curve analysis was used to validate whether primers yielded a single PCR product.
Table 1. Sequence of Primers Used in This Study.
| Primer | Sequence | Product Size, bp |
|---|---|---|
| Β2ACTIN | 105 | |
| Forward | AGATGAGTATGCCTGCCGTG | |
| Reverse | GCGGCATCTTCAAACCTCCA | |
| SFRP2 | 162 | |
| Forward | ACCGAGGAAGCTCCAAAGGT | |
| Reverse | GCTCTTGGTCTCCAGGATGATT | |
| TBLR1 | 141 | |
| Forward | GGGAGGAGAATGGAGCACAT | |
| Reverse | CAGGGTTCCAGGCACAGATA | |
| DVL3 | 130 | |
| Forward | TGGACGACGATTTCGGAGTG | |
| Reverse | TTATCAGCACAGAAGGGGGC | |
| CCND1 | 180 | |
| Forward | GAGGCGGAGGAGAACAAACA | |
| Reverse | GAGGCGGTAGTAGGACAGGA | |
| DKK3 | 112 | |
| Forward | CCTGGCAAACTTACCTCCC | |
| Reverse | AGTCTGGTTGTTGGTTATCTTGT | |
| TWIST2 | 142 | |
| Forward | GTGACATCGGACAGAAGA | |
| Reverse | CAAACATAAGACCCAGAAGAAA | |
| RICTOR | 195 | |
| Forward | ACAACAGAGCAACGAGGTA | |
| Reverse | TCTGGATTCTGAAGTGCTAGTT | |
| S6K1 | 167 | |
| Forward | TGCTTAATCACCAAGGTCAT | |
| Reverse | TCCCAAACTCCACCAATC | |
| EIF4E | 159 | |
| Forward | CCAGGGCCAAACGGACATA | |
| Reverse | GGGATTAGGAGTAGGGGTGGT | |
| PRKCA | 131 | |
| Forward | TGCAAAGGACTGATGACCAAAC | |
| Reverse | GGCTGGATCTCCCTGTTCTC | |
| MTOR | 168 | |
| Forward | TGGGGACTGCTTTGAGGTTG | |
| Reverse | ACACTGTCCTTGTGCTCTCG |
3.6. Statistical Analysis
Relative expression software tool (REST©) was applied for comparison of the total expression ratio of the genes between treated and control cells using a randomization test.
Mann-Whitney test was used for comparison of IC50 (concentration giving half-maximal inhibition) of cells treated with lactobacilli culture supernatants and pH- and lactate adjusted as well as pretreated controls in SPSS software (version 16.0). All data were expressed as a mean ± SE of three separate experiments. P < 0.05 was considered as statistically significant.
4. Results
4.1. The Effects of LCS and LRS on HeLa, MDA-MB-231 and HT-29 Cell Proliferation
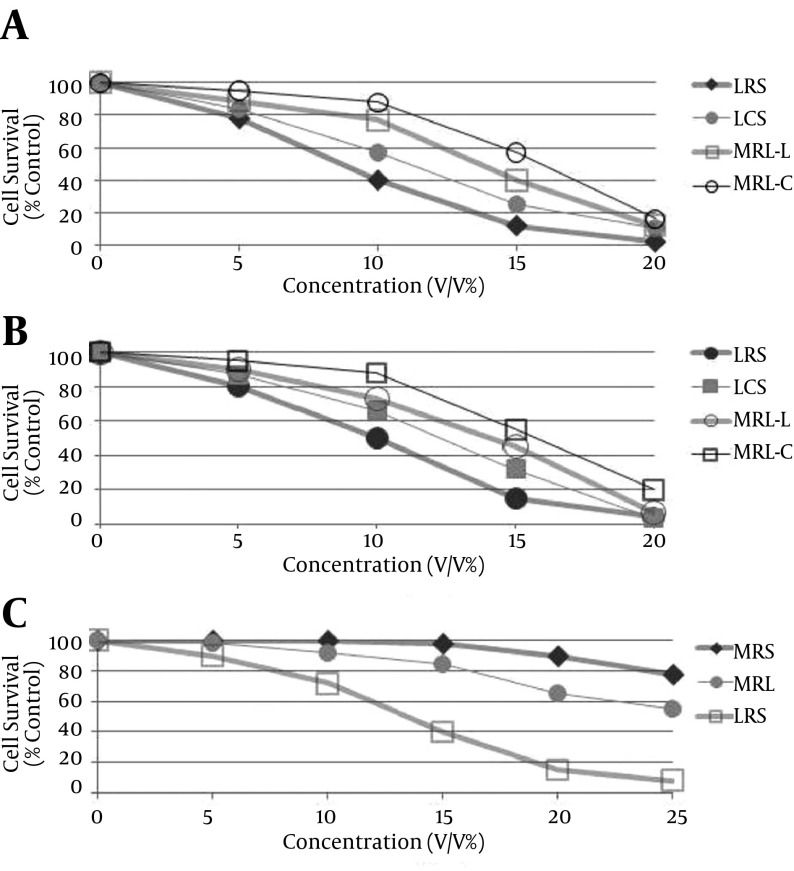
LCS and LRS had no toxicity against MRC5 cells as we reported previously (30). The IC50 values of LRS against HeLa and MDA-MB-231 cells were 9 and 10% (v/v) and those of LCS were 11 and 13% (v/v) respectively. The cytotoxic effects of LCS and LRS against HeLa and MDA-MB-231 cells were higher than those of MRS and MRL (MRS with pH adjusted to that of LCS and LRS) (P < 0.05) (Figures 2A and 2B). LRS but not LCS has cytotoxic effects against HT-29 cell (Figure 2C). The IC50 of LRS against HT-29 cells was 14% (v/v). These results showed that the main cause of cancer cell death was not the acidity. This cytotoxicity against cancer cells can be attributed to a substance other than lactate in the supernatant of the lactobacilli. In addition, cytotoxicity effect of LRS was significantly higher than LCS in three cancer cell lines examined (P < 0.01).
Figure 2. Cell Growth Inhibitory Effects of Different Concentrations of Lactobacillus crispatus Culture Supernatant (LCS), Lactobacillus rhamnosus Culture Supernatant (LRS), MRS and MRL on 2A, HeLa; 2B, MDA-MB-231; 2C, HT-29 Cells Respectively.
4.2. The Effects of LCS and LRS on Expression of mTOR and Wnt/ β-Catenin Pathways Genes
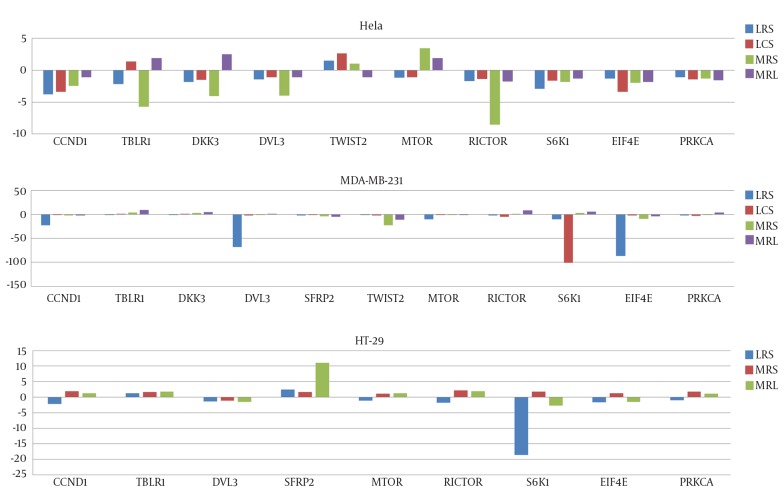
mRNA levels of mentioned genes in cancer cell lines were measured by qRT-PCR before and after treatment with LCS and LRS. After 4 hours treatment of cancer cells with certain percentages (v/v) of culture supernatants (based on the observed IC50 for each LS against a cell line), the effects of LRS and MRS on genes expression was compared with MRS and MRL. All genes have been expressed in three cell line before treatment except for SFRP2 whose expression has not been detected in HeLa cells before treatment but has been significantly upregulated following both LRS and LCS treatments. Figure 3 shows the effects of LCS and LRS on the expression of mTOR and Wnt/ β-catenin pathways genes in three cell lines respectively.
Figure 3. The Effects of Lactobacillus crispatus Culture Supernatant (LCS), Lactobacillus rhamnosus Culture Supernatant (LRS), MRS and MRL on Expression of Selected Genes From mTOR and Wnt/ β-Catenin Pathways in HeLa, MDA-MB-231 and HT-29 Cells Respectively.
5. Discussion
In this experiment, we have demonstrated downregulation of some mTOR related genes following LS treatment in MDA-MB-231, HeLa and HT-29 cell lines. Of note, the effects of LS on expression of genes have been strain specific as well as cell line specific. For instance, expression of EIF4E has been decreased in MDA-MB-231 cells following LRS treatment by the factors 87. However, LCS has resulted in a 100 fold reduction in S6K1 expression in the same cell line. mTOR inhibitors have shown anti-tumor activity against various human cancers. Combinations of mTOR inhibitors with other treatment strategies such as cytotoxic chemotherapy as well as a variety of targeted molecular agents have shown promising results in many patients (15). Among mTOR inhibitors which are currently in clinical use are rapamycin and its analogs. These drugs have been shown to bind to a domain rather than the catalytic site and inhibit various mTOR functions. A potential drawback of these drugs is that they may activate an mTOR-dependent survival pathway resulting in treatment failure. However, small molecules that compete with ATP in the catalytic site have been shown to inhibit all of the kinase-dependent functions of mTOR without activating the survival pathway (31). Here we have shown that supernatants from two lactobacilli cultures significantly downregulates expression of some genes in mTOR pathway and can be regarded as a mechanism by which these lactobacilli exert their cytotoxic effects against cancer cells. As we have shown the cytotoxic effects of these lactobacilli on cancer cells, the possibility of activating the survival pathway by these lactobacilli is probably ruled out. In addition, considering the role of mTOR pathway in resistance to target specific therapies in breast cancer, downregulation of some mTOR genes in triple negative MDA-MB-231 cells by lactobacilli may be of therapeutic value.
In order to translate the result of these kinds of studies into the clinical use, it is necessary to find the fraction of culture supernatant which is responsible for such effect. However, it is possible that different fractions have synergic effects. Future studies should focus on identification of lactobacilli fractions which confers cytotoxic effects against cancer cells as well as those modulate cancer-related pathways. Furthermore, in this study we just evaluated expression of these targets at mRNA level. As phosphorylation status of different proteins in mTOR pathway is important in regulation of this pathway, future studies should investigate the effect of lactobacilli-derived products on phosphorylation of these proteins.
In addition, we have demonstrated modulation of different parts of Wnt/ β-catenin pathway following lactobacilli treatment in different cell lines. Of note, SFRP2 expression has not been detected in HeLa cells before lactobacilli treatment, but considerably has been upregulated following treatment. As revealed by a former study, restoration of the expression of SFRP2 has resulted in decreased Wnt signaling in CaSki cervical cancer cells, decreased abnormal accumulation of free β-catenin in the nucleus, and inhibited cancer cell growth. In addition, SFRP2 inhibited the expression of three transcription factors involved in the EMT program including TWIST (32). However, in our experiment, upregulation of SFRP2 in HeLa cells has not been accompanied by downregulation of TWIST2 expression. In addition, treatment of HT-29 cells with LRS has increased the expression of SFRP2 while decreased the expression of CCND1. CCND1 has been regarded as an unfavorable prognostic factor for colorectal cancer (33), so its downregulation following LS treatment may be of clinical value. As SFRP genes are regarded as targets of cancer specific hypermethylation in the colon (34, 35), upregulation of SFRP2 expression in HT-29 cells following LRS treatment implies a role for lactobacilli in epigenetic regulation of gene expression, which should be evaluated in future studies.
Additionally, we have demonstrated downregulation of TBLR1, a prognostic marker in cervical cancer with a critical role in the invasion and metastasis (36) in HeLa cells following LRS treatment. However, as such effect has been seen following MRS treatment as well, it is not considered as significant. In addition, CCND1 has been downregulated in HeLa cells after LS treatment. As CCND1 is regarded as a marker of poor prognosis in early stage cervical cancer (37), its downregulation by LS may have a clinical significance.
A previous study has shown that the transformation of HPV expressing human keratinocytes needs activation of the Wnt pathway (38). Furthermore, E6 and E7 have been shown to be involved in β-catenin nuclear accumulation and activation of Wnt signaling in HPV-induced cancers (39). Downregulation of Wnt-β catenin pathway in cervical cancer following lactobacilli treatment in addition to our previous data regarding down regulation of HPV E6 oncogene by lactobacilli in these cells (3) implies that certain lactobacilli strains can defeat cervical cancer by various means.
In addition, we have demonstrated that treatment with LRS can result in downregulation of CCND1 and DVL3 expressions in MDA-MB-231 cells by the factors 22 and 68 respectively. Downregulation of these Wnt agonists by lactobacilli provide a possible explanation for beneficial effects of lactobacilli in the treatment of breast cancer patients. However, DKK3 is regarded as a putative Wnt signaling inhibitor (40) whose expression has not been significantly changed following LS treatments.
5.1. Conclusions
Lactobacilli can modulate expression of mTOR and Wnt/ β-catenin pathways genes in cancer cell lines in a strain specific as well as cell type specific manner. Considering the role of lactobacilli in cancer prevention and treatment, understanding how the lactobacilli-derived products inhibit cancer-related signaling pathways may shed new insights on design and development of novel anti cancer strategies.
Acknowledgments
This study was supported by a grant from Shahid Beheshti University of Medical Sciences and done as M.Sc. project of the first two authors.
Footnotes
Authors’ Contribution:Zahra Taherian-Esfahani and Atieh Abedin-Do contributed equally to this manuscript. Zahra Taherian-Esfahani and Atieh Abedin-Do carried out the experiments. Zahra Nouri helped in cell culture, Reza Mirfakhraie assisted in gene expression analyses. Soudeh Ghafouri-Fard and Elahe Motevaseli designed the study and supervised the project. Soudeh Ghafouri-Fard wrote the manuscript.
Conflict of Interests:None declared.
Financial Disclosure:None declared.
Funding/Support:None declared.
References
- 1.Motevaseli E, Shirzad M, Raoofian R, Hasheminasab SM, Hatami M, Dianatpour M, et al. Differences in vaginal lactobacilli composition of Iranian healthy and bacterial vaginosis infected women: a comparative analysis of their cytotoxic effects with commercial vaginal probiotics. Iran Red Crescent Med J. 2013;15(3):199–206. doi: 10.5812/ircmj.3533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Motevaseli E, Shirzad M, Akrami SM, Mousavi AS, Mirsalehian A, Modarressi MH. Normal and tumour cervical cells respond differently to vaginal lactobacilli, independent of pH and lactate. J Med Microbiol. 2013;62(Pt 7):1065–72. doi: 10.1099/jmm.0.057521-0. [DOI] [PubMed] [Google Scholar]
- 3.Azam R, Ghafouri-Fard S, Tabrizi M, Modarressi MH, Ebrahimzadeh-Vesal R, Daneshvar M, et al. Lactobacillus acidophilus and Lactobacillus crispatus culture supernatants downregulate expression of cancer-testis genes in the MDA-MB-231 cell line. Asian Pac J Cancer Prev. 2014;15(10):4255–9. doi: 10.7314/apjcp.2014.15.10.4255. [DOI] [PubMed] [Google Scholar]
- 4.Zabihollahi R, Motevaseli E, Sadat SM, Azizi-Saraji AR, Asaadi-Dalaie S, Modarressi MH. Inhibition of HIV and HSV infection by vaginal lactobacilli in vitro and in vivo. Daru. 2012;20(1):53. doi: 10.1186/2008-2231-20-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Murosaki S, Muroyama K, Yamamoto Y, Yoshikai Y. Antitumor effect of heat-killed Lactobacillus plantarum L-137 through restoration of impaired interleukin-12 production in tumor-bearing mice. Cancer Immunol Immunother. 2000;49(3):157–64. doi: 10.1007/s002620050615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abedin-Do A, Taherian-Esfahani Z, Ghafouri-Fard S, Ghafouri-Fard S, Motevaseli E. Immunomodulatory effects of Lactobacillus strains: emphasis on their effects on cancer cells. Immunotherapy. 2015;7(12):1307–29. doi: 10.2217/imt.15.92. [DOI] [PubMed] [Google Scholar]
- 7.Ahmadi MA, Ebrahimi MT, Mehrabian S, Tafvizi F, Bahrami H, Dameshghian M. Antimutagenic and anticancer effects of lactic acid bacteria isolated from Tarhana through Ames test and phylogenetic analysis by 16S rDNA. Nutr Cancer. 2014;66(8):1406–13. doi: 10.1080/01635581.2014.956254. [DOI] [PubMed] [Google Scholar]
- 8.Zhang M, Wang F, Jiang L, Liu R, Zhang L, Lei X, et al. Lactobacillus salivarius REN inhibits rat oral cancer induced by 4-nitroquioline 1-oxide. Cancer Prev Res (Phila). 2013;6(7):686–94. doi: 10.1158/1940-6207.CAPR-12-0427. [DOI] [PubMed] [Google Scholar]
- 9.Chavez KJ, Garimella SV, Lipkowitz S. Triple negative breast cancer cell lines: one tool in the search for better treatment of triple negative breast cancer. Breast Dis. 2010;32(1-2):35–48. doi: 10.3233/BD-2010-0307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shokrollahi Barough M, Hasanzadeh H, Barati M, Pak F, Kokhaei P, Rezaei-Tavirani M. Apoptosis/Necrosis Induction by Ultraviolet, in ER Positive and ER Negative Breast Cancer Cell Lines. Iran J Cancer Prev. 2015;8(6):e5369. doi: 10.17795/ijcp-4193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Adey A, Burton JN, Kitzman JO, Hiatt JB, Lewis AP, Martin BK, et al. The haplotype-resolved genome and epigenome of the aneuploid HeLa cancer cell line. Nature. 2013;500(7461):207–11. doi: 10.1038/nature12064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Motevaseli E, Azam R, Akrami SM, Mazlomy M, Saffari M, Modarressi MH, et al. The Effect of Lactobacillus crispatus and Lactobacillus rhamnosusCulture Supernatants on Expression of Autophagy Genes and HPV E6 and E7 Oncogenes in The HeLa Cell Line. Cell J. 2016;17(4):601–7. doi: 10.22074/cellj.2016.3833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang SM, Zhang LW, Fan RB, Han X, Yi HX, Zhang LL, et al. Induction of HT-29 cells apoptosis by lactobacilli isolated from fermented products. Res Microbiol. 2014;165(3):202–14. doi: 10.1016/j.resmic.2014.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Paplomata E, O'Regan R. The PI3K/AKT/mTOR pathway in breast cancer: targets, trials and biomarkers. Ther Adv Med Oncol. 2014;6(4):154–66. doi: 10.1177/1758834014530023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Husseinzadeh N, Husseinzadeh HD. mTOR inhibitors and their clinical application in cervical, endometrial and ovarian cancers: a critical review. Gynecol Oncol. 2014;133(2):375–81. doi: 10.1016/j.ygyno.2014.02.017. [DOI] [PubMed] [Google Scholar]
- 16.Francipane MG, Lagasse E. mTOR pathway in colorectal cancer: an update. Oncotarget. 2014;5(1):49–66. doi: 10.18632/oncotarget.1548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Meric-Bernstam F, Gonzalez-Angulo AM. Targeting the mTOR signaling network for cancer therapy. J Clin Oncol. 2009;27(13):2278–87. doi: 10.1200/JCO.2008.20.0766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kang JH. Protein kinase C (PKC) isozymes and cancer. N J Sci. 2014:231418. [Google Scholar]
- 19.Lonne GK, Cornmark L, Zahirovic IO, Landberg G, Jirstrom K, Larsson C. PKCalpha expression is a marker for breast cancer aggressiveness. Mol Cancer. 2010;9:76. doi: 10.1186/1476-4598-9-76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang W, Xue L, Liu H, Wang P, Xu P, Cai Y. Aberrant changes of Wnt2/beta-catenin signaling pathway induced by sodium nitroprusside in human esophageal squamous cell carcinoma cell lines. Cancer Invest. 2010;28(3):230–41. doi: 10.3109/07357900903095698. [DOI] [PubMed] [Google Scholar]
- 21.Chang E, Park DI, Kim YJ, Kim BK, Park JH, Kim HJ, et al. Detection of colorectal neoplasm using promoter methylation of ITGA4, SFRP2, and p16 in stool samples: a preliminary report in Korean patients. Hepatogastroenterology. 2010;57(101):720–7. [PubMed] [Google Scholar]
- 22.Chung MT, Sytwu HK, Yan MD, Shih YL, Chang CC, Yu MH, et al. Promoter methylation of SFRPs gene family in cervical cancer. Gynecol Oncol. 2009;112(2):301–6. doi: 10.1016/j.ygyno.2008.10.004. [DOI] [PubMed] [Google Scholar]
- 23.Veeck J, Noetzel E, Bektas N, Jost E, Hartmann A, Knuchel R, et al. Promoter hypermethylation of the SFRP2 gene is a high-frequent alteration and tumor-specific epigenetic marker in human breast cancer. Mol Cancer. 2008;7:83. doi: 10.1186/1476-4598-7-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Luu HH, Zhang R, Haydon RC, Rayburn E, Kang Q, Si W, et al. Wnt/beta-catenin signaling pathway as a novel cancer drug target. Curr Cancer Drug Targets. 2004;4(8):653–71. doi: 10.2174/1568009043332709. [DOI] [PubMed] [Google Scholar]
- 25.Wang T, Li Y, Wang W, Tuerhanjiang A, Wu Z, Yang R, et al. Twist2, the key Twist isoform related to prognosis, promotes invasion of cervical cancer by inducing epithelial-mesenchymal transition and blocking senescence. Hum Pathol. 2014;45(9):1839–46. doi: 10.1016/j.humpath.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 26.Li J, Wang CY. TBL1-TBLR1 and beta-catenin recruit each other to Wnt target-gene promoter for transcription activation and oncogenesis. Nat Cell Biol. 2008;10(2):160–9. doi: 10.1038/ncb1684. [DOI] [PubMed] [Google Scholar]
- 27.Polakis P. Wnt signaling and cancer. Genes Dev. 2000;14(15):1837–51. [PubMed] [Google Scholar]
- 28.Esfandiary A, Taherian-Esfahani Z, Abedin-Do A, Mirfakhraie R, Shirzad M, Ghafouri-Fard S, et al. Lactobacilli Modulate Hypoxia-Inducible Factor (HIF)-1 Regulatory Pathway in Triple Negative Breast Cancer Cell Line. Yakhteh. 2016;18(2) doi: 10.22074/cellj.2016.4319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zare-Mirakabadi A, Sarzaeem A, Moradhaseli S, Sayad A, Negahdary M. Necrotic Effect versus Apoptotic Nature of Camptothecin in Human Cervical Cancer Cells. Iran J Cancer Prev. 2012;5(3):109–16. [PMC free article] [PubMed] [Google Scholar]
- 30.Nouri Z, Karami F, Neyazi N, Modarressi MH, Karimi R, Khorramizadeh MR. Dual anti-metastatic and anti-proliferative activity assessment of two probiotics on HeLa and HT-29 cell lines. Yakhteh. 2016;18(2):127–34. doi: 10.22074/cellj.2016.4307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ballou LM, Lin RZ. Rapamycin and mTOR kinase inhibitors. J Chem Biol. 2008;1(1-4):27–36. doi: 10.1007/s12154-008-0003-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chung MT, Lai HC, Sytwu HK, Yan MD, Shih YL, Chang CC, et al. SFRP1 and SFRP2 suppress the transformation and invasion abilities of cervical cancer cells through Wnt signal pathway. Gynecol Oncol. 2009;112(3):646–53. doi: 10.1016/j.ygyno.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 33.Li Y, Wei J, Xu C, Zhao Z, You T. Prognostic significance of cyclin D1 expression in colorectal cancer: a meta-analysis of observational studies. PLoS One. 2014;9(4):e5369. doi: 10.1371/journal.pone.0094508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mori Y, Olaru AV, Cheng Y, Agarwal R, Yang J, Luvsanjav D, et al. Novel candidate colorectal cancer biomarkers identified by methylation microarray-based scanning. Endocr Relat Cancer. 2011;18(4):465–78. doi: 10.1530/ERC-11-0083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Safaei A, Sobhi S, Rezaei-Tavirani M, Zali MR. Genomic and epigenetic instability in colorectal cancer. Iran J Cancer Prevent. 2013;6:54–63. [Google Scholar]
- 36.Wang J, Ou J, Guo Y, Dai T, Li X, Liu J, et al. TBLR1 is a novel prognostic marker and promotes epithelial-mesenchymal transition in cervical cancer. Br J Cancer. 2014;111(1):112–24. doi: 10.1038/bjc.2014.278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bae DS, Cho SB, Kim YJ, Whang JD, Song SY, Park CS, et al. Aberrant expression of cyclin D1 is associated with poor prognosis in early stage cervical cancer of the uterus. Gynecol Oncol. 2001;81(3):341–7. doi: 10.1006/gyno.2001.6196. [DOI] [PubMed] [Google Scholar]
- 38.Uren A, Fallen S, Yuan H, Usubutun A, Kucukali T, Schlegel R, et al. Activation of the canonical Wnt pathway during genital keratinocyte transformation: a model for cervical cancer progression. Cancer Res. 2005;65(14):6199–206. doi: 10.1158/0008-5472.CAN-05-0455. [DOI] [PubMed] [Google Scholar]
- 39.Rampias T, Boutati E, Pectasides E, Sasaki C, Kountourakis P, Weinberger P, et al. Activation of Wnt signaling pathway by human papillomavirus E6 and E7 oncogenes in HPV16-positive oropharyngeal squamous carcinoma cells. Mol Cancer Res. 2010;8(3):433–43. doi: 10.1158/1541-7786.MCR-09-0345. [DOI] [PubMed] [Google Scholar]
- 40.Veeck J, Bektas N, Hartmann A, Kristiansen G, Heindrichs U, Knuchel R, et al. Wnt signalling in human breast cancer: expression of the putative Wnt inhibitor Dickkopf-3 (DKK3) is frequently suppressed by promoter hypermethylation in mammary tumours. Breast Cancer Res. 2008;10(5):R82. doi: 10.1186/bcr2151. [DOI] [PMC free article] [PubMed] [Google Scholar]