Abstract
Background & Aims
Monocyte and macrophage (MΦ) activation contributes to the pathogenesis of chronic hepatitis C virus (HCV) infection. Disease pathogenesis is regulated by both liver-resident MΦs and monocytes recruited as precursors of MΦs into the damaged liver. Monocytes differentiate into M1 (classic/proinflammatory) or M2 (alternative/anti-inflammatory) polarized MΦs in response to tissue microenvironment. We hypothesized that HCV-infected hepatoma cells (infected with Japanese fulminant hepatitis-1 [Huh7.5/JFH-1]) induce monocyte differentiation into polarized MΦs.
Methods
Healthy human monocytes were co-cultured with Huh7.5/JFH-1 cells or cell-free virus for 7 days and analyzed for MΦ markers and cytokine levels. A similar analysis was performed on circulating monocytes and liver MΦs from HCV-infected patients and controls.
Results
Huh7.5/JFH-1 cells induced monocytes to differentiate into MΦs with increased expression of CD14 and CD68. HCV-MΦs showed M2 surface markers (CD206, CD163, and Dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN)) and produced both proinflammatory and anti-inflammatory cytokines. HCV-induced early interleukin 1β production promoted transforming growth factor (TGF)β production and MΦ polarization to an M2 phenotype. TGF-β secreted by M2-MΦ led to hepatic stellate cell activation indicated by increased expression of collagen, tissue inhibitor of metalloproteinase 1, and α-smooth muscle actin. In vivo, we observed a significant increase in M2 marker (CD206) expression on circulating monocytes and in the liver of chronic HCV-infected patients. Furthermore, we observed the presence of a unique collagen-expressing CD14+CD206+ monocyte population in HCV patients that correlated with liver fibrosis.
Conclusions
We show an important role for HCV in induction of monocyte differentiation into MΦs with a mixed M1/M2 cytokine profile and M2 surface phenotype that promote stellate cell activation via TGF-β. We also identified circulating monocytes expressing M2 marker and collagen in chronic HCV infection that can be explored as a biomarker.
Keywords: Collagen, Fibrocytes, CD206, Biomarkers
Abbreviations used in this paper: α-SMA, α-smooth muscle actin; APC, antigen-presenting cell; COL, collagen; FITC, fluorescein isothiocyanate; HCV, hepatitis C virus; HSC, hepatic stellate cell; Huh7.5/JFH-1, Huh7.5 cells infected with JFH-1 (HCV); IL, interleukin; IL1RA, IL1-receptor antagonist; JFH-1, Japanese fulminant hepatitis-1; MΦ, macrophage; MFI, mean fluorescence intensity; mRNA, messenger RNA; NEAA, nonessential amino acid; PBMC, peripheral blood mononuclear cell; PE, Phycoerythrin; TGF, transforming growth factor; TIMP, tissue inhibitor of metalloproteinase; TNF, tumor necrosis factor
Summary.
It is not known whether hepatitis C virus (HCV) infection causes monocytes to differentiate into polarized macrophages. We showed that HCV-infected hepatoma cells or cell-free HCV induce monocyte differentiation into M2-polarized macrophages, which activate hepatic stellate cells and validated our findings in monocytes from chronic HCV patients.
Hepatitis C virus (HCV) is a prevalent pathogen worldwide and is a leading cause of morbidity and mortality. According to recent estimates, more than 185 million people are infected with HCV and more than 350,000 people die annually from HCV-related liver diseases.1, 2 Extensive research has led to the development of various therapeutic drugs for the disease.3 The US Food and Drug Administration recently approved 3 new drugs: sofosbuvir (RNA polymerase inhibitor), simeprevir (protease inhibitor), and sofosbuvir plus ledipasvir (direct-acting antivirals); thus marking a new era in HCV treatment because these drugs achieve sustained virologic response rates greater than 95% in most patients with shorter treatments and minimal adverse effects.4 HCV establishes chronicity in 50%–80% of infected individuals, leading to liver inflammation and fibrosis. Clearance, persistence, and immunopathology of HCV infection depends on the interplay between the innate and adaptive immune responses in the liver.
Innate immune cells are important for recognizing HCV and influence the outcome of the viral infection.5, 6, 7, 8, 9, 10 Prolonged immune cell activation during chronic infections such as HCV and human immunodeficiency virus can drive viral replication and viral disease progression.11, 12 Monocytes are attracted to the site of infection and are exposed to viral RNA and protein, which can lead to their activation.9, 13, 14, 15 Monocytes are characterized by considerable plasticity. Monocytes traffic to sites of infection and differentiate into macrophages (MΦ) with distinct phenotypes, and also replenish the Kupffer cells in the liver.14, 16, 17, 18 MΦs may undergo polarization into classically activated (M1) or alternatively activated (M2) phenotypes.19, 20 During chronic infection, immune activation can lead to an anti-inflammatory response to limit immunopathology, which can be characterized by the presence of M2-like macrophages. M1 MΦs express CD86, major histocompatibility complex II, CD40, and CD16 molecules on their surface, whereas the CD206 (mannose receptor), Dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN), and CD163 (scavenger receptor) characterize the M2 signature.21, 22
Chronic HCV infection leads to progressive hepatic fibrosis, however, there is a lack of consensus on whether fibrosis is a direct consequence of the virus infection or a result of chronic hepatic inflammation.23 Although proinflammatory cytokines including tumor necrosis factor (TNF)α and interleukin (IL)1β have been implicated in the fibrosis process, anti-inflammatory transforming growth factor (TGF)-β is the most potent profibrogenic cytokine.24, 25, 26, 27 A study by Li et al28 showed that TGF-β produced by HCV-specific intrahepatic lymphocytes (T-regulatory cells) correlated inversely with liver fibrosis. This implies the presence of yet another cellular source of TGF-β, which leads to hepatic stellate cell (HSC) activation and fibrosis in chronic HCV infection. Because of the lack of small animal models of HCV infection, in vitro culture systems have been used to study the activation and function of immune cells.29
In this study, we evaluated changes in the phenotype and function of healthy human monocytes cultured in the presence of HCV-infected hepatoma cells or cell-free HCV. We present evidence that HCV can induce circulating monocytes to differentiate into macrophages with a mixed M1/M2 cytokine production and M2 surface marker expression. We also show that chronic presence of HCV can program monocytes and induce HSC activation via TGF-β during HCV infection. Finally, we delineated a unique population of circulating monocytes with both M2 marker and collagen expression in chronic HCV-infected patients that correlated with the presence of liver fibrosis.
Materials and Methods
Biological Materials and Reagents
Huh7.5 cells were kindly provided by Dr Charles Rice (Rockefeller University, New York, NY) and were maintained in low-glucose Dulbecco’s modified Eagle medium containing 10% fetal bovine serum (Hyclone, Logan, UT), 10 μg/mL ciprofloxacin, and supplemented with nonessential amino acids (NEAA) (Gibco, Grand Island, NY). The Japanese fulminant hepatitis-1 (JFH-1) (HCV) construct was provided by Dr Takaji Wakita (National Institute of Infectious Diseases, Tokyo, Japan). In vitro synthesized JFH-1 RNA constructs were transfected and JFH-1 virus stock were produced as previously described.30 HCV viral supernatants were collected from Huh7.5 cells highly infected with JFH-1 cell culture–derived HCV particles and determined by flow cytometry.30 The supernatant was passed through 0.22-μm filters and was concentrated 20–30 times using an Amicon Ultra-15 100K centrifugal filter unit, and virus stock was preserved at -80°C (Millipore, Billerica, MA).30 Uninfected Huh7.5 cell supernatant was collected, concentrated, and was used as controls. Human liver biopsy specimens evaluated from 12 patients with chronic HCV (Supplementary Table 1) and 11 controls were received as anonymized samples by the National Institutes of Health–Funded Liver Tissue Procurement and Cell Distribution System. Human peripheral blood was collected from healthy donors and HCV-infected patients (Supplementary Table 2) with approval from the Institutional Review Board for the Protection of Human Subjects in Research at the University of Massachusetts Medical School with written informed consent. All authors had access to the study and reviewed and approved the final manuscript.
Dulbecco’s modified Eagle medium and RPMI 1640 cell culture media, antibiotics, and NEAA were purchased from Gibco. Recombinant TGF-β was purchased from Peprotech (Rocky Hill, NJ). Human (latency associated peptide) TGF-β1 antibody and mouse IgG1 isotype control were purchased from R&D Systems (Minneapolis, MN). Human antibodies (CD16 Allophycocyanin [APC], CD14 fluorescein isothiocyanate [FITC], CD40 FITC, and CD86 Phycoerythrin [PE]) were purchased from eBioscience (San Diego, CA), and CD14 APC, CD14 PE, CD163 PE, CD11c APC, CD68 PE, CD206 APC, DC-SIGN-FITC, CD45 PE, CD14 PE-Cy7, and isotype control antibodies were purchased from BD Pharmingen (Franklin Lakes, NJ). α-Smooth muscle actin (α-SMA) antibody, CD68 antibody, CD206 antibody, and FITC-conjugated donkey anti-rabbit IgG were purchased from Abcam (Cambridge, MA).
Human Monocyte Isolation and Co-culture Experiments
Human peripheral blood was collected from healthy donors, monocytes were isolated to more than 95% purity from peripheral blood mononuclear cells (PBMCs) by using CD14 microbeads, an MS Column, and a MiniMACS separator (Miltenyi Biotec, Inc, San Diego, CA), and were confirmed by flow cytometry (Supplementary Figure 1). Monocytes were cultured in RPMI 1640 medium (Gibco), supplemented with 10% fetal bovine serum, 100 U/mL penicillin, 100 mg/mL streptomycin, and NEAA as described previously.15 Informed consent was obtained and approved by the Institutional Review Board for the Protection of Human Subjects in Research at the University of Massachusetts Medical School. Huh7.5 or Huh7.5/JFH-1 cells (48 hours after infection) were plated on 12-well plates (2.5 × 105 cells/well) and co-cultured with monocytes (5 × 105 cells) in a 37°C, 5% CO2 incubator for 7 days. Monocytes were treated with HCV concentrate and Huh7.5 concentrate for 7 days. Supernatants, cells, and RNA were collected from all these experiments at the indicated time points and surface markers and cytokine expression were studied. To study stellate cell activation, the HSC cell line LX2 (2 × 105 cells/well) was cultured in the presence of supernatant obtained from 7-day (monocyte-Huh7.5/JFH-1) co-culture experiments and the profibrogenic gene and protein expressions were evaluated.
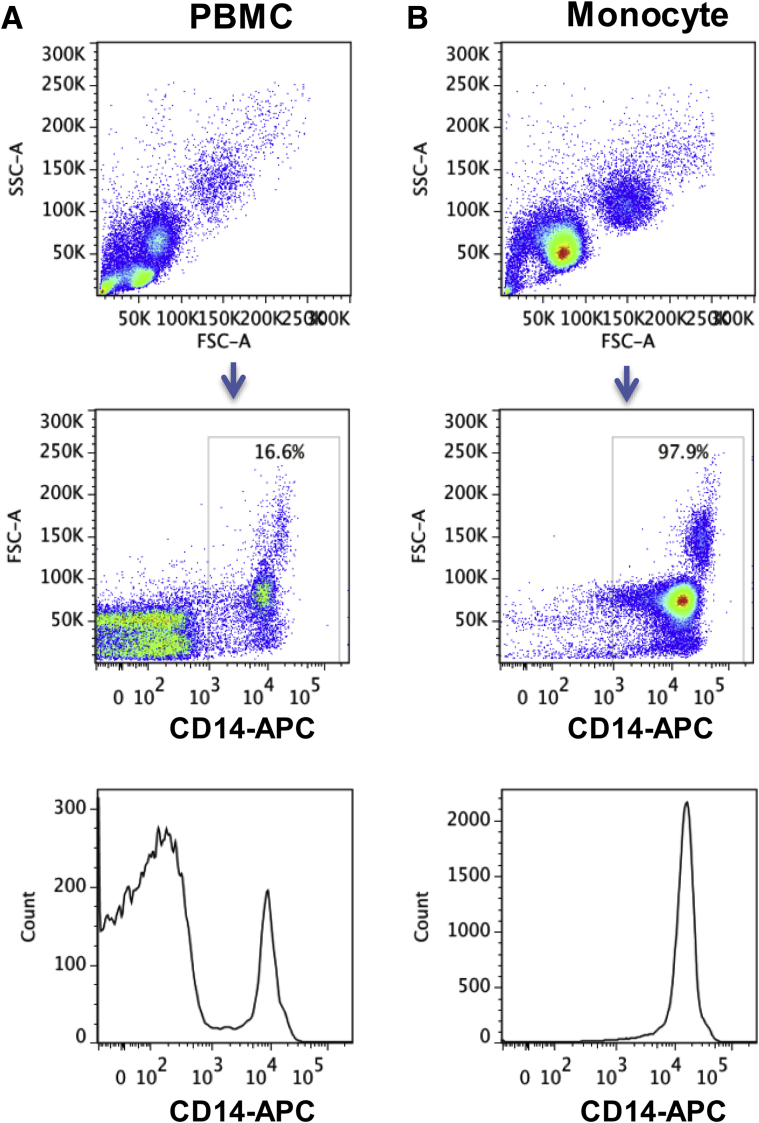
Supplementary Figure 1.
Purification of CD14+ monocytes from human PBMCs. Monocytes were isolated from human PBMCs using CD14 microbeads, an MS Column, and a MiniMACS separator. (A) PBMCs and (B) monocytes after isolation were stained with CD14-APC antibody. Representative dot plots and histograms are shown.
Enzyme-Linked Immunosorbent Assay
Levels of TNFα and IL1β were quantified in cell culture supernatant using commercially available enzyme-linked immunosorbent assay kits from BD Biosciences (Franklin Lakes, NJ). Enzyme-linked immunosorbent assay kits from eBioscience were used to measure IL10 and TGF-β in the cell culture supernatant.
RNA Extraction and Quantification
RNA was extracted using the RNeasy Mini kit (Qiagen, Valencia, CA) and 2-step real-time polymerase chain reaction as described previously30 using primers described in Supplementary Table 3. The level of the target gene expression was measured by the ΔΔCt method using the ratio of the fold-change in target gene expression vs the fold-change in reference gene expression (glyceraldehyde-3-phosphate dehydrogenase or 18S ribosomal RNA).
Flow Cytometry
To analyze cell surface marker expression, monocytes were immunostained for 30 minutes at 4°C by using appropriate antibody or isotype control as described previously.30 Monocytes were identified on the basis of CD14 expression, and the expression of other cell surface markers was analyzed on CD14+ cells. For intracellular detection of α-SMA, LX2 cells were fixed with 1% paraformaldehyde, permeabilized with 1% Tween 20, and stained with α-SMA antibody.31 Data acquisition was performed on a BD-LSR II (BD Biosciences, San Diego, CA), and the data were analyzed with FlowJo software (Tree Star, Ashland, OR). We determined the mean fluorescence intensity (MFI) for the gated positive population for each of the respective monocyte/macrophage markers.
Amnis Flowsight (EMD Millipore, Billerica, MA), which combines flow cytometry with real-time confocal imaging, was used for studying the expression of collagen in monocytes. Briefly, CD14 monocytes were isolated from both healthy controls and HCV-infected patients' peripheral blood. Approximately 105 cells were suspended in buffer containing Fc receptor-blocking agent (Miltenyi Biotec, Inc). Cells then were stained with 4′,6-diamidino-2-phenylindole, anti-CD45 PE, anti-CD206 APC, and anti-CD14 PE-Cy7, and then washed, fixed, and permeabilized with the CytoFix/cytoperm kit (BD Pharmingen). Cells then were incubated with rat-anti-human pro-Col-Iα or isotype control, washed, and finally stained with FITC-conjugated secondary antibody. The samples were acquired on the Amnis Flowsight, and analysis of the data was performed with IDEAS software (EMD Millipore, Seattle, WA).
Western Blot Analysis
Western Blot analysis was performed for CD68, CD206, and β-actin as previously described.32
Statistical Analysis
All values are expressed as means ± SEM obtained from 3 or more independent experiments. The Student t test and 1-way or 2-way analysis of variance test was used to compare means of multiple groups. A P value less than .05 was considered statistically significant. GraphPad Prism software (La Jolla, CA) was used for statistical analysis.
Results
HCV-Infected Hepatoma Cells Induce Differentiation of Monocytes Into Polarized Macrophages
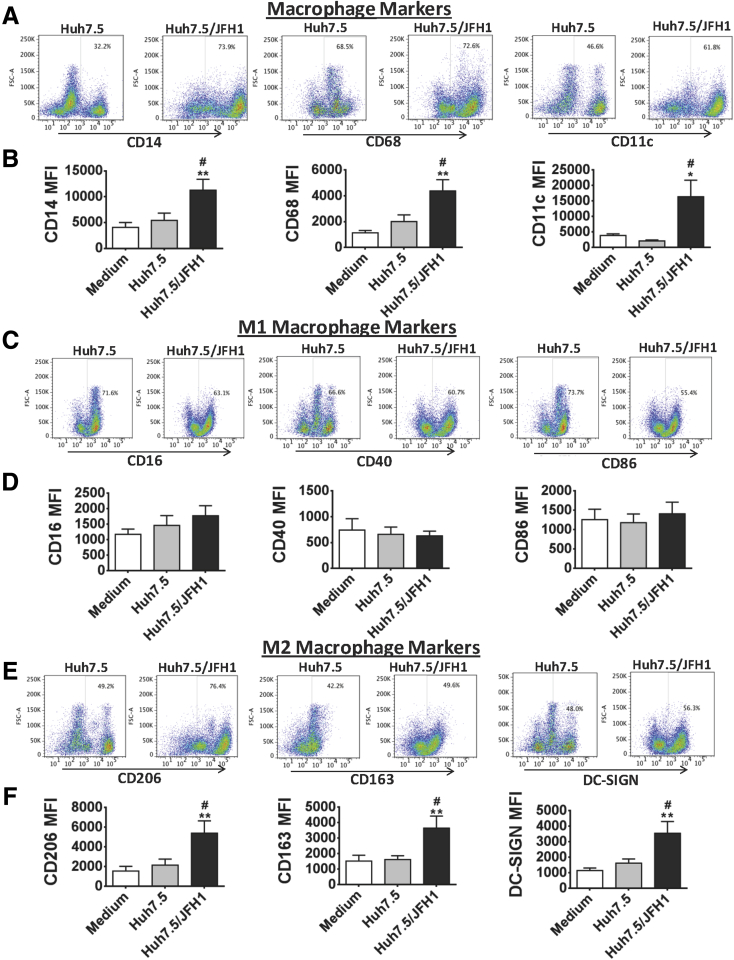
Monocytes play important roles in inflammatory and innate immune responses during HCV infection.33, 34 Monocytes from HCV-infected patients express TNFα on exposure to HCV proteins and show hyper-responsiveness to Toll-like receptor 2 and 4 ligands.15, 35 Monocytes are highly plastic and home to tissues where they differentiate to macrophages in response to the local environment. To test the hypothesis that HCV infection can lead to monocyte differentiation into macrophages, we performed in vitro co-cultures of HCV-naive healthy monocytes with HCV-infected Huh7.5/JFH-1 cells. We observed that exposure to Huh7.5/JFH-1 cells for 7 days induced monocyte differentiation into MΦs characterized by increased CD14, CD68, and CD11c expression compared with cells co-cultured with control Huh7.5 cells (Figure 1A and Supplementary Figure 2). The MFI of CD14, CD68, and CD11c also was increased in HCV MΦ compared with controls, indicating the MΦ phenotype (Figure 1B).
Figure 1.
Long-term co-culture of healthy human monocytes with HCV-infected hepatoma cells leads to monocyte differentiation into macrophages characterized with increased expression of M2 markers. Huh7.5 cells or Huh7.5/JFH-1 cells were co-cultured with monocytes obtained from healthy donors for 7 days. The cells were harvested and the phenotypic characteristics of monocytes after 7 days of co-culture were evaluated by flow cytometry. The dot plots show the expression of (A) MΦ markers (CD14, CD68, and CD11c), (C) M1 MΦ markers (CD16, CD40, and CD86), and (E) M2 MΦ markers (CD206, CD163, and DC-SIGN). The data are representative of 8–10 independent experiments. The expression levels of the MΦ markers in terms of MFI are shown for different co-culture conditions. The bar graphs represent the levels of (B) CD14, CD68, and CD11c, (D) M1 markers CD16, CD40, and CD86, and (F) M2 markers CD206, CD163, and DC-SIGN. Results are expressed as means ± SEM (N = 8–10). (B, D, and F) *P ≤ .05 and **P ≤ .01 compared with medium control. #P ≤ .05 compared with Huh7.5 co-culture. All P values were determined by 1-way analysis of variance.
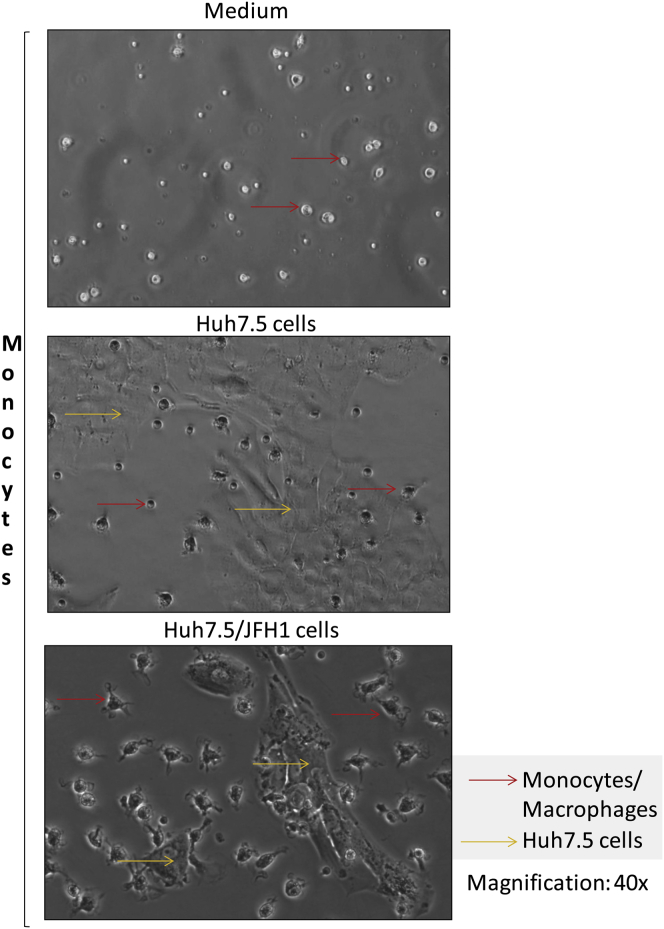
Supplementary Figure 2.
Monocytes differentiate into macrophages in the presence of Huh7.5/JFH-1 cells. Monocytes were isolated from healthy donors and were co-cultured with Huh7.5 cells with or without JFH-1 infection. After 7 days of culture, photographs of the cells were taken with a Nikon DS-QiMc (Nikon Instruments Inc, Melville, NY) camera attached to a Nikon Eclipse TS100 microscope at 40× magnification. The experiments were repeated 8–10 times and representative images of the co-culture conditions have been shown.
Next, we sought to identify the phenotype of the HCV MΦ. MΦs generated from monocytes in the presence of granulocyte-macrophage colony–stimulating factor or a combination of interferon-γ plus lipopolysaccharide were positive controls for M1 MΦs (classically activated) that had significantly increased expression of CD16, CD40, and CD86 (Supplementary Figure 3A and B). Macrophage colony–stimulating factor– or IL4-induced M2 MΦs (alternatively activated) had high CD206, CD163, and DC-SIGN expressions36, 37 (Supplementary Figure 3A and C). However, we also observed CD40 and DC-SIGN expression in both M1- and M2-positive controls, indicating that some of the markers are expressed under both polarizing conditions. HCV MΦ showed increased expression of M2 markers (CD206, CD163, and DC-SIGN) and no significant change in the expression of M1 markers (CD16, CD40, and CD86) compared with monocytes co-cultured with Huh7.5 cells (Figure 1C–F). These results suggest that HCV-infected hepatoma cells promote monocyte differentiation into MΦ with predominant M2 MΦ marker expression.
Supplementary Figure 3.
Expression of M1 and M2 markers on monocytes differentiated in the presence of various M1 and M2 polarizing agents. Monocytes were cultured in the presence of macrophage colony–stimulating factor (M-CSF) (50 ng/mL) or granulocyte-macrophage colony–stimulating factor (GM-CSF) (50 ng/mL) for 7 days or were cultured in the presence of M-CSF for 6 days and then treated with interferon (IFN)γ (20 ng/mL) + lipopolysaccharide (LPS) (100 ng/mL) or IL4 (20 ng/mL) for 18 hours to generate M1 and M2 MΦs, respectively. The cells were harvested and the phenotypic characteristics were evaluated by flow cytometry. (A–C) The expression levels of the MΦ markers in terms of MFI are shown for different co-culture conditions. The bar graphs represent the levels of (A) CD14, CD68, and CD11c, (B) M1 markers (CD16, CD40, and CD86), and (C) M2 markers (CD206, CD163, and DC-SIGN). Results are expressed as means ± SEM, n = 8–10. (A–C) *P ≤ .05, **P ≤ .01, and ***P ≤ .001 compared with medium control. All P values were determined by 1-way analysis of variance.
Monocytes Co-cultured With HCV-Infected Hepatoma Cells Results in Production of Both M1 and M2 Polarizing Cytokines
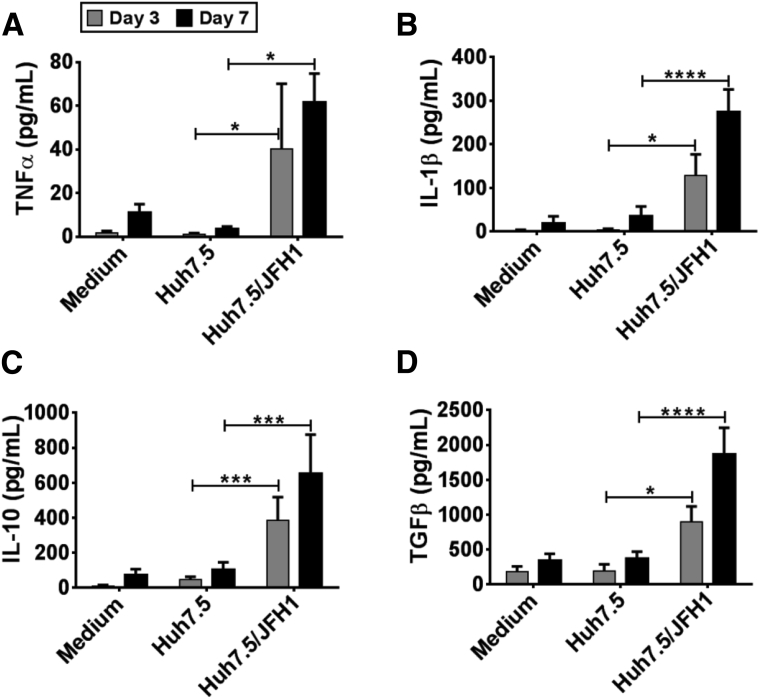
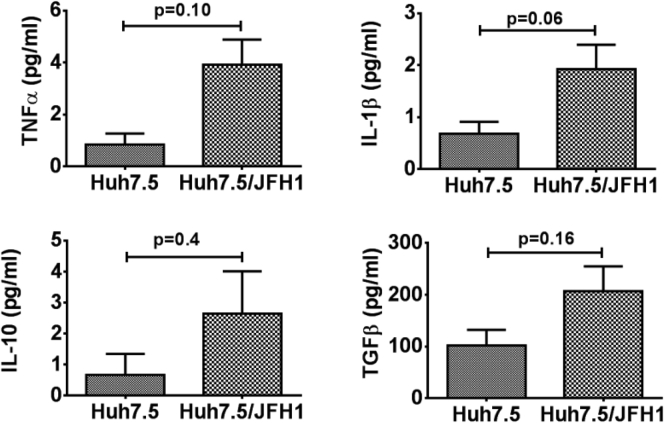
Next, we evaluated the cytokine secretion pattern of monocytes co-cultured with HCV-infected hepatoma cells. We observed that monocytes co-cultured with Huh7.5/JFH-1 cells secreted TNFα and IL1β by day 3, which was sustained even on day 7 (Figure 2A and B). However, monocyte secretion of the M2-polarizing cytokines, IL10 and TGF-β, also significantly increased in the presence of Huh7.5/JFH-1 cells over time, peaking on day 7 (Figure 2C and D). Huh7.5 cells alone secreted negligible levels of these cytokines regardless of HCV infection (Supplementary Figure 4). These results showed that monocytes co-cultured in the presence of Huh7.5/JFH-1 cells secreted both proinflammatory and anti-inflammatory cytokines (Figure 2).
Figure 2.
HCV-induced M2-like macrophages secrete proinflammatory and anti-inflammatory cytokines. Monocytes obtained from healthy donors were co-cultured with or without Huh7.5 or Huh7.5/JFH-1 cells for 7 days. Cell culture supernatant was collected on days 3 and 7 of the experiment and enzyme-linked immunosorbent assay was performed for (A) TNFα, (B) IL1β, (C) IL10, and (D) TGF-β. Results are expressed as means ± SEM (n = 4–7; *P ≤ .05, ***P ≤ .001, and ****P ≤ .0001). All P values were determined by 2-way analysis of variance.
Supplementary Figure 4.
Cytokine secretion by HCV-infected Huh7.5 cells was comparable with uninfected Huh7.5 cells. Huh7.5 cells were either infected with JFH-1 or left uninfected. Culture supernatant was collected after 7 days and TNFα, IL1β, IL10, and TGF-β levels were determined by enzyme-linked immunosorbent assay. The data are represented as means ± SEM, n = 4. The P values were determined by an unpaired t test.
Despite the increased expression of M1-polarizing cytokines, TNFα and IL1β, we observed an increase in the M2 MΦ markers on the HCV MΦ (Figure 1). This led us to assess whether proinflammatory cytokines induced in the early phase of monocyte-Huh7.5/JFH-1 co-culture could contribute to the anti-inflammatory response characterized by TGF-β production. IL1β regulates TGF-β signaling in human kidney epithelial cells where prolonged IL1 stimulation (or chronic inflammation) leads to enhanced TGF-β signaling.38 We found that addition of various doses of IL1–receptor antagonist (IL1RA), anakinra, to co-cultures of monocytes with HCV-infected hepatoma cells attenuated the HCV-induced M2-like MΦ phenotype (Figure 3). Secretion of IL1β was reduced in a dose-dependent manner in monocytes cultured with Huh7.5/JFH-1 cells in the presence of IL1RA (Figure 3A). That was expected given the autoregulatory feedback loop of IL1 production.39 Importantly, IL1RA treatment inhibited secretion of TGF-β, but not IL10, in the monocyte-Huh7.5/JFH-1 co-cultures (Figure 3B and C). Furthermore, when we evaluated the expression of M2 MΦ markers on IL1RA-treated co-cultures of monocytes with Huh7.5/JFH-1, we observed an attenuation of CD206, CD163, and DC-SIGN expression (Figure 3D–F). The levels of DC-SIGN were decreased to a greater extent compared with CD206 and CD163 expression, which suggests that inhibiting IL1 does not completely abolish M2–MΦ polarization. Thus, these results showed that the induction of IL1β in the presence of HCV-infected hepatoma cells promoted monocyte differentiation into TGF-β producing MΦ.
Figure 3.
IL1-receptor antagonist, anakinra, inhibits TGF-β secretion in monocytes co-cultured with HCV-infected hepatoma cells. Monocytes were co-cultured with Huh7.5 or Huh7.5/JFH-1 cells in the presence of anakinra (1–100 μg/mL) for 7 days. Culture supernatants were collected and assayed for (A) IL1β, (B) TGF-β, and (C) IL10. Flow cytometry analysis of the M2 markers (D) CD206, (E) CD163, and (F) DC-SIGN also were performed on the monocytes from the co-cultures. Results are represented as means ± SEM from 3–7 independent experiments. *P ≤ .05, **P ≤ .01, ***P ≤ .001, and ****P ≤ .0001. All P values were determined by 2-way analysis of variance.
TGF-β Released by HCV MΦ Leads to Hepatic Stellate Cell Activation
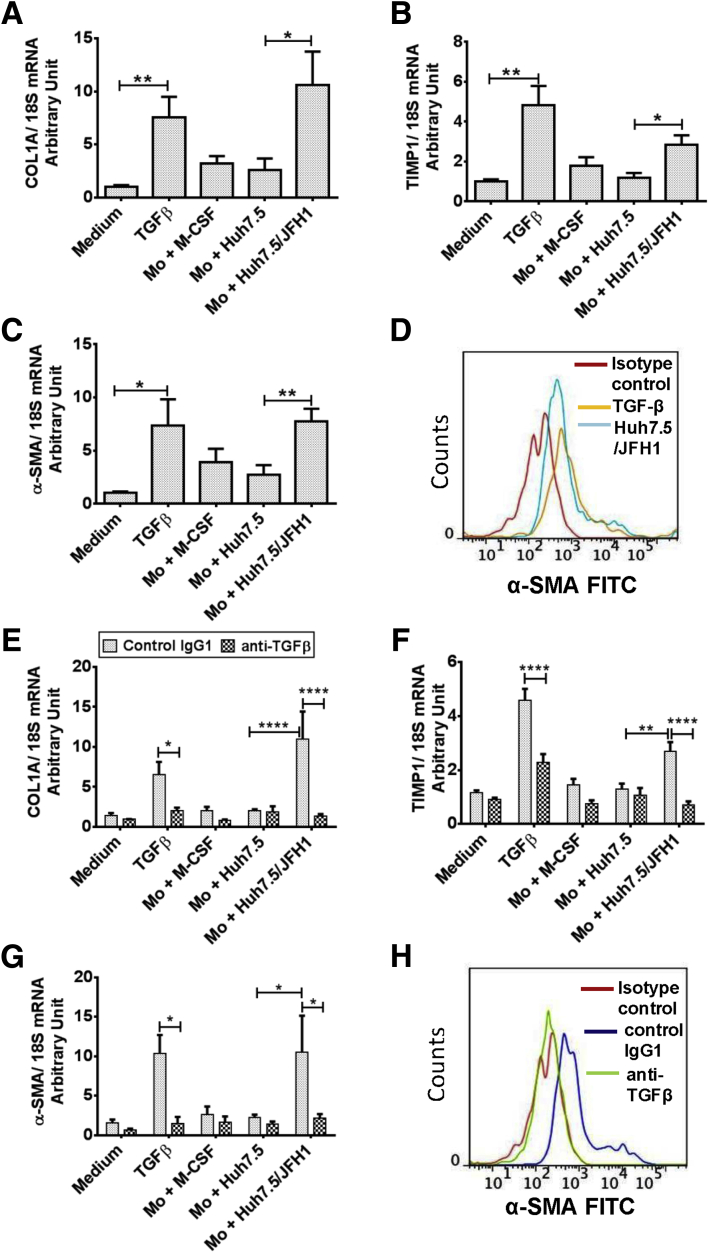
To assess the functional role of these HCV MΦs, we asked whether they could induce HSC activation. LX2, a human HSC line, was treated with the supernatants collected from the 7-day monocyte-Huh7.5/JFH-1 co-culture experiments. We studied the expression of collagen (COL)1A, tissue inhibitor of metalloproteinase (TIMP)1, and α-SMA messenger RNA (mRNA) and observed significant increases at day 6 (Figure 4A–C). TGF-β, as a positive control, increased mRNA levels of COL1A, TIMP1, and α-SMA in LX2 cells. Supernatants of Huh7.5/JFH-1 cells (without monocytes) resulted in a minimal increase in COL1A, TIMP1, and α-SMA mRNA expression in LX2 cells, suggesting that HCV-infected hepatoma cells do not directly activate LX2 cells. We also measured the levels of α-SMA protein in HSCs treated with supernatants from the monocyte Huh7.5/JFH-1 and observed increased α-SMA, which was comparable with TGF-β–treated LX2 cells (Figure 4D).
Figure 4.
TGF-β secreted from HCV-educated monocytes mediates profibrogenic effects on HSCs. LX2 cells were cultured for 6 days in the presence of supernatant obtained from a 7-day monocyte (Mo)-Huh7.5 or Huh7.5/JFH-1 co-culture experiment. LX2 cells also were treated with supernatant from Huh7.5 or Huh7.5/JFH-1 cells. LX2 cells also were treated with or without TGF-β (2.5 ng/mL). Total RNA was isolated, complementary DNA was transcribed, and real-time polymerase chain reaction was performed. Relative mRNA expression of (A) COL-1A, (B) TIMP1, and (C) α-SMA was determined. The data are represented as means ± SEM, N = 5–7, where *P ≤ .05 and **P ≤ .01 (unpaired t test). (D) LX2 cells were cultured for 6 days as described earlier and intracellular α-SMA levels were determined by flow cytometry. The representative histogram depicts the expression of α-SMA in LX2 cells treated with Mo-Huh7.5/JFH-1 co-culture supernatants and TGF-β–treated LX2 cells. LX2 cells were cultured in the presence of control IgG1 or neutralizing anti–TGF-β antibody under similar conditions as described earlier for 6 days and the relative mRNA expression of (E) COL-1A, (F) TIMP1, and (G) α-SMA was measured. (H) α-SMA protein levels also were determined by flow cytometry and a representative histogram is shown for the LX2 cells treated with Mo-Huh7.5/JFH-1 supernatants in the presence of anti–TGF-β neutralizing antibodies and control IgG1. (E–G) The data are representative of means ± SEM, N = 4, where *P ≤ .05, **P ≤ .01, and ****P ≤ .0001 (2-way analysis of variance). (D and H) Data are representative of 2 independent experiments.
Because TGF-β levels were increased in the 7-day co-cultures of monocyte Huh7.5/JFH-1, we studied the effect of TGF-β neutralization on LX2 activation. We observed that the profibrotic effect of the supernatants derived from the monocyte-Huh7.5/JFH-1 co-cultures was prevented by TGF-β neutralization (Figure 4E–G). LX2 cells pretreated with anti–TGF-β–neutralizing antibodies and cultured in the presence of monocyte-Huh7.5/JFH-1 co-culture supernatant had decreased α-SMA protein levels compared with control IgG1-treated LX2 cells (Figure 4H). These results showed that TGF-β released from HCV MΦs leads to HSC activation.
Monocyte to Macrophage Differentiation Is Induced by Cell-Free HCV and Does Not Require Cell–Cell Contact
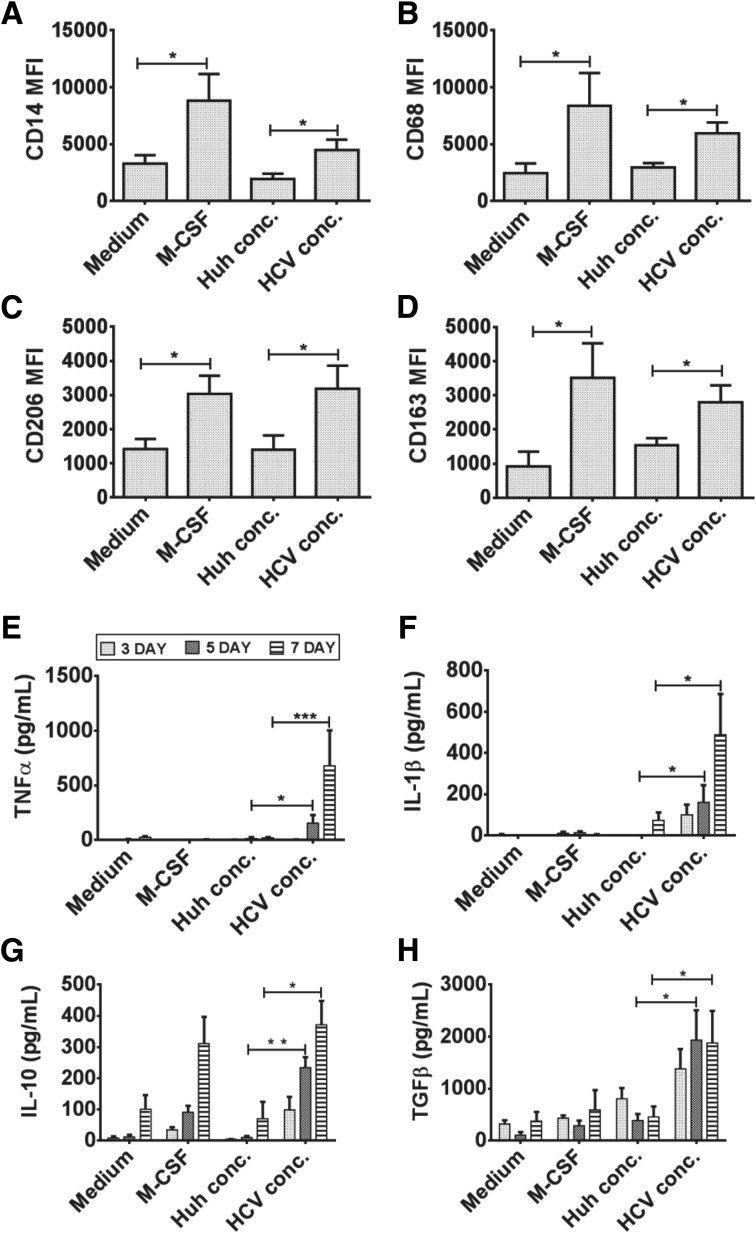
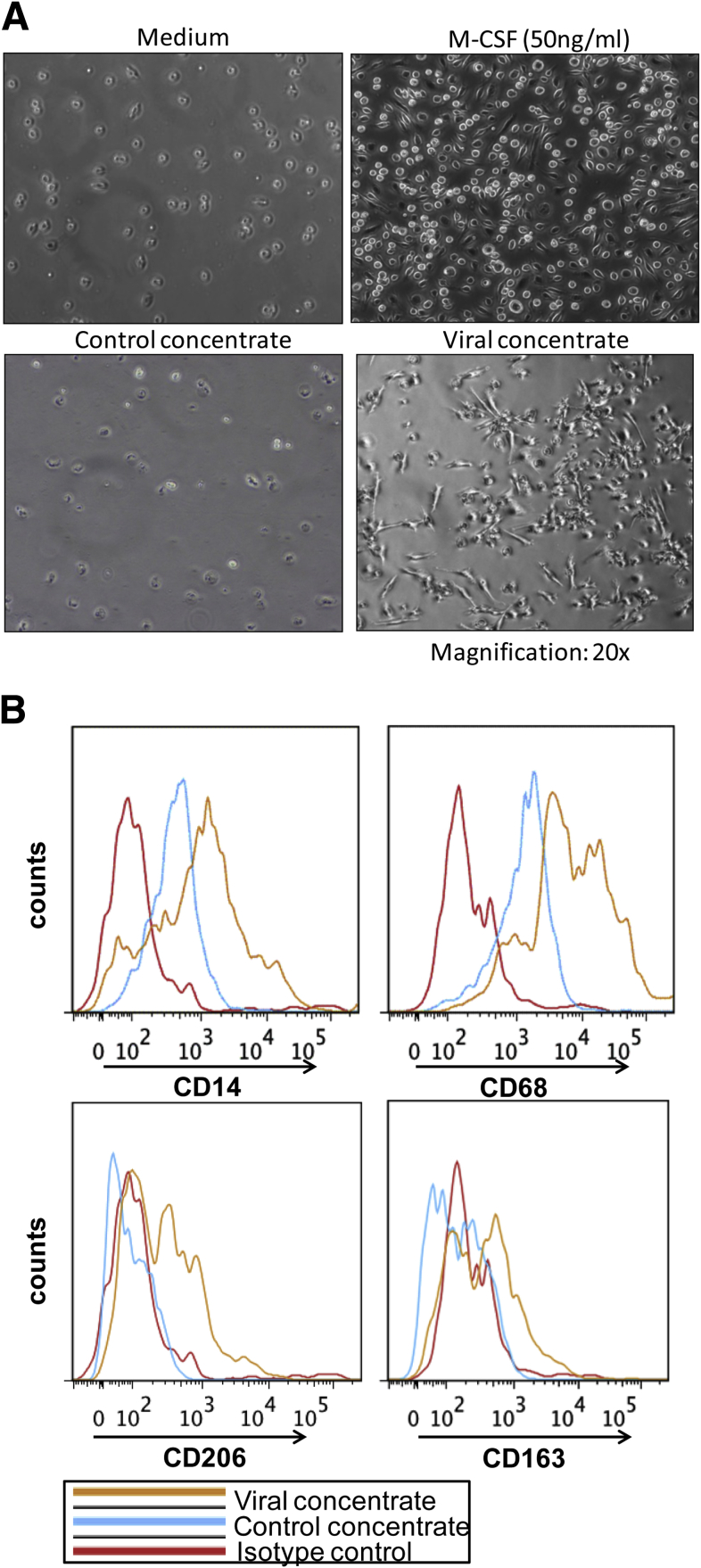
Next, we performed experiments to evaluate whether cell-free HCV can have similar effects on monocyte activation as observed in the presence of HCV-infected hepatoma cells. Because JFH-1–infected Huh7.5 cells release HCV into the culture supernatant we tested effects of cell-free HCV on monocytes.40 Similar to HCV-infected hepatoma cells, the addition of isolated HCV directly to monocytes increased CD14, CD68, CD206, and CD163 expression and changed monocyte morphology over a 7-day period (Figure 5A–D and Supplementary Figure 5A and B). TNFα and IL1β production increased in monocytes cultured with HCV, and a time-dependent increase in IL10 and TGF-β levels suggested that cell-free HCV can induce the monocyte to MΦ differentiation (Figure 5E–H). Collectively, these results showed that without cell–cell contact, cell-free HCV robustly and directly influenced monocyte differentiation into MΦs that expressed M2 surface markers and a mixed M1/M2 cytokine secretion pattern.
Figure 5.
Cell-free HCV induces monocyte differentiation. Monocytes were cultured in the presence of macrophage colony–stimulating factor (M-CSF), Huh7.5 culture supernatant concentrate, or HCV concentrate for 7 days and immunostained for (A) CD14, (B) CD68, (C) CD206, and (D) CD163. (E) TNFα, (F) IL1β, (G) IL10, and (H) TGF-β levels were measured on days 3, 5, and 7 by enzyme-linked immunosorbent assay from culture supernatant. The data are represented as means ± SEM, N = 4–6, *P ≤ .05, **P ≤ .01, ***P ≤ .001.
Supplementary Figure 5.
HCV induces monocyte differentiation into macrophages. Monocytes were isolated from healthy donors and cultured alone or with Huh or HCV concentrate for 7 days. (A) Photographs of the cells were taken with a Nikon DS-QiMc camera attached to a Nikon Eclipse TS100 microscope at a magnification of 20×. A representative image is shown. (B) Representative histogram plots of CD14, CD68, CD206, and CD163 on monocytes alone and cells treated with Huh concentrate or viral concentrate.
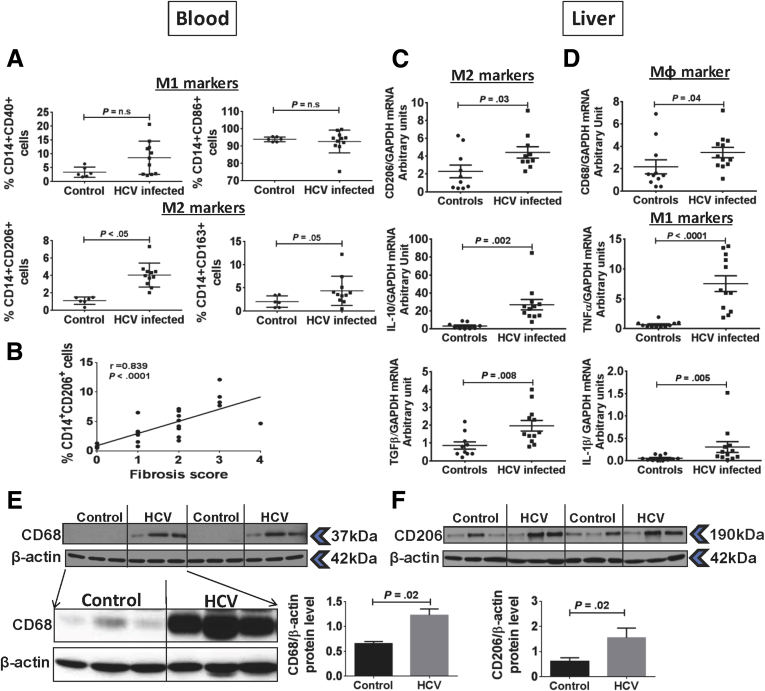
M2 MΦ Markers Are Expressed on Circulating Monocytes in the Blood and in the Liver in Chronic HCV-Infected Patients
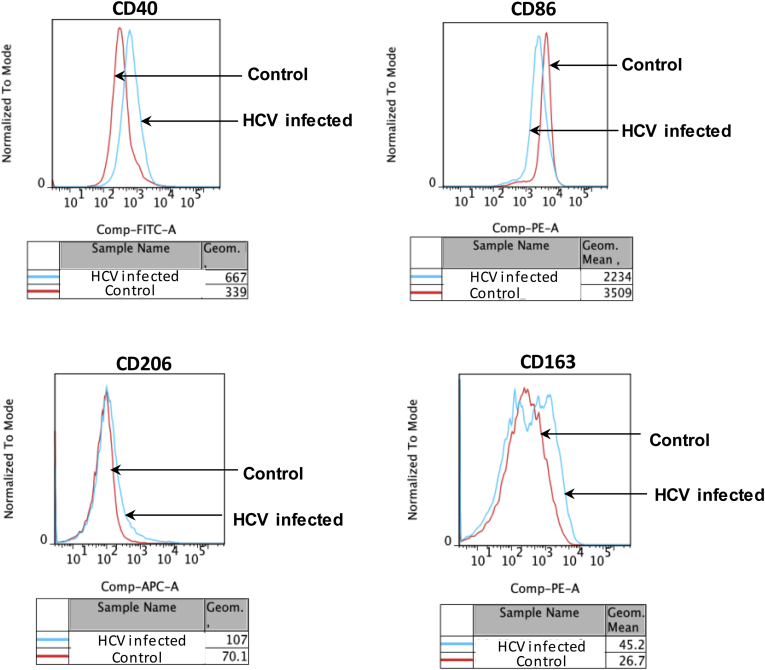
Our in vitro results showed the role of HCV in mediating monocyte differentiation to MΦs. During chronic HCV infection, monocytes continuously are exposed to HCV and viral RNA/protein in circulation.35 Thus, we wanted to evaluate the phenotype of chronic HCV patients’ monocytes based on expression of M1 (CD40, CD86) and M2 (CD206, CD163) markers. We found a significantly increased percentage of M2 marker CD206 expression in HCV-infected patients compared with controls (Figure 6A). Expression of M1 marker CD40 and M2 marker CD163 were not increased significantly as compared with controls (Figure 6A). There was no change in the percentage of CD14+CD86+ cells in the patient vs control group (Figure 6A). The MFI of CD40, CD206, and CD163 increased, but that of CD86 decreased in the HCV-infected group as compared with controls (Supplementary Figure 6). Next, we performed a correlation analysis between the percentage of CD14+CD206+ circulating monocytes with the extent of liver fibrosis (Metavir score) in patients with HCV infection. As shown in Figure 6B, Spearman analysis showed a positive correlation of CD14+CD206+ circulating monocyte percentage with the fibrosis score (r = 0.839; P < .0001).
Figure 6.
M2 macrophage markers are up-regulated on circulating monocytes and liver during chronic HCV infection. (A) CD14+ monocytes from controls and HCV-infected patients were immunophenotyped by flow cytometry and the percentage of cells expressing CD40, CD86, CD206, and CD163 markers are shown. The data are represented as means ± SEM (controls, N = 7; HCV, N = 11). (B) Correlation analysis of the percentage of CD14+CD206+ circulating monocytes and the fibrosis stage is shown (HCV, N = 26). The statistical analysis was performed using Spearman correlation. (C) mRNA expression of CD206, IL10, and TGF-β, and (D) CD68, TNFα, and IL1β were determined in liver biopsy specimens from controls or HCV-infected patients by real-time polymerase chain reaction. The data are represented as means ± SEM (controls, N = 10–11; HCV, N = 12). The P value was determined by unpaired t test. (E and F) Protein level expression of CD68 and CD206 in the liver of control and HCV-infected patients is shown. Western blot showing the expression of CD68 with digital imaging at higher exposure for liver samples (controls, N = 3; HCV, N = 3). β-actin was used as the loading control. The bar graph shows the fold-change in CD68 or CD206 levels as compared with β-actin (controls, N = 6; HCV, N = 6). The P value was determined by an unpaired t test. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Supplementary Figure 6.
M2-macrophage markers are up-regulated on circulating monocytes. CD14+ monocytes from healthy controls and HCV-infected patients were immunophenotyped by flow cytometry for CD40, CD86, CD206, and CD163. The representative histogram plots and the MFI are shown.
Next, we evaluated the expression of MΦ markers in the livers of HCV-infected patients. Previous studies have shown increased number and activation of MΦs in the liver in chronic HCV infection.15, 35 We found increased levels of M2 MΦ markers; CD206, IL10, and TGF-β mRNA in the HCV-infected liver were observed as compared with controls (Figure 6C). We also observed that the MΦ marker, CD68, was up-regulated along with increased mRNA expression of M1 MΦ markers, TNFα and IL1β, compared with control livers (Figure 6D). There was a significant increase in the protein expression of MΦ markers, CD68 and CD206, in the liver of HCV-infected patients as compared with controls (Figure 6E and F). Together, these results showed that the frequency and expression of M2 MΦ markers was increased in circulating monocytes and the expression of these markers also was increased in the liver of HCV-infected patients.
HCV Infection Leads to Generation of Collagen-Expressing Monocytes In Vivo and In Vitro
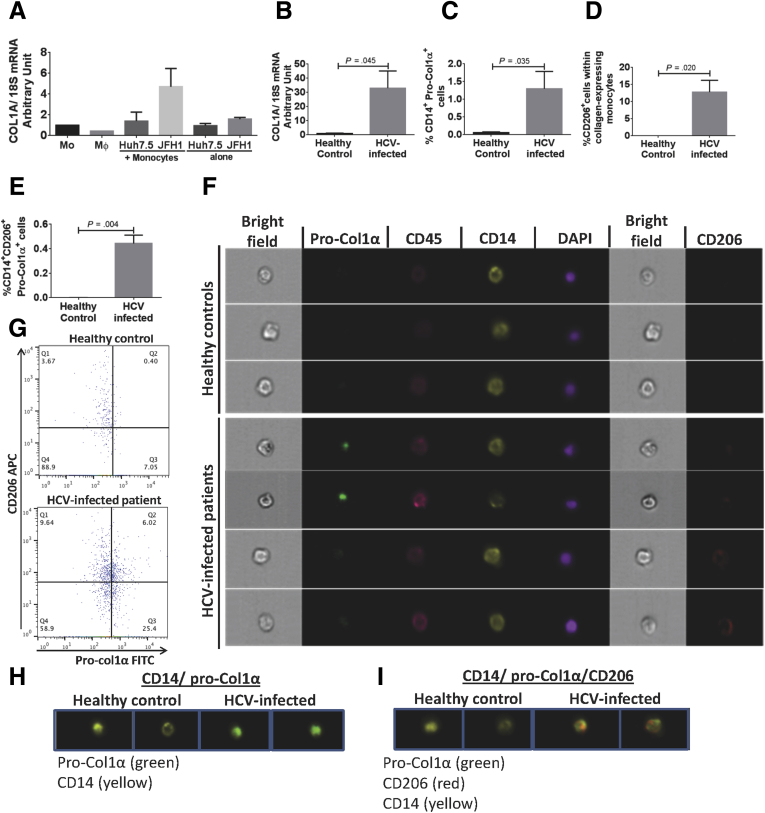
The presence of monocytes expressing M2-MΦ markers prompted us to further investigate the characteristics of these circulating monocytes. Previous studies indicated that CD14+ peripheral blood monocytes can differentiate into cells that express collagen, which are attracted to the sites of tissue injury and mediate tissue repair and fibrosis.41, 42 Here, we found increased mRNA levels of collagen (COL1A) in the monocytes co-cultured with Huh7.5/JFH-1 cells (Figure 7A). To determine the in vivo relevance of this finding, we next tested collagen expression in the PBMCs of HCV-infected patients. There was a significant increase in the mRNA levels of COL1A in PBMCs in HCV-infected patients as compared with healthy controls (Figure 7B).
Figure 7.
Collagen-expressing monocytes showing M2 phenotype identified in HCV-infected patients. (A) Isolated monocytes from healthy donors were co-cultured with Huh7.5 or Huh7.5/JFH-1 cells for 8 days. mRNA levels of COL-1A were measured by real-time polymerase chain reaction. The data are represented as means ± SEM, n = 3 experiments. (B) PBMCs were isolated from healthy donors (N = 2) and HCV-infected patients (N = 6), and mRNA levels of COL-1A were measured. (C–G) Isolated monocytes from healthy donors (N = 5) and HCV-infected patients (N = 10) were immunostained with antibodies against CD14, CD45, and CD206. Cells were fixed and permeabilized to stain with pro-col-1α antibody. Expression of these markers was studied by flow cytometry and Amnis Flowsight. (C) Frequency of CD14+ Pro-Col1α+ in controls and HCV-infected patients was determined. (D) Frequency of CD206-expressing CD14+ Pro-Col1α+ cells in controls and HCV-infected patients was measured. (E) Frequency of CD206+CD14+ Pro-Col1α+ cells in circulation. P value was determined by unpaired t test. (F–I) Confocal images of monocytes expressing CD14 (yellow), pro-Col1α (green), CD45 (pink), CD206 (red), and 4′,6-diamidino-2-phenylindole (DAPI) (purple) staining for nucleus is shown (original magnification, ×20) for 3 representative healthy donors and 4 representative HCV-infected patients. (G) Representative dot plots for the circulating monocytes expressing CD206 and proCol1-α is shown for a healthy control and HCV-infected patient. (H) Co-localization of CD14 (yellow) and pro-Col1α (green) (original magnification, ×20) is shown for healthy controls (N = 2) and HCV-infected patients (N = 2). (I) Co-localization of CD14 (yellow), CD206 (red), and pro-Col1α (green) (original magnification, ×20) for 2 representative healthy donors and HCV-infected subjects (N = 2) have been shown.
We found a significantly increased number of CD14+ monocytes expressing procollagen 1α in HCV-infected patients compared with controls (Figure 7C). Procollagen 1α is packaged in the Golgi bodies and undergoes post-translational modification before it is secreted from a cell.43 Because monocytes from the HCV-infected patients also showed an M2 phenotype we studied the co-expression of pro-Col1α and CD206 in CD14+ monocytes. We observed that 10%–15% of monocytes that expressed collagen also expressed CD206 in monocytes from HCV-infected patients, which was approximately 0.4% of the total circulating cells, although there was no collagen expression in monocytes from controls (Figure 7D and E). Finally, to visualize the presence of collagen in monocytes from HCV-infected patients we used Amnis image stream technology (Amnis Flowsight). We observed that circulating monocytes from HCV-infected patients expressed pro-Col1α but not from healthy controls (Figure 7G and H). We also found observed co-localization of pro-Col1α and CD206 on CD14+ monocytes from HCV-infected patients (Figure 7I). These results showed that during HCV infection, collagen-producing CD14+ monocytes are present in the circulation in vivo and may contribute to liver fibrogenesis.
Discussion
Innate immune responses play a critical role in the control and resolution of acute HCV infection and contribute to chronic inflammation and liver fibrosis.5, 44 The mechanisms by which HCV-infected hepatocytes affect circulating monocytes and liver MΦs and the effects of HCV on monocyte differentiation are only partially understood. Because of the lack of animal models of HCV infection, most of the studies are performed in vitro using the well-established HCV infection culture system.29, 40 By using co-cultures of human monocytes and HCV-infected hepatoma cells or cell-free HCV we showed that HCV induced differentiation of monocytes to MΦ with mixed M1/M2 cytokine production and M2-polarized surface marker expression. Our data indicated that M2-MΦ–derived TGF-β acted as a mediator of HSC activation and fibrosis. We also found increased frequency of circulating monocytes expressing M2 marker in the blood of chronic HCV-infected patients and identified CD14+CD206+collagen+ monocytes in the circulation that correlated positively with liver fibrosis in HCV-infected patients.
Peripheral blood monocytes constantly enter the liver where they interact with HCV-infected hepatocytes, cell-free virus, and replenish liver MΦs.35 Experimental animal models of liver diseases have shown that the disease pathogenesis is mediated partially by Kupffer cells, but largely is dependent on the recruitment of monocytes into the injured and inflamed liver.45 Thus, in this study we performed co-culture experiments to understand the effect of HCV-infected hepatoma cells on the phenotype and function of monocytes. Consistent with previous studies that showed activation of Kupffer cells, monocytes, and MΦs in chronic HCV infection, we found increased activation and production of cytokines by monocytes exposed to HCV.15, 46 Our studies showed that HCV-infected hepatoma cells triggered monocytes to differentiate into a MΦ phenotype (CD14high, CD68high, CD11chigh) with increased M2-marker (CD206+, CD163+, DC-SIGN+) expression.
In patients with viral hepatitis, increased numbers of Kupffer cells (CD14+CD68+) are found around the region of damage and fibrosis that has been associated with liver injury.45 A detailed study from liver tissues obtained from nonviral hepatitis patients showed that a specific monocyte population accumulated within the liver, which had high phagocytic activity and released both inflammatory and profibrogenic cytokines.14 Thus, it is likely that the chronic presence of virus in the circulation and in the liver during HCV infection may trigger accumulation of recruited monocytes and alter the phenotype of the resident MΦs in the liver. Persistent inflammation generally is accompanied by negative feedback mechanisms, and during chronic viral infections M2 MΦs showing potent anti-inflammatory, wound healing, and fibrotic capacity may accumulate in the liver.45, 47 Although M2 MΦ are essential to restore homeostasis in acute inflammation, they also produce growth factors and cytokines, including TGF-β1 and platelet-derived growth factors, which stimulate fibroblasts.48 Our results support a model in which chronic HCV presence leads to activation of profibrotic MΦs. We found that HCV-infected hepatoma cells triggered early induction of proinflammatory cytokines (TNFα and IL1β) whereas TGF-β and IL10 were predominant in the late cytokine response. Although production of anti-inflammatory cytokines (IL10, TGF-β) is a component of the natural evolution of late-phase inflammation, the results from our experiments using IL1RA, anakinra, indicate that the early induction of IL1β contributes to increased TGF-β production in HCV-induced MΦ. Further, anakinra inhibited production of TGF-β and not IL10, which in turn led to decreased expression of M2 markers on the monocytes co-cultured with Huh7.5/JFH-1. This suggested the direct role of TGF-β in inducing monocyte differentiation and/or macrophage polarization during HCV infection. This indicates an autoregulatory loop by which HCV skews innate immune response to its advantage. IL1β is a profibrotic mediator in liver and lung fibrosis whereas TGF-β signaling plays a pivotal role in cellular responses by promoting cell proliferation, differentiation, and apoptosis.23, 25, 43 HCV-infected patients express high levels of TGF-β in serum and liver, which correlates with the degree of fibrosis.23 Expression of HSC activation markers, COL1A, TIMP-1, and α-SMA, in LX2 cells was decreased with TGF-β inhibition in monocyte and Huh7.5/JFH-1 co-culture supernatants. Further experiments with primary HSCs could validate our finding with LX2 cells on the effects of monocyte polarization during HCV infection on HSC activation.
The effects of the HCV-infected hepatoma cells as well as cell-free virus on the monocyte phenotype and function were observed in an in vitro culture system, however, this may not entirely mimic the complex environment of the liver where the phenotype of the monocyte/MΦs are influenced by the presence of other immune and nonimmune cells. The limitation of our study was that it characterized functions of circulating monocytes that should be interpreted carefully regarding the function of monocytes in the HCV-infected liver. Nonetheless, our in vivo results underscore the importance of the in vitro findings because we show that during HCV infection there is a significant increase in the expression of CD14+ monocytes expressing the M2 marker CD206. This increase in the M2 marker expressing monocytes correlated positively with liver fibrosis in the HCV-infected patients. The mRNA and protein expression suggested the presence of M2 MΦs in HCV-infected livers. Thus, these in vivo results further underscore the role of HCV in influencing monocyte differentiation and polarization.
CD14+ monocytes are precursors not only for MΦs, but also for fibrocytes.41 Fibrocytes have features of both MΦs and fibroblasts and express markers of both hematopoietic cells (CD34, CD43, CD45, Lymphocyte-specific protein 1, major histocompatibility complex class II) and stromal cells (collagen I and III).41, 42, 49 In addition, a population of CD45+/CD14+/Col-I+ monocytes also has been described in the circulation of patients with systemic sclerosis.50 Although monocytes give rise to fibrocytes, the expression of CD14 on fibrocytes is controversial and thus, the term fibrocytes is used for the cells that are CD14-, whereas CD14+ cells are referred to as collagen-producing monocytes.50, 51 Increased numbers of fibrocytes are reported in diseases characterized by chronic inflammation; animal models also have implicated fibrocytes in the development of tissue fibrosis involving lungs, liver, heart, and kidney.42, 52, 53 Collagen-producing monocytes are detected in the circulation and the lungs of systemic sclerosis–interstitial lung disease patients and are important in the progression of lung fibrosis.51 Nunnari et al54 showed the correlation of circulating fibrocytes CD45+/CD34+/Col-I+ with liver fibrosis in chronic HCV patients. We detected collagen-producing monocytes in the blood of chronic HCV patients, but not in healthy subjects. We also showed that a significant percentage of collagen-producing monocytes from HCV-infected patients also expressed the M2 marker CD206. This suggested that, first, during HCV infection, some monocytes acquire a profibrotic characteristic and the frequency of profibrotic monocytes is increased in the circulation compared with normal controls. Second, in HCV-infected patients, the collagen-producing CD14+ monocytes comprised a fraction of the circulating M2 monocyte population of chronic HCV-infected patients, which could function as a biomarker. Our results also suggested that the presence of M2 markers in the liver and blood during chronic HCV infection positively correlated with liver fibrosis in HCV-infected patients. In the present study, we analyzed a small number of HCV-infected patients, thus it is important to screen for these markers in a larger cohort of patients at different stages of liver fibrosis to establish CD14+CD206+ cells as potential biomarkers of fibrosis.
In summary, our data show a new aspect of HCV and host interactions in which HCV-infected hepatocytes promote monocyte differentiation into MΦs with a mixed M1/M2 cytokine profile and M2 surface phenotype. We found that HCV-mediated monocyte differentiation leads to a MΦ phenotype that is profibrotic, with characteristics of M2 (alternatively activated) MΦs, leading to HSC activation. Furthermore, our results suggest that the circulating monocyte phenotype from HCV patients can act as a marker for liver fibrosis in chronic hepatitis C infection.
Acknowledgments
The authors thank Dr Takaji Wakita (National Institute of Infectious Diseases, Tokyo, Japan) and Dr Charles M. Rice (Rockefeller University, New York, NY) for kindly providing the infectious JFH-1 molecular clone and Huh7.5 cells; Dr Scott L. Friedman (Mount Sinai Hospital, New York, NY) for the LX2 cell line; Dr Melanie Trombly and Dr Shuye Zhang (Shanghai Public Health Clinical Center, Shanghai, China) for critical reading of the manuscript; and Donna Giansiracusa, Sharon Balcom, and Vo Trang from the GI research office and Dr Michal Ganz for assistance with the clinical samples. The authors greatly appreciate the participation of the patients and volunteers for this study.
Footnotes
Conflicts of interest The authors disclose no conflicts.
Funding Supported by National Institutes of Health grant R37 AA014372 (G.S.). Human liver samples were provided by the National Institutes of Health–Funded Liver Tissue Procurement and Cell Distribution System (N01-DK-7-0004/HHSN26700700004C).
Supplementary Material
Supplementary Table 1.
Clinical Characteristics of the HCV-Infected Patients
| Serial number | Genotype | Viral load, log IU/mL | Fibrosis stage |
|---|---|---|---|
| 1 | 1a | 5.13 | Grade 4/4 |
| 2 | NA | 4.01 | Grade 4/4 |
| 3 | NA | 6.95 | Grade 1/4 |
| 4 | NA | 5.71 | Grade 4/4 |
| 5 | 3 | 6.86 | Grade 4/4 |
| 6 | NA | 2.84 | Grade 4/4 |
| 7 | NA | NA | Grade 3/4 |
| 8 | NA | 5.50 | Grade 4/4 |
| 9 | NA | NA | Grade 4/4 |
| 10 | NA | 5.46 | Grade 4/4 |
| 11 | NA | 3.11 | Grade 4/4 |
| 12 | NA | 5.68 | NA |
NOTE. The HCV genotype, viral load, and the fibrosis stage are shown for the HCV-infected patients who had a liver biopsy. Metavir scoring was used for fibrosis scoring. The fibrosis was graded on a 5-point scale from 0 to 4. Fibrosis scores were as follows: F0, no fibrosis; F1, portal fibrosis without septa; F2, portal fibrosis with few septa; F3, numerous septa without cirrhosis; and F4, cirrhosis. NA, not available.
Supplementary Table 2.
Clinical Characteristics of the HCV-Infected Patients
| Serial number | Genotype | Viral load, log IU/mL | Fibrosis stage |
|---|---|---|---|
| 1 | 2b | 6.40 | Grade 0/4 |
| 2 | 1a | 6.61 | Grade 2/4 |
| 3 | 1a | 5.71 | Grade 1/4 |
| 4 | 1b | 5.72 | Grade 2/4 |
| 5 | 1a | 4.69 | Grade 0/4 |
| 6 | 1b | 5.79 | Grade 1/4 |
| 7 | 3a | 6.12 | Grade 3/4 |
| 8 | 1a | 6.71 | Grade 0/4 |
| 9 | 1a | 5.06 | Grade 0/4 |
| 10 | 1a | 6.07 | Grade 2/4 |
| 11 | 1a | 5.73 | Grade 1/4 |
| 12 | 1a | 6.51 | Grade 2/4 |
| 13 | 2b | 6.55 | Grade 2/4 |
| 14 | 1a | 7.92 | Grade 4/4 |
| 15 | 1a | 6.5 | Grade 1/4 |
| 16 | 3a | 6.12 | Grade 2/4 |
| 17 | 1a | 5.71 | Grade 1/4 |
| 18 | 3a | 5.93 | Grade 3/4 |
| 19 | 1a | 6.61 | Grade 2/4 |
| 20 | 1b | 4.6 | Grade 3/4 |
| 21 | 1a | 1.2 | Grade 2/4 |
| 22 | 1b | 6.03 | Grade 3/4 |
| 23 | 2b | 6.6 | Grade 0/4 |
| 24 | 3a | 5.93 | Grade 24 |
| 25 | NA | 2.56 | Grade 0/4 |
| 26 | 1a | 7 | Grade 1/4 |
NOTE. The HCV genotype, viral load, and the fibrosis stage are shown for the HCV-infected patients who provided blood samples for the study. Metavir scoring was used for fibrosis scoring. The fibrosis was graded on a 5-point scale from 0 to 4. Fibrosis scores were as follows: F0, no fibrosis; F1, portal fibrosis without septa; F2, portal fibrosis with few septa; F3, numerous septa without cirrhosis; and F4, cirrhosis. NA, not available.
Supplementary Table 3.
Primer Sequences Used for Real-Time Polymerase Chain Reaction Analysis
| Gene symbol | Forward primer (5'-3') | Reverse primer (5'-3') |
|---|---|---|
| GAPDH | GAAGGTGAAGGTCGGAGTC | GAAGATGGTGATGGGATTTC |
| 18s | GTAACCCGTTGAACCCCATT | CCATCCAATCGGTAGTAGCG |
| CD68 | GAACCCCAACAAAACCAA | GATGAGAGGCAGCAAGAT |
| IL1β | ATGATGGCTTATTACAGTGGCAA | GTCGGAGATTCGTAGCTGGA |
| TNFα | ATCTTCTCGAACCCCGAGTGA | CGGTTCAGCCACTGGAGCT |
| IL10 | ATGCCCCAAGCTGAGAACCAAGACCCA | TCTCAAGGGGCTGGGTCAGCTATCCCA |
| TGF-β | CACCGGAGTTGTGCGGCAGT | GGCCGGTAGTGAACCCGTTGAT |
| CD206 | ACAAAAGCTGACACAAGGAAGATG | TCAGGAGGATCACAATGATGACTAC |
| COL-1A | GGCGGCCAGGGCTCCGAC | AATTCCTGGTCTGGGGCACC |
| TIMP1 | CACCCACAGACGGCCTTCT | TCTGGTGTCCCCACGAACTT |
| α-SMA | CTGTTCCAGCCATCCTTCAT | CGGCTTCATCGTATTCCTGT |
NOTE. The forward and reverse primer sequences used for real-time polymerase chain reaction assay in the study are shown.
References
- 1.Mohd Hanafiah K., Groeger J., Flaxman A.D. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57:1333–1342. doi: 10.1002/hep.26141. [DOI] [PubMed] [Google Scholar]
- 2.Messina J.P., Humphreys I., Flaxman A. Global distribution and prevalence of hepatitis C virus genotypes. Hepatology. 2015;61:77–87. doi: 10.1002/hep.27259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Webster D.P., Klenerman P., Dusheiko G.M. Hepatitis C. Lancet. 2015;385:1124–1135. doi: 10.1016/S0140-6736(14)62401-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rein D.B., Wittenborn J.S., Smith B.D. The cost-effectiveness, health benefits, and financial costs of new antiviral treatments for hepatitis C virus. Clin Infect Dis. 2015;61:157–168. doi: 10.1093/cid/civ220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bekeredjian-Ding I., Roth S.I., Gilles S. T cell-independent, TLR-induced IL-12p70 production in primary human monocytes. J Immunol. 2006;176:7438–7446. doi: 10.4049/jimmunol.176.12.7438. [DOI] [PubMed] [Google Scholar]
- 6.Medzhitov R., Janeway C.A., Jr. Decoding the patterns of self and nonself by the innate immune system. Science. 2002;296:298–300. doi: 10.1126/science.1068883. [DOI] [PubMed] [Google Scholar]
- 7.Wilkins C., Gale M., Jr. Recognition of viruses by cytoplasmic sensors. Curr Opin Immunol. 2010;22:41–47. doi: 10.1016/j.coi.2009.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu B.S., Groothuismink Z.M., Janssen H.L. Role for IL-10 in inducing functional impairment of monocytes upon TLR4 ligation in patients with chronic HCV infections. J Leukoc Biol. 2011;89:981–988. doi: 10.1189/jlb.1210680. [DOI] [PubMed] [Google Scholar]
- 9.Cros J., Cagnard N., Woollard K. Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity. 2010;33:375–386. doi: 10.1016/j.immuni.2010.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Saha B., Bala S., Hosseini N. Krüppel-like factor 4 is a transcriptional regulator of M1/M2 macrophage polarization in alcoholic liver disease. J Leukoc Biol. 2015;97:963–973. doi: 10.1189/jlb.4A1014-485R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cassol E., Cassetta L., Alfano M. Macrophage polarization and HIV-1 infection. J Leukoc Biol. 2010;87:599–608. doi: 10.1189/jlb.1009673. [DOI] [PubMed] [Google Scholar]
- 12.Lawn S.D., Butera S.T., Folks T.M. Contribution of immune activation to the pathogenesis and transmission of human immunodeficiency virus type 1 infection. Clin Microbiol Rev. 2001;14:753–777. doi: 10.1128/CMR.14.4.753-777.2001. table of contents. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu B.S., Janssen H.L., Boonstra A. Type I and III interferons enhance IL-10R expression on human monocytes and macrophages, resulting in IL-10-mediated suppression of TLR-induced IL-12. Eur J Immunol. 2012;42:2431–2440. doi: 10.1002/eji.201142360. [DOI] [PubMed] [Google Scholar]
- 14.Liaskou E., Zimmermann H.W., Li K.K. Monocyte subsets in human liver disease show distinct phenotypic and functional characteristics. Hepatology. 2013;57:385–398. doi: 10.1002/hep.26016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dolganiuc A., Norkina O., Kodys K. Viral and host factors induce macrophage activation and loss of toll-like receptor tolerance in chronic HCV infection. Gastroenterology. 2007;133:1627–1636. doi: 10.1053/j.gastro.2007.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Auffray C., Sieweke M.H., Geissmann F. Blood monocytes: development, heterogeneity, and relationship with dendritic cells. Annu Rev Immunol. 2009;27:669–692. doi: 10.1146/annurev.immunol.021908.132557. [DOI] [PubMed] [Google Scholar]
- 17.Shi C., Pamer E.G. Monocyte recruitment during infection and inflammation. Nat Rev Immunol. 2011;11:762–774. doi: 10.1038/nri3070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Klein I., Cornejo J.C., Polakos N.K. Kupffer cell heterogeneity: functional properties of bone marrow derived and sessile hepatic macrophages. Blood. 2007;110:4077–4085. doi: 10.1182/blood-2007-02-073841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mosser D.M., Edwards J.P. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008;8:958–969. doi: 10.1038/nri2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Martinez F.O., Gordon S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep. 2014;6:13. doi: 10.12703/P6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rey-Giraud F., Hafner M., Ries C.H. In vitro generation of monocyte-derived macrophages under serum-free conditions improves their tumor promoting functions. PLoS One. 2012;7:e42656. doi: 10.1371/journal.pone.0042656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lolmede K., Campana L., Vezzoli M. Inflammatory and alternatively activated human macrophages attract vessel-associated stem cells, relying on separate HMGB1- and MMP-9-dependent pathways. J Leukoc Biol. 2009;85:779–787. doi: 10.1189/jlb.0908579. [DOI] [PubMed] [Google Scholar]
- 23.Schuppan D., Krebs A., Bauer M. Hepatitis C and liver fibrosis. Cell Death Differ. 2003;10(Suppl 1):S59–S67. doi: 10.1038/sj.cdd.4401163. [DOI] [PubMed] [Google Scholar]
- 24.Schuppan D., Schattenberg J.M. Non-alcoholic steatohepatitis: pathogenesis and novel therapeutic approaches. J Gastroenterol Hepatol. 2013;28(Suppl 1):68–76. doi: 10.1111/jgh.12212. [DOI] [PubMed] [Google Scholar]
- 25.Kolb M., Margetts P.J., Anthony D.C. Transient expression of IL-1beta induces acute lung injury and chronic repair leading to pulmonary fibrosis. J Clin Invest. 2001;107:1529–1536. doi: 10.1172/JCI12568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oikonomou N., Harokopos V., Zalevsky J. Soluble TNF mediates the transition from pulmonary inflammation to fibrosis. PLoS One. 2006;1:e108. doi: 10.1371/journal.pone.0000108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shirasaki T., Honda M., Shimakami T. Impaired IFN signaling in chronic hepatitis C patients with advanced fibrosis via the TGF-beta signaling pathway. Hepatology. 2014;60:1519–1530. doi: 10.1002/hep.27277. [DOI] [PubMed] [Google Scholar]
- 28.Li S., Vriend L.E., Nasser I.A. Hepatitis C virus-specific T-cell-derived transforming growth factor beta is associated with slow hepatic fibrogenesis. Hepatology. 2012;56:2094–2105. doi: 10.1002/hep.25951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Serti E., Werner J.M., Chattergoon M. Monocytes activate natural killer cells via inflammasome-induced interleukin 18 in response to hepatitis C virus replication. Gastroenterology. 2014;147:209–220.e3. doi: 10.1053/j.gastro.2014.03.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang S., Kodys K., Li K. Human type 2 myeloid dendritic cells produce interferon-lambda and amplify interferon-alpha in response to hepatitis C virus infection. Gastroenterology. 2013;144:414–425.e7. doi: 10.1053/j.gastro.2012.10.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Castilho-Fernandes A., de Almeida D.C., Fontes A.M. Human hepatic stellate cell line (LX-2) exhibits characteristics of bone marrow-derived mesenchymal stem cells. Exp Mol Pathol. 2011;91:664–672. doi: 10.1016/j.yexmp.2011.09.002. [DOI] [PubMed] [Google Scholar]
- 32.Hou W., Bukong T.N., Kodys K. Alcohol facilitates HCV RNA replication via up-regulation of miR-122 expression and inhibition of cyclin G1 in human hepatoma cells. Alcohol Clin Exp Res. 2013;37:599–608. doi: 10.1111/acer.12005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dolganiuc A., Oak S., Kodys K. Hepatitis C core and nonstructural 3 proteins trigger toll-like receptor 2-mediated pathways and inflammatory activation. Gastroenterology. 2004;127:1513–1524. doi: 10.1053/j.gastro.2004.08.067. [DOI] [PubMed] [Google Scholar]
- 34.Chattergoon M.A., Latanich R., Quinn J. HIV and HCV activate the inflammasome in monocytes and macrophages via endosomal Toll-like receptors without induction of type 1 interferon. PLoS Pathog. 2014;10:e1004082. doi: 10.1371/journal.ppat.1004082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Heydtmann M. Macrophages in hepatitis B and hepatitis C virus infections. J Virol. 2009;83:2796–2802. doi: 10.1128/JVI.00996-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Verreck F.A., de Boer T., Langenberg D.M. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc Natl Acad Sci U S A. 2004;101:4560–4565. doi: 10.1073/pnas.0400983101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Martinez F.O., Gordon S., Locati M. Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J Immunol. 2006;177:7303–7311. doi: 10.4049/jimmunol.177.10.7303. [DOI] [PubMed] [Google Scholar]
- 38.Luo D.D., Fielding C., Phillips A. Interleukin-1 beta regulates proximal tubular cell transforming growth factor beta-1 signalling. Nephrol Dial Transplant. 2009;24:2655–2665. doi: 10.1093/ndt/gfp208. [DOI] [PubMed] [Google Scholar]
- 39.Granowitz E.V., Clark B.D., Vannier E. Effect of interleukin-1 (IL-1) blockade on cytokine synthesis: I. IL-1 receptor antagonist inhibits IL-1-induced cytokine synthesis and blocks the binding of IL-1 to its type II receptor on human monocytes. Blood. 1992;79:2356–2363. [PubMed] [Google Scholar]
- 40.Wakita T., Pietschmann T., Kato T. Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat Med. 2005;11:791–796. doi: 10.1038/nm1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pilling D., Fan T., Huang D. Identification of markers that distinguish monocyte-derived fibrocytes from monocytes, macrophages, and fibroblasts. PLoS One. 2009;4:e7475. doi: 10.1371/journal.pone.0007475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Reilkoff R.A., Bucala R., Herzog E.L. Fibrocytes: emerging effector cells in chronic inflammation. Nat Rev Immunol. 2011;11:427–435. doi: 10.1038/nri2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wree A., Eguchi A., McGeough M.D. NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology. 2014;59:898–910. doi: 10.1002/hep.26592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Barth H., Rybczynska J., Patient R. Both innate and adaptive immunity mediate protective immunity against hepatitis C virus infection in chimpanzees. Hepatology. 2011;54:1135–1148. doi: 10.1002/hep.24489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Boltjes A., Movita D., Boonstra A. The role of Kupffer cells in hepatitis B and hepatitis C virus infections. J Hepatol. 2014;61:660–671. doi: 10.1016/j.jhep.2014.04.026. [DOI] [PubMed] [Google Scholar]
- 46.Hosomura N., Kono H., Tsuchiya M. HCV-related proteins activate Kupffer cells isolated from human liver tissues. Dig Dis Sci. 2011;56:1057–1064. doi: 10.1007/s10620-010-1395-y. [DOI] [PubMed] [Google Scholar]
- 47.Sica A., Invernizzi P., Mantovani A. Macrophage plasticity and polarization in liver homeostasis and pathology. Hepatology. 2014;59:2034–2042. doi: 10.1002/hep.26754. [DOI] [PubMed] [Google Scholar]
- 48.Murray P.J., Wynn T.A. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol. 2011;11:723–737. doi: 10.1038/nri3073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yang L., Scott P.G., Giuffre J. Peripheral blood fibrocytes from burn patients: identification and quantification of fibrocytes in adherent cells cultured from peripheral blood mononuclear cells. Lab Invest. 2002;82:1183–1192. doi: 10.1097/01.lab.0000027841.50269.61. [DOI] [PubMed] [Google Scholar]
- 50.Mathai S.K., Gulati M., Peng X. Circulating monocytes from systemic sclerosis patients with interstitial lung disease show an enhanced profibrotic phenotype. Lab Invest. 2010;90:812–823. doi: 10.1038/labinvest.2010.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tourkina E., Bonner M., Oates J. Altered monocyte and fibrocyte phenotype and function in scleroderma interstitial lung disease: reversal by caveolin-1 scaffolding domain peptide. Fibrogenesis Tissue Repair. 2011;4:15. doi: 10.1186/1755-1536-4-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Niedermeier M., Reich B., Rodriguez Gomez M. CD4+ T cells control the differentiation of Gr1+ monocytes into fibrocytes. Proc Natl Acad Sci U S A. 2009;106:17892–17897. doi: 10.1073/pnas.0906070106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kisseleva T., Uchinami H., Feirt N. Bone marrow-derived fibrocytes participate in pathogenesis of liver fibrosis. J Hepatol. 2006;45:429–438. doi: 10.1016/j.jhep.2006.04.014. [DOI] [PubMed] [Google Scholar]
- 54.Nunnari G., Vancheri C., Gilli E. Circulating fibrocytes as a marker of liver fibrosis in chronic hepatitis C. Front Biosci (Elite Ed) 2010;2:1241–1245. doi: 10.2741/e184. [DOI] [PubMed] [Google Scholar]