Abstract
Introduction:
The aim of this study was to investigate if defective repair of DNA double-strand break (DSB) in head and neck squamous cell carcinoma (HNSCC) could be used as an early predictor of treatment response.
Methods:
Tumour biopsy 24–36 h following induction chemotherapy (IC) and pre-treatment biopsies were stained for RAD51 and geminin (S-phase marker) for immunofluorescence in patients with HNSCC. The difference between RAD51 score (percentage of geminin-positive cells that were also positive for RAD51) was calculated for the two specimens. Tumours with a percentage difference of⩽10% were deemed to have repaired IC-induced DSBs, and were classified as ‘RAD51 negative'. Response at 3 months post treatment and human papilloma virus (HPV) status were assessed.
Results:
Thirteen pairs of samples were available for analyses. Three samples were classified as RAD51 negative and 10 as RAD51 positive at 24 h post IC. All of the three patients with tumours classified as RAD51 negative had partial response or progressive disease and the 10 patients with tumours deemed RAD51 positive had a complete response. 100% of the HPV-positive tumours were RAD51 positive and had a complete response.
Conclusions:
We have demonstrated that impaired DSB DNA repair may underlie enhanced treatment sensitivity of HPV-positive HNSCC and repair capacity following platinum-induced DNA damage predicts response in HNSCC. This has potential as a biomarker for patient selection in trials of DNA damage response pathway modulation.
Keywords: deficient DNA DSB repair, treatment response, HNSCC, HPV positive
Radiotherapy with concomitant chemotherapy (CRT) is a standard-of-care for the non-surgical management of head and neck squamous cell carcinoma (HNSCC). Unrepaired DNA double-strand breaks (DSB) are the principal lesions that determine responses in tumours and normal tissues, following treatment with radiation and/or several cytotoxic agents (Willers et al, 2004). Error-free DSB repair is done via homologous recombination (HR) using sister chromatid DNA as a template (Jackson and Bartek, 2009). The protein RAD51 is a key component of HR and forms foci at the site of DSBs that can be visualised using immunofluorescence (Graeser et al, 2010). HR forms one component of a complex network of DNA damage response (DDR) pathways, functional assessment of which in tumours might predict treatment sensitivity or resistance (Willers et al, 2015).
Two studies in breast cancer have proven that RAD51 foci, as seen on immunofluorescence in tumour biopsies taken after induction of DNA damage, can be used as a functional marker of DDR (Graeser et al, 2010; Naipal et al, 2014). In addition, Graeser et al demonstrated that delayed HR as determined by non-resolution of RAD51 foci predict for response to neo-adjuvant chemotherapy in patients with breast cancer. To our knowledge, there have been no similar clinical studies in HNSCC. The aim of this study was to investigate if delayed HR-mediated repair of DNA DSB can be used as a predictive biomarker of response to CRT in HNSCC.
Materials and methods
Eligible patients had stage III/IV SCC of the oropharynx, with primary tumours that were visible trans-orally. All patients were scheduled to receive IC followed by CRT according to a standard regimen that has been previously described (Bhide et al, 2007; Wilkins et al, 2013). Informed consent was obtained from all subjects and the relevant research and development and ethics committees approved the study.
Data on stage (AJCC 2007), smoking and human papilloma virus (HPV) status (p16 immunohistochemistry (IHC)) and response at 3 months post treatment were collected. Response at 3 months after treatment was assessed using PET-CT scans. Residual disease as diagnosed on PET-CT scans was biopsy confirmed. HPV status was confirmed using PCR with reverse transcription (RT–PCR) for E6 mRNA in the diagnostic biopsy (Smeets et al, 2007).
Study intervention
All patients underwent a punch biopsy of the primary tumour under local anaesthetic between 24–30 h after administration of platinum during the first cycle of IC. The biopsy specimen was immediately fixed in a 10% neutral buffered formalin solution (Sigma-Aldrich, St Louis, MO, USA) and transferred to the pathology department at our institution for preparation of formalin-fixed paraffin-embedded (FFPE) tumour blocks. Haematoxylin-eosin stained slides prepared from the FFPE material were reviewed by a consultant pathologist (KT) to confirm the presence of tumour. Directly contiguous slides were cut for the RAD51 and phosphorylated H2AX (γH2AX) analyses. γH2AX staining was performed to confirm that the biopsied tumours had been exposed to chemotherapy in concentrations sufficient to induce DNA damage.
Pre-treatment FFPE tumour blocks, that were used for primary diagnosis and hence had presence of tumour confirmed by a specialist head and neck pathologist at relevant institution were also obtained.
RAD51 immunofluorescence assay
The pre-treatment and post-IC tumour blocks were stained using identical procotols previously validated at our institution (Graeser et al, 2010). Irradiated FFPE mouse xenograft was used as a positive control. Immunofluorescence analysis was carried out on 3 μm sections of FFPE tumour material. Following antigen retrieval by microwaving at pH 9 (DAKO pH 9 buffer) for 18 min, followed by 20 min cooling in buffer, sections were treated with Triton 0.2% for permeabilization for 20 min, washed in phosphate-buffered saline (PBS), treated with 100 μl of DNAse I (Roche) 1 : 10 000 for 1 h at 37 °C and blocked with immunofluorescence buffer (IFF; 1% bovine serum albumin, 2% fetal bovine serum; PAA gold in PBS) for 30 min at room temperature. Sections were stained with geminin antibody (10802-1-AP, ProteinTech Group). 1 : 400 in IFF for 1 h at room temperature, washed with PBS, followed by anti-rabbit Alexa488 (Invitrogen, #A21121) conjugate 1 : 1000 in IFF for 1 h at room temperature, washed with PBS, fixed with 4% paraformaldehyde (PFA) solution for 15 min, stained with RAD51 antibody (Clone 14B4; GeneTex) 1 : 100 in IFF for 1 h at room temperature, washed with PBS, followed by anti-mouse Alexa555 conjugate (Invitrogen, #A21429) 1 : 1000 in IFF for 1 h at room temperature, washed in PBS with 4', 6 diamidino 2 phenylindole (DAPI; 1 : 10000) for 15 min, and sections were fixed again with 4% paraformaldehyde (Sigma-Aldrich). The protocol for γH2AX staining was similar, with primary antibody 1 : 500 (phospho- Histone H2AX Ser139 Clone JBW301; Millipore) in IFF for 1 h at RT.
Scoring RAD51 foci
Images were captured on a Leica TCS confocal microscope (Leica Microsystems, Milton Keynes, Bucks, UK) set up with the laser outputs controlled via the Acousto-Optical Tunable Filter and the four collection windows using the Acousto-Optical Beam Splitter as follows; 403 nm laser (25%) window 410–483 nm, 488 nm laser (25%) window 493–538 nm, 543 nm laser (100%) window 548–628 nm and 633 nm laser (25%) window 638–700 nm. Images were collected using the microscope in sequential mode with a line average of 4 and a format of 1024 Å∼1024 pixels. The image acquisition protocol for the study samples was calibrated based on the positive control. Three investigators blinded to the time-point of the sample and all clinical data, including clinical response, counted the foci independently. The staining of nuclear foci was scored as follows. Between 100 and 500 cells in the sub-epithelial layer (invasive tumour) with any nuclear geminin staining (S-phase marker), were counted at representative areas across the section. A cell with any nuclear geminin staining was counted as being positive and in S-phase of the cell cycle. Any geminin positive cell in the sub-epithelial layer would be a proliferating cell and by definition a tumour cell. RAD51 foci were only counted in the geminin-positive cells. A cell was counted as being RAD51 positive if there was at least one distinct focus per nucleus. γH2AX staining was performed on a contiguous section, with a cell at least one focus considered positive. This threshold was selected as per the previous study by Graeser et al (2010). The mean of the foci counted by the three independent investigators was calculated. The percentage of geminin-positive cells that were also positive for RAD51 was calculated as the RAD51 score for pre-treatment and 24 h post-treatment specimens. The difference between the RAD51 score for the two specimens was then calculated. Tumour specimens with a percentage difference of ⩽10% between pre- and post-treatment specimens were deemed to have repaired the IC-induced DSBs, and were classified as RAD51 negative. The remaining patients were classified as RAD51 positive. This limit was set, as per Graeser et al (2010), before unblinding the response data.
Results
Fifteen patients were recruited into the study. The pre-treatment diagnostic biopsy was unavailable for one patient and geminin immunofluorescence staining was deemed to be of insufficient quality in one post-IC biopsy sample. Therefore, 13 pairs of samples were available for detailed analysis. Table 1 details the demographic data for these patients. IHC and RT–PCR were in agreement and confirmed that five tumours were HPV-negative and eight were HPV-positive.
Table 1. Clinical characteristics, RAD 51 status and treatment response of patients.
| Trial no. | Stage | HPV status | Smoking status (pack years) | Prognostic group | Difference in RAD51 foci pre- and post-IC (%) | RAD 51 status | Treatment response |
|---|---|---|---|---|---|---|---|
| 1002 | T2N2B | −ve | 30 | Poor | 31 | Positive | CR |
| 1003 | T4N2B | +ve | 20 | Intermediate | 25 | Positive | CR |
| 1004 | T4N2B | −ve | 40 | Poor | 7 | Negative | PR |
| 1005 | T2N2B | −ve | 20 | Poor | 3 | Negative | PR |
| 1006 | T2N2B | +ve | Never | Good | 62 | Positive | CR |
| 1007 | T2N1 | +ve | Never | Good | 44 | Positive | CR |
| 1008 | T3N2B | +ve | 30 | Intermediate | 71 | Positive | CR |
| 1009 | T3N0 | +ve | 15 | Good | 74 | Positive | CR |
| 1010 | T4N2C | +ve | 30 | Intermediate | 27 | Positive | CR |
| 1011 | T3N2B | +ve | 30 | Intermediate | 72 | Positive | CR |
| 1013 | T4N2B | −ve | 40 | Poor | 3 | Negative | PR |
| 1014 | T2N2B | −ve | 40 | Poor | 27 | Positive | CR |
| 1015 | T3N2B | +ve | Never | Good | 57 | Positive | CR |
Abbreviations: CR=complete response; HPV=human papilloma virus; PR=partial response.
The median RAD51 scores for the pre- and post-IC samples were 12 and 50%, respectively. Based on a cut-off of persistence of RAD51 foci at<or >10% above baseline, three tumours were classified as RAD51 negative (<10%) and ten as RAD51 positive (>10%) at 24-hours post-IC. Figure 1 shows example confocal images for RAD51 negative (1A) and RAD51 positive (1B) tumours. Pre- and post-IC samples demonstrated 20% average increase in percentage of γH2AX foci (Supplementary Figure 1). There was no difference in the tumours samples from RAD51 negative (58% increase) and positive (52% increase) patients (Supplementary Figures 2 and 3).
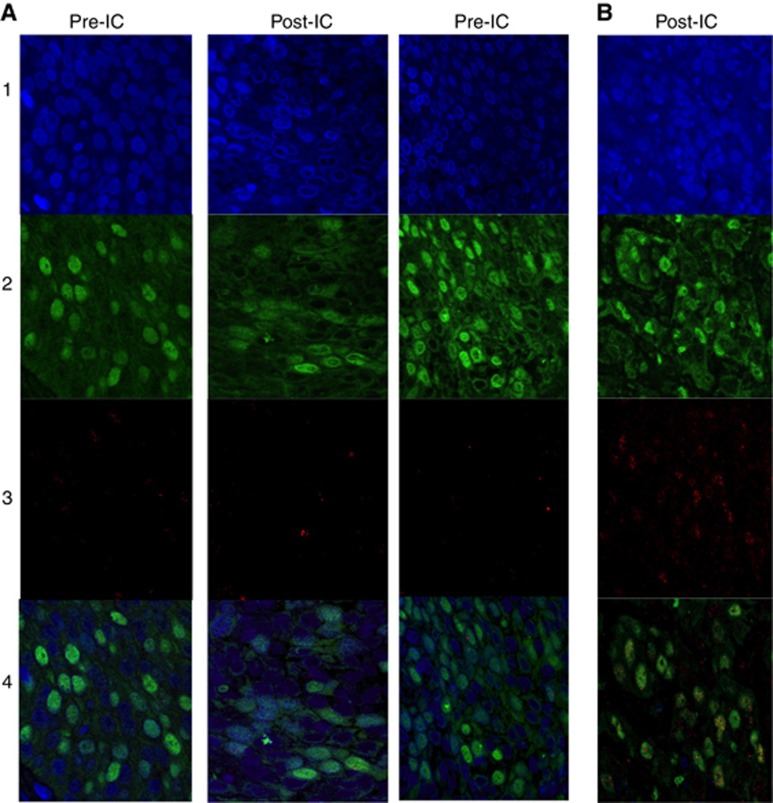
Figure 1.
Shows typical example of confocal images with immunofluorescence for DAPI (blue, 1), geminin (green, 2) RAD51 (red, 3) and overlaid (4). (A) (RAD51-negative tumours) shows similar proportion of geminin positive with Rad51 foci in post-IC compared with pre-IC images and (B) (RAD51-positive tumours) shows increased proportion of cells with RAD51 foci in post-IC images.
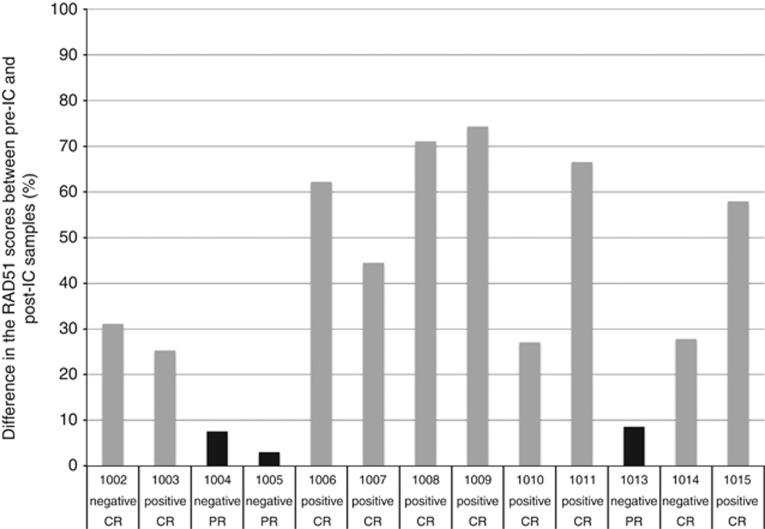
Treatment outcomes at 3 months post CRT are detailed in Table 1. Complete clinical and radiological responses (CR) to CRT at this time were achieved for 10/13 (77%) patients; the remaining 3/13 (23%) patients had evidence of persistent or progressive disease (PD). All of the three patients classified as having RAD51-negative tumours had persistent or PD and the 10 patients with RAD51-positive tumours had a CR (Table 1 and Figure 2). Two out of the five (40%) patients with HPV-negative disease and 100% of the patients with HPV-positive disease had RAD51-positive tumours and had a CR. All three RAD51-negative tumours with PD were HPV-negative and would have been predicted to be in the poor prognosis group according to the Ang classification (Ang et al, 2010).
Figure 2.
Shows the relationship between RAD51-negative tumours (black), RAD51-positive tumours (grey), treatment response and HPV status. X-Axis Legend: 100…=patient trial number; CR=complete response; PR=partial response/progressive disease; negative=HPV negative; and positive=HPV positive.
Discussion
This study demonstrates that HR functional status, as determined by resolution of RAD51 foci following induction of DNA damage by platinum-containing chemotherapy, predicts treatment response after CRT for HNSCC.
We were able to classify the patients with HPV-negative tumours into responders (40%) and non-responders (60%) based on the repair of RAD51 foci of their tumours. There was no difference in the γH2AX foci induction in RAD51-negative and -positive patients, indicating that the differences in the RAD51 foci were as a result of differences in tumour biology and opposed to poor chemotherapy exposure. HNSCC can be divided into good, intermediate and poor prognosis groups based on HPV status, tumour stage and smoking history (Ang et al, 2010). Clinical trials of treatment de-escalation in the good prognosis group (HPV-positive) are currently underway (Masterson et al, 2014) and dose-intensification strategies are being explored in the poor prognosis group (Dillon and Harrington, 2015). One approach to treatment intensification is through DDR pathway modulation, based on the high prevalence of TP53 mutations and the resulting dysfunction in downstream DDR pathways in HPV-negative HNSCC (Dillon et al, 2014). DNA DSB repair status, as determined in this study, could potentially be used as a biomarker to identify patients with RAD51 negative tumours who might benefit from DDR modulation during CRT.
All of the HPV-positive tumours in this study were RAD51 positive and had CR following treatment. This suggests that impaired DNA DSB repair might underlie significantly higher response rates observed in HPV-positive HNSCC. Previous in vitro studies have shown that impaired DNA DSB repair might underlie enhanced treatment sensitivity in HPV-positive HNSCC (Rieckmann et al, 2013; Park et al, 2014). Ours is the first study to provide clinical proof-of-principle for this hypothesis by assessing DSB repair in tumour biopsies before and after DNA-damaging therapy.
Previous IHC-based studies have correlated RAD51 expression to outcomes in HNSCC (Connell et al, 2006). However, HR protein expression at a single time-point does not provide information on the HR function DSB repair following DNA damage. This can be considered to be a major drawback when considering this as a predictive biomarker for treatment personalisation. The study by Graeser et al (2010) is the only other study that assessed DSB repair following induction of DNA damage, albeit in patients with breast cancer.
The Fanconi Anaemia pathway status for tumour samples was not known. The Fanconi Anaemia pathway consists of a network of up to 15 proteins, encoded by different genes (Thompson and Hinz, 2009). These proteins control DNA replication and mutagenic responses to endogenous and exogenous DNA damage. The functional status of this pathway can be affected by altered gene expression either through mutations or altered methylation status. Therefore, the complete characterisation of Fanconi Anaemia pathway status would have required assessment of the mutational and methylation status of all of the pathway genes. Unfortunately adequate tumour material was not available to undertake such an analyses. Not knowing the Fanconi Anaemia pathway status in these tumours can be considered a drawback of this study, nevertheless, somatic mutations in this pathway are highly uncommon in HPV-positive and HPV-negative head and neck SCC (Agrawal et al, 2011; Stransky et al, 2011).
This was a pilot study with a small sample size. Therefore formal statistical tests for comparisons were not undertaken. However, we have used validated scientific methodology with adequate controls and have accounted for cell cycle (S) phase dependence of the DSB repair pathway using geminin staining. DNA damage was induced in this study by platinum-containing IC. However, the use of IC in clinical practice continues to decline, in light of evidence of only marginal benefits when compared with CRT alone (Pignon et al, 2009; O'Sullivan et al, 2013). Therefore, it will be important to repeat this study on tumour samples taken before and after CRT. However, larger studies are required to validate our current findings and this is likely to be challenging given the invasive nature of repeated biopsies (especially during treatment). Preliminary studies have demonstrated the potential utility of analysing HR function in microRNA and circulating tumour DNA samples obtained at venesection. Such samples could be used for larger validation studies (Nel et al, 2013; Liu et al, 2015).
Conclusion
We have demonstrated that the failure to resolve RAD51 foci following platin-induced DNA damage can be used as an index of DSB repair and can predict response following CRT for HNSCC. This assay holds promise as a potential biomarker for patient selection in trials of DDR pathway modulation. In addition, this study provides direct clinical evidence that delayed DSB repair may explain the enhanced treatment sensitivity of HPV-positive HNSCC.
Acknowledgments
This work was undertaken in The Royal Marsden NHS Foundation Trust which received a proportion of its funding from the NHS Executive; the views expressed in this publication are those of the authors and not necessarily those of the NHS Executive. This work was supported by Cancer Research UK Programme Grants C46/A10588 and C7224/A13407. The authors acknowledge Dr Nicholas Turner (The Institute of Cancer Research) for his input into study design and David Robertson (The Institute of Cancer Research) for his help on confocal microscope. We also acknowledge the support of the National Institute for Health Research Royal Marsden and Institute of Cancer Research Biomedical Research Centre and the Clinical Research Facility.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on British Journal of Cancer website (http://www.nature.com/bjc)
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Material
References
- Agrawal N, Frederick MJ, Pickering CR, Bettegowda C, Chang K, Li RJ, Fakhry C, Xie TX, Zhang J, Wang J, Zhang N, El-Naggar AK, Jasser SA, Weinstein JN, Trevino L, Drummond JA, Muzny DM, Wu Y, Wood LD, Hruban RH, Westra WH, Koch WM, Califano JA, Gibbs RA, Sidransky D, Vogelstein B, Velculescu VE, Papadopoulos N, Wheeler DA, Kinzler KW, Myers JN (2011) Exome sequencing of head and neck squamous cell carcinoma reveals inactivating mutations in NOTCH1. Science 333(6046): 1154–1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tan PF, Westra WH, Chung CH, Jordan RC, Lu C, Kim H, Axelrod R, Silverman CC, Redmond KP, Gillison ML (2010) Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med 363(1): 24–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhide S, Clark C, Harrington K, Nutting CM (2007) Intensity modulated radiotherapy improves target coverage and parotid gland sparing when delivering total mucosal irradiation in patients with squamous cell carcinoma of head and neck of unknown primary site. Med. Dosim. 32(3): 188–195. [DOI] [PubMed] [Google Scholar]
- Connell PP, Jayathilaka K, Haraf DJ, Weichselbaum RR, Vokes EE, Lingen MW (2006) Pilot study examining tumor expression of RAD51 and clinical outcomes in human head cancers. Int J Oncol 28(5): 1113–1119. [PubMed] [Google Scholar]
- Dillon MT, Good JS, Harrington KJ (2014) Selective targeting of the G2/M cell cycle checkpoint to improve the therapeutic index of radiotherapy. Clin Oncol (R Coll Radiol) 26(5): 257–265. [DOI] [PubMed] [Google Scholar]
- Dillon MT, Harrington KJ (2015) Human papillomavirus-negative pharyngeal cancer. J Clin Oncol 33(29): 3251–3261. [DOI] [PubMed] [Google Scholar]
- Graeser M, McCarthy A, Lord CJ, Savage K, Hills M, Salter J, Orr N, Parton M, Smith IE, Reis-Filho JS, Dowsett M, Ashworth A, Turner NC (2010) A marker of homologous recombination predicts pathologic complete response to neoadjuvant chemotherapy in primary breast cancer. Clin Cancer Res 16(24): 6159–6168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson SP, Bartek J (2009) The DNA-damage response in human biology and disease. Nature 461(7267): 1071–1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu G, Yang D, Rupaimoole R, Pecot CV, Sun Y, Mangala LS, Li X, Ji P, Cogdell D, Hu L, Wang Y, Rodriguez-Aguayo C, Lopez-Berestein G, Shmulevich I, De Cecco L, Chen K, Mezzanzanica D, Xue F, Sood AK, Zhang W (2015) Augmentation of response to chemotherapy by microRNA-506 through regulation of RAD51 in serous ovarian cancers. J Natl Cancer Inst e-pub ahead of print 20 May 2015 doi:10.1093/jnci/djv108. [DOI] [PMC free article] [PubMed]
- Masterson L, Moualed D, Liu ZW, Howard JE, Dwivedi RC, Tysome JR, Benson R, Sterling JC, Sudhoff H, Jani P, Goon PK (2014) De-escalation treatment protocols for human papillomavirus-associated oropharyngeal squamous cell carcinoma: a systematic review and meta-analysis of current clinical trials. Eur J Cancer 50(15): 2636–2648. [DOI] [PubMed] [Google Scholar]
- Naipal KA, Verkaik NS, Ameziane N, van Deurzen CH, Ter Brugge P, Meijers M, Sieuwerts AM, Martens JW, O'Connor MJ, Vrieling H, Hoeijmakers JH, Jonkers J, Kanaar R, de Winter JP, Vreeswijk MP, Jager A, van Gent DC (2014) Functional ex vivo assay to select homologous recombination-deficient breast tumors for PARP inhibitor treatment. Clin Cancer Res 20(18): 4816–4826. [DOI] [PubMed] [Google Scholar]
- Nel I, Gauler TC, Eberhardt WE, Nickel AC, Schuler M, Thomale J, Hoffmann AC (2013) Formation and repair kinetics of Pt-(GpG) DNA adducts in extracted circulating tumour cells and response to platinum treatment. Br J Cancer 109(5): 1223–1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Sullivan B, Huang SH, Siu LL, Waldron J, Zhao H, Perez-Ordonez B, Weinreb I, Kim J, Ringash J, Bayley A, Dawson LA, Hope A, Cho J, Irish J, Gilbert R, Gullane P, Hui A, Liu FF, Chen E, Xu W (2013) Deintensification candidate subgroups in human papillomavirus-related oropharyngeal cancer according to minimal risk of distant metastasis. J Clin Oncol 31(5): 543–550. [DOI] [PubMed] [Google Scholar]
- Park JW, Nickel KP, Torres AD, Lee D, Lambert PF, Kimple RJ (2014) Human papillomavirus type 16 E7 oncoprotein causes a delay in repair of DNA damage. Radiother Oncol 113(3): 337–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pignon JP, le Maitre A, Maillard E, Bourhis J (2009) Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17 346 patients. Radiother Oncol 92(1): 4–14. [DOI] [PubMed] [Google Scholar]
- Rieckmann T, Tribius S, Grob TJ, Meyer F, Busch CJ, Petersen C, Dikomey E, Kriegs M (2013) HNSCC cell lines positive for HPV and p16 possess higher cellular radiosensitivity due to an impaired DSB repair capacity. Radiother Oncol 107(2): 242–246. [DOI] [PubMed] [Google Scholar]
- Smeets SJ, Hesselink AT, Speel EJ, Haesevoets A, Snijders PJ, Pawlita M, Meijer CJ, Braakhuis BJ, Leemans CR, Brakenhoff RH (2007) A novel algorithm for reliable detection of human papillomavirus in paraffin embedded head and neck cancer specimen. Int J Cancer 121(11): 2465–2472. [DOI] [PubMed] [Google Scholar]
- Stransky N, Egloff AM, Tward AD, Kostic AD, Cibulskis K, Sivachenko A, Kryukov GV, Lawrence MS, Sougnez C, McKenna A, Shefler E, Ramos AH, Stojanov P, Carter SL, Voet D, Cortes ML, Auclair D, Berger MF, Saksena G, Guiducci C, Onofrio RC, Parkin M, Romkes M, Weissfeld JL, Seethala RR, Wang L, Rangel-Escareno C, Fernandez-Lopez JC, Hidalgo-Miranda A, Melendez-Zajgla J, Winckler W, Ardlie K, Gabriel SB, Meyerson M, Lander ES, Getz G, Golub TR, Garraway LA, Grandis JR (2011) The mutational landscape of head and neck squamous cell carcinoma. Science 333(6046): 1157–1160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson LH, Hinz JM (2009) Cellular and molecular consequences of defective Fanconi anemia proteins in replication-coupled DNA repair: mechanistic insights. Mutat Res 668(1-2): 54–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkins AC, Rosenfelder N, Schick U, Gupta S, Thway K, Nutting CM, Harrington KJ, Newbold K, Bhide SA (2013) Equivalence of cisplatin and carboplatin-based chemoradiation for locally advanced squamous cell carcinoma of the head and neck: a matched-pair analysis. Oral Oncol 49(6): 615–619. [DOI] [PubMed] [Google Scholar]
- Willers H, Dahm-Daphi J, Powell SN (2004) Repair of radiation damage to DNA. Br J Cancer 90(7): 1297–1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willers H, Gheorghiu L, Liu Q, Efstathiou JA, Wirth LJ, Krause M, von Neubeck C (2015) DNA damage response assessments in human tumor samples provide functional biomarkers of radiosensitivity. Semin Radiat Oncol 25(4): 237–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.