Abstract
Background
We characterized cardiac surgery-induced dynamic changes of the corrected QT (QTc) interval and tested the hypothesis that genetic factors are associated with perioperative QTc prolongation independent of clinical and procedural factors.
Methods
All study subjects were ascertained from a prospective study of patients who underwent elective cardiac surgery during August 1999 to April 2002. We defined a prolonged QTc interval as >440 msec, measured from 24-hr pre- and postoperative 12-lead electrocardiograms. The association of 37 single nucleotide polymorphisms (SNPs) in 21 candidate genes – involved in modulating arrhythmia susceptibility pathways with postoperative QTc changes–was investigated in a two-stage design with a stage I cohort (n = 497) nested within a stage II cohort (n = 957). Empirical P values (Pemp) were obtained by permutation tests with 10,000 repeats.
Results
After adjusting for clinical and procedural risk factors, we selected four SNPs (P value range, 0.03-0.1) in stage I, which we then tested in the stage II cohort. Two functional SNPs in the pro-inflammatory cytokine interleukin-1β (IL1β), rs1143633 (odds ratio [OR], 0.71; 95% confidence interval [CI], 0.53 to 0.95; Pemp = 0.02) and rs16944 (OR, 1.31; 95% CI, 1.01 to 1.70; Pemp = 0.04), remained independent predictors of postoperative QTc prolongation. The ability of a clinico-genetic model incorporating the two IL1B polymorphisms to classify patients at risk for developing prolonged postoperative QTc was superior to a clinical model alone, with a net reclassification improvement of 0.308 (P = 0.0003) and an integrated discrimination improvement of 0.02 (P = 0.000024).
Conclusion
The results suggest a contribution of IL1β in modulating susceptibility to postoperative QTc prolongation after cardiac surgery.
Résumé
Contexte
Nous avons caractérisé les changements dynamiques de l’intervalle QT corrigé (QTc) induits par une chirurgie cardiaque et testé l’hypothèse selon laquelle des facteurs génétiques sont associés à une prolongation périopératoire du QTc indépendamment de facteurs cliniques ou procéduraux.
Méthode
Tous les patients évalués dans cette étude ont été sélectionnés à partir d’une étude prospective portant sur des patients ayant subi une chirurgie cardiaque non urgente entre août 1999 et avril 2002. Nous avons défini un intervalle QTc prolongé en tant que >440 msec, mesuré à l’aide d’électrocardiogrammes à 12 dérivations 24 h préet postopératoires. L’association de 37 polymorphismes nucléotidiques simples (PNS) dans 21 gènes candidats – impliqués dans la modulation des voies susceptibles à l’arythmie avec les changements postopératoires du QTc – a été examinée dans un modèle à deux stades, avec une cohorte de stade I (n = 497) imbriquée dans la cohorte de stade II (n = 957). Les valeurs P empiriques (Pemp) ont été obtenues grâce à des tests de permutation à 10 000 répétitions.
Résultats
Après avoir procédé à des ajustements pour tenir compte des facteurs de risque cliniques et procéduraux, nous avons sélectionné quatre PNS (plage de valeurs P, 0,03-0,1) dans le stade I, que nous avons testé par la suite dans la cohorte de stade II. Deux PNS fonctionnels de cytokine pro-inflammatoire interleukine-1β (IL1β), soit le rs1143633 (rapport de cotes [RC], 0, 71; intervalle de confiance [IC] 95 %, 0,53 à 0,95; Pemp = 0,02) et le rs16944 (RC, 1,31; IC 95 %, 1,01 à 1,70; Pemp = 0,04), sont demeurés des prédicteurs indépendants de prolongation postopératoire du QTc. La capacité d’un modèle clinico-génétique incorporant les deux polymorphismes d’IL1β à classifier les patients à risque de manifester un QTc postopératoire prolongé était supérieure à celle d’un modèle clinique seul, avec une amélioration de reclassification nette de 0,308 (P = 0,0003) et une amélioration de la discrimination intégrée de 0,02 (P = 0,000024).
Conclusion
Selon ces résultats, l’IL1β pourrait contribuer à moduler la susceptibilité à une prolongation postopératoire du QTc après une chirurgie cardiaque.
Introduction
Abnormal cardiac repolarization, as indicated by electrocardiographic corrected QT (QTc) interval prolongation,1 is of significant interest to perioperative clinicians in light of its frequent occurrence after major surgeries in both adult and pediatric populations,2-5 the relationship with postoperative ventricular arrhythmias (e.g., torsades de pointes),6 and the prognostic implications for sudden cardiac death.7-11 Acquired QTc prolongation results from a combination of environmental stress and pharmacological effects on a genetic predisposition background that reduces an individual’s repolarization reserve.12 The duration of the QTc interval, like other electrocardiographic indices, is highly heritable in the general population, with 35-45% of its variation attributed to genetic factors as evidenced by family linkage, twin, and genome-wide association studies.13,14 A spectrum of genetic variants has been identified as modifiers of changes in QTc intervals in response to environmental stressors or upon drug exposure, which can serve as second hits to unmask subclinical genetic susceptibilities.15 Cardiac surgery provides such a robust environmental stressor, with a combination of myocardial ischemia-reperfusion, inflammation, oxidative stress, hypothermia, alterations in autonomic tone, and electrolyte disturbances, which we postulate would collectively increase a patient’s vulnerability for acquired QTc prolongation. In addition, cardiac surgical patients face simultaneous exposure to inotropes and an ever-growing list of other QTc-prolonging drugs.16-18 Nevertheless, the role of superimposed genetic effects in modulating inter-individual variability in myocardial repolarization abnormalities following surgery remains unknown, leaving potentially important implications for perioperative risk stratification, surveillance, and management. Most of the genetic diversity in the population is attributable to widespread DNA sequence variants, typically single nucleotide base substitutions (i.e., single nucleotide polymorphisms [SNPs]). Polymorphisms may directly alter the amino acid sequence and therefore potentially influence protein function or alter regulatory DNA sequences that modulate protein expression. Genetic association studies examine whether the frequency of specific genetic polymorphisms differs between diseased subjects and healthy controls. Such studies have been successful in identifying susceptibility for a variety of perioperative adverse outcomes, including myocardial infarction, neurocognitive dysfunction, stroke, and acute kidney injury.19
This novel study aimed to characterize the acute perioperative changes in QTc duration following cardiac surgery and to test the hypothesis that genetic variants are associated with postoperative QTc prolongation independent of clinical and procedural risk factors.
Methods
We performed a joint two-stage candidate gene association study to identify genetic variants predisposing to postoperative QTc prolongation.20 Furthermore, we followed the recommendations of the “Strengthening the Reporting of Genetic Association Studies” for reporting the study results.21
Description of study populations
After obtaining Institutional Review Board approval and written informed consent, we enrolled patients 18 years of age and older and undergoing cardiac surgery into the Perioperative Genetics and Safety Outcomes Study (PEGASUS),22 an ongoing longitudinal single-site study initiated in 1989 at Duke University Medical Center (Durham, NC, USA). The PEGASUS study used comprehensive strategies (e.g., candidate genes, pharmacogenomic analyses, and gene expression profiling) in an effort to identify genetic variations associated with increased susceptibility for major adverse perioperative events following cardiac surgery.22,23 The present substudy targeted a cohort of consecutive patients who underwent cardiac surgery from August 1999 to April 2002 and obtained detailed perioperative QTc interval information from electronically stored 12-lead electrocardiograms. Patients were excluded from enrolment in PEGASUS if they had a history of renal failure, active liver disease, bleeding disorders, autoimmune diseases, or immunosuppressive therapy. We further excluded patients from this substudy cohort if they required postoperative pacing, developed perioperative atrial fibrillation/ flutter or bundle branch block, or received perioperative drugs known to be associated with QTc interval prolongation (e.g., antiarrhythmics - amiodarone, sotalol, procainamide; antibiotics – erythromycin, ciprofloxacin; antiemetics – droperidol, promethazine, ondansetron; antipsychotics – haloperidol; and antihistamines). Based on these criteria, 91 patients were excluded. This study design attempted to minimize the confounding effects of factors outside perioperative stress that might have influenced the QTc interval directly or indirectly. Nevertheless, detailed information was not available for patients previously diagnosed with or undergoing treatment for congenital long QT syndrome. The cohort identified for the current substudy has a limited overlap with participants in prior published analyses involving other postoperative outcomes, some of which reflect more recently enrolled patients.24-26
Intraoperative anesthetic, perfusion, and cardioprotective management were standardized and overall consistent with contemporary practice. General anesthesia was maintained with a combination of fentanyl and isoflurane. Perfusion support consisted of non-pulsatile cardiopulmonary bypass (30-32°C), crystalloid prime, pump flow rates > 2.4 L·min−1·m−2, cold blood cardioplegia, α-stat blood gas management with heparin to maintain activated clotting times > 450 sec, ε-aminocaproic acid infusion routinely administered, and serial hematocrits kept at > 0.18.
Data collection and endpoint definition
Patient demographics, comorbidities, medications, intraoperative variables, and postoperative outcomes were collected and curated during the index hospitalization using the Duke Information System for Cardiovascular Care, an integral part of the Duke Databank for Cardiovascular Disease. Postoperative ventricular arrhythmias were defined as new-onset sustained ventricular tachycardia (lasting ≥ 30 sec or requiring cardioversion) or ventricular fibrillation. Two independent investigators blinded to the clinical and genetic data (Supplemental data Ia; available as Electronic Supplementary Material) performed the QT interval analysis within the 24-hr period before and after surgery using electronically stored 12-lead electrocardiograms (Philips TraceMaster, Andover, MA, USA. The QT interval was corrected for heart rate using the Bazett formula,27 and the QTc interval duration was calculated as the mean duration of all QTc intervals measured. Prolonged QTc interval was defined as a postoperative QTc > 440 msec.28
Rationale for candidate polymorphism selection
We selected 45 SNPs within a set of 24 candidate genes based on current understanding of normal cardiac electrophysiology, the molecular pathophysiology of QTc prolongation, and responses to cardiac surgery-induced inflammatory and oxidative stress.12,29-33 Emphasis was placed on common SNPs that were functionally relevant to the physiological disturbances underlying arrhythmia susceptibility. This was evidenced by preliminary functional characterization of variant proteins in vitro, in genetically modified animals, in computer simulations, as well as in previous genetic epidemiological analyses. A list of the candidate genes and polymorphisms studied is provided in Table S1 (available as Electronic Supplementary Material).
Genotyping and quality controls
Genomic DNA was isolated from whole blood using standard procedures. Genotyping was performed in a single batch by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry on a Sequenom™ system (Sequenom, Inc., San Diego, CA, USA) at a core facility (Agencourt Bioscience Corporation, Beverly, MA, USA).23 The primers used and details of polymorphisms can be found in Table S2 (available as Electronic Supplementary Material). A SNP call rate threshold of > 85% was imposed on all variants genotyped. The accuracy of genotyping was validated at > 99% by scoring a panel of six SNPs in 100 randomly selected patients using ABI PRISM® 3700 capillary sequencer (Applied Biosystems, Foster City, CA, USA). As an additional quality control, Hardy-Weinberg equilibrium (HWE) was evaluated in controls using an exact test. Eight of the 45 candidate SNPs deviated from HWE, and we excluded these from subsequent analyses to avoid biasing the estimates of genetic effects.
Considering that population stratification can confound genetic association studies, a reference panel of 54 ancestry-informative unlinked markers was genotyped to assess and control for population structure using a modified Eigenstrat method, as previously described (Supplemental data Id and Tables S3 and S4; available as Electronic Supplementary Material).34
Joint genotype and phenotype information was available in 957 patients. In stage I, the cohort consisted of the 497 earliest enrolled patients in chronological order of their index surgery date. In stage II, we tested the top genetic variants in a joint analysis dataset within which the discovery cohort was nested and expanded by 460 subsequently enrolled patients, for 957 patients overall. Such a joint analysis strategy of all available samples has been shown to result in increased power to detect genetic associations when compared with the alternative standard replication-based strategies.35
Statistical analysis
We evaluated clinical risk factors for postoperative QTc interval prolongation, including age, sex, self-reported ethnicity, comorbidities, medication use at hospital admission, type of cardiac surgical procedure, and duration of aortic cross-clamp. Differences in these demographic, clinical, and procedural characteristics between cases (average postoperative QTc > 440 msec) and controls (average postoperative QTc ≤ 440 msec) were assessed by χ2 tests. Within the stage I cohort, covariates with two-tailed nominal P ≤ 0.1 in univariable analyses were used to fit a multivariable logistic regression model using forward stepwise selection guided by the Akaike information criterion. Allelic associations with postoperative QTc prolongation were first analyzed univariately using χ2 tests for each of the 37 SNPs, assuming additive, dominant, or recessive inheritance patterns. Subsequently, a multivariable logistic regression analysis was implemented to adjust genetic association tests for clinical and procedural covariates, self-reported ethnicity, and the top significant principal components to form the population stratification analysis (Supplemental data Id and Table S4; available as Electronic Supplementary Material). Potential susceptibility SNPs with two-tailed P ≤ 0.1 were tested in the stage II cohort using the same multivariable logistic regression model. To correct for multiple testing, family-wise error rates were estimated for 37 SNP comparisons within each model by permutation (10,000 iterations), and empirical P values (Pemp) were reported for the stage II analyses.36 We conducted haplotype analyses for the top SNPs identified in the stage II analyses using Haplo Stats (http://mayoresearch.mayo.edu/mayo/research/schaid_lab/software.cfm). Comparative performance of risk models, including clinical and clinico-genetic information to predict postoperative QTc prolongation was assessed using receiver operating characteristic, global measures of model fit, and reclassification analysis (Supplemental data Ie; available as Electronic Supplementary Material).
The relevance of postoperative QTc prolongation on the incidence of postoperative ventricular arrhythmias and perioperative (30-day), one-year, and five-year mortality was assessed using logistic regression analyses, adjusted for EuroSCORE.22,37 All statistical analyses were performed using SAS/Genetics® version 9.2 (SAS® Inc, Cary, NC, USA) or R version 2.15.1 (The R Foundation for Statistical Computing, Vienna, Austria).
Mechanistic subset analysis
Based on positive genetic associations, we serially measured circulating levels of interleukin-1β (IL-1β) and interleukin-1 receptor antagonist (IL-1Ra) in a subset of 288 patients. The patients’ blood had been collected and handled (before induction of anesthesia and at four, 24, and, 48 hr after aortic cross-clamp removal) using PEGASUS standard operating procedures, centrifuged within 60 min at 4°C with serum aliquots, and stored at −80°C. Frozen aliquots from this cohort that had not undergone freeze-thaw cycles were shipped in a single batch to a core laboratory (Biosite, Inc., Waltham, MA, USA) within three years of storage, where a panel of cytokines and other biomarkers were measured using proprietary sandwich enzyme-linked immunosorbent assays (ELISA). Serial data were evaluated by analysis of variance (Kruskal-Wallis test, with Dunn’s post hoc multiple comparison test). To determine whether polymorphisms affect gene expression in response to cardiac surgery, the association between time-dependent changes in serum levels of IL-1β, IL-1Ra, and IL1B genotypes was tested using generalized estimating equations based on a first-order autoregressive variance-covariance matrix model.38
Results
Clinical risk factors for postoperative QTc prolongation
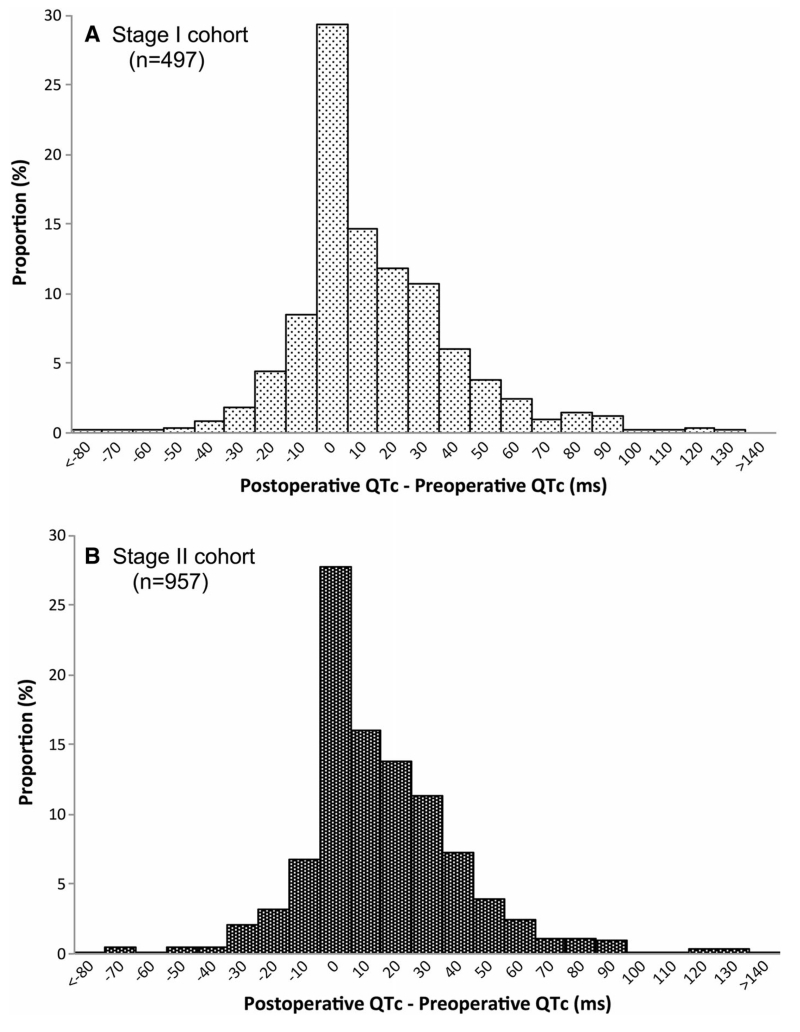
Fig. 1 shows the distribution of perioperative QTc interval changes (frequency distribution), and Fig. S1 depicts the distribution as a scatter plot (available as Electronic Supplementary Material). Postoperative QTc prolongation was observed in 151 (30.4%) and 255 (26.6%) patients in the stage I and stage II cohorts, respectively. Demographic, clinical, and procedural characteristics of the study cohorts are presented in Table 1. Of all factors univariably associated with postoperative QTc prolongation, female sex, preoperative left ventricular ejection fraction (LVEF), preoperative QTc prolongation, and duration of aortic cross-clamp remained independent clinical risk predictors in multivariable logistic regression analysis (Table 2). Preoperative and postoperative QTc interval durations were moderately correlated (Spearman r = 0.58, 95% confidence interval [CI], 0.54 to 0.62; P < 0.001), consistent with prolonged preoperative QTc being the strongest predictor of postoperative QTc prolongation. Although self-reported ethnicity was not statistically significant in either univariable or multivariable analyses (P = 0.59), it was included as a covariate in the final genetic association model in addition to the top six principal components identified in the population stratification analysis (Supplemental data Id, Table S4; available as Electronic Supplementary Material).
Fig. 1.
Frequency distributions of changes (in msec) from preoperative to postoperative QTc intervals in patients undergoing cardiac surgery. (A) stage I cohort (n = 497); (B) stage II cohort (n = 957)
Table 1.
Characteristics of the study populations by postoperative QTc duration
| Characteristics | Stage I cohort (n = 497) |
Stage II cohort (n = 957) |
||||||
|---|---|---|---|---|---|---|---|---|
| QTc ≤ 440 msec (n = 346) |
QTc > 440 msec (n = 151) |
OR (95% CI) | P value | QTc ≤ 440 msec (n = 702) |
QTc ≤ 440 msec (n = 255) |
OR (95% CI) | P value | |
| Demographic | ||||||||
| Age (yr), mean (SD) | 64.0 (11.3) | 66.1 (11.3) | 1.03 (1.01 to 1.04)a | 0.007 | 62.5 (11.0) | 65.2 (11.2) | 1.02 (1.01 to 1.04) | 0.001 |
| Female sex, n (%) | 90 (26.0) | 56 (37.1) | 1.67 (1.12 to 2.52) | 0.013 | 182 (25.9) | 104 (40.8) | 1.97 (1.46 to 2.66) | <0.0001 |
| Self-reported ethnicity, n (%) | 0.74 | 0.63 | ||||||
| White | 275 (79.5) | 124 (82.1) | 1.0 | 586 (83.5) | 211 (82.7) | 1.0 | ||
| Black | 61 (17.6) | 24 (15.9) | 0.87 (0.52 to 1.46) | 90 (12.8) | 37 (14.5) | 1.14 (0.76 to 1.73) | ||
| Other | 10 (2.9) | 3 (2.0) | 0.67 (0.18 to 2.46) | 26 (3.7) | 7 (2.7) | 0.75 (0.32 to 1.75) | ||
| Medical history, n (%) | ||||||||
| Chronic obstructive pulmonary disease | 44 (12.7) | 18 (11.9) | 0.93 (0.52 to 1.67) | 0.8 | 78 (11.1) | 34 (13.3) | 1.23 (0.80 to 1.89) | 0.35 |
| Chronic renal dysfunction | 25 (7.2) | 19 (12.6) | 1.85 (0.98 to 3.47) | 0.06 | 35 (5.0) | 24 (9.4) | 1.98 (1.15 to 3.40) | 0.01 |
| Diabetes mellitus | 114 (32.9) | 49 (32.5) | 0.98 (0.65 to 1.47) | 0.91 | 255 (32.1) | 84 (32.9) | 1.04 (0.77 to 1.41) | 0.8 |
| Extracardiac arteriopathy | 64 (18.5) | 25 (16.6) | 0.87 (0.53 to 1.45) | 0.6 | 119 (17.0) | 43 (16.9) | 0.99 (0.6 to −1.46) | 0.97 |
| Preoperative LVEF (%), mean (SD) | 51.4 (16.2) | 45.9 (16.5) | 0.98 (0.97 to 0.99)b | 0.0007 | 53.0 (14.0) | 48.5 (15.1) | 0.98 (0.97 to 0.99) | <0.0001 |
| Preoperative QTc > 440 msec | 22 (6.4) | 60 (39.7) | 9.71 (5.65 to 16.68) | <0.0001 | 30 (4.3) | 91 (35.7) | 12.43 (7.95 to 19.43) | <0.0001 |
| Medication use at hospital admission, n (%) | ||||||||
| Angiotensin-converting enzyme inhibitors | 168 (48.6) | 69 (45.7) | 0.89 (0.61 to 1.31) | 0.63 | 282 (40.2) | 116 (45.5) | 1.24 (0.93 to 1.66) | 0.15 |
| Acetylsalicylic Acid | 239 (69.1) | 101 (66.9) | 0.90 (0.60 to 1.36) | 0.63 | 512 (72.9) | 183 (71.8) | 0.94 (0.69 to 1.30) | 0.72 |
| Beta-blockers | 194 (56.1) | 90 (59.6) | 1.15 (0.78 to 1.70) | 0.46 | 428 (61.0) | 159 (62.4) | 1.06 (0.80 to 1.42) | 0.7 |
| Calcium channel blockers | 55 (15.9) | 24 (15.9) | 1.00 (0.59 to 1.69) | 1.0 | 113 (16.1) | 41 (16.1) | 0.99 (0.68 to 1.48) | 1.0 |
| Diuretics | 77 (22.3) | 48 (31.8) | 1.63 (1.06 to 2.49) | 0.02 | 143 (20.4) | 81 (31.8) | 1.82 (1.32 to 2.51) | 0.0003 |
| Nitrates | 105 (30.3) | 51 (33.8) | 1.17 (0.78 to 1.76) | 0.45 | 231 (32.9) | 90 (35.3) | 1.11 (0.82 to 1.50) | 0.49 |
| Statins | 154 (44.5) | 61 (40.4) | 0.85 (0.57 to 1.25) | 0.4 | 303 (43.2) | 103 (40.4) | 0.90 (0.67 to 1.19) | 0.44 |
| Procedural factors, n (%) | ||||||||
| Aortic cross-clamp time (min), mean (SD) | 69.9 (40.0) | 84.3 (44.7) | 1.008 (1.004 to l.0l)c | 0.001 | 66.0 (34.3) | 80.5 (45.4) | 1.01 (1.0 to 1.01) | <0.0001 |
| Procedure type | 0.004 | <0.0001 | ||||||
| Off-pump CABG | 4 (1.2) | 1 (0.7) | 0.38 (0.04 to 3.84) | 14 (2.0) | 6 (2.4) | 0.53 (0.18 to 1.58) | ||
| On-pump CABG | 296 (85.5) | 110 (72.8) | 0.57 (0.28 to 1.19) | 626 (89.2) | 190 (74.5) | 0.38 (0.22 to 0.65) | ||
| CABG and valve | 26 (7.5) | 27 (17.9) | 1.60 (0.66 to 3.86) | 31 (4.4) | 34 (13.3) | 1.36 (0.66 to 2.79) | ||
| Valve | 20 (5.8) | 13 (8.6) | 1.0 | 31 (4.4) | 25 (9.8) | 1.0 | ||
| Genotype, n (%) | ||||||||
| IL1B rs 16994 | 1.29 (0.98 to 1.7) | 0.07 | 1.33 (1.06 to 1.67) | 0.01 | ||||
| CC | 144 (42.5) | 49 (32.7) | 263 (39.0) | 77 (30.8) | ||||
| CT | 146 (43.1) | 75 (50.0) | 347 (51.4) | 140 (56.0) | ||||
| TT | 49 (14.5) | 26 (17.3) | 65 (9.6) | 33 (13.2) | ||||
| IL1B rs1143633 | 0.71 (0.51 to 0.99) | 0.04 | 0.75 (0.58 to 0.96) | 0.02 | ||||
| GG | 128 (53.6) | 62 (63.9) | 267 (49.3) | 106 (55.8) | ||||
| GA | 67 (28.0) | 25 (25.8) | 205 (37.8) | 72 (37.9) | ||||
| AA | 44 (18.4) | 10 (10.3) | 70 (12.9) | 12 (6.3) | ||||
Continuous variables presented as mean (SD), categorical variables as % frequencies. QTc = corrected QT interval; CABG = coronary artery bypass graft surgery; IL1B = interleukin-1β; LVEF = left ventricular ejection fraction; OR (95% CI) = univariate odds ratio (95% confidence interval) evaluating the odds in cases (prolonged postoperative QTc group) compared with the odds in controls. ORs for continuous variables reflect: a, per each additional year; b, per each additional LVEF percentage point; c, per each additional minute. ORs for self-reported ethnicity evaluate the odds of prolonged postoperative QTc in patients self-identified as black or of other ancestry compared with the odds in white patients as a reference group (set as 1). ORs for procedure type evaluate the odds of prolonged postoperative QTc in patients undergoing CABG or CABG/valve surgery compared with the odds in patients undergoing isolated valve surgery as a reference (set as 1)
Table 2.
Multivariable predictors of postoperative QTc prolongation in logistic regression models
| Model variables | Stage I Cohort (n = 497) |
Stage II Cohort (n = 957) |
Risk model performance* |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | Empirical P value† |
Model P value |
Model AIC | Model BIC | Model –2LLR | C-statistic | |
| I. Clinical Covariate Model Demographics | <0.0001 | 945.78 | 1,004.15 | 921.78 | 0.749 | |||||
| Age, yr | 1.01 (0.99 to 1.03) | 0.19 | 1.01 (1.00 to 1.03) | 0.12 | ||||||
| Female sex | 1.48 (0.92 to 2.40) | 0.11 | 1.79 (1.26 to 2.54) | 0.001 | ||||||
| Self-reported ethnicity | 0.59 | 0.77 | ||||||||
| Black vs White | 0.77 (0.42 to 1.40) | 0.90 (0.56 to 1.45) | ||||||||
| Other vs White | 0.62 (0.13 to 2.89) | 0.74 (0.28 to 1.98) | ||||||||
| Medical history | ||||||||||
| Preoperative LVEF, % | 0.99 (0.97 to 1.0) | 0.05 | 0.99 (0.98 to 0.99) | 0.02 | ||||||
| Preoperative QTc >440 msec | 7.78 (4.42 to 13.68) | <0.0001 | 9.72 (6.10 to 15.50) | <0.0001 | ||||||
| Medication use at hospital admission | ||||||||||
| Diuretics | 1.18 (0.72 to 1.94) | 0.52 | 1.25 (0.85 to 1.83) | 0.25 | ||||||
| Procedural factors | ||||||||||
| Aortic cross-clamp time, min | 1.0 (0.99 to 1.01) | 0.15 | 1.01 (1.0 to 1.01) | 0.04 | ||||||
| Procedure type | 0.66 | 0.06 | ||||||||
| Off-pump CABG vs valve | 0.98 (0.09 to 10.92) | 1.15 (0.33 to 4.02) | ||||||||
| On-pump CABG vs valve | 0.63 (0.27 to 1.48) | 0.50 (0.26 to 0.96) | ||||||||
| CABG and valve vs valve | 0.83 (0.30 to 2.30) | 0.68 (0.30 to 1.59) | ||||||||
| II. Clinico-genetic model by individual SNP‡ | ||||||||||
| IL1B (rs1143633) | 0.73 (0.49 to 1.08) | 0.1 | 0.71 (0.53 to 0.95) | 0.02 | 0.02 | <0.0001 | 704 | 782 | 670 | 0.771 |
| IL1B (rs16944) | 1.35 (0.98 to 1.85) | 0.06 | 1.31 (1.01 to 1.7) | 0.04 | 0.01 | <0.0001 | 916 | 998 | 882 | 0.762 |
| III. Clinico-genetic model with both SNPs added‡ | ||||||||||
| IL1B (rs1143633) and IL1B (rs16944) | <0.001 | 640 | 772 | 654 | 0.773 | |||||
QTc = corrected QT interval; AIC = Akaike information criterion; BIC = Bayesian information criterion; CABG = coronary artery bypass graft surgery; CI = confidence interval; LVEF = left ventricular ejection fraction; LLR = log likelihood ratio; OR = multivariate odds ratio; SNP = single nucleotide polymorphism
Based on stage II cohort analyses.
Permutation-adjusted (see Methods)
Final logistic regression model variables included female sex, LVEF, preoperative QTc prolongation, aortic cross-clamp time, procedure type, self-reported ethnicity, top six principal components from population stratification analysis, and SNP. SNPs in IL1RN and CAT were not significant after covariate adjustment (not shown)
Genetic effects on postoperative QTc interval duration
Among the 37 candidate SNPs examined, four were selected in stage I covariate and population stratification adjusted analysis (P ≤ 0.1 in additive inheritance models) for follow-up in stage II: rs16944 and rs1143633 in IL1B (interleukin-1β), rs315952 in IL1RN (interleukin-1 receptor antagonist), and rs769214 in CAT (catalase) (Table S5; available as Electronic Supplementary Material). Power calculations for stage I analyses are reported in Supplemental data IIc and Table S6 (available as Electronic Supplementary Material).
From the four SNPs tested in the stage II cohort, only rs16944 and rs1143633 in IL1B remained significant in covariate and permutation-adjusted analysis (Pemp= 0.01 and Pemp= 0.02, respectively, based on additive inheritance models, Table 2). Adding IL1B genotypes to the clinical logistic regression model improved the area under the receiver operating characteristic curve from 0.749-0.773 (P < 0.001) as well as global measures of model fit (Table 2), suggesting genetic regulation of inflammation as an independent contributor to postoperative QTc interval prolongation. Nevertheless, since the same genetic effects may contribute to inter-individual variability in both the natural preoperative QTc interval and in the acquired postoperative QTc interval in response to cardiac surgical perturbations, we further assessed associations of IL1B variants with preoperative QTc prolongation as well as with modelled postoperative QTc prolongation without adjustment for preoperative QTc. Neither IL1B variant was associated with preoperative QTc prolongation in either cohort analysis (data not shown). Removing preoperative QTc from the logistic regression model while retaining the other significant covariates yielded similar results for both rs1143633 (odds ratio [OR] = 0.38; P = 0.005) and rs16944 (OR = 1.31; P = 0.03).
We chose to dichotomize the postoperative QTc interval using a 440-msec threshold as the primary outcome variable to increase relevance for the practicing clinician. Even so, we conducted a secondary analysis using the trait as a continuous variable in order to avoid controversies in threshold selection for defining QTc prolongation, maximize power to detect subtle genetic effects, estimate genetic effect sizes on postoperative QTc duration, and allow comparisons with similar studies conducted in ambulatory populations. In a linear regression model of rank-transformed postoperative QTc interval duration in the stage II cohort, using the same covariate structure as for the binary outcome analysis, IL1B rs1143633 (recessive model) was associated with a postoperative QTc interval duration (beta = −0.2; P = 0.03), whereas rs16944 (dominant model) conferred a trend for an increased postoperative QTc duration (beta = 0.1; P = 0.06). The average genetic effect size –estimated as the difference between the mean postoperative QTc interval duration of rs1143633 AA vs GA+GG genotypes (recessive model)– was −6.9 msec (minor allele homozygotes AA had shorter postoperative QTc intervals). In contrast, for rs16944 TT+CT vs CC genotypes (dominant model), the difference between the mean postoperative QTc interval duration was 3.1 msec (minor allele T carriers had longer postoperative QTc intervals). Generally, these post hoc sensitivity analyses confirm the independent effects of IL1B variants on postoperative QTc.
The IL1B haplotype bearing both risk alleles (rs1143633G and rs16944T), present in 32% of controls and 40% of cases, was associated with increased risk for postoperative QTc prolongation (OR = 1.57; 95% CI, 1.14 to 2.18; P = 0.006) (Table 3). Haplotype analyses were also conducted for the two IL1B markers (rs1143633 and rs16944) and an IL1RN marker (rs315952), considering that both genes are located in the same IL1 gene cluster and cytokine family and that the balance between IL-1 and its specific receptor antagonist IL-1Ra is ultimately determining IL-1 bioactivity. The G-T-T haplotype of rs1143633-rs16944-rs315952 (26% of controls, 31% of cases) was associated with postoperative QTc prolongation (OR = 1.95; 95% CI, 1.24 to 3.40; P = 0.018) (Table 3).
Table 3.
Associations of IL1B and IL1RN haplotypes with postoperative QTc interval prolongation
| IL1B | IL1B | IL1RN | Haplotype | Haplotype frequency in controls |
Haplotype frequency in cases |
‡OR (95% CI) | P value |
|---|---|---|---|---|---|---|---|
| rs1143633 | rs16944 | A-C* | 0.30 | 0.25 | 1.0 | ||
| rs1143633 | rs16944 | A-T | 0.03 | 0.008 | 0.24 (0.05 to 1.26) | 0.091 | |
| rs1143633 | rs16944 | G-C | 0.35 | 0.33 | 1.12 (0.79 to 1.59) | 0.523 | |
| rs1143633 | rs16944 | G-T | 0.32 | 0.40 | 1.57 (1.14 to 2.18) | 0.006 | |
| rs1143633 | rs16944 | rs315952 | A-C-C* | 0.12 | 0.09 | 1.0 | |
| rs1143633 | rs16944 | rs315952 | A-C-T | 0.17 | 0.16 | 1.43 (0.68 to 2.97) | 0.344 |
| rs1143633 | rs16944 | rs315952 | A-T-C** | – | – | – | – |
| rs1143633 | rs16944 | rs315952 | A-T-T | 0.03 | 0.009 | 0.35 (0.07 to 1.85) | 0.217 |
| rs1143633 | rs16944 | rs315952 | G-C-C | 0.12 | 0.10 | 1.41 (0.64 to 3.09) | 0.397 |
| rs1143633 | rs16944 | rs315952 | G-C-T | 0.23 | 0.23 | 1.40 (0.80 to 2.46) | 0.234 |
| rs1143633 | rs16944 | rs315952 | G-T-C | 0.07 | 0.09 | 1.92 (0.87 to 4.28) | 0.109 |
| rs1143633 | rs16944 | rs315952 | G-T-T | 0.26 | 0.31 | 1.95 (1.24 to 3.40) | 0.018 |
QTc = corrected QT interval;
Haplotypes “A-C” and “A-C-C” were used as reference, respectively.
The frequency of “A-T-C” haplotype was too low (not reported).
Adjusted for female sex, left ventricular ejection fraction, preoperative QTc prolongation, aortic cross-clamp time, procedure type, and self-reported ethnicity
Genetic effects on IL-1β production following cardiac surgery
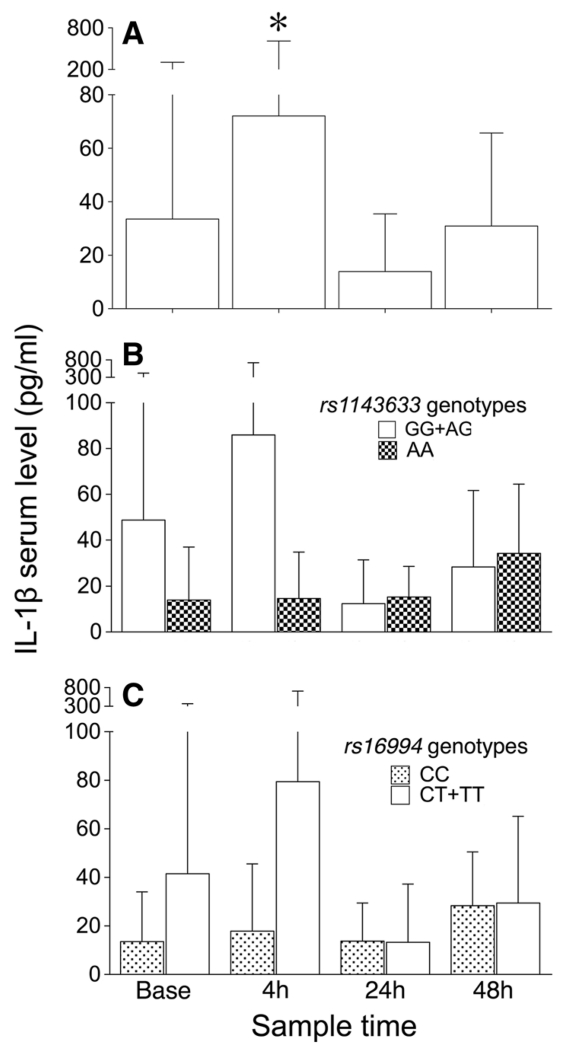
To provide functional evidence in support of the observed associations between IL1B polymorphisms and postoperative QTc prolongation, we conducted a time-course analysis of perioperative changes in serum IL-1β and assessed genotypic effects of the two SNPs of interest on circulating IL-1β levels in response to cardiac surgery as an intermediate molecular phenotype. Serum IL-1β and IL-1Ra showed large inter-individual variability and increased at four hours post aortic cross-clamp release relative to preoperative values [Fig. 2A; (Fig. S2; available as Electronic Supplementary Material); P < 0.001]. Autoregressive models using generalized estimating equations identified significant changes over time in the serum levels of IL-1β by IL1B genotypes. For rs1143633, lower change rates were observed in AA homozygotes compared with AG and GG genotype carriers (recessive model, P = 0.04; Fig. 2B). In contrast, for rs16994, higher change rates were observed in CT and TT genotype carriers compared with CC homozygotes (dominant model, P = 0.03; Fig. 2C), consistent with the allelic effects seen on the electrocardiographic trait.
Fig. 2.
Perioperative serum concentrations of interleukin-1β in patients undergoing cardiac surgery (n = 288). Sampling time points are preoperative (base), and four hours, 24 hr, and 48 hr after aortic cross-clamp removal. Results at each time point are expressed as mean (SD) in all patients (A), stratified by IL1B rs1143633 genotypes (homozygous major allele GG and heterozygous AG vs homozygous minor allele AA carriers, recessive inheritance model) (B), and by IL1B rs16994 genotypes (homozygous minor allele TT and heterozygous CT vs homozygous major allele CC carriers, dominant inheritance pattern) (C). Patients carrying either the rs1143633G allele and/or the rs16994T allele are high interleukin-1β producers. *P < 0.01 difference compared with preoperative (base) values. IL = interleukin
Since the bioactivity of IL-1β also involves the antagonistic effects of IL-1Ra, we assessed the association of IL1B and IL1RN genotypes on IL1-Ra circulating levels and found no significant differences (data not shown). Together, these preliminary results suggest that the IL1B variants specifically alter the amount of bioactive IL-1β produced in response to cardiac surgery.
Risk prediction of postoperative QTc prolongation
Given the limitations of using receiver operating characteristic curve analysis to assess the added usefulness of new biomarkers in risk prediction models, we used reclassification analysis to quantify improvement offered by the two IL1B genetic markers beyond clinical and procedural factors.39,40 Overall, the ability of a clinico-genetic risk model that included rs16994 and rs1143633 to reclassify patients at risk for developing postoperative QTc prolongation improved significantly, with a net reclassification improvement of 0.308 (P = 0.0003) and an integrated discrimination improvement of 0.02 (P = 0.000024) compared with the clinical covariate risk model. We then examined reclassification of individual patients between risk categories by addition of the ILB1 genotypes. Seven hundred seven patients had non-missing genotype information at both SNPs (187 with prolonged postoperative QTc and 520 with normal postoperative QTc). Of the 187 patients with postoperative QTc prolongation, 109 were correctly reclassified as being high risk, while 78 were incorrectly reclassified as being low risk. Conversely, of the 520 patients with normal postoperative QTc, 297 were correctly reclassified, while 223 were incorrectly reclassified (Supplemental data IId; available as Electronic Supplementary Material). For the entire cohort, this translates to 57.4% ([109+297]/707) of patients correctly reclassified, offset by the 42.6% ([78+223]/707) of patients incorrectly reclassified, for a net of 14.8% of patients correctly reclassified after adding genetic information.
Postoperative QTc prolongation and adverse clinical outcomes
Although the current study was not powered to assess hard clinical endpoints, we conducted further exploratory analyses evaluating the association of postoperative QTc interval prolongation with the incidence of postoperative ventricular arrhythmias and perioperative (30-day), one-year, and five-year mortality. Complete long-term follow-up survival information was available in 859 of the 957 patients. Postoperative ventricular arrhythmias occurred in 43 of the 859 patients (5.01%). Mortality rates at 30 days, one year, and five years were 1.63%, 5.12%, and 19.32%, respectively. Postoperative QTc prolongation was not associated with the incidence of postoperative ventricular arrhythmias or perioperative mortality. Nevertheless, a trend for increased one-year mortality and a significant increase in five-year mortality were seen in patients with prolonged postoperative QTc intervals, after adjustment for EuroSCORE (Table 4). In post hoc analyses, there was no significant association between the IL1B polymorphisms studied and the incidence of postoperative ventricular arrhythmias or any of the mortality endpoints (data not shown).
Table 4.
Association of postoperative QTc prolongation with major adverse events
| Outcome | QTc ≤ 440 msec (n = 644) |
QTc > 440 msec (n = 215) |
OR (95%CI)* | P value* |
|---|---|---|---|---|
| Postoperative ventricular arrhythmias | 31 (4.8) | 12 (5.6) | 1.17 (0.6 to 2.32) | 0.65 |
| 30-day mortality | 10 (1.6) | 4 (1.9) | 1.2 (0.37 to 3.87)** | 0.76** |
| 1-yr mortality | 28 (4.4) | 16 (7.4) | 1.76 (0.94 to 3.34)** | 0.07** |
| 5-yr mortality | 109 (16.9) | 57 (26.5) | 1.77 (1.22 to 2.55)** | 0.003** |
Variables are presented as n (%). QTc = corrected QT interval; OR (95% CI) = odds ratio (95% confidence interval)
From logistic regression models;
Adjusted for EuroSCORE
Discussion
In this retrospective analysis of patients undergoing cardiac surgery, we show that early postoperative QTc prolongation is frequent and can be predicted by clinical and procedural risk factors as well as genetic polymorphisms in IL-1β. These findings add to previous epidemiological studies suggesting that genetic regulation of inflammation may be a potentially clinically useful determinant of the risk for perioperative cardiovascular morbidity after cardiac surgery.23,32,41
Interleukin-1β is a strong candidate for the observed association. As a major pro-inflammatory cytokine and powerful inducer of innate immune responses, IL-1β has been implicated in the pathogenesis of myocardial dysfunction following ischemia-reperfusion associated with cardiac surgery,41,42 cardiac remodelling after acute myocardial infarction, and heart failure progression.43 Tissue injury leads to a local surge of IL-1β, which significantly amplifies the inflammatory response by recruiting additional inflammatory cells, inducing the expression of secondary inflammatory mediators, and stimulating metalloproteinase activities. This ultimately results in inflammatory cell death. Moreover, preclinical studies have identified pro-arrhythmic effects of IL-1β, which mechanistically involve increased spontaneous Ca2+ leak from the sarcoplasmic reticulum, leading to cell depolarization.44
Two SNPs in IL1B were associated with postoperative QTc prolongation. The minor alleles of these SNPs had opposite phenotypic effects; the rs1143633A allele was associated with decreased risk, while the rs16944T allele was associated with increased risk for postoperative QTc prolongation. The rs1143633 polymorphism, located within an intron of the IL1B gene (IL1B 5810G/A), is likely to influence messenger ribonucleic acid (RNA) stability, resulting in lower IL1B gene expression and an attenuated inflammatory response45 to cardiac surgery in carriers. Conversely, the rs16944 polymorphism is located within the promoter region of IL1B gene (IL1B -511 C/T) and has been associated with increased inter-individual variability in inflammatory responses.45,46 The functional consequences of rs16944, however, have been inconsistent across studies. Some studies reported pro-inflammatory effects (increased IL-1β production)46-48 and genetic epidemiological associations with increased severity of inflammatory conditions, including atherosclerotic plaque formation49 and systolic hypertension.50 Other studies found associations with a lower risk for myocardial infarction and ischemic stroke45 and reduced susceptibility to sepsis after severe trauma.51 It was therefore important to assess the effects of these polymorphisms on changes in circulating levels of IL-1β, specifically in response to cardiac surgical stress. First, we confirmed previous reports of significant increases in IL-1β in the early (four hours) postoperative period.52 Furthermore, we provided new preliminary functional evidence that perioperative hourly changes in IL-1β serum levels were higher in carriers of the rs16944T allele and lower in carriers of the rs1143633A allele. Inclusion of an intermediate phenotype showing allele-specific differences in perioperative IL-1β production adds biological plausibility and strengthens the observed genetic association with the electrocardiographic phenotype.
We also found that haplotypes bearing the high IL-1β expressing alleles (rs1143633G and rs16944T) and the IL1RN risk allele (rs315952T) cumulatively conferred further increased risk for postoperative QTc prolongation compared with individual genotype effects. This finding is consistent with increasing evidence suggesting that the balance between IL-1β and its naturally occurring antagonist IL-1Ra plays an essential role in a variety of cardiovascular conditions.
For the practicing anesthesiologist, identification of patients at risk for developing adverse postoperative outcomes remains an unmet clinical need. Risk prediction models are essential tools in the era of precision medicine, providing estimates of prognostic outcomes that can ultimately support decision-making in screening, perioperative optimization, or choice of therapy. Nevertheless, a systematic and rigorous evaluation of the clinical validity and utility for using genetic tests to improve risk prediction is required prior to their adoption in healthcare decision-making. A particular focus is placed on the “actionability” of specific genetic variants – in this context, whether the identification of a variant preoperatively in an asymptomatic individual can lead to an intervention that can mitigate harm before a clinical diagnosis is made.53 To put the perioperative genotypic effect sizes observed in this study into perspective, other common genetic variants have been associated with modest increments (1-4 msec·allele−1) in the duration of the QTc interval in ambulatory populations.13,14 In contrast, the Food and Drug Administration mandates that the threshold level of regulatory concern is a mean increase in QTc duration of >5 msec in response to a drug in healthy volunteers, triggering an expanded safety evaluation during the later stages of drug development.28 Nevertheless, the impact of any such increases in QTc duration on clinical risk/benefit decisions for an individual patient remains to be determined.54 In evaluating the predictive ability of IL1B genetic polymorphisms, we found minimal increases in the area under the receiver operating characteristic curve compared with the clinical risk model, similar to many previous biomarker studies.55 We therefore used reclassification analysis to quantify the prediction increment of the clinico-genetic model more effectively. The net reclassification improvement of 14.8% of patients observed here is consistent with existing reports evaluating the contribution of novel biomarkers on cardiovascular events56 or prediction of the risk of perioperative morbidity.22,25,57
Although we found a contribution of common genetic variants in IL1B, individually and in aggregate, on postoperative QTc interval prolongation, and confirmed the relationship between QTc prolongation and long-term mortality following cardiac surgery, we found no association between QTc prolongation and the occurrence of postoperative ventricular arrhythmias or perioperative death. Our limited analysis does not address the potential contributions of incomplete revascularization or heart failure on the observed long-term mortality. Furthermore, in post hoc analyses, there was no evidence in our cohort of a relationship between genetic variation in IL1B and the above-mentioned postoperative clinical endpoints. This is consistent with similar analyses in ambulatory populations58 and may result from lack of power, lack of true effect, or because not all QT-prolonging alleles actually increase mortality risk. Considering that the study objective was to assess the contribution of cardiac surgical stress and genetic susceptibility to postoperative QTc prolongation, we minimized confounding factors by excluding patients who required postoperative pacing, developed perioperative bundle branch block or atrial fibrillation/flutter, and importantly received major known culprits for drug-induced QTc prolongation (with the notable exception of volatile anesthetics and inotropes). This amounted to almost 10% of the initial cohort, further limiting the power to detect associations with genetic variants, postoperative QTc prolongation, and relatively infrequent hard clinical endpoints.
Several additional limitations should be kept in mind when interpreting these findings. Our analysis set is dated and institution-specific, thus generalizability to a more contemporary cardiac surgical population could be limited. Nevertheless, this study cohort reflects clinical practice at a tertiary academic medical centre and substantially differs from contemporary cardiac surgical cohorts at our institution only with respect to surgical indications (e.g., increased proportions of valve, coronary artery bypass grafting combined with valve procedures, and advanced heart failure therapies/assist devices). This study cohort does not differ substantially with respect to anesthetic (fentanyl/volatile anesthesia, similar use of pulmonary artery catheters) and perfusion management. The focus of our investigation was on the epidemiology of acute perioperative QTc interval prolongation following cardiac surgery and, as such, lacked a control group of patients undergoing percutaneous revascularization or major non-cardiac surgery. Moreover, the observation period for assessing the QTc interval was narrow, restricted to the first postoperative day. Power calculations show that, based on the current sample size and the incidence of postoperative QTc prolongation, our study is powered to detect only very common genetic variants with relatively large effect sizes.
In conclusion, high IL-1β expressing genotypes are independently associated with alterations in myocardial repolarization reserve that acted in response to perioperative stressors occurring during cardiac surgery. If validated in independent studies, the IL1B variants reported herein could potentially assist the practicing clinician. Conceivably, they could inform prescription behaviour in the perioperative period (e.g., more strict avoidance of QTc-prolonging drugs) and could foster preemptive use of targeted antiinflammatory therapies in patients predicted to be high IL-1β producers in response to surgery (especially given involvement of IL-1β in the pathogenesis of other perioperative complications).43,59 Furthermore, they could facilitate targeting perioperative QTc surveillance, provision of critical care, and selection of high-risk patients who would benefit from an implantable cardioverter-defibrillator.60 Although these findings are based on a cohort of cardiac surgical patients, postoperative QTc prolongation is also a significant concern for patients undergoing non-cardiac surgery, and thus, the clinical implications for risk prediction may have broader ramifications for perioperative medicine.
Supplementary Material
Acknowledgements
We sincerely thank the members of the Duke Perioperative Genetics and Safety Outcomes (PEGASUS) Investigative Team for their contributions to the conduct of this study: Cooter Mary, Danseshmand Mani, Funk Bonita, Gaca Jeffrey G, Ghadimi Kamrouz, Ginsburg Geoffrey S, Glower Donald D, Hauser Elizabeth, Jones Robert, Kertai Miklos D, Laskowitz Daniel T, Li Yi-Ju, Lodge Andrew J, Mathew Joseph P, Milano Carmelo A, Newman Mark F, Quinones Quintin J, Podgoreanu Mihai V, Schroder Jacob, Smith Michael P, Smith Peter K, Stafford-Smith Mark, Swaminathan Madhav, Welsby Ian J.
Financial support This work was supported in part by National Institutes of Health grants R01-HL075273 and R01-HL092071 (to Dr. Podgoreanu) and by American Heart Association grants 0256342U and 9951185U (to Dr. Mathew) and 0120492U (to Dr. Podgoreanu).
Footnotes
Author contributions: Mihai V. Podgoreanu contributed to the conception and design of the study. Miklos D. Kertai, Yi-Ju Li, Yunqi Ji, and Mihai V. Podgoreanu contributed to the data analysis. Miklos D. Kertai, Yi-Ju Li, and Mihai V. Podgoreanu contributed to data interpretation. Miklos D. Kertai and Yunqi Ji drafted the manuscript. Yi-Ju Li contributed to the critical revision of the manuscript. Yunqi Ji contributed to data quality control. Joseph P. Mathew, James P. Daubert, and Mihai V. Podgoreanu contributed to data acquisition and critical review of the manuscript.
Conflicts of interest None declared.
Contributor Information
Miklos D. Kertai, Duke Perioperative Genomics Program, Department of Anesthesiology, Duke University Medical Center, DUMC 3094, Durham, NC 27710, USA.
Yunqi Ji, Department of Biostatistics and Bioinformatics, Duke Molecular Physiology Institute, Duke University Medical Center, Durham, NC, USA.
Yi-Ju Li, Duke Perioperative Genomics Program, Department of Anesthesiology, Duke University Medical Center, DUMC 3094, Durham, NC 27710, USA; Department of Biostatistics and Bioinformatics, Duke Molecular Physiology Institute, Duke University Medical Center, Durham, NC, USA.
Joseph P. Mathew, Duke Perioperative Genomics Program, Department of Anesthesiology, Duke University Medical Center, DUMC 3094, Durham, NC 27710, USA.
James P. Daubert, Clinical Electrophysiology Section, Department of Medicine, Duke University Medical Center, Durham, NC, USA; Duke Clinical Research Institute, Durham, NC 27710, USA.
Mihai V. Podgoreanu, Duke Perioperative Genomics Program, Department of Anesthesiology, Duke University Medical Center, DUMC 3094, Durham, NC 27710, USA; Duke Clinical Research Institute, Durham, NC 27710, USA.
References
- 1.Roden DM. Clinical practice. Long-QT syndrome. N Engl J Med. 2008;358:169–76. doi: 10.1056/NEJMcp0706513. [DOI] [PubMed] [Google Scholar]
- 2.Myredal A, Karlsson AK, Johansson M. Elevated temporal lability of myocardial repolarization after coronary artery bypass grafting. J Electrocardiol. 2008;41:698–702. doi: 10.1016/j.jelectrocard.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 3.Cagli K, Ozbakir C, Ergun K, Bakuy V, Circi R, Circi P. Electrocardiographic changes after coronary artery surgery. Asian Cardiovasc Thorac Ann. 2006;14:294–9. doi: 10.1177/021849230601400406. [DOI] [PubMed] [Google Scholar]
- 4.Punn R, Lamberti JJ, Balise RR, Seslar SP. QTc prolongation in children following congenital cardiac disease surgery. Cardiology Young. 2011;21:400–10. doi: 10.1017/S1047951111000175. [DOI] [PubMed] [Google Scholar]
- 5.Nagele P, Pal S, Brown F, Blood J, Miller JP, Johnston J. Postoperative QT interval prolongation in patients undergoing noncardiac surgery under general anesthesia. Anesthesiology. 2012;117:321–8. doi: 10.1097/ALN.0b013e31825e6eb3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Johnston J, Pal S, Nagele P. Perioperative torsade de pointes: a systematic review of published case reports. Anesth Analg. 2013;117:559–64. doi: 10.1213/ANE.0b013e318290c380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vrtovec B, Ryazdanbakhsh AP, Pintar T, Collard CD, Gregoric ID, Radovancevic B. QTc interval prolongation predicts postoperative mortality in heart failure patients undergoing surgical revascularization. Tex Heart Inst J. 2006;33:3–8. [PMC free article] [PubMed] [Google Scholar]
- 8.Kinoshita T, Asai T, Suzuki T, Matsubayashi K, Horie M. Time course and prognostic implications of QT interval in patients with coronary artery disease undergoing coronary bypass surgery. J Cardiovasc Electrophysiol. 2012;23:645–9. doi: 10.1111/j.1540-8167.2011.02244.x. [DOI] [PubMed] [Google Scholar]
- 9.Straus SM, Kors JA, De Bruin ML, et al. Prolonged QTc interval and risk of sudden cardiac death in a population of older adults. J Am Coll Cardiol. 2006;47:362–7. doi: 10.1016/j.jacc.2005.08.067. [DOI] [PubMed] [Google Scholar]
- 10.Bonnemeier H, Hartmann F, Wiegand UK, Bode F, Katus HA, Richardt G. Course and prognostic implications of QT interval and QT interval variability after primary coronary angioplasty in acute myocardial infarction. J Am Coll Cardiol. 2001;37:44–50. doi: 10.1016/s0735-1097(00)01061-5. [DOI] [PubMed] [Google Scholar]
- 11.Zhang Y, Post WS, Blasco-Colmenares E, Dalal D, Tomaselli GF, Guallar E. Electrocardiographic QT interval and mortality: a meta-analysis. Epidemiology. 2011;22:660–70. doi: 10.1097/EDE.0b013e318225768b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Roden DM. Long QT syndrome: reduced repolarization reserve and the genetic link. J Intern Med. 2006;259:59–69. doi: 10.1111/j.1365-2796.2005.01589.x. [DOI] [PubMed] [Google Scholar]
- 13.Newton-Cheh C, Eijgelsheim M, Rice KM, et al. Common variants at ten loci influence QT interval duration in the QTGEN Study. Nat Genet. 2009;41:399–406. doi: 10.1038/ng.364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pfeufer A, Sanna S, Arking DE, et al. Common variants at ten loci modulate the QT interval duration in the QTSCD Study. Nat Genet. 2009;41:407–14. doi: 10.1038/ng.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sauer AJ, Newton-Cheh C. Clinical and genetic determinants of torsade de pointes risk. Circulation. 2012;125:1684–94. doi: 10.1161/CIRCULATIONAHA.111.080887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kannankeril P, Roden DM, Darbar D. Drug-induced long QT syndrome. Pharmacol Rev. 2010;62:760–81. doi: 10.1124/pr.110.003723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Smithburger PL, Seybert AL, Armahizer MJ, Kane-Gill SL. QT prolongation in the intensive care unit: commonly used medications and the impact of drug-drug interactions. Expert Opin Drug Saf. 2010;9:699–712. doi: 10.1517/14740331003739188. [DOI] [PubMed] [Google Scholar]
- 18.Scuderi PE. Sevoflurane and QTc prolongation: an interesting observation, or a clinically significant finding? Anesthesiology. 2010;113:772–5. doi: 10.1097/ALN.0b013e3181f2b088. [DOI] [PubMed] [Google Scholar]
- 19.Podgoreanu MV, Schwinn DA. New paradigms in cardiovascular medicine: emerging technologies and practices: perioperative genomics. J Am Coll Cardiol. 2005;46:1965–77. doi: 10.1016/j.jacc.2005.08.040. [DOI] [PubMed] [Google Scholar]
- 20.Zuo Y, Zou G, Wang J, Zhao H, Liang H. Optimal two-stage design for case-control association analysis incorporating genotyping errors. Ann Hum Genet. 2008;72:375–87. doi: 10.1111/j.1469-1809.2007.00419.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Little J, Higgins JP, Ioannidis JP, et al. STrengthening the REporting of Genetic Association studies (STREGA): an extension of the STROBE Statement. Ann Intern Med. 2009;150:206–15. doi: 10.7326/0003-4819-150-3-200902030-00011. [DOI] [PubMed] [Google Scholar]
- 22.Lobato RL, White WD, Mathew JP, et al. Thrombomodulin gene variants are associated with increased mortality after coronary artery bypass surgery in replicated analyses. Circulation. 2011;124(11 Suppl):S143–8. doi: 10.1161/CIRCULATIONAHA.110.008334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Podgoreanu MV, White WD, Morris RW, et al. Inflammatory gene polymorphisms and risk of postoperative myocardial infarction after cardiac surgery. Circulation. 2006;114(1 Suppl):I275–81. doi: 10.1161/CIRCULATIONAHA.105.001032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stafford-Smith M, Li YJ, Mathew JP, et al. Genome-wide association study of acute kidney injury after coronary bypass graft surgery identifies susceptibility loci. Kidney Int. 2015;66:623–32. doi: 10.1038/ki.2015.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kertai MD, Li YJ, Li YW, et al. Genome-wide association study of perioperative myocardial infarction after coronary artery bypass surgery. BMJ Open. 2015;5:e006920. doi: 10.1136/bmjopen-2014-006920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kertai MD, Li YW, Li YJ, et al. G protein-coupled receptor kinase 5 gene polymorphisms are associated with postoperative atrial fibrillation after coronary artery bypass grafting in patients receiving beta-blockers. Circ Cardiovasc Genet. 2014;7:625–33. doi: 10.1161/CIRCGENETICS.113.000451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bazett HC. An analysis of the time-relations of electrocardiograms. Heart. 1920;7:353–70. [Google Scholar]
- 28.U.S. Department of Health and Human Services. Food and Drug Administration. Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation and Research (CBER) [accessed October 2015];Guidance for Industry. E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs - October 2005. Available from URL: http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm073153.pdf.
- 29.Anderson ME, Al-Khatib SM, Roden DM, Califf RM. Duke Clinical Research Institute/American Heart Journal Expert Meeting on Repolarization Changes. Cardiac repolarization: current knowledge, critical gaps, and new approaches to drug development and patient management. Am Heart J. 2002;144:769–81. doi: 10.1067/mhj.2002.125804. [DOI] [PubMed] [Google Scholar]
- 30.Newton-Cheh C, Shah R. Genetic determinants of QT interval variation and sudden cardiac death. Current Opin Genet Develop. 2007;17:213–21. doi: 10.1016/j.gde.2007.04.010. [DOI] [PubMed] [Google Scholar]
- 31.Roden DM, Viswanathan PC. Genetics of acquired long QT syndrome. J Cliin Invest. 2005;115:2025–32. doi: 10.1172/JCI25539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tomic V, Russwurm S, Moller E, et al. Transcriptomic and proteomic patterns of systemic inflammation in on-pump and off-pump coronary artery bypass grafting. Circulation. 2005;112:2912–20. doi: 10.1161/CIRCULATIONAHA.104.531152. [DOI] [PubMed] [Google Scholar]
- 33.Ruel M, Bianchi C, Khan TA, et al. Gene expression profile after cardiopulmonary bypass and cardioplegic arrest. J Thorac Cardiovasc Surg. 2003;126:1521–30. doi: 10.1016/s0022-5223(03)00969-3. [DOI] [PubMed] [Google Scholar]
- 34.Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38:904–9. doi: 10.1038/ng1847. [DOI] [PubMed] [Google Scholar]
- 35.Skol AD, Scott LJ, Abecasis GR, Boehnke M. Optimal designs for two-stage genome-wide association studies. Genet Epidemiol. 2007;31:776–88. doi: 10.1002/gepi.20240. [DOI] [PubMed] [Google Scholar]
- 36.Good P. PermutationTests: A Practical Guide to Resampling Methods for Testing Hypotheses. Springer; New York: 2000. [Google Scholar]
- 37.Nashef SA, Roques F, Hammill BG, et al. Validation of European System for Cardiac Operative Risk Evaluation (EuroSCORE) in North American cardiac surgery. Eur J Cardiothorac Surg. 2002;22:101–5. doi: 10.1016/s1010-7940(02)00208-7. [DOI] [PubMed] [Google Scholar]
- 38.Lange C, Whittaker JC. Mapping quantitative trait Loci using generalized estimating equations. Genetics. 2001;159:1325–37. doi: 10.1093/genetics/159.3.1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cook NR. Use and misuse of the receiver operating characteristic curve in risk prediction. Circulation. 2007;115:928–35. doi: 10.1161/CIRCULATIONAHA.106.672402. [DOI] [PubMed] [Google Scholar]
- 40.Pencina MJ, D’Agostino RB, Sr, Steyerberg EW. Extensions of net reclassification improvement calculations to measure usefulness of new biomarkers. Stat Med. 2011;30:11–21. doi: 10.1002/sim.4085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Paparella D, Yau TM, Young E. Cardiopulmonary bypass induced inflammation: pathophysiology and treatment. An update. Eur J Cardiothorac Surg. 2002;21:232–44. doi: 10.1016/s1010-7940(01)01099-5. [DOI] [PubMed] [Google Scholar]
- 42.Cain BS, Meldrum DR, Dinarello CA, et al. Tumor necrosis factor-alpha and interleukin-1beta synergistically depress human myocardial function. Crit Care Med. 1999;27:1309–18. doi: 10.1097/00003246-199907000-00018. [DOI] [PubMed] [Google Scholar]
- 43.Van Tassell BW, Toldo S, Mezzaroma E, Abbate A. Targeting interleukin-1 in heart disease. Circulation. 2013;128:1910–23. doi: 10.1161/CIRCULATIONAHA.113.003199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Duncan DJ, Yang Z, Hopkins PM, Steele DS, Harrison SM. TNF-alpha and IL-1βeta increase Ca2+ leak from the sarcoplasmic reticulum and susceptibility to arrhythmia in rat ventricular myocytes. Cell Calcium. 2010;47:378–86. doi: 10.1016/j.ceca.2010.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Iacoviello L, Di Castelnuovo A, Gattone M, et al. Polymorphisms of the interleukin-1beta gene affect the risk of myocardial infarction and ischemic stroke at young age and the response of mononuclear cells to stimulation in vitro. Arterioscler Thromb Vasc Biol. 2005;25:222–7. doi: 10.1161/01.ATV.0000150039.60906.02. [DOI] [PubMed] [Google Scholar]
- 46.Hall SK, Perregaux DG, Gabel CA, et al. Correlation of polymorphic variation in the promoter region of the interleukin-1 beta gene with secretion of interleukin-1 beta protein. Arthritis Rheum. 2004;50:1976–83. doi: 10.1002/art.20310. [DOI] [PubMed] [Google Scholar]
- 47.El-Omar EM, Carrington M, Chow WH, et al. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature. 2000;404:398–402. doi: 10.1038/35006081. [DOI] [PubMed] [Google Scholar]
- 48.Eklund C, Jahan F, Pessi T, Lehtimaki T, Hurme M. Interleukin 1B gene polymorphism is associated with baseline C-reactive protein levels in healthy individuals. Eur Cytokine Netw. 2003;14:168–71. [PubMed] [Google Scholar]
- 49.Kornman KS, Pankow J, Offenbacher S, Beck J, di Giovine F, Duff GW. Interleukin-1 genotypes and the association between periodontitis and cardiovascular disease. J Periodontal Res. 1999;34:353–7. doi: 10.1111/j.1600-0765.1999.tb02265.x. [DOI] [PubMed] [Google Scholar]
- 50.Huang G, Niu T, Peng SJ, et al. Association between the interleukin-1 beta C(-511)T polymorphism and blood pressure in a Chinese hypertensive population. Immunol Lett. 2004;91:159–62. doi: 10.1016/j.imlet.2003.11.009. [DOI] [PubMed] [Google Scholar]
- 51.Wen AQ, Gu W, Wang J, et al. Clinical relevance of IL-1βeta promoter polymorphisms (−1470, −511, and −31) in patients with major trauma. Shock. 2010;33:576–82. doi: 10.1097/SHK.0b013e3181cc0a8e. [DOI] [PubMed] [Google Scholar]
- 52.Roth-Isigkeit A, Hasselbach L, Ocklitz E, et al. Inter-individual differences in cytokine release in patients undergoing cardiac surgery with cardiopulmonary bypass. Clin Exp Immunol. 2001;125:80–8. doi: 10.1046/j.1365-2249.2001.01521.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Institute of Medicine of the National Academies . Assessing Genomic Sequencing Information for Health Care Decision Making: Workshop Summary. The National Academies Press; Washington, DC: 2014. [PubMed] [Google Scholar]
- 54.Institute of Medicine of the National Academies . Accelerating the Development of Biomarkers for Drug Safety: Workshop Summary. The National Academies Press; Washington, DC: 2009. [PubMed] [Google Scholar]
- 55.Pencina MJ, D’Agostino RB, Sr, D’Agostino RB, Jr, Vasan RS. Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med. 2008;27:1, 57–72. doi: 10.1002/sim.2929. discussion 207-12. [DOI] [PubMed] [Google Scholar]
- 56.Gulati A, Jabbour A, Ismail TF, et al. Association of fibrosis with mortality and sudden cardiac death in patients with nonischemic dilated cardiomyopathy. JAMA. 2013;309:896–908. doi: 10.1001/jama.2013.1363. [DOI] [PubMed] [Google Scholar]
- 57.Parikh CR, Coca SG, Thiessen-Philbrook H, et al. Postoperative biomarkers predict acute kidney injury and poor outcomes after adult cardiac surgery. J Am Soc Nephrol. 2011;22:1748–57. doi: 10.1681/ASN.2010121302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Noseworthy PA, Havulinna AS, Porthan K, et al. Common genetic variants, QT interval, and sudden cardiac death in a Finnish population-based study. Circ Cardiovasc Genet. 2011;4:305–11. doi: 10.1161/CIRCGENETICS.110.959049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dinarello CA, Simon A, van der Meer JW. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov. 2012;11:633–52. doi: 10.1038/nrd3800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fazal IA, Bates MG, Matthews IG, Turley AJ. Do implantable cardioverter defibrillators improve survival in patients with severe left ventricular systolic dysfunction after coronary artery bypass graft surgery? Interact Cardiovasc Thorac Surg. 2011;12:1010–6. doi: 10.1510/icvts.2010.259465. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.