Abstract
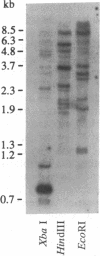
The initial step in the conversion of the isoprenoid intermediate farnesyl diphosphate to the sesquiterpenoid phytoalexin capsidiol in elicitor-treated tobacco tissues is catalyzed by an inducible sesquiterpene cyclase [5-epi-aristolochene synthase (EAS)]. Two independent cDNA clones (cEAS1 and cEAS2) encoding EAS were isolated from an elicitor-induced tobacco cDNA library by differential hybridization and subsequently were characterized by hybrid selection--in vitro translation. Insertion of cEAS1, a partial cDNA clone encoding 175 C-terminal amino acids, into an Escherichia coli expression vector resulted in accumulation of a fusion protein immunodetectable with EAS-specific polyclonal antibodies. The cDNA clones were used to isolate two full-length EAS genes that mapped 5 kilobases (kb) apart on one 15-kb genomic clone. The nucleotide sequences of the structural gene components were identical from 388 base pairs (bp) upstream of the transcription initiation site to 40 bp downstream of the translation termination codon, suggesting a relatively recent duplication event. The genes consist of 1479-bp open reading frames, each containing five introns and specifying 56,828-Da proteins. The N-terminal amino acid sequence deduced from the genomic clones was identical to the first 16 amino acids of the EAS protein identifiable by Edman degradation. RNA blot hybridization with cEAS1 demonstrated a mRNA induction time course consistent with the induction of the EAS mRNA translational activity with maximum levels 4-6 h after elicitation. EAS mRNA was not detected in control cells. DNA blot-hybridization analysis of genomic DNA revealed a copy number of approximately 12-15 for EAS-like genes in the tetraploid tobacco genome. The conservation of a putative allelic prenyl diphosphate binding motif is also discussed.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alwine J. C., Kemp D. J., Parker B. A., Reiser J., Renart J., Stark G. R., Wahl G. M. Detection of specific RNAs or specific fragments of DNA by fractionation in gels and transfer to diazobenzyloxymethyl paper. Methods Enzymol. 1979;68:220–242. doi: 10.1016/0076-6879(79)68017-5. [DOI] [PubMed] [Google Scholar]
- Anderson M. S., Yarger J. G., Burck C. L., Poulter C. D. Farnesyl diphosphate synthetase. Molecular cloning, sequence, and expression of an essential gene from Saccharomyces cerevisiae. J Biol Chem. 1989 Nov 15;264(32):19176–19184. [PubMed] [Google Scholar]
- Chappell J., Nable R. Induction of sesquiterpenoid biosynthesis in tobacco cell suspension cultures by fungal elicitor. Plant Physiol. 1987 Oct;85(2):469–473. doi: 10.1104/pp.85.2.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dittrich H., Kutchan T. M. Molecular cloning, expression, and induction of berberine bridge enzyme, an enzyme essential to the formation of benzophenanthridine alkaloids in the response of plants to pathogenic attack. Proc Natl Acad Sci U S A. 1991 Nov 15;88(22):9969–9973. doi: 10.1073/pnas.88.22.9969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards K., Cramer C. L., Bolwell G. P., Dixon R. A., Schuch W., Lamb C. J. Rapid transient induction of phenylalanine ammonia-lyase mRNA in elicitor-treated bean cells. Proc Natl Acad Sci U S A. 1985 Oct;82(20):6731–6735. doi: 10.1073/pnas.82.20.6731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glisin V., Crkvenjakov R., Byus C. Ribonucleic acid isolated by cesium chloride centrifugation. Biochemistry. 1974 Jun 4;13(12):2633–2637. doi: 10.1021/bi00709a025. [DOI] [PubMed] [Google Scholar]
- Goldstein J. L., Brown M. S. Regulation of the mevalonate pathway. Nature. 1990 Feb 1;343(6257):425–430. doi: 10.1038/343425a0. [DOI] [PubMed] [Google Scholar]
- Hanahan D., Meselson M. Plasmid screening at high colony density. Gene. 1980 Jun;10(1):63–67. doi: 10.1016/0378-1119(80)90144-4. [DOI] [PubMed] [Google Scholar]
- Herrmann A., Schulz W., Hahlbrock K. Two alleles of the single-copy chalcone synthase gene in parsley differ by a transposon-like element. Mol Gen Genet. 1988 Apr;212(1):93–98. doi: 10.1007/BF00322449. [DOI] [PubMed] [Google Scholar]
- Hohn T. M., Beremand P. D. Isolation and nucleotide sequence of a sesquiterpene cyclase gene from the trichothecene-producing fungus Fusarium sporotrichioides. Gene. 1989 Jun 30;79(1):131–138. doi: 10.1016/0378-1119(89)90098-x. [DOI] [PubMed] [Google Scholar]
- Jennings S. M., Tsay Y. H., Fisch T. M., Robinson G. W. Molecular cloning and characterization of the yeast gene for squalene synthetase. Proc Natl Acad Sci U S A. 1991 Jul 15;88(14):6038–6042. doi: 10.1073/pnas.88.14.6038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leary J. J., Brigati D. J., Ward D. C. Rapid and sensitive colorimetric method for visualizing biotin-labeled DNA probes hybridized to DNA or RNA immobilized on nitrocellulose: Bio-blots. Proc Natl Acad Sci U S A. 1983 Jul;80(13):4045–4049. doi: 10.1073/pnas.80.13.4045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lois A. F., West C. A. Regulation of expression of the casbene synthetase gene during elicitation of castor bean seedlings with pectic fragments. Arch Biochem Biophys. 1990 Jan;276(1):270–277. doi: 10.1016/0003-9861(90)90038-z. [DOI] [PubMed] [Google Scholar]
- Murray M. G., Thompson W. F. Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res. 1980 Oct 10;8(19):4321–4325. doi: 10.1093/nar/8.19.4321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryder T. B., Hedrick S. A., Bell J. N., Liang X. W., Clouse S. D., Lamb C. J. Organization and differential activation of a gene family encoding the plant defense enzyme chalcone synthase in Phaseolus vulgaris. Mol Gen Genet. 1987 Dec;210(2):219–233. doi: 10.1007/BF00325687. [DOI] [PubMed] [Google Scholar]
- Vögeli U., Chappell J. Induction of sesquiterpene cyclase and suppression of squalene synthetase activities in plant cell cultures treated with fungal elicitor. Plant Physiol. 1988 Dec;88(4):1291–1296. doi: 10.1104/pp.88.4.1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vögeli U., Chappell J. Regulation of a sesquiterpene cyclase in cellulase-treated tobacco cell suspension cultures. Plant Physiol. 1990 Dec;94(4):1860–1866. doi: 10.1104/pp.94.4.1860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vögeli U., Freeman J. W., Chappell J. Purification and characterization of an inducible sesquiterpene cyclase from elicitor-treated tobacco cell suspension cultures. Plant Physiol. 1990 May;93(1):182–187. doi: 10.1104/pp.93.1.182. [DOI] [PMC free article] [PubMed] [Google Scholar]