Abstract
The functional outcome of schizophrenia is heterogeneous and markers of the course are missing. Functional outcome is associated with social cognition and negative symptoms. Gesture performance and nonverbal social perception are critically impaired in schizophrenia. Here, we tested whether gesture performance or nonverbal social perception could predict functional outcome and the ability to adequately perform relevant skills of everyday function (functional capacity) after 6 months. In a naturalistic longitudinal study, 28 patients with schizophrenia completed tests of nonverbal communication at baseline and follow-up. In addition, functional outcome, social and occupational functioning, as well as functional capacity at follow-up were assessed. Gesture performance and nonverbal social perception at baseline predicted negative symptoms, functional outcome, and functional capacity at 6-month follow-up. Gesture performance predicted functional outcome beyond the baseline measure of functioning. Patients with gesture deficits at baseline had stable negative symptoms and experienced a decline in social functioning. While in patients without gesture deficits, negative symptom severity decreased and social functioning remained stable. Thus, a simple test of hand gesture performance at baseline may indicate favorable outcomes in short-term follow-up. The results further support the importance of nonverbal communication skills in subjects with schizophrenia.
Key words: nonverbal communication, negative symptoms, psychosis, hand gestures, social perception
Introduction
The outcome of schizophrenia is very heterogeneous, ranging from functional recovery to substantial decline.1–3 While remission, that is, a low level of positive and negative symptoms, is frequently achieved with adequate treatment in the first episodes, only 40% of patients may achieve social or functional recovery, that is, vocational and social functioning in the normal range.2 The achievement of functional recovery is not necessarily linked to remission of symptoms.4 Meta-analyses demonstrated poor functional outcome to be related to impaired cognition, with slightly stronger effects of social cognition than neurocognition on outcomes.5 The poor long-term outcome observed in some patients with schizophrenia is a result of several factors, including the course of the illness, social adversities, and availability of mental health care.2 In addition, severe negative symptoms in the early course have frequently been reported in subjects with poor outcome.3 Clinicians today have no reliable and readily available markers of treatment outcomes in schizophrenia. However, to truly individualize treatment efforts, markers are clearly needed.6
Two symptom domains share a putative association with poor outcome in schizophrenia, that is, negative symptoms and impaired social cognition. Indeed, negative symptoms contribute to poor functional outcome and may also moderate the effect of cognitive impairment on functional outcome.7 This is particularly true for the early course of schizophrenia.8 Among negative symptoms, motivational deficits are particularly relevant for poor functional outcome in schizophrenia.9–11 Negative symptoms tend to be stable over time, also in the early course.12–14 Even though, findings of a recent meta-analysis suggest that negative symptoms may substantially improve in outpatient settings.15
Social cognition is another important factor contributing to functional outcome in schizophrenia.16 It includes processes involved in social interaction, such as perceiving, interpreting, and reacting to social cues. Besides deficits in perceiving and imitating affective facial expression17,18 or emotional prosody,19 schizophrenia patients also share impaired perception, interpretation, and performance of gestures.20–23
Schizophrenia patients and subjects at risk for psychosis use hand gestures less frequently during social interaction.24–26 In addition, up to 67% of patients present with clear deficits performing gestures.21,27,28 Perception and performance of gestures are tightly coupled in schizophrenia, that is, poor perception is linked to impaired performance.21 In subjects at risk for psychosis, gesturing is more frequently associated with wrong content.29 These deficits in gesture perception and performance in schizophrenia are likely to impair real-life social interaction and therefore hamper social functioning. Cross-sectionally, hand gesture performance was linked to positive and negative symptoms,18,21,22,25,29 while nonverbal social perception was predominantly associated with positive symptoms.21,30 Whether gesture deficits are predictive of poor functional outcome or negative symptom severity, however, remains unknown.
The current study tested whether the performance of hand gestures was predictive of symptomatic and functional outcome after 6 months. We hypothesized poor gesture performance at baseline to be associated with poor functioning (social and global), impairments in performance-based measures of outcome, and increased negative symptoms at follow-up. Furthermore, we tested whether nonverbal social perception was predictive of symptomatic and functional outcome after 6 months. Here, we hypothesized poor nonverbal perception at baseline to be associated with poor functioning but not with increased negative symptoms at follow-up.
Methods
Participants
This study was part of a larger investigation on gesture control in schizophrenia. Baseline data have already been reported.21 In total, 28 patients with schizophrenia spectrum disorders participated in the 6-month follow-up tests. All patients were treated within an outpatient program, except one who was completely remitted and did not receive any treatment at the longitudinal assessment. Patients received continued outpatient treatment including pharmacotherapy, visits with psychiatrists, and case management. No specific psychosocial intervention was offered during outpatient care. Exclusion criteria were substance abuse or dependence other than nicotine; past or current medical conditions impairing motor function such as idiopathic parkinsonism, dystonia, or stroke; history of head trauma with concurrent loss of consciousness; and history of electroconvulsive treatment. Patients were interviewed using the Mini International Neuropsychiatric Interview31 as well as the Comprehensive Assessment of Symptoms and History.32 According to DSM-5 at baseline, 25 patients suffered from schizophrenia and 3 from schizophreniform disorder. All participants provided written informed consent. The protocol had been approved by the local ethics committee.
Procedures
Comprehensive assessment of psychopathology, gesture performance, and nonverbal social perception has been conducted at baseline.21 Baseline measures of negative symptoms included the Clinical Assessment Interview for Negative Symptoms (CAINS)33 and the Scale for the Assessment of Negative Symptoms (SANS).34 Follow-up assessments included gesture performance, nonverbal social perception, the Positive And Negative Syndrome Scale (PANSS),35 as well as measures of functional outcome. All clinical and psychopathological assessments at any time point were performed by one experienced clinical psychiatrist (K.S.), who had previously been trained by the senior (S.W.) to achieve interrater reliability of κ ≥ .8. In order to attain high attrition rates during follow-up, patients were approached by telephone up to five times to schedule assessment at 6 months. Furthermore, they received financial compensation for travel costs and participation in the follow-up assessments.
TULIA
The Test of Upper Limb Apraxia (TULIA) is a comprehensive assessment of gesture performance in two domains: following demonstration by the examiner (imitation) or on verbal command (pantomime).36 Evaluation of the 48 items rated content and temporal-spatial errors from video recordings. Total scores range from 0 to 240. All ratings were performed by a single rater (S.W.), blind to diagnoses, clinical presentation, and assessment time point, who had been trained by the test developers (T.V. and S.B.). Intraclass correlations prior to the start of the study exceeded .83. Based on cutoff scores,28 patients were classified into those with gesture deficits and those without gesture deficits at baseline (demographic and clinical data are given in supplementary table S1). The original cutoff score of 194 (2 SD below the mean of controls) separated patients with apraxia from healthy older adults (mean age 61 years).36 We have determined a cutoff score of 210 from an age- and gender-matched control group that fit to typical samples of schizophrenia patients (mean age 40 years).28 The cutoff is across both TULIA domains, thus may balance clear deficits in one domain. Patients with gesture deficits demonstrate content or temporospatial errors in the majority of gestures. In addition, patients with gesture deficits are older, are chronic, and have more impaired frontal lobe function and more motor symptoms.27,37
PONS
We applied the Mini Profile of Nonverbal Sensitivity (Mini-PONS)38 to test nonverbal social perception. The Mini-PONS includes 64 scenes from the original PONS,39 in which short videos of 2s each present a white woman with altering facial expression, voice intonation, and/or bodily gestures. Patients had to choose from two options the one that best describes the observed situation immediately after watching the video, for example, saying a prayer or talking to a lost child. The total scores range from 0 to 64. In one participant, PONS was not assessed at baseline.
Outcome
Functioning was assessed with the Social and Occupational Functioning (SOFAS)40 and the Global Assessment of Functioning (GAF) from DSM-IV, mainly based on self-report. In addition, we assessed functional capacity, that is, the ability to adequately perform everyday skills that are relevant to functioning. Functional capacity was measured with the brief version of the University of California San Diego Performance-Based Assessment (UPSA brief).41 The brief version of the UPSA is limited to 10- to 15-min administration and consists of role-play tasks focusing on finances (eg, counting change or writing a check) and communication (eg, calling to reschedule an appointment). Scores range from 0 to 100, with higher scores indicating superior real world functioning. The UPSA brief has proven great validity in clinical samples.41,42
Statistical Analyses
Descriptive and clinical characteristics were compared between time points using paired t-tests. Simple correlation analyses were performed between psychopathological and outcome measures as well as TULIA and PONS scores at baseline. To test the categorical impact of gesture performance at baseline (patients with or without gesture deficits) on the course of psychopathology and outcome, we conducted repeated measures ANCOVAs testing the effects of time, group, and the Time × Group interaction including age and duration of illness as covariates.
To test the dimensional impact of gesture behavior on functional and symptomatic outcome (GAF, SOFAS, UPSA brief, and PANSS scores at follow-up), simple linear regression analyses were applied. Finally, we tested the additional contribution of baseline TULIA or PONS scores on the course of PANSS negative, PANSS positive, GAF, and SOFAS, applying hierarchical regression analyses. Here, we explored the effect of the baseline values (first step) and the additional contribution of baseline TULIA or PONS scores (second step) on follow-up values of the same rating scales. Within this approach, the magnitude of the R 2 change (∆R 2) was tested for significance. All analyses were performed with SPSS-22.
Results
Over the course of 6 months, positive symptoms (PANSS positive), total PANSS scores, and nonverbal perception (PONS) improved (table 1). No changes were noted in negative symptoms (PANSS negative), GAF, SOFAS, medication dose, or gesture performance (TULIA). When classifying subjects according to the proportional change of scores into those with improving (≥10% better), declining (≥ 10% worse), or stable course (within 10% change), 14 patients (50%) improved, 5 (18%) declined, and 9 (32%) remained unchanged in the PANSS negative. Likewise, for the SOFAS course, 7 (25%) improved, 13 (46%) declined, and 8 (29%) remained unchanged.
Table 1.
Clinical and Demographic Characteristics
| Baseline | 6 Months | Range of Proportional Change From Baseline (%) | Statistic | P Values | |
|---|---|---|---|---|---|
| Gender (men / women) | 20 / 8 | ||||
| Age (years) | 38.4 (10.8) | ||||
| Education (years) | 13.8 (3.1) | ||||
| Duration of illness (years) | 13.5 (11.5) | ||||
| PANSS positive | 18.8 (5.7) | 15.0 (6.2) | 43–167 | t(27) = 3.1 | .004 |
| PANSS negative | 18.3 (5.4) | 16.7 (7.0) | 48–139 | t(27) = 1.7 | .09 |
| PANSS total | 73.0 (16.4) | 64.2 (21.0) | 52–135 | t(27) = 3.0 | .006 |
| CPZ (mg) | 294.8 (262.3) | 327.5 (314.9) | t(27) = .9 | .38 | |
| GAF | 53.2 (18.2) | 48.5 (22.5) | 40–175 | t(27) = 1.4 | .17 |
| SOFAS | 53.9 (18.0) | 49.6 (21.9) | 40–184 | t(27) = 1.4 | .18 |
| TULIA | 201.5 (33.1) | 195.1 (35.8) | 78–142 | t(27) = 1.8 | .09 |
| PONS | 42.6 (5.9) | 44.8 (5.7) | 78–135 | t(26) = −2.5 | .02 |
Note: CPZ = chlorpromazine equivalents; GAF = Global Assessment of Functioning; PANSS = Positive And Negative Syndrome Scale; PONS = Profile of Nonverbal Sensitivity; SOFAS = Social and Occupational Functioning; TULIA = Test of Upper Limb Apraxia.
Typical hand gesture errors included body-part-as-object errors (eg, use of the extended index finger to indicate the use of a toothbrush instead of the correct hand position during brushing the teeth) and errors of spatial orientation (ie, incorrect hand/finger posture relative to body or face). Furthermore, we observed errors of wrong content and incorrect movement sequences.
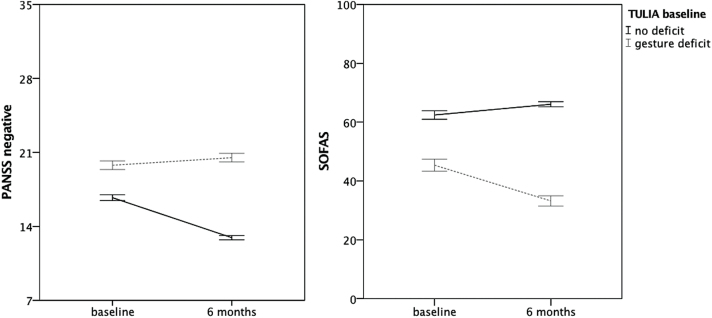
Poorer baseline gesture performance and nonverbal perception correlated with higher symptom severity at follow-up and with impaired functional outcome at 6-month follow-up (table 2). In fact, patients with a gesture deficit at baseline experienced decline in SOFAS and stable PANSS negative syndrome scores (figure 1 and supplementary table S2). Furthermore, performance-based measures of functional capacity at 6-month follow-up were worse in patients with gesture deficits at baseline, particularly in the communication domain (table 3).
Table 2.
Correlations of Baseline Gesture Performance and Clinical Measures
| Baseline | ||||
|---|---|---|---|---|
| TULIA (n = 28) | PONS (n = 27) | |||
| r | P Values | r | P Values | |
| Baseline measures | ||||
| PANSS negative | −.59 | <.001 | −.26 | .18 |
| CAINS | −.45 | .02 | −.25 | .20 |
| SANS | −.48 | .01 | −.18 | .38 |
| GAF | .67 | <.001 | .57 | .002 |
| SOFAS | .63 | <.001 | .59 | .001 |
| Follow-up measures | ||||
| PANSS negative | −.74 | <.001 | −.50 | .01 |
| GAF | .65 | <.001 | .51 | .01 |
| SOFAS | .71 | <.001 | .52 | .01 |
| UPSA brief | .74 | <.001 | .59 | .001 |
Note: Abbreviations are explained in the first footnote to table 1. CAINS = Clinical Assessment Interview for Negative Symptoms; SANS = Scale for the Assessment of Negative Symptoms; UPSA = University of California San Diego Performance-Based Assessment.
Measures of clinical rating scales for each assessment (baseline or follow-up) were correlated here.
Fig. 1.
Baseline gesture performance and course of social functioning and negative symptoms. Lines indicate means and standard errors of the mean (SEM). Baseline TULIA scores were used to dichotomize subjects into those with gesture deficits (n = 14) and those without gesture deficits (n = 14). Group × Time interactions were detected for PANSS negative (F 2, 28 = 7.0, P = .01) and SOFAS (F 2, 28 = 9.8, P = .004), both co-varied for age and duration of illness. PANSS = Positive And Negative Syndrome Scale; SOFAS = social and occupational functioning; TULIA = Test of Upper Limb Apraxia.
Table 3.
Performance-Based Measures at Follow-up and Baseline TULIA Performance
| Gesture Deficit | No Deficit | t | P Values | |
|---|---|---|---|---|
| UPSA finances | 39.0 (12.9) | 46.4 (4.1) | 2.1 | .06 |
| UPSA communication | 26.2 (9.4) | 35.3 (9.6) | 2.5 | .02 |
| UPSA brief total | 65.1 (20.1) | 81.7 (11.2) | 2.7 | .01 |
Functional outcome was associated with gesture performance in both categorical and dimensional ratings of gesture performance. Linear regression analyses indicated that TULIA at baseline predicted UPSA brief at 6 months ( = .53, F = 32.3, β = .74, P < .001), GAF at 6 months ( = .40, F = 18.6, β = .65, P < .001), SOFAS at 6 months ( = .49, F = 26.7, β = .71, P < .001), and PANSS negative at 6 months ( = .53, F = 32.3, β = .74, P < .001). Hierarchical linear regression analyses tested whether TULIA at baseline predicted the course of functioning and psychopathology (table 4, top). Better TULIA performance at baseline accounted for 8% of the variance for 6-month GAF, 13% of the variance for 6-month SOFAS, and 14% of the variance for 6-month PANSS negative.
Table 4.
Hierarchical Regression Analyses
| Block I: Baseline Variables | Block II: TULIA at Baseline | |||||||||
| R 2 | df | F | β | P Values | ∆R 2 | df | F | β | P Values | |
| Variables at 6 months | ||||||||||
| PANSS positive | .19 | 1, 26 | 6.21 | .44 | .02 | .04 | 1, 25 | 1.25 | –.20 | .27 |
| PANSS negative | .55 | 1, 26 | 32.01 | .74 | <.001 | .14 | 1, 25 | 10.88 | –.46 | .003 |
| GAF | .42 | 1, 26 | 19.00 | .65 | <.001 | .08 | 1, 25 | 4.01 | .38 | .06 |
| SOFAS | .46 | 1, 26 | 22.51 | .68 | <.001 | .13 | 1, 25 | 8.15 | .47 | .01 |
| Block I: Baseline Variables | Block II: PONS at Baseline | |||||||||
| R 2 | df | F | β | P Values | ∆R 2 | df | F | β | P Values | |
| Variables at 6 months | ||||||||||
| PANSS positive | .19 | 1, 25 | 6.21 | .44 | .02 | .04 | 1, 24 | 1.25 | −.20 | .27 |
| PANSS negative | .55 | 1, 25 | 30.32 | .74 | <.001 | .10 | 1, 24 | 6.58 | −.32 | .02 |
| GAF | .43 | 1, 25 | 18.61 | .65 | <.001 | .03 | 1, 24 | 1.28 | .21 | .27 |
| SOFAS | .47 | 1, 25 | 22.08 | .69 | <.001 | .02 | 1, 24 | 1.08 | .19 | .31 |
Note: Abbreviations are explained in the first footnote to table 1.
Hierarchical regression analyses applied the same instruments at baseline and follow-up.
Likewise, PONS at baseline predicted UPSA brief at 6 months (
= .32, F = 13.0, β = .59, P = .001), GAF at 6 months (
= .23, F = 8.8, β = .51, P = .007), SOFAS at 6 months (
= .25, F = 9.5, β = .52, P = .005), and PANSS negative at 6 months (
= .22, F = 8.2, β = −.50, P = .009). Hierarchical regression analyses also tested whether PONS at baseline predicted the course of functioning and symptoms (table 4, bottom). Better PONS performance at baseline accounted for 10% of the variance for 6-month PANSS negative. But PONS had no predictive value for functioning at follow-up.
Discussion
The outcome of schizophrenia is a complex issue for which there are currently no predictive markers available. The results of this study indicate that gesture performance and nonverbal social perception at baseline were associated with symptoms, functional capacity, and functional outcome after 6 months. Furthermore, hierarchical regression analyses demonstrated that both gesture performance and nonverbal social perception predicted the course of negative symptoms. But only gesture performance at baseline predicted functional outcome beyond the baseline measures. Thus, if these findings are replicated, a bedside test of hand gesture accuracy may become a marker of functional outcome in schizophrenia spectrum disorders after 6 months.
We applied two sets of outcome measures. First, the performance-based measure UPSA brief, which evaluates actual problem solving. UPSA brief performance was predicted by both baseline measures TULIA and PONS, which explained different proportions of the UPSA brief variance (53% vs 32%). The UPSA brief scores are well in line with those reported in the literature from Swedish or US samples.41,43,44 Second, we applied the GAF and SOFAS, which are rated mainly based on patients’ self-report. TULIA and PONS at baseline predicted a substantial proportion of GAF and SOFAS variance at follow-up, with better performance at baseline indicating superior functioning at follow-up. GAF and SOFAS were entered into hierarchical regression analyses and were predicted by gesture performance but not nonverbal social perception. Therefore, our results not only suggest that nonverbal communication skills were predictive of functional outcome, but further indicate that nonverbal performance skills are more useful than nonverbal perceptual skills when predicting the functional outcome at 6-month follow-up.
The main finding of our study is well in line with reports on the contribution of social cognitive impairments to poor functional outcome in schizophrenia.5,16,45–47 One has to keep in mind that we applied rather direct measures of social cognition, that is, hand gesture performance and nonverbal social perception, both of which tended to slightly improve over time in our sample. However, more complex measures of social cognition demonstrated temporal stability in a 12-month longitudinal study as well as in a cross-sectional study in first episode schizophrenia.47,48 While poor social functioning at baseline predicted poor social function at 6-month follow-up, the baseline ability to perform hand gestures correctly added further information on social functioning at follow-up. Thus, an actual performance measure of nonverbal social skills may become a useful predictor of functional outcome. Indeed, those with poor gesture performance at baseline faced a decline of social functioning during follow-up.
Both gesture performance and nonverbal social perception at baseline predicted negative symptom severity at follow-up, even when controlling for baseline negative symptoms. This is in line with the notion that negative symptoms comprise two main factors, one of which is diminished expression49 that should be related to poor gesture performance, the other is avolition. In fact, the association of gesture performance and negative symptom severity has been reported before,22,29 even though the correlation between impaired gesture performance and negative symptom severity was weak in some studies.21,27,28 In contrast, nonverbal social perception was unrelated to negative symptoms in cross-sectional studies.50,51 However, broader measures of social cognition correlated with negative symptom severity.47,48 In the present study, TULIA correlated with several negative symptom ratings at baseline and follow-up, while PONS was only associated with negative symptoms at follow-up. Thus, negative symptom severity seems to be linked to nonverbal communication skills. Strikingly, poor baseline gesture performance indicated a group of patients with stable course of negative symptoms, while good performance was associated with amelioration of negative symptoms during follow-up.
The correct performance of hand gestures may critically contribute to nonverbal communication and therefore benefit social skills.20 In addition, incorrect interpretation of hand gestures would interfere with communication, particularly, as schizophrenia patients are more likely to interpret accidental gestures as threatening or self-referential.23,30 Importantly, gesture performance and nonverbal social perception are strongly correlated in schizophrenia, that is, deficits in one domain are associated with impairments in the other.21 Previous work of our lab and others has established that correct hand gesture performance relies on motor abilities, frontal lobe functions, and to some extent on working memory performance.18,21,22,27,28 The cross-sectional analysis indicated that both impairments in gesture performance and poor nonverbal social perception were associated with the factor motor/frontal lobe dysfunction and with the factor positive symptoms/working memory.21 In the course of 6-month treatment, positive symptoms significantly improved. At the same time, scores on the Mini-PONS increased, suggesting that nonverbal social perception may improve as positive symptoms are controlled. However, at group level we failed to observe changes in gesture performance. Neither gesture performance nor nonverbal social perception at baseline explained any variance of PANSS positive at follow-up when controlling for the baseline PANSS positive score. Thus, any association between positive symptoms and nonverbal skills was cross-sectional.
The baseline TULIA scores were used to separate patients without gesture deficits from patients with gesture deficits based on cutoff scores.28 In line with our previous studies, patients with gesture deficits were older and had been suffering longer from schizophrenia (supplementary table S1).27 Gesture deficits are more frequently observed in patients with multiple episodes of the disorder.37 Still, the Group × Time interactions for negative symptoms and social functioning (figure 1 and supplementary table S2) in the repeated measures ANCOVAs remained significant even when controlling for age and duration of illness. Furthermore, the dimensional assessment of gesture performance yielded similar results as the categorical one in the hierarchical regression analyses. Findings hold also true when restricting analyses to subjects with less than 15 years of illness duration ( supplementary table S5).
If these findings can be replicated in larger studies with rigorous longitudinal statistics accounting for various associated clinical factors, baseline performance of hand gestures may help to identify subjects who require even more effort in treating or preventing negative symptoms and functional decline. Schizophrenia patients with clear gesture deficits have reduced gray matter within the left inferior frontal gyrus compared to schizophrenia patients without gesture deficits.52 Thus, structural brain alterations within the semantic networks implicated in gesture and speech processing53,54 may contribute to poor hand gesture performance.
Social cognitive training has been effective in the domains of facial affect recognition and theory of mind.55 Future studies need to establish whether the hand gesture deficits may be alleviated with specific nonverbal communication training.20 Our findings suggest that this could also improve functional outcome.
Some limitations of the current study require discussion. First, the assessment of functional capacity with the UPSA brief has only been performed at follow-up. Baseline measures would have enabled hierarchical regression analyses also for this important outcome variable. However, linear regression analyses clearly supported an association of baseline gesture performance and social perception with follow-up functional capacity. Second, we applied the Mini-PONS to assess the accuracy of nonverbal perception. It contains various nonverbal stimuli that are not gestures. In future studies, a specific test of gesture perception should be applied. Third, the small sample size limits the number of factors that may be entered in the regression analyses. Thus, the potential contribution of other clinical factors to social outcome in schizophrenia could not be explored. Fourth, due to the exploratory nature, analyses were uncorrected for multiple comparisons. Thus, findings have to be interpreted with caution and require replication. Fifth, this was a naturalistic longitudinal study in which patients received treatment as usual. Therefore, we cannot infer whether particular treatment strategies may have improved social functioning or outcome of negative symptoms. However, no single specific treatment option has emerged to effectively target negative symptoms or social outcome. Finally, a common problem to longitudinal studies is decline in attrition rate, ultimately leading to selection bias. However, at baseline no relevant differences emerged between patients completing the study and patients lost to follow-up (supplementary table S4).
Conclusion
In a longitudinal study, two tasks of nonverbal communication, that is, hand gesture performance and nonverbal social perception, predicted negative symptoms, functional capacity, and functional outcome after 6 months. The performance of hand gestures at baseline was of added value predicting social functioning at follow-up beyond the baseline measure. Further longitudinal studies need to test whether simple tests of nonverbal communication skills may have the potential to become readily available outcome markers in schizophrenia.
Supplementary Material
Supplementary material is available at http://schizophreniabulletin.oxfordjournals.org.
Funding
Bangerter-Rhyner Foundation (to Dr Walther); Swiss National Science Foundation (SNF grant 152619 to Drs Walther and Bohlhalter).
Supplementary Material
Acknowledgment
The authors have declared that there are no conflicts of interest in relation to the subject of this study.
References
- 1. van Os J, Kapur S. Schizophrenia. Lancet 2009;374:635–645. [DOI] [PubMed] [Google Scholar]
- 2. Zipursky RB, Reilly TJ, Murray RM. The myth of schizophrenia as a progressive brain disease. Schizophr Bull. 2013;39:1363–1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lang FU, Kösters M, Lang S, Becker T, Jäger M. Psychopathological long-term outcome of schizophrenia—a review. Acta Psychiatr Scand. 2013;127:173–182. [DOI] [PubMed] [Google Scholar]
- 4. Harvey PD, Bellack AS. Toward a terminology for functional recovery in schizophrenia: is functional remission a viable concept? Schizophr Bull. 2009;35:300–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Fett AK, Viechtbauer W, Dominguez MD, Penn DL, van Os J, Krabbendam L. The relationship between neurocognition and social cognition with functional outcomes in schizophrenia: a meta-analysis. Neurosci Biobehav Rev. 2011;35:573–588. [DOI] [PubMed] [Google Scholar]
- 6. Murck H, Laughren T, Lamers F, Picard R, Walther S, Goff D, Sainati S. Taking personalized medicine seriously: biomarker approaches in phase IIb/III studies in major depression and schizophrenia. Innov Clin Neurosci. 2015;12:26S–40S. [PMC free article] [PubMed] [Google Scholar]
- 7. Ventura J, Hellemann GS, Thames AD, Koellner V, Nuechterlein KH. Symptoms as mediators of the relationship between neurocognition and functional outcome in schizophrenia: a meta-analysis. Schizophr Res. 2009;113:189–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ventura J, Subotnik KL, Gitlin MJ, et al. Negative symptoms and functioning during the first year after a recent onset of schizophrenia and 8 years later. Schizophr Res. 2015;161:407–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Faerden A, Barrett EA, Nesvåg R, et al. Apathy, poor verbal memory and male gender predict lower psychosocial functioning one year after the first treatment of psychosis. Psychiatry Res. 2013;210:55–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fervaha G, Foussias G, Agid O, Remington G. Motivational deficits in early schizophrenia: prevalent, persistent, and key determinants of functional outcome. Schizophr Res. 2015;166:9–16. [DOI] [PubMed] [Google Scholar]
- 11. Galderisi S, Rossi A, Rocca P, et al. The influence of illness-related variables, personal resources and context-related factors on real-life functioning of people with schizophrenia. World Psychiatry. 2014;13:275–287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. McGlashan TH, Fenton WS. Subtype progression and pathophysiologic deterioration in early schizophrenia. Schizophr Bull. 1993;19:71–84. [DOI] [PubMed] [Google Scholar]
- 13. Arndt S, Andreasen NC, Flaum M, Miller D, Nopoulos P. A longitudinal study of symptom dimensions in schizophrenia. Prediction and patterns of change. Arch Gen Psychiatry. 1995;52:352–360. [DOI] [PubMed] [Google Scholar]
- 14. Heilbronner U, Samara M, Leucht S, Falkai P, Schulze TG. The longitudinal course of schizophrenia across the lifespan: clinical, cognitive, and neurobiological aspects. Harv Rev Psychiatry. 2016;24:118–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Savill M, Banks C, Khanom H, Priebe S. Do negative symptoms of schizophrenia change over time? A meta-analysis of longitudinal data. Psychol Med. 2015;45:1613–1627. [DOI] [PubMed] [Google Scholar]
- 16. Green MF, Horan WP, Lee J. Social cognition in schizophrenia. Nat Rev Neurosci. 2015;16:620–631. [DOI] [PubMed] [Google Scholar]
- 17. Kohler CG, Walker JB, Martin EA, Healey KM, Moberg PJ. Facial emotion perception in schizophrenia: a meta-analytic review. Schizophr Bull. 2010;36:1009–1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Park S, Matthews N, Gibson C. Imitation, simulation, and schizophrenia. Schizophr Bull. 2008;34:698–707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Bach DR, Buxtorf K, Grandjean D, Strik WK. The influence of emotion clarity on emotional prosody identification in paranoid schizophrenia. Psychol Med. 2009;39:927–938. [DOI] [PubMed] [Google Scholar]
- 20. Walther S, Mittal VA. Why we should take a closer look at gestures. Schizophr Bull. 2016;42:259–261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Walther S, Stegmayer K, Sulzbacher J, et al. Nonverbal social communication and gesture control in schizophrenia. Schizophr Bull. 2015;41:338–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Matthews N, Gold BJ, Sekuler R, Park S. Gesture imitation in schizophrenia. Schizophr Bull. 2013;39:94–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. White TP, Borgan F, Ralley O, Shergill SS. You looking at me?: Interpreting social cues in schizophrenia. Psychol Med. 2016;46:149–160. [DOI] [PubMed] [Google Scholar]
- 24. Troisi A, Spalletta G, Pasini A. Non-verbal behaviour deficits in schizophrenia: an ethological study of drug-free patients. Acta Psychiatr Scand. 1998;97:109–115. [DOI] [PubMed] [Google Scholar]
- 25. Lavelle M, Healey PG, McCabe R. Is nonverbal communication disrupted in interactions involving patients with schizophrenia? Schizophr Bull. 2013;39:1150–1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Mittal VA, Tessner KD, McMillan AL, Delawalla Z, Trotman HD, Walker EF. Gesture behavior in unmedicated schizotypal adolescents. J Abnorm Psychol. 2006;115:351–358. [DOI] [PubMed] [Google Scholar]
- 27. Walther S, Vanbellingen T, Müri R, Strik W, Bohlhalter S. Impaired pantomime in schizophrenia: association with frontal lobe function. Cortex. 2013;49:520–527. [DOI] [PubMed] [Google Scholar]
- 28. Walther S, Vanbellingen T, Müri R, Strik W, Bohlhalter S. Impaired gesture performance in schizophrenia: particular vulnerability of meaningless pantomimes. Neuropsychologia. 2013;51:2674–2678. [DOI] [PubMed] [Google Scholar]
- 29. Millman ZB, Goss J, Schiffman J, Mejias J, Gupta T, Mittal VA. Mismatch and lexical retrieval gestures are associated with visual information processing, verbal production, and symptomatology in youth at high risk for psychosis. Schizophr Res. 2014;158:64–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Bucci S, Startup M, Wynn P, Baker A, Lewin TJ. Referential delusions of communication and interpretations of gestures. Psychiatry Res. 2008;158:27–34. [DOI] [PubMed] [Google Scholar]
- 31. Sheehan DV, Lecrubier Y, Sheehan KH, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. 1998;59(suppl 20):22–33. [PubMed] [Google Scholar]
- 32. Andreasen NC, Flaum M, Arndt S. The Comprehensive Assessment of Symptoms and History (CASH). An instrument for assessing diagnosis and psychopathology. Arch Gen Psychiatry. 1992;49:615–623. [DOI] [PubMed] [Google Scholar]
- 33. Forbes C, Blanchard JJ, Bennett M, Horan WP, Kring A, Gur R. Initial development and preliminary validation of a new negative symptom measure: the Clinical Assessment Interview for Negative Symptoms (CAINS). Schizophr Res. 2010;124:36–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Andreasen NC. The Scale for the Assessment of Negative Symptoms (SANS): conceptual and theoretical foundations. Br J Psychiatry Suppl. 1989:49–58. [PubMed] [Google Scholar]
- 35. Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13:261–276. [DOI] [PubMed] [Google Scholar]
- 36. Vanbellingen T, Kersten B, Van Hemelrijk B, et al. Comprehensive assessment of gesture production: a new test of upper limb apraxia (TULIA). Eur J Neurol. 2010;17:59–66. [DOI] [PubMed] [Google Scholar]
- 37. Stegmayer K, Moor J, Vanbellingen T, et al. Gesture performance in first- and multiple-episode patients with schizophrenia spectrum disorders. Neuropsychobiology. 2016;73:201–208. [DOI] [PubMed] [Google Scholar]
- 38. Banziger T, Scherer KR, Hall JA, Rosenthal R. Introducing the MiniPONS: a short multichannel version of the profile of nonverbal sensitivity (PONS). J Nonverbal Behav. 2011;35:189–204. [Google Scholar]
- 39. Rosenthal R, Hall JA, DiMatteo MR, Rogers PL, Archer D. Sensitivity to Nonverbal Communication: The PONS Test. Baltimore, MD: John Hopkins University Press; 1979. [Google Scholar]
- 40. Goldman HH, Skodol AE, Lave TR. Revising axis V for DSM-IV: a review of measures of social functioning. Am J Psychiatry. 1992;149:1148–1156. [DOI] [PubMed] [Google Scholar]
- 41. Mausbach BT, Harvey PD, Goldman SR, Jeste DV, Patterson TL. Development of a brief scale of everyday functioning in persons with serious mental illness. Schizophr Bull. 2007;33:1364–1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Mausbach BT, Depp CA, Bowie CR, et al. Sensitivity and specificity of the UCSD Performance-based Skills Assessment (UPSA-B) for identifying functional milestones in schizophrenia. Schizophr Res. 2011;132:165–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Harvey PD, Helldin L, Bowie CR, et al. Performance-based measurement of functional disability in schizophrenia: a cross-national study in the United States and Sweden. Am J Psychiatry. 2009;166:821–827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Mausbach BT, Harvey PD, Pulver AE, et al. Relationship of the Brief UCSD Performance-based Skills Assessment (UPSA-B) to multiple indicators of functioning in people with schizophrenia and bipolar disorder. Bipolar Disord. 2010;12:45–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Schmidt SJ, Mueller DR, Roder V. Social cognition as a mediator variable between neurocognition and functional outcome in schizophrenia: empirical review and new results by structural equation modeling. Schizophr Bull. 2011;37(suppl 2):S41–S54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Mueller DR, Schmidt SJ, Roder V. One-year randomized controlled trial and follow-up of integrated neurocognitive therapy for schizophrenia outpatients. Schizophr Bull. 2015;41:604–616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Horan WP, Green MF, DeGroot M, et al. Social cognition in schizophrenia, Part 2: 12-month stability and prediction of functional outcome in first-episode patients. Schizophr Bull. 2012;38:865–872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Green MF, Bearden CE, Cannon TD, et al. Social cognition in schizophrenia, Part 1: performance across phase of illness. Schizophr Bull. 2012;38:854–864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Kaiser S, Heekeren K, Simon JJ. The negative symptoms of schizophrenia: category or continuum? Psychopathology. 2011;44:345–353. [DOI] [PubMed] [Google Scholar]
- 50. Toomey R, Schuldberg D, Corrigan P, Green MF. Nonverbal social perception and symptomatology in schizophrenia. Schizophr Res. 2002;53:83–91. [DOI] [PubMed] [Google Scholar]
- 51. Giannitelli M, Xavier J, Francois A, et al. Facial, vocal and cross-modal emotion processing in early-onset schizophrenia spectrum disorders. Schizophr Res. 2015;168:252–259. [DOI] [PubMed] [Google Scholar]
- 52. Stegmayer K, Bohlhalter S, Vanbellingen T, et al. Structural brain correlates of defective gesture performance in schizophrenia. Cortex. 2016;78:125–137. [DOI] [PubMed] [Google Scholar]
- 53. Straube B, Green A, Sass K, Kirner-Veselinovic A, Kircher T. Neural integration of speech and gesture in schizophrenia: evidence for differential processing of metaphoric gestures. Hum Brain Mapp. 2013;34:1696–1712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Straube B, Green A, Sass K, Kircher T. Superior temporal sulcus disconnectivity during processing of metaphoric gestures in schizophrenia. Schizophr Bull. 2014;40:936–944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Kurtz MM, Richardson CL. Social cognitive training for schizophrenia: a meta-analytic investigation of controlled research. Schizophr Bull. 2012;38:1092–1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.