Abstract
The encoding and storage of experience by the hippocampus is essential for the formation of episodic memories and the transformation of individual experiences into semantic structures such as maps and schemas. The rodent hippocampus compresses ongoing experience into repeating theta sequences, but the factors determining the content of theta sequences are not understood. Here we first show that the spatial paths represented by theta sequences in rats extend farther in front of the rat during acceleration and higher running speeds and begin farther behind the rat during deceleration. Second, the length of the path is directly related to the length of the theta cycle and the number of gamma cycles in it. Finally, theta sequences represent the environment in segments or ‘chunks’. These results imply that information encoded in theta sequences is subject to powerful modulation by behavior and task variables. Furthermore, these findings suggest a potential mechanism for the cognitive ‘chunking’ of experience.
The hippocampus has a central role in episodic memory1, navigation2 and episodic future thinking3,4. An essential aspect of hippocampal contributions to these behavioral phenomena is the notion that the hippocampus encodes and stores a record of experience5. If we are ultimately to uncover the role the hippocampus plays in navigation, imagination and planning processes, it is necessary to understand its information processing characteristics. How is experiential information encoded and subsequently recalled within the hippocampal system? Which features of the encoding process are invariant under, and which ones depend on, the behavioral state of the subject and the structure of the task?
The leading systems-level model for memory encoding in the hippocampus is provided by recordings from hippocampal place cells in freely moving rodents2,6. These cells tend to fire when the animal is in specific locations in an environment, such that a particular trajectory taken by the animal corresponds to a unique sequence of active place cells—the experience to be stored. Notably, the hippocampus compresses sequences of active place cells, continually repeating them at a speed of 5–10× during each cycle of the theta oscillation7 as the animal runs8–10. These theta sequences not only provide a repetition of ongoing experience but also bring the spike timing of successively activated place cells into the range conducive to spike timing–dependent plasticity8,11–14. As such, the content of theta sequences provides a unique representational window into what the hippocampus encodes and recalls during active experience.
Previous studies have concentrated on this phenomenon from the perspective of phase precession, examining how the phase of spiking of individual cells and cell assemblies changes8,13–16. Studies of single passes through a place field17 and of the timing intervals between pairs of cells spiking within a single theta cycle9,18 indicate that the sequence within the cycle itself is more reliable than the phase of any single cell’s spiking, suggesting that it would be fruitful to directly examine the relationship between sequences themselves and behavior.
In this study, instead of measuring the relationship of single cell firing to phase of theta, we used the multicell sequence represented within each individual theta cycle to determine the spatial path represented and examined the properties of that path in relation to behavior and task structure. We found that paths represented by theta sequences extended farther ahead of the animal as it left landmarks and accelerated, whereas sequences started farther behind as the animal approached landmarks and decelerated. Even after taking these effects into account, we found that theta sequences more frequently represented the space between adjacent landmarks. Thus, the environment was represented in chunks, implying a task-related segmentation of information processing in hippocampus.
RESULTS
To investigate the relationship between behavior and the paths represented by theta sequences, we recorded neural ensembles from the dorsal CA1 region of hippocampus in six rats trained on a spatial decision task19 (Fig. 1a). On a given day the rats had to learn whether the rewarded policy was alternation, left turns, or right turns at turn 2 on the maze. The rewarded policy changed approximately halfway through the session. Rats determined both the initial policy and the switched policy quickly19.
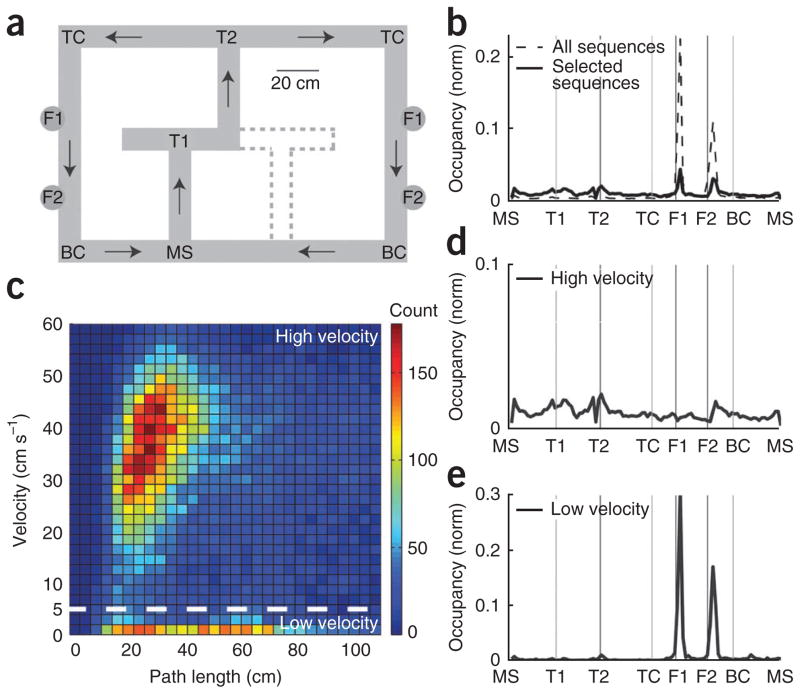
Figure 1.
Distinct populations of theta sequences on the two-choice T maze. (a) The two-choice T maze. The maze had two possible physical configurations, the second indicated by dashed lines. Noteworthy landmarks on the maze include the maze start (MS), turn 1 (T1), turn 2 (T2), top corner (TC), feeder 1 (F1), feeder 2 (F2) and bottom corner (BC). Arrows indicate the direction of maze traversal. (b) Overall and selected sequence count over the linearized maze. Overall sequence count over the maze (n = 618,408 sequences) is a reflection of the amount of time rats spent at each location on the maze. Selected sequence count (n = 33,397 sequences) indicates the number of sequences at each location on the maze that passed the inclusion criteria described in the main text. (c) Theta sequence histogram of rat velocity versus path length, defined as the distance (in centimeters) from the decoded start of the sequence of firing within the theta cycle to the decoded end of the sequence (see Online Methods). The color of each pixel indicates the number of theta sequences with a specified path length (x axis) recorded while a rat was running at a particular velocity (y axis). The horizontal dashed line separates low- from high-velocity theta sequences. (d) High-velocity sequence count (n = 29,351 sequences) over the linearized maze. The number of high-velocity sequences (sequences above horizontal line in c) at each location on the maze. (e) Low-velocity sequence count (n = 4,046 sequences) over the linearized maze. Occupancy is defined as time spent at each location on the maze.
To determine what spatial path was represented by each theta sequence, the beginning and end of each theta cycle were identified (see Online Methods) and the neural activity during each cycle was analyzed. Theta cycles with three or more active cells, a mean sharp wave ripple (SWR) power <1 s.d. above the mean (to exclude awake replay sequences20–22) and theta power >1 s.d. below the mean were included for analysis. Results were similar with thresholds of four or five active cells and with theta power threshold set to the mean theta power or to 2 s.d. below the mean (data not shown). Of 618,408 theta cycles, 168,770 met these criteria. These theta cycles were analyzed for sequential spiking structure, and only cycles with statistically significant sequential structure (defined in Online Methods) were used in this study (33,397 out of 168,770; 19.8%) (Supplementary Fig. 1). Using a Bayesian decoding approach, we identified the spatial path represented during each theta cycle (see Online Methods). An alternative, non-Bayesian approach for decoding spatial paths23 showed qualitatively similar results to those presented here. Theta sequences occurred at all locations on the maze (Fig. 1b). As expected, rats spent the most time at feeder locations, and a smaller proportion of theta cycles were significant at feeder locations compared with other locations on the maze.
From the start and end locations of each represented path and the rat’s location, we calculated three key distances: the distance from the rat’s location to the end of the path (ahead length), the distance from the start of the path to the rat’s location (behind length) and the total length of the path (path length). We examined the relationship between rat velocity and path length by creating a two-dimensional histogram of velocity as a function of path length. The theta sequences naturally grouped into high- and low-velocity families (Fig. 1c). The high-velocity sequences were evenly distributed across the maze (Fig. 1d), whereas the low-velocity sequences were preferentially observed when rats were at the feeder locations and the choice point (Fig. 1e). For the remainder of the analyses presented here, we focus on high-velocity theta sequences (29,351 sequences), although all results are qualitatively unchanged by the inclusion of low-velocity sequences.
Ahead and behind representations
We observed that some sequences represented a spatial path extending farther ahead of the rat, whereas other sequences represented paths beginning more behind the rat’s location. Ahead sequences were observed at the choice point (Fig. 2), potentially analogous to the forward sweeps previously observed in CA3 while rats were paused and deliberating at a choice point24. However, we also observed ahead-extending sequences at other locations on the maze (Fig. 3). Examples of ahead and behind representations at various locations on the maze are shown in Figures 2 and 3. Ahead sequences tended to occur as rats left physical landmarks on the maze. Behind sequences tended to occur as rats approached landmarks. In between sequences tended to occur during journeys between those landmarks.
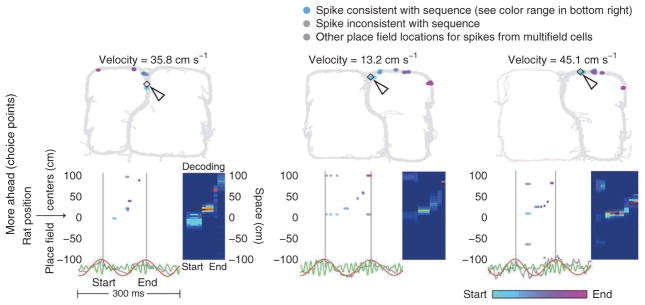
Figure 2.
Examples of ahead sequences while rats were located at the choice point. Top, the rat’s average velocity over the theta cycle. Each place field center represented by a spike in the sequence (colored point in corresponding bottom panel) is plotted on the two-dimensional maze at the location of its two-dimensional place field center. The arrow shows the rat’s location. Bottom left, spikes plotted by place field center location (space) relative to the rat’s position (along either a left or right loop of the maze) over a single theta cycle (see Online Methods). LFPs filtered between 6 Hz and 12 Hz (red), 40 Hz and 100 Hz (green) and unfiltered (gray) are plotted below. Colored points indicate spikes that contribute positively to the sequence score according to the sequence detection algorithm (for visualization purposes only). The color of the spike indicates its relative time within the sequence (light blue, early; light purple, late). For cells with multiple place fields, gray points are plotted at every place field center belonging to the cell (colored points occupy the place field center that contributes maximally to the score). Spikes that do not contribute positively to the sequence score are also plotted in gray. Bottom right, the Bayesian decoded spatial probability distributions computed over the theta cycle (see Online Methods). Red indicates high probability, blue low probability. These examples likely correspond to the previously reported ‘sweeps’24.
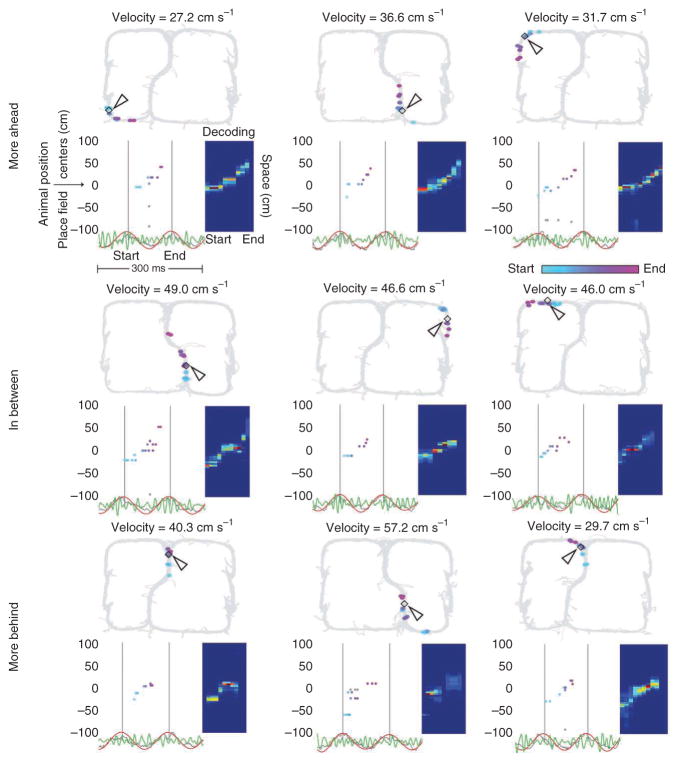
Figure 3.
Examples of ahead and behind sequences. Top, sequences representing paths more ahead of the rat while rats were at various locations on the maze. Middle, sequences representing paths that are behind and ahead of the rat. Bottom, paths more behind the rat. See Figure 2 caption for details.
Ahead and behind length are inversely related
Ahead, behind and overall path length define the spatial information represented by each given theta cycle. Ahead length measures how far the multicell sequence extends into the space ahead of the rat, behind length measures how far behind the rat the multicell sequence begins and path length measures the distance covered by the entire sequence. (see Online Methods.) To determine the relationships among these three lengths, we plotted each mean length with respect to the others. As path length increased, both ahead and behind length increased (Fig. 4), as expected, because the path length of each theta sequence is the sum of its ahead and behind lengths. On the other hand, as ahead length increased, behind length decreased. Thus, ahead length and behind length were inversely related; spatial paths that extended farther forward did not begin as far back and vice versa.
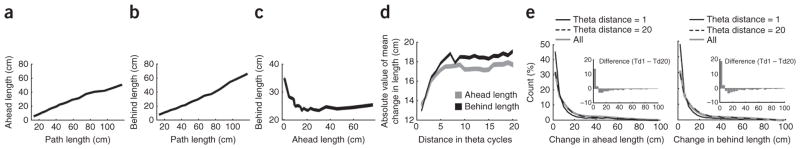
Figure 4.
Characteristics of theta sequence ahead and behind length. (a) Mean ahead length as a function of path length. (b) Mean behind length as a function of path length. (c) Mean behind length as a function of ahead length. Note the inverse relationship between ahead length and behind length. (d) Ahead and behind length changes as a function of theta cycle separation. The absolute value of the average change in both ahead and behind length increased from successive theta cycles (beginning with 1 on the x axis) to a separation of about seven theta cycles and then leveled off. This indicates that the spaces mapped by neural activity in adjacent theta cycles are more similar than the spaces mapped in nonadjacent cycles. (e) Adjacent sequences tend to have related ahead and behind lengths but also occasionally include lengths that are substantially different. Sequences that are more separated (twenty theta cycles shown) tend to have larger changes in ahead and behind length. The subtraction between these two distributions is shown in an inset. Also shown is the distribution of all differences, ignoring the theta cycle distance between the two cycles. Sequences separated by 20 cycles approach random chance, whereas sequences separated by only a single theta cycle are more likely to be similar to each other. These analyses included 29,351 theta cycles. Line width in a–d indicate s.e.m. Note that some y axes have non-zero origins.
To determine how quickly prospective and retrospective representations changed on this task, we measured the changes in ahead and behind length as a function of theta-cycle separation. The ahead and behind lengths of individual theta cycles showed similarities with the preceding and following theta cycles; however, they could also shift substantially from one theta cycle to the next. For adjacent theta cycles, 63% had ahead-length differences <10 cm. The average change in the distance represented by a theta sequence ahead or behind the rat’s location increased from successive theta cycles to a separation of about seven theta cycles and then leveled off (Fig. 4d). Because theta sequences generally represent positions near the rat, one would expect adjacent theta sequences to represent similar information; this analysis shows that the prospective or retrospective nature of adjacent theta cycles were more similar than those of nonadjacent theta cycles. However, examination of the distribution of changes in ahead and behind length between adjacent theta cycles showed that there were cases in which large differences occurred, which implies that the hippocampal representation can quickly shift from more behind to more ahead of the rat or from more ahead to more behind. These results may explain the improvement in observations looking at phase precession on a trial-by-trial basis17 and the more reliable timing of cell pairs and sequences within a theta cycle than what would be observed if cells phase precessed independently9.
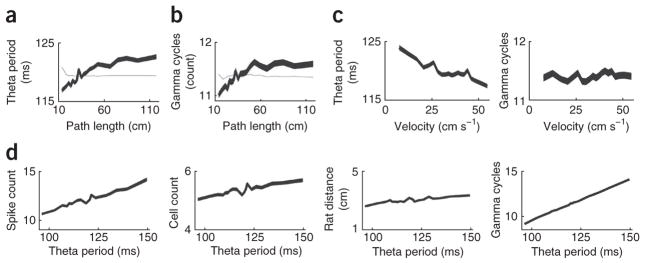
Theta period and gamma cycles increase with path length
As path length increased, we observed that the theta period and the number of gamma cycles per theta period also increased (Fig. 5a,b). Using the previously reported relationship between theta period and velocity (ref. 2 and Fig. 5c) and the observed relationship between velocity and path length (Fig. 1c), we determined the relationship between theta period and path length if the theta period were purely a function of velocity and compared it with the observed relationship (Fig. 5a). The relationship between velocity and theta period did not account for the increase in theta period with increasing path length (Fig. 5a). Similarly, the number of gamma cycles increased with increasing path length (Fig. 5b). Thus, longer theta periods and more gamma cycles per theta cycle were associated with longer paths represented by theta sequences. Longer theta periods were also associated with more active place cells, more spikes and more gamma cycles (Fig. 5d). Although it is possible that the increased cellular activity during longer theta cycles contributed to the representation of longer paths, it should be noted that there was only a slight increase in distance traveled by the rats with increasing theta period, and controlling for velocity does not affect the relationships shown in Figure 5a,b.
Figure 5.
Theta period and gamma cycles vary as functions of path length. (a–c) The thick black curves show mean theta period (a) or mean number of gamma cycles (b) (y axis) with increasing path length (x axis). The gray curves were generated by multiplying the average velocity versus path length relationship (Fig. 8b) by the theta period versus velocity relationship or gamma cycle versus velocity relationship (c). Thus, the gray curve is the expected relationship if the theta period (a) or the number of gamma cycles (b) were merely a function of the relationship between theta period and velocity and path length and velocity. (d) Mean spike count, cell count, rat distance traveled and gamma cycles as functions of theta period. This analysis included 29,351 theta sequences. Line width indicates s.e.m. for a–d. Note that some y axes have non-zero origins.
Average ahead and behind length over space
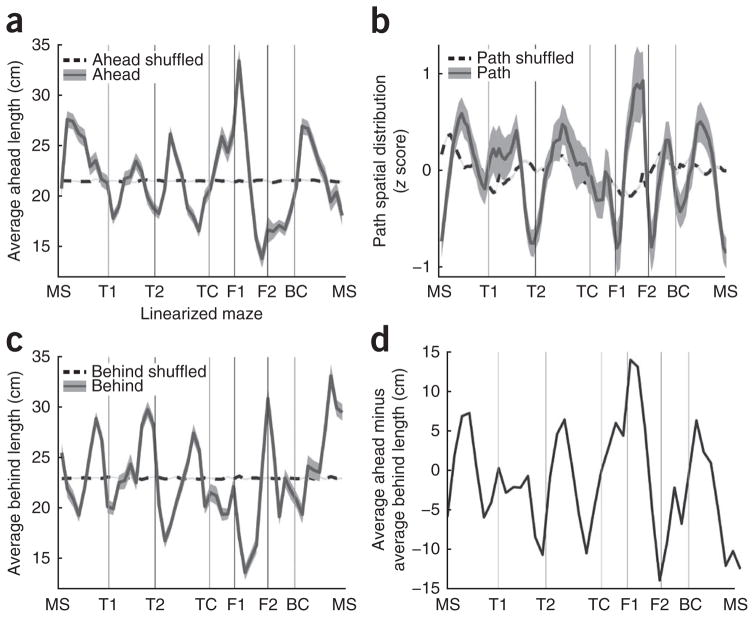
To determine whether theta sequences preferentially represented space ahead or behind the rat at certain locations on the maze, the average ahead length and average behind length were plotted as a function of linearized location on the maze (Fig. 6). We observed clear peaks and troughs in average ahead length over space and average behind length over space (Fig. 6a,c). Furthermore, average ahead length and average behind length were anticorrelated over space (r = −0.34, P < 0.05), with ahead length peaks aligning with behind length troughs and vice versa. This observation is consistent with, but not a necessary result of, the inverse relationship between ahead and behind length presented above (Fig. 4). Thus, at certain locations on the maze, the hippocampus represented more space ahead of the rat and less space behind, and at other locations on the maze, the hippocampus represented more space behind the rat and less space ahead.
Figure 6.
Ahead length, behind length and path distribution vary according to landmarks on the maze. (a) Ahead length as a function of location on maze. Peaks indicate locations at which the represented path extended farther ahead of the rat, on average. Troughs indicate locations where the represented path ended closer to the rat’s location. (b) Density of represented paths as a function of location on maze. Peaks indicate regions of the maze that were overrepresented by theta sequences. The y axis is the average z score (z score computed over each session). (c) Behind length as a function of location on maze. Peaks indicate locations at which the represented path began farther behind the rat, on average. Troughs indicate locations where the represented path began closer to the rat’s location. (d) Average ahead length minus average behind length. This plot demonstrates the alternating pattern of representations that are more ahead and more behind the rat, as a function of location on the maze. This analysis included 29,351 theta sequences. Line width indicates s.e.m. Controls in a–c include a curve produced by randomly reassigning theta sequences with different rat locations. This shuffling procedure was done one hundred times, and the average shuffled distribution is plotted alongside the real data. Note that some y axes have non-zero origins.
The peaks of ahead length and peaks of behind length had a relationship to physical landmarks on the maze (turns and feeders). Ahead length peaks were generally observed just after landmarks (for example, maze start, turn 2 (the choice point), feeder 1, bottom corner) and behind length peaks were observed just before landmarks. Thus, as rats entered a segment of the maze defined by two physical landmarks, the spatial representation was more ahead of the rat; as they exited the segment, the representation was more behind the rat. In effect, the path representations adjusted so as to focus on the segment of space between the two landmarks.
Spatial distribution of represented paths
The alternating pattern of ahead and behind representations resulted in a sequence of segment representations, as reflected in the spatial distribution of represented paths (Fig. 6b). The distribution demonstrated that paths represented by theta sequences preferentially occurred between landmarks on the maze, instead of across them. The distribution shown was normalized such that the paths represented at each location on the maze contributed equally to the distribution (occupancy normalization). This ensured that factors such as the rat consistently spending more time at a particular location on the maze did not misleadingly influence the distribution around that point. This distribution was robust across rats (Supplementary Fig. 2).
The observed distribution was compared to a distribution created by a shuffling procedure. Keeping the rat location for each theta cycle constant, we shuffled the decoded paths and randomly assigned them to the rat locations. This was done 100 times, and the average spatial distribution was plotted along with the observed distribution (Fig. 6b). As would be expected from the occupancy normalization, the shuffled distribution was relatively flat, showing that the observed spatial distribution of paths was a product of specific sequences occurring at particular locations on the maze. The shuffling procedure was also done for the average ahead and behind length over space plot (Fig. 6a,c), and, as expected, the shuffled distribution was flat.
Hippocampal cells show spatially localized firing (place fields)2,6. Place fields were defined as contiguous linear firing of a given cell (Online Methods). We compared the spatial distribution of represented paths (and ahead and behind lengths over space) shown in Figure 6 with the place field density, linearized place field length and the rat’s velocity. Neither place field density, place field length nor average velocity over space matched the shape of the path distribution or ahead and behind length (Supplementary Fig. 3). Place field length, plotted by location on the maze, showed some peaks in between landmarks; however, the overall shape and magnitude was not consistent with the ahead, behind and spatial path distributions. For example, peaks in the ahead and behind distributions between the maze start and turn 1 had peak-to-trough distances of ~11 cm and 10 cm, respectively (Fig. 6a,c), whereas the peak-to-trough distance of the place field length distribution was approximately half that distance (6 cm; Supplementary Fig. 3). Although some changes in ahead and behind length occurred at physical turns on the maze (such as the second choice point), other changes occurred on straight paths, without corresponding changes in orientation. For example, a large peak was seen in the ahead distribution just after the first feeder, and a large peak was seen in the behind distribution just at the second feeder, leading to an increased likelihood of representing the between-feeder zone, in the absence of a peak in the place field length distribution.
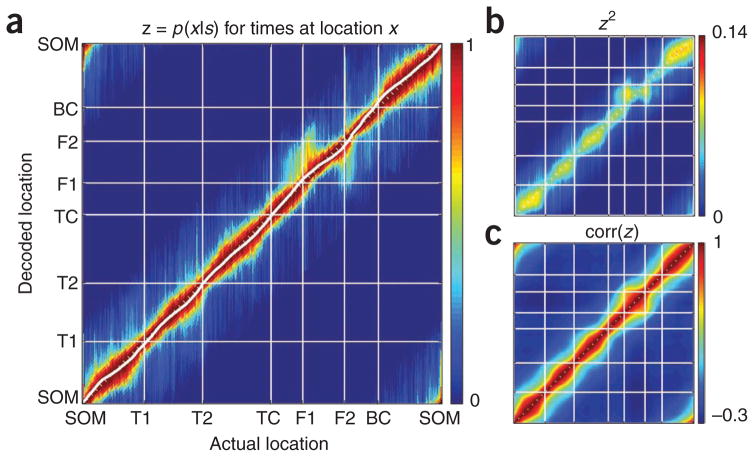
To further examine the representation of spatial paths as a function of location, we calculated the distribution of positions represented by all theta cycles occurring at that location. This produced a two-dimensional plot with each column representing the average region of space covered by theta sequences occurring at each maze location. Taking the circular mean of the locations covered by this plot produced small deviations above the x = y line where rats left landmarks and below the x = y line where they approached them (Fig. 7a). Deviations above the line indicate maze locations where representations were more ahead of the rat, and deviations below the line indicate locations where representations were more behind the rat.
Figure 7.
Segmented representation of space by theta sequences. (a) Average decoded spatial path during a theta sequence as a function of the rat’s location on the maze. Each column corresponds to the average decoded path when the rat was at a particular location on the maze. Thus, each column corresponds to the mean p(x|s), where x is the position along the maze and s is the spike sequence. The solid white line shows the weighted circular mean of each column. Dotted white line indicates the x = y line. (b) The chunking process can be seen in the pinches in spatial correlation between chunks (at landmark locations marked by intersections of white lines) found when we calculated the full joint probability by squaring the decoding matrix or by calculating the cross-spatial correlations of the decoding matrix (c). This analysis included 29,351 theta sequences.
These small deviations were amplified by creating the full joint probability distribution plot (Fig. 7b, which measures the probability that the rat is at location x and the decoded location is y given the sequence S, calculated by squaring the matrix in Fig. 7a) and by creating the correlation plot (Fig. 7c, which correlates each column of Fig. 7a with each column in Fig. 7a). The correlation plot is analogous to one used previously25, except that the correlation here was carried out on the decoding relationship rather than the raw population spiking vectors.
Given that theta sequences over-represented the space between landmarks and occur at a fast time scale, one would expect to see greater similarity in the population of cells firing in the space between landmarks, compared with the space across landmarks. This was tested by correlating the average population activity vectors at each location on the maze25. This approach did in fact show increased similarity of the average spatial population vectors in the space between landmarks (Supplementary Fig. 4). However, the segmentation of space by theta sequences (Fig. 7) indicates that not only was segmentation occurring when considering the average cellular activity over many experiences, but it occurred during each theta cycle, at a time scale that allowed for spike-timing dependent plasticity.
In summary, the spatial distribution of paths represented by theta sequences reflected an over-representation of certain segments or ‘chunks’ of the environment that were bounded by physical landmarks on the maze.
Lengths showed a complex relationship with acceleration
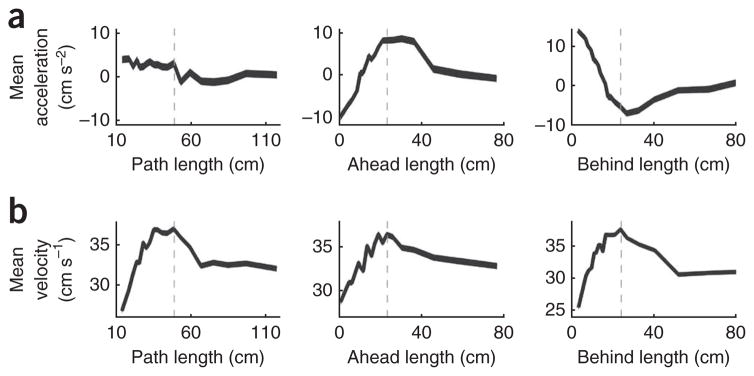
Whereas the spatial distribution of paths was only weakly visible in measures of place field density, place field length and rat velocity (see Supplementary Fig. 3), it did show striking similarities with rat acceleration (compare Fig. 6b with Supplementary Fig. 5a). The peaks and troughs of acceleration were generally aligned with peaks in ahead length and peaks in behind length, respectively (Fig. 6a,c), and ahead length was positively correlated (r = 0.41, P = 0.016), and behind length negatively correlated (r = −0.74, P < 0.01), with acceleration over space.
When looking at acceleration as a function of ahead and behind length, for shorter length sequences, as ahead length increased, acceleration also increased; conversely, as behind length increased, acceleration decreased (Fig. 8a). In contrast, acceleration remained relatively constant with increasing path length. Although these relationships were strong for shorter ahead and behind lengths, they leveled off to zero acceleration for longer lengths. This peak and the subsequent drop-off were also seen in the relationship between velocity and path, ahead and behind lengths (Fig. 8b); therefore, the peak of the velocity versus length distributions were identified and marked on both the acceleration and velocity plots. Ahead length and behind length for shorter sequences (70% of all sequences) showed a linear relationship with acceleration, but they were more variable as a function of acceleration for longer sequences.
Figure 8.
Relationships between rat behavior and the represented path. (a) Relationship between acceleration and path, ahead and behind lengths. (b) Relationship between velocity and path, ahead and behind lengths. Dashed lines in a and b indicate the peak of the velocity versus length relationships. As seen in a, acceleration is relatively constant with increasing path length. However, acceleration and ahead length are positively correlated, and acceleration and behind length are negatively correlated for shorter sequences (left of dashed line). This relationship changes for longer sequences (right of dashed line). Similarly, in b, velocity is positively correlated with path, ahead and behind length for shorter sequences, but the relationship changes for longer sequences. This analysis included 29,351 theta sequences. Line width indicates s.e.m. Note that some y axes have non-zero origins.
To determine whether the relationship between ahead and behind length and acceleration could account for the spatial distribution of represented paths, we built a model data set using acceleration and deceleration within each theta cycle to create ahead and behind representations. Using the actual behavioral data and the observed relationship between acceleration and ahead and behind length, we assigned each theta cycle an ahead and behind length on the basis of the rat’s acceleration during the cycle. We then plotted the spatial distribution of represented paths on the basis of this model and found that it did not match the segmented nature of the spatial distribution from the actual data (compare Fig. 6b with Supplementary Fig. 5b). This difference was not unexpected given the complex relationship between ahead and behind length and acceleration shown in Figure 8a.
Lengths showed a complex relationship with velocity
To determine the relationship between rat velocity and theta sequence lengths, we plotted velocity as a function of path, ahead and behind lengths (Fig. 8b). As each of the three lengths increased, there was initially an increase in velocity and subsequently a drop off. For shorter theta sequences, path, ahead and behind length all increased with increasing velocity; however, for longer sequences, velocity decreased or was relatively constant with changes in path, ahead and behind length. Thus, the relationship between velocity and path length cannot be completely explained by a greater sampling of place fields at higher speeds. Changes in cell firing with velocity also cannot explain the ahead-shifted and behind-shifted representations.
This observation and the presence of low- and high-velocity theta sequences presented earlier (Fig. 1c) indicate that there was not a simple relationship between velocity and the paths represented during theta sequences. Instances of both short and long paths occurred while rats were stationary or moving at low velocities in the environment, as previously observed24. Sequences also occurred as rats were moving at higher velocities. These sequences could be divided into a set of short-path sequences that increased in length with increasing rat velocity similar to those seen by others16,26 and a set of long-path sequences that decreased or remained constant in length with increasing velocity. It should be noted that theta, beta, gamma and SWR power were relatively constant with increasing path, ahead and behind length (Supplementary Fig. 6); thus, the local field potential (LFP) profile did not change as the represented path increased in length.
DISCUSSION
Hippocampal neural activity during each theta cycle sequentially represents a short path that begins just behind the animal and ends just in front of the animal8,10,13; however, we found considerable variability in the actual paths represented on each theta cycle, comparable to the differences seen on each phase-precession pass17. By examining each theta cycle independently, we found interesting relationships between the spatial representations and velocity, acceleration and physical landmarks on the maze. Most notably, we found that theta sequences represented the maze in segments. Consistent with this, we found that more space was represented ahead of the rat as it left physical landmarks on the maze, and more space behind the rat as it approached those same physical landmarks, leading to a segmentation of the spatial information. These results have implications for how the rat might organize its spatial and task knowledge.
Phase precession and neural sequences
As an animal passes through a cell’s place field toward a goal, the cell initially fires late in the theta cycle and then gradually fires earlier and earlier in the theta cycle as the animal traverses the place field13,15. At the population level, phase precession implies that compressed sequences of place cell activity emerge during each theta cycle8–14,16,26.
As noted in the introduction, evidence suggests that the compressed sequences are actually occurring independently on each theta cycle9,27. Our analyses exploit this and differ from previous studies by taking a dynamic view of the theta sequences; rather than averaging across passes, trials or cells, we calculated the path represented by each theta cycle sequence individually and then examined the distribution of properties of the individual sequences.
These sequences included both a set of sequences that were observed to represent longer spatial paths with increasing velocity (consistent with recent reports16,26) and a set of sequences that represented a variety of path lengths independent of velocity. This second set tended to occur while rats were paused in the environment, consistent with previously observed sweep sequences in CA3 (ref. 24), but this second set of sequences also occurred during movement. In general, we found a more complex relationship between the represented path and the rat’s behavior than have been observed in previous studies9,10,14,16,24,26. For example, long ahead representations were previously observed while animals were paused at a choice point24. In contrast, we observed that these longer sequences occurred at other locations on the maze as well, and they did not occur only when rats were paused. These longer sequences sometimes represented more space ahead of the rat and at other times represented more space behind the rat. Our analyses found that representations can start farther behind the rat or end farther ahead of the rat on particular theta cycles. Whereas adjacent theta cycle sequences are often very similar, they can on occasion differ substantially from theta cycle to theta cycle. These results indicated that theta sequences are not always a simple function of animal behavior and can flexibly represent different information on different theta cycles.
The sequence data presented in this paper suggested that there may be subtle changes in single-cell phase precession as animals approach or leave landmarks. As rats approached landmarks, sequences tended to include more space behind the rat (Fig. 6). Thus, we predict that single cells participating in these sequences would be more likely to be firing in the late aspects of their place fields and early phases of theta as animals approach landmarks. Similarly, as rats accelerated away from landmarks, sequences tended to include more space ahead of the rat, suggesting that cells participating in these sequences would be more likely to be firing during late phases of theta. Hints of such single cell effects may be seen in Supplementary Figure 7; however, they should be clearest in phase-precessing cells whose firing field covers a sufficiently large space to be bisected by a landmark, such as those in the ventral hippocampus or ventral striatum28,29. Indeed, single phase-precessing cells in the ventral striatum show transient phase changes that could be indicative of sequence effects29. However, a systematic test of this idea should establish if these cells participate in theta sequences, but this requires ensemble sizes beyond those currently available for ventral regions.
Representations of past and future
A number of studies have investigated whether hippocampal activity is reflective of the past and/or the future. Some of these studies have focused on the spatial path represented during neural activity observed in each theta cycle9,10,24,26. Other studies have analyzed hippocampal activity with respect to the animal’s past and future behaviors and have shown that the activity of individual place cells or place cell ensembles (at relatively slow time scales) is sometimes more reflective of past behaviors and is sometimes more reflective of future behaviors30–33. Thus, there is evidence that the hippocampus represents more than just the current state of the animal during behavior and does so at multiple timescales34.
We observed a diversity of sequence representations during theta, sometimes reflective of a ‘look ahead’ function and at other times reflective of ‘look behind’. We found that when an animal accelerates, the paths represented are shifted forward in space, possibly in preparation or anticipation of reaching a desired location, whereas when the animal decelerates, the paths represented shift backward in space, potentially in review of the experience. It is possible that the ahead representations observed here reflect a predictive recall process to aid in navigation26,27 whereas the behind representations reflect an encoding process to store what was just experienced35,36. This type of recall process could be cued by sensory input from upcoming landmarks, which were previously associated with locations in space37. However, it should be noted that the link between multicell sequences in hippocampus and memory processes such as encoding, retrieval, planning and anticipation is an active area of research and has not been proven.
Additionally, place fields are smaller when spikes are shifted to anticipate the animal’s location by 120 ms38, and approaches in two dimensions overlap only at the late phases of theta39,40, which suggests that portions of the place field may be thought of as representing approach to a future position. We add to this body of work by showing that at the theta time scale, the representation of future space and past space is dissociable and dependent on the animal’s acceleration and landmarks in the environment.
Information processing: theta and gamma
It has been proposed that the information represented during each theta cycle is discretized into parts by each cycle of gamma41,42. In this view, the information capacity of a given cycle of theta would be determined by the number of gamma cycles that occur over the course of the theta cycle. In support of these proposals, we observed a linear increase in theta period and the number of gamma cycles per theta cycle as the length of the represented path increased. It is possible that the increase in gamma cycles per theta cycle allows for an increase in the number of information processing steps, thus contributing to longer represented paths.
Behavioral and neural chunking
Chunking refers to a phenomenon in which information is grouped into intuitive, information-rich units. Such recoding is thought to increase information-handling abilities43 and to enable planning through subgoals44,45. It has been observed that individual ventral striatal neurons, which receive hippocampal projections, have receptive fields that represent navigationally relevant segments in an environment46. We did find some evidence that the hippocampal spatial place field density and place field length distributions reflected discrete segments (Supplementary Fig. 3), but the effect was small compared to the changes in ahead and behind length (Fig. 6). Instead, we found that sequences of place cell activity preferentially represented particular, navigationally relevant chunks of the environment. Thus, chunking was evident from the neural representation of space in the hippocampus.
Several studies have suggested that these neural sequences could be related to navigation8,26,47, planning24,42 or memory35–37. For example, evidence exists that systemic cannabinoid agonists disrupt both phase precession and behavioral performance while leaving average place fields unchanged47. However, it is unknown whether phase precession and behavioral performance are causally related and whether the disruption of phase precession extends to theta sequences. Nevertheless, our data suggest that the information being processed during each neural sequence expressed in each cycle of theta reflects cognitive segments of the task at hand.
From a behavioral perspective, chunking was also evident on the 2T task from the rat’s acceleration-deceleration profile (Supplementary Fig. 5). This was not surprising because landmarks on the maze were either turns or feeders, and it would be expected for the rats to slow down as they approached a turn and speed up afterwards. However, we also observed an increase in acceleration before arrival at the top corner of the maze, which implies that the behavioral chunking did not always perfectly align with the physical landmarks.
Although theta sequences preferentially represented segments of the environment, such segmentation was not seen in replay sequences during sharp wave ripples, occurring while animals were awake and paused at feeder locations on the 2T maze19. It was recently found that replay sequences can represent long paths in space and can occur across multiple ripple events, suggesting that replay events consist of chains of subsequences48. Thus, it is possible that theta sequences preferentially encode particular segments of experience, which are then concatenated during extended, multiripple replay events.
Segmentation of the environment by hippocampal theta sequences has implications for the encoding of experience and for how the hippocampus represents space. Because neural activity during theta represents experience as it occurs and operates at the timescale required for spike timing–dependent plasticity, it is thought that synaptic learning during each theta cycle is the basis for storing experience. Thus, theta sequences representing the environment in segments may result in the encoding of experience in chunks. Storing experience this way could be a coding scheme that the brain uses to organize vast amounts of information in a way that is efficient and behaviorally useful to animals.
ONLINE METHODS
Methods including the details about the subjects, surgery, recording, histology and the 2T task have been described previously19.
Subjects
Six male Fisher-Brown-Norway hybrid rats, aged 7–10 months at the start of recording sessions. All procedures were in accordance with the US National Institutes of Health guidelines for animal care and were approved by the Institutional Animal Care and Use Committee at the University of Minnesota. Rats were housed individually and maintained on a 12-h light-dark cycle, with the lights on during the day, when training and recording sessions took place.
Surgery, recording and histology
Three rats were each implanted with a single bundle 12-tetrode, 2-reference microdrive (Neuro-Hyperdrive; Kopf) directed toward the CA1 hippocampal (HC) subfield (3.8 mm posterior and 2.5 mm right-lateral from bregma). Another three rats were each implanted with a double bundle 12-tetrode, 2-reference microdrive directed toward CA1 and ventral striatum (only HC data analyzed here). One reference was placed in the corpus callosum for common-mode noise rejection. The other reference was placed in the hippocampal fissure. All theta LFP signals were analyzed from the second reference electrode (a tetrode with all four channels shorted together) in the hippocampal fissure. The results presented here are qualitatively similar when sequences included for analysis were only allowed one cell per tetrode.
Additional details of the surgery were as previously presented49. Neural activity was recorded, and spikes were sorted into putative cells49. All units were examined to have a reasonable isolation distance and L ratio50 and no spikes during the 2-ms refractory period. The median isolation distance and L ratio were 21 and 0.09, respectively. Only well-isolated units were included in the analyses. Recordings from an example tetrode are shown in Supplementary Figure 8.
After the recording phase of the experiment, rats were overdosed with sodium pentobarbital (150 mg/kg, Nembutal) and perfused intracardially with formalin. Their brains were then transferred to a 30% (wt/vol) sucrose-formalin solution, sectioned, and stained with cresyl violet with standard procedures. All HC recording locations were verified to lie in the CA1 region of the dorsal HC (see Supplementary Fig. 9).
The 2T task
The task was a continuous T maze with two choice points arranged in sequence (Fig. 1a). Food delivery occurred at two sites on each return rail, contingent on the rat’s choice at the second turn. Training and recording sessions lasted 40 min. Training on the task was carried out in three phases. In phase 1, rats were trained to run laps on one side of the maze, while the other side was blocked. Phase 1 continued until rats ran at least 40 laps on two consecutive days. In phase 2, the blocks were removed and on any given day, rats had to run all left laps (L), all right laps (R) or alternating left and right laps (A) in order to receive reward. After consistently getting 80% of the laps correct on all three task contingencies, rats were implanted with hyperdrives. After recovery from surgery, phase two of training resumed until the rats regained proficiency and tetrodes were in the cell layer. At this point, a 6-d sequence of recording sessions began (phase 3). During recording sessions, the task contingency changed approximately halfway through the session (mean, 18.07 min ± 1.13 min s.d.). There were six recording sessions to allow for all possible pairings of the three tasks (L-R, R-L, L-A, A-L, R-A, A-R).
Data analysis: place fields
Cells firing more than 15,000 spikes over the 40-min session (6.25 Hz) were excluded to filter putative inhibitory interneurons. Additionally, cells with <100 spikes (~0.04 Hz) were excluded. Sessions with >40 cells were considered for analysis (31 out of the 36 recording sessions). A total of 2,046 cells were isolated from 31 recording sessions; 1,881 cells satisfied the number-of-spikes criterion, and 1,470 cells contained at least one place field on the maze. Position along the maze was linearized separately for left and right laps, such that the rat’s position along a lap could be described by a scalar value51. Place fields were then identified as contiguous linear pixels (one linear pixel is ~3.5 cm along the linearized maze) with average activity >5% of the maximum rate observed over the session for any cell at any pixel (cells could have more than one place field, although place fields separated by only a single pixel were merged). Additionally, cells were required to have at least one place field including three adjacent pixels of >2 Hz to be included. Activity during low animal velocity was not excluded in place field identification. From the 1,470 place cells, 2,729 place fields (and place field centers) were identified. Centers were ordered from maze start to maze start in the direction the rats traveled around the maze.
Data analysis: detecting theta cycles with significant sequences
All theta signals were measured from the electrode placed in the hippocampal fissure taken relative to the electrode placed in the callosum. Individual theta cycles were separated peak to peak, which is the point of minimal cell firing. Peaks of the theta oscillation were identified by first filtering the recording from the reference electrode placed in the hippocampal fissure with a fourth-order Butterworth filter (pass-band between 6–12 Hz) to identify the theta oscillation. The peaks of the filtered signal were then identified by finding the maxima in the filtered signal. The point of minimal cell firing was consistent for the significant theta sequences used in analysis (see below) and was consistent with changes in ahead, behind, and path length (Supplementary Fig. 7). A sequence detection algorithm was then used to identify theta cycles whose spike sequence showed significant spatial and temporal structure. The algorithm was nearly identical to the one used previously to analyze replay sequences19, except that sequences were restricted to begin and end within the same theta cycle. The full algorithmic details are provided below. The algorithm used here and previously19 detected spatial and temporal structure in the pattern of place cell activity by comparing the times and place field centers of spike pairs. The algorithm was run twice for each session, once using left lap place field centers and once using right lap centers19. The resulting scores for each spike sequence were then analyzed using two independent bootstrapping procedures to identify sequences with significant spatial and temporal structure occurring during theta cycles with low sharp-wave ripple power. The sequence score procedure was only used to identify the set of candidate theta cycles to study.
Data analysis: sequence identification
The identification of theta sequences with significant structure was carried out in two steps. The first step was to compute the sequence score based on the neural activity in each theta cycle. The second step was to compare the score of each sequence against two distributions created using two separate bootstrapping procedures. These steps are described below.
Step 1: Sequence scoring
The algorithm described here is nearly identical to the algorithm used previously19. In this study, the algorithm was used to detect coherent sequences of place cell activity during each cycle of the theta oscillation, rather than replay events during the awake state. The sequence score of place cell activity within each theta cycle was calculated as follows. Using the place field centers and spike times, each spike in the theta cycle was pairwise compared with other spikes occurring in the same theta cycle. If the place field center corresponding to spike A was traversed before the place field center for spike B and spike A occurred before spike B, the sequence score was +1, otherwise the score was −1. For all spikes in a given theta cycle, all pairwise comparisons were summed to determine the cumulative score of the sequence.
The basic sequence scoring algorithm was as follows.
Given:
Spike times: T vector: [nSpikes × 1]
Spike place field centers: C vector: [nSpikes × 1]
Compute:
Time difference for each spike pair: ΔT matrix: [nSpikes × nSpikes]
Field center distance for each spike pair: ΔC matrix: [nSpikes × nSpikes]
Element-wise multiply ΔT and ΔC to create ΔTC matrix: [nSpikes × nSpikes]
Binarize ΔTC. For each element in ΔTC: if element is positive, replace with ‘+1’; if element is negative, replace with ‘−1’.
Sum over all elements in ΔTC to get the sequence score.
A positive sequence score indicates that the sequence is ‘forward’ ordered.
The scoring presented above requires that each spike be associated with a single place field. For cells with multiple place fields, it is unclear which place field is being represented by a spike. To address this issue, the algorithm assigned each spike from a multifield cell to the place field which best fit with the rest of the firing sequence. This was done by maximizing the score over each place field available to a spike. Always picking the place field that optimizes the sequence score could result in artificially high sequence scores. To provide a fair and statistically justified comparison, this same score maximization procedure was conducted for the control sequences produced in the bootstrapping procedure described below.
Step 2: Sequence significance testing
Next, each theta sequence was individually analyzed to identify significant structure using two independent bootstrapping procedures. The first method preserved the identity of the cell to which each spike belonged and randomly shuffled the spike times for each event 300 times. Each shuffled event had its sequence score recomputed, once again with spikes from cells with multiple place fields being assigned to the place field center that maximized the sequence score. This ensured that there was no bias resulting from assigning spikes to fields that maximized the sequence score. The second bootstrapping procedure preserved the spike trains, but randomly reassigned each spike train to a different cell’s firing field(s)48. Each event was shuffled 300 times, once again with spikes from cells with multiple place fields being assigned to the place field center that maximized the sequence score. If the unshuffled sequence score was >95% of both independent sets of shuffled sequences, the sequence was deemed significant and included for analysis. These methods ensured that only significant (P < 0.05) theta sequences were included in the analysis.
Data analysis: Bayesian decoding
For theta cycles with significant sequential structure, the represented path in space was determined using a Bayesian decoding approach52. Decoding was carried out on each theta cycle using a 40-ms sliding time window shifted by 10 ms at each step. Decoding was restricted to an ~190-cm region (half a lap) centered at the rat’s location. Thus, decoding over each theta cycle resulted in an nTimeBins × nSpaceBins matrix of probabilities, with each row containing the probability distribution over space for a particular time bin. All spikes were included in the Bayesian analyses, whether they contributed to the sequence score or not.
Data analysis: ahead, behind and path lengths
To detect the beginning of the spatial path represented, the probability distributions over space for the first half of the theta cycle (matrix size: nTimeBins/2 × nSpaceBins) were summed to create a single probability distribution over space (an nSpaceBins-element vector). The 5% tail of this distribution (tail on the end of the distribution that is behind the rat) was located, providing the beginning location of the spatial path. The distance between this point and the location of the rat was defined as the behind length, as it measures the distance the sequence travels from behind the rat to the rat’s location. Similarly, the end of the represented path was determined by summing the probability distributions over space for the second half of the theta cycle and locating the 5% tail of that distribution (tail on the end of the distribution that is ahead of the rat). The distance between the location of the rat and this point was defined as the ahead length, as it measures the distance ahead of the rat’s location that the sequence progresses. Finally, the total distance from the start of the sequence to the end of the sequence was defined as the path length.
The Bayesian decoding results were robust to the use of other time windows (20 and 30 ms) or other tail sizes (10%, 15% and 20%) and were similar when the first and last time bin were used for determining the ends of the spatial path or when the spatial distributions were summed over the entire theta cycle rather than over the first and second half separately. Furthermore, the results obtained using this Bayesian decoding approach were qualitatively replicated using a separate, non-Bayesian approach for detecting the represented path23.
Data analysis: location, velocity and acceleration
The rat’s location was defined as the location at the middle of the theta cycle (at which cell firing was maximal, Supplementary Fig. 7); however, all results are qualitatively unchanged if the rat’s location was taken as at another point (beginning or end) of the cycle. Velocity was measured as the derivative of the sequence of locations53, and acceleration was measured as the derivative of the velocity sequences53.
Supplementary Material
Acknowledgments
We thank C. Boldt, K.D. Seeland and A.P. Steiner for technical assistance and A. Johnson and the members of the Redish lab for discussion. This work was supported by US National Institutes of Health grants (R01 MH-080318 and F30 MH-091821), the Pennsylvania Department of Health and a US National Science Foundation Integrative Graduate Education and Research Traineeship grant (DGE-0549352).
Footnotes
Note: Supplementary information is available in the online version of the paper.
AUTHOR CONTRIBUTIONS
A.S.G., M.A.A.v.d.M. and A.D.R. designed the experiment; D.S.T. and A.D.R. supervised the project; A.S.G. and M.A.A.v.d.M. carried out the experiments; A.S.G. analyzed the data; and A.S.G., M.A.A.v.d.M., D.S.T. and A.D.R. wrote the paper.
COMPETING FINANCIAL INTERESTS
The authors declare no competing financial interests.
References
- 1.Cohen NJ, Eichenbaum H. Memory, Amnesia, and the Hippocampal System. MIT Press; Cambridge, Massachusetts, USA: 1993. [Google Scholar]
- 2.O’Keefe J, Nadel L. The Hippocampus as a Cognitive Map. Clarendon Press; Oxford, UK: 1978. [Google Scholar]
- 3.Hassabis D, Kumaran D, Vann SD, Maguire EA. Patients with hippocampal amnesia cannot imagine new experiences. Proc Natl Acad Sci USA. 2007;104:1726–1731. doi: 10.1073/pnas.0610561104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schacter DL, Addis DR, Buckner RL. Remembering the past to imagine the future: the prospective brain. Nat Rev Neurosci. 2007;8:657–661. doi: 10.1038/nrn2213. [DOI] [PubMed] [Google Scholar]
- 5.Morris G, Nevet A, Arkadir D, Vaadia E, Bergman H. Midbrain dopamine neurons encode decisions for future action. Nat Neurosci. 2006;9:1057–1063. doi: 10.1038/nn1743. [DOI] [PubMed] [Google Scholar]
- 6.Redish AD. Beyond the Cognitive Map: From Place Cells to Episodic Memory. MIT Press; Cambridge, Massachusetts, USA: 1999. [Google Scholar]
- 7.Vanderwolf CH. Hippocampal electrical activity and voluntary movement in the rat. Electroencephalogr Clin Neurophysiol. 1969;26:407–418. doi: 10.1016/0013-4694(69)90092-3. [DOI] [PubMed] [Google Scholar]
- 8.Skaggs WE, McNaughton BL, Wilson MA, Barnes CA. Theta phase precession in hippocampal neuronal populations and the compression of temporal sequences. Hippocampus. 1996;6:149–172. doi: 10.1002/(SICI)1098-1063(1996)6:2<149::AID-HIPO6>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 9.Dragoi G, Buzsaki G. Temporal encoding of place sequences by hippocampal cell assemblies. Neuron. 2006;50:145–157. doi: 10.1016/j.neuron.2006.02.023. [DOI] [PubMed] [Google Scholar]
- 10.Foster DJ, Wilson MA. Hippocampal theta sequences. Hippocampus. 2007;17:1093–1099. doi: 10.1002/hipo.20345. [DOI] [PubMed] [Google Scholar]
- 11.Tsodyks MV, Skaggs WE, Sejnowski TJ, McNaughton BL. Population dynamics and theta rhythm phase precession of hippocampal place cell firing: a spiking neuron model. Hippocampus. 1996;6:271–280. doi: 10.1002/(SICI)1098-1063(1996)6:3<271::AID-HIPO5>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 12.Jensen O, Lisman JE. Hippocampal CA3 region predicts memory sequences: accounting for the phase precession of place cells. Learn Mem. 1996;3:279–287. doi: 10.1101/lm.3.2-3.279. [DOI] [PubMed] [Google Scholar]
- 13.Maurer AP, McNaughton BL. Network and intrinsic cellular mechanisms underlying theta phase precession of hippocampal neurons. Trends Neurosci. 2007;30:325–333. doi: 10.1016/j.tins.2007.05.002. [DOI] [PubMed] [Google Scholar]
- 14.Malhotra S, Cross RWA, van der Meer MAA. Theta phase precession beyond the hippocampus. Rev Neurosci. 2012;23:39–65. doi: 10.1515/revneuro-2011-0064. [DOI] [PubMed] [Google Scholar]
- 15.O’Keefe J, Recce M. Phase relationship between hippocampal place units and the EEG theta rhythm. Hippocampus. 1993;3:317–330. doi: 10.1002/hipo.450030307. [DOI] [PubMed] [Google Scholar]
- 16.Geisler C, Robbe D, Zugaro M, Sirota A, Buzsáki G. Hippocampal place cell assemblies are speed-controlled oscillators. Proc Natl Acad Sci USA. 2007;104:8149–8154. doi: 10.1073/pnas.0610121104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schmidt R, et al. Single-trial phase precession in the hippocampus. J Neurosci. 2009;29:13232–13241. doi: 10.1523/JNEUROSCI.2270-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Geisler C, et al. Temporal delays among place cells determine the frequency of population theta oscillations in the hippocampus. Proc Natl Acad Sci USA. 2010;107:7957–7962. doi: 10.1073/pnas.0912478107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gupta AS, Van der Meer MAA, Touretzky DS, Redish AD. Hippocampal replay is not a simple function of experience. Neuron. 2010;65:695–705. doi: 10.1016/j.neuron.2010.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Foster DJ, Wilson MA. Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature. 2006;440:680–683. doi: 10.1038/nature04587. [DOI] [PubMed] [Google Scholar]
- 21.Diba K, Buzsáki G. Forward and reverse hippocampal place-cell sequences during ripples. Nat Neurosci. 2007;10:1241–1242. doi: 10.1038/nn1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Carr MF, Jadhav SP, Frank LM. Hippocampal replay in the awake state: a potential substrate for memory consolidation and retrieval. Nat Neurosci. 2011;14:147–153. doi: 10.1038/nn.2732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gupta AS. PhD thesis. Carnegie Mellon Univ; 2011. Behavioral Correlates of Hippocampal Neural Sequences. [Google Scholar]
- 24.Johnson A, Redish AD. Neural ensembles in CA3 transiently encode paths forward of the animal at a decision point. J Neurosci. 2007;27:12176–12189. doi: 10.1523/JNEUROSCI.3761-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gothard KM, Hoffman KL, Battaglia FP, McNaughton BL. Dentate gyrus and CA1 ensemble activity during spatial reference frame shifts in the presence and absence of visual input. J Neurosci. 2001;21:7284–7292. doi: 10.1523/JNEUROSCI.21-18-07284.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Maurer AP, Burke SN, Lipa P, Skaggs WE, Barnes CA. Greater running speeds result in altered hippocampal phase sequence dynamics. Hippocampus. 2012;22:737–747. doi: 10.1002/hipo.20936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lisman J, Redish AD. Prediction, sequences and the hippocampus. Philos Trans R Soc Lond B Biol Sci. 2009;364:1193–1201. doi: 10.1098/rstb.2008.0316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Royer S, Sirota A, Patel J, Buzsáki G. Distinct representations and theta dynamics in dorsal and ventral hippocampus. J Neurosci. 2010;30:1777–1787. doi: 10.1523/JNEUROSCI.4681-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.van der Meer MAA, Redish AD. Theta phase precession in rat ventral striatum links place and reward information. J Neurosci. 2011;31:2843–2854. doi: 10.1523/JNEUROSCI.4869-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wood ER, Dudchenko PA, Robitsek RJ, Eichenbaum H. Hippocampal neurons encode information about different types of memory episodes occurring in the same location. Neuron. 2000;27:623–633. doi: 10.1016/s0896-6273(00)00071-4. [DOI] [PubMed] [Google Scholar]
- 31.Frank LM, Brown EN, Wilson M. Trajectory encoding in the hippocampus and entorhinal cortex. Neuron. 2000;27:169–178. doi: 10.1016/s0896-6273(00)00018-0. [DOI] [PubMed] [Google Scholar]
- 32.Ferbinteanu J, Shapiro ML. Prospective and retrospective memory coding in the hippocampus. Neuron. 2003;40:1227–1239. doi: 10.1016/s0896-6273(03)00752-9. [DOI] [PubMed] [Google Scholar]
- 33.Ji D, Wilson MA. Firing rate dynamics in the hippocampus induced by trajectory learning. J Neurosci. 2008;28:4679–4689. doi: 10.1523/JNEUROSCI.4597-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yartsev MM. Dissociating the effects of past and future on neural encoding of sequences in the hippocampus. J Neurosci. 2008;28:8383–8384. doi: 10.1523/JNEUROSCI.2442-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Buzsáki G. Theta rhythm of navigation: link between path integration and landmark navigation, episodic and semantic memory. Hippocampus. 2005;15:827–840. doi: 10.1002/hipo.20113. [DOI] [PubMed] [Google Scholar]
- 36.Hasselmo ME. The role of hippocampal regions CA3 and CA1 in matching entorhinal input with retrieval of associations between objects and context: Theoretical comment on Lee et al. Behav Neurosci. 2005;119:342–345. doi: 10.1037/0735-7044.119.1.342. [DOI] [PubMed] [Google Scholar]
- 37.Hasselmo ME. A model of episodic memory: mental time travel along encoded trajectories using grid cells. Neurobiol Learn Mem. 2009;92:559–573. doi: 10.1016/j.nlm.2009.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Muller RU, Kubie JL. The firing of hippocampal place cells predicts the future position of freely moving rats. J Neurosci. 1989;9:4101–4110. doi: 10.1523/JNEUROSCI.09-12-04101.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Battaglia FP, Sutherland GR, McNaughton BL. Local sensory cues and place cell directionality: additional evidence of prospective coding in the hippocampus. J Neurosci. 2004;24:4541–4550. doi: 10.1523/JNEUROSCI.4896-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huxter JR, Senior TJ, Allen K, Csicsvari J. Theta phase–specific codes for two-dimensional position, trajectory and heading in the hippocampus. Nat Neurosci. 2008;11:587–594. doi: 10.1038/nn.2106. [DOI] [PubMed] [Google Scholar]
- 41.Lisman JE, Idiart MA. Storage of 7 ± 2 short-term memories in oscillatory subcycles. Science. 1995;267:1512–1515. doi: 10.1126/science.7878473. [DOI] [PubMed] [Google Scholar]
- 42.Lisman J. The theta/gamma discrete phase code occurring during the hippocampal phase precession may be a more general brain coding scheme. Hippocampus. 2005;15:913–922. doi: 10.1002/hipo.20121. [DOI] [PubMed] [Google Scholar]
- 43.Miller GA. The magical number seven, plus or minus two: some limits on our capacity for processing information. Psychol Rev. 1956;63:81–97. [PubMed] [Google Scholar]
- 44.Newell A. Unified Theories of Cognition. Harvard University Press; 1990. [Google Scholar]
- 45.Newell A, Simon H. Human Problem Solving. Prentice-Hall; Englewood Cliffs, New Jersey, USA: 1972. [Google Scholar]
- 46.Mulder AB, Tabuchi E, Wiener SI. Neurons in hippocampal afferent zones of rat striatum parse routes into multi-pace segments during maze navigation. Eur J Neurosci. 2004;19:1923–1932. doi: 10.1111/j.1460-9568.2004.03301.x. [DOI] [PubMed] [Google Scholar]
- 47.Robbe D, Buzsáki G. Alteration of theta timescale dynamics of hippocampal place cells by a cannabinoid is associated with memory impairment. J Neurosci. 2009;29:12597–12605. doi: 10.1523/JNEUROSCI.2407-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Davidson TJ, Kloosterman F, Wilson MA. Hippocampal replay of extended experience. Neuron. 2009;63:497–507. doi: 10.1016/j.neuron.2009.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jackson JC, Johnson A, Redish AD. Hippocampal sharp waves and reactivation during awake states depend on repeated sequential experience. J Neurosci. 2006;26:12415–12426. doi: 10.1523/JNEUROSCI.4118-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schmitzer-Torbert NC, Jackson J, Henze D, Harris K, Redish AD. Quantitative measures of cluster quality for use in extracellular recordings. Neuroscience. 2005;131:1–11. doi: 10.1016/j.neuroscience.2004.09.066. [DOI] [PubMed] [Google Scholar]
- 51.Schmitzer-Torbert N, Redish AD. Neuronal activity in the rodent dorsal striatum in sequential navigation: separation of spatial and reward responses on the multiple T task. J Neurophysiol. 2004;91:2259–2272. doi: 10.1152/jn.00687.2003. [DOI] [PubMed] [Google Scholar]
- 52.Zhang K, Ginzburg I, McNaughton BL, Sejnowski TJ. Interpreting neuronal population activity by reconstruction: unified framework with application to hippocampal place cells. J Neurophysiol. 1998;79:1017–1044. doi: 10.1152/jn.1998.79.2.1017. [DOI] [PubMed] [Google Scholar]
- 53.Janabi-Sharifi F, Hayward V, Chen CSJ. Discrete-time adaptive windowing for velocity estimation. IEEE Trans Control Syst Technol. 2000;8:1003–1009. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.