Abstract
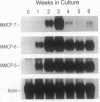
A gene that encodes mouse mast cell protease (mMCP) 7 (also known as mouse mast cell tryptase 2) was isolated by genomic cloning with a cDNA that encodes mMCP-6, a tryptase in serosal mast cells. cDNAs encoding mMCP-7 were isolated from a bone-marrow-derived mast cell cDNA library. The mMCP-7 gene spans 2.3 kilobases and contains five exons rather than six, as found in the mMCP-6 and human mast cell tryptase I genes. Comparison of the 5' end of the transcript with the genomic sequence indicated that the region corresponding to the first intron in the mMCP-6 and human tryptase I genes is not spliced during transcription of mMCP-7 mRNA because of a point mutation at the intron 1 acceptor splice site; this results in a 5' untranslated region of 195 nucleotides, which is longer than that of any other known mast cell-specific transcript. mMCP-7 is 71-76% homologous with mMCP-6 and with dog and human mast cell tryptases, and it is the most acidic mast cell protease, with an overall net charge of -10. RNA blot analyses revealed that the mMCP-7 gene is transcribed in bone-marrow-derived mast cells but is not transcribed in mature serosal mast cells or in mucosal mast cell-enriched intestinal tissue of Trichinella spiralis-infected mice. Transcription of the mMCP-7 gene by differentiating bone-marrow-derived mast cells occurred within 1 week of bone-marrow culture but decreased dramatically after 3 weeks. Thus, the mMCP-7 gene displays a number of unusual structural characteristics and is distinctive in its transient and selective expression in immature mast cells maintained in interleukin 3-enriched medium.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bories D., Raynal M. C., Solomon D. H., Darzynkiewicz Z., Cayre Y. E. Down-regulation of a serine protease, myeloblastin, causes growth arrest and differentiation of promyelocytic leukemia cells. Cell. 1989 Dec 22;59(6):959–968. doi: 10.1016/0092-8674(89)90752-6. [DOI] [PubMed] [Google Scholar]
- Breathnach R., Chambon P. Organization and expression of eucaryotic split genes coding for proteins. Annu Rev Biochem. 1981;50:349–383. doi: 10.1146/annurev.bi.50.070181.002025. [DOI] [PubMed] [Google Scholar]
- Campbell E. J., Cury J. D., Shapiro S. D., Goldberg G. I., Welgus H. G. Neutral proteinases of human mononuclear phagocytes. Cellular differentiation markedly alters cell phenotype for serine proteinases, metalloproteinases, and tissue inhibitor of metalloproteinases. J Immunol. 1991 Feb 15;146(4):1286–1293. [PubMed] [Google Scholar]
- Chen E. Y., Seeburg P. H. Supercoil sequencing: a fast and simple method for sequencing plasmid DNA. DNA. 1985 Apr;4(2):165–170. doi: 10.1089/dna.1985.4.165. [DOI] [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Cromlish J. A., Seidah N. G., Marcinkiewicz M., Hamelin J., Johnson D. A., Chrétien M. Human pituitary tryptase: molecular forms, NH2-terminal sequence, immunocytochemical localization, and specificity with prohormone and fluorogenic substrates. J Biol Chem. 1987 Jan 25;262(3):1363–1373. [PubMed] [Google Scholar]
- Ghildyal N., McNeil H. P., Gurish M. F., Austen K. F., Stevens R. L. Transcriptional regulation of the mucosal mast cell-specific protease gene, MMCP-2, by interleukin 10 and interleukin 3. J Biol Chem. 1992 Apr 25;267(12):8473–8477. [PubMed] [Google Scholar]
- Ghildyal N., McNeil H. P., Stechschulte S., Austen K. F., Silberstein D., Gurish M. F., Somerville L. L., Stevens R. L. IL-10 induces transcription of the gene for mouse mast cell protease-1, a serine protease preferentially expressed in mucosal mast cells of Trichinella spiralis-infected mice. J Immunol. 1992 Sep 15;149(6):2123–2129. [PubMed] [Google Scholar]
- Gurish M. F., Ghildyal N., McNeil H. P., Austen K. F., Gillis S., Stevens R. L. Differential expression of secretory granule proteases in mouse mast cells exposed to interleukin 3 and c-kit ligand. J Exp Med. 1992 Apr 1;175(4):1003–1012. doi: 10.1084/jem.175.4.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang R. Y., Blom T., Hellman L. Cloning and structural analysis of MMCP-1, MMCP-4 and MMCP-5, three mouse mast cell-specific serine proteases. Eur J Immunol. 1991 Jul;21(7):1611–1621. doi: 10.1002/eji.1830210706. [DOI] [PubMed] [Google Scholar]
- Johnson D. A., Barton G. J. Mast cell tryptases: examination of unusual characteristics by multiple sequence alignment and molecular modeling. Protein Sci. 1992 Mar;1(3):370–377. doi: 10.1002/pro.5560010309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNeil H. P., Austen K. F., Somerville L. L., Gurish M. F., Stevens R. L. Molecular cloning of the mouse mast cell protease-5 gene. A novel secretory granule protease expressed early in the differentiation of serosal mast cells. J Biol Chem. 1991 Oct 25;266(30):20316–20322. [PubMed] [Google Scholar]
- Miller J. S., Moxley G., Schwartz L. B. Cloning and characterization of a second complementary DNA for human tryptase. J Clin Invest. 1990 Sep;86(3):864–870. doi: 10.1172/JCI114786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller J. S., Westin E. H., Schwartz L. B. Cloning and characterization of complementary DNA for human tryptase. J Clin Invest. 1989 Oct;84(4):1188–1195. doi: 10.1172/JCI114284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nabel G., Galli S. J., Dvorak A. M., Dvorak H. F., Cantor H. Inducer T lymphocytes synthesize a factor that stimulates proliferation of cloned mast cells. Nature. 1981 May 28;291(5813):332–334. doi: 10.1038/291332a0. [DOI] [PubMed] [Google Scholar]
- Nakano T., Sonoda T., Hayashi C., Yamatodani A., Kanayama Y., Yamamura T., Asai H., Yonezawa T., Kitamura Y., Galli S. J. Fate of bone marrow-derived cultured mast cells after intracutaneous, intraperitoneal, and intravenous transfer into genetically mast cell-deficient W/Wv mice. Evidence that cultured mast cells can give rise to both connective tissue type and mucosal mast cells. J Exp Med. 1985 Sep 1;162(3):1025–1043. doi: 10.1084/jem.162.3.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce J. H., Di Fiore P. P., Aaronson S. A., Potter M., Pumphrey J., Scott A., Ihle J. N. Neoplastic transformation of mast cells by Abelson-MuLV: abrogation of IL-3 dependence by a nonautocrine mechanism. Cell. 1985 Jul;41(3):685–693. doi: 10.1016/s0092-8674(85)80049-0. [DOI] [PubMed] [Google Scholar]
- Razin E., Ihle J. N., Seldin D., Mencia-Huerta J. M., Katz H. R., LeBlanc P. A., Hein A., Caulfield J. P., Austen K. F., Stevens R. L. Interleukin 3: A differentiation and growth factor for the mouse mast cell that contains chondroitin sulfate E proteoglycan. J Immunol. 1984 Mar;132(3):1479–1486. [PubMed] [Google Scholar]
- Reynolds D. S., Gurley D. S., Austen K. F., Serafin W. E. Cloning of the cDNA and gene of mouse mast cell protease-6. Transcription by progenitor mast cells and mast cells of the connective tissue subclass. J Biol Chem. 1991 Feb 25;266(6):3847–3853. [PubMed] [Google Scholar]
- Reynolds D. S., Serafin W. E., Faller D. V., Wall D. A., Abbas A. K., Dvorak A. M., Austen K. F., Stevens R. L. Immortalization of murine connective tissue-type mast cells at multiple stages of their differentiation by coculture of splenocytes with fibroblasts that produce Kirsten sarcoma virus. J Biol Chem. 1988 Sep 5;263(25):12783–12791. [PubMed] [Google Scholar]
- Reynolds D. S., Stevens R. L., Lane W. S., Carr M. H., Austen K. F., Serafin W. E. Different mouse mast cell populations express various combinations of at least six distinct mast cell serine proteases. Proc Natl Acad Sci U S A. 1990 Apr;87(8):3230–3234. doi: 10.1073/pnas.87.8.3230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serafin W. E., Reynolds D. S., Rogelj S., Lane W. S., Conder G. A., Johnson S. S., Austen K. F., Stevens R. L. Identification and molecular cloning of a novel mouse mucosal mast cell serine protease. J Biol Chem. 1990 Jan 5;265(1):423–429. [PubMed] [Google Scholar]
- Serafin W. E., Sullivan T. P., Conder G. A., Ebrahimi A., Marcham P., Johnson S. S., Austen K. F., Reynolds D. S. Cloning of the cDNA and gene for mouse mast cell protease 4. Demonstration of its late transcription in mast cell subclasses and analysis of its homology to subclass-specific neutral proteases of the mouse and rat. J Biol Chem. 1991 Jan 25;266(3):1934–1941. [PubMed] [Google Scholar]
- Spiegelman B. M., Frank M., Green H. Molecular cloning of mRNA from 3T3 adipocytes. Regulation of mRNA content for glycerophosphate dehydrogenase and other differentiation-dependent proteins during adipocyte development. J Biol Chem. 1983 Aug 25;258(16):10083–10089. [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5201–5205. doi: 10.1073/pnas.77.9.5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trong H. L., Newlands G. F., Miller H. R., Charbonneau H., Neurath H., Woodbury R. G. Amino acid sequence of a mouse mucosal mast cell protease. Biochemistry. 1989 Jan 10;28(1):391–395. doi: 10.1021/bi00427a054. [DOI] [PubMed] [Google Scholar]
- Vanderslice P., Ballinger S. M., Tam E. K., Goldstein S. M., Craik C. S., Caughey G. H. Human mast cell tryptase: multiple cDNAs and genes reveal a multigene serine protease family. Proc Natl Acad Sci U S A. 1990 May;87(10):3811–3815. doi: 10.1073/pnas.87.10.3811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanderslice P., Craik C. S., Nadel J. A., Caughey G. H. Molecular cloning of dog mast cell tryptase and a related protease: structural evidence of a unique mode of serine protease activation. Biochemistry. 1989 May 16;28(10):4148–4155. doi: 10.1021/bi00436a004. [DOI] [PubMed] [Google Scholar]
- Yurt R. W., Leid R. W., Jr, Austen K. F. Native heparin from rat peritoneal mast cells. J Biol Chem. 1977 Jan 25;252(2):518–521. [PubMed] [Google Scholar]
- von Heijne G. How signal sequences maintain cleavage specificity. J Mol Biol. 1984 Feb 25;173(2):243–251. doi: 10.1016/0022-2836(84)90192-x. [DOI] [PubMed] [Google Scholar]