Abstract
Context:
Phosphaturic mesenchymal tumors (PMTs) are small, typically difficult to localize, and express somatostatin receptors. Recent work suggests imaging studies using 68Gallium (68Ga)-conjugated somatostatin peptide analogues, such as 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA)TATE, which enables somatostatin receptor imaging with positron emission tomography (PET), may be useful at identifying these tumors.
Objective:
Our objective was to evaluate the use of 68Ga-DOTATATE PET/computed tomography (CT) for tumor localization in tumor-induced osteomalacia (TIO).
Design:
This was a single-center prospective study of patients with TIO.
Setting:
The study was conducted at the National Institutes of Health Clinical Center between February 2014 and February 2015.
Subjects:
Eleven subjects (six females, five males) with TIO were included.
Intervention:
Subjects underwent 68Ga-DOTATATE PET/CT in addition to 111In-pentetreotide single-photon emission CT (Octreoscan- SPECT/CT) and fluorodeoxyglucose-PET/CT (18F FDG-PET/CT) scan.
Main Outcome Measures:
Localization of PMTs on the previously described imaging modalities were determined.
Results:
The tumor was successfully localized in 6/11 (54.5%) subjects (one was metastatic). The tumor was identified by 68Ga-DOTATATE in all six cases. Both Octreoscan-SPECT/CT and 18F FDG-PET each identified the tumor in 4/6. In no cases was 68Ga-DOTATATE the only imaging study to identify the tumor.
Conclusions:
In this first prospective study comparing 68Ga-DOTATATE PET/CT to Octreoscan-SPECT/CT and 18F FDG-PET in TIO localization, 68Ga-DOTATATE PET/CT demonstrated the greatest sensitivity and specificity, suggesting that it may be the best single study for localization of PMTs in TIO.
Tumor-induced osteomalacia is caused by FGF23-secreting tumors that are notoriously difficult to localize. In this first, prospective study of patients with TIO, we have found that 68Ga-DOTATATE as a single study is superior to Octreoscan or FDGPET/ CT.
Tumor-induced osteomalacia (TIO) is a paraneoplastic disorder caused by small mesenchymal tumors that produce high levels of the hormone fibroblast-growth-factor 23 (FGF23). Complete surgical resection leads to cure; however, these tumors are notoriously difficult to locate due to their small size.
In our recent series of subjects with TIO, we found that successful localization was achieved in only 61% of subjects, even with a combination of localization studies that included two functional imaging modalities, 111In-pentetreotide single-photon emission computed tomography (SPECT) (Octreoscan-SPECT/CT) and fluorodeoxyglucose-positron emission tomography (18F FDG-PET/CT), anatomical localization studies (magnetic tomography imaging [MRI] and CT), and as needed, selective venous sampling (1). Recent reports suggest 68Gallium (68Ga)-conjugated somatostatin peptide analogues, such as DOTATATE PET/CT may be useful at identifying phosphaturic mesenchymal tumors (PMTs), but in none of these reports was there a direct comparison to conventional studies, including both Octreoscan-SPECT/CT and 18F FDG-PET together (2–4). The possible superiority of 68Ga- DOTATATE lies in its higher affinity than Octreoscan for somatostatin receptors 2 and 5, which PMTs express (3, 5). Furthermore, the optimal physical characteristics of 68Ga (PET emitter) enables faster acquisition of images and lower radiation dose, in addition to the vastly superior performance of PET imaging over scintigraphy in the case of Octreoscan-SPECT/CT. In this prospective study, we compared 68Ga-DOTATATE PET/CT vs Octreoscan SPECT/CT and 18F FDG-PET/CT scan in subjects with TIO referred to a tertiary referral center for tumor localization over a 12-month period.
Subjects and Methods
The diagnosis of TIO was based upon an acquired condition of hypophosphatemia from renal phosphate wasting and an elevated blood FGF23. The onset of symptoms (weakness, multiple fractures) occurred within 2–24 years of presentation, with a median of 7 years (Table 1). All patients had a biochemical profile consistent with TIO, including low serum phosphorus and 1,25-dihdroxyvitamin D, low tubular reabsorption of phosphorus, and an elevated blood FGF23, as measured by an intact FGF23 ELISA assay (Kainos or Immutopics) or C-terminus ELISA assay (Immutopics) for patient 1. All subjects had failed tumor localization at least once before referral. Four subjects had had prior surgical attempts. Subject 4, who was ultimately shown to have metastatic disease, had undergone four failed surgical attempts previously (all in the right mandible, where the primary tumor originated). Subject 5 had had an unsuccessful attempt at tumor resection in the right foot based on false-positive 18F FDG-PET/CT imaging. Subject 6 had had a PMT resected from the left femur that resulted in normalization of blood phosphate and FGF23. However, tumor margins were not identified and there was local recurrence, which resulted in the need for reoperation. Subject 9 had had a PMT resected from the maxilla, and although tumor margins showed PMT, blood phosphate and FGF23 initially normalized and she was deemed cured. Disease recurred several years later at the same location.
Table 1.
Characteristics and Imaging Results of 11 Subjects With Tumor-Induced Osteomalacia
| Sex (Age of Onset, y) | Symptom Duration (mo) | Initial FGF23a | 68Ga-DOTATATE PET/CT | OctreoScan SPECT/CT | 18F FDG-PET/CT | Outcome on Discharge |
|---|---|---|---|---|---|---|
| 1. Male (60) | 60 | 247 | Negative | Negative | Negative | Tumor not found |
| 2. Male (19) | 19 | 1787 | Right acetabulum | Negative | Right acetabulum | Cured |
| 3. Male (43) | 43 | 1063 | Right femur | Right femur | Right femur | Cured |
| 4. Male (38)b | 38 | 5939 | Metastatic | Metastatic | Metastatic | Widespread disease |
| 5. Female (21)b | 21 | 860 | Negative | Negative | Right footc | Tumor not found |
| 6. Female (52)b | 52 | 1156 | Left femur | Negative | Left femur | Cured |
| 7. Female (44) | 44 | 218 | Negative | Negative | Negative | Tumor not found |
| 8. Male (47) | 47 | 1515 | Right inferior pelvis | Right inferior pelvis | Negative | Cured |
| 9. Female (27)b | 27 | 105 | Left maxilla | Left maxilla | Negative | Cured |
| 10. Female (17) | 17 | 353 | Negative | Negative | Negative | Tumor not found |
| 11. Female (48) | 48 | 286 | Right clavicle and pelvisc | Right clavicle and pelvisc | Right clavicle and pelvisc | Tumor not found |
Bold data represent discrepancy between 68Ga-DOTATATE PET/CT and OctreoScan SPECT/CT.
With the exception of subject 1 who underwent measurement of C-terminus FGF23 (normal range <180 RU/ml), all patients underwent measurement of intact FGF23 (normal range 8–78 pg/ml).
Subject had one or more surgical procedures to resect tumor before the study.
Ultimately considered to be false positive as determined by biopsy and/or venous sampling, and/or surgical resection.
Subjects were admitted to the National Institutes of Health (NIH) Clinical Center between February 2014 and February 2015 and gave informed consent to be enrolled in studies that were approved by the National Institute of Dental and Craniofacial Research and the National Cancer Institute Institutional review boards. Biochemical evaluation to confirm TIO included blood phosphate, FGF23, 25-hydroxy- and 1,25-dihydroxyvitamin D, PTH, and calculation of tubular reabsorption of phosphate, as previously described (1). Imaging studies included 68Ga-DOTATATE PET/CT (as described later), Octreoscan-SPECT/CT scan, and 18F FDG-PET/CT (in two cases, subjects 9 and 10, the 18F FDG-PET/CT study had been previously performed recently and was not repeated). Anatomical imaging with MRI and CT, and selective venous sampling were performed to confirm tumor localization before resection, as needed. Surgical resection was performed if the tumor was localized, with normalization of blood phosphate off of supplementation following tumor resection to prove cure (Table 1).
Imaging
DOTATATE is an amide of the acid DOTA (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid), which acts as a chelator for the radionuclide to (Tyr3)-octreotate, a derivative of octreotide. 68Ga-DOTATATE PET/CT was recently approved by the US Food and Drug Administration for clinical use (6); however, at the time of the study, it was classified as an investigational agent in the United States, therefore the study was performed under a research protocol approved by the National Cancer Institute Institutional Review Board and the NIH Radiation Safety Committee (NCT01967537) under an Investigational New Drug application from the US Food and Drug Administration. Through a peripheral vein, approximately 5 mCi of 68Ga-DOTATATE was administered. After approximately 60 minutes, the patient was positioned in the supine position in a PET/CT scanner (Siemens mCT), and images from the toes to top of the skull, including extremities, were obtained. A low-dose, noncontrast CT was used for attenuation correction and anatomic localization. Maximum standardized uptake values (SUVmax) were normalized by body weight. An Octreoscan-SPECT/CT with imaging at 4 and 24 hours after IV administration of approximately 6 mCi (222 MBq) of 111In-pentetreotide was performed within 6 weeks of 68Ga-DOTATATE PET/CT to allow for direct comparison. All subjects underwent whole-body 18F FDG-PET/CT either during the same admission (nine of 11) or during a previous evaluation at the NIH (two of 11). For anatomical confirmation additional imaging studies, included CT and MRI of suspicious areas were performed.
Images were reviewed by independent radiologists and nuclear medicine physicians in a blinded fashion. All imaging studies were analyzed and correlated with clinical information by a multidisciplinary team to determine the optimal treatment options based on the functional status of the tumors and/or the presence of metastatic disease.
Results
The cohort consisted of 11 subjects (six females, five males), with mean age of age of 38 (range, 19–60) (Table 1). Seven of 11 subjects had findings on 68Ga-DOTATATE suggestive of PMT. After anatomical confirmation by CT and MRI, four underwent surgical resection and were cured (patients 3, 6, 8, 9). After confirmation by anatomical imaging and selective venous sampling, the location of the tumor in subject 2 was judged to be such that that surgery would have been highly morbid. Therefore, he underwent catheter-guided embolization and cryoablation and was cured. Subject 4 had unresectable, widely metastatic disease and was enrolled in clinical trial with the fibroblast growth receptor-inhibitor NVP-BGJ398 (NCT02160041) with a promising response (7). Subject 11 had findings that were ultimately considered to be false positive after anatomical imaging and venous sampling failed to support the 68Ga-DOTATATE finding as that of a PMT.
Five of the 11 subjects had suggestive findings on Octreoscan-SPECT/CT. In all five cases, the lesion was also seen on 68Ga-DOTATATE imaging. One was subject 11, mentioned previously, in whom all three imaging modalities gave a false-positive reading. Six of 11 subjects had positive 18F FDG-PET/CT scans; five were also positive on 68Ga-DOTATATE. In total, two18F FDG- PET/CT scans were considered to be false positive. 68Ga-DOTATATE showed suspicious lesions that were not seen with Octreoscan-SPECT/CT in two cases, both of which eventually proved to be the culprit tumor, and both of which were also seen on 18F FDG-PET/CT scan. Comparison of images of all three modalities in subjects 2, 3, and 9 are shown in Figures 1, 2, and 3 respectively.
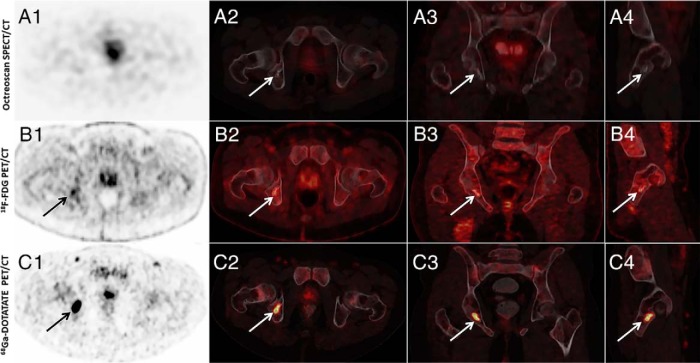
Figure 1.
Example of a subject in whom 111In-pentetreotide SPECT/CT (Octreoscan) was negative and 18F FDG-PET/CT and 68Ga-DOTATATE PET/CT (DOTATATE) were positive (subject 2). (A1) Axial SPECT, (A2) axial SPECT/CT, (A3) coronal SPECT/CT, and (A4) sagittal SPECT/CT images of whole-body 111In Pentreotide-Octreotide SPECT/CT scan that did not show abnormal uptake by the right acetabulum region. (B1) Axial PET, (B2) axial PET/CT, (B3) coronal PET/CT, (B4) sagittal PET/CT images of whole-body 18F-FDG PET/CT scan showing increased activity (SUVmax, 4.05) in the right posterior inferior acetabulum associated with bony changes. (C1) axial PET, (C2) axial PET/CT, (C3) coronal PET/CT, and (C4) sagittal PET/CT images of whole-body 68Ga-DOTATATE PET/CT scan showing increased uptake (SUVmax, 23.8) by the right acetabulum.
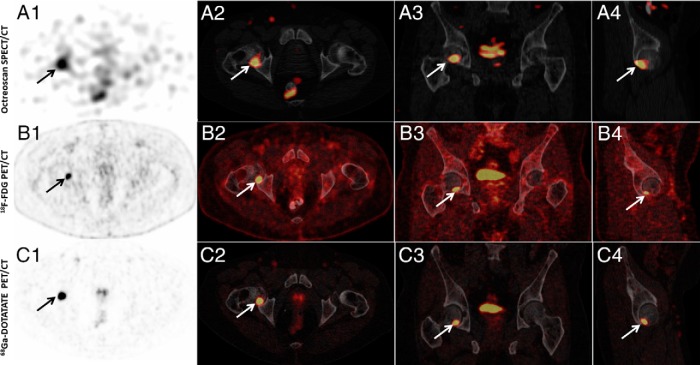
Figure 2.
Example of a study in which all three imaging studies, 111In-pentetreotide SPECT/CT (Octreoscan), 18F FDG-PET/CT, and 68Ga-DOTATATE PET/CT (DOTATATE) were positive (subject 3). (A1) Axial SPECT, (A2) axial SPECT/CT, (A3) coronal SPECT/CT, and (A4) sagittal SPECT/CT images of whole-body 111In Pentreotide-Octreotide SPECT/CT scan that showed focus of abnormally increased uptake by the posterior inferior right femoral head. (B1) Axial PET, (B2) axial PET/CT, (B3) coronal PET/CT, and (B4) sagittal PET/CT images of whole-body 18F FDG-PET/CT scan showing increased activity (SUVmax, 13.8) in the right femoral head. (C1) Axial PET, (C2) axial PET/CT, (C3) coronal PET/CT, and (C4) sagittal PET/CT images of whole-body 68Ga-DOTATATE PET/CT scan showing increased uptake (SUVmax, 54.1) by the right femoral head.
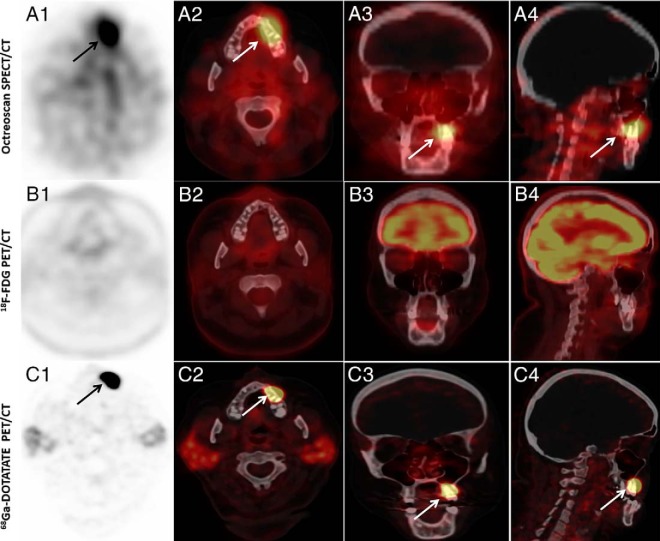
Figure 3.
An example of a subject in whom 111In-pentetreotide SPECT/CT (Octreoscan) and 68Ga-DOTATATE PET/CT (DOTATATE) were positive, but was negative (subject 9). (A1) Axial SPECT, (A2) axial SPECT/CT, (A3) coronal SPECT/CT, and (A4) sagittal SPECT/CT images of whole-body 111In Pentreotide-Octreotide SPECT/CT scan showing focus of abnormally increased uptake by left maxilla. (B1) Axial PET, (B2) axial PET/CT, (B3) coronal PET/CT, and (B4) sagittal PET/CT images of whole-body 18F FDG-PET/CT scan that did not show abnormally increased activity in the left maxilla region. (C1) Axial PET, (C2) axial PET/CT, (C3) coronal PET/CT, and (C4) sagittal PET/CT images of whole-body 68Ga-DOTATATE PET/CT scan showing intensely increased uptake (SUVmax, 86) by the left maxilla.
Discussion
The culprit tumor was successfully localized and treated in 54.5% (6/11) of subjects, including one patient with metastatic disease; and not localized in 45.5% (5/11). 68Ga-DOTATATE imaging correlated with Octreoscan-SPECT/CT in 9/11 cases, and detected lesions in 2/11 cases not seen on Octreoscan-SPECT/CT but seen on 18F FDG-PET/CT, both of which were confirmed to represent the culprit tumor (Table 1). Interestingly, four of five subjects with localized tumors had intact FGF23 levels higher than 1000 pg/ml (subjects 2, 3, 6, 8), suggesting that markedly elevated levels of FGF23 may be associated with larger and/or tumors that have stronger somatostatin receptor (SSTR) uptake on imaging. Findings on 68Ga-DOTATATE imaging were accurate in localizing tumor in six of seven cases, and proved to be false positive in one; Octreoscan-SPECT/CT was accurate in four of the five positive imaging studies and false positive in one of five; 18F FDG-PET/CT was accurate in four of six positive studies and false negative in two of six. Based on these findings, the sensitivity and specificity of 68Ga-DOTATATE was 54.5% and 85.7%; Octreoscan-SPECT/CT 36.3% and 80%; and 18F FDG-PET/CT 36.3% and 86%, respectively.
PMTs are reported to express a variety of somatostatin receptors (SSTR1, 2A, 2B, 3,4,5). 68Ga-DOTATATE, like octreotide, is an antagonist of the SSTR, which upon receptor binding is internalized resulting in accumulation of radioactivity in tumor cells (8). However, 68Ga-DOTATATE is a positron emitter, a type of beta decay, which affords greater resolution than the gamma ray emitting 111Indium isotope. In vitro, somatostatin analogues have been shown to have variable affinity for different receptors: SSTR2 > SSTR5 for 68Ga-DOTATATE, SSTR5 > SSTR2 for DOTATOC, and SSTR2,3,5 for DOTANOC (9, 10). Octreoscan-SPECT/CT imaging on the other hand has high affinity for SSTR 2,5 (11). Breer et al reported immunohistochemical staining on 15 PMTs from 14 patients with TIO, and found diffuse and strongly positive SSTR2A staining in all tumors (3). Thus, the proposed superiority of 68Ga-DOTATATE imaging over Octreoscan-SPECT/CT imaging may be based on the higher affinity of 68Ga-DOTA-conjugated peptides for SSTR2 receptors as compared to Octreoscan-SPECT/CT (5). In addition to the higher affinity of 68Ga-DOTATATE for SSTR2, Octreoscan-SPECT/CT is limited by relatively poor spatial resolution compared to PET, thus accounting for why SSTR-imaging using 68Ga-DOTATATE enables better visualization of organs with higher octreotide-physiologic uptake (eg, liver). Additionally, 68Ga-DOTATATE imaging provides advantages in shorter acquisition time and lower radiation exposure because of its short half-life of 68 minutes, compared with 2.8 days for 111In, the isotope used in Octreoscan-SPECT/CT imaging (5). The presumed greater sensitivity of 68Ga-DOTATATE as compared to Octreoscan is accounted for both the radiotracer (68Ga), which is a positron emitter that enables STTR imaging with PET and the use of higher affinity to somatostatin receptors compounds like DOTATATE.
In the largest retrospective study published to date evaluating 68Ga-DOTATATE imaging in TIO, Zhang and colleagues found that 68Ga-DOTATATE imaging was positive in 32 of 32 subjects who had TIO confirmed pathologically; however, patients with negative 68Ga-DOTATATE imaging were excluded from the analysis as not having TIO, whereas the cause of the hypophosphatemia in at least half of the patients was unclear (12). No comparison to other imaging modalities was performed.
Clifton-Bligh et al were the first to study the localization of TIO tumors using 68Ga-DOTATATE imaging and successfully localized TIO tumors in six of six cases (10). However, Octreoscan-SPECT/CT was performed in only one case, along with other imaging modalities such as 18F FDG-PET (performed in two of six patients) or Tc-Sestamibi scan (performed in one of six) (only 68Ga-DOTATATE imaging localized the tumors) (10). Although the study found 100% sensitivity and specificity for 68Ga-DOTATATE, the study was retrospective and included only a subset of patients with presumed TIO. It is unclear if the other subjects with suspected TIO and in whom the tumor was not localized had 68Ga-DOTATATE imaging. In a study similar to ours, Breer et al compared the efficacy of 68Ga-DOTATATE to Octreoscan-SPECT/CT for tumor localization in five patients with TIO (three females, two males) (3). Whereas only one of five cases showed focal uptake by Octreoscan-SPECT/CT, all five cases showed focal activity on 68Ga-DOTATATE imaging. All subjects were cured after surgical resection. However, the low sensitivity of Octreoscan-SPECT/CT in this study is likely due to the fact that SPECT was not performed from head to feet as in our study, but confined to suspicious regions identified on planar images. Two studies from India, one with six subjects (Agrawal et al) and one with nine subjects (Jahdhav et al) used 68Ga-DOTATATE and 18F FDG-PET; in both studies, the sensitivity and specificity of 18F FDG-PET was 50%, sensitivity of 68Ga-DOTATATE was 83.3–100%, and specificity of 68Ga-DOTATATE was 100% (2, 4). Jadhav et al also used 99Tc-HYNIC-Octreoscan, which had 100% sensitivity and specificity in all six patients undergoing this imaging (4). A recent case series also described the successful use of 68Ga-DOTA peptide PET/CT imaging in localizing PMT tumors in all three of three patients presenting with TIO (13). 68Ga-DOTANOC has been evaluated in one study of 10 TIO patients showing a sensitivity and specificity of 90% (9). Several case reports also report successful localization using this radiopharmaceutical (14–16). 68Ga-DOTATOC has also been described in the successful localization of PMT in an adolescent boy (17). Other imaging modalities studied include 99Tc-HYNIC-Octreoscan, with a sensitivity and specificity of 86.3% and 99.1%, respectively (18), and 99Tc-octretide in which 49% had positive imaging and 41% were found to have TIO (19).
In this cohort, we found that 68Ga-DOTATATE as a single study provided the highest sensitivity/specificity. However it was not superior to our previously reported sensitivity and specificity of combined Octreoscan-SPECT/CT and 18F FDG-PET in localization of PMTs (1). Although our study showed high specificity for all three imaging modalities used, it showed low sensitivity for all three. In the case of 68Ga-DOTATATE, the sensitivity of 54.5% for 68Ga-DOTATATE is the lowest reported among other published studies, which have reported sensitivities of 83–100% (2–4, 10, 12). This difference is probably best explained by the fact this cohort consisted of subjects who had previously failed extensive, sometimes multiple, localization attempts before being seen at our center, and in some cases had failed localization at our center before the availability of 68Ga-DOTATATE imaging. Therefore, this likely represents a group of tumors that are extremely small and difficult to localize. Although comprehensive testing was performed to rule out other acquired forms of hypophosphatemia, such as Fanconi syndrome, heavy metal exposure, etc., other as-yet unknown causes of this disorder may exist. An important feature of this study is the fact that it was prospective. Previously published studies were retrospective and, as such, may have excluded subjects with negative imaging and/or reclassified them as having alternative causes of hypophosphatemia. This would significantly raise the sensitivity found in such studies. In this prospective study, we consider negative images as failed localization attempts, thus significantly reducing the sensitivities of the reported imaging modalities.
An important feature of this study and previous work in this area from our institution is the manner in which the Octreoscan-SPECT/CT is performed. SPECT imaging is typically performed from head to toe, an approach that can require 5–6 hours of scanning time over 2 days; an approach that is undertaken at few other centers, but necessary to maximize the sensitivity of Octreoscan-SPECT/CT in localizing PMTs. Because 68Ga-DOTATATE was only recently approved by the US Food and Drug Administration for clinical use in the detection of rare neuroendocrine tumors (6), and a cost structure has not been established; therefore, a cost-benefit analysis at this time is not possible. Finally, an important limitation in this study is the small number of subjects. Potential studies characterizing SSTR pattern and its correlation to imaging response may prove useful. Future studies with similar comparison imaging modalities will be needed to definitively define the ideal single imaging study for identifying PMTs.
Conclusion
In this first prospective study of SSTR imaging using 68Ga-DOTATATE in TIO, 68Ga-DOTATATE PET/CT had greater sensitivity and specificity than either Octreoscan-SPECT/CT or 18F FDG-PET/CT but was not more sensitive than Octreoscan-SPECT/CT and 18F FDG-PET/CT combined. Future prospective studies, including a larger sample size, and possibly the use of radiolabeled somatostatin analogues with different SSTR affinities, such as DOTATNOC and DOTATOC, may show added benefits over functional imaging modalities such as combined Octreoscan-SPECT/CT and 18F FDG-PET/CT.
Acknowledgments
This work was supported by the Division of Intramural Research, the National Cancer Institute and National Institute of Dental and Craniofacial Research, National Institutes of Health, Bethesda, MD.
Disclosure Summary: None of the authors have nothing to disclose.
Footnotes
- DOTA
- 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid
- 18F FDG-PET/CT
- fluorodeoxyglucose-positron emission tomography-computed tomography
- FGF23
- fibroblast-growth-factor 23
- 68Ga
- 68Gallium
- NIH
- National Institutes of Health
- Octreoscan-SPECT/CT
- 111In-pentetreotide single-photon emission/computed tomography
- PMT
- phosphaturic mesenchymal tumors
- SSTR
- somatostatin receptors
- SUV
- standardized uptake value
- TIO
- tumor-induced osteomalacia.
References
- 1. Chong WH, Andreopoulou P, Chen CC, et al. Tumor localization and biochemical response to cure in tumor-induced osteomalacia. J Bone Miner Res. 2013;28:1386–1398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Agrawal K, Bhadada S, Mittal BR, et al. Comparison of 18F-FDG and 68Ga DOTATATE PET/CT in localization of tumor causing oncogenic osteomalacia. Clin Nucl Medicine. 2015;40:e6–e10. [DOI] [PubMed] [Google Scholar]
- 3. Breer S, Brunkhorst T, Beil FT, et al. 68Ga DOTA-TATE PET/CT allows tumor localization in patients with tumor-induced osteomalacia but negative 111In-octreotide SPECT/CT. Bone. 2014;64:222–227. [DOI] [PubMed] [Google Scholar]
- 4. Jadhav S, Kasaliwal R, Lele V, et al. Functional imaging in primary tumour-induced osteomalacia: relative performance of FDG PET/CT vs somatostatin receptor-based functional scans: a series of nine patients. Clin Endocrinol. 2014;81:31–37. [DOI] [PubMed] [Google Scholar]
- 5. Hofman MS, Lau WF, Hicks RJ. Somatostatin receptor imaging with 68Ga DOTATATE PET/CT: clinical utility, normal patterns, pearls, and pitfalls in interpretation. Radiographics. 2015;35:500–516. [DOI] [PubMed] [Google Scholar]
- 6. US Food and Drug Administration. FDA approves new diagnostic imaging agent to detect rare neuroendocrine tumors. 2016; http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm504524.htm Accessed June 10, 2016.
- 7. Collins MT, Bergwitz C, Aitcheson G, et al. Striking response of tumor-induced osteomalacia to the FGFR inhibitor NVP-BGJ398. Paper presented at: American Society of Bone and Mineral Research Annual Meeting 2015; Seattle, WA. [Google Scholar]
- 8. Dalm SU, Nonnekens J, Doeswijk GN, et al. Comparison of the therapeutic response to treatment with a 177Lu-labeled somatostatin receptor agonist and antagonist in preclinical models. J Nucl Med. 2016;57:260–265. [DOI] [PubMed] [Google Scholar]
- 9. Bhavani N, Reena Asirvatham A, et al. Utility of gallium-68 DOTANOC PET/CT in the localization of tumour-induced osteomalacia. Clin Endocrinol. 2016;84:134–140. [DOI] [PubMed] [Google Scholar]
- 10. Clifton-Bligh RJ, Hofman MS, Duncan E, et al. Improving diagnosis of tumor-induced osteomalacia with gallium-68 DOTATATE PET/CT. J Clin Endocrinol Metab. 2013;98:687–694. [DOI] [PubMed] [Google Scholar]
- 11. Intenzo CM, Jabbour S, Lin HC, et al. Scintigraphic imaging of body neuroendocrine tumors. Radiographics. 2007;27:1355–1369. [DOI] [PubMed] [Google Scholar]
- 12. Zhang J, Zhu Z, Zhong D, et al. 68Ga DOTATATE PET/CT is an accurate imaging modality in the detection of culprit tumors causing osteomalacia. Clin Nucl Med. 2015;40:642–646. [DOI] [PubMed] [Google Scholar]
- 13. Ho CL. Ga68-DOTA peptide PET/CT to detect occult mesenchymal tumor-inducing osteomalacia: a case series of three patients. Nucl Med Mol Imaging. 2015;49:231–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hesse E, Moessinger E, Rosenthal H, et al. Oncogenic osteomalacia: exact tumor localization by co-registration of positron emission and computed tomography. J Bone Miner Res. 2007;22:158–162. [DOI] [PubMed] [Google Scholar]
- 15. Naswa N, Sharma P, Kumar R, Malhotra A, Bal C. Successful localization of residual culprit tumor in a case of tumor-induced osteomalacia using 68Ga-DOTANOC PET/CT. Clin Nucl Med. 2013;38:639–640. [DOI] [PubMed] [Google Scholar]
- 16. von Falck C, Rodt T, Rosenthal H, et al. (68)Ga-DOTANOC PET/CT for the detection of a mesenchymal tumor causing oncogenic osteomalacia. Eur J Nucl Med Mol Imaging. 2008;35:1034. [DOI] [PubMed] [Google Scholar]
- 17. Haeusler G, Freilinger M, Dominkus M, et al. Tumor-induced hypophosphatemic rickets in an adolescent boy–clinical presentation, diagnosis, and histological findings in growth plate and muscle tissue. J Clin Endocrinol Metab. 2010;95:4511–4517. [DOI] [PubMed] [Google Scholar]
- 18. Jing H, Li F, Zhuang H, et al. Effective detection of the tumors causing osteomalacia using [Tc-99m]-HYNIC-octreotide (99mTc-HYNIC-TOC) whole body scan. Eur J Radiol. 2013;82:2028–2034. [DOI] [PubMed] [Google Scholar]
- 19. Jiang Y, Xia WB, Xing XP, et al. Tumor-induced osteomalacia: an important cause of adult-onset hypophosphatemic osteomalacia in China: Report of 39 cases and review of the literature. J Bone Mineral Res. 2012;27:1967–1975. [DOI] [PubMed] [Google Scholar]