Abstract
Background
Ginseng extracts are known to have angiogenic effects. However, to date, only limited information is available on the molecular mechanism underlying the angiogenic effects and the main components of ginseng that exert these effects. Human umbilical-vein endothelial cells (HUVECs) are used as an in vitro model for screening therapeutic agents that promote angiogenesis and wound healing. We recently isolated gintonin, a novel ginseng-derived lysophosphatidic acid (LPA) receptor ligand, from ginseng. LPA plays a key role in angiogenesis and wound healing.
Methods
In the present study, we investigated the in vitro effects of gintonin on proliferation, migration, and tube formation of HUVECs, which express endogenous LPA1/3 receptors.
Results
Gintonin stimulated proliferation and migration of HUVECs. The LPA1/3 receptor antagonist, Ki16425, short interfering RNA against LPA1 or LPA3 receptor, and the Rho kinase inhibitor, Y-27632, significantly decreased the gintonin-induced proliferation, migration, and tube formation of HUVECs, which indicates the involvement of LPA receptors and Rho kinase activation. Further, gintonin increased the release of vascular endothelial growth factors from HUVECs. The cyclooxygenase-2 inhibitor NS-398, nuclear factor kappa B inhibitor BAY11-7085, and c-Jun N-terminal kinase inhibitor SP600125 blocked the gintonin-induced migration, which shows the involvement of cyclooxygenase-2, nuclear factor kappa B, and c-Jun N-terminal kinase signaling.
Conclusion
The gintonin-mediated proliferation, migration, and vascular-endothelial-growth-factor release in HUVECs via LPA-receptor activation may be one of in vitro mechanisms underlying ginseng-induced angiogenic and wound-healing effects.
Keywords: gintonin, human umbilical-vein endothelial cells, LPA receptors, Panax ginseng
1. Introduction
Wound healing is a physiological process that involves cell proliferation and migration to restore normal state after injury of the skin, blood vessels, and other tissues. This process includes hemostasis, inflammation, angiogenesis, collagen deposition, epithelialization, and remodeling [1]. Angiogenesis is a critical component in wound healing and involves a series of steps, including proliferation and migration of endothelial cells [1], [2]. Endothelial dysfunction causes impairment in wound healing and angiogenesis. The therapeutic effects of angiogenesis have been investigated in repairing and minimizing tissue damage due to cardiovascular diseases, such as coronary heart diseases and peripheral arterial diseases, and wound-healing disorders [3], [4], [5], [6].
Ginseng, the root of Panax ginseng, is a traditional herbal medicine used as a tonic for invigorating the body or for alleviating a variety of diseases, including cardiovascular diseases, rheumatoid arthritis, diabetes mellitus, and cancer [7], [8], [9], [10]. Ginseng contains several bioactive components, such as ginsenosides, acidic polysaccharides, and other unidentified components. Previous studies have shown that the ginseng extract and the ginseng saponin fraction, which contain a mixture of ginsenosides and other unidentified ingredients, stimulate angiogenesis [9], [11], [12]. Each of the ginsenosides has different effects on angiogenesis or wound healing. While ginsenoside Rg1 stimulates angiogenesis [12], [13], ginsenosides Rg3 and compound K inhibit in vitro angiogenesis [14], [15]. Although ginsenoside Rb1 and Rd seem to stimulate wound healing of the skin [16], [17], the angiogenic effects of those ginsenosides are controversial depending on experimental models [16], [18]. In addition, ginseng extracts and the total saponin fraction of ginseng include additional unidentified ingredients. Therefore, the active component responsible for the effects of ginseng remains to be clarified.
Recently, we isolated gintonin, a lysophosphatidic-acid (LPA) receptor ligand, from ginseng [19], [20]. Gintonin consists of a complex of ginseng proteins and LPAs, and potently activates LPA receptors in animal cells. LPA-receptor activation plays a role in diverse cellular effects, including proliferation and migration of cells, vascular development, and neurite retraction [21]. LPA-receptor-mediated cellular effects are further coupled to biological activities, such as brain development, angiogenesis, embryo implantation, spermatogenesis, and wound healing [21].
Human umbilical-vein endothelial cells (HUVECs) express endogenous LPA-receptor subtypes, LPA1 and LPA3 [22]. LPA induces proliferation and migration of HUVECs, and silencing of LPA1/3 by short interfering RNA (siRNA) markedly suppresses the LPA-induced proliferation and migration of HUVECs, which are essential steps for angiogenesis [23]. In addition, LPA stimulates the release of vascular endothelial growth factor (VEGF), which in turn facilitates the angiogenic processes [24]. In addition, cyclooxygenase-2 (COX-2) and nuclear factor kappa B (NF-κB) are involved in the increased release of VEGF by various stimulators [25], [26]. On the basis of the findings reported previously, we assumed that activation of the LPA receptors by gintonin can be a molecular basis of ginseng-extract-induced angiogenesis.
In the present study, we examined the in vitro angiogenic effects of gintonin in HUVECs. Gintonin stimulated the proliferation, migration, and tube formation of HUVECs. In particular, we discussed the molecular mechanisms underlying gintonin-induced wound-healing effect with the evidence of LPA-receptor activation, VEGF release, and activation of COX-2 and NF-κB by gintonin in HUVECs. Our results show that gintonin can induce in vitro angiogenesis and wound healing through the activation of LPA receptors and VEGF signaling pathways.
2. Materials and methods
2.1. Materials
Crude gintonin was isolated from P. ginseng as described previously [19]. Gintonin is a glycolipoprotein containing ginseng protein complexed with LPA [20]. Ginsenosides were purchased from the LKT Laboratories Inc. (St. Paul, MN, USA). VEGF, basic fibroblast growth factor, and Quantikine human VEGF immunoassay kit were purchased from R&D Systems (Minneapolis, MN, USA). M199 medium and 0.1% gelatin solution were purchased from WelGENE (Daegu-si, Korea). Matrigel (growth factor reduced) and collagen type 1 were purchased from BD Biosciences (Bedford, MA, USA). All other reagents used were purchased from Sigma-Aldrich (St. Louis, MO, USA).
2.2. Cell culture
HUVECs were isolated from human umbilical cord veins by collagenase treatment as described previously [27], and cultured in M199 medium supplemented with 20% (volume/volume) fetal bovine serum (FBS), 5 units/mL heparin, 3 ng/mL basic fibroblast growth factor, 100 units/mL penicillin, and 100 μg/mL streptomycin. The cultures were maintained at 37°C in humidified conditions under 5% CO2. The cells at passages 2–7 were used in all the experiments.
2.3. Cell proliferation
Proliferation of HUVECs was determined using a sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt (XTT)-based assay, which measures the cell viability based on the activity of mitochondrial enzyme [28]. Cells were seeded at 3 × 103 cells per well into 96-well plates coated with 0.1% gelatin solution. After 24 h, the cells were washed with M199 medium and incubated for 6 h with M199 containing 1% FBS. The cells were washed with fresh M199 (1% FBS) again, and incubated with gintonin, ginsenosides, or VEGF at the specific concentrations. Inhibitors were added 30 min before the incubation period. After the indicated incubation time, cell proliferation was assessed using the XTT assay as described previously [28]. The culture medium of cells in each well of the 96-well plate was replaced with 200 μL of serum-free medium without phenol red. Then, 50 μL of XTT reaction solution (containing 1 mg/mL XTT and 0.0306 mg/mL phenazine methosulfate) was added to each well. After incubation for 2 h, the absorbance was measured at 450 nm, which correlates with the cell viability.
2.4. Migration assay
The chemotactic motility of HUVECs was measured using the modified Boyden chamber (Neuro Probe, Gaithersburg, MD, USA) as described previously [20]. Briefly, a polycarbonate membrane with an 8-μm pore size (Neuro Probe) was coated with 0.1 mg/mL of collagen type I from the rat tail (BD Biosciences, San Jose, CA, USA). Gintonin, VEGF, or ginsenosides in M199 (0.1% bovine serum albumin) were added to the lower chambers. The Boyden chamber was assembled by laying the membrane and the top chamber on the lower chambers. Cells (5 × 104 cells/well) were loaded to the top chambers and incubated for 70–80 min at 37°C. In some experiments, the cells were placed in the lower chambers with or without the inhibitors. The Boyden chamber was assembled and placed upside down, and incubated for 60 min. Then, the chamber was returned to the upright position, and gintonin or VEGF in M199 (0.1% bovine serum albumin) was added to the upper chambers, followed by incubation for an additional 120 min. The cells on the membrane were fixed and stained with Diff-Quik (Sysmex, Kobe, Japan). The migrated cells in the four fields each well (16 fields each group) were counted under a microscope (light microscopy) at a magnification of 200×. The images were photographed using a dark-field microscope (Eclipse 80i; Nikon, Tokyo, Japan).
2.5. Scratch wound-healing assay
We performed an in vitro wound-healing assay as described previously [22]. Briefly, the HUVECs were seeded at a density of 2 × 105 cells/well in 24-well plates precoated with gelatin. The cells were incubated in serum-free media for 6 h and wounded in a line across the well using a 200-μL pipette tip, followed by washing twice with a serum-free medium. The cells were incubated with different concentrations of gintonin in M199 containing 1% FBS for 20 h. The image of the wounded area was captured, and recovery of the area was analyzed using an inverted fluorescence microscope (Axiovert 200; Carl Zeiss AG, Oberkochen, Germany) at a magnification of 100×. The cell-free area in three photos each group was quantified by the software AxioVision (Zeiss; Carl Zeiss AG, Oberkochen, Germany). The cell-free area at 20 h was determined as a percentage of the initial wounded area.
2.6. Tube-formation assay
Tube formation was examined as reported previously with some modification [9]. The HUVECs were serum starved in M199 containing 1% FBS for 6 h, and seeded at a density of 2 × 105 cells/well in 24-well plates precoated with growth factor-reduced Matrigel (250 μL/well). The cells were incubated with gintonin or VEGF in M199 containing 1% FBS at 37°C. In some experiments, the inhibitors were added 30 min before the incubation period. After 4 h, tube formation was examined using an inverted fluorescent microscope at a magnification of 100×. Tube formation was quantified by counting the number of branches of the tubes in fields at 4 h.
2.7. siRNA
To suppress the expression of LPA receptors in HUVECs, we transfected the cells with siRNAs of LPA1 and LPA3. In addition, scrambled siRNA was used as a control (Santa Cruz Biotechnology, Santa Cruz, CA, USA). After the medium was replaced with serum-free M199, siRNA was added to a final concentration of 50nM siRNA using Lipofectamine 2000 (Life Technologies, Grand Island, NY, USA), according to the manufacturer's instructions.
2.8. VEGF enzyme-linked immunosorbent assay
VEGF was assayed as reported previously with some modification [29]. Briefly, the HUVECs were starved in M199 containing 1% FBS for 6 h, and incubated with M199 containing 1% FBS in the presence or absence of gintonin at the indicated concentrations for 24 h. The supernatant was collected and concentrated 50-fold at 4°C using Amicon centrifugal filter devices (EMD Millipore, Billerica, MA, USA). The concentrated medium was assayed for VEGF content using an enzyme-linked-immunosorbent-assay kit (R&D Systems) according to the manufacturer's instructions.
2.9. Statistical analysis
Data are expressed as means ± standard deviation. Statistical comparisons between controls and treated experimental groups were performed using the Student t test. Statistical evaluation was performed using GraphPad prism, version 5.0 (GraphPad Software, San Diego, CA, USA). We considered p < 0.05 to be statistically significant.
3. Results
3.1. Effects of gintonin on in vitro proliferation and migration of HUVECs
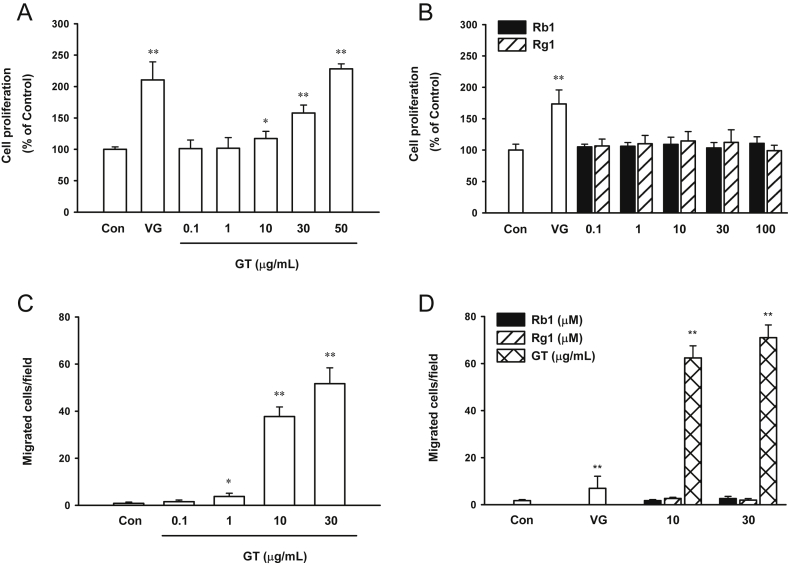
Cells treated with gintonin (10–50 μg/mL) showed a dose-dependent increase in viability by XTT assay; the viability of the treated cells was twice that of the untreated control cells (Fig. 1A). The half-maximal effective concentration (EC50) value for cell proliferation was 32.20 ± 2.76 μg/mL. Ginsenosides Rb1 and Rg1 had no effects on cell proliferation (Fig. 1B). VEGF was used as a positive control (Fig. 1A, B). The proliferation of cells treated with 10 ng/mL VEGF was approximately 1.7–2-fold of that of the control cells.
Fig. 1.
Effect of ginsenosides and gintonin on proliferation and migration of human umbilical-vein endothelial cells. (A, B) Cells were incubated either with vascular endothelial growth factor (positive control, 20 ng/mL), ginsenosides Rb1 and Rg1, or gintonin at the indicated concentrations in M199 containing 1% fetal bovine serum for 48 h. Cell proliferation was examined using a sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt-based cell-viability assay as described in the Materials and Methods section. (C, D) Effect of vascular endothelial growth factor (as a positive control, 20 ng/mL), ginsenosides Rb1 and Rg1, or gintonin on the chemotactic motility of human umbilical-vein endothelial cells was assessed using the modified Boyden chamber as described in the Materials and Methods section. Results of the sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt assay show that, compared to the untreated control cells, (A) the cells treated with gintonin showed increased proliferation, while (B) those treated with Rb1 and Rg1 did not stimulate proliferation. (C) Gintonin stimulate cell migration in a dose-dependent manner, while (D) ginsenosides Rb1 and Rg1 did not stimulate cell migration at 10μM and 30μM. The data represent the means ± SD of the results of four to six independent experiments. *p < 0.05. **p < 0.001 compared with control. Con, control; GT, gintonin; VG, vascular endothelial growth factor.
Further, we examined the effect of ginsenosides Rb1 and Rg1 and gintonin on the chemotactic motility using the modified Boyden chamber. Gintonin stimulated the migration of HUVECs in a concentration-dependent manner. Treatment with 10 μg/mL and 30 μg/mL gintonin stimulated the migration of HUVECs by up to 40-fold compared to that of the control cells. The EC50 value for cell migration was 6.11 ± 0.43 μg/mL (Fig. 1C). This value is much lower than that of cell proliferation by gintonin, which shows that the migration of HUVECs is much more sensitive to gintonin than proliferation. VEGF (20 ng/mL) stimulated the migration of HUVECs by four-fold compared to that of the control cells (Fig. 1D). However, ginsenosides (Rb1 and Rg1) at concentrations of 10μM and 30μM had no effect on the migration of HUVECs. Thus, gintonin, but not ginsenosides, stimulated the proliferation and migration of HUVECs.
3.2. Effects of gintonin on in vitro wound healing and tube formation of HUVECs
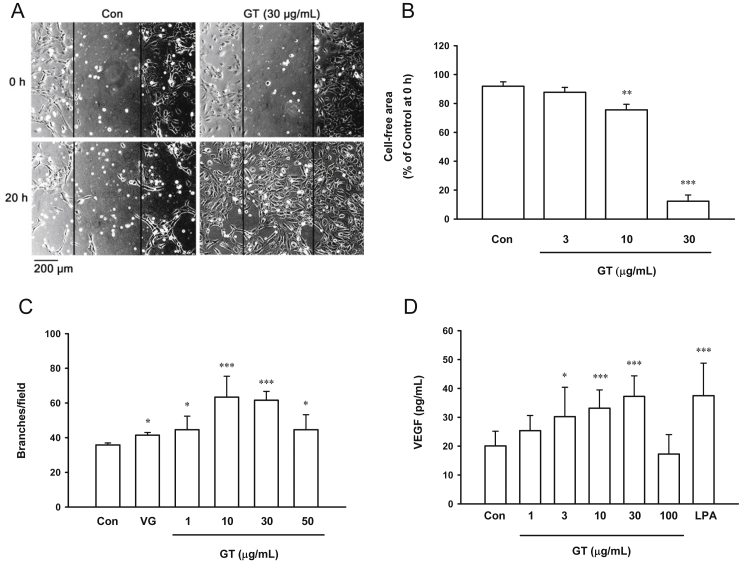
We examined the effect of gintonin on wound healing using HUVEC monolayers. The remaining cell-free area at 20 h was calculated as a percentage of the initial wounded area. In the control cells, the cell-free wound healed very slowly, showing 91.9 ± 1.8% of cell-free area at 20 h (Fig. 2A, B). Compared to the untreated control cells, the cells treated with 10 μg/mL and 30 μg/mL of gintonin significantly decreased the cell-free area (75.6 ± 2.2% and 12.3 ± 2.5%, respectively; Fig. 2B). These results showed that gintonin stimulated wound closure in HUVECs.
Fig. 2.
Effect of gintonin on scratch wound healing, tube formation, and release of vascular endothelial growth factor in human umbilical-vein endothelial cells. (A, B) Cells were scratched and incubated with either vehicle or gintonin at the indicated concentrations in M199 containing 1% fetal bovine serum for 20 h. Relative cell-free area of each field was calculated compared to that at 0 h. Gintonin stimulated wound healing in a dose-dependent manner at 10 μg/mL and 30 μg/mL. (C) Cells were incubated with gintonin at the indicated concentrations in M199 medium containing 1% fetal bovine serum for 4 h on the plates coated with Matrigel, and tube formation was examined as described in the Materials and Methods section. Vascular endothelial growth factor (10 ng/mL) was also used as the positive control. (D) For vascular-endothelial-growth-factor assay, cells were incubated with gintonin at the indicated concentrations for 24 h. The conditioned medium was collected and concentrated 50-fold, and the vascular-endothelial-growth-factor level was determined using enzyme-linked immunosorbent assay as described in the Materials and Methods section. The data represent the means ± SD of the results of three to nine independent experiments. *p < 0.05. **p < 0.01. ***p < 0.001 compared with untreated control. Con, control; GT, gintonin; LPA, lysophosphatidic acid; VEGF and VG, vascular endothelial growth factor.
An in vitro tube-formation assay of HUVECs was examined to investigate the effect of compounds on the remodeling of endothelial cells. Gintonin stimulated tube formation at 1–50 μg/mL (Fig. 2C). The maximum effects of gintonin were observed at 10 μg/mL and 30 μg/mL. In addition, a positive control, VEGF (10 ng/mL), stimulated tube formation in HUVECs. VEGF acts on vascular endothelial cells as a chemotactic and mitogenic factor. To determine whether gintonin stimulates the release of VEGF, we performed VEGF assay in the conditioned media of HUVECs incubated with gintonin at different concentrations or LPA. Treatment with 30 μg/mL gintonin stimulated the release of VEGF by HUVECs by 1.85 ± 0.12-fold compared to that by the control cells (Fig. 2D). Similarly, 10μM LPA increased the production of VEGF by 1.87 ± 0.19-fold compared to that by the control cells.
3.3. Gintonin-mediated migration and tube formation of HUVECs are partially mediated by the activation of LPA receptor
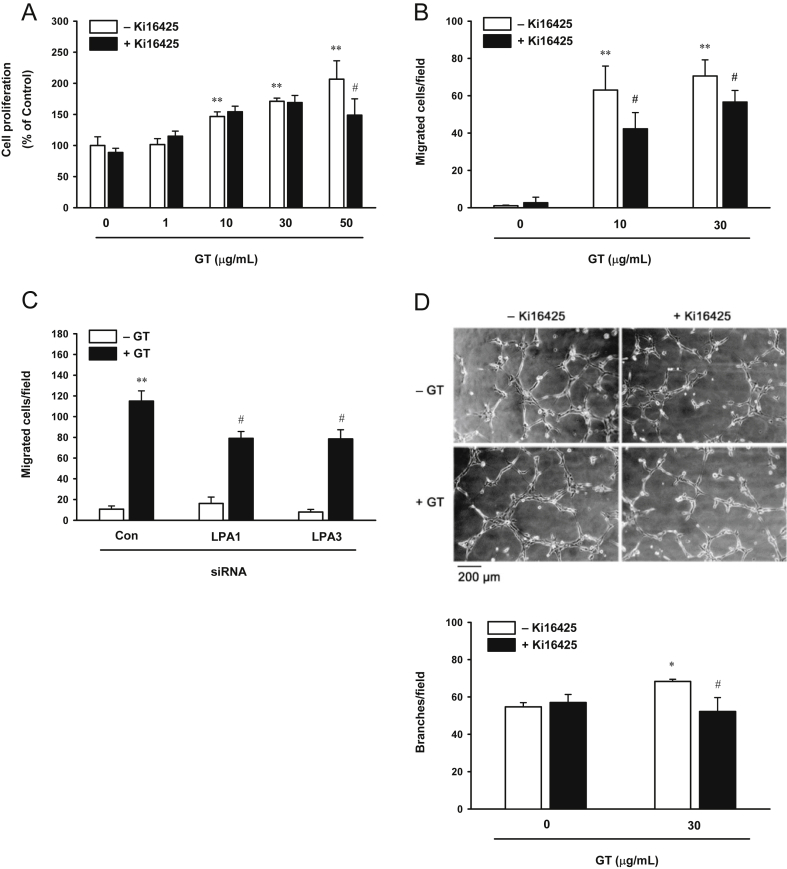
To study whether the LPA receptor is involved in gintonin-induced cell proliferation, migration, and tube formation, we used the LPA-receptor inhibitor, Ki16425. Incubation of HUVECs with Ki16425 slightly decreased the cell proliferation induced by 50 μg/mL of gintonin (Fig. 3A). However, Ki16425 partially attenuated the gintonin-induced migration of HUVECs by 33% and 17% at 10 μg/mL and 30 μg/mL of gintonin, respectively (Fig. 3B). In addition, to further clarify the role of the LPA receptor on gintonin-induced HUVEC migration, LPA1 and 3 in HUVECs were silenced with siRNA. In the LPA-receptor silenced HUVECs, the gintonin-induced migration decreased by ∼30% compared to that in HUVECs transfected with control siRNA (Fig. 3C). Moreover, pretreatment of HUVECs with Ki16425 attenuated the gintonin-induced tube formation by approximately 24% (Fig. 3D). These results showed that gintonin-induced migration and tube formation of HUVECs are at least partially mediated by the activation of LPA receptors.
Fig. 3.
Lysophosphatidic acid 1/3 receptor inhibition attenuates gintonin-induced (A) proliferation, (B and C) migration, and (D) tube formation. (A) Cells were incubated in the presence or absence of lysophosphatidic acid 1/3 receptor inhibitor, Ki16425 (10μM), for 30 min, and then incubated with gintonin at different concentrations for another 48 h before the sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt assay. Cell proliferation was examined using a sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt-based cell-viability assay as described in the Materials and Methods section. (B) For the migration assay, cells were incubated with gintonin (10 μg/mL and 30 μg/mL) in the presence or absence of the lysophosphatidic acid 1/3 receptor inhibitor, Ki16425 (10μM), for 70 min. Migration of human umbilical-vein endothelial cells was assessed using the modified Boyden chamber as described in the Materials and Methods section. (C) Cells were transfected with 50nM control or with lysophosphatidic acid 1 or lysophosphatidic acid 3 short interfering RNA in serum-free M199 for 6 h, and incubated for an additional 24 h in growth media. The cells transfected with short interfering RNA were trypsinized, and cell migration induced by gintonin (10 μg/mL) was assessed using the modified Boyden-chamber assay. (D) Inhibition of lysophosphatidic acid 1/3 receptor attenuates gintonin-induced tube formation in human umbilical-vein endothelial cells. Cells were incubated in the presence or absence of the lysophosphatidic acid 1/3 receptor inhibitor, Ki16425 (10μM). (A, B, D) The data represent the means ± SD of the results of four to six independent experiments. *p < 0.05. **p < 0.001 compared with control. #p < 0.05 compared to without Ki16425. (C) The data represent the means ± SD of the results of four independent experiments. **p < 0.001 compared to without gintonin. #p < 0.05 compared with control short interfering RNA. Con, control; GT, gintonin; LPA, lysophosphatidic acid; siRNA, short interfering RNA.
3.4. Gintonin-mediated in vitro proliferation, migration, and tube formation of HUVECs are partially mediated by the activation of Rho kinase
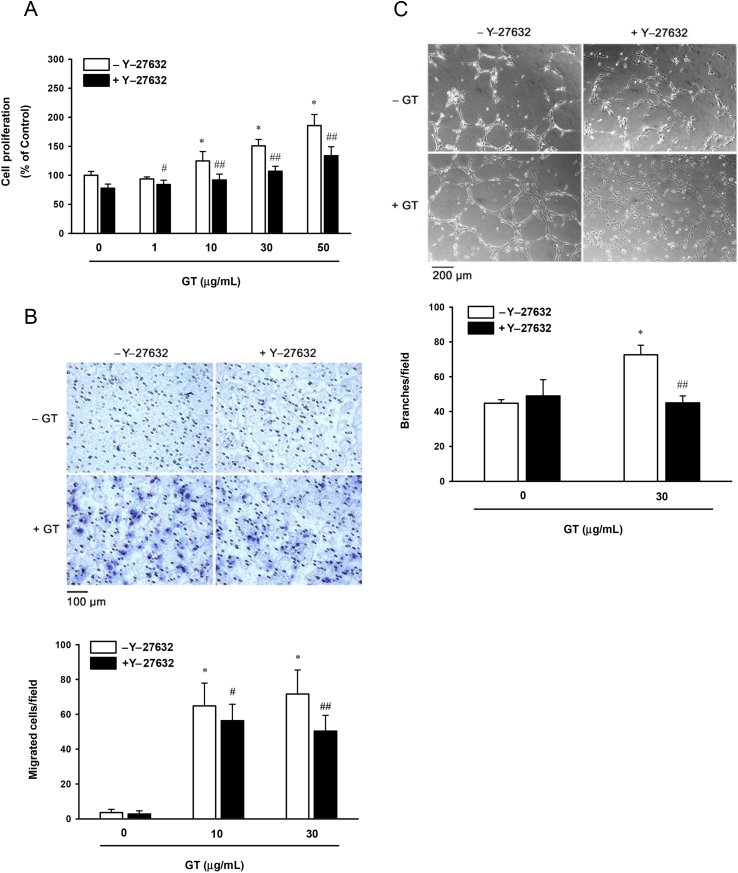
Rho kinase is one of the downstream signaling molecules of LPA-receptor activation involved in the migration of HUVECs. To investigate the involvement of Rho kinase signaling in gintonin-induced cellular events, we used the Rho kinase inhibitor, Y-27632. Incubation of HUVECs with Y-27632 decreased the cell viability induced by 1–50 μg/mL of gintonin by 10–28% (Fig. 4A). Y-27632 decreased the migration of HUVECs induced by 10 μg/mL and 30 μg/mL of gintonin by 13% and 30%, respectively (Fig. 4B). In addition, Y-27632 decreased the tube formation in HUVECs induced by 30 μg/mL of gintonin by 38% (Fig. 4C). These results support the fact that gintonin-induced proliferation, migration, and tube formation of HUVECs are partially mediated by the activation of Rho kinase.
Fig. 4.
A Rho kinase inhibitor attenuates gintonin-induced proliferation, migration, and tube formation. (A) Cells were incubated in the presence or absence of the Rho kinase inhibitor, Y-27632 (10μM), for 30 min, and then incubated with gintonin at different concentrations for another 48 h before the sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt assay. Cell proliferation was examined using a sodium 2,3,-bis(2-methoxy-4-nitro-5-sulfophenyl)-5-[(phenylamino)-carbonyl]-2H-tetrazolium inner salt-based cell-viability assay as described in the Materials and Methods section. (B) For the migration assay, cells were incubated with gintonin (10 μg/mL and 30 μg/mL) in the presence or absence of the Rho kinase inhibitor, Y-27632 (10μM), for 70 min. Migration of human umbilical-vein endothelial cells was assessed using the modified Boyden chamber as described in the Materials and Methods section. (C) Rho kinase inhibitor, Y-27632 (10μM), for 30 min on the plates coated with Matrigel, and then incubated with gintonin (30 μg/mL) for another 4 h. The data represent the means ± SD of the results of four to 12 independent experiments. *p < 0.001 compared with untreated control (gintonin 0 μg/ml). #p < 0.05. ##p < 0.001 compared to without Y-27632. Con, control; GT, gintonin.
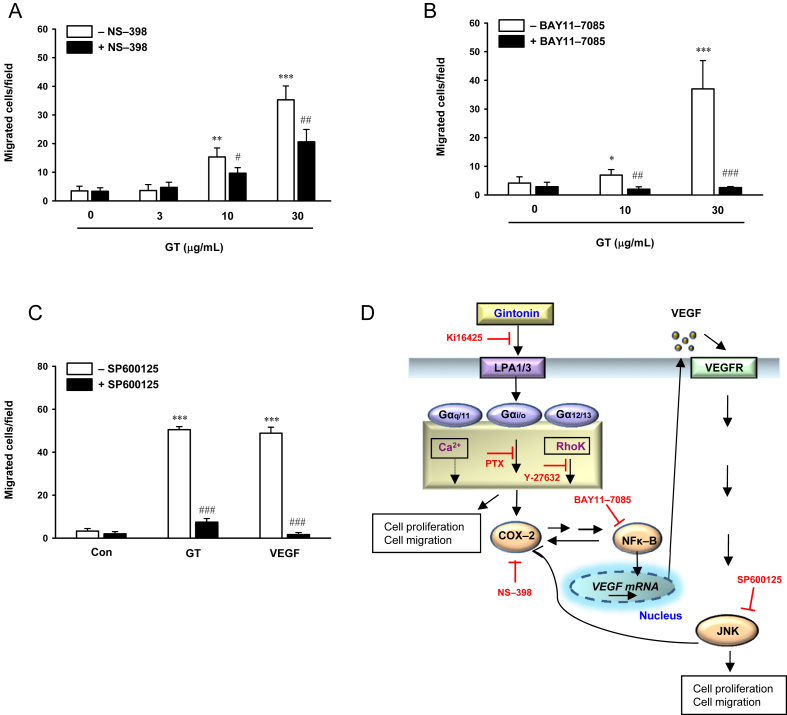
3.5. Gintonin-induced migration of HUVECs is mediated by the activation of COX-2, NF-κB, and c-Jun N-terminal kinase
COX-2 and NF-κB regulate the release and the level of VEGF expression [30]. In addition, COX-2 plays a role in VEGF-induced proliferation of HUVECs via the activation of c-Jun N-terminal kinase (JNK) [31]. To determine whether COX-2, NF-κB, and JNK signaling are involved in the gintonin-induced migration of HUVECs, we examined the effects of NS-398, a COX-2 inhibitor, BAY11-7085, an NF-κB inhibitor, and SP600125, a JNK inhibitor, on the migration of HUVECs using the modified Boyden-chamber assay. Treatment with NS-398 decreased the migration of HUVECs induced by 10 μg/mL and 30 μg/mL of gintonin by 13% and 30%, respectively (Fig. 5A). BAY11-7085 and SP600125 completely inhibited the migration of HUVECs induced by 10 μg/mL and 30 μg/mL of gintonin (Fig. 5B, C). These results indicate that gintonin-mediated migration of HUVECs involves the activation of COX-2/NF-κB and JNK signaling.
Fig. 5.
Attenuation of gintonin-induced migration by the inhibition of cyclooxygenase-2, nuclear factor kappa B, and c-Jun N-terminal kinase, and the proposed mechanism underlying the regulation of endothelial cell proliferation and migration by gintonin. (A–C) Cells were preincubated in the presence or absence of the (A) cyclooxygenase-2 inhibitor NS-398 (50μM), (B) nuclear factor kappa B inhibitor BAY11-7085 (10μM), and (C) c-Jun N-terminal kinase inhibitor SP600125 (20μM) for 60 min in the modified Boyden chamber. After incubation for an additional 120 min either with or without gintonin (3 μg/mL, 10 μg/mL, and 30 μg/mL), the effect of these inhibitors on gintonin-induced migration of human umbilical-vein endothelial cells was evaluated as described in the Materials and Methods section. (C) Gintonin (30 μg/mL), vascular endothelial growth factor (positive control, 200 ng/mL). The data represent the means ± SD of the results of four independent experiments. (D) Activation of the lysophosphatidic-acid receptor by gintonin stimulates the expression of vascular endothelial growth factor via cyclooxygenase-2 and nuclear factor kappa B. Vascular-endothelial-growth-factor receptor activation by vascular endothelial growth factor induces stimulation of c-Jun N-terminal kinase. Activated c-Jun N-terminal kinase, in turn, increases cyclooxygenase-2 expression. Enhanced lysophosphatidic-acid-receptor activation and vascular-endothelial-growth-factor receptor signaling have been related to the proliferation and migration of endothelial cells. *p < 0.05. **p < 0.01. ***p < 0.001 compared with control. #p < 0.05. ##p < 0.01. ###p < 0.001 compared to without inhibitors. Con, control; COX-2, cyclooxygenase-2; GT, gintonin; JNK, c-Jun N-terminal kinase; LPA, lysophosphatidic acid; NF-κB, nuclear factor kappa B; PTX, pertussis toxin; VEGF, vascular endothelial growth factor; VEGFR, vascular-endothelial-growth-factor receptor.
4. Discussion
Angiogenesis is a multistep process that involves the activation of receptors on the endothelial cells by growth factors, destruction of the basement membrane by proteases secreted from endothelial cells, proliferation and migration of endothelial cells, and tube formation by endothelial cells [11], [32]. Fibroblast growth factor, VEGF, platelet-derived growth factor, and transforming growth factors are the main angiogenic factors [33]. In addition, LPA stimulates proliferation, migration, mitogen-activated-protein-kinase phosphorylation, Rho activation, and Ca2+ mobilization through the activation of LPA1/3 receptors in HUVECs, which show angiogenic effects [22], [34], [35], [36]. Lipid molecules, like LPA and S1P, exert their effects indirectly and through the release of peptides, such as transforming growth factor [37] and VEGF [25], [38].
In a previous study, we showed that gintonin is a novel LPA–ginseng protein complex derived from ginseng [20]. Ginseng and LPA have angiogenic effects [9], [11], [22], and gintonin contains approximately 9.5% of LPA [20]; therefore, we examined the in vitro effects of gintonin on HUVECs. Similar to LPA, gintonin induced Ca2+ mobilization in rat B103 neuroblastoma cells transfected with LPA-receptor subtypes in our previous study [20]. Our previous study showed that gintonin stimulates bromodeoxyuridine incorporation, migration, and extracellular-related-kinase (ERK) phosphorylation in a dose-dependent manner in HUVECs [20]. Pertussis toxin (PTX) completely blocked the gintonin-stimulated migration and ERK phosphorylation [20].
In this study, we found that, while gintonin stimulated the proliferation and migration of HUVECs, which are essential steps for angiogenesis, ginsenosides Rb1 and Rg1 did not affect these processes (Fig. 1). The in vitro tube-formation assay for angiogenesis showed that gintonin (10 μg/mL and 30 μg/mL) stimulated the formation of capillary-like tubes on a Matrigel matrix (Fig. 2C). The activation of LPA receptor and Rho kinase is involved in gintonin-stimulated migration of HUVECs. These effects of gintonin are very similar to those of LPA observed in previous studies [22], [34], [35], [36].
LPA-receptor subtypes exert their roles by coupling with different types of G proteins, such as Gα12/13, Gαi/o, and Gαq11 [21], [39]. LPA receptors coupled with Gα12/13 are involved in Rho activation and morphological change [40]. Ca2+ mobilization through LPA-receptor activation is mediated mainly by Gαq11 and Gαi/o, followed by phospholipase C activation, Ca2+ release from intracellular organelles, or influx of Ca2+ into cells. Depending on the cellular system, the specific LPA-receptor subtypes or G proteins are expressed and activated to different extents. HUVECs predominantly express LPA1 and LPA3 [22]. LPA1 can effectively couple with Gαi/o and Gα12/13 than with Gαq11 [21], [23], [39], whereas LPA3 can mainly couple with Gαq11 [21], [23], [39]. Our results showed that gintonin-induced migration was decreased by the LPA1/3 receptor antagonist, Ki16425, and silencing LPA1/3 with siRNA; thus, the activation of LPA1/3 receptor may be involved in gintonin-induced migration (Fig. 5D). The inhibition of gintonin-induced migration and ERK phosphorylation in HUVECs by PTX suggested that these effects of gintonin on HUVECs are mediated through LPA receptors coupled with the PTX-sensitive G protein Gαi/o [20]. Thus, gintonin might mainly stimulate the migration of HUVECs through the activation of the LPA-receptor subtype LPA1 coupled with Gαi/o (Fig. 5D). The inhibition of gintonin-induced migration by a Rho kinase inhibitor indicated the possible involvement of Gα12/13.
LPA upregulates VEGF via the transactivation of epidermal growth factor receptor, and the activation of COX-2 and NF-κB in cancer cells, as well as in HUVECs [25]. Treatment of HUVECs with VEGF induces the expression of COX-2 [31]. COX-2 expression induced by VEGF was accompanied with cell proliferation, and was inhibited by the JNK inhibitor, SP600125, which indicated that COX-2 plays a key role in VEGF-induced wound healing via p38/JNK activation [31]. VEGF seems to stimulate autotaxin, which is the chemotactic-motility factor, and produces LPA from lysophosphatidylcholine [41]. Although gintonin and LPA itself can suppress the autotaxin activity by negative feedback [42], [43], the overexpression of VEGF can play a role as a potent mitogenic factor in certain types of cells like endothelial cells by positive feedback with autotaxin [41], [44].
Our results confirmed that gintonin also stimulates VEGF release in HUVECs, which indicates that gintonin-induced proliferation and migration could also be due to gintonin-stimulated VEGF release (Fig. 2D). We could observe a decrease in gintonin-induced migration by the COX-2 inhibitor (Fig. 5A). In addition, the NF-κB inhibitor and JNK inhibitor potently suppressed gintonin-induced migration (Fig. 5B, C). Our data suggest that gintonin might exert its angiogenic effect via LPA-receptor activation coupled with G proteins Gαi/o and Gα12/13 (Fig. 5D). The gintonin-stimulated migration of HUVECs seems to be dependent on the expression of COX-2 and NF-κB. The gintonin-stimulated VEGF release might be due to the activation of COX-2 and NF-κB, which are downstream signaling pathways in the activation of LPA receptors by LPA. The VEGF released can activate the VEGF receptor and stimulate the migration of HUVECs via JNK activation. JNK might activate COX-2 for further enhanced cell migration and proliferation similar to that observed previously in endothelial cells [31].
In conclusion, our study showed that gintonin stimulated in vitro proliferation, migration, VEGF release, and tube formation of HUVECs. In addition, our results also indicated that LPA-receptor activation and Rho kinase activation, and VEGF, COX-2, NF-κB, and JNK might be coupled to the angiogenic effects of gintonin. In a future study, if we show that gintonin can exhibit in vivo angiogenic and wound-healing effects, gintonin can be a potential therapeutic candidate for angiogenesis and wound healing.
Conflicts of interest
All authors have no conflicts of interest to declare.
Acknowledgments
This work was supported by the Basic Science Research Program (NRF-2014R1A1A2054538), which is funded by the Ministry of Education, Science, and Technology, and by the BK21 Plus Project fund to S.-Y.N.
References
- 1.Bao P., Kodra A., Tomic-Canic M., Golinko M.S., Ehrlich H.P., Brem H. The role of vascular endothelial growth factor in wound healing. J Surg Res. 2009;153:347–358. doi: 10.1016/j.jss.2008.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lamalice L., Le Boeuf F., Huot J. Endothelial cell migration during angiogenesis. Circ Res. 2007;100:782–794. doi: 10.1161/01.RES.0000259593.07661.1e. [DOI] [PubMed] [Google Scholar]
- 3.Post M.J., Laham R., Sellke F.W., Simons M. Therapeutic angiogenesis in cardiology using protein formulations. Cardiovasc Res. 2001;49:522–531. doi: 10.1016/s0008-6363(00)00216-9. [DOI] [PubMed] [Google Scholar]
- 4.Emanueli C., Madeddu P. Changing the logic of therapeutic angiogenesis for ischemic disease. Trends Mol Med. 2005;11:207–216. doi: 10.1016/j.molmed.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 5.Khurana R., Simons M., Martin J.F., Zachary I.C. Role of angiogenesis in cardiovascular disease: a critical appraisal. Circulation. 2005;112:1813–1824. doi: 10.1161/CIRCULATIONAHA.105.535294. [DOI] [PubMed] [Google Scholar]
- 6.Ziebart T., Yoon C.H., Trepels T., Wietelmann A., Braun T., Kiessling F., Stein S., Grez M., Ihling C., Muhly-Reinholz M. Sustained persistence of transplanted proangiogenic cells contributes to neovascularization and cardiac function after ischemia. Circ Res. 2008;103:1327–1334. doi: 10.1161/CIRCRESAHA.108.180463. [DOI] [PubMed] [Google Scholar]
- 7.Mochizuki M., Yoo Y.C., Matsuzawa K., Sato K., Saiki I., Tono-oka S., Samukawa K., Azuma I. Inhibitory effect of tumor metastasis in mice by saponins, ginsenoside-Rb2, 20(R)- and 20(S)-ginsenoside-Rg3, of red ginseng. Biol Pharm Bull. 1995;18:1197–1202. doi: 10.1248/bpb.18.1197. [DOI] [PubMed] [Google Scholar]
- 8.Zhou W., Chai H., Lin P.H., Lumsden A.B., Yao Q., Chen C.J. Molecular mechanisms and clinical applications of ginseng root for cardiovascular disease. Med Sci Monit. 2004;10:RA187–RA192. [PubMed] [Google Scholar]
- 9.Kim Y.M., Namkoong S., Yun Y.G., Hong H.D., Lee Y.C., Ha K.S., Lee H., Kwon H.J., Kwon Y.G., Kim Y.M. Water extract of Korean Red Ginseng stimulates angiogenesis by activating the PI3K/Akt-dependent ERK1/2 and eNOS pathways in human umbilical vein endothelial cells. Biol Pharm Bull. 2007;30:1674–1679. doi: 10.1248/bpb.30.1674. [DOI] [PubMed] [Google Scholar]
- 10.Hong Y.J., Kim N., Lee K., Sonn C.H., Lee J.E., Kim S.T., Baeg I.H., Lee K.M. Korean Red Ginseng (Panax ginseng) ameliorates type 1 diabetes and restores immune cell compartments. J Ethnopharmacol. 2012;144:225–233. doi: 10.1016/j.jep.2012.08.009. [DOI] [PubMed] [Google Scholar]
- 11.Morisaki N., Watanabe S., Tezuka M., Zenibayashi M., Shiina R., Koyama N., Kanzaki T., Saito Y. Mechanism of angiogenic effects of saponin from ginseng Radix rubra in human umbilical vein endothelial cells. Br J Pharmacol. 1995;115:1188–1193. doi: 10.1111/j.1476-5381.1995.tb15023.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yue P.Y., Mak N.K., Cheng Y.K., Leung K.W., Ng T.B., Fan D.T., Yeung H.W., Wong R.N. Pharmacogenomics and the yin/yang actions of ginseng: anti-tumor, angiomodulating and steroid-like activities of ginsenosides. Chin Med. 2007;2:6. doi: 10.1186/1749-8546-2-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yue P.Y., Wong D.Y., Ha W.Y., Fung M.C., Mak N.K., Yeung H.W., Leung H.W., Chan K., Liu L., Fan T.P. Elucidation of the mechanisms underlying the angiogenic effects of ginsenoside Rg1 in vivo and in vitro. Angiogenesis. 2005;8:205–216. doi: 10.1007/s10456-005-9000-2. [DOI] [PubMed] [Google Scholar]
- 14.Yue P.Y., Wong D.Y., Wu P.K., Leung P.Y., Mak N.K., Yeung H.W., Liu L., Cai Z., Jiang Z.H., Fan T.P. The angiosuppressive effects of 20(R)-ginsenoside Rg3. Biochem Pharmacol. 2006;72:437–445. doi: 10.1016/j.bcp.2006.04.034. [DOI] [PubMed] [Google Scholar]
- 15.Shin K.O., Seo C.H., Cho H.H., Oh S., Hong S.P., Yoo H.S., Hong J.T., Oh K.W., Lee Y.M. Ginsenoside compound K inhibits angiogenesis via regulation of sphingosine kinase-1 in human umbilical vein endothelial cells. Arch Pharm Res. 2014;37:1183–1192. doi: 10.1007/s12272-014-0340-6. [DOI] [PubMed] [Google Scholar]
- 16.Kimura Y., Sumiyoshi M., Kawahira K., Sakanaka M. Effects of ginseng saponins isolated from red ginseng roots on burn wound healing in mice. Br J Pharmacol. 2006;148:860–870. doi: 10.1038/sj.bjp.0706794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim W.K., Song S.Y., Oh W.K., Kaewsuwan S., Tran T.L., Kim W.S., Sung J.H. Wound-healing effect of ginsenoside Rd from leaves of Panax ginseng via cyclic AMP-dependent protein kinase pathway. Eur J Pharmacol. 2013;702:285–293. doi: 10.1016/j.ejphar.2013.01.048. [DOI] [PubMed] [Google Scholar]
- 18.Leung K.W., Cheung L.W., Pon Y.L., Wong R.N., Mak N.K., Fan T.P., Au S.C., Tombran-Tink J., Wong A.S. Ginsenoside Rb1 inhibits tube-like structure formation of endothelial cells by regulating pigment epithelium-derived factor through the oestrogen beta receptor. Br J Pharmacol. 2007;152:207–215. doi: 10.1038/sj.bjp.0707359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pyo M.K., Choi S.H., Hwang S.H., Shin T.J., Lee B.H., Lee S.M., Lim Y., Kim D., Nah S.Y. Novel glycoproteins from ginseng. J Ginseng Res. 2011;35:92–103. [Google Scholar]
- 20.Hwang S.H., Shin T.J., Choi S.H., Cho H.J., Lee B.H., Pyo M.K., Lee J.H., Kang J.K., Kim H.J., Park C.W. Gintonin, newly identified compounds from ginseng, is novel lysophosphatidic acids–protein complexes and activates G protein-coupled lysophosphatidic acid receptors with high affinity. Mol Cells. 2012;33:151–162. doi: 10.1007/s10059-012-2216-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chun J., Hla T., Lynch K.R., Spiegel S., Moolenaar W.H. International Union of Basic and Clinical Pharmacology. LXXVIII. Lysophospholipid receptor nomenclature. Pharmacol Rev. 2010;62:579–587. doi: 10.1124/pr.110.003111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee H., Goetzl E.J., An S. Lysophosphatidic acid and sphingosine 1-phosphate stimulate endothelial cell wound healing. Am J Physiol Cell Physiol. 2000;278:C612–C618. doi: 10.1152/ajpcell.2000.278.3.C612. [DOI] [PubMed] [Google Scholar]
- 23.Lin C.I., Chen C.N., Lin P.W., Chang K.J., Hsieh F.J., Lee H. Lysophosphatidic acid regulates inflammation-related genes in human endothelial cells through LPA1 and LPA3. Biochem Biophys Res Commun. 2007;363:1001–1008. doi: 10.1016/j.bbrc.2007.09.081. [DOI] [PubMed] [Google Scholar]
- 24.Sako A., Kitayama J., Shida D., Suzuki R., Sakai T., Ohta H., Nagawa H. Lysophosphatidic acid (LPA)-induced vascular endothelial growth factor (VEGF) by mesothelial cells and quantification of host-derived VEGF in malignant ascites. J Surg Res. 2006;130:94–101. doi: 10.1016/j.jss.2005.08.007. [DOI] [PubMed] [Google Scholar]
- 25.Lin C.I., Chen C.N., Huang M.T., Lee S.J., Lin C.H., Chang C.C., Lee H. Lysophosphatidic acid upregulates vascular endothelial growth factor-C and tube formation in human endothelial cells through LPA(1/3), COX-2, and NF-kappaB activation- and EGFR transactivation-dependent mechanisms. Cell Signal. 2008;20:1804–1814. doi: 10.1016/j.cellsig.2008.06.008. [DOI] [PubMed] [Google Scholar]
- 26.Zhao L., Wu Y., Xu Z., Wang H., Zhao Z., Li Y., Yang P., Wei X. Involvement of COX-2/PGE2 signalling in hypoxia-induced angiogenic response in endothelial cells. J Cell Mol Med. 2012;16:1840–1855. doi: 10.1111/j.1582-4934.2011.01479.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gimbrone M.A., Jr., Cotran R.S., Folkman J. Human vascular endothelial cells in culture. Growth and DNA synthesis. J Cell Biol. 1974;60:673–684. doi: 10.1083/jcb.60.3.673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Scudiero D.A., Shoemaker R.H., Paull K.D., Monks A., Tierney S., Nofziger T.H., Currens M.J., Seniff D., Boyd M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988;48:4827–4833. [PubMed] [Google Scholar]
- 29.Al-Nedawi K., Meehan B., Kerbel R.S., Allison A.C., Rak J. Endothelial expression of autocrine VEGF upon the uptake of tumor-derived microvesicles containing oncogenic EGFR. Proc Natl Acad Sci U S A. 2009;106:3794–3799. doi: 10.1073/pnas.0804543106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Symowicz J., Adley B.P., Woo M.M., Auersperg N., Hudson L.G., Stack M.S. Cyclooxygenase-2 functions as a downstream mediator of lysophosphatidic acid to promote aggressive behavior in ovarian carcinoma cells. Cancer Res. 2005;65:2234–2242. doi: 10.1158/0008.5472.CAN-04-2781. [DOI] [PubMed] [Google Scholar]
- 31.Wu G., Mannam A.P., Wu J., Kirbis S., Shie J.L., Chen C., Laham R.J., Sellke F.W., Li J. Hypoxia induces myocyte-dependent COX-2 regulation in endothelial cells: role of VEGF. Am J Physiol Heart Circ Physiol. 2003;285:H2420–H2429. doi: 10.1152/ajpheart.00187.2003. [DOI] [PubMed] [Google Scholar]
- 32.Pepper M.S. Role of the matrix metalloproteinase and plasminogen activator–plasmin systems in angiogenesis. Arterioscler Thromb Vasc Biol. 2001;21:1104–1117. doi: 10.1161/hq0701.093685. [DOI] [PubMed] [Google Scholar]
- 33.Folkman J., Shing Y. Angiogenesis. J Biol Chem. 1992;267:10931–10934. [PubMed] [Google Scholar]
- 34.English D., Kovala A.T., Welch Z., Harvey K.A., Siddiqui R.A., Brindley D.N., Garcia J.G. Induction of endothelial cell chemotaxis by sphingosine 1-phosphate and stabilization of endothelial monolayer barrier function by lysophosphatidic acid, potential mediators of hematopoietic angiogenesis. J Hematother Stem Cell Res. 1999;8:627–634. doi: 10.1089/152581699319795. [DOI] [PubMed] [Google Scholar]
- 35.Panetti T.S., Nowlen J., Mosher D.F. Sphingosine-1-phosphate and lysophosphatidic acid stimulate endothelial cell migration. Arterioscler Thromb Vasc Biol. 2000;20:1013–1019. doi: 10.1161/01.atv.20.4.1013. [DOI] [PubMed] [Google Scholar]
- 36.van Meeteren L.A., Ruurs P., Stortelers C., Bouwman P., van Rooijen M.A., Pradère J.P., Pettit T.R., Wakelam M.J., Saulnier-Blache J.S., Mummery C.L. Autotaxin, a secreted lysophospholipase D, is essential for blood vessel formation during development. Mol Cell Biol. 2006;26:5015–5022. doi: 10.1128/MCB.02419-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Piazza G.A., Ritter J.L., Baracka C.A. Lysophosphatidic acid induction of transforming growth factors alpha and beta: modulation of proliferation and differentiation in cultured human keratinocytes and mouse skin. Exp Cell Res. 1995;216:51–64. doi: 10.1006/excr.1995.1007. [DOI] [PubMed] [Google Scholar]
- 38.Lin C.I., Chen C.N., Huang M.T., Lee S.J., Lin C.H., Chang C.C., Lee H. Lysophosphatidic acid up-regulates vascular endothelial growth factor-C and lymphatic marker expressions in human endothelial cells. Cell Mol Life Sci. 2008;65:2740–2751. doi: 10.1007/s00018-008-8314-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Meyer zu Heringdorf D., Jakobs K.H. Lysophospholipid receptors: signalling, pharmacology and regulation by lysophospholipid metabolism. Biochim Biophys Acta. 2007;1768:923–940. doi: 10.1016/j.bbamem.2006.09.026. [DOI] [PubMed] [Google Scholar]
- 40.Kranenburg O., Poland M., van Horck F.P., Drechsel D., Hall A., Moolenaar W.H. Activation of RhoA by lysophosphatidic acid and Galpha12/13 subunits in neuronal cells: induction of neurite retraction. Mol Biol Cell. 1999;10:1851–1857. doi: 10.1091/mbc.10.6.1851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ptaszynska M.M., Pendrak M.L., Stracke M.L., Roberts D.D. Autotaxin signaling via lysophosphatidic acid receptors contributes to vascular endothelial growth factor-induced endothelial cell migration. Mol Cancer Res. 2010;8:309–321. doi: 10.1158/1541-7786.MCR-09-0288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hwang S.H., Lee B.H., Kim H.J., Cho H.J., Shin H.C., Im K.S., Choi S.H., Shin T.J., Lee S.M., Nam S.W. Suppression of metastasis of intravenously-inoculated B16/F10 melanoma cells by the novel ginseng-derived ingredient, gintonin: involvement of autotaxin inhibition. Int J Oncol. 2013;42:317–326. doi: 10.3892/ijo.2012.1709. [DOI] [PubMed] [Google Scholar]
- 43.van Meeteren L.A., Ruurs P., Christodoulou E., Goding J.W., Takakusa H., Kikuchi K., Perrakis A., Nagano T., Moolenaar W.H. Inhibition of autotaxin by lysophosphatidic acid and sphingosine 1-phosphate. J Biol Chem. 2005;280:21155–21161. doi: 10.1074/jbc.M413183200. [DOI] [PubMed] [Google Scholar]
- 44.Ptaszynska M.M., Pendrak M.L., Bandle R.W., Stracke M.L., Roberts D.D. Positive feedback between vascular endothelial growth factor-A and autotaxin in ovarian cancer cells. Mol Cancer Res. 2008;6:352–363. doi: 10.1158/1541-7786.MCR-07-0143. [DOI] [PMC free article] [PubMed] [Google Scholar]