Abstract
Background
Drug-paired environments can act as stimuli that elicit drug craving. In humans, drug craving is influenced by the amount of time abstinent, number of past periods of abstinence, and inadvertent exposure to the previously abused drug. The current experiments were designed to determine the effects of (a) the duration of abstinence on expression of EtOH-seeking; (b) EtOH priming following a short and long abstinence period; and (c) repeated deprivation cycles on relapse drinking and EtOH-seeking.
Methods
Rats were allowed to self-administer 15% ethanol (EtOH), processed through extinction training, maintained in a home cage for a designated EtOH-free period, and then reintroduced to the operant context in the absence of EtOH. The experiments examined the effects of: 1) various home cage duration periods (1 to 8 weeks), 2) priming injections of EtOH in the Pavlovian Spontaneous Recovery (PSR; 14 days after extinction) and Reinstatement of Responding (RoR; I day after extinction) models, and 3) exposure to repeated cycles of EtOH access-deprivation on relapse drinking and EtOH-seeking behavior.
Results
Highest expression of EtOH-seeking was observed following 6 weeks of home-cage maintenance. Priming injections of EtOH were more efficacious at stimulating/enhancing EtOH-seeking in the PSR than RoR model. Exposure to repeated cycles of EtOH deprivation and access enhanced and prolonged relapse drinking and the expression of EtOH-seeking (318 ± 22 responses), which was not observed in rats given equivalent consistent exposure to EtOH (66 ± 11 responses).
Discussion
Overall, the data indicated that the PSR model has ecological validity; factors that enhance EtOH craving in humans enhance the expression of EtOH seeking in the PSR test. The data also detail factors that need to be examined to determine the biological basis of EtOH-seeking (e.g., neuroadaptations that occur during the incubation period and following repeated cycles of EtOH drinking and abstinence).
Keywords: Alcohol-preferring (P) rats, alcohol-seeking behavior, repeated alcohol deprivations, alcohol relapse
INTRODUCTION
Alcohol addiction is characterized as a chronic reoccurring illness. In humans, the effects of environmental cues to elicit drug-seeking behaviors after a prolonged abstinence period may contribute to the chronic relapsing nature of alcohol addiction (O’Brien et al., 1998). Cues associated with alcohol use can readily enhance self-reported craving for alcohol, and this effect is heightened in alcohol-dependent patients (Gauggel et al., 2010).
Clinical findings have shown that real-world drug-associated environments have an important role in precipitating craving in abstinent drug users (Conklin et al., 2008; 2009). Context in which alcohol has been consumed has also been shown to have a significant impact on Pavlovian-conditioned alcohol-seeking in non-selected rats (Chaudhri et al., 2008a, b; 2009; Janak and Chaudhri, 2010). In these studies, alcohol consumption occurred in a specific environment (context A), extinction training was conducted in a different context (context B) and then animals are returned the original context (A) for renewal context seeking. The renewal paradigm can readily elicit EtOH-seeking and was readily observed with non-selected stock rats 21 days after the last extinction session (Chaudhri et al., 2008a,b; 2009; Janak and Chaudhri, 2010). In alcoholics undergoing treatment, relapse can occur after weeks to months of abstinence (Ferri et al., 2006). Therefore, it would be important to determine the impact of prolonged abstinence on environmental context-induced EtOH-seeking in a suitable animal model of alcoholism (McBride et al., 2014).
Context has been shown to influence extinction learning and reinstatement of previously learned behaviors (Bouton, 2002). In a spontaneous recovery paradigm, subjects are allowed to self-administer in a specific environment, the behavior is extinguished in the same environment, the subjects are withheld from that environment for a certain time, and behavior is recorded when the animals are returned to the original environment. The expression of a spontaneous recovery is directly correlated to reward saliency (Honig and Staddon, 1977), contextual cues associated with first-learned signals, and the amount of first- and second-learned associations (Brooks, 2000). In general, the spontaneous recovery phenomenon has been asserted to be the result of an intrinsic shift away from the recent extinction (second-) learning to the initial reinforced learning responses, which reflects an intrinsic motivation to obtain the previously administered reward (Rescorla, 2001, Bouton, 2002, 2004). Since spontaneous recovery is defined in the alcohol clinical literature as the cessation of alcohol consumption in human alcoholics without treatment intervention, we have altered the term to Pavlovian Spontaneous Recovery (PSR). PSR is a unique phenomenon in that it is time dependent, and the behavior appears to be dependent on the re-exposure of the organism to cues in the behavioral environment previously associated with the reinforcer. The PSR model has been used to study environmental context-induced EtOH-seeking behavior following a prolonged period of rest after extinction training (Rodd-Henricks et al., 2002a, b; Rodd et al., 2006; Hauser et al., 2011) and the expression of PSR can be enhanced by exposure to EtOH odor cues or EtOH priming (Rodd-Henricks et al. 2002a, b). This model measures EtOH-seeking after physical signs of withdrawal have dissipated and may be a better measure of craving-like behavior that persists in humans who have been abstinent for many weeks to months. Thus far, the duration of the ‘rest’ period following extinction on expression of EtOH-seeking has not been fully explored.
Many alcoholics go through repeated periods of high alcohol drinking and periods of abstinence (Burish et al., 1981; Hilbrom 1990; McMillen 1997). Repeated cyclic patterns of consumption and deprivation may have severe consequences, as suggested by findings that multiple previous detoxifications are associated with a reduction in the response to treatment of withdrawal symptoms and heavier drinking during relapse (Malcolm et al., 2000). Repeated alcohol deprivation cycles have been reported to progressively increase EtOH self-administration with each cycle (Rodd et al., 2003). However, the effects of repeated deprivation cycles on expression of EtOH-seeking have not been adequately addressed.
Inadvertent exposure to the past abused drug or a compound that produces similar intrinsic effects can stimulate drug-seeking in humans (Field et al., 2008:; de Wit 1996). Stewart and colleagues paralleled this phenomenon when creating the Reinstatement of Responding (RoR) model of drug craving (de Wit and Stewart, 1981; Stewart, 1983, Shaham et al., 1997). Briefly, in the RoR model, animals are allowed to self-administer a drug for a certain time period and then processed through extinction training in the same environment. Testing for drug-seeking occurs following experimenter administration of the past self-administered drug or another compound immediately prior to reintroduction into the test environment. Priming doses of EtOH or exposure to stress can elicit EtOH-seeking in the RoR model (Le et al., 1998, 1999). In the PSR model, P rats readily demonstrate EtOH-seeking behavior without priming or use of stress, whereas this is not the case in the RoR model. A main difference between the two procedures is the length of the interval between the last extinction session and testing for EtOH-seeking, with the results suggesting that during the extended abstinence period neuroadaptations are occurring which enhance the expression of EtOH-seeking behavior. If this is the case, then EtOH-priming would be expected to have a greater impact on EtOH-seeking in the PSR model than the RoR model.
The objectives of the current study are to (1) determine the effects of the duration of abstinence on expression of EtOH-seeking in the PSR test; (2) compare the effects of EtOH priming on expression of EtOH-seeking in the RoR vs. PSR model; and (3) determine the effects of repeated deprivation cycles on expression of EtOH-seeking in the PSR test. The overall hypotheses to be tested are that robust EtOH-seeking behavior will still be observed in alcohol-preferring (P) rats after 4 or more weeks of abstinence, EtOH-priming will have more profound effects in the PSR than RoR model, and repeated cycles of deprivation will progressively increase relapse drinking and expression of EtOH-seeking behavior.
MATERIALS AND METHODS
Animals
Adult male P rats from the 66th – 68th generations weighing 250–325g at the start of the experiment were used. Rats were maintained on a 12-hr reversed light-dark cycle (lights off at 0900 hr). Food and water were available ad libitum throughout the experiment, except during operant testing. The animals used in these experiments were maintained in facilities fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care (AAALAC). All research protocols were approved by the institutional animal care and use committee and are in accordance with the guidelines of the Institutional Care and Use Committee of the National Institute on Drug Abuse, National Institutes of Health, and the Guide for the Care and Use of Laboratory Animals (Research Institute for Laboratory Animal Research, 2011).
Operant Training
EtOH self-administration procedures were conducted in standard two-lever experimental chambers (Coulbourn Instruments, Allentown, PA), as previously described (Rodd et al., 2006). Rats were placed into the operant chamber to self-train. Operant sessions were 60-min in duration and occurred daily for 10 weeks (Fig. 1; Hauser et al., 2011, Rodd et al. 2006). The EtOH concentration used for operant administration was 15% (vol/vol) and the rats received 0.1 ml of the EtOH per dipper presentation. During the initial 4 weeks of daily operant access, both solutions (water and EtOH) were reinforced on an FR-1 schedule. At the end of this time, the response requirement for EtOH was increased to an FR-3 schedule for 3 weeks, and then to FR-5 schedule for 3 weeks. The water lever was maintained on the FR1 schedule to allow easy access to water. Water was available so that the rats always had a choice between water and 15% EtOH. After the P rats had established stable levels of responding on the FR5 schedule for EtOH and FR1 for water, they underwent 7 days of extinction (60 min/day), when neither water nor EtOH was available (Hauser et al., 2011; Rodd et al. 2006). With the exception of no fluid being presented, the delivery system operated exactly as the preceding EtOH self-administration sessions.
Figure 1.
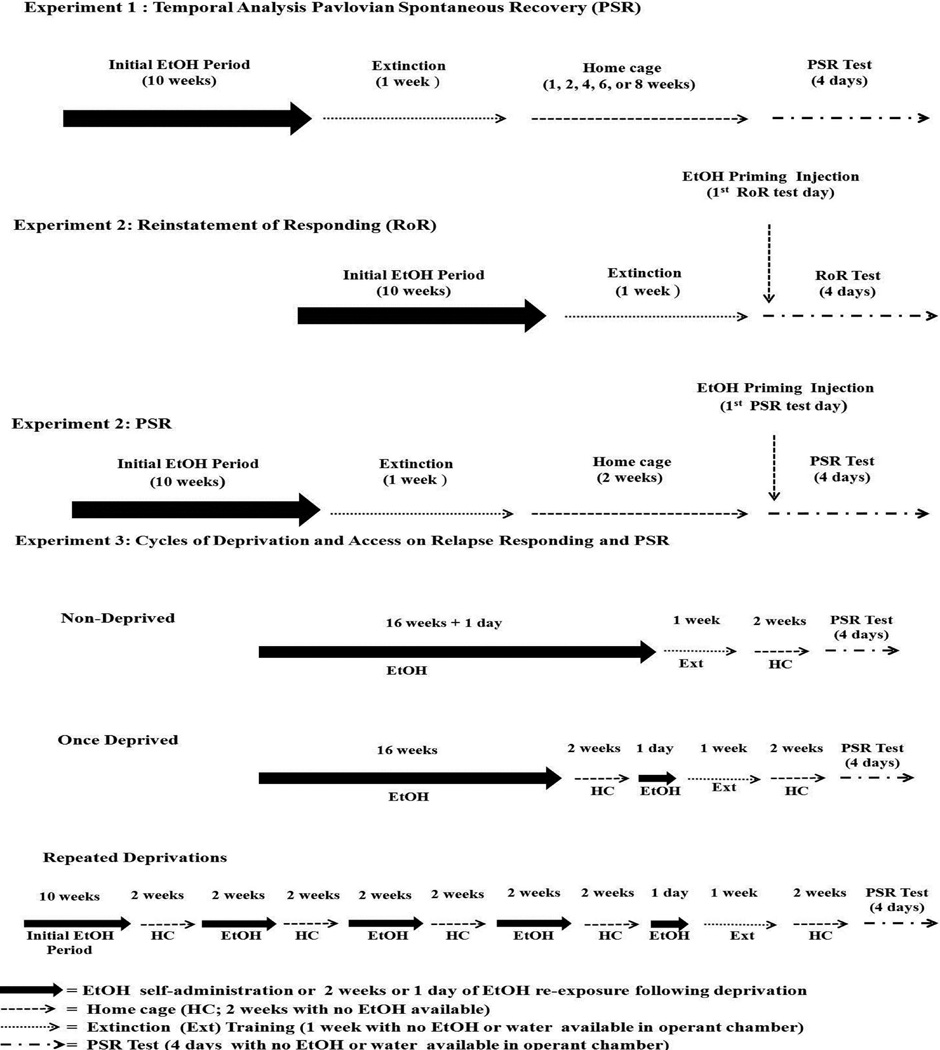
Depicts the experimental time lines for the PSR, RoR, and deprivation experiments.
Experiment 1 – Temporal Analysis of the Development of Context-Induced EtOH-Seeking
After extinction training, rats were maintained in the home cages for 1, 2, 4, 6, or 8 weeks (Fig. 1; n = 61, 11–13/group), before being returned to the operant chambers for PSR testing (no EtOH or water present) for 4 consecutive sessions (Hauser et al., 2011; Rodd et al., 2006). There are 4 consecutive PSR sessions because previous studies have shown that exposure to EtOH odor cues or EtOH priming (Rodd-Henricks et al., 2002a, b) and some drugs (Dhaher et al., 2010) may enhance PSR responding for more than one session.
Experiment 2 – Priming Injections of Ethanol in the PSR and RoR Models
In the RoR model, expression of EtOH-seeking is usually observed following priming or stress, whereas a robust expression of EtOH-seeking by P rats can be observed in the PSR model without these manipulations. This experiment was conducted to determine the effects of EtOH priming on EtOH-seeking behavior the day after the last extinction session (RoR procedure) and 2 weeks after the last extinction session (PSR procedure) (Fig. 1). All rats were given 10 weeks of daily operant access to EtOH, followed by 7 consecutive extinction sessions. For the RoR rats, the effects of an EtOH priming dose was tested on the day following the last extinction session (n = 59; n = 7–12/injection condition). Rats were treated with 0, 0.5 or 1.0 g/kg immediately or 30 min prior to EtOH-seeking testing (6 groups total). The immediate and 30 min time points prior to PSR were chosen to tease out any stimulatory effects of EtOH on locomotor activity (LMA) because previous studies have shown that low doses of EtOH can stimulate LMA during the 1st 30 minutes on the ascending end of the dose response curve but after the 1st 30 minutes LMA is relative to baseline on the descending end of dose response curve (Waller et al., 1986; Rodd et al., 2004). For PSR rats, the standard 2 week home cage maintenance period following the last extinction session occurred prior to examining the effects of EtOH priming dose on EtOH-seeking (n = 58; n = 8–12/injection condition). For both RoR and PSR testing, saline group sizes were 7–8, while for all EtOH doses tested, the group sizes were 10–12. The rats received a single injection prior to the 1st seeking session, but seeking testing (no EtOH or water present) occurred over 4 consecutive sessions.
Experiment 3 – Effects of Repeated Cycles of Deprivation and Access on Relapse Responding and Context-Induced EtOH-Seeking
Consistent findings have indicated that exposure to repeated deprivation cycles enhance responding for EtOH under operant conditions in a step-wise manner. To control for a history of high amount of lever presses, an additional control group was added to this experiment. This group was given an FR1 schedule for weeks 1–3, FR5 for weeks 3–4, and FR10 from week 5 onward.
There were 4 test groups in this experiment (n = 46; 10–12/group). Two groups of rats were never deprived of EtOH prior to extinction (an FR5 and FR10 constant group). These rats were given consecutive daily operant sessions for 16 weeks + 1 day. A once deprived group (FR5) received 16 weeks of consecutive daily operate sessions deprived for 2 weeks, reinstated for a single session, and then given extinction training. The repeated deprivation group (FR5) was given 10 weeks of daily access to EtOH, 2 weeks deprivation, 2 weeks access, 2 weeks deprivation, 2 weeks access, 2 weeks deprivation, 2 weeks access, 2 weeks deprivation, 1 day reinstatement, and then given 1 week of extinction training (Fig 1). All rats received a total of 16 weeks +1 day of operant EtOH access. PSR testing (no EtOH or water present) occurred 2 weeks after completion of extinction training and was conducted over 4 consecutive sessions (Hauser et al., 2011; Rodd et al., 2006).
Statistical Analyses
Overall operant responding (60 min) on EtOH and water levers data were analyzed with a mixed factorial ANOVA with a between subject factors of dose and time point and a repeated measure of ‘session’. The baseline measure for the factor of ‘session’ was the average number of responses on the EtOH (or water) lever for the last 3 extinction sessions. Operant EtOH responding data were also analyzed in 10-minute blocks, which required the additional repeated measure of time. Post-hoc Tukey’s b tests were performed to determine group differences.
Results
Experiment 1 – Temporal Analysis of the Development of Context-Induced EtOH-Seeking
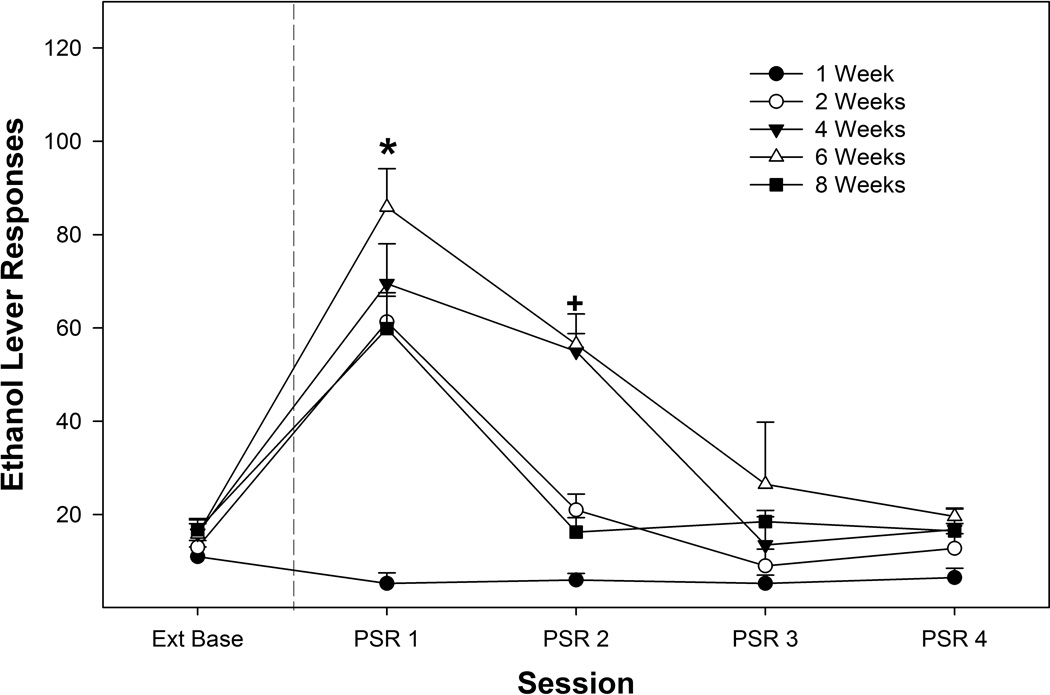
Examining the average number of responses on the lever previously associated with the delivery of EtOH between extinction average and the 4 PSR test sessions revealed a significant ‘Session’ by ‘Group’ interaction (Fig. 1, F16,224 = 4.90; p < 0.001; Figure 2). There was no significant effect of ‘Group’ for extinction baseline or the 3rd and 4th PSR test sessions (F4,58 values < 1.27; p values > 0.29). In contrast, during the 1st and 2nd PSR test session, there were significant differences between temporal ‘Groups’ (F4,58 values > 13.58; p values < 0.001). Post-hoc comparisons revealed that, during the 1st PSR test session, rats given 2, 4, 6 or 8 weeks of home cage maintenance responded more than rats given a single week of home cage maintenance, and rats maintained in the home cage for 6 weeks responded more than rats given a home cage period of 2 or 8 weeks. During the 2nd PSR test session the rats exposed to a home cage period of 4 or 6 weeks responded significantly more than all other groups. Paired t-tests revealed that rats maintained in their home cage for 2, 4, 6 or 8 weeks responded more during the 1st PSR test session than compared to extinction lever of responding (p values < 0.002) and the 4 or 6 week groups responded more during the 2nd PSR test session compared to extinction lever of responding (p values < 0.013). There were no significant differences for responses on the lever previously associated with the delivery of water for any experiment (data not shown).
Figure 2.
Depicts the mean (± SEM) responses on the lever previously associated with the delivery of EtOH during PSR testing as a function of homecage duration period. * indicates that rats in the 2-, 4-, 6- and 8- week groups responded more than extinction baseline (Ext Base) and the 6-week group responded significantly more than the 2- and 8- week groups. + indicates that 4- and 6- week groups are elevated compared to extinction baseline and are significantly different than all other groups.
Experiment 2 – Priming Injections of Ethanol in the PSR and RoR Models
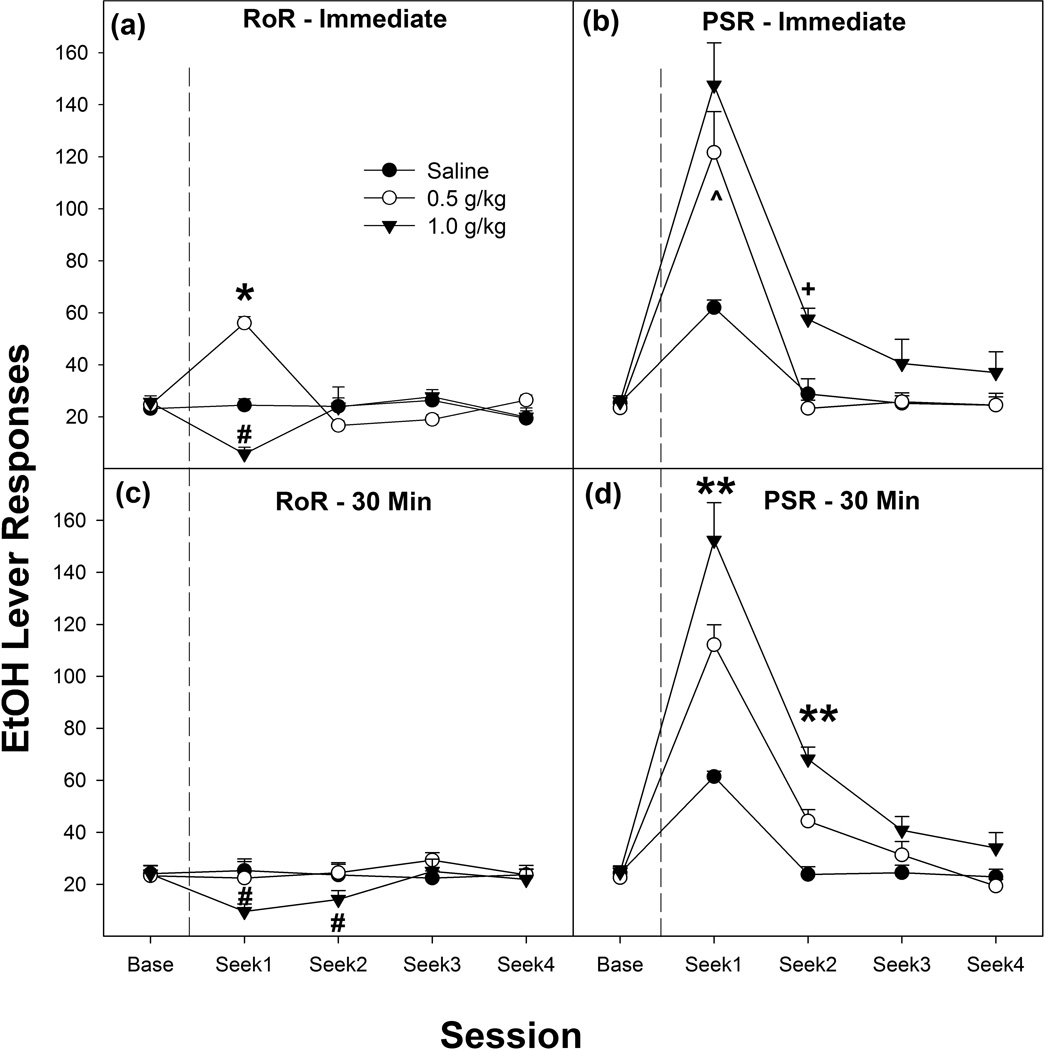
The overall analysis revealed a significant ‘Model’ (PSR vs RoR) × ‘Dose’ X ‘Time of Injection’ × ‘Session’ interaction (F8, 420 = 4.15; p < 0.001; Figure 3). Reducing the analysis to examine responding on the lever previously associated with the delivery of EtOH immediately following the EtOH priming injection (top two panels, Figures. 3a and 3b) revealed a significant ‘Model’ X ‘Dose’ X ‘Session” interaction (F8, 216 = 7.29; p < 0.0001). In P rats administered EtOH immediately prior to RoR testing, there was a significant ‘Dose’ × ‘Session’ interaction (F8, 108 = 17.75; p < 0.0001). P rats administered 0.5 g/kg EtOH immediately prior to RoR testing displayed an increase in responding on the lever previously associated with the delivery of EtOH, while rats administered 1.0 g/kg EtOH displayed a reduction in responding, compared to saline control groups (top left panel, Fig 3a).
Figure 3.
Depicts the mean (± SEM) responses on the lever previously associated with the delivery of EtOH during RoR (Figs 3a & c) or PSR (Figs 3b & 3d) testing as a function of a priming injection of 0, 0.5 or 1.0 g/kg i.p. EtOH given immediately or 30 min prior to the 1st test session. # indicates that the 1.0 g/kg EtOH groups is significantly less than saline controls. * indicates that 0.5 g/kg treated rats are significantly greater than saline controls and extinction baseline. ^ indicates that both the 0.5 and 1.0 g/kg EtOH groups are significantly greater than saline controls and extinction baseline. + indicates that the 1.0 g/kg group are significantly greater than all other groups and extinction baseline. ** indicates that all groups are different from each other.
In P rats administered EtOH immediately prior to PSR testing there was a significant ‘Dose’ × ‘Session’ interaction (F8, 108 = 3.26; p = 0.002).There were significant effects of ‘Dose’ during the 1st and 2nd seeking session (F2,270 values > 7.6; p values < 0.002). P rats administered 0.5 or 1.0 g/kg EtOH immediately prior to PSR testing displayed an increase in responding on the lever previously associated with the delivery of EtOH during Seek1, while rats administered 1.0 g/kg EtOH displayed an increase in responding during Seek2, compared to saline control groups (top right panel, Figure 3b).
In P rats administered EtOH 30 min prior to RoR testing, there was a significant ‘Dose’ × ‘Session’ interaction (F8, 104 = 2.32; p = 0.024). There were significant effects of ‘Dose’ during the 1st and 2nd seeking session (F2,26 values > 5.33; p values < 0.011). P rats administered 1.0 g/kg EtOH 30 minutes prior to seeking testing responded less on the lever previously associated with the delivery of EtOH compared to saline and 0.5 g/kg EtOH groups during sessions 1 and 2 (bottom left panel, Figure 3c).
In P rats administered EtOH 30 min prior to PSR testing, there was a significant ‘Dose’ × ‘Session’ interaction (F8, 100 = 4.93; p < 0.001). There were significant effects of ‘Dose’ during the 1st and 2nd seeking session (F2,25 values > 8.63; p values < 0.001). P rats administered 0.5 or 1.0 g/kg EtOH 30 minutes prior to the 1st PSR testing responded more on the lever previously associated with the delivery of EtOH compared to saline and 1.0 g/kg group responded more than the 0.5 g/kg group (bottom right panel, Figure 3d) during sessions 1 and 2.
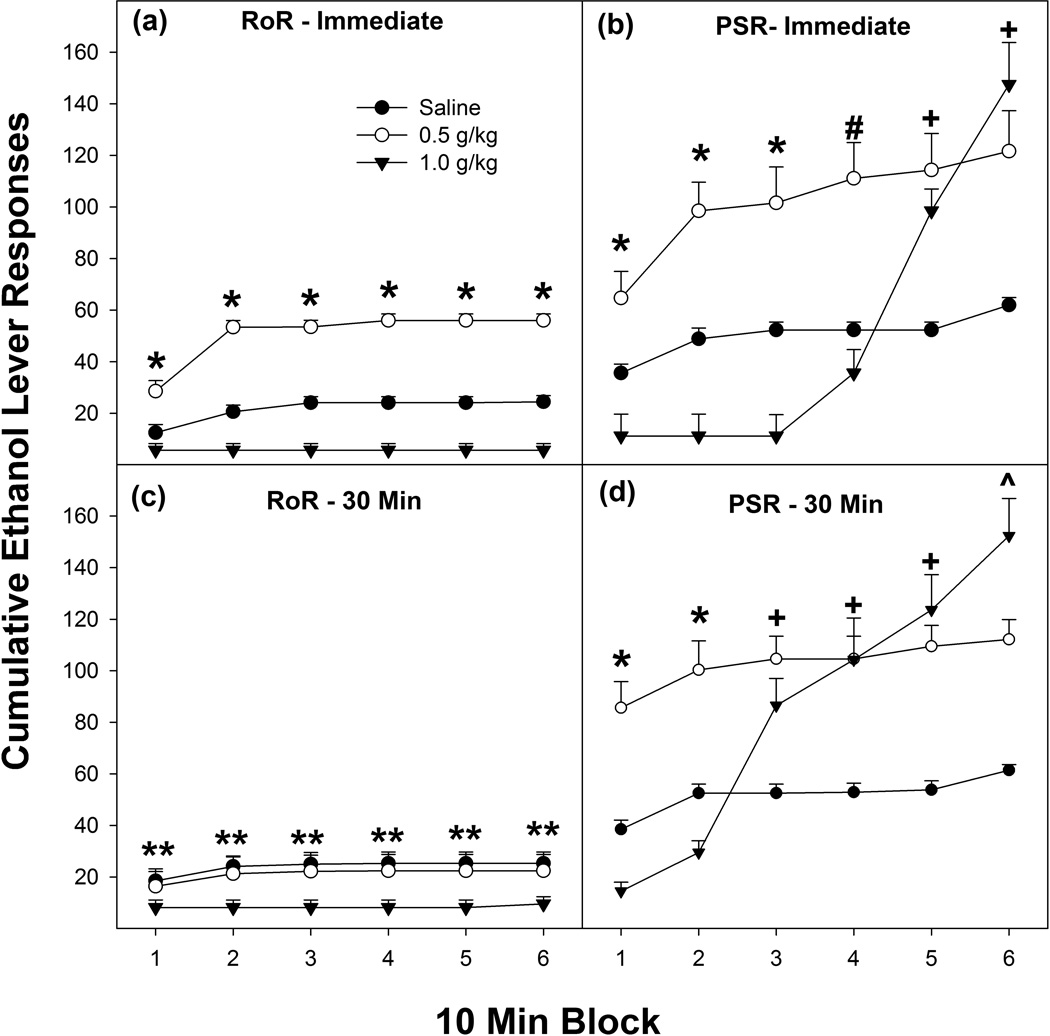
The temporal responding during the 1st EtOH-seeking session (Seek1) was examined in all groups (Fig. 3). The overall analysis revealed a significant ‘Model’ (PSR vs RoR) × ‘Dose’ X ‘Time of Injection’ × ‘Time Block’ interaction (F10,525 = 3.39; p < 0.001). Administration of 0.5 g/kg EtOH immediately prior to RoR or PSR testing resulted in significantly more EtOH lever responding during the first 20 min of testing (98.4% and 83.4% of total responding respectively; top panel Figures 4a and 4b). The major EtOH lever response difference was between the 1.0 g/kg EtOH immediate RoR and PSR groups. Responding on the lever previously associated with the delivery of EtOH in rats administered 1.0 g/kg EtOH immediately prior to RoR testing was suppressed for the whole 1 hr test session (Fig. 4a). In contrast, administration of 1.0 g/kg EtOH immediately prior to PSR testing resulted in suppression of responding for the initial 40 min, but, during the last 20 min period, EtOH lever responding was significantly increased (Fig. 4b). Statistical analysis between these two groups revealed only significant differences in the 5th and 6th time period (p < 0.0001).
Figure 4.
Depicts the mean (± SEM) cumulative responses on the lever previously associated with the delivery of EtOH during RoR (Figs 4a & 4c) or PSR (4c & 4d) testing as a function of a priming injection of EtOH during the 1st EtOH-seeking test session separated into 10-min blocks. * indicates all groups are significantly different from each other (0.5 g/kg > saline > 1.0 g/kg). # indicates that the 0.5 g/kg group is significantly higher than saline and 1.0 g/kg EtOH group. + indicates that the 0.5 and 1.0 g/kg EtOH groups are significantly higher than saline controls. ^ indicates that all groups are significantly different from each other (1.0 g/kg > 0.5 g/kg > saline). ** indicates that responding in the 1.0 g/kg group is significantly lower than the saline and 0.5 g/kg groups.
EtOH administered 30 min prior altered only the PSR responding on the lever previously associated with the delivery of EtOH when examined in 10 min blocks of activity (F10, 125 = 6.58; p < 0.0001; Fig. 4d). Rats administered 0.5 g/kg EtOH 30 min prior to PSR testing displayed a significantly higher level of EtOH-seeking during the 1st 10 min period compared to saline treated rats. Rats administered 1.0 g/kg EtOH 30 min prior to PSR testing displayed a reduction in EtOH-seeking behaviors for the initial 20 min of testing, but during the 3rd 10 min block the amount of EtOH-seeking expressed was greater than all other groups. Post-hoc comparisons indicated that all groups were different (0.5 g/kg > saline > 1.0 g/kg) during the initial two 10 min blocks, and, during the 3rd – 5th 10-min block periods, EtOH treated rats were significantly higher than saline treated rats, and, during the 6th 10-min block period, all groups were significantly different from each other (1.0 g/kg > 0.5 g/kg > saline).
Experiment 3 – Effects of Repeated Cycles of Deprivation and Access on Relapse Responding and EtOH-Seeking
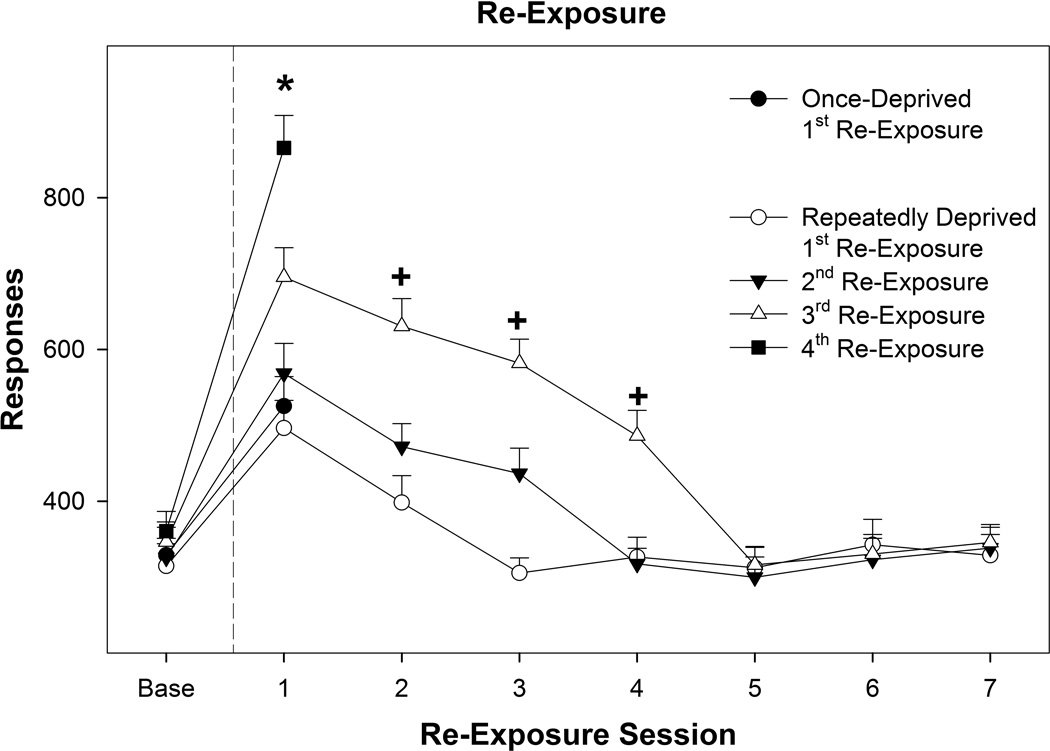
In rats exposed to repeated cycles of EtOH access-deprivation (Figure 5), the amount of EtOH self-administration for the 3 sessions prior to deprivation (Base) did not differ across cycles (F3,9 = 1.24; p = 0.35). Examining the effects of repeated cycles of EtOH access-deprivations (all 4 cycles) for alcohol consumption during the 1st re-exposure session indicated a significant effect of ‘cycle’ (F3,9 = 33.5; p < 0.0001). Responding following the 3rd deprivation cycle was increased compared to the 1st and 2nd cycle, and self-administration following the 4th deprivation period was higher than all other deprivation cycle. T-test comparisons revealed that there was a significant ADE following each deprivation cycle (p values < 0.0001). An additional analysis examining the amount of EtOH self-administered across the re-exposure sessions following the 1st – 3rd access-deprivation periods revealed a significant ‘cycle’ by ‘session’ interaction (F10, 110 = 10.06; p < 0.0001). The estimated baseline EtOH intake for the groups was between 2.1–2.2 g/kg. The estimated intake for 1st session of re-exposure for the once-deprived group was 3.24 g/kg and for repeated deprivation the estimate intake for 1st session was 3.36 g/kg (1st re-exposure), 4.43 g/kg (2nd re-exposure), 4.69 g/kg (3rd re-exposure), and 5.3 g/kg (4th re-exposure).
Figure 5.
Depicts the mean (± SEM) EtOH responses during re-exposure as a function of deprivation cycle. * indicates that all groups are significantly greater than baseline and that responding following the 4th deprivation period was greater than all other groups, and responding following the 3rd deprivation period was greater than following the 1st and 2nd deprivation period. + indicates that EtOH responding following the 3rd deprivation period was elevated compared to baseline and significantly different from that observed following the 1st and 2nd deprivation period.
Blood EtOH concentrations (BECs) or amount of fluid consumed were measured in repeatedly deprived rats only at one time point (following the initial re-instatement session after the 3rd deprivation period). BECs were determined to be 209 ± 12 mg%. In addition, utilizing the same protocol to estimate EtOH fluid intake reported previously in our lab (Rodd et al., 2003) the amount of fluid consumed versus the amount of predicted consumption during this session was 93%. This is slightly lower than previously reported (97%; Rodd et al., 2003). Yet, this slight reduction may be the result of multiple causes; a) reduction in accuracy of obtaining the EtOH solution by highly intoxicated rats and/or b) rats adding saliva to the EtOH trough Furthermore, to eliminate a possible reduction in %fluid consumed between the FR5 and FR10 non-deprived groups, fluid intake was assessed in these rats during week 14. Similar to the past report (Rodd et al., 2003), the amount of predicted EtOH consumed was 95 and 97% for the FR5 and FR10 groups, respectively.
EtOH self-administration between each ‘cycle’ during individual sessions revealed that self-administration following the 3rd deprivation cycle was greater during re-exposure sessions 1–4 compared to that observed following the 1st and 2nd deprivation period. Additionally, there was a significant effect of ‘session’ following each deprivation period (p values < 0.001). Paired t-tests revealed that following the 1st and 2nd deprivation period, EtOH self-administration was increased during re-exposure session 1. Following the 3rd deprivation period, EtOH self-administration was increased during re-exposure sessions 1–4. In rats given a single deprivation period, there was a significant increase in EtOH self-administration during the single re-exposure period (p < 0.001).
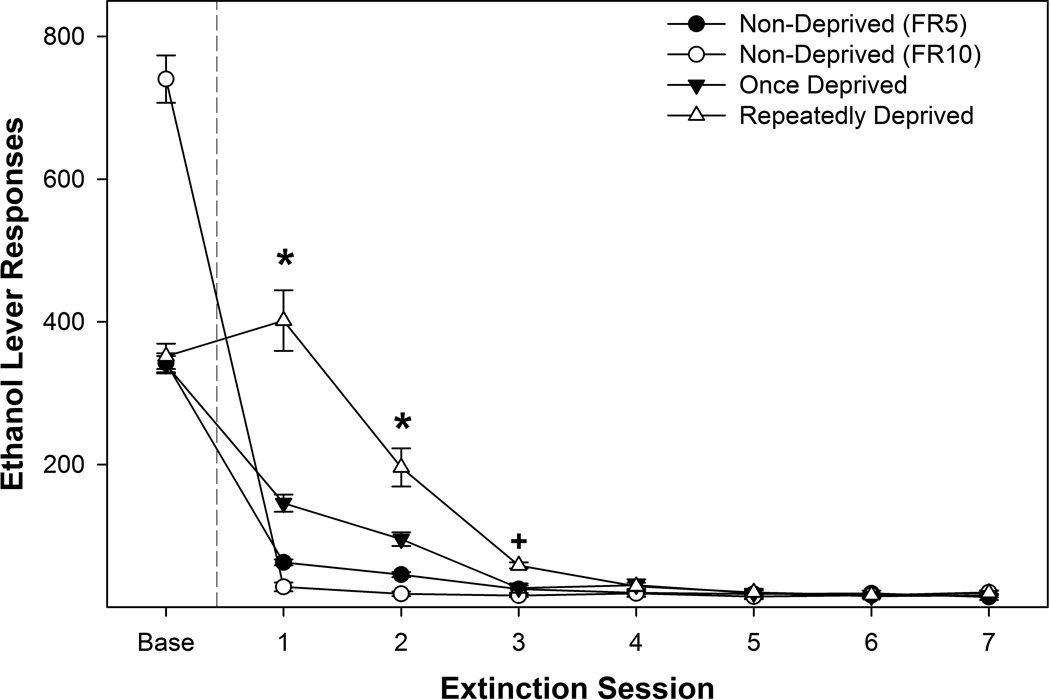
Extinction training responding was contrasted in all 4 groups (Figure 6). Responding during baseline (average of the last 3 sessions prior to extinction) was significantly different between the groups (F3, 42 = 18.2; p < 0.001). Logically, rats self-administering EtOH on the FR10 schedule of reinforcement responded more on the EtOH lever than all other groups, but received an identical amount of reinforcers. Overall, there was a significant ‘group’ by ‘session’ interaction for responding on the lever previously associated with the delivery of EtOH during extinction training (F21, 294 = 3.12; p < 0.001). Examining the extinction responding during each extinction session revealed significant group differences during extinction sessions 1–3 (F3, 42 values > 13.8; p values < 0.001). During the 1st and 2nd extinction session, all groups were significantly different from each other. During the 3rd extinction session, the repeatedly deprived group was significantly higher than all other groups.
Figure 6.
Depicts the mean (± SEM) responses on the lever previously associated with the delivery of EtOH during extinction training as a function of past drinking history. * indicates that all groups are statistically different from each other. + indicates that rats repeatedly deprived responded more than all other groups.
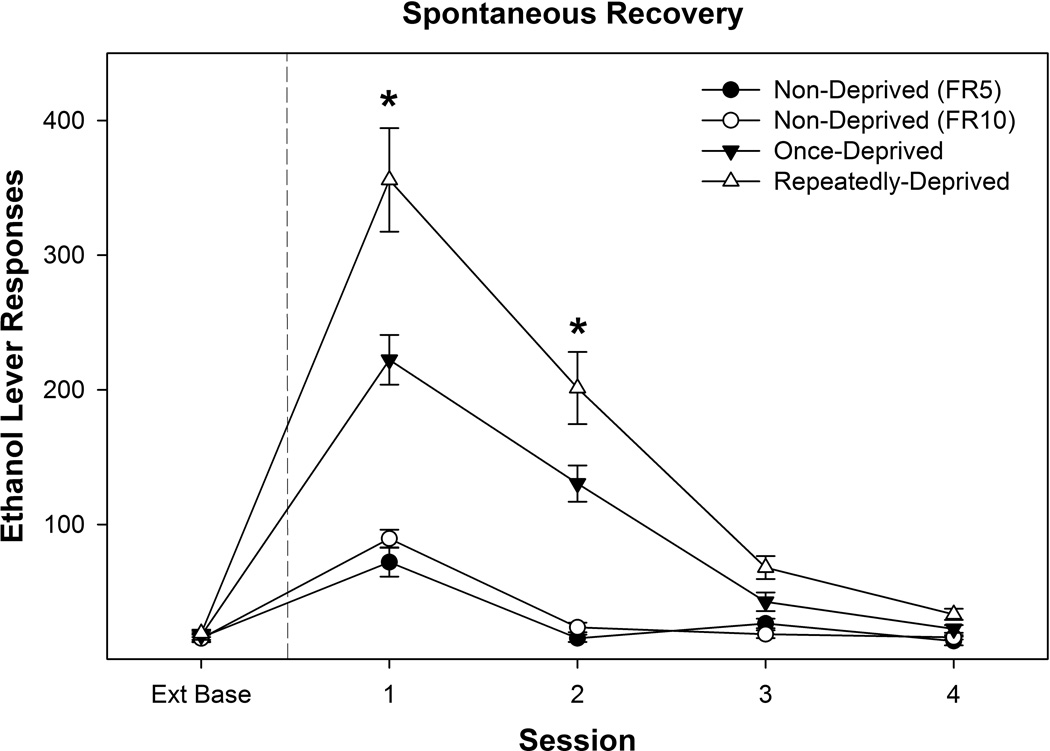
Examining the effects of past drinking history on the expression of context-induced EtOH-seeking (PSR; Figure 7) revealed a significant ‘group’ by ‘session’ interaction (F12, 168 = 4.7; p < 0.001). Examining the responses on the lever previously associated with delivery of EtOH during the 1st PSR test session indicated a significant effect of ‘group’ (F3, 42 = 23.4; p < 0.001). Rats exposed to repeated cycles of EtOH access-deprivation responded more on the lever previously associated with the delivery of EtOH than all other groups and the level of responding was greater in the once deprived group then observed in rats that were non-deprived . Similar differences between groups were observed during the 2nd PSR test session. Responding during the 1st PSR test session was elevated in the FR5 and FR10 non-deprived groups compared to baseline (p values < 0.01), but there was no difference between these two groups. In rats repeated deprived or once deprived, responding on the lever previously associated with the delivery of EtOH was increased compared to extinction baseline for the 1st and 2nd PSR test session. There were no significant differences on water lever responses between groups for any of the experiments.
Figure 7.
Depicts the mean (± SEM) responses on the lever previously associated with the delivery of EtOH during PSR testing as a function of past drinking history. * indicates that all groups are significantly different from extinction baseline (Ext Base), responding in repeatedly deprived rats is greater than all other groups, and rats exposed to a single deprivation period is greater than both non-deprived groups.
DISCUSSION
The major findings of this study are that (1) robust context-induced EtOH-seeking was observed following 8 weeks of EtOH abstinence (Fig. 2); (2) priming doses of EtOH were more effective in enhancing expression of EtOH seeking when given 14 days (PSR procedure) than 1 day (RoR procedure) after the last extinction session. (Fig. 3), (3) repeated cycles of deprivation progressively enhanced relapse self-administration (Fig. 5) and expression of context-induced EtOH-seeking (Fig. 7), and increased resistance to extinction (Fig. 6).
EtOH-seeking was observed in a time-dependent manner (Fig. 1). The observation that expression of EtOH-seeking was not observed after one week of home-cage rest but was observed after 2 weeks suggests that neuronal alterations were taking places during this period which reduced the effects of the extinction training and/or enhanced the effects of EtOH self-administration conditioning. The fact that P rats, maintained in the home cage for 4 and 6 weeks, continued to respond in the 2nd PSR session, whereas rats in the home cage for 2 weeks did not, suggests that additional neuronal alterations may have taken place during the longer ‘rest’ periods to strengthen the association of the operant chamber context with EtOH self-administration. Maximal expression of EtOH-seeking was observed following 6 weeks of abstinence (Fig. 2).Although robust EtOH-seeking was observed after 8 weeks of abstinence, the effect did not persist into the 2nd PSR session, suggesting some reductions in the neuronal alterations associated with EtOH self-administration had occurred.
Overall, the results of this study suggest that the motivation to obtain EtOH was maintained for at least 8 weeks in the absence of access to EtOH or further experience in the operant chambers. The retention of the rewarding actions of EtOH after 7 weeks of abstinence has been demonstrated to be present in the posterior VTA (Rodd et al., 2005). However, in this study, extinction training was not done and repeated deprivation cycles were used. These findings with PSR model are in agreement with the learning phenomenon termed the incubation model that demonstrated operant drug-seeking responding following prolong periods of abstinence can be time-dependent, long-lasting, but not permanent, resulting in an inverted U-shape curve (Grimm et al., 2001;Lu et al., 2004; Lu et al., 2009). Bienkowski et al. (2004) using the incubation model showed that EtOH-seeking induced by context and discrete EtOH-associated cues reached maximal levels after 4 and/or 8 weeks in home cage/abstinence phase compared to 24 hours in non-selective rats. In addition, a recent clinical study provided evidence for incubation of alcohol craving in humans (Li et al., 2015). Li et al. (2015) examined cue-induced alcohol craving after 4 time-points (7, 14, 30 and 60 days of abstinence) and found that cue-induced alcohol craving measures were the highest at 60 days of abstinence. Taken together with the current study, these finding are in agreement with Gawin and Kleber (1986) hypothesis that suggests time points beyond the acute withdrawal phase may increase the vulnerability to cue- or context- induced craving behaviors.
Similar to the current findings, EtOH-seeking was also observed 14 and 21 days after extinction training in the renewal model of context-induced drug-seeking (Janak and Chaudhri, 2010). In the present study, extinction training was conducted in the same context as EtOH self-administration, suggesting that the environmental context plays an important role in determining the impact of extinction training on EtOH-seeking. The ability of a drug-paired context to elicit seeking has been reported in the clinical literature in that real-world drug-associated environments can induce robust, persistent craving longer than the more general drug-specific cues (Fatseas et al., 2015). In addition, real-world drug-associated environments were associated with higher craving levels under conditions of abstinence as well as paralleling with increases of brain signals in areas of the brain involved in drug-environment associations (McClernon et al., 2016).
Examination of the RoR model in the current study showed that unless the P rats are given a moderate priming dose of EtOH immediately prior to the session, they do not demonstrate EtOH-seeking (Fig. 3a). These results are similar to findings reported by others using non-selected rats (Le et al., 1998; 1999). In contrast to the results with the RoR model, context-induced EtOH-seeking was observed in the PSR model with saline and with both doses of EtOH (Fig. 3b and 3d). These results are consistent with neuronal alterations occurring during the 2-week home cage rest period that strengthened susceptibility to enhanced EtOH-seeking.
In the RoR model, the 1.0 g/kg dose of EtOH reduced responding throughout the 60 min session (Fig. 4a and 4c), suggesting that this dose may be producing sedative or motor impairing effects. In the PSR model, the 1.0 g/kg EtOH dose suppressed responding for the first 30 min (Fig. 4c), but responding during the last 30 min recovered to the high level of responding observed for the P rats receiving 0.5 g/kg EtOH, suggesting the brain EtOH levels decreased below sedation and into the stimulating levels. This idea is supported by the findings with the P rats given the 1.0 g/kg dose of EtOH 30 min before the test session. In this case, reduced responding was observed within the first 20 min; however, by 30 min, responding had recovered to the high levels observed for the 0.5 g/kg group (Fig. 4d), and exceeded the responding of the 0.5 g/kg group by the end of the session. The mechanisms underlying the stimulating effects of EtOH on EtOH-seeking in the PSR model have not been established. However, local microinjection of EtOH into the posterior VTA of P rats significantly enhanced expression of EtOH-seeking in the PSR model (Hauser et al., 2011), supporting the idea that the stimulating effects of EtOH on VTA dopamine neurons may be contributing to the EtOH-enhanced expression of EtOH-seeking.
Repeated cycles of deprivation produced profound effects on EtOH self-administration (Fig. 5), resistance to extinction training (Fig. 6) and expression of EtOH-seeking (Fig. 7). These results indicate that repeated deprivation cycles produced robust increases in EtOH self-administration that persisted across 4 sessions. These results are similar to results previously reported (Rodd et al., 2003), in which estimated intakes of approximately 4 g/kg body wt were attained in the 1-hr session on the first re-exposure day after repeated deprivation cycles; this level of EtOH intake would produce BACs in excess of 120 mg% (McBride et al., 2013). These results suggest that, with repeated deprivation cycles and progressive increases in the number of EtOH reinforcements, the rewarding actions of EtOH may have been enhanced and tolerance to the high dose non-rewarding (aversive, sedative) actions of EtOH may have developed. In support of this idea, an intra-cranial self-administration study reported repeated cycles of alcohol deprivation and access produced a prolonged increase in the sensitivity and response of the posterior VTA of P rats to the rewarding actions of EtOH, such that low and high concentrations of EtOH, which were not self-infused into the posterior VTA of the water control group, were now self-infused into the posterior VTA following repeated cycles of deprivation, even after 7 weeks of abstinence following the last EtOH drinking episode (Rodd et al., 2005). Both the non-deprived FR5 and FR10 groups essentially extinguished responding in the 1st extinction session (Fig. 6). This rapid extinction after approximately 10 weeks of EtOH self-administration was somewhat surprising. The P rats may have quickly learned that EtOH was not available and did not respond on either lever.
In contrast to the non-deprived groups, the repeatedly deprived group exhibited resistance to extinction (Fig. 6). With repeated cycles of deprivation and progressively higher EtOH intakes, associative learning between environmental context cues and EtOH reinforcement may have been enhanced (Katner et al., 1999; Macintosh 1977), the reward saliency of EtOH may have been enhanced (Macintosh 1977), and/or tolerance to the emotional aspects of the extinction paradigm may have developed (Azrin et al., 1966). The finding that responding in the PSR test was significantly higher in the repeatedly deprived group compared to the other groups suggests that neuronal alterations associated with EtOH-seeking behavior was strengthened with repeated cycles of deprivation. Furthermore, these results suggest that the reward saliency of EtOH increased with cycles of deprivation and the progressively higher EtOH intakes during relapse.
Overall, the results suggest a single period of deprivation produces neuronal alterations that make it difficult to extinguish drinking behavior and also enhance expression of EtOH-seeking. Furthermore, repeated cycles of deprivation and elevated relapse drinking will produce neuronal alterations that will make it progressively more difficult to resist relapse drinking. These findings are in agreement with human clinical findings (Malcolm et al., 2000), and patterns of binge drinking observed in human alcoholics (Finney and Moos 1991; Nezlek et al., 1994).
It is important to note the relevance of the FR10 control group in these experiments. The above listed effects of repeated cycles of alcohol access-deprivations was not based upon the fact that these animals had periods of heightened level of responding compared to the non-deprived and single deprived groups. The FR10 control group responded on the EtOH lever approximately 800/session, which is equivalent to the amount of Repeatedly-Deprived group during the 4th re-exposure period (Figs 5 and 6). Unlike the single session of responding at this level for the Repeatedly-Deprived group, the FR10 group responded at this level for EtOH (but for approximately 50% of the reinforcers) for 12 consecutive weeks. In addition, the FR10 control group extinguished EtOH responding at a higher rate than all other groups. This finding parallels the learning field concept of negative contrast of extinction training (Macintosh, 1977). In general, negative contrast is a well replicated learning phenomenon in which animals with the highest work requirement for a reinforcer extinguish responding at a greater rate compared to animals that had self-administer a reinforcer at a lower work requirement. Further, the finding that the FR5 and FR10 control groups responded equivalently during the PSR testing validates this model in that seeking is not predicated upon past work requirement history but the saliency of the reinforcer.
In summary, the results of the current study indicate that context-induced EtOH-seeking can be exhibited by P rats even after a prolonged (2-month) period of EtOH abstinence, suggesting retention of the reward saliency of EtOH. In addition, the reward saliency of EtOH is markedly increased with repeated cycles of deprivation and EtOH drinking, as indicated by increased resistance to extinction and enhanced expression of EtOH-seeking with repeated deprivation cycles.
Acknowledgments
This study was supported in part by NIH grants AA020908, AA022287, AA007611 and AA007462. The content of the manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Alcohol Abuse and Alcoholism or NIH.
Footnotes
None of the authors has a conflict of interest associated with this research.
REFERENCES
- Azrin NH, Hutchinson RR, Hake DF. Extinction-induced aggression. J Exp Anal Behav. 1966;9:191–204. doi: 10.1901/jeab.1966.9-191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bienkowski P, Rogowski A, Korkosz A, Mierzejewski P, Radwanska K, Kaczmarek L, Bogucka-Bonikowska A, Kostowski W. Time-dependent changes in alcohol-seeking behaviour during abstinence. Eur Neuropsychopharmacol. 2004;14:355–360. doi: 10.1016/j.euroneuro.2003.10.005. [DOI] [PubMed] [Google Scholar]
- Bouton ME. Context, ambiguity, and unlearning: sources of relapse after behavioral extinction. Biol Psychiatry. 2002;52:976–986. doi: 10.1016/s0006-3223(02)01546-9. [DOI] [PubMed] [Google Scholar]
- Bouton ME. Context and behavioral processes in extinction. Learn Mem. 2004;11:485–494. doi: 10.1101/lm.78804. [DOI] [PubMed] [Google Scholar]
- Brooks DC. Recent and remote extinction cues reduce spontaneous recovery. Q J Exp Psychol B. 2000;53:25–58. doi: 10.1080/027249900392986. [DOI] [PubMed] [Google Scholar]
- Burish TG, Maisto SA, Cooper AM, Sobell MB. Effects of voluntary short-term abstinence from alcohol on subsequent drinking patterns of college students. J Stud Alcohol. 1981;42:1013–1020. doi: 10.15288/jsa.1981.42.1013. [DOI] [PubMed] [Google Scholar]
- Chaudhri N, Sahuque LL, Janak PH. Context-induced relapse of conditioned behavioral responding to ethanol cues in rats. Biol Psychiatry. 2008a;64:203–210. doi: 10.1016/j.biopsych.2008.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudhri N, Sahuque LL, Cone JJ, Janak PH. Reinstated ethanol-seeking in rats is modulated by environmental contex and requires the nucleus accumbens core. Eup J Neurosci. 2008b;28:2288–2298. doi: 10.1111/j.1460-9568.2008.06517.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudhri N, Sahuque LL, Janak PH. Ethanol seeking triggered by environmental context is attenuated by blocking dopamine D1 receptors in the nucleus accumbens core and shell in rats. Psychopharmacology. 2009;207:303–314. doi: 10.1007/s00213-009-1657-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conklin CA, Robin N, Perkins KA, Salkeld RP, McClernon FJ. Proximal versus distal cues to smoke: the effects of environments on smokers’ cue-reactivity. Exp Clin Psychopharmacol. 2008;16:207–214. doi: 10.1037/1064-1297.16.3.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conklin CA, Perkins KA, Robin N, McClernon FJ, Salkeld RP. Bringing the real world into the laboratory: personal smoking and nonsmoking environments. Drug Alcohol Depend. 2009;111:58–63. doi: 10.1016/j.drugalcdep.2010.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhaher R, Hauser SR, Getachew B, Bell RL, McBride WJ, McKinzie DL, Rodd ZA. The Orexin-1 Receptor Antagonist SB-334867 Reduces Alcohol Relapse Drinking, but not Alcohol Seeking, in Alcohol-Preferring (P) Rats. J Addict Med. 2010;4:153–159. doi: 10.1097/ADM.0b013e3181bd893f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wit H. Priming effects with drugs and other reinforcers. Exp Clin Psychopharmacol. 1996;4:5–10. [Google Scholar]
- de Wit H, Stewart J. Reinstatement of cocaine-reinforced responding in the rat. Psychopharmacology (Berl) 1981;75:134–143. doi: 10.1007/BF00432175. [DOI] [PubMed] [Google Scholar]
- Fatseas M, Serre F, Alexandre JM, Debrabant R, Auriacombe M, Swendsen J. Craving and substance use among patients with alcohol, tobacco, cannabis or heroin addiction: a comparison of substance- and person-specific cues. Addiction. 2015;110:1035–1042. doi: 10.1111/add.12882. [DOI] [PubMed] [Google Scholar]
- Ferri M, Amato L, Davoli M. Alcoholics Anonymous and other 12-step programmes for alcohol dependence. Cochrane Database Syst Rev. 2006;3:CD005032. doi: 10.1002/14651858.CD005032.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Field M, Schoenmakers T, Wiers RW. Cognitive processes in alcohol binges: a review and research agenda. Curr Drug Abuse Rev. 2008;1:263–279. doi: 10.2174/1874473710801030263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finney JW, Moos RH. The long-term course of treated alcoholism: I. Mortality, relapse and remission rates and comparisons with community controls. J Stud Alcohol. 1991;52:44–54. doi: 10.15288/jsa.1991.52.44. [DOI] [PubMed] [Google Scholar]
- Gauggel S, Heusinger A, Forkmann T, Boecker M, Lindenmeyer J, Cox WM, Staedtgen M. Effects of alcohol cue exposure on response inhibition in detoxified alcohol-dependent patients. Alcohol Clin Exp Res. 2010;34:1584–1589. doi: 10.1111/j.1530-0277.2010.01243.x. [DOI] [PubMed] [Google Scholar]
- Gawin FH, Kleber HD. Abstinence symptomatology and psychiatric diagnosis in cocaine abusers. Clinical observations. Arch Gen Psychiatry. 1986;43:107–113. doi: 10.1001/archpsyc.1986.01800020013003. [DOI] [PubMed] [Google Scholar]
- Grimm JW, Hope BT, Wise RA, Shaham Y. Neuroadaptation. Incubation of cocaine craving after withdrawal. Nature. 2001;412:141–142. doi: 10.1038/35084134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauser SR, Ding ZM, Getachew B, Toalston JE, Oster SM, McBride WJ, Rodd ZA. The posterior ventral tegmental area mediates alcohol-seeking behavior in alcohol-preferring rats. J Pharmacol Exp Ther. 2011;336:857–865. doi: 10.1124/jpet.110.168260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilbrom ME. Alcohol withdrawal seizures and binge versus chronic drinking. In: Port RJ, Mattson RH, Cramer JA, Diamond I, editors. Alcohol and Seizures: Basic Mechanisms and Clinical Concepts. Philadelphia: FA Davis; 1990. pp. 206–215. [Google Scholar]
- Honig WK, Staddon JER. Handbook of operant behavior. Englewood Cliffs, N.J.: Prentice-Hall; 1977. [Google Scholar]
- Janak PH, Chaudhri N. The potent effect of environmental contex on relapse to alcohol-seeking after extinction. Open Addict J. 2010;3:76–87. doi: 10.2174/1874941001003010076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katner SN, Magalong JG, Weiss F. Reinstatement of alcohol-seeking behavior by drug-associated discriminative stimuli after prolonged extinction in the rat. Neuropsychopharmacology. 1999;20:471–479. doi: 10.1016/S0893-133X(98)00084-0. [DOI] [PubMed] [Google Scholar]
- Le AD, Quan B, Juzytch W, Fletcher PJ, Joharchi N, Shaham Y. Reinstatement of alcohol-seeking by priming injections of alcohol and exposure to stress in rats. Psychopharmacology. 1998;135:169–174. doi: 10.1007/s002130050498. [DOI] [PubMed] [Google Scholar]
- Le AD, Poulos CX, Harding S, Watchus J, Juzytch W, Shaham Y. Effects of naltrexone and fluoxetine on alcohol self-administration and reinstatement of alcohol-seeking induced by priming injections of alcohol and exposure to stress. Neuropsychopharmacology. 1999;21:435–444. doi: 10.1016/S0893-133X(99)00024-X. [DOI] [PubMed] [Google Scholar]
- Li P, Wu P, Xin X, Fan YL, Wang GB, Wang F, Ma MY, Xue MM, Luo YX, Yang FD, Bao YP, Shi J, Sun HQ, Lu L. Incubation of alcohol craving during abstinence in patients with alcohol dependence. Addict Biol. 2015;20:513–522. doi: 10.1111/adb.12140. [DOI] [PubMed] [Google Scholar]
- Lu L, Grimm JW, Hope BT, Shaham Y. Incubation of cocaine craving after withdrawal: a review of preclinical data. Neuropharmacology. 2004;47(Suppl 1):214–226. doi: 10.1016/j.neuropharm.2004.06.027. [DOI] [PubMed] [Google Scholar]
- Lu L, Wang X, Wu P, Xu C, Zhao M, Morales M, Harvey BK, Hoffer BJ, Shaham Y. Role of ventral tegmental area glial cell line-derived neurotrophic factor in incubation of cocaine craving. Biol Psychiatry. 2009;66:137–145. doi: 10.1016/j.biopsych.2009.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macintosh JJ. Stimulus control: Attentional factors. In: Honig WK, Staddon JER, editors. Handbook on Operant Behavior. Englewood Cliffs, NJ: Prentice-Hall; 1977. pp. 162–241. [Google Scholar]
- Malcolm R, Roberts JS, Wang W, Myrick H, Anton RF. Multiple previous detoxifications are associated with less responsive treatment and heavier drinking during an index outpatient detoxification. Alcohol. 2000;22:159–164. doi: 10.1016/s0741-8329(00)00114-2. [DOI] [PubMed] [Google Scholar]
- McBride WJ, Kimpel MW, McClintick JN, Ding Z-M, Hauser SR, Edenberg HJ, Bell RL, Rodd ZA. Changes in gene expression within the ventral tegmental area following repeated excessive binge-like alcohol drinking by alcohol-preferring (P) rats. Alcohol. 2013;47:367–380. doi: 10.1016/j.alcohol.2013.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBride WJ, Rodd ZA, Bell RL, Lumeng L, Li T-K. The alcohol-preferring (P) and high-alcohol-drinking (HAD) rats - animal models of alcoholism. Alcohol. 2014;48:209–215. doi: 10.1016/j.alcohol.2013.09.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClernon FJ, Conklin CA, Kozink RV, Adcock RA, Sweitzer MM, Addicott MA, Chou YH, Chen NK, Hallyburton MB, DeVito AM. Hippocampal and Insular Response to Smoking-Related Environments: Neuroimaging Evidence for Drug-Context Effects in Nicotine Dependence. Neuropsychopharmacology. 2016;41:877–885. doi: 10.1038/npp.2015.214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMillen BA. Toward a definition of a valid animal model of alcoholism: Multiple animal models for multiple diseases. Alcohol. 1997;14:409–419. doi: 10.1016/s0741-8329(97)90012-4. [DOI] [PubMed] [Google Scholar]
- Nezlek JB, Pilkington CJ, Bilbro KG. Moderation in excess: binge drinking and social interaction among college students. J Stud Alcohol. 1994;55:342–351. doi: 10.15288/jsa.1994.55.342. [DOI] [PubMed] [Google Scholar]
- O’Brien CP, Childress AR, McLellan T, Ehrman R. Integrating systemic cue exposure with standard treatment in recovering drug dependent patients. Addict Behav. 1998;15:355–365. doi: 10.1016/0306-4603(90)90045-y. [DOI] [PubMed] [Google Scholar]
- Research Institute for Laboratory Animal Research. Guide for the Care and Use of Laboratory Animals. 8th. Washington, D.C.: The National Academies Press; 2011. [PubMed] [Google Scholar]
- Rescorla RA. Retraining of extinguished Pavlovian stimuli. J Exp Psychol Anim Behav Process. 2001;27:115–124. [PubMed] [Google Scholar]
- Rodd ZA, Bell RL, Kuc KA, Murphy JM, Lumeng L, Li T-K, McBride WJ. Effects of repeated alcohol deprivations on operant ethanol self-administration by alcohol-preferring (P) rats. Neuropsychopharmacology. 2003;28:1614–1621. doi: 10.1038/sj.npp.1300214. [DOI] [PubMed] [Google Scholar]
- Rodd ZA, Bell RL, McKinzie DL, Webster AA, Murphy JM, Lumeng L, Li TK, McBride WJ. Low-dose stimulatory effects of ethanol during adolescence in rat lines selectively bred for high alcohol intake. Alcohol Clin Exp Res. 2004;28:535–543. doi: 10.1097/01.alc.0000122107.08417.d0. [DOI] [PubMed] [Google Scholar]
- Rodd ZA, Bell RL, McQueen VK, Davids MR, Hsu CC, Murphy JM, Li T-K, Lumeng L, McBride WJ. Prolonged increase in the sensitivity of the posterior ventral tegmental area to the reinforcing effects of ethanol following repeated exposure to cycles of ethanol access and deprivation. J Pharmacol Exp Ther. 2005;315:648–657. doi: 10.1124/jpet.105.084350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodd ZA, McKinzie DL, Bell RL, McQueen VK, Murphy JM, Schoepp DD, McBride WJ. The metabotropic glutamate 2/3 receptor agonist LY404039 reduces alcohol-seeking but not alcohol self-administration in alcohol-preferring (P) rats. Behav Brain Res. 2006;171:207–215. doi: 10.1016/j.bbr.2006.03.032. [DOI] [PubMed] [Google Scholar]
- Rodd-Henricks ZA, Bell RL, Kuc KA, Murphy JM, McBride WJ, Lumeng L, Li T-K. Effects of ethanol exposure on subsequent acquisition and extinction of ethanol self-administration and expression of alcohol-seeking behavior in adult alcohol-preferring (P) rats: I. Periadolescent exposure. Alcohol Clin Exp Res. 2002a;26:1632–1641. doi: 10.1097/01.ALC.0000036301.36192.BC. [DOI] [PubMed] [Google Scholar]
- Rodd-Henricks ZA, Bell RL, Kuc KA, Murphy JM, McBride WJ, Lumeng L, Li T-K. Effects of ethanol exposure on subsequent acquisition and extinction of ethanol self-administration and expression of alcohol-seeking behavior in adult alcohol-preferring (P) rats: II. Adult exposure. Alcohol Clin Exp Res. 2002b;26:1642–1652. doi: 10.1097/01.ALC.0000036302.73712.9D. [DOI] [PubMed] [Google Scholar]
- Shaham Y, Adamson LK, Grocki S, Corrigall WA. Reinstatement and spontaneous recovery of nicotine seeking in rats. Psychopharmacology (Berl) 1997;130:396–403. doi: 10.1007/s002130050256. [DOI] [PubMed] [Google Scholar]
- Stewart J. Conditioned and unconditioned drug effects in relapse to opiate and stimulant drug self-administration. Prog Neuropsychopharmacol Biol Psychiatry. 1983;7:591–597. doi: 10.1016/0278-5846(83)90030-1. [DOI] [PubMed] [Google Scholar]
- Waller MB, Murphy JM, McBride WJ, Lumeng L, Li TK. Effect of low dose ethanol on spontaneous motor activity in alcohol-preferring and non-preferring lines of rats. Pharmacol Biochem Behav. 1986;24:617–623. doi: 10.1016/0091-3057(86)90567-8. [DOI] [PubMed] [Google Scholar]