Abstract
Our study aimed to develop embryo models to evaluate the impact of oxidative stress on embryo development. Mouse zygotes, which stayed at G1 phase, were treated with prepared culture medium (containing 0.00, 0.01, 0.02, 0.03, 0.04, 0.05, or 0.1 mM hydrogen peroxide (H2O2)) for 30 min in experiment 1. The dose-effects of H2O2 on embryo development were investigated via comparisons of the formation rate at each stage (2- and 4-cell embryos and blastocysts). Experiment 2 was carried out to compare behaviors of embryos in a mild oxidative-stressed status (0.03 mM H2O2) with those in a control (0 mM H2O2). Reactive oxygen species (ROS) levels, variation of mitochondrial membrane potential (MMP), expression of γH2AX, and cell apoptosis rate of blastocyst were detected. We observed a dose-dependent decrease on cleavage and blastocyst rates. Besides, higher level of ROS, rapid reduction of MMP, and the appearance of γH2AX revealed that embryos are injured early in mild oxidative stress. Additionally, γH2AX may involve during DNA damage response in early embryos. And the apoptotic rate of blastocyst may significantly increase when DNA damage repair is inadequate. Most importantly, our research provides embryo models to study cell cycle regulation and DNA damage response under condition of different levels of oxidative stress.
1. Introduction
In aerobic respiration cell, ROS, such as H2O2, hydroxyl radical (•OH) and superoxide anion radical (O2 −), appears as a by-products of cell metabolism [1]. Physiological concentration of ROS is necessary to maintain normal embryo development [2, 3]. However, excessive ROS, produced in improper culture conditions, such as culture systems [4], light conditions [5], and gas atmosphere [1, 6–8], may cause developmental arrest, DNA damage, and cell apoptosis [1]. In many species studied, evidences have been found that ROS compromises embryo development [4–8]. In contrast with gassed bags, incubator chamber is more efficient in generating higher number and better quality of bovine embryos due to a lower level of ROS [4]. Hamster embryos will stop dividing at two-cell stage when zygotes are exposed to cool white fluorescent light [5]. Besides, the number of cleaved embryos is similar when embryos are cultured under 5% or 20% O2 [7, 8]. But there is a decline in the percentage of blastocyst formation [7, 8]. We can learn that human embryos on day 3 are apparently normal in both groups, but embryo quality is prior in 5% O2 [8]. Those studies revealed that ROS-induced damage correlates with the phenomenon above.
ROS displays roles of double-edged sword in many physiological and pathological processes in cell [9]. It affects embryo development by altering gene expression [1], inducing lipid peroxidation and DNA damage [1, 9], containing nuclear and mitochondrial DNA strand breaks [9, 10]. Numerous studies used H2O2 to simulate deleterious effects of ROS on embryo development. H2O2, a form of highly active ROS, readily penetrates the cellular envelope and forms •OH which is a more active form of ROS. Liu et al. chose an intensive (1 mM H2O2 for 1.5 h) and a mild (0.2 mM for 15 min) stimulus to treat zygotes (fertilized in vivo); embryos from both groups do not divide till 96 hpi [11]. In another experiment, 2-cell embryos were treated with different doses of H2O2 (0–50 μM) for 30 or 60 min; the results showed a negative dose-effect on blastocyst rate [12]. We may observe a decline in blastocyst rate when treating zygotes with 35 μM H2O2 for 15 min [13]. Researches on embryos injured by ROS are common, but most of their embryos was collected from in vivo fertilization, and/or part of the oxidative stimulus started at a later stage of embryo development. Those are inconsistent with clinical assisted reproductive techniques which eggs fertilize in vitro and embryos suffer oxidative stress from the very first.
Studies state that accumulation of ROS caused developmental arrest of embryos by altering activities and structures of mitochondria [10, 11, 14]. Firstly, alteration of mitochondria is an early event in the process of oxidative damage [11]. Secondly, mild oxidative stress may induce a decline in MMP and dysfunction of mitochondria. Then cell cycle arrest and programmed cell death follow [10, 11, 15]. Lastly, mitochondria play an important role in mediating embryo development and apoptosis in oxidative status [10]. Thus, we regard decrease of MMP as an indicator of mitochondrial damage and the early phase of apoptosis.
Another important inducement of embryo developmental retardation is DNA damage. Our previous studies found that γH2AX, the marker of DNA damage [16–18], appeared at 1- and 4-cell embryos fertilized with H2O2-treated sperm [19]. Further experiments revealed that oxidative stress in sperm activated G2/M delay by cascade of ATM → Chk1 → Cdc25B/Cdc25C to repair damaged DNA [19–21]. It has been identified that damage response checkpoints include G1/S, intra-S, and G2/M [22]. Cell cycle delay gains time for DNA repair. Besides, γH2AX provides other molecules such as ATM, Rad 50, and Rad 51 with a recognition site to activate DNA damage response [16–18, 23]. Those molecules are key to repair DNA damage. But specific molecular mechanisms of developmental delay in embryos with oxidative-stressed damage are obscure. Also, cell cycle checkpoint and apoptosis share some common molecules [24]. To some extent, cells will end with death if DNA damage does not repair [25].
The phenomenon above pushes us to research the molecular mechanisms of developmental retardation in zygotes with oxidative-stressed damage. Because of the limitation of clinical researches on human embryos, we treated mouse zygotes with different doses of exogenous H2O2 to simulate effects of oxidative stress on embryonic development. In order to clarify the molecular mechanisms of DNA damage response activated by oxidative stress and cell cycle arrest in early embryos in subsequent experiment, we aim to develop alternative embryo models with oxidative damage in this research.
2. Materials and Methods
2.1. Animals
Adult Kun-Ming mice (3–6 weeks old) were purchased from the Animal Center of Shantou University Medical College and treated in compliance with the Guide for the Care of Use of Laboratory Animal by the US National Institutes of Health (NIH Publication number 85-23, revised 1996) and the rules of the National Animal Protection of China. All experimental protocols were approved by the Laboratory Animal Ethics Committee of our institution (SUMC2014-014). This study was approved by the Institutional Animal Care and Use Committee of Shantou University Medical College.
2.2. Reagents and Media
All reagents were purchased from Sigma (St. Louis, MO, USA) unless otherwise stated. Rabbit antibody for anti-phospho-histone H2AX (γH2AX; Ser 139) and goat anti-rabbit Alexa Fluor 488 secondary antibody were from Abcam (England). The In Situ Cell Death Detection kit (Fluorescein) was from Roche (Switzerland). PI and DAPI were from Panera (France). Human tubal fluid-HEPES (HTF-HEPES) was from Cooper Surgical (USA). Phosphate-buffered saline (PBS) was dissolved in Milli-Q water: 136.9 mM NaCl, 2.7 mM KCl, 0.9 mM CaCl2, 0.5 mM MgCl2, 7 mM Na2HPO4, 1.25 mM NaH2PO4, 1.5 mM KH2PO4, and 1 g/L PVA (290 mOsm/kg; pH 7.2). Pancreatin solution was diluted to 0.1% with PBS, the pH adjusted to 3.0 with HCl, filtered, aliquoted, and stored at 4°C. The embryo culture medium was made by adding 0.4% BSA and 10% fetal bovine serum to HTF solution. And the medium of the treated group was made by addition of H2O2 to a final concentration of 0.01, 0.02, 0.03, 0.04, 0.05, or 0.1 mM and then preequilibrated them for 1 hour before use. Stationary liquid was 4% paraformaldehyde in PBS. Membrane liquid was PBS + 0.5% Triton X-100. Sperm capacitation liquid (HTF solution + 1.5% bovine serum albumin), fertilization liquid (HTF solution + 0.4% BSA), and embryo culture medium were placed at 37°C in a 5% CO2 incubator for 4 h. Blastocysts were cultured in blastocyst medium (Cooper Surgical, Inc.).
2.3. Epididymal Sperm Preparation, Collection, and Culture of Oocytes and Embryos
As described in our previous study, sperm was obtained from the tail of the epididymis of mature male mice and incubated in capacitation medium at 37°C in a 5% CO2 incubator for 1 h [19]. Female mice were intraperitoneally injected with consecutive injections of 10 IU pregnant mare serum gonadotropin (PMSG) to promote ovulation. And 10 IU human chorionic gonadotropin (HCG) 48 h apart. Then mice were euthanized by cervical dislocation at 13 to 15 h after HCG administration. Cumulus oocytes were collected from the ovaries of the mice, placed in 37°C PBS, then moved to 37°C fertilization liquid containing 10 μL capacitated sperm, and incubated for 6 h to permit fertilization. Zygotes were washed three times and cultured in new medium after fertilization.
2.4. Experiment 1: Effects of Zygotes Oxidative Stress on Embryonic Development
According to studies by Liu et al. [11] and our preliminary experiments, zygotes (7 hpi) were placed in culture medium with different concentrations of H2O2 for 30 minutes, then rinsed three times, and cultured in medium without H2O2. Embryos were incubated at 37°C in a 5% CO2 atmosphere. The medium was renewed daily. Development of two-cell and four-cell embryos and blastocysts was observed, respectively, at 24 hpi, 48 hpi, and 96 hpi with an inverted microscope (Olympus Inc., Japan). Cleavage and blastocyst formation rates were defined as the total number of embryos from corresponding stage divided by total number of zygotes. The total number of zygotes included in 0.01, 0.02, 0.03, 0.04, 0.05, and 0.1 mM H2O2 was 519, 363, 316, 489, 365, 326, and 50, respectively.
2.5. Experiment 2: Effects of Oxidative Stress Induction on Embryo Injured Related Variables
2.5.1. Determination of ROS Products
The intracellular ROS level was detected by 2′, 7′-dichlorofluorescein fluorescence (DCFH-DA). A stock solution of DCFH-DA (1 × 10−3 mol/L in DMSO) was added to the embryo culture medium to a final concentration of 10 μmol/L, and zygotes were incubated at 37°C in 5% CO2 for 30 minutes. Embryos were rinsed three times (5 min each time) in fresh culture fluid to remove the residual dye and then placed on a glass slide with 20 μL culture fluid. Fluorescence staining was observed immediately under a fluorescence microscope with exciting light of 495 nm and emissive light of 520 nm (Nikon Eclipse 90 Ni-E). The fluorescence intensity was recorded at 5 s after exciting the zygotes. Image Pro Plus 6.0 was used to quantify the fluorescence (Media Cybernetics Inc., Bethesda, MD, USA). 34 zygotes from the treated group and 29 zygotes from the untreated group were detected in this part.
2.5.2. Detection of Mitochondrial Membrane Potential (MMP)
MMP was detected at 1-cell embryos with the lipophilic, cationic probe 5, 5′, 6, 6′-tetrachloro-1, 1′ 3, 3′-tetraethylbenzimidazolylcarbocyanine iodide (JC-1, stock 5 mg/mL). Then, PBS was added to the solution of JC-1 to a final concentration of 1.25 μmol/L. Zygotes were washed three times with PBS (5 min each time) after staining for 20 min and placed on a glass slide. Images were recorded immediately by using a fluorescent microscope (Nikon Eclipse 90 Ni-E) at an excitation wavelength of 488 nm and beam path control setting at LP 585 nm for Ch1 and BP 505–530 nm for Ch2. Analysis of data was done with Image Pro Plus 6.0 software (Media Cybernetics Inc., Bethesda, MD, USA). The number of treated and untreated zygotes at each hour was shown as follows: 18 and 20 for 0 h, 17 and 25 for 1 h, 22 and 19 for 2 h, 22 and 21 for 4 h, and 19 and 16 for 6 h.
2.5.3. Assay of γH2AX
Embryos were rinsed with TPBS (PBS + 0.05% Tween 20), and zona pellucidae were removed from embryos with pancreatin (0.1%) for 30 s. Then embryos were fixed with 4% paraformaldehyde in TPBS for 30 min, permeabilized at room temperature for 30 min, and blocked for 1 h at room temperature in blocking solution (PBS + 0.05% Tween 20 + 3% BSA + 10% normal goat serum). Before each step, embryos were washed with TPBS three times, 5 minutes per time. After blocking, embryos were incubated with rabbit anti-γH2AX primary antibody (1 : 1000 dilution) for 12 h to 15 h at 4°C, then washed, and incubated with goat anti-rabbit Alexa Fluor 488 secondary antibody (1 : 500 dilution). The immunostained embryos were washed with TPBS, counterstained with propidium iodide, and observed under a fluorescence microscope (Nikon Eclipse 90 Ni-E).
2.5.4. TUNEL Assay
TUNEL assay was performed to analyze apoptosis of blastocysts, using the In Situ Cell Death Detection kit (fluorescein) in accordance with manufacturer's instructions. Embryos were withdrawn at 96 hpi to collect blastocysts according to our previous experiment [20]. Zona pellucidae were removed from the embryos at first. Then embryos were washed three times with TPBS, fixed in 4% paraformaldehyde at room temperature for 30 min, and mounted on polylysine slides and washed again with TPBS and permeabilized at room temperature for 30 min. Embryos were washed with TPBS three times again and incubated with fluorescein-conjugated dUTP and terminal deoxynucleotidyl transferase at 37°C in dark for 1 h. The reaction was terminated by washing in TPBS three times, 5 min each time, and then the embryos were counterstained with DAPI, rinsed with TPBS, and sealed with coverslips. Observation was done under an Olympus FluoView FV 1000 confocal microscope (Olympus Inc., Japan). According to previous studies [20], the apoptotic rate was expressed as the percentage of TUNEL-positive cell number relative to the total cell number of blastocysts. 24 blastocysts from the control group and 34 from the treated group were used for the TUNEL assay.
2.6. Statistical Analysis
Results were collected from at least 3 independent experiments and data were analyzed by SPSS 17.0 software (SPSS Inc., Chicago, IL). Data showed as percentages were analyzed using Chi square test. Values expressed as mean ± SD of treated and untreated groups was compared, using Student's t-test. P < 0.05 was considered statistically significant.
3. Results
3.1. Experiment 1: A Dose-Dependent Decline of Embryonic Development at Each Stage
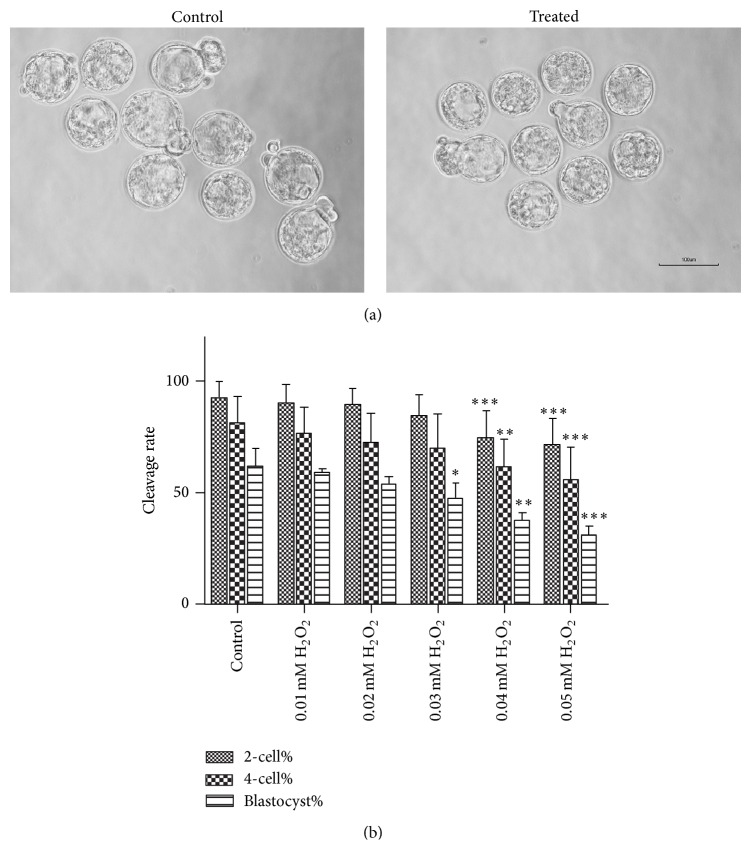
We could learn from Figure 1(a) that blastocysts have a bigger shape in control group than those in 0.03 mM H2O2 group. And we characterized the dose-response of H2O2 on embryo development. No zygotes divided at 60 hpi when they were treated with 0.1 mM H2O2. H2O2 at 0.01 mM or 0.02 mM had no significant effects on the percentages of two- and four-cell embryos or blastocyst formation at 24, 48, and 96 hpi, respectively (Figure 1(b)) while H2O2 at 0.03 mM exposure produced a 14.50% reduction in blastocyst formation rate (P < 0.05, Figure 1(b)). Although the two- and four-cell embryo formation rates were not different from the control group, 0.03 mM H2O2 was the concentration of our treated group in subsequent experiments. When mouse zygotes were exposed to H2O2 at 0.04 mM or 0.05 mM, the proportion of blastocyst formation and cleavage rates decreased (P < 0.05, Figure 1(b)) and exhibited a dose-dependent decline (Figure 1(b)).
Figure 1.
Comparisons of embryo development between zygotes from groups with different concentration of H2O2 and control group. (a) Representative images of embryos at 96 hpi from control group and 0.03 mM H2O2 group. (b) Counting out the cleavage rates from each group, blastocyst formation rate declined from 0.03 mM H2O2 group; early embryos showed stagnation from 0.04 mM H2O2 group. Data are presented as mean ± SD in six independent experiments for each group at least. Differences between the groups were calculated using Chi square test. ∗ P < 0.05; ∗∗ P < 0.01; ∗∗∗ P < 0.001 compared with the control group.
3.2. Experiment 2: Oxidative Stress Implies the Variations in Embryos
3.2.1. ROS Concentrations in Embryos
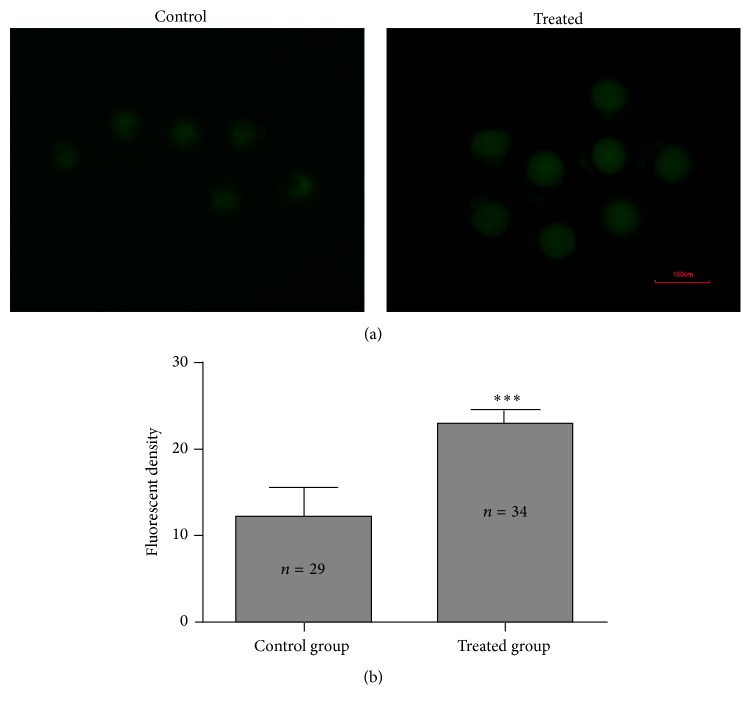
The comparison of the yields of ROS between treated and untreated group was via DCFH-DA fluorescence intensity in zygotes (7.5 hpi) (Figure 2(a)). The mean fluorescence intensity of ROS in zygotes of the treated group (23.01 ± 1.47) was about 2-fold higher than that in the control group (12.19 ± 3.44) (P < 0.05, Figure 2(b)).
Figure 2.
Comparisons of ROS levels between control group and H2O2 treated group. (a) Representative pictures of ROS levels. (b) Average fluorescence intensity per zygote. Data are showed as mean ± SD, collected from three independent experiments, and each experiment had 5 zygotes at least. n shows the number of zygotes. ∗∗∗ P < 0.001, the treated group compared with the untreated group (Student's t-test).
3.2.2. Change in MMP of Zygotes with Time
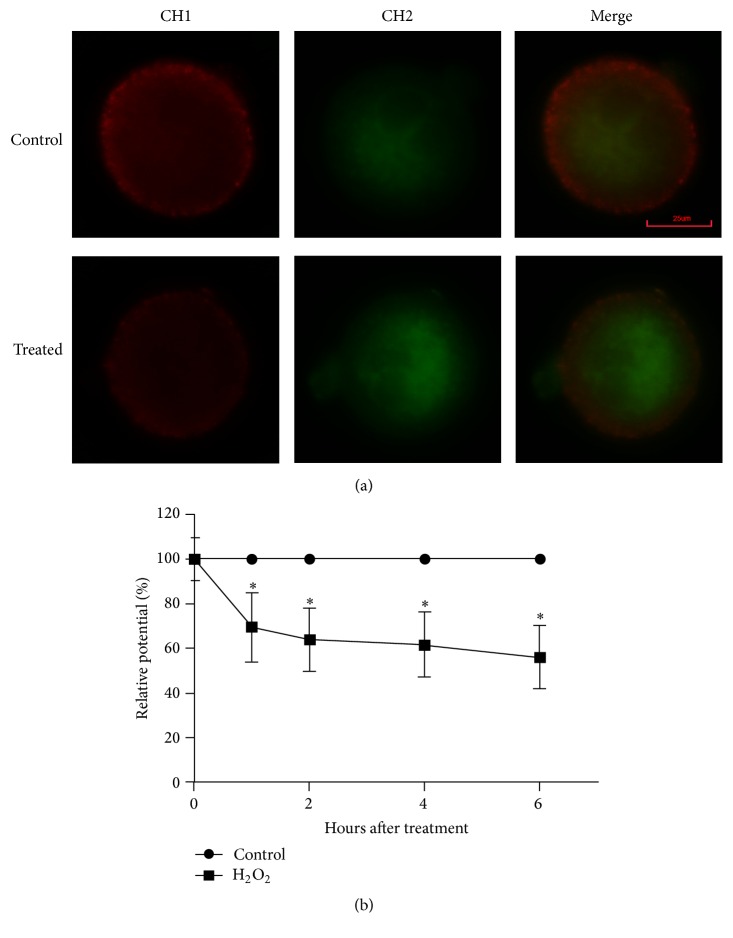
To further characterize the harmful effects of mild oxidative stress on embryo development in vitro, we detected the variation of MMP after treatment of zygotes with 0.03 mM H2O2 at 0, 1, 2, 4, and 6 h. The MMP was higher in control group than treated group at all check points (Figure 3(a)). The relative ratio of red to green fluorescence intensity indicated that MMP dropped rapidly within the first hour and did not recover within 6 hours after treatment (P < 0.05, Figure 3(b)). That is to say, the relative pixel ratio intensity, which reflected the MMP, was lower in H2O2-treated zygotes within 1 h after the treatment of H2O2 compared with the control group.
Figure 3.
Dysfunction of mitochondria in mouse zygotes induced by H2O2. (a) Representative images of MMP in mouse zygotes at 6 h from treated group and control group; red fluorescence from channel 1 represented J-aggregates (high polarized mitochondria); green fluorescence from channel 2 represented monomer form of JC-1 (low polarized mitochondria). (b) The analysis of MMP was via the comparisons of relative fluorescence intensity and set the average value of red/green fluorescence intensity at each time point from control group as 100%; MMP in treated group was showed relative to control group at the corresponding point time. MMP in treated zygotes declined during the first hour after treatment by 0.03 mM H2O2 for 30 min and kept on dropping over the subsequent hours. Data are presented as mean ± SD in three independent experiments. ∗ P < 0.05, the treated group versus the control group (Student's t-test).
3.2.3. Expression of γH2AX in Embryos with Oxidative Stress Injury
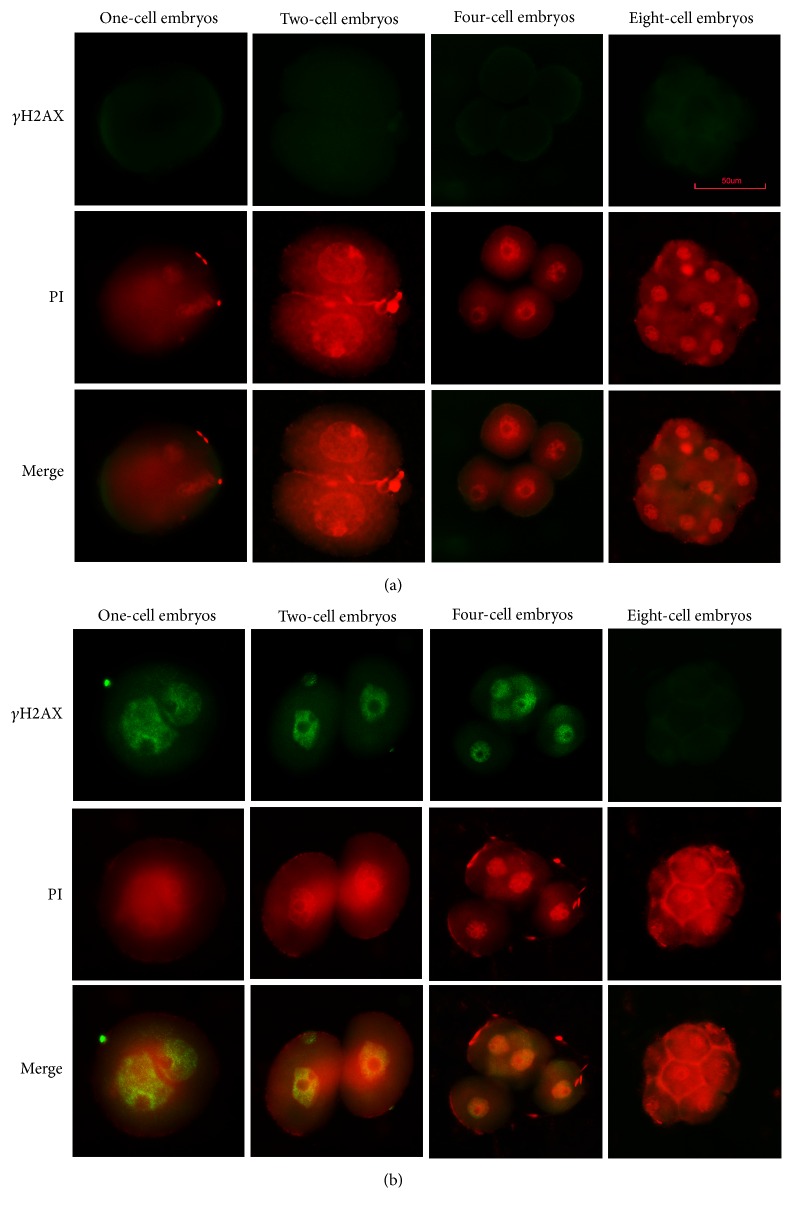
We discovered that H2O2-treated embryos expressed γH2AX at the one-, two-, and four-cell embryos. However, there is no γH2AX staining in control group (Figures 4(a) and 4(b)). Therefore, DNA damage existed in one-, two-, and four-cell embryos that developed from oxidative damaged zygotes. Representative images are shown in Figures 4(a) and 4(b).
Figure 4.
Expression of γH2AX. (a) Control group; no positive signal. (b) 0.03 mM H2O2 treated group; positive signal at one-, two- and four-cell stages. PI = propidium iodide staining.
3.2.4. Apoptosis of Blastocysts
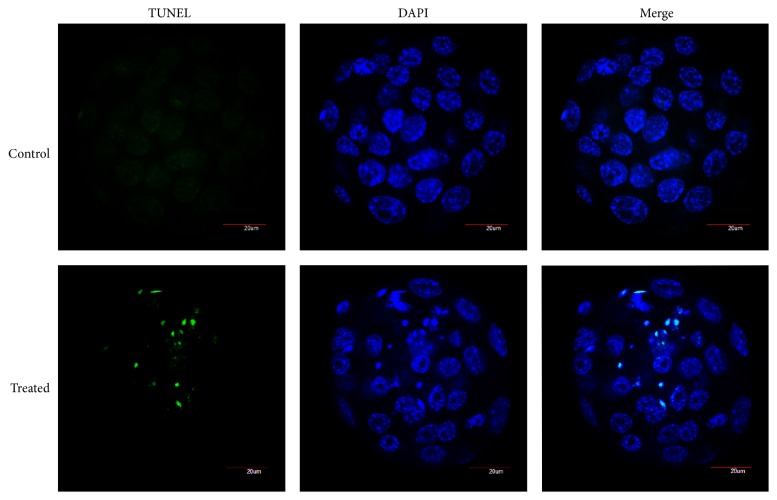
The average cell number per blastocyst in the treated group was 37.77 ± 3.94 and 46.74 ± 3.05 in the control group (P < 0.05). At the same time, the average apoptotic cell counts was over 2-fold higher in the treated group relative to the untreated group (6.24 ± 2.42 (treated) versus 2.27 ± 0.70 (control)) (P < 0.05). Moreover, the mean apoptotic rate was (15.9 ± 5.8)% for each H2O2-treated embryo versus (4.9 ± 1.3)% for the control. Representative images of normal and apoptotic mouse blastocysts are illustrated in Figure 5.
Figure 5.
Hydrogen peroxide-induced apoptosis. Representative images of normal and apoptotic cells in mouse blastocysts; no positive signals (Green stains) appeared in control group; several positive signals could be seen in treated group. Nuclei were stained with DAPI (blue).
4. Discussion
Embryos cultured in vitro are subjected to oxidative stress. We used H2O2 to mimic oxidative stress. Finally, we gain a series of embryo models which suffer different levels of oxidative stress. And we found that a slightly high level of ROS could lead to a low formation rate of blastocyst with less cells by reducing MMP of zygotes and increasing DNA damage at one-, two-, and four-cell embryos.
Bain and colleagues reported that effects of H2O2 on blastocyst formation became more severe during the treatment of later stages of development [26]. Another important factor is embryos subjecting to oxidative stress from early stage in clinic. Thus, we chose zygotes rather than 2- or 4-cell embryos to be treated to avoid excessively adverse effects on embryos. Ciray et al. and Kasterstein et al. have shown that embryos in clinic (5% or 20% O2 concentration) have a similar cleavage rate at day 3 but decreasing blastocyst formation rate at day 5 or 6 in 20% O2 atmosphere [7, 8]. So we use small concentration gradient (0–0.1 mM) and low doses of H2O2 to treat zygotes for 30 minutes according to our preliminary experiment and other researches [10, 11, 13, 26], just for embryo model with similar developmental pattern above. Furthermore, cells undergoing G1→S transition are very sensitive to low levels of stimulus [22]. Many embryos under oxidative stress step into a transient cell cycle arrest which is activated by DNA damage response before apoptosis [11]. In order to gain a suitable model to research molecular mechanisms about DNA damage repair, cell cycle, and apoptosis in our subsequent experiments, we utilized H2O2 to treat zygotes at 7 hpi as they are in G1 phase [20], which is the first checkpoint of cell cycle to keep genetic stability. Also, that may provide models for us to study molecular mechanisms which activate G2/M phase delay.
Based on the theory above, we implemented experiment 1 and the results showed that zygotes presented no division in 0.1 mM H2O2 at 60 hpi. And there will be a lower percentage of embryo formation rate at each stage with treatment of a higher concentration of H2O2. To put it another way, embryo developmental patterns in 0.01–0.05 mM H2O2 could be used, as alternate models, to research influences of mild oxidative stress on developmental retardation. Developmental conditions of 0.01 or 0.02 mM H2O2 group could be used to study embryo development by Peng et al. No obvious differences were found in cleavage or high quality embryo rate at day 3 [27]. Moreover, the embryo model in 0.04 or 0.05 mM H2O2 group may suit studying the human embryo development with low formation rate at day 3 and day 5 or 6 [28]. Most importantly, we selected 0.03 mM H2O2 in our subsequent studies as we found the developmental style was similar to clinic and other researches [7, 8]. That is, there are no obvious differences in the percentage of cleavage rates between 20% and 5% O2 concentration, but 5% O2 group have more top-quality embryos on day 3 and more blastocysts on day 5 or 6 [7, 8]. Our data presented that embryo formation rates at 24 h and 48 h were approximate, but more blastocysts with less apoptotic cells were in control group than 0.03 mM H2O2. The concurrent research found G2/M delay of zygotes in 0.03 mM H2O2 group [29]. As is known human embryos at 4- to 8-cell and mouse embryos at 1- to 2-cell are undergoing ZGA (zygotic gene activation) phase, which is a key process of maternal to zygotic transition [29, 30]. Embryos develop the ability to support proper development of early stage during ZGA phase [30]. The above prompt is that potential damage may exist in early mouse embryos (1- and/or 2-cell embryos) in 0.03 mM H2O2 group and it may be adverse to the formation of blastocyst. Further investigations will be done to determine the molecular mechanisms about cell cycle arrest. As to reasons for the otherness of human embryo development aforementioned, it may be the inconsistent ovulation induction protocols, different lab conditions, technologist's experiences, technologies and skills, and many other uncontrollable factors. Additionally, we have observed that the shape of embryos in control group is larger than that of the treated group and that outcome may be induced by high apoptotic rate in blastocyst of treated group. Nevertheless, it needs more endeavor to unveil the influences of the above phenomenon on blastocyst quality. Our results showed a higher production of intracellular ROS (Figure 2(b)) which indicates that embryos are in oxidative stress status. Moreover, we could draw a conclusion from the sharp decline of MMP that zygotes were damaged during 1 h after treated by 0.03 mM H2O2. Embryos with continuous high MMP can reactive the arrest development [31]. Thus, embryos may be damaged by ROS at the first hour after treatment by 0.03 mM H2O2.
Our previous study revealed that development of zygotes from fresh spermatozoa and sperm treated with 1 mM H2O2 for 1 h was similar [19]. But blastocyst formation rate was significantly declined when treating zygotes with lower concentration of H2O2 and shorter processing time. It states that oxidative-stressed damage put more negative influences on zygotes than on sperm. And the reasons might be as follows: (1) two pronuclei were exposed to oxidative stress and both might be damaged; (2) embryonic cytoplasm was affected by ROS, especially mitochondria. Almost the whole cytoplasm is from oocyte and cytoplasm plays a major role in regulating early embryo development [10]. That finding further suggests that maternal resources are crucial in mediating early embryo development which conforms to other researches [29, 30].
To further delineate the effects of oxidative stress on DNA, we firstly detected the expression of γH2AX. Our results showed that γH2AX appeared at the one-, two-, and four-cell embryos in the treated group. Luo et al. found that mouse embryos exposed to 50-Hz sinusoidal electromagnetic fields (EMFs) for 24 h elicited the presence of γH2AX foci in two-cell embryos or γH2AX expressed in two- and eight-cell embryos with exposure for 48 h [32]. UV irradiation caused expression of γH2AX at later embryo stage only [33]. Those behaviors of γH2AX may be induced by various types of DNA damage or embryos diversely in response to different degree of DNA damage. Further investigations are necessary to better unveil that unsolved puzzle. G2/M checkpoint of zygotes was delayed at 0.03 mM H2O2 group as compared with control group [29]. And DNA damage exists in early embryos. There were no significant differences in the percentages of 2- and 4-cell embryo formation. One possible reason could be that G2/M phase is from 17 hpi to 20 hpi in control group [20], but we recorded the cleavage rate at 24 hpi, the last 4 hours before recording provide embryos of treated group with time to catch up. Another reason might be that oxidative stress, increasing the metabolism with superoxide detoxification, might accelerate the speed of the first cleavage [26]. Also DNA damage response may play a positive role in repair damaged DNA of early embryos, and it may benefit from the functions of γH2AX below: firstly, γH2AX may work as it collects other signal or repair proteins to injured sites [16, 17, 23]; secondly, it localizes at kinetochores and calls together MDC1, MDC2, and CDC20 for assembling an integrated mitotic checkpoint complexes [34]; finally, dynamic γH2AX in cell cycle reminds us of its specific role in the process of mitosis [18, 35]. Recently, it has been demonstrated that PIk3-mediated activation of γH2AX subsequently regulates the cell cycle progression and cell fate in human corneal epithelial cells [36]. Besides, γH2AX accumulated beyond its role in DNA damage and as a regulator of cell cycle progression by inhibitor DNA replication [37, 38]. Those remind us that γH2AX may involve in DNA damage response to regulate cell cycle in zygotes with oxidative-stress damage, too. We may learn that normal zygotes mediate G2/M by protein kinase A (PKA), WEE1B, 14-3-3ε, and M-phase promoting factors [39–41]. But the specific molecular pathways regulate cell cycle of normal zygotes and zygotes with oxidative-stressed damage are unclear, and whether DNA damage in zygotes activates cascade of ATM → Chk1 → Cdc25B/Cdc25C during G2/M to repair the damage depends on our further study. Moreover, compared with our previous study, in which γH2AX appeared at one- and four-cell stage [19], the above phenomenon verifies that zygotes with oxidative-stressed damage exert more negative effects on embryo development than zygotes derived from ova fertilized with H2O2-treated sperm again. In the later stage that did not obviously express γH2AX, we assume that damaged cells in those embryos were removed following either apoptosis or permanently developmental arrest or repairing their damaged DNA. Our results illustrated that ROS gives rise to the formation of blastocysts with less cells. Although DNA damage response may produce positive roles at one-, two-, and four-cell embryo stages, the cell counts of blastocyst and the blastocyst formation rate were still significantly lower than the untreated group, indicating that activation of DNA repair mechanisms and/or cell cycle arrest failed to completely repair the damage. Our results strongly state that adverse effects of ROS on embryo development, at least partly, act through DNA damage.
5. Conclusions
In brief, effects of oxidative stress on early embryos are dose-dependent and lasting to blastocyst. Also, the decline of MMP caused by oxidative stress in zygotes may forecast the compromised development of embryos. γH2AX may play an important role in DNA repair of early embryo stages, but cells with unrepaired DNA will reduce the blastocyst formation rate. Therefore, our embryo models should be considered when evaluating the hazardous effects of environmental ROS in human embryos and search for effective approaches for prevention.
Acknowledgments
The authors are thankful for the support from Center for Neuroscience, Shantou University Medical College for the utilization of the laser confocal microscopy. Those studies were supported by National Natural Science Foundation of China (nos. 30872771, 81070542, 81471522, and 81671536); Natural Science Foundation of Guangdong Province of China (nos. 8151503102000010, 10151503102000020, and 2014A030313482); Guangdong Provincial Science and Technology Project (2016A020218015).
Competing Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
References
- 1.Takahashi M. Oxidative stress and redox regulation on in vitro development of mammalian embryos. The Journal of Reproduction and Development. 2012;58(1):1–9. doi: 10.1262/jrd.11-138n. [DOI] [PubMed] [Google Scholar]
- 2.D'Autréaux B., Toledano M. B. ROS as signalling molecules: mechanisms that generate specificity in ROS homeostasis. Nature Reviews Molecular Cell Biology. 2007;8(10):813–824. doi: 10.1038/nrm2256. [DOI] [PubMed] [Google Scholar]
- 3.Groeger G., Quiney C., Cotter T. G. Hydrogen peroxide as a cell-survival signaling molecule. Antioxidants & Redox Signaling. 2009;11(11):2655–2671. doi: 10.1089/ars.2009.2728. [DOI] [PubMed] [Google Scholar]
- 4.Arias M. E., Sanchez R., Felmer R. Evaluation of different culture systems with low oxygen tension on the development, quality and oxidative stress-related genes of bovine embryos produced in vitro. Zygote. 2012;20(3):209–217. doi: 10.1017/S0967199411000025. [DOI] [PubMed] [Google Scholar]
- 5.Takenaka M., Horiuchi T., Yanagimachi R. Effects of light on development of mammalian zygotes. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(36):14289–14293. doi: 10.1073/pnas.0706687104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Leoni G. G., Rosati I., Succu S., et al. A low oxygen atmosphere during IVF accelerates the kinetic of formation of in vitro produced ovine blastocysts. Reproduction in Domestic Animals. 2007;42(3):299–304. doi: 10.1111/j.1439-0531.2006.00783.x. [DOI] [PubMed] [Google Scholar]
- 7.Ciray H. N., Aksoy T., Yaramanci K., Karayaka I., Bahceci M. In vitro culture under physiologic oxygen concentration improves blastocyst yield and quality: a prospective randomized survey on sibling oocytes. Fertility and Sterility. 2009;91(4):1459–1461. doi: 10.1016/j.fertnstert.2008.07.1707. [DOI] [PubMed] [Google Scholar]
- 8.Kasterstein E., Strassburger D., Komarovsky D., et al. The effect of two distinct levels of oxygen concentration on embryo development in a sibling oocyte study. Journal of Assisted Reproduction and Genetics. 2013;30(8):1073–1079. doi: 10.1007/s10815-013-0032-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gille J. J. P., Joenje H. Cell culture models for oxidative stress: superoxide and hydrogen peroxide versus normobaric hyperoxia. Mutation Research/DNAging. 1992;275(3-6):405–414. doi: 10.1016/0921-8734(92)90043-o. [DOI] [PubMed] [Google Scholar]
- 10.Liu L., Keefe D. L. Cytoplasm mediates both development and oxidation-induced apoptotic cell death in mouse zygotes. Biology of Reproduction. 2000;62(6):1828–1834. doi: 10.1095/biolreprod62.6.1828. [DOI] [PubMed] [Google Scholar]
- 11.Liu L., Trimarchi J. R., Keefe D. L. Involvement of mitochondria in oxidative stress-induced cell death in mouse zygotes. Biology of Reproduction. 2000;62(6):1745–1753. doi: 10.1095/biolreprod62.6.1745. [DOI] [PubMed] [Google Scholar]
- 12.Cebral E., Carrasco I., Vantman D., Smith R. Preimplantation embryotoxicity after mouse embryo exposition to reactive oxygen species. Biocell. 2007;31(1):51–59. [PubMed] [Google Scholar]
- 13.Yu S., Long H., Lyu Q.-F., et al. Protective effect of quercetin on the development of preimplantation mouse embryos against hydrogen peroxide-induced oxidative injury. PLoS ONE. 2014;9(2, article e89520) doi: 10.1371/journal.pone.0089520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Velez-Pardo C., Morales A. T., Rio M. J. D., Olivera-Angel M. Endogenously generated hydrogen peroxide induces apoptosis via mitochondrial damage independent of NF-κB and p53 activation in bovine embryos. Theriogenology. 2007;67(7):1285–1296. doi: 10.1016/j.theriogenology.2007.01.018. [DOI] [PubMed] [Google Scholar]
- 15.Igosheva N., Abramov A. Y., Poston L., et al. Maternal diet-induced obesity alters mitochondrial activity and redox status in mouse oocytes and zygotes. PLoS ONE. 2010;5(4, article e10074) doi: 10.1371/journal.pone.0010074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Redon C. E., Nakamura A. J., Zhang Y.-W., et al. Histone γH2AX and poly(ADP-ribose) as clinical pharmacodynamic biomarkers. Clinical Cancer Research. 2010;16(18):4532–4542. doi: 10.1158/1078-0432.ccr-10-0523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li A., Yu Y., Lee S.-C., Ishibashi T., Lees-Miller S. P., Ausió J. Phosphorylation of histone H2A.X by DNA-dependent protein kinase is not affected by core histone acetylation, but it alters nucleosome stability and histone H1 binding. The Journal of Biological Chemistry. 2010;285(23):17778–17788. doi: 10.1074/jbc.m110.116426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Turinetto V., Giachino C. Survey and summary multiple facets of histone variant H2AX: a DNA double-strand-break marker with several biological functions. Nucleic Acids Research. 2015;43(5):2489–2498. doi: 10.1093/nar/gkv061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xiao J., Liu Y., Li Z., et al. Effects of the insemination of hydrogen peroxide-treated epididymal mouse spermatozoa on γH2AX repair and embryo development. PLoS ONE. 2012;7(6) doi: 10.1371/journal.pone.0038742.e38742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang B., Li Z., Wang C., et al. Zygotic G2/M cell cycle arrest induced by ATM/Chk1 activation and DNA repair in mouse embryos fertilized with hydrogen peroxide-treated epididymal mouse sperm. PLoS ONE. 2013;8(9, article e73987) doi: 10.1371/journal.pone.0073987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Song Y., Li Z., Wang B., Xiao J., Wang X., Huang J. Phospho-Cdc25 correlates with activating G2/M checkpoint in mouse zygotes fertilized with hydrogen peroxide-treated mouse sperm. Molecular and Cellular Biochemistry. 2014;396(1-2):41–48. doi: 10.1007/s11010-014-2140-1. [DOI] [PubMed] [Google Scholar]
- 22.Deckbar D., Jeggo P. A., Löbrich M. Understanding the limitations of radiation-induced cell cycle checkpoints. Critical Reviews in Biochemistry and Molecular Biology. 2011;46(4):271–283. doi: 10.3109/10409238.2011.575764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tanaka T., Halicka H. D., Huang X., Traganos F., Darzynkiewicz Z. Constitutive histone H2AX phosphorylation and ATM activation, the reporters of DNA damage by endogenous oxidants. Cell Cycle. 2006;5(17):1940–1945. doi: 10.4161/cc.5.17.3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mladenov E., Magin S., Soni A., Iliakis G. DNA double-strand-break repair in higher eukaryotes and its role in genomic instability and cancer: cell cycle and proliferation-dependent regulation. Seminars in Cancer Biology. 2016;37-38:51–64. doi: 10.1016/j.semcancer.2016.03.003. [DOI] [PubMed] [Google Scholar]
- 25.Li H., Mitchell J. R., Hasty P. DNA double-strand breaks: a potential causative factor for mammalian aging? Mechanisms of Ageing and Development. 2008;129(7-8):416–424. doi: 10.1016/j.mad.2008.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bain N. T., Madan P., Betts D. H. The early embryo response to intracellular reactive oxygen species is developmentally regulated. Reproduction, Fertility and Development. 2011;23(4):561–575. doi: 10.1071/RD10148. [DOI] [PubMed] [Google Scholar]
- 27.Peng Z.-F., Shi S.-L., Jin H.-X., et al. Impact of oxygen concentrations on fertilization, cleavage, implantation, and pregnancy rates of in vitro generated human embryos. International Journal of Clinical and Experimental Medicine. 2015;8(4):6179–6185. [PMC free article] [PubMed] [Google Scholar]
- 28.Guo N., Li Y., Ai J., Gu L., Chen W., Liu Q. Two different concentrations of oxygen for culturing precompaction stage embryos on human embryo development competence: a prospective randomized sibling-oocyte study. International Journal of Clinical and Experimental Pathology. 2014;7(9):6191–6198. [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang Y., Qian D., Li Z., et al. Oxidative stress-induced DNA damage of mouse zygotes triggers G2/M checkpoint and phosphorylates Cdc25 and Cdc2. Cell Stress and Chaperones. 2016;21(4):687–696. doi: 10.1007/s12192-016-0693-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Langley A. R., Smith J. C., Stemple D. L., Harvey S. A. New insights into the maternal to zygotic transition. Development. 2014;141(20):3834–3841. doi: 10.1242/dev.102368. [DOI] [PubMed] [Google Scholar]
- 31.Van Blerkom J., Cox H., Davis P. Regulatory roles for mitochondria in the peri-implantation mouse blastocyst: possible origins and developmental significance of differential Δψm. Reproduction. 2006;131(5):961–976. doi: 10.1530/rep.1.00458. [DOI] [PubMed] [Google Scholar]
- 32.Luo Q., Yang J., Zeng Q.-L., Zhu X.-M., Qian Y.-L., Huang H.-F. 50-Hertz electromagnetic fields induce gammaH2AX foci formation in mouse preimplantation embryos in vitro. Biology of Reproduction. 2006;75(5):673–680. doi: 10.1095/biolreprod.106.052241. [DOI] [PubMed] [Google Scholar]
- 33.Mu X. F., Jin X. L., Farnham M. M. J., Li Y., O'Neill C. DNA damage-sensing kinases mediate the mouse 2-cell embryo's response to genotoxic stress. Biology of Reproduction. 2011;85(3):524–535. doi: 10.1095/biolreprod.110.089334. [DOI] [PubMed] [Google Scholar]
- 34.Eliezer Y., Argaman L., Kornowski M., Roniger M., Goldberg M. Interplay between the DNA damage proteins MDC1 and ATM in the regulation of the spindle assembly checkpoint. The Journal of Biological Chemistry. 2014;289(12):8182–8193. doi: 10.1074/jbc.m113.532739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.An J., Huang Y.-C., Xu Q.-Z., et al. DNA-PKcs plays a dominant role in the regulation of H2AX phosphorylation in response to DNA damage and cell cycle progression. BMC Molecular Biology. 2010;11, article 18 doi: 10.1186/1471-2199-11-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang L., Dai W., Lu L. Osmotic stress-induced phosphorylation of H2AX by polo-like kinase 3 affects cell cycle progression in human corneal epithelial cells. The Journal of Biological Chemistry. 2014;289(43):29827–29835. doi: 10.1074/jbc.m114.597161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fernando R. N., Eleuteri B., Abdelhady S., Nussenzweig A., Andäng M., Ernfors P. Cell cycle restriction by histone H2AX limits proliferation of adult neural stem cells. Proceedings of the National Academy of Sciences of the United States of America. 2011;108(14):5837–5842. doi: 10.1073/pnas.1014993108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Turinetto V., Orlando L., Sanchez-Ripoll Y., et al. High basal γH2AX levels sustain self-renewal of mouse embryonic and induced pluripotent stem cells. STEM CELLS. 2012;30(7):1414–1423. doi: 10.1002/stem.1133. [DOI] [PubMed] [Google Scholar]
- 39.Cui C., Ren X., Liu D., et al. 14-3-3 epsilon prevents G2/M transition of fertilized mouse eggs by binding with CDC25B. BMC Developmental Biology. 2014;14(1, article 33) doi: 10.1186/s12861-014-0033-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu C., Liu Y., Liu Y., et al. Ser 15 of WEE1B is a potential PKA phosphorylation target in G2/M transition in one-cell stage mouse embryos. Molecular Medicine Reports. 2013;7(6):1929–1937. doi: 10.3892/mmr.2013.1437. [DOI] [PubMed] [Google Scholar]
- 41.Yu B., Wang Y., Liu Y., et al. Protein kinase A regulates cell cycle progression of mouse fertilized eggs by means of MPF. Developmental Dynamics. 2005;232(1):98–105. doi: 10.1002/dvdy.20205. [DOI] [PubMed] [Google Scholar]