Abstract
Pheochromocytoma, a rarely reported adrenal gland neoplasm in Old World primates, was diagnosed in five rhesus macaques (Macaca mulatta) and two African green monkeys (Chlorocebus aethiops) from three research institutions. Age and sex were available for six primates. Two males and four females were affected ranging in age from nine to 31 years. All neoplasms were unilateral and, in the cases reporting the affected gland, four involved the right adrenal gland and two involved the left. Diagnosis was established by characteristic histologic features. Immunohistochemically, neoplastic cells in all cases expressed chromogranin A and met-enkephalin and were negative for melan-A and inhibin. Six of seven tumors were positive for beta-endorphin. Pulmonary metastases were present in two rhesus macaques and portal vein invasion in one African green monkey. To the authors’ knowledge, this is the first report of malignant pheochromocytoma in Old World primates.
Keywords: adrenal gland neoplasm, African green monkey, Chlorocebus aethiops, immunohistochemistry, Macaca mulatta, pheochromocytoma, rhesus macaque, Old World primate
Pheochromocytomas are neuroendocrine neoplasms originating from catecholamine-secreting chromaffin cells of the adrenal medulla. They have rarely been reported in macaques (Macaca sp.)4,5,8,9,10 and have not been reported in African green monkeys (Chlorocebus aethiops).
In humans, pheochromocytoma is an uncommon tumor that affects all ages. Most are sporadic, unilateral, solitary and benign. There is no sex predilection. Many pheochromocytomas are functional and secrete catecholamines, causing vasoconstriction and thus sustained or paroxysmal hypertension, tachycardia, cardiac hypertrophy, pulmonary edema, sweating, and headaches. Detection of urinary catecholamines or catabolites allows diagnosis of functional pheochromocytomas.7 Grossly, pheochromocytomas vary in size and are dark red-brown often with foci of hemorrhage, necrosis, and cystic degeneration. Microscopically, tumors generally exhibit a characteristic neuroendocrine pattern. Prediction of the clinical behavior can be challenging unless metastases are present.7 Nuclear pleomorphism is not reliable as a determinant of malignancy. Hematogenous or lymphatic metastases to the liver, lymph node, lung, and bone have been reported in humans. Reported cardiovascular changes secondary to the effects of elevated catecholamine levels include cardiomyocyte necrosis, interstitial myocardial fibrosis, myocarditis, and arteriosclerosis.7 Histochemical staining and ultrastructural analysis to detect neurosecretory granules aid in the diagnosis of pheochromocytoma. Immunoreactivity is expected for catecholamines, chromogranin, catecholamine-synthesizing enzymes, neuron-specific enolase, synaptophysin, and opioid peptides (met-enkephalin, leu-enkephalin, beta-endorphin, dynorphin B).7 In humans, pheochromocytomas may carry germline or, less frequently, somatic mutations in multiple genes.2,7 Several familial tumor syndromes such as multiple endocrine neoplasia type 2 (MEN-2), type 1 neurofibromatosis, and von Hippel-Lindau syndrome may include pheochromocytoma. Genetic forms tend to occur at a younger age and may be multifocal.7 Germline mutation of the succinate dehydrogenase complex, subunit B (SDHB) gene is considered a strong risk factor for malignancy.2 GATA binding protein 3 (GATA3), a transcription factor, is used immunohistochemically to identify pheochromocytomas and is used to differentiate them from tumors exhibiting similar morphologic features.6
This report describes seven pheochromocytomas diagnosed in Old World primates at necropsy, affecting five rhesus macaques (Macaca mulatta) and two African green monkeys. The animals were maintained in research colonies at the Oregon National Primate Research Center (ONPRC), the Harlow Center for Biological Psychology (HCBP), and the Behavioural Science Foundation (BSF). Animal care and procedures were approved by the Institutional Animal Care and Use Committee at the ONPRC and HCBP and were conducted to ensure compliance with the U.S. Animal Welfare Act and other prevailing statutes, regulations and guidelines. Animal care and use at the BSF was approved by the Animal Care Committee of the BSF acting under the auspices of the Canadian Council on Animal Care. Complete necropsies were performed and representative tissues were collected for microscopic examination. Tissues were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 5 microns, and stained with hematoxylin and eosin; two were stained with the Churukian-Schenk method for argyrophil granules. Tissue microarrays were constructed with tissue from each case and immunohistochemistry for chromogranin A, met-enkephalin, beta-endorphin, melan-A and inhibin was performed (Supplemental Methods 1). Separately, immunohistochemistry was performed for synaptophysin, chromogranin A, GATA3, and SDHB, on case 7 (Supplemental Table 1 and Supplemental Methods 2). Supplemental material may be viewed on the journal website.
Case summaries are presented in Table 1. All neoplasms were unilateral with four affecting the right adrenal gland, two affecting the left adrenal gland, and one not specified (case 4). Adrenal gland enlargement was observed grossly in five cases (1,2,5,6,7) and ranged from 3.0-4.5 cm in greatest dimension. Affected glands were distorted and exhibited a variegated appearance with foci of hemorrhage and necrosis (Fig. 1). On microscopic examination, neoplastic cells often replaced the medulla and compressed the cortex; tumors were composed of polygonal cells arranged in nests, packets, and cords supported by a fine, fibrovascular stroma (Fig. 2). Cell borders were distinct with a moderate to abundant, granular, eosinophilic to basophilic cytoplasm. The nucleus was irregularly round with lightly stippled chromatin and a solitary, variably distinct nucleolus. Anisocytosis and anisokaryosis ranged from mild to marked. Occasional cells had giant, irregular, hyperchromatic nuclei (cases 1,5,6,7) or multiple (up to four) nuclei (cases 5,6,7). Cytoplasmic invagination was frequent in case 7 (Fig. 2, inset). Mitoses ranged from ≤1 to 3 per 10 400X fields. Variably sized, blood-filled spaces were present in all but case 3. Metastatic pheochromocytoma was observed in the lungs of two rhesus macaques (cases 1,5; Fig. 3) and portal vein invasion occurred in one African green monkey (case 6).
Table 1.
Signalment, Clinical and Pathologic Findings in Seven Old World Primates with Pheochromocytoma.
| Case | Species1 | Age2 | Sex | Facility4 | Reason for death | Diagnosis | Co-morbid conditions |
|---|---|---|---|---|---|---|---|
| 1 | M. mulatta | 20 Y | F | ONPRC | Gastric outflow obstruction | Right adrenal gland, pheochromocytoma with pulmonary metastasis | Aspiration of gastric contents; renal tubular necrosis |
| 2 | M. mulatta | >31 Y | F | ONPRC | Poor anesthetic recovery with respiratory compromise | Right adrenal gland, pheochromocytoma, benign | Chronic interstitial pneumonia, prior lung mite infection; ovarian granulosa cell tumor; endometrial polyp; cardiovascular lesions; mammary gland adenoma |
| 3 | M. mulatta | 9 Y | M | ONPRC | Reactive stifle arthritis | Right adrenal gland, pheochromocytoma, benign | Chronic typhlocolitis |
| 4 | M. mulatta | >18 Y | M | ONPRC | Weight loss, utilized for tissue distribution | Adrenal gland, left or right not specified, pheochromocytoma, benign | Chronic colitis; submandibular salivary gland mixed tumor, infarcted; thyroid C cell hyperplasia and adenoma; active lung mite infection; subacute cholecystitis |
| 5 | M. mulatta | 17 Y | F | HCBP | Spontaneous death after birth of healthy infant | Left adrenal gland, pheochromocytoma with pulmonary metastasis | Undelivered placenta; right adrenocortical hyperplasia |
| 6 | C. aethiops | Adult | U3 | BSF | Not recorded | Right adrenal gland, pheochromocytoma with portal vein invasion | Not recorded |
| 7 | C. aethiops | 9 Y | F | BSF | Facial malignant melanoma | Left adrenal gland, pheochromocytoma, benign | Mild chronic, interstitial nephritis, glomerular sclerosis and obsolescence |
M. mulatta, Macaca mulatta, rhesus macaque; C. aethiops, Chlorocebus aethiops, African green monkey
Y, years
U, unknown
ONPRC, Oregon National Primate Research Center, Beaverton, OR; HCBP, Harlow Center for Biological Psychology, Madison, WI; BSF, The Behavioral Science Foundation, St. Kitts, West Indies
Figure 1.
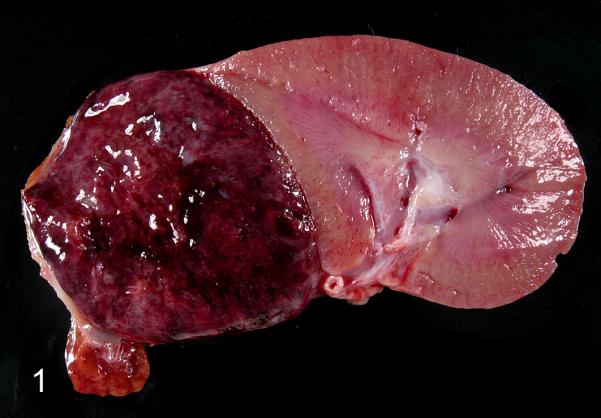
Pheochromocytoma, adrenal gland, rhesus macaque, case 1. On cross-section, the pheochromocytoma is heterogeneous, replaces the adrenal gland, and compressed the cranial pole of the kidney.
Figure 2.
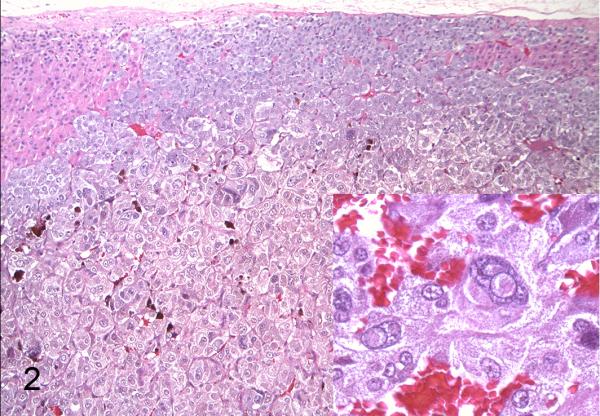
African green monkey, case 7. The tumor compresses the cortex, approaches the capsule, and consists of polygonal cells arranged in nests and packets separated by a fine, fibrovascular stroma. Anisokaryosis is marked. Inset: Karyomegalic cells with cytoplasmic invagination. Hematoxylin and eosin (HE).
Figure 3.
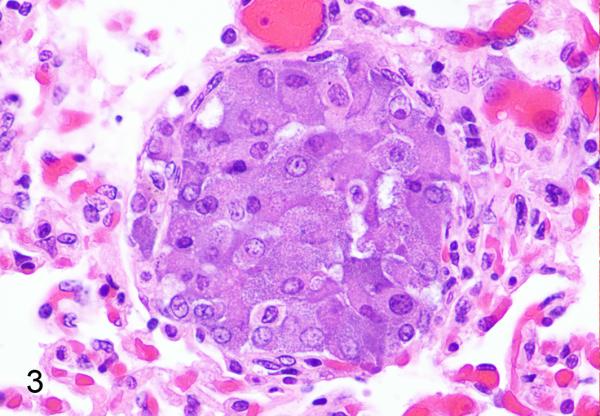
Metastatic pheochromocytoma, lung, rhesus macaque, case 5. HE. A nest of neoplastic cells expands the pulmonary vasculature.
The Churukian-Schenk method demonstrated myriad brown to black cytoplasmic granules in the two cases evaluated (cases 1,2). Immunohistochemically, neoplastic cells of all cases exhibited cytoplasmic labeling for chromogranin A and met-enkephalin and six cases were positive for beta-endorphin (cases 1-5,7; Supplemental Table 2). Adjacent adrenocortical tissue did not label with these markers. Neoplastic cells failed to label with inhibin and melan-A, whereas adrenocortical cells labeled well. In case 7, neoplastic cells had diffuse intense immunopositivity for chromogranin A (Fig. 4) and variably intense labeling for synaptophysin, while reactivity for melan-A was limited to adrenocortical cells. GATA3 expression was not detected in case 7; SDHB expression was intact.
Figure 4.
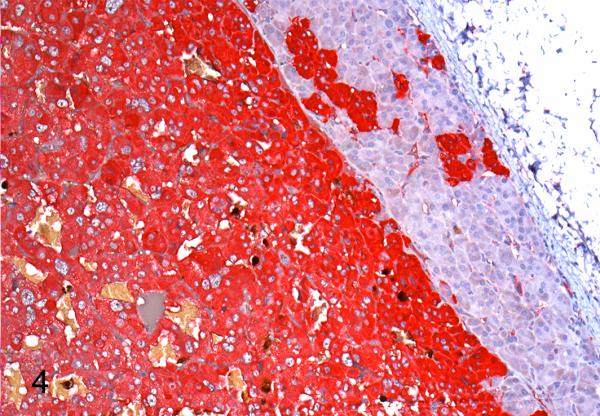
Pheochromocytoma, adrenal gland, African green monkey, case 7. Neoplastic tissue is strongly and diffusely immunoreactive for chromogranin A. The adjacent adrenocortical tissue does not label with this marker.
Available tissues were examined microscopically for cardiovascular abnormalities to help identify catecholamine-secreting pheochromocytomas. Mild to moderate myocardial fibrosis (cases 2,7), aortic atherosclerosis (cases 1,2) with mineralization (case 2), mild intimal sclerosis and fibromyxomatous medial change and fragmentation of the elastic lamina of intrarenal arteries (cases 1,2), afferent glomerular arteriolar changes including hyperplasia of the tunica media (cases 1,2,5,6), intimal sclerosis (case 2), and minimal splitting of the internal elastic lamina (case 5) were observed. Systemic arteriosclerosis affecting the kidneys, heart, lung, gastrointestinal tract, mesentery, uterus, right adrenal gland and pancreas was characterized by hyalinosis of vessel walls and variable medial hyperplasia, hypertrophy, and fibrosis in case 7. No vascular lesions were identified in cases 3 or 4.
Few pheochromocytomas have been reported in Old World primates.1,3,4,5,8,9,10 Most were identified as incidental lesions at necropsy, their endocrine activity unknown. Malignant pheochromocytomas have not been reported. Three retrospective studies of nonhuman primate neoplasms include eight pheochromocytomas in macaques.4,5,8,9 One report described an angiomatous pheochromocytoma in an aged rhesus macaque with a history of chronic heart disease.10 Pheochromocytomas have been reported in six baboons (Papio cynocephalus) and a patas monkey (Erythrocebus patas).1,3,4 Cardiovascular lesions noted in the rhesus macaque diagnosed with an angiomatous pheochromocytoma included cardiomyofiber atrophy, hypertrophy, and lysis with associated inflammation and replacement fibrosis.10 Based on these findings, the authors presumed a functional pheochromocytoma with catecholamine excess and a consequent cardiotoxicity. Clinical signs of hypertension were observed in most of the baboons diagnosed with pheochromocytomas at necropsy; pulmonary congestion with increased heart rate and dyspnea were exhibited by four animals.3 Cardiovascular histologic findings were not discussed.
Diagnosis for all cases in this series was established by identification of characteristic microscopic features on routine hematoxylin and eosin staining and was supported by special histochemical and immunohistochemical staining. The Churukian-Schenk method demonstrated argyrophilic granules consistent with neuroendocrine granules in the two cases evaluated. Neoplastic cells were positive in all cases for chromogranin A confirming the neuroendocrine nature of the tumors. Additionally, neoplastic tissue was immunoreactive for opioid peptide markers produced by the adrenal medulla: metenkephalin in all cases and beta-endorphin in six of seven cases. GATA3 has been shown to be a useful marker for pheochromocytomas in humans with one study reporting immunohistochemical positivity in 20/21 tumors.6 In contrast, it was not expressed in the one pheochromocytoma tested (case 7). Intact SDHB expression in this same tumor suggests that there was no germline mutation of SDHB.
As the pheochromocytomas in this series were identified at necropsy, no antemortem hormonal assessments were performed to confirm excessive catecholamine production. However, cardiovascular lesions were variably present in five cases and included regional and systemic arteriosclerosis, myocardial fibrosis, and aortic atherosclerosis. These morphologic changes suggest hypertension and possible catecholamine hypersecretion, particularly in the relatively young, nine-year-old African green monkey (case 7) with systemic arteriosclerosis and myocardial fibrosis. Cardiovascular changes observed in three of the rhesus macaques are likely multifactorial. The average age of these affected animals was 22.9 years; thus age-associated vascular changes and stress were considered as potential contributing factors to the cardiovascular lesions as well as the possibility of elevated catecholamines. The spontaneous death in case 5 was attributed to the combination of presumptively high circulating catecholamines from the pheochromocytoma and high circulating glucocorticoids at parturition.
In this series, an ovarian granulosa cell tumor (case 2) and thyroid gland parafollicular cell (C cell) adenoma and multifocal C cell hyperplasia (case 4) were concurrently diagnosed, suggesting the possibility of multiple endocrine neoplasia-like syndrome. Multiple endocrine neoplasms including adrenal carcinoma, pituitary adenoma, parathyroid adenoma, and ovarian granulosa cell tumor, have been associated with pheochromocytomas in baboons.1,3 MEN-2, a heritable condition caused by mutation of the receptor tyrosine kinase proto-oncogene (RET), is associated with three primary types of tumors in humans: pheochromocytoma, medullary thyroid carcinoma, and parathyroid tumor.7 None of our cases match the current definition of MEN-2, and mutations in RET have not been described in nonhuman primates.
Histologic diagnosis of pheochromocytoma in this case series was straightforward on routine evaluation of hematoxylin and eosin-stained sections. Immunohistochemical panels were useful in confirming adrenal tumors of medullary origin. Cardiovascular lesions suggesting possible hypertension secondary to a functional pheochromocytoma were noted in five of the seven cases. This case series should heighten awareness of pheochromocytomas in Old World primates and reviews the associated signs that may be detected during clinical assessment. It adds seven cases of spontaneous pheochromocytoma to the existing literature and documents the occurrence of this neoplasm in two African green monkeys, a species in which pheochromocytoma has not been previously reported. To the authors’ knowledge, this is also the first report of malignant pheochromocytoma in Old World primates.
Supplementary Material
Acknowledgements
We thank Dr. Amy Beierschmitt for providing cases 6 and 7 in this series, Natalie Keirstead for her microscopic evaluation of case 6, Dr. Josepha DeLay for immunohistochemical analysis of tumor 7 and Dr. Michelle Hirsch for performing and interpreting SDHB and GATA3 immunohistochemistry on tumor 7.
This work was supported in part by grant P51OD011092 from the National Institutes of Health.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship and or publication of this article.
Case 5 was presented at the 40th Annual Workshop of the Association of Primate Veterinarians, November 2012, St. Paul, Minnesota. Case 7 was presented as a poster at the combined 65th Annual Meeting of the American College of Veterinary Pathologists (ACVP) and 49th Annual Meeting of the American Society for Veterinary Clinical Pathology, November 2014, Atlanta, Georgia. An oral presentation of Case 1 was given during the Primate Pathology Post-Meeting Workshop of the same meeting.
References
- 1.Cianciolo E, Butler SD, Eggers JS, et al. Spontaneous neoplasia in the baboon (Papio spp.). J Med Primatol. 2007;36:61–79. 2. doi: 10.1111/j.1600-0684.2006.00202.x. [DOI] [PubMed] [Google Scholar]
- 2.Gill AJ, Benn DE, Chou A, et al. Immunohistochemistry for SDHB triages genetic testing of SDHB, SDHC, and SDHD in paraganglioma-pheochromocytoma syndromes. Human Pathology. 2010;41:805–814. doi: 10.1016/j.humpath.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 3.Guardado-Mendoza R, Dick EJ, Jimeniz-Ceja LM, et al. Spontaneous pathology of the baboon endocrine system. J Med Primatol. 2009;38:383–389. doi: 10.1111/j.1600-0684.2009.00384.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lowenstine LJ. Neoplasms and proliferative disorders in nonhuman primates. In: Benirschke K, editor. The Road to Self-Sustaining Populations. Springer-Verlag; New York, NY: 1986. pp. 781–814. [Google Scholar]
- 5.McClure HM. Neoplastic diseases in nonhuman primates: literature review and observations in an autopsy series of 2176 animals. In: Montali RJ, Migaki G, editors. The Comparative Pathology of Zoo Animals. Smithsonian Institution Press; Washington, DC: 1979. pp. 549–565. [Google Scholar]
- 6.Nonaka D, Wang BY, Edmondson D, et al. A Study of GATA3 and PHOX2b expression in tumors of the autonomic nervous system. Am J Surg Pathol. 2013;37(8):1236–1241. doi: 10.1097/PAS.0b013e318289c765. [DOI] [PubMed] [Google Scholar]
- 7.Rosai J. Adrenal gland and other paraganglia. In: Rosai J, editor. Rosai and Ackerman's Surgical Pathology. 10th ed. Vol. 1. Mosby; Edinburgh, U.K.: 2011. pp. 1057–1100. [Google Scholar]
- 8.Seibold HR, Wolf RH. Neoplasms and proliferative lesions in 1065 nonhuman primate necropsies. Lab Anim Sci. 1973;23(4):533–539. [PubMed] [Google Scholar]
- 9.Squire RA, Goodman DG, Valerio MG, et al. Tumors. In: Benirschke K, Garner FM, Jones TC, editors. Pathology of Laboratory Animals. II. Springer-Verlag; New York, New York: 1978. p. 1234. [Google Scholar]
- 10.Vogel P, Fritz D. Cardiomyopathy Associated with Angiomatous Pheochromocytoma in a Rhesus Macaque (Macaca mulatta). Vet Pathol. 2003;40:468–473. doi: 10.1354/vp.40-4-468. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


