Abstract
Objective
Attention-deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder that negatively affects different areas of life. We aimed to evaluate the associations between the Val66Met polymorphism of brain-derived neurotrophic factor (BDNF) and ADHD and to assess the effect of the BDNF polymorphism on the neurocognitive profile and clinical symptomatology in ADHD.
Methods
Two hundred one ADHD cases and 99 typically developing subjects (TD) between the ages of 8 and 15 years were involved in the study. All subjects were evaluated using a complete neuropsychological battery, Child Behavior Checklist, the Teacher's Report Form (TRF) and the DSM-IV Disruptive Behavior Disorders Rating Scale-teacher and parent forms.
Results
The GG genotype was significantly more frequent in the patients with ADHD than in the TD controls, and the GG genotype was also significantly more frequent in the ADHD-combined (ADHD-C) subtype patients than in the TDs. However, there were no significant associations of the BDNF polymorphism with the ADHD subtypes or neurocognitive profiles of the patients. The teacher-assessed hyperactivity and inattention symptom count and the total score were higher, and the appropriately behaving subtest score of the TRF was lower in the GG genotypes than in the GA and AA (i.e., the A-containing) genotypes.
Conclusion
We found a positive association between the BDNF gene Val66Met polymorphism and ADHD, and this association was observed specifically in the ADHD-C subtype and not the ADHD-predominantly inattentive subtype. Our findings support that the Val66Met polymorphism of BDNF gene might be involved in the pathogenesis of ADHD. Furthermore Val66Met polymorphism of BDNF gene may be more closely associated with hyperactivity rather than inattention.
Keywords: Attention-deficit hyperactivity disorder, Brain-derived neurotrophic factor, Gene polymorphism
INTRODUCTION
Attention-deficit hyperactivity disorder (ADHD) is a common and heritable neurodevelopmental disorder that affects 8% to 12% of school-aged children.1 ADHD is characterized by inattention, hyperactivity, and impulsivity and leads to many problems related to social, academic and behavioral areas of life. The etiology of ADHD is not completely understood, but the estimated heritability is approximately 75%, and thus it is generally thought to have a having a genetic basis.2 Dysregulations of the catecholaminergic neurotransmitter systems are thought to play important roles in ADHD, and the dopamine and norepinephrine systems have been studied extensively.3
Brain-derived neurotrophic factor (BDNF) is a member of the neurotrophin superfamily and plays critical roles in the serotonergic,4 glutamatergic,5 noradrenergic,6 and dopaminergic7 neurotransmitter systems, which are important for the pathogenesis of the ADHD. Chen et al.8 reported that the plasma concentrations of BDNF are significantly lower in patients than in healthy controls, although these authors found that the BDNF gene polymorphism does not affect plasma BDNF levels.
Polymorphisms of the human BDNF gene, which is located on chromosome 11p13, have been studied in several psychiatric disorders. The most frequent is the Val66Met (rs6265) guanine (G)-to-adenine (A) single-nucleotide polymorphism (SNP) at nucleotide 196 that results in an amino acid substitution of methionine (Met) for valine (Val).9 This polymorphism impairs the packaging of proBDNF and results in reduced depolarization-induced secretion of BDNF from neurosecretory cells and cortical neurons. However, in the same study, the authors showed that the constitutive secretion of mature BDNF is not affected by this polymorphism and that the basal release of BDNF continues.10 The Val (G) allele of the Val66Met polymorphism has been reported to be associated with bipolar disorder and schizophrenia in previous studies.11,12 Additionally the Met allele of the Val66Met polymorphism has been reported to be associated with the restrictive type of anorexia nervosa.13 Sen et al.14 reported that the Met allele has a protective effect in depression.
Previous studies have reported inconsistent results regarding the relationship between the BDNF gene and ADHD. Some of the studies have reported a positive association between the Val66Met polymorphism of BDNF and ADHD. One of these studies was conducted by Lanktree et al.15 who reported a positive association of the Val allele with ADHD in adult patients. In another study that consisted of a family based-sample of 341 ADHD probands also found a relationship between the Val allele and ADHD.16 In contrast to these results, some studies have reported no association between the BDNF Val66Met polymorphism and ADHD.17,18,19
Studies of the cognitions related to ADHD have shown that ADHD children often perform poorly on tasks of working memory and exhibit impairments in spatial and verbal components.20,21 Lee et al.22 reported no association between BDNF alleles and working memory abilities in their sample of ADHD families. Mice with an inactivated BDNF gene have exhibit significantly more locomotor activity when stressed than normal mice.23 Additionally, animal studies have revealed that BDNF is critical for learning and working memory.24,25
In the present study, we hypothesized that this BDNF gene polymorphism might be involved in the pathogenesis of ADHD and influence clinical presentations including neurocognition and symptoms. Therefore, we aimed to evaluate the associations between the Val66Met polymorphism of the BDNF gene and ADHD and to assess the effects of this BDNF polymorphism on the neurocognitive profile and clinical symptomatology of ADHD.
METHODS
Study design
The present study included 201 ADHD subjects [101 ADHD-combined (ADHD-C) and 100 ADHD-predominantly inattentive type (ADHD-I)] and 99 typically developing (TD) subjects aged 8 to 15 years who were consecutively assessed in the child and adolescent psychiatry clinic of Ege University from December 2011 to March 2013. The parents provided written informed consent. The Ethics Committee of Pamukkale University approved the study protocol.
Participants
The inclusion criteria for both the ADHD and TD subjects were the following: 1) an estimated intelligence quotient (IQ) greater than 80, 2) no history of head injury with loss of consciousness, 3) no neurological or other serious medical disease, 4) no prior use of stimulants, and 5) no use of psychotropic medication within the last six months. For the ADHD subjects, the additional criterion of a lack of history of any psychiatric disorder other than oppositional defiant disorder (ODD) was also included. For the TD controls, the additional criterion of a lack of any psychiatric disorder was included.
Diagnostic procedures
Initially, the families and teachers completed the Child Behavior Checklist (CBCL),26 the Teacher's Report Form (TRF),26 and the DSM-IV Disruptive Behavior Disorders Rating Scaleteacher and parent forms.27 The subjects with inattentive scores that were 1 SD greater than the age norms for these scales were invited to participate in the diagnostic portion of the study. This procedure was performed to guarantee that the inattentive symptoms were adequately represented in all of the ADHD types assessed. Thereafter, a semi-structured interview (i.e., the Kiddie-Schedule for Affective Disorders and Schizophrenia, present and life time version-K-SADSPL) was applied to parents by a senior child psychiatry resident.28 Additionally, we assessed the participants estimated IQs using the vocabulary and block design subtests of the WISC-R.
Two experienced child psychiatrists who were blinded to the first diagnostic assessment conducted confirmatory second diagnostic interviews with the participants with positive ADHD diagnoses in the first KSDAS-PL interview. The parents and subjects were also interviewed according to the KSADS-PL. "A best estimate procedure" was used to determine the final diagnoses.29 Three subjects refused to attend the second diagnostic interview, and disagreements the between interviewers regarding the ADHD types/comorbid diagnoses occurred in 10 cases. Thirteen subjects were excluded from the second diagnostic part of the study. Unrelated healthy controls were also recruited from the same community and were matched as well possible to the ADHD patients in terms of age and gender. The exact same diagnostic procedure was applied for the assessments of the controls. However, we required that the TD subjects exhibited inattentive scores that were one standard deviation below the means for the child's age on the CBCL, TRF, and ADHD-RS-IV scales. Consequently, 101 ADHD-C, 100 ADHD-I type and 99 TD subjects were included in the study.
Neuropsychological evaluation
All cases in the study were assessed using a standardized neuropsychological battery (CNS-VS) that included tests addressing the neuropsychological constructs known to be associated with ADHD. The battery included tests that measured verbal and visual memory, finger tapping, digit symbol coding, the Stroop test, a test of shifting attention and the continuous performance test (CPT). Six main scores, including the neurocognitive index, total memory, reaction time, complex attention, cognitive flexibility, and psychomotor speed, were obtained.30
DNA extraction and genotype determination
DNA was extracted from saliva samples for genotyping. Genotypes were determined using a TaqMan™ fluorogenic 5'-nuclease assay with TaqMan Probes. All reactions were carried out following the manufacturer's protocol. Primer Express 3.0 (Applied Biosystems) was used to design both the PCR primers and the TaqMan probes. For BDNF Gene Val66Met (rs6265) polymorphisms, custom made primers and probes are as follows: Forward Primer 5'-AGGCAGGTTCA AGAGGCTTGA-3', Reverse Primer 5'-TTCTGGTCCTCAT CCAACAGCT-3', Probe G: 5'-Yakima Yellow-TGA(pdC) A(pdC)TTT(pdC)GAA(pdC)ACGTGATA-BHQ-1-3', Probe A: 5'-FAM-TGA(pdC)A(pdC)TTT(pdC)GAA(pdC)A(pdC) ATGATA-BHQ-1-3'. Single nucleotide polymorphism amplification assays were performed according to the manufacturer's instructions.In brief, 25 µL of reaction solution containing 30 ng of DNA was mixed with 12.5 µL of 2X TaqMan Universal PCR Master Mix (Applied Biosystems) and 900 nmol of each primer, 200 nmol of each probe. Reaction conditions consisted of preincubation at 60℃ for 1 minute and at 95℃ for 10 minute, followed by 40 cycles at 95℃ for 15 second and at 60℃ for 1 minute. Amplifications and analysis were performed in an ABI Prism 7500 Real-Time PCR System (Applied Biosystems), using the SDS 2.0.6 software for allelic discrimination (Applied Biosystems).
Statistical analyses
The data analyses were performed using the statistical package for the SPSS 17.0 version for PC, and p-values <0.05 were considered statistically significant. Chi-square tests were applied to the categorical variables, and t-tests were applied to the continuous variables for the genotype group comparisons. We performed the multivariate analysis of variance (MANOVA) by assigning the age and gender as covariates to control the effects of these parameters on ADHD-related symptoms. Posthoc comparisons were carried out using Bonferroni correction and lower than 0.025 p values were accepted as statistically significant for MANOVA because of the presence of two comparison genotype groups.
RESULTS
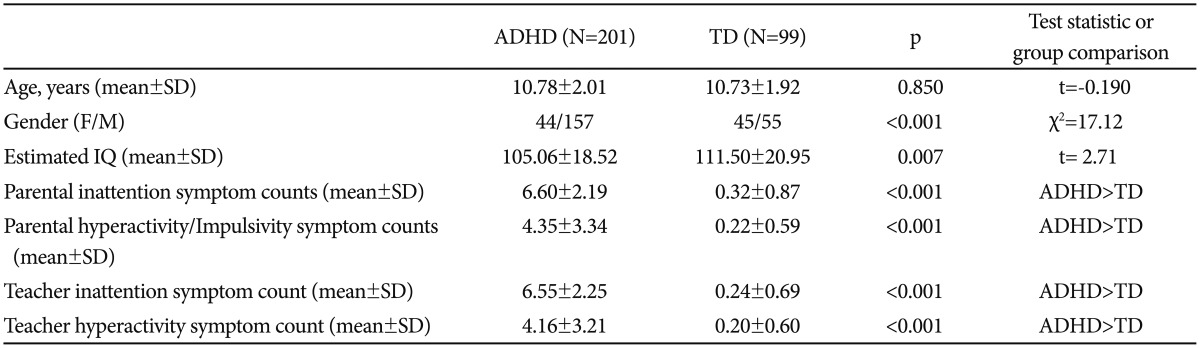
Two hundred and one patients with ADHD and 99 TD subject were involved the present study. The mean age of the ADHD group was 10.78±2.01 years, and the mean age of the TD group was 10.73±1.92 years. There was no difference in mean age (t=-0.190, p=0.85) between the groups. Although male predominance was provided in both groups of ADHD and TD, there was a significant difference between the patients and TD groups in terms of gender distribution. Sociodemographic and clinical characteristic of the participants were shown in Table 1.
Table 1. Socio-demographic characteristics and ADHD rating scale scores.
Data are presented as the means±standard deviations (SD) or n, as appropriate. ADHD: attention-deficit hyperactivity disorder, TD: typically developing children, IQ: intelligence quotient
Genetic assessments
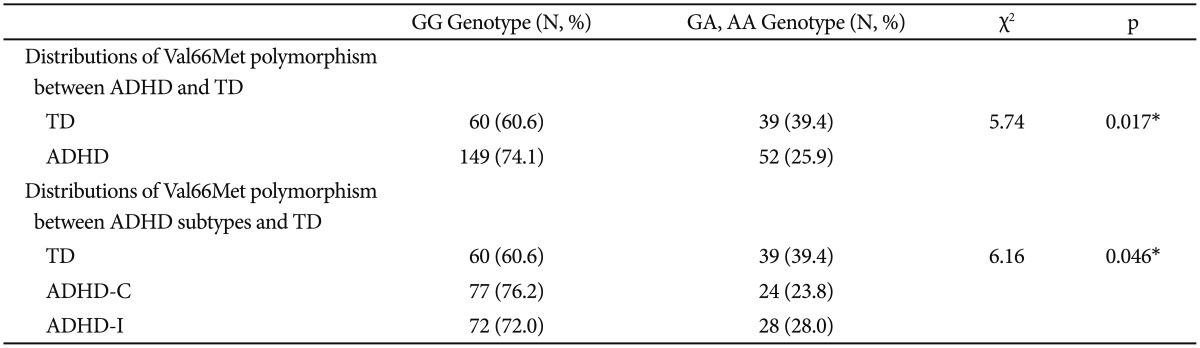
The genotype determinations revealed that only 10 subjects (3.3%) among both the ADHD and TD groups had the AA genotype. Therefore, the sample was grouped into GG genotypes and A-containing (GA+AA) genotypes (Table 2). The data analyses revealed significant differences in the genotypic frequency distributions of the Val66Met polymorphism between the ADHD and TD groups. The GG genotype was significantly more frequent among the patients with ADHD than the TD participants (χ2=6.16, p=0.017). Additionally, the genotype frequency distributions of Val66Met polymorphism between the TD groups, ADHD-C subtype and ADHD-I subtype groups were found to be significantly different (χ2=6.16, p=0.046). The GG genotype was significantly more frequent in the ADHD-C group than the TD group. However, the frequency distributions of the GG genotype and A-containing genotypes between the subtypes of ADHD did not differ (Table 2).
Table 2. Distributions of Val66Met polymorphism.
Chi-square test. *statistically significance p<0.05. ADHD: attention-deficit hyperactivity disorder, ADHD-C: ADHD-combined type, ADHDI: ADHD predominantly inattentive type, TD: typically developing subjects
Assessments of ADHD-related symptoms
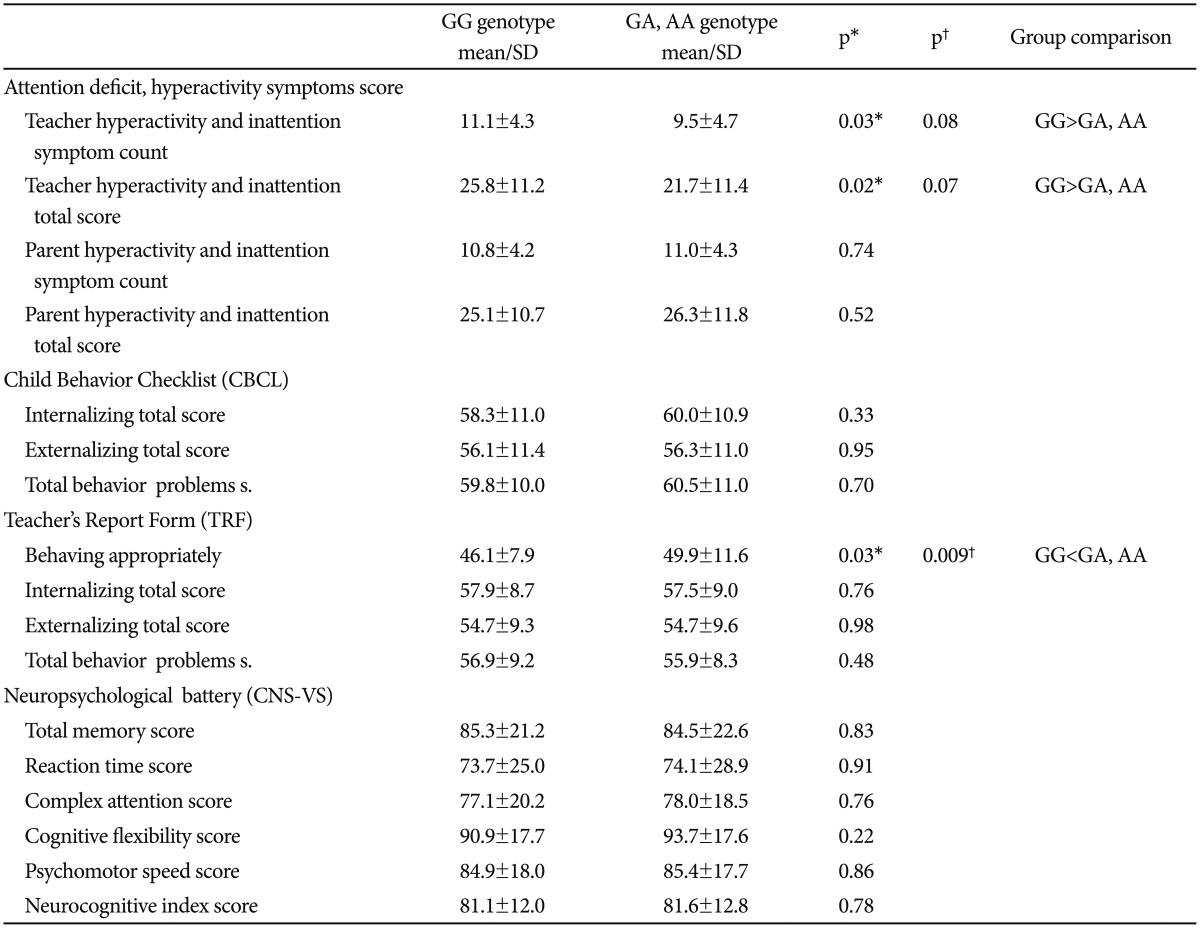
In our study, the teacher-rated hyperactivity and inattention symptom counts and total scores were higher among the GG genotype group than the A-containing genotype group (p=0.031, p=0.025). Regarding the CBCL scores, there were no significant differences between the Val66Met polymorphism subgroups. However, the appropriate behavior score of the TRF was significantly lower in the GG genotype group than the A-containing genotypes group (p=0.031) (Table 3). When we performed the multivariate analysis of variance (MANOVA) by assigning the age and gender as covariates to control the effects of these parameters on the ADHD-related symptoms, and we found that the appropriate behavior score of the TRF remained to be statistically significantly different between the polymorphism groups in patients with ADHD (λ=.957, F=2.92, p=0.009) (Table 3).
Table 3. Comparison of genotype groups in terms of symptoms and neuropsychological findings in patients.
*independent simples t-test, statistically significance p<0.05, †multivariate analysis of variance (MANOVA) by assigning the age and gender as covariates (Post-hoc comparisons were carried out using Bonferroni correction and less than 0.025 p values were accepted as statistically significant)
Neuropsychological findings
The neuropsychological performances, including verbal and visual memory, finger tapping, digit symbol coding, the Stroop test, the test of shifting attention, and the vocabulary and block design subtests of the WISC-R and the CPT of the Val66Met polymorphism groups was not found to be significantly different (p>0.05). Additionally, the neuropsychological battery scores did not differ within the entire group (ADHD and TD) or within the TD group according to the BDNF polymorphism (p>0.05) (Table 3).
DISCUSSION
In the present study, we found a positive association between the BDNF gene Val66Met SNP polymorphism and ADHD that the GG genotype was more frequent in the patients with ADHD and the ADHD-C subtype than in the TD group. BDNF is a member of the neurotrophins family that has a crucial role in neuronal developmental process, neuroplasticity and neuronal function.31 Thus investigation of effects of BDNF gene polymorphism on ADHD which is a neurodevelopmental disorder is great importance. The Val66Met polymorphism of BDNF can affect the intracellular trafficking and can reduce depolarization-induced secretion of the BDNF. However, Chen et al.10 reported that the mature BDNF is not affected by the polymorphism and its basal release continues. The neuropsychiatric studies suggested that the levels of circulating BDNF which is reflect the level of braintissue BDNF32 was effected by the polymorphism. Reduced BDNF levels were reported in Met carriers patients with depression33 and schizophrenia.34 Conversely, studies demonstrated that the healthy human with Met allele carriers have increased circulating BDNF levels.35,36 Li et al.37 indicated that the females who were Met/Met genotype carriers with ADHD had a tendency of increased plasma BDNF levels than Val allele carriers however the difference was not statistically significant. Taken together these data and our result, it may be suggested that the Val allele may contribute to pathogenesis of ADHD.
The relationship between ADHD and the BDNF gene has been investigated in many studies. In accordance with our findings, Kent et al.16 reported a positive association with the Val (G) allele, particularly when this allele is paternally transmitted, in children with ADHD. Similarly, Aureli et al.37 reported that the G allele of the Val66Met polymorphism is significantly associated with ADHD. In another study conducted by Li et al.,38 a higher frequency of the Val allele was found in females with ADHD than in controls. Another study conducted by Lanktree et al.,15 which has currently only been reported in abstract form, described evidence for the involvement of the Val allele of the Val66Met polymorphism in both case control and trio analyses of adult ADHD patients. In light of these results, it may be suggested that there is a positive association between the BDNF polymorphism and ADHD and moreover that this association is more prominent in terms of hyperactivity than inattention. Furthermore, we speculated that the Val allele of the Val66Met polymorphism is a risk factor for ADHD. Conversely, in some previous studies, no evidence of an association between the Val66Met polymorphism and ADHD has been found.17,18,19,39,40 These contradictory findings may be due to genetic and clinical heterogeneity, the use of different diagnostic practices, differences of in the ADHD subtype distributions across the studies, differences in comorbidities, and the insufficient sample sizes of the studies.40 It is possible that the above mentioned reasons have masked a real association between this disorder and the BDNF gene polymorphism. Additionally, epigenetic factors or mechanisms, such as environmental factors, abnormal imprinting of the genes, and neuroanatomically tissue-specific expression patterns, might affect the pathogenesis of ADHD.16 A better understanding of the relationships of variations in genes, such as BDNF, with the pathogenesis of ADHD may require analyses of gene-gene and gene-environment interactions.
The distributions of the Val66Met polymorphism did not differ between the ADHD-C and ADHD-I subtypes in our study. Similarly, in a meta-analysis, Sánchez-Mora et al.40 reported that there is no statistically significant effect of the Val66Met polymorphism of the BDNF gene on ADHD subtypes. This finding may indicate that the contribution of BDNF to ADHD is not subtype-specific40 or, alternatively, that the small sample sizes of the studies, including our own, have been insufficient to uncover an association.
In our study, the teacher-rated hyperactivity and inattention symptom counts and total scores were higher, and the appropriate behavior scores of the TRF were significantly lower in the GG genotype group than in the A-containing genotype groups. However the multivariate analysis of variance by assigning the age and gender as covariates have shown that appropriate behavior score of the TRF remained to be statistically significantly lower in GG genotype group of patients with ADHD. As mentioned previously, the GG genotype was observed more frequently in the patients than the controls in the present study, which is consistent with the results of some of the previous studies. Therefore, it may be suggested that the GG genotype may have a role in the pathogenesis of ADHD and may also affect the clinical symptomatology of ADHD. There are few studies that have assessed BDNF polymorphisms and the symptoms related to ADHD. Consistent with our findings, Lee et al.22 reported that parents and teachers rate the inattention and hyperactivity/impulsivity scores of ADHD patients with G alleles higher than those of patients with A alleles, but this difference was not statistically significant. Conversely, Gadow et al.41 reported that the BDNF Met66-group (A alleles) elicited more severe parental ratings of ADHD symptoms. Bergman et al.,42 reported that the Met allele (A) of the Val66Met polymorphism is associated with the symptoms of ADHD at the ages 8–9 and that this effect on hyperactivity-impulsivity symptoms in ADHD persists at the ages of 13–14 years. These contradictory findings may be due to that fact that the symptomatology of ADHD is characterized by developmental heterogeneity.43 In contrast, differential genetic components may have a role in childhood ADHD with or without the remission of the phenotype across the life span.40
In animal studies, BDNF has been related to learning and memory functions.24,25 Eagan et al.44 reported that the Val66Met polymorphism of the BDNF gene may influence human hippocampal function and memory. For these reasons, we sought to evaluate the neurocognitive functions of children with ADHD according to the Val66Met polymorphism, but we were unable to find any differences between the polymorphism groups. We found only one study that investigated the relationship between the Val66Met polymorphism and cognitive functions in ADHD. This study was conducted by Lee et al.,22 and these authors reported that no association was found between BDNF alleles and working memory abilities in their sample of ADHD families. Foltynie et al.45 found that Met allele carriers performed better in the Tower of London test, which is a measure of planning ability that is used for Parkinson's disease. Conversely, Rybakowski et al.46 reported that the Met allele was associated with worse performance compared with the Val/Val genotype in the Wisconsin card sorting test, which is used to measure PFC function in bipolar patients. Similarly, studies performed in healthy populations have also obtained inconsistent results regarding hippocampal function. Two studies indicated decreased hippocampal activation in Met carriers.47,48 Another study reported increased hippocampal activation in Met carriers.49 One study, which supports our findings, found not differences between Val66Met groups in hippocampal activity during a face recognition task.50 These contradictory findings may be due to heterogeneity of the study sample diagnoses. Moreover, the differential effects of the BDNF polymorphism may potentially be explained by the complex interactions between BDNF and other candidate gene complexes related to cognitive functions.
There are several limitations to the present study. Our study included only two ADHD subtypes, i.e., ADHD-I and ADHD-C, because ADHD-H is rarely observed clinically. Future studies that include the ADHD-H subtype would provide more comprehensive information related the psychopathology of ADHD. We did not exclude ODD, and the potential presence of comorbid ODD in some patients may have created the confounding effects. Additionally, we assessed estimated IQ using only the vocabulary and block design subtests of the WISC-R. Thus, other potential ADHD neuropsychological domains were not assessed (e.g., delay aversion and RT variability). We did not match the ADHD and TD groups in terms of gender although we provided the male predominance in both groups. Finally, we used saliva samples as the DNA source; however, the use of peripheral blood samples could have increased the efficiency of the genotyping.
As a conclusion, in the present study, the GG genotype was more frequently observed in the ADHD and ADHD-C subtype patients than in the TDs. Consistent with these findings, the hyperactivity and inattention symptom counts and total scores were higher, and the appropriate behavior scores were lower in the subjects with GG genotypes than in those with A-containing genotypes. Consequently, our findings support the hypothesis that the Val66Met polymorphism of BDNF is involved in the pathogenesis of ADHD. Moreover, the polymorphism of BDNF may influence the clinic features of ADHD. Future studies with larger sample size are needed to clarify the relationship between BDNF Val66Met polymorphism and pathogenesis of ADHD.
References
- 1.Biederman J, Faraone SV. Attention-deficit hyperactivity disorder. Lancet. 2005;366:237–248. doi: 10.1016/S0140-6736(05)66915-2. [DOI] [PubMed] [Google Scholar]
- 2.Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, et al. Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2005;57:1313–1323. doi: 10.1016/j.biopsych.2004.11.024. [DOI] [PubMed] [Google Scholar]
- 3.Banaschewski T, Becker K, Scherag S, Franke B, Coghill D. Molecular genetics of attention-deficit/hyperactivity disorder: an overview. Eur Child Adolesc Psychiatry. 2010;19:237–257. doi: 10.1007/s00787-010-0090-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mossner R, Daniel S, Albert D, Heils A, Okladnova O, Schmitt A, et al. Serotonin transporter function is modulated by brain-derived neurotrophic factor (BDNF) but not nerve growth factor (NGF) Neurochem Int. 2000;36:197–202. doi: 10.1016/s0197-0186(99)00122-9. [DOI] [PubMed] [Google Scholar]
- 5.Carvalho A, Caldeira M, Santos S, Duarte C. Role of the brain-derived neurotrophic factor at glutamatergic synapses. Br J Pharmacol. 2008;153(Suppl 1):S310–S324. doi: 10.1038/sj.bjp.0707509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Traver S, Marien M, Martin E, Hirsch EC, Michel PP. The phenotypic differentiation of locus ceruleus noradrenergic neurons mediated by brain-derived neurotrophic factor is enhanced by corticotropin releasing factor through the activation of a cAMP-dependent signaling pathway. Mol Pharmacol. 2006;70:30–40. doi: 10.1124/mol.106.022715. [DOI] [PubMed] [Google Scholar]
- 7.Guillin O, Diaz J, Carroll P, Griffon N, Schwartz JC, Sokoloff P. BDNF controls dopamine D3 receptor expression and triggers behavioural sensitization. Nature. 2001;411:86–89. doi: 10.1038/35075076. [DOI] [PubMed] [Google Scholar]
- 8.Chen SL, Lee SY, Chang YH, Wang TY, Chen SH, Chu CH, et al. The BDNF Val66Met polymorphism and plasma brain-derived neurotrophic factor levels in Han Chinese heroin-dependent patients. Sci Rep. 2015;5:8148. doi: 10.1038/srep08148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cargill M, Altshuler D, Ireland J, Sklar P, Ardlie K, Patil N, et al. Characterization of single-nucleotide polymorphisms in coding regions of human genes. Nat Genet. 1999;22:231–238. doi: 10.1038/10290. [DOI] [PubMed] [Google Scholar]
- 10.Chen ZY, Jing D, Bath KG, Ieraci A, Khan T, Siao CJ, et al. Genetic variant BDNF (Val66Met) polymorphism alters anxiety-related behavior. Science. 2006;314:140–143. doi: 10.1126/science.1129663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Geller B, Badner JA, Tillman R, Christian SL, Bolhofner K, Cook EH., Jr Linkage disequilibrium of the brain-derived neurotrophic factor Val66-Met polymorphism in children with a prepubertal and early adolescent bipolar disorder phenotype. Am J Psychiatry. 2004;161:1698–1700. doi: 10.1176/appi.ajp.161.9.1698. [DOI] [PubMed] [Google Scholar]
- 12.Neves-Pereira M, Cheung JK, Pasdar A, Zhang F, Breen G, Yates P, et al. BDNF gene is a risk factor for schizophrenia in a Scottish population. Mol Psychiatry. 2005;10:208–212. doi: 10.1038/sj.mp.4001575. [DOI] [PubMed] [Google Scholar]
- 13.Ribasés M, Gratacòs M, Armengol L, de Cid R, Badía A, Jiménez L, et al. Met66 in the brain-derived neurotrophic factor (BDNF) precursor is associated with anorexia nervosa restrictive type. Mol Psychiatry. 2003;8:745–751. doi: 10.1038/sj.mp.4001281. [DOI] [PubMed] [Google Scholar]
- 14.Sen S, Nesse RM, Stoltenberg SF, Li S, Gleiberman L, Chakravarti A, et al. A BDNF coding variant is associated with the NEO personality inventory domain neuroticism, a risk factor for depression. Neuropsychopharmacology. 2003;28:397–401. doi: 10.1038/sj.npp.1300053. [DOI] [PubMed] [Google Scholar]
- 15.Lanktree M, Muglia P, Squassina A, Krinsky M, Jain U, Macciadi F, et al. A potential role for brain derived neurotrophic factor (BDNF) in adult ADHD. Abstract in Am J Med Gent Part B. 2004;130B:96–97. [Google Scholar]
- 16.Kent L, Green E, Hawi Z, Kirley A, Dudbridge F, Lowe N, et al. Association of the paternally transmitted copy of common Valine allele of the Val66Met polymorphism of the brainderived neurotrophic factor (BDNF) gene with susceptibility to ADHD. Mol Psychiatry. 2005;10:939–943. doi: 10.1038/sj.mp.4001696. [DOI] [PubMed] [Google Scholar]
- 17.Friedel S, Fontenla Horro F, Wermter AK, Geller F, Dempfle A, Reichwald K, et al. Mutation screen of the brain derived neurotrophic factor gene (BDNF): identification of several genetic variants and association studies in patients with obesity, eating disorders and attention-deficit/hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet. 2005;132B:96–99. doi: 10.1002/ajmg.b.30090. [DOI] [PubMed] [Google Scholar]
- 18.Brookes K, Xu X, Chen W, Zhou K, Neale B, Lowe N, et al. The analysis of 51 genes in DSM-IV combined type attention deficit hyperactivity disorder: Association signals in DRD4, DAT1 and 16 other genes. Mol Psychiatry. 2006;11:934–953. doi: 10.1038/sj.mp.4001869. [DOI] [PubMed] [Google Scholar]
- 19.Lee YH, Song GG. BDNF 196 G/A and COMT Val158Met polymorphisms and susceptibility to ADHD: a meta-analysis. J Atten Disord. doi: 10.1177/1087054715570389. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 20.Hervey AS, Epstein JN, Curry JF. Neuropsychology of adults with attention-deficit/hyperactivity disorder: a meta-analytic review. Neuropsychology. 2004;18:485–503. doi: 10.1037/0894-4105.18.3.485. [DOI] [PubMed] [Google Scholar]
- 21.Martinussen R, Hayden J, Hogg-Johnson S, Tannock R. A metaanalysis of working memory impairments in children with attentiondeficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2005;44:377–384. doi: 10.1097/01.chi.0000153228.72591.73. [DOI] [PubMed] [Google Scholar]
- 22.Lee J, Laurin N, Crosbie J, Ickowicz A, Pathare T, Malone M, et al. Association study of the brain derived neurotropic factor (BDNF) gene in attention deficit hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet. 2007;144B:976–981. doi: 10.1002/ajmg.b.30437. [DOI] [PubMed] [Google Scholar]
- 23.Rios M, Fan G, Fekete C, Kelly J, Bates B, Kuehn R, et al. Conditional deletion of brain-derived neurotrophic factor in the postnatal brain leads to obesity and hyperactivity. Mol Endocrinol. 2001;15:1748–1757. doi: 10.1210/mend.15.10.0706. [DOI] [PubMed] [Google Scholar]
- 24.Kiprianova I, Sandkuhler J, Schwab S, Hoyer S, Spranger M. Brain derived neurotrophic factor improves long-term potentiation and cognitive functions after transient forebrain ischemia in the rat. Exp Neurol. 1999;159:511–519. doi: 10.1006/exnr.1999.7109. [DOI] [PubMed] [Google Scholar]
- 25.Mizuno M, Yamada K, Olariu A, Nawa H, Nabeshima T. Involvement of brain-derived neurotrophic factor in spatial memory formation and maintenance in a radial arm maze test in rats. J Neurosci. 2000;20:7116–7121. doi: 10.1523/JNEUROSCI.20-18-07116.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Achenbach TM. Integrative Guide for the CBCL/4-18, YSR, and TRF Profiles. Burlington: Department of Psychiatry, University of Vermont; 1991. [Google Scholar]
- 27.Ercan ES, Amado S, Somer O, Çıkoğlu S. [Development of a test battery for the assessment of attention deficit hyperactivity disorder] Turk J Child Adoles Ment Health. 2001;8:132–144. [Google Scholar]
- 28.Gökler B, Ünal F, Pehlivantürk B, Kültür EÇ, Akdemir D, Taner Y. Reliability and validity of schedule for affective disorders and schizophrenia for school age children-present and lifetime version-turkish version (KSADS-PL-T) Turk J Child Adolesc Ment Health. 2004;11:109–116. [Google Scholar]
- 29.Leckman JF, Sholomskas D, Thompson D, Belanger A, Weissman MM. Best estimate of lifetime psychiatric diagnosis: a methodological study. Arch Gen Psychiatry. 1982;39:879–883. doi: 10.1001/archpsyc.1982.04290080001001. [DOI] [PubMed] [Google Scholar]
- 30.Gualtieri CT, Johnson LG. Age-related cognitive decline in patients with mood disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32:962–967. doi: 10.1016/j.pnpbp.2007.12.030. [DOI] [PubMed] [Google Scholar]
- 31.Huang EJ, Reichardt LF. Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci. 2001;24:677–736. doi: 10.1146/annurev.neuro.24.1.677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pan W, Banks WA, Fasold MB, Bluth J, Kastin AJ. Transport of brainderived neurotrophic factor across the blood-brain barrier. Neuropharmacology. 1998;37:1553–1561. doi: 10.1016/s0028-3908(98)00141-5. [DOI] [PubMed] [Google Scholar]
- 33.Ozan E, Okur H, Eker Ç, Eker ÖD, Gönül AS, Akarsu N. The effect of depression, BDNF gene val66met polymorphism and gender on serum BDNF levels. Brain Res Bull. 2010;81:61–65. doi: 10.1016/j.brainresbull.2009.06.022. [DOI] [PubMed] [Google Scholar]
- 34.Zhang XY, Chen da C, Xiu MH, Haile CN, Luo X, Xu K, et al. Cognitive and serum BDNF correlates of BDNF Val66Met gene polymorphism in patients with schizophrenia and normal controls. Human Genet. 2012;131:1187–1195. doi: 10.1007/s00439-012-1150-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lang UE, Hellweg R, Sander T, Gallinat J. The Met allele of the BDNF Val66Met polymorphism is associated with increased BDNF serum concentrations. Mol Psychiatry. 2009;14:120–122. doi: 10.1038/mp.2008.80. [DOI] [PubMed] [Google Scholar]
- 36.Bus BA, Arias-Vasquez A, Franke B, Prickaerts J, de Graaf J, Voshaar RC. Increase in serum brain-derived neurotrophic factor in met allele carriers of the BDNF Val66Met polymorphism is specific to males. Neuropsychobiology. 2012;65:183–187. doi: 10.1159/000336997. [DOI] [PubMed] [Google Scholar]
- 37.Aureli A, Del Beato T, Sebastiani P, Marimpietri A, Melillo CV, Sechi E, et al. Attention-deficit hyperactivity disorder and intellectual disability: a study of association with brain-derived neurotrophic factor gene polymorphisms. Int J Immunopathol Pharmacol. 2010;23:873–880. doi: 10.1177/039463201002300323. [DOI] [PubMed] [Google Scholar]
- 38.Li H, Liu L, Tang Y, Ji N, Yang L, Qian Q, et al. Sex-specific association of brain-derived neurotrophic factor (BDNF) Val66Met polymorphism and plasma BDNF with attention-deficit/hyperactivity disorder in a drug-naïve Han Chinese sample. Psychiatry Res. 2014;217:191–197. doi: 10.1016/j.psychres.2014.03.011. [DOI] [PubMed] [Google Scholar]
- 39.Schimmelmann BG, Friedel S, Dempfle A, Warnke A, Lesch KP, Walitza S, et al. No evidence for preferential transmission of common valine allele of the Val66Met polymorphism of the brain-derived neurotrophic factor gene [BDNF] in ADHD. J Neural Transm (Vienna) 2007;114:523–526. doi: 10.1007/s00702-006-0616-1. [DOI] [PubMed] [Google Scholar]
- 40.Sánchez-Mora C, Ribasés M, Ramos-Quiroga JA, Casas M, Bosch R, Boreatti-Hümmer A, et al. Meta-analysis of brain-derived neurotrophic factor p.Val66Met in adult ADHD in four European populations. Am J Med Genet B Neuropsychiatr Genet. 2010;153B:512–523. doi: 10.1002/ajmg.b.31008. [DOI] [PubMed] [Google Scholar]
- 41.Gadow KD, Roohi J, DeVincent CJ, Kirsch S, Hatchwell E. Association of COMT (Val158Met) and BDNF (Val66Met) Gene polymorphisms with anxiety, ADHD and tics in children with autism spectrum disorder. J Autism Dev Disord. 2009;39:1542–1551. doi: 10.1007/s10803-009-0794-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bergman O, Westberg L, Lichtenstein P, Eriksson E, Larsson H. Study on the possible association of brain-derived neurotrophic factor polymorphism with the developmental course of symptoms of attention deficit and hyperactivity. Int J Neuropsychopharmacol. 2011;14:1367–1376. doi: 10.1017/S1461145711000502. [DOI] [PubMed] [Google Scholar]
- 43.Willoughby MT. Developmental course of ADHD symptomatology during the transition from childhood to adolescence: a review with recommendations. J Child Psychol Psychiatry. 2003;44:88–106. doi: 10.1111/1469-7610.t01-1-00104. [DOI] [PubMed] [Google Scholar]
- 44.Egan M, Kojima M, Callicott J, Goldberg T, Kolachana B, Bertolino A, et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell. 2003;112:257–269. doi: 10.1016/s0092-8674(03)00035-7. [DOI] [PubMed] [Google Scholar]
- 45.Foltynie T, Lewis SGJ, Goldberg TE, Blackwell AD, Kolachana BS, Weinberger DR, et al. The BDNF Val66Met polymorphism has a gender specific influence on planning ability in Parkinson’s disease. J Neurol. 2005;252:833–838. doi: 10.1007/s00415-005-0756-5. [DOI] [PubMed] [Google Scholar]
- 46.Rybakowski JK, Borkowska A, Czerski PM, Skibinska M, Hauser J. Polymorphism of the brain-derived neurotrophic factor gene and performance on a cognitive prefrontal test in bipolar patients. Bipolar Disord. 2003;5:468–472. doi: 10.1046/j.1399-5618.2003.00071.x. [DOI] [PubMed] [Google Scholar]
- 47.Hariri AR, Goldberg TE, Mattay VS, Kolachana BS, Callicott JH, Egan MF, et al. Brain-derived neurotrophic factor val66met polymorphism affects human memory related hippocampal activity and predicts memory performance. J Neurosci. 2003;23:6690–6694. doi: 10.1523/JNEUROSCI.23-17-06690.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hashimoto R, Moriguchi Y, Yamashita F, Mori T, Nemoto K, Okada T, et al. Dose-dependent effect of the Val66Met polymorphism of the brain-derived neurotrophic factor gene on memory-related hippocampal activity. Neurosci Res. 2008;61:360–367. doi: 10.1016/j.neures.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 49.Dennis Na, Cabeza R, Need AC, Waters-Metenier S, Goldstein DB, Labar KS. Brain-derived neurotrophic factor val66met polymorphism and hippocampal activity on during episodic encoding and retrieval tasks. Hippocampus. 2010;21:980–989. doi: 10.1002/hipo.20809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van Wingen G, Rijpkema M, Franke B, vanEijndhoven P, Tendolkar I, Verkes RJ, et al. The brain-derived neurotrophic factor Val66Met polymorphism affects memory formation and retrieval of biologically salient stimuli. Neuroimage. 2010;50:1212–1218. doi: 10.1016/j.neuroimage.2010.01.058. [DOI] [PubMed] [Google Scholar]