Abstract
Several studies have demonstrated that allelic variants related to inflammation and the immune system may increase the risk for major depressive disorder (MDD) and reduce patient responsiveness to antidepressant treatment. Proteasomes are fundamental complexes that contribute to the regulation of T-cell function. Only one study has shown a putative role of proteasomal PSMA7, PSMD9 and PSMD13 genes in the susceptibility to an antidepressant response, and sparse data are available regarding the potential alterations in proteasome expression in psychiatric disorders such as MDD. The aim of this study was to clarify the role of these genes in the mechanisms underlying the response/resistance to MDD treatment. We performed a case-control association study on 621 MDD patients, of whom 390 were classified as treatment-resistant depression (TRD), and we collected peripheral blood cells and fibroblasts for mRNA expression analyses. The analyses showed that subjects carrying the homozygous GG genotype of PSMD13 rs3817629 had a twofold greater risk of developing TRD and exhibited a lower PSMD13 mRNA level in fibroblasts than subjects carrying the A allele. In addition, we found a positive association between PSMD9 rs1043307 and the presence of anxiety disorders in comorbidity with MDD, although this result was not significant following correction for multiple comparisons. In conclusion, by confirming the involvement of PSMD13 in the MDD treatment response, our data corroborate the hypothesis that the dysregulation of the complex responsible for the degradation of intracellular proteins and potentially controlling autoimmunity- and immune tolerance–related processes may be involved in several phenotypes, including the TRD.
Introduction
Major depressive disorder (MDD) is a major health problem associated with gradual and often incomplete recovery and with significant limitations in functioning and well-being. Unfortunately, several studies have shown that only about one-third of patients experience full remission after first-choice treatment, and that for many patients achieving remission requires repeated trials of sufficiently dosed antidepressant medication.1, 2 In addition, 15–30% of MDD patients are classified as having treatment-resistant depression (TRD).3, 4
Carvalho et al.5, 6 reported that increased activation of the inflammatory system is concordant with hyperactivity of the hypothalamic–pituitary–adrenal axis and with impaired glucocorticoid receptor (GR) sensitivity in TRD patients. These findings were corroborated by genetics studies indicating that functional allelic variants in the interleukin (IL)-1beta (IL-1b), tumor necrosis factor-α and C-reactive protein genes, as well as genetic variations affecting T-cell function, may increase the risk for depression. Moreover, single nucleotide polymorphisms (SNPs) in the IL-1b, IL-6 and IL-11 genes and in those regulating T-cell function may be associated with reduced responsiveness to antidepressant therapy.7
Although the aforementioned ILs and other factors have been extensively studied in MDD and in the response to its treatment, few studies of T-cell function are available to date.
Fundamental complexes that contribute to regulate T-cell function are proteasomes. In fact, proteasomes are the major multi-catalytic enzyme complexes involved in the intracellular degradation of ubiquitinated proteins and in the production of small protein fragments that can be used by major histocompatibility complex class I to present antigens to the immune system. Protein degradation is fundamental for many important biological functions, such as immune surveillance, cell cycle progression, apoptosis and synaptic reorganization; therefore, the disruption or alteration of this mechanism due to genetic variants can have a relevant impact on several diseases.8
Based on genetic studies, the ubiquitin–proteasome system has been identified as a canonical pathway that is associated with neuropsychiatric disorders such as Alzheimer disease,9 psychosis10 and bipolar disorder.11, 12, 13 In particular, two studies showed that several proteasome 26S subunits are significantly downregulated in schizophrenia,14, 15 and Bousman et al.11 suggested that the dysregulation of the ubiquitin–proteasome system may partially determine the manifestation and the severity of psychotic symptoms in schizophrenia and mood disorders.
Only one study has shown a putative role of proteasomal subunit genes in the susceptibility to MDD and the antidepressant response.16 The authors observed that the proteasome subunit β4 (PSMD4) gene is involved in etiopathogenesis of MDD, although this result was not replicated in the largest genome-wide association study on MDD.17 Alternatively, Wong et al.16 found significant associations of the treatment response with the 26S proteasome non-ATPase subunit 9 (PSMD9), proteasome alpha type 7 subunit (PSMA7) and PSMD13 genes. Recently, the PSMD9 gene has been found to be associated with generalized anxiety disorder.18
Finally, although data are available regarding the proteasome expression levels in human peripheral tissues in MDD patients,19, 20 to the best of our knowledge, no data have been reported concerning the relationship between the expression pattern of the ubiquitin–proteasome system and the antidepressant treatment response. Peripheral blood cells (PBCs) are frequently used to identify depression biomarkers;21 however, recent evidence has indicated that fibroblasts represent a promising model for studying several processes related to MDD22 because many possible confounding factors that are often associated with patient PBCs, such as diet, lifestyle, smoking status and drug treatment, may be virtually eliminated after several cycles of cell division.23
Based on the above findings, to better understand the role of proteasome genes in the mechanisms underlying the MDD treatment response, we (1) investigated the potential roles of three genes (PSMA7, PSMD9 and PSMD13) previously found to be associated with drug responses,16 (2) performed single-gene re-analysis on the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) data set and (3) analyzed the expression profiles of PBCs and fibroblasts.
Materials and methods
Sample
About 621 MDD patients with at least moderate to severe depression, who met Diagnostic and Statistical Manual of Mental Disorders-IV (DSM-IV) classification system criteria were voluntarily enrolled in the study. All of them had been referred to the Villa S. Chiara Psychiatric Hospital in Verona or to the Psychiatry Rehabilitation Unit of IRCCS Centro Fatebenefratelli ‘S. Giovanni di Dio' in Brescia.
Diagnosis of unipolar depression was confirmed using the Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) diagnostic scale. The exclusion criteria were as follows: (a) mental retardation or cognitive disorder; (b) a lifetime history of schizophrenic, schizoaffective or bipolar disorder; (c) personality disorder, substance abuse, alcohol abuse or dependency, obsessive-compulsive disorder or post-traumatic stress disorder as the primary diagnosis; and (d) comorbidity with an eating disorder.
About 147 patients (23.7%) showed psychotic symptoms; 258 (41.5%) showed current comorbidity in Axis I (generalized anxiety disorder, panic attacks or anxiety disorder not otherwise specified), 139 (22.4%) showed symptoms of Axis II disorders (paranoid, dependent, obsessive-compulsive or histrionic personality disorder) and 24 (3.9%) alcohol abuse, as a secondary diagnosis (the total number exceeded the number of subjects due to the presence of comorbidities).
About 390 subjects were classified as TRD patients (Group 1: TRD). Treatment resistance to antidepressants was defined as the failure to respond to two or more adequate trials of two or more different classes of antidepressants and to an adequate trial of a tricyclic antidepressant drug; such patients were classified as Stage III according to the Thase and Rush staging method.24
The second group was composed of 231 MDD patients experiencing a depressive episode who responded to their current treatment (Group 2: non-TRD).
The control sample consisted of 467 unrelated healthy volunteers who were screened for DSM-IV Axis I disorders by expert psychologists using the Mini-International Neuropsychiatric Interview (M.I.N.I.).25 Only healthy volunteers without a history of drug or alcohol abuse or dependence and without a personal or first-degree family history of psychiatric disorders were enrolled in the study. Furthermore, subjects who obtained a score <27/30 on the Mini Mental State Examination (M.M.S.E.)26 were excluded from the study.
All socio-demographic and clinical characteristics for all groups are shown in Table 1.
Table 1. Demographic and clinical characteristics of both the control and MDD patient groups, as well as the non-TRD and TRD patient subgroups.
| Characteristics | Controls (N=467) | MDD (N=621) | P-value |
|---|---|---|---|
| Age (years), mean (s.d.) | 46.9 (16.3) | 56.2 (13.7) | <0.001 |
| Gender (F), n (%) | 260 (55.7) | 422 (68.0) | <0.001 |
| Education (years), mean (s.d.) | 12.7 (4.7) | 9.2 (4.0) | <0.001 |
| TRD (N=390) | Non-TRD (N=231) | ||
| Age (years), mean (s.d.) | 56.8 (13.4) | 55.2 (14.1) | 0.16 |
| Gender (F), n (%) | 260 (66.7) | 162 (70.1) | 0.37 |
| Education (years), mean (s.d.) | 8.9 (3.9) | 9.8 (4.1) | 0.007 |
| % Of recurrent MDD | 90.5 | 70.1 | <0.001 |
| % Of severe vs moderate MDD | 87.9 | 45.0 | <0.001 |
| % Psychotic symptoms | 35.6 | 3.5 | <0.001 |
| % Comorbidity with personality disorders | 26.7 | 15.2 | 0.001 |
| % Comorbidity with anxiety disorders | 43.6 | 38.1 | 0.18 |
| % Comorbidity with alcohol abuse | 3.3 | 4.8 | 0.37 |
Abbreviations: F, female; MDD, major depressive disorder; PBC, peripheral blood cell; TRD, treatment-resistant depression.
We performed transcriptional analyses on 59 PBCs (from 37 MDD patients, including 20 non-TRD and 17 TRD patients, and 22 controls) and 48 fibroblasts samples (from 27 MDD patients, including 17 non-TRD and 10 TRD patients, and 21 controls). The socio-demographic and clinical features for both tissue samples are shown in Table 2.
Table 2. Demographic and clinical characteristics of both the control and MDD patient groups, as well as the TRD and non-TRD subgroups, from which fibroblast and PBC samples were collected.
|
Fibroblasts |
PBCs |
|||||
|---|---|---|---|---|---|---|
| Characteristics | Controls (N=21) | MDD (N=27) | P-value | Controls (N=22) | MDD (N=37) | P-value |
| Age (years), mean (s.d.) | 46.6 (11.9) | 51.2 (12.8) | 0.66 | 32.1 (5.1) | 49.9 (12.4) | <0.001 |
| Gender (% F) | 47.6 | 66.7 | 0.18 | 50.0 | 73.0 | 0.08 |
| Education (years), mean (s.d.) | 13.7 (5.8) | 8.6 (3.5) | 0.001 | 16.8 (4.1) | 10.6 (4.6) | <0.001 |
| BMI | 24.8 (2.5) | 26.1 (6.0) | 0.41 | 23.3 (2.6) | 25.3 (4.3) | 0.07 |
| % Smokers | 4.8 | 40.9 | 0.005 | 22.7 | 50.0 | 0.04 |
| TRD (N=10) | Non-TRD (N=17) | TRD (N=17) | Non-TRD (N=20) | |||
| Age (years), mean (s.d.) | 48.6 (9.7) | 52.7 (14.4) | 0.43 | 52.3 (11.9) | 47.7 (12.7) | 0.25 |
| Gender (% F) | 64.7 | 70.0 | 0.78 | 64.7 | 80.0 | 0.30 |
| Education (years), mean (s.d.) | 8.7 (4.2) | 8.6 (3.2) | 0.94 | 10.8 (5.9) | 10.4 (3.2) | 0.78 |
| BMI | 28.5 (6.1) | 25.2 (6.0) | 0.36 | 25.2 (3.6) | 25.5 (5.0) | 0.85 |
| % Smokers | 33.3 | 46.2 | 0.55 | 35.3 | 64.7 | 0.09 |
| % Of recurrent MDD | 100.0 | 82.4 | 0.16 | 100.0 | 45.0 | <0.001 |
| % Of severe vs moderate MDD | 50.0 | 47.1 | 0.88 | 88.2 | 45.0 | 0.006 |
| % Psychotic symptoms | 0.0 | 5.9 | 0.43 | 70.6 | 0.0 | <0.001 |
| % Comorbidity with personality disorders | 40.0 | 35.3 | 0.81 | 76.5 | 25.0 | 0.002 |
| % Comorbidity with anxiety disorders | 40.0 | 29.4 | 0.57 | 64.7 | 25.0 | 0.02 |
| % Comorbidity with alcohol abuse | 20.0 | 0.0 | 0.06 | 5.9 | 0.0 | 0.27 |
Abbreviations: BMI, body mass index; F, female; MDD, major depressive disorder; PBC, peripheral blood cell; TRD, treatment-resistant depression.
Only for transcriptional analyses, we used the following additional exclusion criteria: (1) age >70 years old, because fibroblasts have a shorter life span in culture; (2) presence of metabolic disorders (for example, diabetes); and (3) presence of specific skin diseases (for example, skin cancer or psoriasis).
Both patients and controls were Caucasians of Italian descent for at least two generations, residing in north Italy and unrelated to other participants.
The study was approved by the Local Ethics Committees (CEIOC IRCCS Istituto Centro San Giovanni di Dio Fatebenefratelli, Brescia N: 50/2008 and Ethics Committee of the province of Verona N: 4997/09.11.01), and written informed consent was obtained.
Genotyping
PSMD13 rs3817629, PSMD9 rs1043307 and PSMA7 rs2057168 were genotyped using the BeadXpress System and the VeraCode Assay according to the manufacturer's instructions (http://www.illumina.com). The raw BeadXpress data were processed using the Illumina BeadStudio software suite (genotyping module 3.3.7; San Diego, CA, USA), producing report files containing normalized intensity data and SNP genotypes.
The genotyping of PSMD13 rs3817629 and PSMD9 rs1043307 was repeated via the SNaPshot assay as a quality control. All details regarding the SNaPshot assay are available on request.
LD analysis
For the intronic SNP PSMD13 rs3817629, that was positively associated with response, we tested for linkage disequilibrium (LD) with other variants selected for their putative effects on protein function. Using the 1000 Genome database data, we computed the LD coefficient (r2) between the PSMD13 rs3817629 and all SNPs mapping inside PSMD13 gene and 10 kb downstream and upstream the gene. Then we selected those displaying r2>0.8. For coding variants, functional prediction was performed using the Variant Annotation Integrator (http://genome.ucsc.edu/index.html), and for noncoding variants, functional prediction was performed using the Regulome database (http://regulomedb.org/).27
STAR*D data sets replication study
We evaluated genetic data from data set 2 and 3 of the STAR*D study28 (https://www.nimhgenetics.org/) – distribution 3.02, selecting only patients defined as ‘White' we categorized these patients into the non-TRD and TRD groups. We defined any MDD patient who did not respond to two or more trials of different classes of antidepressants as TRD. We excluded subjects who responded to citalopram at Level 1 of the study from the non-TRD group to avoid placebo effects.29, 30 The population consisted of 259 non-TRD and 186 TRD patients. Logistic regression analyses were performed to evaluate differences in the genotype frequency distribution between the two groups. To correct for ethnic heterogeneity, we performed principal component analysis, and we used the first three components as covariates in the regression analyses. None of the analyzed SNPs was directly genotyped in the STAR*D cohort. Therefore, for the SNP PSMA7 rs2057168, the association was indirectly tested by analyzing the SNP rs1535669, which displayed high LD (r2=0.95) with rs2057168 in the European population accordingly to the NetAffx database of Affymetrix (http://www.affymetrix.com). For PSMD13 rs3817629 and PSMD9 rs1043307, because no SNPs displaying LD were available in the STARD data set, the association was tested using dosage data of the imputed genotypes. The SNPs within chromosomal regions (Hg19) chr11:236,808–252,984 and chr20:122,326,812–122,386,604 were imputed using Beagle software31 and using the 1000 Genome database.
All logistic regression analyses were performed using Plink software (v.1.9).32, 33
Cell culture and RNA isolation
Skin biopsies (3 mm2) were collected under local anesthesia from the scapular region under aseptic conditions and were immediately immersed in saline solution (phosphate-buffered saline). All primary cultures were grown in Eagle's minimum essential medium (Invitrogen-Life Technologies, Carlsbad, CA, USA) supplemented with fetal bovine serum (10%), penicillin (100 U ml−1), streptomycin (100 μg ml−1), non-essential amino acids (1% v/v) and glutamine (1% v/v) under optimal conditions (37 °C and 5% CO2). The medium was replaced every 3 days. When the fibroblasts reached confluence, the cells were subcultured into larger tissue culture dishes or were frozen in 20% foetal bovine serum and 10% DMSO. All fibroblasts were cultured until the fifth passage. Fibroblast cell cultures were screened for mycoplasma infection and mycoplasma removal agents were used to avoid and prevent the risk of contamination.
RNA was extracted from fibroblasts using the NucleoSpin kit (Carlo Erba Reagenti, Milan, Italy) according to the manufacturer's instructions.
Blood samples were collected between 0800 and 0900 hours after an overnight fast via venepuncture using PaxGene Tubes (Qiagen, Crawley, UK). RNA isolation was performed using the PaxGene Blood RNA Kit (Qiagen) according to the manufacturer's instructions.
One microgram of total RNA was used for complementary DNA synthesis using random hexamer primers (Invitrogen-Life Technologies) and Superscript II Reverse Transcriptase (Invitrogen-Life Technologies) according to the manufacturer's instructions.
Real-time PCR analyses
For those genes in which a positive genotype association was found (PSMD13 and PSMD9), the mRNA expression level was evaluated in both fibroblasts and PBCs.
The transcriptional expression levels were determined with TaqMan assays (Applied Biosystems, Foster City, CA, USA) performed on a StepOnePlus instrument (Applied Biosystems) according to the manufacturer's protocol.
In a pilot study in fibroblasts, we found that GAPDH is among the most stable reference genes in this tissue (data not shown). Moreover, GAPDH has been already used as a reference in MDD studies using fibroblasts and PBCs.34, 35, 36 Therefore, the relative expression levels of the target genes in the patient groups were calculated according to the comparative Ct method (−ΔΔCt method)37 using GAPDH as the reference gene. Each determination was repeated in duplicate.
Statistical analyses
Analysis of variance was used to detect possible differences in age and education between controls and MDD, as well as between TRD and non-TRD. χ2-tests were used to detect differences in demographic and clinical variables between the groups (Table 1). The differences in genotype frequency distribution for the three SNPs between MDD and controls were tested using a logistic regression analysis. Since the two groups differed in terms of gender, age and education (see Table 1) these variables were considered as covariates in the model. The same analyses were performed to test differences in genotype frequency distribution between TRD and non-TRD, and education (Table 1) was considered as covariate in the model. Moreover, we performed secondary analyses by logistic regression to investigate the putative influences of these three SNPs on comorbidity with anxiety disorders and the presence of psychotic symptoms. The additive, dominant and recessive genotype models were tested. All P-values were adjusted applying Bonferroni correction for all comparisons (N=36). Odds ratios and corresponding 95% confidence intervals (CIs) were used to quantify the association. All the genotype analyses were performed with Plink (v. 1.9) software.
According to G*Power Calculator software38, 39 in our cohorts of TRD e non-TRD patients, we have a power greater than 95% (for α<0.0014) of detecting the same allele differences (odds ratios<0.44), reported in the study by Wong et al.16 between responder and nonresponder.
The Kruskal–Wallis and Mann–Whitney U non-parametric tests were used to evaluate differences in PSMD13 and PSMD9 mRNA levels between MDD and controls, as well as between TRD and non-TRD, and to evaluate the association between the expression levels of the same genes with the relative polymorphisms. Pearson's coefficient was used to evaluate bivariate correlations between mRNA expression levels and the socio-demographic and clinical variables reported in Table 2.
Parametric and non-parametric tests were used meeting relative assumptions (for example, distribution, sample size).
All statistical analyses were conducted using SPSS version 17.0 (SPSS, Chicago, IL, USA).
Results
Genetic analysis results for our sample
As reported in Table 3, none of the analyzed SNPs showed a significantly different genotype frequency distribution between the controls and the patients using the additive model. The identical results were observed using the dominant and recessive models (data not reported).
Table 3. Logistic regression results for the genotype frequency distributions between the CTRL and the MDD patients.
| SNP | Major homozygous frequency | Heterozygous frequency | Minor homozygous frequency | Regression P-value additive modela | PGC P-value |
|---|---|---|---|---|---|
| PSMA7 rs2057168 | AA | AG | GG | ||
| CTRL | 296 (0.64) | 148 (0.32) | 21 (0.04) | 0.59 | 0.48 |
| MDD | 399 (0.65) | 186 (0.30) | 27 (0.04) | ||
| PSMD9 rs1043307 | AA | AG | GG | ||
| CTRL | 207 (0.44) | 219 (0.47) | 40 (0.09) | 0.23 | 0.68 |
| MDD | 303 (0.49) | 255 (0.41) | 59 (0.10) | ||
| PSMD13 rs3817629 | GG | AG | AA | ||
| CTRL | 224 (0.48) | 202 (0.44) | 37 (0.08) | 0.41 | Not genotyped |
| MDD | 311 (0.51) | 261 (0.42) | 42 (0.07) | ||
Abbreviations: CTRL, control; MDD, major depressive disorder; PGC, Psychiatric Genomics Consortium; SNP, single nucleotide polymorphism.
Gender, age and education were considered as covariates in the model.
When the patients were stratified into the TRD and non-TRD groups, differences in the genotype distribution of PSMD13 rs3817629 was observed (P=0.004); GG homozygotes were more frequent in TRD patient group (56%, P=0.00045, OR=1.75, 95% CI: 1.26–2.45) (Table 4). This difference remained significant after adjustment for multiple comparisons (P=0.016).
Table 4. Logistic regression results for the genotype frequency distributions between the non-TRD and TRD patients.
| SNP | Major homozygous frequency | Heterozygous frequency | Minor homozygous frequency | P-valuea additive model | P-valuea dominant model | P-valuea recessive model |
|---|---|---|---|---|---|---|
| PSMA7 rs2057168 | AA | AG | GG | |||
| Non-TRD | 143 (0.62) | 77 (0.33) | 10 (0.04) | 0.35 | 0.22 | 0.79 |
| TRD | 256 (0.67) | 109 (0.29) | 17 (0.04) | |||
| PSMD9 rs1043307 | AA | AG | GG | |||
| Non-TRD | 107 (0.46) | 101 (0.44) | 21 (0.09) | 0.50 | 0.33 | 0.90 |
| TRD | 196 (0.51) | 154 (0.40) | 38 (0.10) | |||
| PSMD13 rs3817629 | GG | AG | AA | |||
| Non-TRD | 94 (0.42) | 115 (0.51) | 16 (0.07) | 0.004 | 0.0005 | 0.87 |
| TRD | 217 (0.56) | 146 (0.38) | 26 (0.07) | |||
Abbreviations: SNP, single nucleotide polymorphism; TRD, treatment-resistant depression.
Education was considered as a covariate in the model. Bold numbers indicate significant P-values (<0.05).
The Regulome database reported that PSMD13 rs3817629 is an intronic SNP displaying minimal evidence of affecting transcription factor-binding sites (score: 4). LD analysis of the genotypic data from the 1000 Genome database revealed that PSMD13 rs3817629 is in high LD with 40 SNPs located inside or close to PSMD13 gene, 6 of which are missense variants mapping to the first or second exon of the PSMD13 gene and predicted as benign using Polyphen (http://genetics.bwh.harvard.edu/pph/) (Supplementary Figure 1S). Moreover, rs10902110 and rs10794302, were identified as intronic variants expected to affect the binding of transcription factors (Regulome score: 2b). Chromatin immunoprecipitation sequencing (chip-seq) analysis indicated that rs10902110, which maps to the first intron of the PSMD13 gene, is located in an open chromatin locus that binds to RNA polymerase II and other transcription factors such as CREBBP, RFX3, MYC, SP1, GABPA and ELF140, 41, 42 (ENCODE project). Alternatively, rs10794302 maps to the first intron of the SIRT3 gene at a locus predicted to have an open chromatin structure and a transcription factor-binding motif. Chip-seq analysis suggested that the GR (NR3C1) binds to this region (ENCODE project). Significant associations were detected between the presence of anxiety disorders in comorbidity with MDD and PSMD9 rs1043307. The genotype frequency distribution of this SNP was significantly different between MDD patients with and without anxiety disorders according to the additive model (P=0.035). In particular, subjects carrying the AA or AG genotype exhibited a 1.76-fold (95% CI 0.98–3.18) and 1.38-fold (95% CI 0.76–2.51) increased risk of anxiety comorbidity relative to subjects carrying the homozygous GG genotype, respectively. However, this result did not remain significant after adjustment for multiple comparisons.
Replication of the genotypic data in the STAR*D cohort
To confirm our results, we replicated the genotypic analyses in subgroups of STAR*D patients. As shown in Table 5, none of the analyzed SNPs showed a significantly different genotype distribution between the TRD and non-TRD groups. We also tested the possible association between PSMD9 and anxiety in 118 patients with and 972 patients without comorbid anxiety disorder. No difference in the rs1043307 genotype frequency distribution was observed between these two groups (P=0.74).
Table 5. Logistic regression results for the genotype frequency distributions between the non-TRD and TRD patients in the STAR*D cohort.
| Gene | SNP tested | P-value additive modela,b | Note |
|---|---|---|---|
| PSMA7 | rs1535669 | 0.76 | LD r2=0.95 with rs2057168 |
| PSMD9 | rs1043307 | 0.30 | Imputed with r2=0.89 |
| PSMD13 | rs3817629 | 0.98 | Imputed with r2=0.94 |
Abbreviations: LD, linkage disequilibrium; PCA, principal component analysis; SNP, single nucleotide polymorphism.
The first three PCAs were considered as covariates in the analysis.
For the imputed SNPs, the B allelic dosage was used as independent variables in the regression.
mRNA expression analysis on fibroblasts
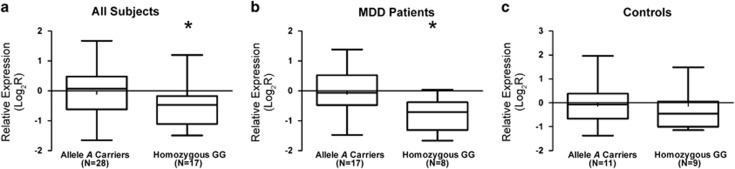
The analyses of the expression levels of PSMD13 mRNA in fibroblasts from MDD subjects and controls revealed no significant differences between the two groups, as well as comparing TRD and non-TRD patients. Furthermore, no significant association with any examined socio-demographic or clinical variables was detected (data not shown). However, we found a trend toward a correlation between the PSMD13 genotype and its mRNA levels (H=5.10; 2 degree of freedom, P=0.08). In particular, the homozygous GG carriers displayed lower expression levels than the A allele carriers (z=−2.20; P=0.03) (Figure 1a). However, after stratifying the subjects into the control and MDD patient groups, this downregulation was observed only in MDD patients (z=−2.40; P=0.02) (Figure 1b), whereas in control sample this association was not obtained (11A allele carriers, 9 GG; z=−0.57; P=0.60) (Figure 1c).
Figure 1.
Relative expression mRNA levels of PSMD13 rs3817629. Box-plots showing the median, quartiles and extreme values of relative expression of PSMD13 rs3817629 A allele carriers with respect to GG subjects. The bold lines represent median values, the box boundaries mark the 25th and 75th percentiles of each distribution, respectively. Whiskers, above and below the hinges, mark the largest and smallest observed values. (a) the analysis was performed in all subjects; (b) only in MDD group; (c) only in control group. '*' Indicates significant difference (P-value<0.05) computed by the Mann–Whitney U non-parametric test. MDD, major depressive disorder.
Regarding the expression of PSMD9, the analyses did not reveal any significant differences in its mRNA levels (data not shown).
mRNA expression analysis on PBCs
The analyses of PSMD13 mRNA levels in PBCs did not show any differences, both when we compared MDD and controls, as well as TRD and non-TRD patients. No association, neither with PSMD13 polymorphism nor with any socio-demographic or clinical variables, was detected.
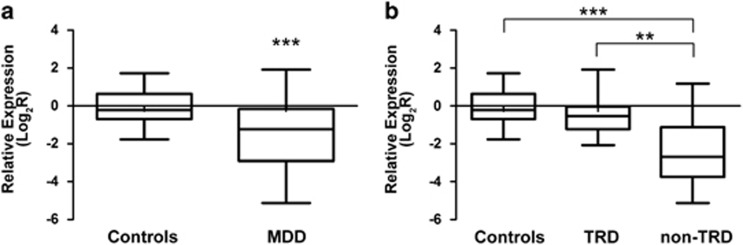
As concerned PSMD9, we observed a significant downregulation in MDD patients compared with healthy controls (z=−3.75; P=1.80 × 10−4) (Figure 2a). Pairwise comparisons indicated high differences between non-TRD patients group and both controls (z=−4.51; P=6.54 × 10−6) and TRD patients (z=−3.38; P=0.001), whereas, no significant difference was observed between the TRD patients and the healthy subjects (z=−1.70; P=0.09) (Figure 2b).
Figure 2.
Relative expression mRNA levels of PSMD9 rs1043307. Box-plots showing the median, quartiles and extreme values of relative expression of PSMD9 rs1043307 in (a) MDD patients and controls; (b) in healthy subjects, TRD and non-TRD. The bold lines represent median values, the box boundaries mark the 25th and 75th percentiles of each distribution, respectively. Whiskers, above and below the hinges, mark the largest and smallest observed values, respectively. '**' Indicates significant difference (P-value<0.01), whereas '***' indicates a P-value <0.001, computed by the Kruskal–Wallis and Mann–Whitney U non-parametric tests.
Our data also revealed significant correlations between PSMD9 mRNA levels with the presence of psychotic symptoms (r=0.43; P=0.008), comorbidity with anxiety disorders (r=0.40; P=0.01) and smoke (r=−0.39; P=0.003). No significant correlation between mRNA levels with PSMD9 rs1043307 SNP was observed.
Discussion
In the present study, we found an association between the PSMD13 rs3817629 G allele and TRD. In particular, homozygotic GG have about a twofold higher risk of developing MDD treatment resistance than A allele carriers. Our result supports the original observation of Wong et al.,16 who found that the A allele is associated with the antidepressant treatment response in a cohort of depressed patients.
LD analysis revealed that although rs3817629 does not appear to affect a binding site, it is in high LD with two SNPs, rs10794302 and rs10902110, which likely affect binding sites according to the Regulome database. In particular, Chip-seq analyses showed that rs10794302 maps to a locus corresponding to a GR-binding site. This result is intriguing because it has been observed that antidepressants exert their clinical effects via the modulation of the GR.43
Moreover, our expression analysis revealed that the homozygous GG genotype was associated with reduced transcription of PSMD13 in MDD fibroblasts. This result suggests that rs3817629 is an expression quantitative trait locus in this tissue. Our finding is corroborated by a recent study that investigated the genetic variability in the regulation of gene expression in 10 regions of the human brain.44 The authors found that rs3817629 is a cis-expression quantitative trait locus in different areas of the brain and that this SNP is associated with the PSMD13 transcriptional expression level (t3315549) in the entire brain (http://www.braineac.org/). Specifically, this result was primarily due to the differential expression observed in eight brain regions: the cerebellar cortex, the hippocampus, the inferior olivary nucleus, the occipital cortex, the putamen, the temporal cortex, the thalamus and the intralobular white matter. In all of these tissues, as in our fibroblasts, the homozygous GG genotype was associated with lower PSMD13 expression providing further evidence that fibroblasts serve as a good peripheral model for the study of biomarkers of the central nervous system.22, 45, 46
This transcriptional finding combined with our genetic main result, that the homozygous GG subjects have about a twofold higher risk of developing MDD treatment resistance, might suggest that this SNP may represent a risk factor for TRD due to alteration of PSMD13 transcription. Unfortunately, the small sample size of our TRD and non-TRD groups for expression analysis, do not allow us to test differences in the mRNA levels between these two groups as concern the GG genotype.
Under inflammatory conditions mediated by IFN-γ, the activation of the mTOR pathway enhances protein translation and induces protein-damaging oxygen radical production; these events contribute to an increase in the synthesis of proteins that fail to fold correctly, referred to as defective ribosomal products and in proteasomal activity.47, 48 We suggest that subjects carrying the GG genotype and, thus, a low expression level of PSMD13 may respond less effectively to conditions of augmented need for defective ribosomal product degradation, such as those caused by stress or an inflammatory response, which are observed in MDD. Therefore, such conditions could lead to more severe phenotypes and to worse drug treatment responses in MDD patients.
Our negative pharmacogenetic results for PSMD9 did not confirm the previous finding reported by Wong et al.;16 however, a recent study has shown that the PSMD9 missense SNP rs1043307 co-segregates with generalized anxiety disorder in type-2 diabetes families.18 Interestingly, we found a positive association between this SNP and the presence of anxiety disorders in comorbidity with MDD. Although our result did not remain significant after correction for multiple comparisons, taken together these data support the putative role of PSMD9 as a risk factor for anxiety phenotypes.
Anxiety disorders co-occur in the same individual, either simultaneously or sequentially, with MDD (in ~40–50% of patients), and it has been widely demonstrated that anxiety impairs depression remission.49 Furthermore, several pieces of evidence led to the hypothesis that these two phenotypes have partially shared genetic bases.49, 50, 51, 52 This observation suggests that PSMD9 variants may be indirectly implicated in the response through an involvement in one of the most important negative predictor of response like anxiety.
The likely involvement of PSMD9 in MDD phenotypes was also suggested by our transcriptional data. In fact, mRNA expression analysis of PBCs revealed that PSMD9 is significantly downregulated in MDD patients, specifically non-TRD patients, compared with controls. This result is in agreement with those observed in one of the largest genome-wide gene expression study in MDD.19 The results of single-gene association with MDD status, available on request from the National Institute of Mental Health (NIMH) Center for Collaborative Genomic Studies on Mental Disorders, (https://www.nimhgenetics.org/access_data_biomaterial.php), indicated that the mRNA expression levels of PSMD9 gene were decreased in whole blood of MDD patients (nominal P-value=0.02).
However, our PBCs transcriptional levels are strongly associated to the presence of psychotic symptoms, the comorbidity with anxiety disorders and smoke. Thus, it is difficult to clarify if rather than being the cause of MDD, the alterations of PSMD9 could probably be a consequence of the disease, since it is well-known that a depressive state has an adverse influence on lifestyle.
For both genes (PSMD13 and PSMD9), we observed a discrepancy between PBCs and fibroblasts expression results. This could be explained by biases due to confounding factors that probably affect PBCs findings. Indeed, fibroblasts are maintained in cultures in a controlled, reproducible environment, and after several rounds of cell division they are minimally affected by confounding factors (for example, lifestyle or medication use) to which, in contrast, PBCs are exposed.22, 35, 46, 53, 54
The genetic analyses performed on the STAR*D cohort were not consistent with the results from our cohort or from Wong et al.16 These contrasting results could be explained by different reasons: (1) neither SNP was directly genotyped in the STAR*D cohort, but, albeit with high confidence, they were imputed, and therefore, the analysis was performed on predicted genotypes rather than on actual genotypes; (2) our samples and those of Wong et al.16 and the STAR*D cohort differed with respect to certain clinical characteristics. For instance, the STAR*D study included only outpatients with non-psychotic MDD, whereas both our sample than the Wong et al.16 one include MDD patients with psychotic symptoms. In addition, only ~11% of the patients in the selected STAR*D cohort presented anxiety disorders in comorbidity, whereas this percentage was 40% in our sample and in the study by Wong et al.16, 55 the patients showed an high severity of anxiety. As mentioned above, anxiety is a relevant negative predictor of the treatment response; moreover, in our cohort and in the STAR*D cohort we stratified in TRD and non-TRD patients, whereas Wong et al.16 stratified in responders and non-responders to desipramine or fluoxetine; and (3) the ethnicity in the STAR*D cohort is more heterogeneous than in the cohort of Wong et al.16 and in ours. This could represent an additional confounding factor.
In conclusion, despite the limitations of this study, our findings provide convergent evidence that the dysregulation of the ubiquitin–proteasome system is involved in MDD response treatment mechanisms. Independent replications are needed to better understand the role of this system in psychiatric disorders.
Acknowledgments
This research was supported by grants from the Italian Ministry of Health (RC and RF2007 Conv. 42). We thank Federica Centin and Barbara Bertasi for laboratory support and Elisabetta Tessari, Maria Abate and Giulio Gainelli for the recruitment of the patients. EM is supported by the Shabbetai Donnolo Fellowship between Italy and Israel. Furthermore, we would like to express our sincere gratitude to all volunteers that participated in the study. Study 18—data and biomaterials were obtained from the limited access data sets distributed from the NIH-supported ‘Sequenced Treatment Alternatives to Relieve Depression' (STAR*D). STAR*D focused on non-psychotic major depressive disorder in adults seen in outpatient settings. The primary purpose of this research study was to determine which treatment works best if the first treatment with medication does not produce an acceptable response. The study was supported by NIMH Contract # N01MH90003 to the University of Texas Southwestern Medical Center. The ClinicalTrials.gov identifier is NCT00021528. Data was provided by Dr Douglas F Levinson. The resources were supported by the National Institutes of Health/National Institute of Mental Health Grants 5RC2MH089916 (PI: Douglas F Levinson, MD; Co-investigators: Myrna M Weissman, PhD; James B Potash, MD, MPH; Daphne Koller, PhD and Alexander E Urban, PhD) and 3R01MH090941 (co-investigator: Daphne Koller, PhD).
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Translational Psychiatry website (http://www.nature.com/tp)
Supplementary Material
References
- Trivedi MH, Rush AJ, Wisniewski SR, Nierenberg AA, Warden D, Ritz L et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry 2006; 163: 28–40. [DOI] [PubMed] [Google Scholar]
- Gaynes BN, Rush AJ, Trivedi MH, Wisniewski SR, Spencer D, Fava M. The STAR*D study: treating depression in the real world. Cleve Clin J Med 2008; 75: 57–66. [DOI] [PubMed] [Google Scholar]
- Thomas L, Kessler D, Campbell J, Morrison J, Peters TJ, Williams C et al. Prevalence of treatment-resistant depression in primary care: cross-sectional data. Br J Gen Pract 2013; 63: e852–e858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berlim MT, Turecki G. Definition, assessment, and staging of treatment-resistant refractory major depression: a review of current concepts and methods. Can J Psychiatry 2007; 52: 46–54. [DOI] [PubMed] [Google Scholar]
- Carvalho LA, Pariante CM. In vitro modulation of the glucocorticoid receptor by antidepressants. Stress 2008; 11: 411–424. [DOI] [PubMed] [Google Scholar]
- Carvalho LA, Torre JP, Papadopoulos AS, Poon L, Juruena MF, Markopoulou K et al. Lack of clinical therapeutic benefit of antidepressants is associated overall activation of the inflammatory system. J Affect Disord 2013; 148: 136–140. [DOI] [PubMed] [Google Scholar]
- Bufalino C, Hepgul N, Aguglia E, Pariante CM. The role of immune genes in the association between depression and inflammation: a review of recent clinical studies. Brain Behav Immun 2013; 31: 31–47. [DOI] [PubMed] [Google Scholar]
- Gomes AV. Genetics of proteasome diseases. Scientifica (Cairo) 2013; 2013: 637629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam YA, Pickart CM, Alban A, Landon M, Jamieson C, Ramage R et al. Inhibition of the ubiquitin-proteasome system in Alzheimer's disease. Proc Natl Acad Sci USA 2000; 97: 9902–9906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee YH, Kim JH, Song GG. Pathway analysis of a genome-wide association study in schizophrenia. Gene 2013; 525: 107–115. [DOI] [PubMed] [Google Scholar]
- Bousman CA, Chana G, Glatt SJ, Chandler SD, May T, Lohr J et al. Positive symptoms of psychosis correlate with expression of ubiquitin proteasome genes in peripheral blood. Am J Med Genet B Neuropsychiatr Genet 2010; 153B: 1336–1341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bousman CA, Chana G, Glatt SJ, Chandler SD, Lucero GR, Tatro E et al. Preliminary evidence of ubiquitin proteasome system dysregulation in schizophrenia and bipolar disorder: convergent pathway analysis findings from two independent samples. Am J Med Genet B Neuropsychiatr Genet 2010; 153B: 494–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Middleton FA, Mirnics K, Pierri JN, Lewis DA, Levitt P. Gene expression profiling reveals alterations of specific metabolic pathways in schizophrenia. J Neurosci 2002; 22: 2718–2729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vawter MP, Barrett T, Cheadle C, Sokolov BP, Wood WH 3rd, Donovan DM et al. Application of cDNA microarrays to examine gene expression differences in schizophrenia. Brain Res Bull 2001; 55: 641–650. [DOI] [PubMed] [Google Scholar]
- Altar CA, Jurata LW, Charles V, Lemire A, Liu P, Bukhman Y et al. Deficient hippocampal neuron expression of proteasome, ubiquitin, and mitochondrial genes in multiple schizophrenia cohorts. Biol Psychiatry 2005; 58: 85–96. [DOI] [PubMed] [Google Scholar]
- Wong ML, Dong C, Maestre-Mesa J, Licinio J. Polymorphisms in inflammation-related genes are associated with susceptibility to major depression and antidepressant response. Mol Psychiatry 2008; 13: 800–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ripke S, Wray NR, Lewis CM, Hamilton SP, Weissman MM, Breen G et al. A mega-analysis of genome-wide association studies for major depressive disorder. Mol Psychiatry 2013; 18: 497–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gragnoli C. Proteasome modulator 9 gene SNPs, responsible for anti-depressant response, are in linkage with generalized anxiety disorder. J Cell Physiol 2014; 229: 1157–1159. [DOI] [PubMed] [Google Scholar]
- Mostafavi S, Battle A, Zhu X, Potash JB, Weissman MM, Shi J et al. Type I interferon signaling genes in recurrent major depression: increased expression detected by whole-blood RNA sequencing. Mol Psychiatry 2014; 19: 1267–1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta D, Menke A, Binder EB. Gene expression studies in major depression. Curr Psychiatry Rep 2010; 12: 135–144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tylee DS, Kawaguchi DM, Glatt SJ. On the outside, looking in: a review and evaluation of the comparability of blood and brain "-omes". Am J Med Genet B Neuropsychiatr Genet 2013; 162B: 595–603. [DOI] [PubMed] [Google Scholar]
- Garbett KA, Vereczkei A, Kalman S, Brown JA, Taylor WD, Faludi G et al. Coordinated messenger RNA/microRNA changes in fibroblasts of patients with major depression. Biol Psychiatry 2014; 77: 256–265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi-Takagi A, Vawter MP, Iwamoto K. Peripheral biomarkers revisited: integrative profiling of peripheral samples for psychiatric research. Biol Psychiatry 2014; 75: 920–928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thase ME, Rush AJ. When at first you don't succeed: sequential strategies for antidepressant nonresponders. J Clin Psychiatry 1997; 58: 23–29. [PubMed] [Google Scholar]
- Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998; 59: 22–33, quiz 34-57. [PubMed] [Google Scholar]
- Folstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12: 189–198. [DOI] [PubMed] [Google Scholar]
- Boyle AP, Hong EL, Hariharan M, Cheng Y, Schaub MA, Kasowski M et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res 2012; 22: 1790–1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garriock HA, Kraft JB, Shyn SI, Peters EJ, Yokoyama JS, Jenkins GD et al. A genomewide association study of citalopram response in major depressive disorder. Biol Psychiatry 2010; 67: 133–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perlis RH, Moorjani P, Fagerness J, Purcell S, Trivedi MH, Fava M et al. Pharmacogenetic analysis of genes implicated in rodent models of antidepressant response: association of TREK1 and treatment resistance in the STAR(*)D study. Neuropsychopharmacology 2008; 33: 2810–2819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh BT, Seidman SN, Sysko R, Gould M. Placebo response in studies of major depression: variable, substantial, and growing. JAMA 2002; 287: 1840–1847. [DOI] [PubMed] [Google Scholar]
- Browning BL, Browning SR. A unified approach to genotype imputation and haplotype-phase inference for large data sets of trios and unrelated individuals. Am J Hum Genet 2009; 84: 210–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 2007; 81: 559–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang CC, Chow CC, Tellier LC, Vattikuti S, Purcell SM, Lee JJ. Second-generation PLINK: rising to the challenge of larger and richer data sets. Gigascience 2015; 4: 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garbett KA, Vereczkei A, Kalman S, Brown JA, Taylor WD, Faludi G et al. Coordinated messenger RNA/microRNA changes in fibroblasts of patients with major depression. Biol Psychiatry 2015; 77: 256–265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabriela Nielsen M, Congiu C, Bortolomasi M, Bonvicini C, Bignotti S, Abate M et al. MTHFR: Genetic variants, expression analysis and COMT interaction in major depressive disorder. J Affect Disord 2015; 183: 179–186. [DOI] [PubMed] [Google Scholar]
- Galecki P, Galecka E, Maes M, Orzechowska A, Berent D, Talarowska M et al. Vascular endothelial growth factor gene (VEGFA) polymorphisms may serve as prognostic factors for recurrent depressive disorder development Prog Neuropsychopharmacol. Biol Psychiatry 2013; 45: 117–124. [DOI] [PubMed] [Google Scholar]
- Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 2008; 3: 1101–1108. [DOI] [PubMed] [Google Scholar]
- Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 2007; 39: 175–191. [DOI] [PubMed] [Google Scholar]
- Faul F, Erdfelder E, Buchner A, Lang AG. Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav Res Methods 2009; 41: 1149–1160. [DOI] [PubMed] [Google Scholar]
- Hollenhorst PC, Chandler KJ, Poulsen RL, Johnson WE, Speck NA, Graves BJ. DNA specificity determinants associate with distinct transcription factor functions. PLoS Genet 2009; 5: e1000778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jolma A, Kivioja T, Toivonen J, Cheng L, Wei G, Enge M et al. Multiplexed massively parallel SELEX for characterization of human transcription factor binding specificities. Genome Res 2010; 20: 861–873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu S, Cui K, Jothi R, Zhao DM, Jing X, Zhao K et al. GABP controls a critical transcription regulatory module that is essential for maintenance and differentiation of hematopoietic stem/progenitor cells. Blood 2011; 117: 2166–2178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anacker C, Zunszain PA, Carvalho LA, Pariante CM. The glucocorticoid receptor: pivot of depression and of antidepressant treatment? Psychoneuroendocrinology 2011; 36: 415–425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramasamy A, Trabzuni D, Guelfi S, Varghese V, Smith C, Walker R et al. Genetic variability in the regulation of gene expression in ten regions of the human brain. Nat Neurosci 2014; 17: 1418–1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattane N, Minelli A, Milanesi E, Maj C, Bignotti S, Bortolomasi M et al. Altered gene expression in schizophrenia: findings from transcriptional signatures in fibroblasts and blood. PLoS One 2015; 10: e0116686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auburger G, Klinkenberg M, Drost J, Marcus K, Morales-Gordo B, Kunz WS et al. Primary skin fibroblasts as a model of Parkinson's disease. Mol Neurobiol 2012; 46: 20–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Deventer S, Neefjes J. The immunoproteasome cleans up after Inflammation. Cell 2010; 142: 517–518. [DOI] [PubMed] [Google Scholar]
- Seifert U, Bialy LP, Ebstein F, Bech-Otschir D, Voigt A, Schroter F et al. Immunoproteasomes preserve protein homeostasis upon interferon-induced oxidative stress. Cell 2010; 142: 613–624. [DOI] [PubMed] [Google Scholar]
- Minelli A, Maffioletti E, Shiloh AR. Genetics of anxiety disorders, In: Anxiety disorders: Risk factors, Genetic Determinants and Cognitive-behavioral Treatment. Nova Science Publishers, Hauppauge, NY, USA, 2014.
- Hettema JM. What is the genetic relationship between anxiety and depression? Am J Med Genet C Semin Med Genet 2008; 148C: 140–146. [DOI] [PubMed] [Google Scholar]
- Gatt JM, Burton KL, Williams LM, Schofield PR. Specific and common genes implicated across major mental disorders: a review of meta-analysis studies. J Psychiatr Res 2015; 60C: 1–13. [DOI] [PubMed] [Google Scholar]
- Blanco C, Rubio J, Wall M, Wang S, Jiu CJ, Kendler KS. Risk factors for anxiety disorders: common and specific effects in a national sample. Depress Anxiety 2014; 31: 756–764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akin D, Manier DH, Sanders-Bush E, Shelton RC. Decreased serotonin 5-HT2A receptor-stimulated phosphoinositide signaling in, fibroblasts from melancholic depressed patients. Neuropsychopharmacology 2004; 29: 2081–2087. [DOI] [PubMed] [Google Scholar]
- Kalman S, Garbett KA, Vereczkei A, Shelton RC, Korade Z, Mirnics K. Metabolic stress-induced microRNA and mRNA expression profiles of human fibroblasts. Exp Cell Res 2014; 320: 343–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Licinio J, O'Kirwan F, Irizarry K, Merriman B, Thakur S, Jepson R et al. Association of a corticotropin-releasing hormone receptor 1 haplotype and antidepressant treatment response in Mexican-Americans. Mol Psychiatry 2004; 9: 1075–1082. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.