Abstract
 (3‘-2‘)-α-l-Threose nucleic acid (TNA) is an unnatural polymer that possesses the rare ability to base-pair with RNA, DNA, and itself. This feature, coupled with its chemical simplicity, makes TNA of interest as a possible progenitor of RNA during the early history of life. To evaluate the functional potential of TNA, we have developed a system for the in vitro selection of TNA. We identified the Therminator DNA polymerase as a remarkably efficient DNA-dependent TNA polymerase capable of polymerizing more than 50 tNTPs. We have also developed a method of covalently linking a DNA template to the TNA strand that it encodes, thus obviating the need for a TNA-dependent DNA polymerase during cycles of selection.
(3‘-2‘)-α-l-Threose nucleic acid (TNA) is an unnatural polymer that possesses the rare ability to base-pair with RNA, DNA, and itself. This feature, coupled with its chemical simplicity, makes TNA of interest as a possible progenitor of RNA during the early history of life. To evaluate the functional potential of TNA, we have developed a system for the in vitro selection of TNA. We identified the Therminator DNA polymerase as a remarkably efficient DNA-dependent TNA polymerase capable of polymerizing more than 50 tNTPs. We have also developed a method of covalently linking a DNA template to the TNA strand that it encodes, thus obviating the need for a TNA-dependent DNA polymerase during cycles of selection.
(3‘-2‘)-α-l-Threose nucleic acid (TNA, Figure 1A) is an unnatural nucleic acid that was identified during an extensive evaluation of alternative sugar−phosphate backbones aimed at explaining the structure of the biological nucleic acids.45364wb00001,45364wb00002 TNA possesses the ability to specifically base-pair with RNA, DNA, and itself.45364wb00002 This capability, together with the chemical simplicity of threose relative to ribose, suggests that TNA could have acted as an evolutionary competitor of RNA or even have preceded RNA as the genetic molecule of life. We are attempting to investigate the functional potential of TNA by implementing an in vitro selection scheme for TNA.45364wb00003,45364wb00004 Here, we show that a mutant archaeal family B DNA polymerase is capable of polymerizing more than 50 nucleotides of TNA on a DNA template. We also demonstrate the display of single-stranded TNA covalently linked to its encoding duplex DNA, thus enabling the selection of functional TNA sequences and the amplification or recovery of the attached DNA.
Figure 1.
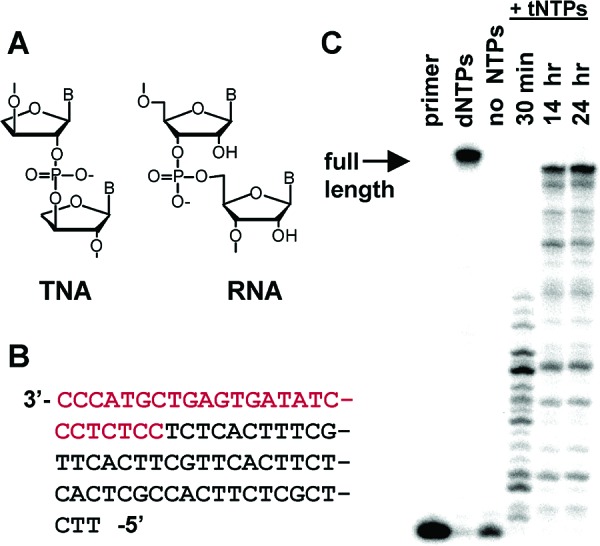
DNA-dependent TNA polymerization with the Therminator DNA polymerase. (A) Structures of TNA and RNA (B) Template sequence. Red = primer binding site; black = ssDNA template. (C) Time course of TNA polymerization. Reaction was performed with 5 nM primer/template, 60 μM tDTP, 31 μM tTTP, 18 μM tCTP, 2 μM tGTP, 1 unit ofTth pyrophosphatase, and 0.5 units of Therminator polymerase in 10 μL of 1× thermopol buffer (NEB) at 75 °C. Samples were boiled in 7 M urea and 400 mM NaOH and analyzed by 20% denaturing PAGE.
We and others have previously shown that certain family B archaeal DNA polymerases possess the ability to synthesize limited stretches of TNA on a DNA template.45364wb00005,45364wb00006 Our recent synthesis of all four TNA triphosphates (tNTPs) enabled us to test polymerases for more extensive activity.45364wb00007 The Therminator DNA polymerase is an engineered exonuclease-deficient form of “9°N” DNA polymerase containing an A485L mutation.45364wb00008 It is capable of efficiently incorporating a wide spectrum of modified nucleotides. We tested the ability of this polymerase to accept tNTPs as substrates using a DNA primer/template construct containing a 50-nucleotide single-stranded template region in which all four DNA nucleobases were represented (Figure 1B). Since previous work had shown that pairing diaminopurine opposite thymine increases the efficiencies of both template-directed ligation and polymerization,45364wb00005,45364wb00009 we used diaminopurine triphosphate (tDTP) instead of tATP. The Therminator polymerase catalyzed the synthesis of >20% full-length 50-nucleotide TNA product within 24 h (Figure 1C).
We reasoned that if transcribed TNA could be covalently linked to its DNA template, we could perform functional selections for TNA molecules and rescue the successful genotypes by PCR amplification of the attached DNA. This approach is analogous to the selection of functional peptides and proteins by mRNA display;45364wb00010 the use of DNA display for peptide and PNA selections has also been proposed.45364wb00010,45364wb00011 By starting with a library of single-stranded DNA hairpins, the 3‘ end of each hairpin could act as a primer for TNA transcription across the randomized DNA template region (Figure 2A). A primer annealed to the loop region of the hairpin could then initiate strand-displacement synthesis, liberating the TNA strand to allow folding and linearizing the DNA template by making it double-stranded.
Figure 2.
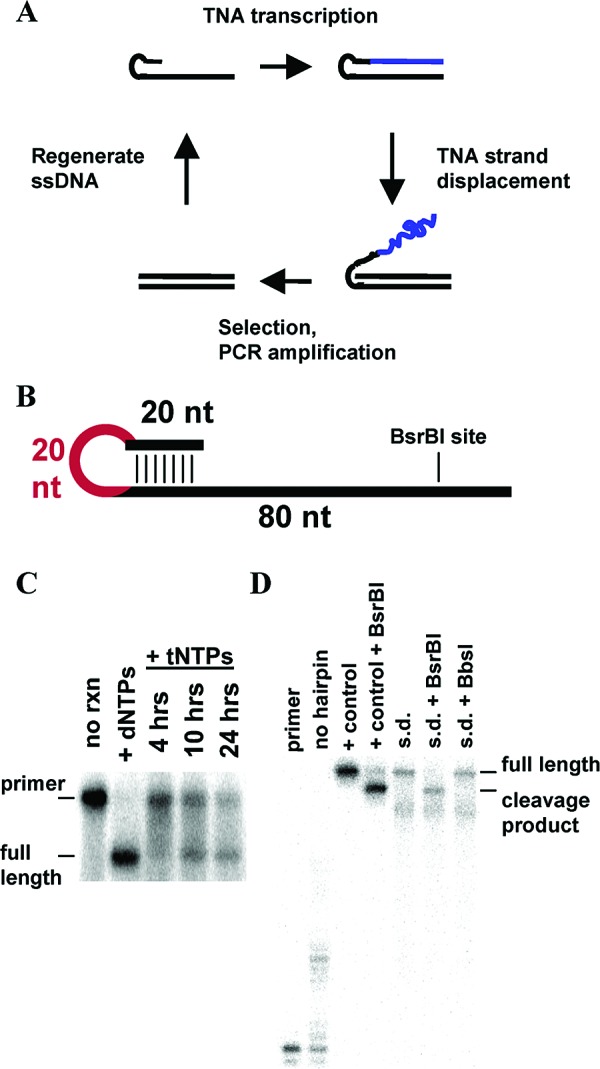
DNA-displayed TNA molecules. (A) In vitro selection scheme for TNA. (B) Synthetic DNA construct. Red represents loop region/strand displacement primer binding site. (C) TNA transcription of hairpin construct. 50 nM hairpin was transcribed with tNTPs for 12 h (∼13% fully extended) as in Figure 1C. Control reaction (+dNTPs) was performed for 10 min. Reactions were analyzed by 12% native PAGE. (D) Strand displacement (s.d.) of TNA strand. Full-length TNA-transcribed hairpins were gel-purified by native PAGE. End-labeled DNA primer (0.1 nM) was annealed to 1 nM transcribed hairpin by heating to 55 °C for 1 min in 1.1× thermopol buffer and 250 μM of each dNTP. One unit of Therminator was added, and the reactions were incubated for 30 min at 55 °C followed by 90 min at 75 °C. Restriction digests: strand displacement reactions were diluted 4-fold in 1× thermopol buffer, and 10 units of restriction enzyme were added for 2 h at 37 °C. Reactions were analyzed by 20% denaturing PAGE. “No hairpin” lane is a primer extension without the hairpin template. “+ control” lane is a primer extension on a single-stranded version of the hairpin constuct.
To test this idea, we synthesized a single-stranded DNA hairpin and transcribed the 60-nucleotide single-stranded region using the Therminator DNA polymerase and tNTPs (Figure 2B). Native gel analysis of the reaction indicated efficient TNA transcription (Figure 2C). Full-length transcripts were purified from the gel and subjected to a strand-displacement reaction with dNTPs, Therminator polymerase, and an end-labeled primer. With the hairpin construct in excess, most of the labeled primer was extended to full length, indicating very efficient strand displacement (Figure 2D). To verify that the resulting DNA−DNA duplexes were stable against strand-invasion by TNA, the strand-displaced constructs were treated with the restriction enzyme BsrBI, which specifically cleaves 15 base pairs from the end of the DNA template. The strand-displaced products were completely digested by BsrBI. BbsI, a control enzyme without a site in the DNA sequence, showed no activity (Figure 2D). BsrBI did not cleave ssDNA (data not shown).
In vitro selection for TNA function will only work if the TNA transcript is a reasonably faithful copy of its DNA template. The number of errors in a full-length TNA transcript is a function of both the misincorporation rate and the rate of primer extension from mismatched sites and is therefore time-dependent (since at longer times, full-length material accumulates by extension of strands that had paused at sites of misincorporation). To study the time- dependent fidelity of TNA synthesis by the Therminator polymerase, we examined the effect of omitting one tNTP at a time from short primer-extension reactions (Supporting Information Figure 1). For each reaction, the DNA template contained only one template site complementary to the omitted tNTP. This site was positioned 11 nucleotides downstream of the DNA primer terminus to ensure that fidelity was measured when the polymerase was fully contacting the TNA strand. A control reaction containing all four tNTPs was performed in parallel for each template. We compared the amount of full-length product in each −tNTP reaction versus control reactions at multiple time points to estimate the fidelity for each base. This assay accounts for both the slow rate of misincorporation as well as the slow rate of extension from mismatched primer termini, but underestimates the actual fidelity because of the lack of competition between correct and incorrect nucleotides. These experiments suggest that the Therminator polymerase incorporates tGTP and tCTP with ∼99 and ∼98% accuracy, respectively, in full-length TNA strands, even at relatively long incubation times (Supporting Information Table 1). The fidelities for incorporation of tDTP and tTTP were much lower (∼94% at 1 h, 10−20% of the primer fully extended), presumably reflecting misincorporation of tGTP and tCTP because of wobble base-pairing. Although the overall fidelity of TNA synthesis is lower than previously measured DNA synthesis fidelities,45364wb00012 more than 10% of TNA pool molecules would be error-free in material purified when TNA synthesis had reached 10−20% as we would do during a selection (Supporting Information Table 2). The actual error rate in material synthesized in the presence of all four tNTPs, and with high D/G and T/C ratios, may be much lower. Since typical in vitro selection experiments involve enrichment factors of more than 100−1000-fold per round, even a 10-fold reduction due to incorrectly transcribed sequences would yield an acceptable enrichment factor of at least 10−100-fold per round.
We have identified the Therminator DNA polymerase as an enzyme capable of reasonably efficient and faithful DNA-templated TNA polymerization. The DNA-display technique developed here for the selection of functional TNA molecules is uniquely suited for the directed evolution of unnatural nucleic acids because it does not require a polymerase that can copy the unnatural nucleic acid back into DNA. Thus, any polymer that can be synthesized by a DNA-templated reaction, either enzymatically or nonenzymatically,45364wb00011 can be used for selection. We hope to investigate the potential of TNA for forming ligand-binding sites or catalytically active structures in order to evaluate its possible role as a progenitor of RNA.
Acknowledgments
We thank R. Krishnamurthy and A. Eschenmoser for the generous gift of advanced intermediates for the synthesis of D, G, and T tNTPs. We are grateful to W. Jack and A. Gardner for very helpful discussions. This work was supported by grants from the NIH (R01 GM053936) and the NASA Astrobiology Institute (NNA04CC12A) to J.W.S. and a grant from the NSF (MCB0077667) to LWM. J.W.S. is an Investigator of the Howard Hughes Medical Institute. J.K.I. was supported in part by a Ford Foundation predoctoral fellowship.
Supporting Information Available
Supporting Information Available
List of oligonucleotide sequences, denaturing PAGE analysis of fidelity assays (Supporting Figure 1), and the fidelity values (Supporting Table 1). This material is available free of charge via the Internet at http://pubs.acs.org.
Funding Statement
National Institutes of Health, United States
Supplementary Material
References
- Eschenmoser A. Science 1999, 284, 2118–2124. 10.1126/science.284.5423.2118. [DOI] [PubMed] [Google Scholar]
- Schoning K.; Scholz P.; Guntha S.; Wu X.; Krishnamurthy R.; Eschenmoser A. Science 2000, 290, 1347–1351. 10.1126/science.290.5495.1347. [DOI] [PubMed] [Google Scholar]
- Carothers J. M.; Oestreich S. C.; Davis J. H.; Szostak J. W. J. Am. Chem. Soc. 2004, 126, 5130–5137. 10.1021/ja031504a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaput J. C.; Ichida J. K.; Szostak J. W. J. Am. Chem. Soc. 2003, 125, 856–857. 10.1021/ja028589k. [DOI] [PubMed] [Google Scholar]
- Chaput J. C.; Szostak J. W. J. Am. Chem. Soc. 2003, 125, 9274–9275. 10.1021/ja035917n. [DOI] [PubMed] [Google Scholar]
- Kempeneers V.; Vastmans K.; Rozenski J.; Herdewijn P. Nucleic Acids Res. 2003, 31, 6221–6226. 10.1093/nar/gkg833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou K.; Horhota A.; Chaput J. C.; Szostak J. W.; McLaughlin L. W. Org. Lett., to be submitted for publication.
- Gardner A. F.; Jack W. E. Nucleic Acids Res. 2002, 30, 605–613. 10.1093/nar/30.2.605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X.; Delgado G.; Krishnamurthy R.; Eschenmoser A. Org. Lett. 2002, 4, 1283–1286. 10.1021/ol020016p. [DOI] [PubMed] [Google Scholar]
- Kurz M.; Gu K.; Al-Gawari A.; Lohse P. A. ChemBioChem 2001, 2, 666–672. 10.1002/1439-7633(20010903)2:9%3C666::AID-CBIC666%3E3.0.CO;2-%23. [DOI] [PubMed] [Google Scholar]
- Rosenbaum D. M.; Liu D. R. J. Am. Chem. Soc. 2003, 125, 13924–13925. 10.1021/ja038058b. [DOI] [PubMed] [Google Scholar]
- Kunkel T. A.; Bebenek K. Annu. Rev. Biochem. 2000, 69, 497–529. 10.1146/annurev.biochem.69.1.497. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


