Abstract
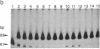
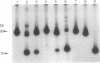
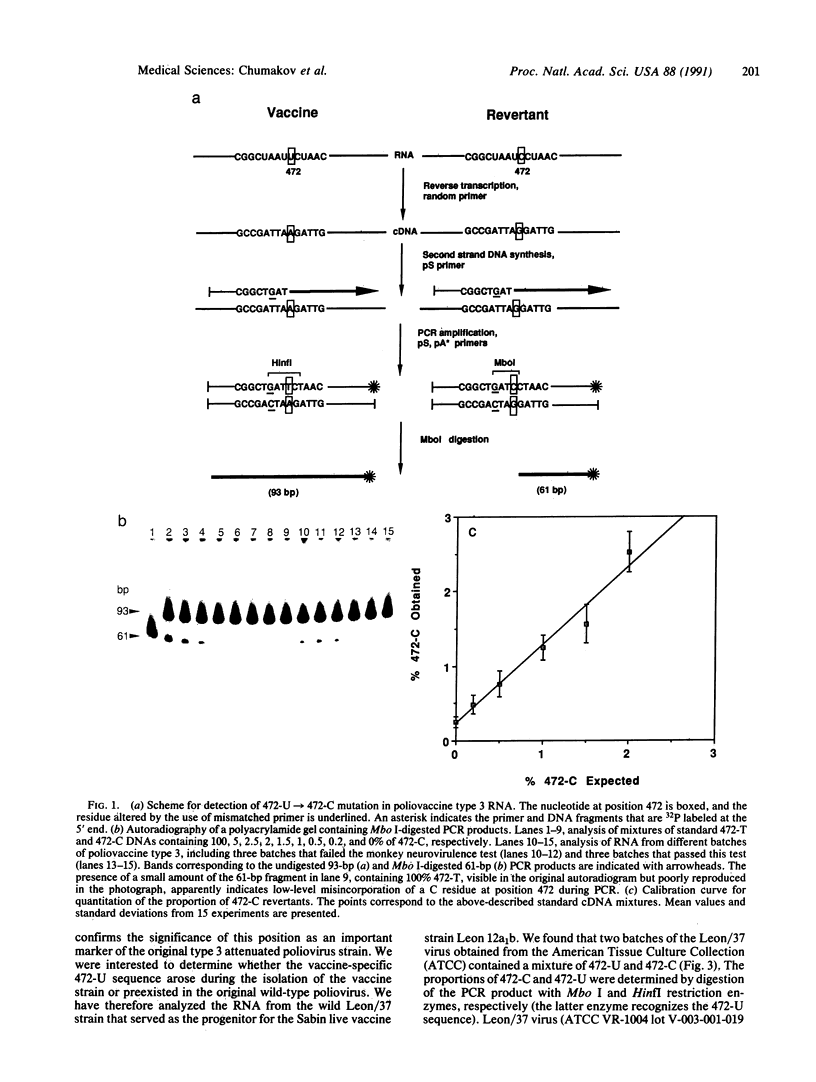
Production of live attenuated oral poliomyelitis vaccine (OPV) requires rigorous neurovirulence safety testing of each vaccine lot, currently carried out in monkeys. It has been reported that a change from 472-U to 472-C in the type 3 OPV RNA is associated with an increased histologic lesion score produced upon intraspinal inoculation of the mutant virus in monkeys. We have developed a method, based on polymerase chain reaction, for measuring the relative abundance of these mutant sequences directly in vaccine preparations and used this method to evaluate the proportion of 472-C in 40 different lots of type 3 OPV. Six vaccine lots that had failed the intraspinal monkey neurovirulence test contained a higher proportion of 472-C than all other lots that had passed this test. OPV type 3 virus containing 472-C was rapidly selected during serial passages in African green monkey kidney cells that are used for manufacturing of the vaccine. We have also found that the wild-type poliovirus type 3 strain Leon/37, from which the vaccine strain was originally derived, contained a mixture of 472-U and 472-C sequences. No other mutations in OPV type 3 RNA have been detected by similar assays at position 2034, also associated with attenuation, or at several other positions reported to be altered in some vaccine preparations. Our results suggest that molecular diagnostics may provide a supplement or a potential alternative to animal testing of live attenuated vaccines.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Evans D. M., Dunn G., Minor P. D., Schild G. C., Cann A. J., Stanway G., Almond J. W., Currey K., Maizel J. V., Jr Increased neurovirulence associated with a single nucleotide change in a noncoding region of the Sabin type 3 poliovaccine genome. Nature. 1985 Apr 11;314(6011):548–550. doi: 10.1038/314548a0. [DOI] [PubMed] [Google Scholar]
- Kawamura N., Kohara M., Abe S., Komatsu T., Tago K., Arita M., Nomoto A. Determinants in the 5' noncoding region of poliovirus Sabin 1 RNA that influence the attenuation phenotype. J Virol. 1989 Mar;63(3):1302–1309. doi: 10.1128/jvi.63.3.1302-1309.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- La Monica N., Racaniello V. R. Differences in replication of attenuated and neurovirulent polioviruses in human neuroblastoma cell line SH-SY5Y. J Virol. 1989 May;63(5):2357–2360. doi: 10.1128/jvi.63.5.2357-2360.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss E. G., O'Neill R. E., Racaniello V. R. Mapping of attenuating sequences of an avirulent poliovirus type 2 strain. J Virol. 1989 May;63(5):1884–1890. doi: 10.1128/jvi.63.5.1884-1890.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noonan K. E., Roninson I. B. mRNA phenotyping by enzymatic amplification of randomly primed cDNA. Nucleic Acids Res. 1988 Nov 11;16(21):10366–10366. doi: 10.1093/nar/16.21.10366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollard S. R., Dunn G., Cammack N., Minor P. D., Almond J. W. Nucleotide sequence of a neurovirulent variant of the type 2 oral poliovirus vaccine. J Virol. 1989 Nov;63(11):4949–4951. doi: 10.1128/jvi.63.11.4949-4951.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SABIN A. B. Characteristics and genetic potentialities of experimentally produced and naturally occurring variants of poliomyelitis virus. Ann N Y Acad Sci. 1955 Sep 27;61(4):924-38; discussion, 938-9. doi: 10.1111/j.1749-6632.1955.tb42551.x. [DOI] [PubMed] [Google Scholar]
- SABIN A. B. Present position of immunization against poliomyelitis with live virus vaccines. Br Med J. 1959 Mar 14;1(5123):663–680. doi: 10.1136/bmj.1.5123.663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabin A. B. Oral poliovirus vaccine: history of its development and use and current challenge to eliminate poliomyelitis from the world. J Infect Dis. 1985 Mar;151(3):420–436. doi: 10.1093/infdis/151.3.420. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Saiki R. K., Scharf S., Faloona F., Mullis K. B., Horn G. T., Erlich H. A., Arnheim N. Enzymatic amplification of beta-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science. 1985 Dec 20;230(4732):1350–1354. doi: 10.1126/science.2999980. [DOI] [PubMed] [Google Scholar]
- Stanway G., Cann A. J., Hauptmann R., Hughes P., Clarke L. D., Mountford R. C., Minor P. D., Schild G. C., Almond J. W. The nucleotide sequence of poliovirus type 3 leon 12 a1b: comparison with poliovirus type 1. Nucleic Acids Res. 1983 Aug 25;11(16):5629–5643. doi: 10.1093/nar/11.16.5629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanway G., Hughes P. J., Mountford R. C., Reeve P., Minor P. D., Schild G. C., Almond J. W. Comparison of the complete nucleotide sequences of the genomes of the neurovirulent poliovirus P3/Leon/37 and its attenuated Sabin vaccine derivative P3/Leon 12a1b. Proc Natl Acad Sci U S A. 1984 Mar;81(5):1539–1543. doi: 10.1073/pnas.81.5.1539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svitkin Y. V., Cammack N., Minor P. D., Almond J. W. Translation deficiency of the Sabin type 3 poliovirus genome: association with an attenuating mutation C472----U. Virology. 1990 Mar;175(1):103–109. doi: 10.1016/0042-6822(90)90190-3. [DOI] [PubMed] [Google Scholar]
- Toyoda H., Kohara M., Kataoka Y., Suganuma T., Omata T., Imura N., Nomoto A. Complete nucleotide sequences of all three poliovirus serotype genomes. Implication for genetic relationship, gene function and antigenic determinants. J Mol Biol. 1984 Apr 25;174(4):561–585. doi: 10.1016/0022-2836(84)90084-6. [DOI] [PubMed] [Google Scholar]
- Westrop G. D., Wareham K. A., Evans D. M., Dunn G., Minor P. D., Magrath D. I., Taffs F., Marsden S., Skinner M. A., Schild G. C. Genetic basis of attenuation of the Sabin type 3 oral poliovirus vaccine. J Virol. 1989 Mar;63(3):1338–1344. doi: 10.1128/jvi.63.3.1338-1344.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]