Abstract
Background
Congenital anomaly wherein the mitral valve leaflets are directly attached to the papillary muscle(s) (PM) with or without short under-developed chords is rarely reported in adults. Patients with two PMs with an intervening fibrous bridge have also been included under this head in previous studies.
Methods
Echocardiography enables accurate evaluation of the morphology and function of valve leaflets, chordae tendineae, and PM. This report describes a series of six patients aged 56–84 years who had abnormal mitral valve with a large solitary and anomalously inserted PM seen over a period of 3 years. Only those patients who had a single pillar or bridge-like PM and either absent tendinous chords or small under-developed chords were included in the analysis.
Results
Among 9600 consecutive echocardiograms performed, six patients met the criteria of an abnormal mitral valve with solitary large PM. Two patients underwent mitral valve replacement with partial excision of the PM wherein echocardiographic observations were confirmed. The patients were previously followed with the diagnosis of hypertrophic cardiomyopathy (3) and rheumatic mitral valve disease (3). Multi-planar reconstruction of 3D echocardiographic images provided incremental value in assessing the detailed patho-anatomy of PMs in these cases.
Conclusion
In adult patients, a high index of suspicion is required to detect congenital mitral stenosis/regurgitation with large solitary PM (resembling a parachute mitral valve) which may masquerade as hypertrophic cardiomyopathy or rheumatic mitral valve disease.
Keywords: Arcade mitral valve, Undifferentiated papillary muscle, Parachute mitral valve, Congenital mitral stenosis
1. Introduction
Congenital malformations of the mitral valve are often complex and affect multiple segments of the valve apparatus. These may occur in isolation or in association with other congenital heart defects. Incomplete differentiation of the leaflets and tensor apparatus produces anomalies of the mitral valve which may resemble an arcade, a hammock, or a parachute. The majority of mitral valve malformations are not classified, and descriptive terms (parachute, hammock, or arcade) often have overlapping patho-anatomy. The malformation of mitral valve in which the chordal apparatus is inserted into a single papillary muscle (PM) or a muscle group is called parachute deformity.1 The “parachute mitral valve” has the usual two mitral valvular leaflets and commissures, but the chordae, instead of diverging to insert into two PM, converge into one major PM. In “true” parachute mitral valve (PMV), mitral valve chordae insert into a solitary central PM. According to Tandon et al., PMV is associated with other congenital anomalies in >95% cases.2 Anomalous mitral arcade was first described by Layman and Edwards in 1967 as “an anomaly of the mitral valve that consisted of connection of the left ventricular (LV) papillary muscles to the anterior mitral valve leaflet, either directly or through the interposition of unusually short tendinous chords”.3 The two PMs resemble two pillars, and the bridging fibrous tissue in between the PM muscles resembles the arch of an arcade. Undifferentiated PM has been defined as a PM with club-like thickening, often attached to the leaflets directly due to a defect or marked under-development of chordae tendineae that leads to compromised flexibility of PM/chordae tendineae/valve leaflet assembly.4 Others have called this anomaly of single PM with, often short and fused tendinous chords and narrow interchordal spaces seen in adults also as the PMV.5 However, a true PMV mitral valve has a single central PM muscle with unifocal attachment of chords, regardless of the anomalies of the chords and is mostly associated with other congenital anomalies. Mitral valve abnormalities with short thick chords or direct attachment of PM to the anterior mitral leaflet have been also reported in the hypertrophic cardiomyopathy6, 7; however, single or undifferentiated PM has not been described. Carpentier et al. classified the congenital mitral valve abnormalities into four types depending upon the presence and type of mitral regurgitation and/or mitral stenosis regardless of the morphology of the tensor apparatus and refrained from using descriptive names.8
This report deals with clinical and echocardiographic spectrum of significant mitral valve dysfunction with undifferentiated solitary PM in the adults seen over a period of 3 years in a busy echocardiographic laboratory.
2. Case 1
A 56-year-old man presented with exertional dyspnea of 4 years duration. He had been previously diagnosed as a case hypertrophic cardiomyopathy and alcohol septal ablation was also suggested. The 12-lead electrocardiogram showed left atrial overload and complete right bundle branch block. The chest skiagram showed cardiomegaly with pulmonary venous congestion. His coronary angiogram revealed mild stenosis of the distal left circumflex artery. Echocardiographic examination showed dilated inferior vena cava, right atrium, and the right ventricle with severe tricuspid regurgitation. Doppler-estimated pulmonary artery systolic pressure was 110 mmHg. The left atrium was enlarged; there was a solitary large PM attached to the lateral and the posterior wall and appeared like a cantilever bridge in the short axis (Fig. 1).
Fig. 1.
2D-echocardiographic short axis view showing dumbbell-shaped anomalous papillary muscle in the left panel. The right panel shows mid-ventricular short axis after excision of the muscle bundle.
This anomalous muscle bundle also had fibromuscular attachments to the interventricular septum (Fig. 2).
Fig. 2.
Modified apical 4-chamber view showing anomalous papillary muscle with attachment to the interventricular septum (yellow arrow). The right panel shows the residual portion of the muscle bundle (yellow arrow) after excision.
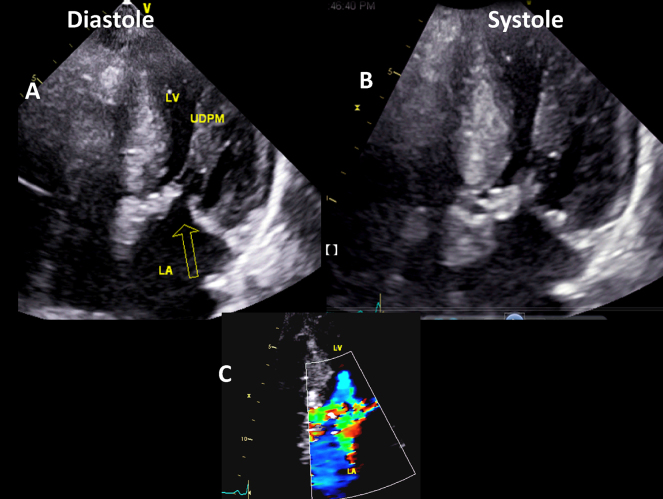
Both mitral valve leaflets were directly attached to this solitary anomalous muscle bundle with no intervening tendinous chords (Fig. 3).
Fig. 3.
Apical 4-chamber view in diastole (left) and 5-chamber view in systole (right panel) showing direct attachment of the mitral leaflets to the papillary muscle.
Mitral valve area by planimetry and the pressure half-time method was 1.2 cm2 with a mean gradient of 6 mmHg. Moderate mitral regurgitation (vena contracta = 5 mm) and an intra-ventricular peak gradient of 30 mmHg were recorded (Fig. 4).
Fig. 4.
Left panel shows anomalous papillary muscle in the modified short axis view. The right upper panel shows continuous-wave Doppler spectrum of mitral regurgitation and mid-cavity obstruction (red arrow). The right lower panel shows continuous-wave Doppler spectrum across the mitral funnel.
The patient underwent cardiac catheterization which showed hardly any gradient between pulmonary wedge and LV diastolic pressures although pulmonary artery pressure was recorded as 115/40 mmHg. The patient was subjected to excision of the muscle bundle along with valve leaflets and was implanted with a #27 St Jude's prosthesis in mitral position. The echocardiographic observations were confirmed on gross pathology.
3. Case 2
This 59-year-old female was being followed as a case of hypertrophic cardiomyopathy for the last 7 years. She had class III exertional dyspnea. Physical examination was conspicuous by a loud parasternal ejection systolic murmur, supine blood pressure of 130/90 mmHg, and no jugular venous distension. The 12-lead ECG showed sinus rhythm, left atrial overload, and LV hypertrophy with strain. The chest skiagram was unremarkable. 2D-echocardiographic examination revealed uniformly thick walls of the left ventricle (14 mm), solitary anomalous PM attached to the apicolateral segment, and both mitral leaflets were thick and club-like and attached to the PM through small chords. Severe eccentric mitral regurgitation and a peak gradient of 50 mmHg were recorded in the LV outflow tract. Trans-mitral peak and mean gradients were 8 and 3 mmHg respectively. The patient underwent extended myomectomy, excision of the PM, and mitral valve replacement (Fig. 5, Fig. 6, Fig. 7).
Fig. 5.
Apical 4-chamber (A) and parasternal long axis (B) views showing thick mitral valve leaflets, short tendinous chords, and a single anomalous papillary muscle. PM, papillary muscle; LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
Fig. 6.
Reduced opening of the mitral orifice in diastole (A) and narrow inter-chordal spaces. Short chords are seen in panel B during systole. Panel C shows severe eccentric mitral regurgitation.
Fig. 7.
Parasternal long axis view (left panel) and the apical 5-chamber view (right panel) showing narrow left ventricular outflow tract due to anteriorly inserted papillary muscle and the abnormal orientation of the thickened anterior leaflet.
4. Case 3
An 84-year-old female was followed as a case of rheumatic mitral valve disease with atrial fibrillation for the last 30 years. She had class II dyspnea and declined any surgery. Physical examination revealed a thin-built elderly lady, BP 130/86 mmHg, irregularly irregular heart rate of 68 BPM, and a mid-diastolic murmur at the apex. The 12-lead electrocardiogram revealed atrial fibrillation and non-specific ST-T changes. The chest skiagram showed mild cardiomegaly and pulmonary venous congestion. Her trans-thoracic 2D echocardiographic examination revealed markedly enlarged left atrium, reduced motion of the mitral leaflets with restricted opening, single large PM attached to the inferolateral and anterolateral walls, short chordae, and mitral valve area of 1.4 cm2 (Fig. 8, Fig. 9, Fig. 10). There was no calcification of the mitral valve leaflets. Moderate mitral regurgitation was observed on color Doppler interrogation.
Fig. 8.
2D-echocardiographic parasternal long axis view in systole (A) and in diastole (B). Note reduced opening of the mitral orifice with short chords.
Fig. 9.
Parasternal biplane views (long axis + short axis) showing near direct attachment of the papillary muscle to the leaflets and reduced oval mitral valve orifice (arrow, right panel).
Fig. 10.
Left lower panel shows multi-plane 3D reconstruction of the left ventricular mid-cavity showing a large anomalous papillary muscle (arrow) attached from 1 o’clock till 6 o’clock position. Right panel shows continuous wave Doppler interrogation of the mitral funnel and a peak gradient of 8 mmHg.
5. Case 4
A 69-year-old female presented with exertional dyspnea of 2 years duration. She had long-standing systemic hypertension and was recently diagnosed with type II diabetes mellitus. Her previous echocardiogram was reported as showing mitral annular calcification and she had the clinical diagnosis of heart failure with preserved ejection fraction and atrial fibrillation. Her echocardiographic examination revealed calcification of the posterior mitral annulus, reduced mitral valve opening with normal thickness of the leaflets (mitral valve area of 1.3 cm2), short tendinous chords, and single large PM attached to the LV free wall. Color Doppler interrogation of the mitral valve revealed flame-shaped ante-grade flow spectrum and moderate mitral regurgitation. Continuous-wave Doppler examination showed trans-mitral peak and mean gradients of 15 and 7 mmHg respectively (Fig. 11, Fig. 12).
Fig. 11.
Left panel (A) shows single large papillary muscle in short axis view (red arrow) while right panel (B) shows short chords, calcification of the posterior annulus, and a large, thick club-like papillary muscle in apical 4-chamber view.
Fig. 12.
Diastolic frames in figures A and B showing normal anterior mitral leaflet, calcified posterior annulus. Figure C shows solitary papillary muscle in short axis. Figure D reveals typical Doppler spectrum of mitral stenosis and regurgitation.
6. Case 5
A 63-year-old male was diagnosed as hypertrophic cardiomyopathy 13 years back and had class II dyspnea. Detailed echocardiographic examination showed thick LV walls, asymmetric septal hypertrophy, solitary PM, and short chords with systolic anterior motion. Peak gradient across mitral valve in diastole was 7 mmHg and moderately severe mitral regurgitation was noted (Fig. 13, Fig. 14).
Fig. 13.
Left panel shows parasternal long axis view in systole with two heads of a single papillary muscle and short chords. Right panel shows single papillary muscle in short axis view.
Fig. 14.
Continuous-wave Doppler interrogation of the mitral valve showing spectrum of mitral regurgitation (upper panel). The lower panel shows pulsed-wave Doppler spectrum with a peak A velocity of 1.6 m/s.
7. Case 6
A 69-year-old female was a known case of mitral stenosis with exertional dyspnea. She was unwilling for any intervention and had been followed as a case of rheumatic heart disease for a very long time. Recent echocardiogram revised the etiology of mitral stenosis. Current echocardiogram shows thickened mitral valve leaflets with no commissural fusion, short chords, and a single large PM placed centrally (Fig. 15, Fig. 16). There was no involvement of other valves and there was no calcification. Mitral valve area by planimetry was 1.1 cm2 and mean diastolic gradient across the mitral valve was 10 mmHg.
Fig. 15.
Upper panel shows continuous wave Doppler spectrum of mitral stenosis with a mean gradient of 10 mmHg. Lower left panel shows narrow jet of mitral diastolic flow and the right lower panel in short axis view shows narrow orifice of the mitral valve with no commissural fusion or calcification.
Fig. 16.
Multi-planar 3D reconstruction showing single papillary muscle in panel A (short axis) and narrow mitral valve orifice as viewed from the left ventricular side (panel B).
8. Discussion
The abnormalities of PM/chordae tendineae of the mitral valve are a rare form of congenital mitral valve dysplasia which may remain asymptomatic in life-time or may present with mitral valve stenosis or regurgitation or both. In the developmental process of mitral valve, chordae tendineae and PM, a prominent horseshoe-shaped myocardial ridge appears from the anterior wall of the LV and extends to the posterior wall of the LV approximately on the 33rd fetal day, and this ridge connects with the endocardial cushion tissue in the atrio-ventricular region.9 Afterwards, the myocardial ridge transforms into two PMs and part of the endocardial cushion tissue differentiates into valve leaflets and chordae tendineae.9 Disturbed delamination of the anterior or posterior part of the trabecular ridge from the ventricular wall, combined with underdevelopment of chordae, seems to be the cause of asymmetric mitral valves. PMV, however, develops when the connection between the posterior and anterior part of the ridge condenses to form one single PM. Oosthoek et al.10 hypothesized that the myocardial ridge of the LV wall connects with the endocardial cushion tissue at the atrio-ventricular septum and differentiates into PM and chordae tendineae at 5–19 fatal weeks. PM are found attached to valve leaflets without the involvement of chordae tendineae in cases of poor differentiation of endocardial cushion into chordae tendineae. They also reported 28 cases of PM–chordae tendineae abnormalities in which one PM was developed from the high level of the LV and attached to AML directly or with slight mediation of chordae tendineae and called them asymmetric parachute-like mitral valves. This report does not include such cases. The spectrum of PMs in four of our cases (cases 1–4) resembles non-delamination and non-separation of the embryonic trabecular ridge.
On the other hand, PMV refers to an anomaly of the mitral valve apparatus where all tendinous cords insert into one PM.1, 2 The other PM is either absent or severely hypoplastic. The mitral valve through the incised LV has a parachute-like appearance because of normally developed tendinous chords. PMV is a rare congenital anomaly of the mitral valve apparatus seen in infants and young children. In most instances, PMV is associated with other congenital anomalies of the heart, in particular obstructive lesions of the mitral inflow (mitral valve ring) and LV outflow tract (subaortic stenosis), and coarctation of aorta and is referred to as Shone's complex or Shone's anomaly.2, 11 Others later observed that in the setting of isolated PM, the tendinous chords are often short and fused with interchordal spaces partially or completely obliterated.5 PMV is commonly classified as a malformation of the PMs. However, that is too simplistic. It is often the associated lesions of mitral valve segments (e.g., commissural underdevelopment, dysplastic leaflets, shortened, and fused tendinous chords) that determine the severity of valvar dysfunction (stenosis and regurgitation) and hence the need for cardiac intervention. The combination of these lesions can give rise to a funnel rather than parachute-like appearance as seen in six of the cases reported in the present work. A series of six such cases presenting as mitral regurgitation and confirmed on surgery have been recently reported.4 Despite two series of ten and six cases,4, 5 sketchy information is available about PMV or undifferentiated PM with mitral valve dysplasia in adults as an isolated anomaly or in association with other congenital cardiac anomalies.12, 13, 14, 15, 16, 17, 18 The clinical course of isolated PMV is varied depending on the presence and severity of mitral stenosis and regurgitation. Four of our patients presented with predominant mitral stenosis while two had significant mitral regurgitation. It has been reported that many patients may remain asymptomatic even in adulthood.19, 20
9. Conclusions
Mitral stenosis or mitral regurgitation associated with undifferentiated PM results from tethering and impaired flexibility of leaflets. It is possible to successfully treat the patients by mitral valve replacement and excision of the PM wholly or in part, unless complex congenital cardiac malformations coexist. Detailed examination of the insertion and bridging of the PM by echocardiography and intraoperative inspection is necessary and surgical techniques should be selected appropriately in each case. From the aspect of long-term prognosis, it is well known that mitral valve repair is superior to mitral valve replacement. However, the results of the mitral valvuloplasty for congenital mitral valve disease in the adult are not incompletely clarified. Repair is feasible in case there is predominant regurgitation with well-developed chords. Recently, repair in one of such cases has been reported.14 It is prudent to follow Carpentier's approach in all such cases.8
Conflicts of interest
The authors have none to declare.
References
- 1.Schiebler G.L., Edwards J.E., Burchell H.B., Dushane J.W., Ongley P.A., Wood E.H. Congenital corrected transposition of the great vessels: a study of 33 cases. Pediatrics. 1961;27:851–854. [PubMed] [Google Scholar]
- 2.Tandon R., Moller J.H., Edwards J.E. Anomalies associated with the parachute mitral valve a pathologic analysis of 52 cases. Can J Cardiol. 1986;2:278–281. [PubMed] [Google Scholar]
- 3.Layman T.E., Edwards J.E. Anomalous mitral arcade: a type of congenital mitral insufficiency. Circulation. 1967;35:389–395. doi: 10.1161/01.cir.35.2.389. [DOI] [PubMed] [Google Scholar]
- 4.Matsumaru I., Hashizume K., Ariyoshi T. Characteristics and treatment strategies of mitral regurgitation associated with undifferentiated papillary muscle. Gen Thorac Cardiovasc Surg. 2012;60:406–410. doi: 10.1007/s11748-012-0055-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hakim F.A., Kendall C.B., Alharthi M., Mancina J.C., Tajik J.A., Mookadam F. Parachute mitral valve in adults – a systematic overview. Echocardiography. 2010;27:581–586. doi: 10.1111/j.1540-8175.2009.01143.x. [DOI] [PubMed] [Google Scholar]
- 6.Cavalcante J.L., Barboza J.S., Lever H.M. Diversity of mitral valve abnormalities in obstructive hypertrophic cardiomyopathy. Prog Cardiovasc Dis. 2012;54:517–522. doi: 10.1016/j.pcad.2012.03.002. [DOI] [PubMed] [Google Scholar]
- 7.Maron M.S., Olivotto I., Harrigan C. Mitral valve abnormalities identified by cardiovascular magnetic resonance represent a primary phenotypic expression of hypertrophic cardiomyopathy. Circulation. 2011;124:40–48. doi: 10.1161/CIRCULATIONAHA.110.985812. [DOI] [PubMed] [Google Scholar]
- 8.Carpentier A., Branchini B., Cour J.C. Congenital malformations of the mitral valve in children: pathology and surgical treatment. J Cardiovasc Surg. 1976;72:854–866. [PubMed] [Google Scholar]
- 9.Ando M. Shinzou-nohattatsu-to-keitai. In: Takao A., editor. Rinshouhattatsusinzoubyogaku. 3rd ed. Chugai-igakusha; Tokyo: 2001. pp. 21–42. [Google Scholar]
- 10.Oosthoek P.W., Wenink A.C., Wisse L.J., Gittenberger-de Groot A.C. Development of the papillary muscles of the mitral valve: morphogenetic background of parachute-like asymmetric mitral valves and other mitral valve anomalies. J Thorac Cardiovasc Surg. 1998;116:36–46. doi: 10.1016/S0022-5223(98)70240-5. [DOI] [PubMed] [Google Scholar]
- 11.Shone J.D., Sellers R.D., Anderson R.C., Adams P., Lillehei C.W., Edwards J.E. The developmental complex of “parachute mitral valve,” supravalvular ring of left atrium subaortic stenosis and coarctation of aorta. Am J Cardiol. 1963;11:714–725. doi: 10.1016/0002-9149(63)90098-5. [DOI] [PubMed] [Google Scholar]
- 12.López-Pardo F., Urbano-Moral J.A., González-Calle A. Three-dimensional transesophageal echocardiography in the anatomical assessment of isolated parachute mitral valve in an adult patient. Echocardiography. 2015 doi: 10.1111/echo.13003. [June 20] [DOI] [PubMed] [Google Scholar]
- 13.Isılak Z., Deveci O.S., Yalcın M., Uz O., Dogan M., Uzun M. A unique case of parachute mitral valve in an adult: an abnormally long chorda tendinea. Echocardiography. 2012;29:E245–E246. doi: 10.1111/j.1540-8175.2012.01778.x. [DOI] [PubMed] [Google Scholar]
- 14.Shiraishi M., Yamaguchi A., Adachi H. Successful surgical repair of the parachute mitral valve with mitral valve regurgitation. Ann Thorac Cardiovasc Surg. 2012;18:569–572. doi: 10.5761/atcs.cr.11.01841. [DOI] [PubMed] [Google Scholar]
- 15.Espinola-Zavaleta N., Chugh R., Ramírez G.M. Parachute mitral valve with severe mitral regurgitation in an adult patient. Echocardiography. 2012;29:E122–E125. doi: 10.1111/j.1540-8175.2011.01620.x. [DOI] [PubMed] [Google Scholar]
- 16.Rybicka J., Dobrowolski P., Kuśmierczyk M., Rózański J., Kowalski M., Hoffman P. Parachute mitral valve in a young adult with recurrent pulmonary oedema. Acta Cardiol. 2011;66:401–403. doi: 10.1080/ac.66.3.2114147. [DOI] [PubMed] [Google Scholar]
- 17.Purvis J., Sharma D. Adult parachute mitral valve detected in pregnancy. Heart. 2011;97:1192. doi: 10.1136/heartjnl-2011-300016. [DOI] [PubMed] [Google Scholar]
- 18.Purvis J.A., Smyth S., Barr S.H. Multi-modality imaging of an adult parachute mitral valve. J Am Soc Echocardiogr. 2011;24:e1–e3. doi: 10.1016/j.echo.2010.07.002. [DOI] [PubMed] [Google Scholar]
- 19.Erdogan O., Aktoz M. Parachute mitral valve abnormality and bicuspid aortic valve in an asymptomatic adult patient. Can J Cardiol. 2008;24:e57. doi: 10.1016/s0828-282x(08)70660-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Patsouras D., Korantzopoulos P., Kountouris E., Siogas K. Isolated parachute mitral valve as an incidental finding in an asymptomatic hypertensive adult. Clin Res Cardiol. 2007;96:38–41. doi: 10.1007/s00392-006-0449-9. [DOI] [PubMed] [Google Scholar]