Abstract
OBJECTIVE
Fibroblast growth factor 21 (FGF21) is involved in the regulation of energy balance and adipose metabolism. Our previous genome-wide association study identified genetic variants in the FGF21 region associated with macronutrient intake preference. We investigated whether the FGF21 genotype modified effects of weight-loss diets varying in macronutrient intake on changes in adiposity in a 2-year randomized diet intervention trial.
RESEARCH DESIGN AND METHODS
We genotyped FGF21 rs838147 in 715 overweight or obese individuals who were assigned to one of four diets varying in macronutrient contents. A DEXA scan was performed to evaluate body composition.
RESULTS
We observed a significant interaction between the FGF21 genotype and carbohydrate/fat intake on 2-year changes in waist circumference (WC), percentage of total fat mass, and percentage of trunk fat (P = 0.049, P = 0.001, and P = 0.003 for interaction, respectively). In response to the low-carbohydrate/high-fat diet, carrying the carbohydrate intake–decreasing C allele of rs838147 was marginally associated with less reduction in WC (P = 0.08) and significantly associated with less reduction of total fat mass (P = 0.01) and trunk fat (P = 0.02). Opposite genetic associations with these outcomes were observed among the high-carbohydrate/low-fat diet group; carrying the C allele was associated with a greater reduction of WC, total body fat mass, and trunk fat.
CONCLUSIONS
Our data suggest that FGF21 genotypes may interact with dietary carbohydrate/fat intake on changes in central adiposity and body fat composition. A low-calorie, high-carbohydrate/low-fat diet was beneficial for overweight or obese individuals carrying the carbohydrate intake–decreasing allele of the FGF21 variant to improve body composition and abdominal obesity.
Introduction
Macronutrients, including fat, carbohydrate, and protein, are primary sources of energy and play a central role in determining energy balance, which regulates body fat accumulation and weight reduction (1,2). In our recent genome-wide association studies (GWAS), we identified sequence polymorphisms close to the fibroblast growth factor 21 (FGF21) gene that were associated with intakes of macronutrients (3,4). Our studies consistently showed that the genetic variation in the FGF21 region was associated with carbohydrate or fat intake (3,4). Prior studies reported that circulating levels of FGF21 increased in response to carbohydrate intake (5,6), and more recent studies provided possible mechanisms for the role of FGF21 in regulating “sweet taste preference” (7–9).
FGF21 has been attracting great interest as a whole-body metabolic regulator of energy balance and adipose metabolism (10,11) and also as a therapeutic potential for treatment of obesity (10). Several studies found that elevated blood concentrations of FGF21 were positively correlated with BMI (12–16), waist circumference (WC) (12,14,16,17), fat mass (13,16), or visceral adipose area (18,19), suggesting obesity as a FGF21-resistant state (20). A study also showed a significant association between circulating FGF21 levels and fasting-mediated weight loss in healthy subjects (21).
Although compelling evidence has shown that dietary interventions with modifications of macronutrients may promote weight loss and improve body fat distribution (2,22,23), considerable interindividual variance has long been noted in response to dietary interventions, and genetic variations may at least partly account for such variance. We hypothesized that the macronutrients-associated FGF21 genotype might modify the effects of dietary interventions varying in macronutrient intakes on changes in adiposity and body composition. The Preventing Overweight Using Novel Dietary Strategies (POUNDS) Lost trial is thus far one of the largest randomized intervention trials comparing diets varying in macronutrients on long-term weight loss (24). In this study, we investigated whether the GWAS-identified macronutrient intake–associated variant in the FGF21 gene modified effects of weight-loss diets on the improvement of adiposity and body composition in the POUNDS Lost trial.
Research Design and Methods
Study Participants
The POUNDS Lost trial was conducted from October 2004 through December 2007 at two sites: Harvard T.H. Chan School of Public Health and Brigham and Women’s Hospital in Boston, MA, and the Pennington Biomedical Research Center of Louisiana State University System in Baton Rouge, LA. Details of the study have been described previously (24). Briefly, this study included 811 overweight or obese individuals who were randomly assigned to one of four energy-reduced diets varying in macronutrient compositions of fat, protein, and carbohydrate to compare their effects on body weight change during a 2-year follow-up time. The targeted percentages of energy derived from fat, protein, and carbohydrates in the four diets were 1) 20, 15, and 65%; 2) 20, 25, and 55%; 3) 40, 15, and 45%; and 4) 40, 25, and 35%, respectively. Thus, two diets were low fat (20%), and the other two diets were high fat (40%); two diets were average protein (15%), and the other two diets were high protein (25%), which constituted a two-by-two factorial design. The low-fat/high-fat diets were the same as the high-carbohydrate/low-carbohydrate diets in the POUNDS Lost Study. The low-carbohydrate/high-fat diet was indicated by 35% or 45% carbohydrate/40% fat of energy; the high-carbohydrate/low-fat diet was indicated by 55% or 65% carbohydrate/20% fat of energy in this study. The study was approved by the human subjects committee at each institution and by a data and safety monitoring board appointed by the National Heart, Lung, and Blood Institute. All participants gave written informed consent.
In the current study, we investigated the data of 715 overweight or obese individuals (mean age 50.9 [SD 9.3] years) who had genotyping data on the FGF21 variant rs838147 at the baseline examination. Of the study participants, 573 (80%) were white, 106 (15%) were African American, 25 (3%) were Hispanic, and 11 (22%) were Asian or other ethnic groups by self-report. WC data were available for 639 individuals at 6 months and for 541 at 2 years. For the assessment of body composition, a random sample of ∼50% of the total study participants were selected to undergo DEXA scans for fat mass and lean mass at baseline and after 6 months and 2 years of randomization (25). The DEXA scan was performed in 373 at the baseline examination, in 297 at 6 months, and in 211 at 2 years.
Measurements
Body weight and WC were measured at baseline, 6, 12, and 18 months, and 2 years. Height was measured at the baseline examination. BMI was calculated as weight in kilograms divided by the square of height in meters (kg/m2). The DEXA scan was performed using a Hologic QDR 4500A (Hologic, Inc.) after an overnight fast. Total fat mass, total lean mass, whole-body total percentage fat mass, and trunk fat percentage were measured at baseline, 6 months, and 2 years of follow-up. Dietary intake was assessed in a random sample of 50% of the total participants by a review of 5-day diet records at baseline and by 24-h recall during a telephone interview on 3 nonconsecutive days at 6 months and at 2 years to assess the adherence to the dietary intervention program.
Genotyping
DNA was extracted from the buffy coat fraction of centrifuged blood using the QIAamp Blood Kit (Qiagen). Our previously reported single-nucleotide polymorphism (SNP) in the FGF21 locus rs838147 was genotyped (3). Genotyping was performed using the OpenArray SNP Genotyping System (BioTrove). The C allele of the rs838147 was associated with lower carbohydrate intake (3). The genotype success rate was 99%. The genotype frequencies were in Hardy-Weinberg equilibrium among all study participants or among two major (white and African American) ethnic groups (P > 0.05).
Statistical Analysis
General linear models for continuous variables and χ2 test for categorical variables were performed for comparison of characteristics across the genotype at baseline examination. The primary outcomes were changes (Δ) in body weight, WC, and body composition assessed by DEXA (total fat, total lean, total fat mass percentage, and trunk fat percentage) among participants who remained in the trial. General linear models were used to compare changes from baseline in these outcomes across genotype groups according to low- or high- carbohydrate diet group at 6-month and 2-year visits with an adjustment of age, sex, ethnicity, and baseline value for the respective outcome (model 1). We also analyzed data with BMI at the baseline examination being further included in the adjusted model (model 2). Additive genetic models were used in the analysis. To test potential gene–diet interactions, a genotype-by-diet product term (e.g., FGF21 genotype × high-/low-carbohydrate diet group) was included in the models. We also performed similar analyses only among white participants. We performed an additional analysis using linear mixed model with time variable as a repeated measurement factor. We tested genetic associations with the trajectory of changes in WC and body composition over the 2-year intervention by including genotype–time interaction terms. Statistical analyses were performed with SAS 9.3 software (SAS Institute Inc.). The statistical significance was considered for P < 0.05.
Results
The frequency of the FGF21 rs838147 carbohydrate intake–decreasing C allele was 44.8% among the study participants. The genotype distribution did not differ by sex or the carbohydrate diet interventions, whereas a significant difference was observed across ethnicity (P < 0.001). No significant difference was found in age, BMI, body weight, WC, and body composition according to the FGF21 rs838147 genotype (Table 1) at the baseline examination; however, the C allele was positively correlated with total energy intake (P = 0.02). We did not observe significant main associations of the FGF21 genotype with changes in body weight, WC, or body composition (total fat, total lean, total fat mass percentage, or trunk fat percentage) during the 2-year intervention after adjustment for age, sex, ethnicity, BMI at baseline, low-/high-carbohydrate diet group, and baseline value for the respective outcome (P > 0.05 for all the outcomes, data not shown).
Table 1.
Characteristics of study participants at the baseline examination according to the FGF21 genetic variant rs838147
| TT | TC | CC | ||
|---|---|---|---|---|
| n = 211 | n = 367 | n = 137 | P value | |
| Age, years | 50.6 (9.1) | 51.1 (9.4) | 50.9 (9.4) | 0.76 |
| Male sex | 75 (35.6) | 147 (40.1) | 57 (41.6) | 0.44 |
| Race or ethnic group | <0.001 | |||
| White | 150 (71.1) | 306 (83.4) | 117 (85.4) | |
| Black | 50 (23.7) | 42 (11.4) | 14 (10.2) | |
| Hispanic or other | 11 (5.2) | 19 (5.2) | 6 (4.4) | |
| Diet group | 0.28 | |||
| Low carbohydrate | 114 (54.0) | 173 (47.1) | 68 (49.6) | — |
| High carbohydrate | 97 (46.0) | 194 (52.9) | 69 (50.4) | — |
| Dietary intake per day* | ||||
| Energy, kcal | 1,892 (545) | 1,976 (555) | 2,099 (617) | 0.02 |
| Protein, % | 18.3 (2.9) | 18.1 (3.6) | 17.6 (2.9) | 0.27 |
| Fat, % | 37.7 (6.3) | 36.8 (5.9) | 36.4 (5.9) | 0.16 |
| Carbohydrate, % | 43.9 (7.3) | 44.7 (7.8) | 45.5 (7.9) | 0.17 |
| BMI, kg/m2 | 32.6 (3.8) | 32.8 (3.8) | 32.5 (4.1) | 0.997 |
| Body weight, kg | 92.2 (14.5) | 93.7 (15.9) | 94.1 (16.6) | 0.24 |
| WC, cm | 102.3 (12.7) | 104.4 (12.9) | 103.8 (13.9) | 0.19 |
| Body composition data** | ||||
| Whole-body fat, kg | 34.4 (7.9) | 35.3 (7.6) | 35.9 (8.5) | 0.20 |
| Whole-body lean, kg | 58.7 (12.6) | 61.0 (13.3) | 61.4 (14.2) | 0.14 |
| Whole-body fat mass, % | 37.1 (7.0) | 36.9 (6.7) | 37.1 (7.3) | 0.96 |
| Trunk fat, % | 38.0 (6.1) | 38.1 (5.8) | 38.1 (6.5) | 0.87 |
Data are mean (SD) or n (%).
*Data for dietary intake per day were available for 366 individuals across the three genotype groups: TT, n = 111; TC, n = 194; and CC, n = 61.
**Data for body composition were available for 373 individuals across the three genotype groups: TT, n = 113; TC, n = 189; and CC, n = 71.
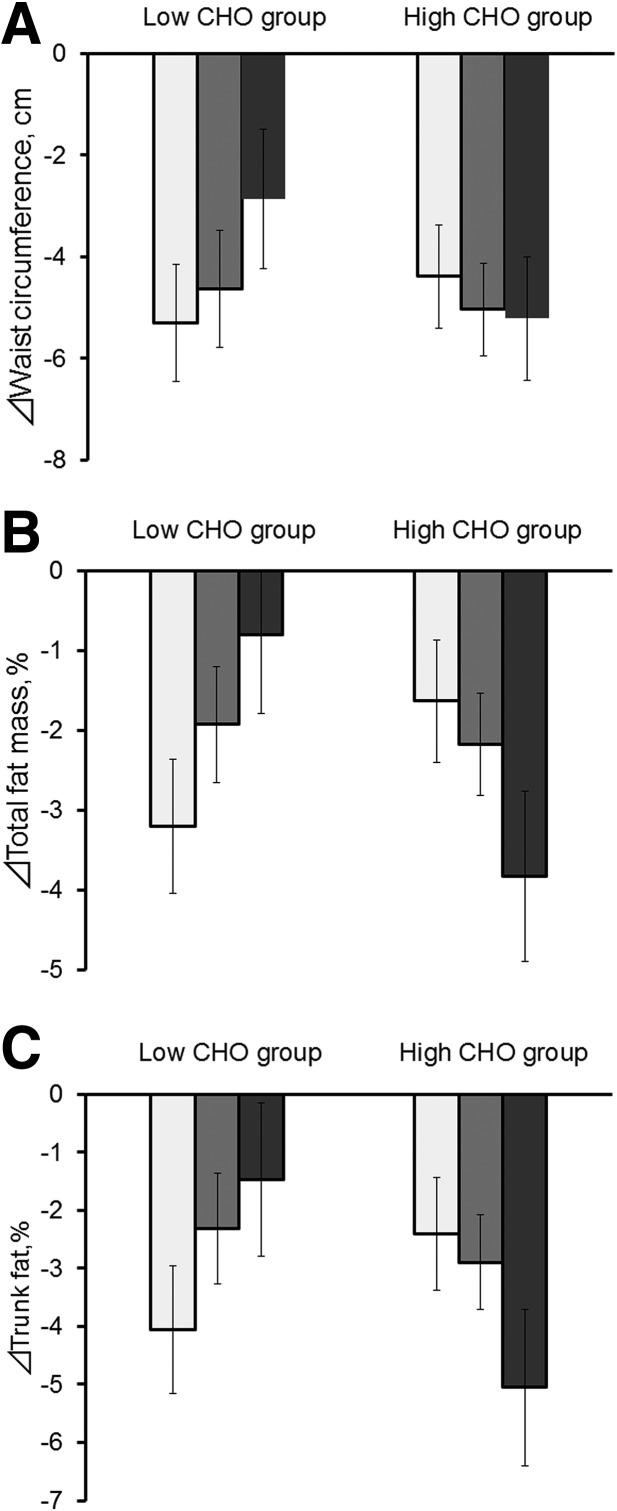
We found significant interactions between the genotype and low-/high-carbohydrate diet on 2-year changes in WC, total fat, total lean, total fat mass percentage, and trunk fat percentage (Pinteraction = 0.049, 0.004, 0.003, 0.001, and 0.003, respectively, in model 1) (Table 2). In response to the low-carbohydrate diet, an increasing number of the C allele of the FGF21 variant was significantly associated with an increase in WC, total fat, total fat mass percentage, and trunk fat percentage at 2 years. Conversely, carrying the C allele was negatively associated with changes in these outcomes in response to the high-carbohydrate diet (Table 2 and Fig. 1). Additional adjustment of BMI at the baseline in model 2 slightly attenuated the association, although P values for interactions between genotype and the low-/high-carbohydrate diet in 2-year changes of body composition remained significant. Further adjustment for total energy intake at baseline did not appreciably change the results (data not shown). When the analysis was performed only among white individuals, we observed fundamentally similar results with significant interactions for 2-year changes in total fat, total lean, total fat mass percentage, and trunk fat percentage (all Pinteraction < 0.05 in model 2).
Table 2.
Effect of the FGF21 genetic variant on changes in body weight, WC, and body composition in response to low- or high-carbohydrate diet at 2 years of diet intervention
| Low carbohydrate |
High carbohydrate |
Pinteraction | |||
|---|---|---|---|---|---|
| β (SE) | P | β (SE) | P | ||
| Model 1 | |||||
| ΔBody weight, kg | 1.46 (0.62) | 0.02 | −0.16 (0.64) | 0.8 | 0.07 |
| ΔWC, cm | 1.22 (0.69) | 0.08 | −0.66 (0.66) | 0.32 | 0.049 |
| ΔTotal fat, g | 1,861 (849) | 0.03 | −1,319 (781) | 0.09 | 0.004 |
| ΔTotal lean, g | 837 (420) | 0.049 | −799 (410) | 0.054 | 0.003 |
| ΔTotal fat mass, % | 1.25 (0.49) | 0.01 | −0.95 (0.50) | 0.06 | 0.001 |
| ΔTrunk fat, % | 1.52 (0.65) | 0.02 | −1.12 (0.64) | 0.08 | 0.003 |
| Model 2 | |||||
| ΔWC, cm | 1.16 (0.68) | 0.09 | −0.44 (0.65) | 0.5 | 0.09 |
| ΔTotal fat, g | 1,848 (854) | 0.03 | −1,221 (786) | 0.12 | 0.006 |
| ΔTotal lean, g | 853 (421) | 0.046 | −806 (414) | 0.054 | 0.003 |
| ΔTotal fat mass, % | 1.21 (0.49) | 0.02 | −0.94 (0.5) | 0.06 | 0.002 |
| ΔTrunk fat, % | 1.35 (0.66) | 0.04 | −1.08 (0.64) | 0.09 | 0.006 |
β represents changes in outcomes for the increasing number of C allele of the SNP rs838147.
Bold values indicate statistical significance (P < 0.05).
Model 1: age, sex, ethnicity, and variable of interest at the baseline examination.
Model 2: model 1 plus BMI at the baseline examination.
Figure 1.
Changes in WC (A), total fat mass percentage (B), and trunk fat percentage (C) according to the FGF21 genetic variant rs838147 and low- or high-carbohydrate diet at 2 years of diet intervention (white bars, FGF21 rs838147 TT genotype; gray bars, TC genotype; black bars, CC genotype). Data are means ± SE values after adjustment for age, sex, ethnicity, and BMI at the baseline examination and value for the respective outcome traits at the baseline examination. CHO, carbohydrate.
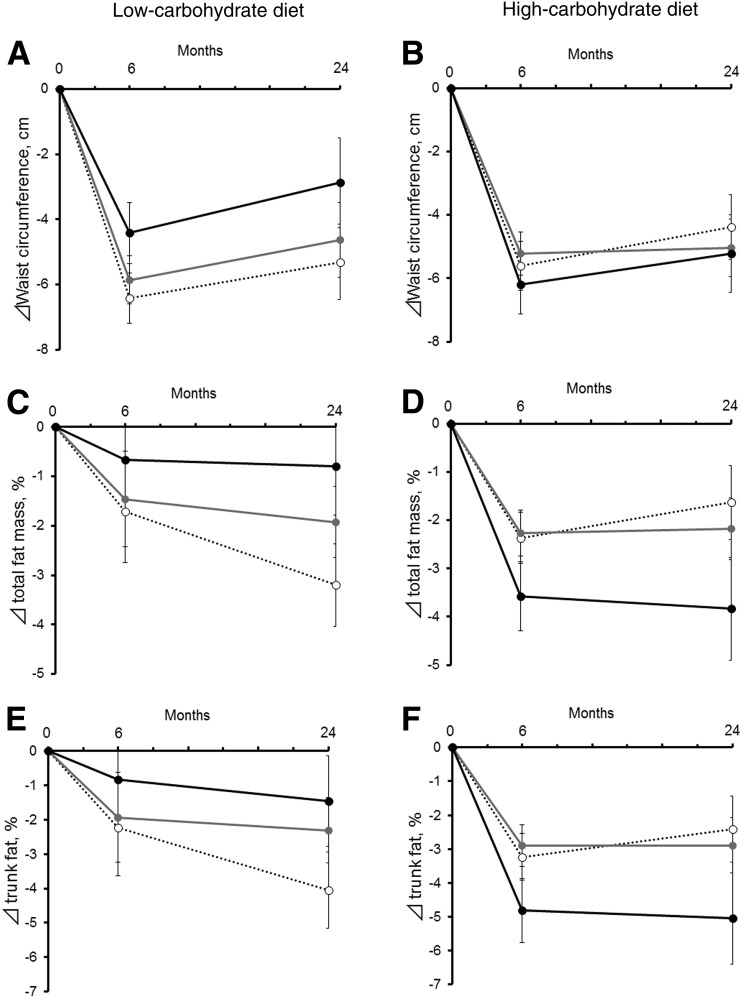
We also performed linear mixed models integrating measures at 6 months and 2 years to assess the trajectory of genetic associations with the outcomes across the course of dietary intervention (Fig. 2). On the one hand, the gene–diet interaction patterns on WC were consistent at 6 months and 2 years, but participants regained WC during the 6 months and 2 years (Fig. 2A and B). On the other hand, we observed significant genotype–time interactions, particularly for changes in total fat mass percentage and trunk fat percentage among the low-carbohydrate diet group (Fig. 2C and E) and the high-carbohydrate diet group (Fig. 2D and F) over the 2-year trajectories. Results of genotype–time interactions were significant even after adjustment for age, sex, ethnicity, BMI, and the values for the respective outcome traits at the baseline examination; the genetic associations with improving in body fat composition appeared to be more pronounced at 2 years than at 6 months (Fig. 2C–F). Particularly, in the low-carbohydrate diet group, individuals without C allele (Fig. 2C and E, dotted line) continued to lose total fat mass and trunk fat from 6 months to 2 years.
Figure 2.
Trajectories of WC (A and B), total body fat mass percentage (C and D), and trunk fat percentage (E and F) according to FGF21 variant rs838147 in response to low- or high-carbohydrate diet during the 2-year intervention assessment (black circle and solid line, CC genotype; gray circle and gray solid line, TC genotype; white circle and dotted line, TT genotype). Data are mean ± SE after adjustment for age, sex, ethnicity, BMI at the baseline examination, and value for the respective outcome traits at the baseline examination. P for interaction (genotype × time) after adjustment for age, sex, ethnicity, and BMI at the baseline examination: P > 0.05 (not significant) for A and B; P < 0.001 for C, D, E, and F. P for interaction (genotype × time) after adjustment for age, sex, ethnicity, BMI, and value for the respective outcome traits at the baseline examination: P > 0.05 (not significant) for A and B; P = 0.006 for C; P = 0.01 for D and E; and P = 0.02 for F.
Conclusions
In this trial, one of the largest randomized dietary intervention trials on weight loss, we found that the macronutrient intake–associated FGF21 genotype significantly interacted with dietary carbohydrate/fat intake on changes in central adiposity and body composition. In response to the low-carbohydrate/high-fat diet, an increasing number of the carbohydrate intake–decreasing C allele of rs838147 was associated with less reduction in WC, whole-body fat, or trunk fat. Conversely, opposite genetic effects on these outcomes were observed in response to the high-carbohydrate/low-fat diet; carrying the C allele was associated with a greater reduction of WC, whole-body fat, and trunk fat.
Our findings are in line with the biological roles of FGF21 in regulating body adiposity and dietary intake. The FGF21 gene encodes FGF21, a circulating hormone-like protein with pleiotropic metabolic actions, such as regulating glucose and fat metabolism, and effect on reducing body weight (26). On the one hand, elevated circulating levels of FGF21 have been associated with a variety of adiposity measures such as WC (12,14,16,17), BMI (12–16), fat mass (13,16), and visceral fat (18,19), with obesity considered an FGF21-resistant state (20). On the other hand, several studies in rodents and humans reported that FGF21 levels were influenced by dietary intake of fat, carbohydrate, or protein (5,6,27–31). In two recent GWAS (3,4), we identified the genetic variants in the region of the FGF21 gene that were associated with dietary macronutrient intakes, and rs838147 showed the strongest association with carbohydrate intake. In the current study, we observed significant interactions between the FGF21 genotype and carbohydrate intake on adiposity measurements. The results from the present study and previous GWAS consistently indicate that FGF21 may play a role in regulating metabolisms of dietary carbohydrate. Because low-carbohydrate intake is usually accompanied by high-fat intake, we could not determine whether carbohydrate or fat would best explain our findings. Nonetheless, two recent studies found that FGF21 acted on the brain to regulate carbohydrate or sweet taste and did not affect response to fat taste preference (7,8), also suggesting that the FGF21 genetic variants might primarily interact with dietary carbohydrate intake on adiposity measures.
In addition, we found that the genetic effects were opposite according to dietary carbohydrate intake on improving adiposity measurements. Such an opposite genetic effect could be partly explained by the “differential susceptibility hypothesis,” (32–34) a theory that suggests that genes may be conceptualized as “plastic,” because genetic risk can be modified by environmental factors, such as dietary factors. Some individuals might be more affected by environmental exposures because of genetic make-up in a for-better-and-for-worse manner, depending on the environment to which they are exposed (33). We observed that carriers of the carbohydrate-decreasing allele showed an opposite response to diet interventions varying in carbohydrate intake; we assume that high- or low-carbohydrate intake may differentially modulate the function or activity of the FGF21 gene with the variant allele. Further studies are warranted to test such postulations.
We also observed significant gene–diet interactions, particularly on body fat composition and marginally on WC. Previous studies suggested that FGF21 may act directly on adipose tissue and that the weight-reducing effect of FGF21 may be partially mediated by induction of thermogenesis in adipose tissue and browning of white adipose tissue (11,26,35). A study has demonstrated that FGF21 levels were slightly better correlated with WC than with BMI and that WC was significantly positively associated with serum FGF21 levels even after adjustment for BMI (12). Plasma concentrations of FGF21 were also reported to be positively associated with both BMI and fat mass, whereas only body fat mass was predictive of plasma FGF21 concentrations when the analysis adjusted for covariates (13). Taken together, these data suggest that the FGF21 genotype may particularly affect certain body fat composition, independent of overall body weight. Whether changes in energy expenditure contribute to explaining our results would need to be further investigated.
Intriguingly, our analysis using the linear mixed models indicated significant genotype–time interaction, supporting that the gene–diet interaction became stronger at 2 years than at 6 months. Participants in the POUNDS Lost trial regained body weight from 6 months to 2 years (24). Nonetheless, we found that carriers without the C allele in the low-carbohydrate/high-fat diet group continued to lose total body fat and trunk fat even though they regained body weight and WC from 6 months to 2 years. Although the mechanisms underlying such observations remained unclear, our data suggest that different pathways might be involved in linking FGF21 to overall body weight and specific body fat composition. Because FGF21 has diverse metabolic functions in multiple target organs, including liver and brain (26,36–38), further investigations are warranted to verify our postulations.
To the best of our knowledge, this is the first study to show significant interactions between the macronutrient intake–associated FGF21 genetic variation and dietary carbohydrate/fat intake on improvement of body composition and central obesity in a large and long-term randomized trial among overweight or obese individuals. Growing evidence shows that FGF21 may be a potential for the treatment of obesity-related metabolic diseases (10,37–39); our study provides novel data to support potential precision dietary interventions considering the FGF21 genotype.
Nonetheless, several limitations of this study should also be considered. We did not measure circulating FGF21 concentrations in this study, which limited our ability to explore potential underlying mechanisms. In addition, 80% of our study participants were white, and the genotype distribution differed across ethnicities. Our subgroup analysis in whites showed similar results; nevertheless, whether our findings are generalizable to other ethnicities needs to be further investigated.
In conclusion, our study showed that dietary carbohydrate/fat intake modified the effect of the FGF21 genotype on changes in central adiposity and body fat composition among overweight or obese individuals during a 2-year dietary intervention. Our findings suggest that a high-carbohydrate/low-fat diet may be beneficial for overweight or obese individuals who carry the carbohydrate intake–decreasing allele of the FGF21 variant to improve body fat composition and abdominal obesity.
Article Information
Acknowledgments. The authors thank all of the participants in the study for their dedication and contribution to the research.
Funding. The study is supported by National Institutes of Health grants from the National Heart, Lung, and Blood Institute (HL-071981, HL-034594, HL-126024), the National Institute of Diabetes and Digestive and Kidney Diseases (DK-091718, DK-100383, DK-078616), the Boston Obesity Nutrition Research Center (DK-46200), and United States–Israel Binational Science Foundation Grant 2011036. Y.H. is a recipient of a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science. L.Q. is a recipient of the American Heart Association Scientist Development Award (0730094N). The sponsors had no role in the design or conduct of the study.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. Y.H. contributed to the study concept and design, analysis and interpretation of data, drafting and revising the manuscript, statistical analysis, and study supervision. W.M. and T.H. contributed to analysis and interpretation of data and to drafting and revising the manuscript. T.W. and Y.Z. contributed to interpretation of data. S.R.S., G.A.B., and F.M.S. contributed to acquisition of data, interpretation of data, and drafting and revising the manuscript. L.Q. contributed to the study concept and design, acquisition of data, analysis and interpretation of data, drafting and revising the manuscript, statistical analysis, funding, and study supervision. All authors were involved in the writing and revising of the manuscript and approved the final version. L.Q. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Clinical trial reg. no. NCT00072995, clinicaltrials.gov.
This article is featured in a podcast available at http://www.diabetesjournals.org/content/diabetes-core-update-podcasts.
References
- 1.Hooper L, Abdelhamid A, Moore HJ, Douthwaite W, Skeaff CM, Summerbell CD. Effect of reducing total fat intake on body weight: systematic review and meta-analysis of randomised controlled trials and cohort studies. BMJ 2012;345:e7666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tobias DK, Chen M, Manson JE, Ludwig DS, Willett W, Hu FB. Effect of low-fat diet interventions versus other diet interventions on long-term weight change in adults: a systematic review and meta-analysis. Lancet Diabetes Endocrinol 2015;3:968–979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tanaka T, Ngwa JS, van Rooij FJ, et al. Genome-wide meta-analysis of observational studies shows common genetic variants associated with macronutrient intake. Am J Clin Nutr 2013;97:1395–1402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chu AY, Workalemahu T, Paynter NP, et al.; CHARGE Nutrition Working Group; DietGen Consortium . Novel locus including FGF21 is associated with dietary macronutrient intake. Hum Mol Genet 2013;22:1895–1902 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dushay JR, Toschi E, Mitten EK, Fisher FM, Herman MA, Maratos-Flier E. Fructose ingestion acutely stimulates circulating FGF21 levels in humans. Mol Metab 2014;4:51–57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sánchez J, Palou A, Picó C. Response to carbohydrate and fat refeeding in the expression of genes involved in nutrient partitioning and metabolism: striking effects on fibroblast growth factor-21 induction. Endocrinology 2009;150:5341–5350 [DOI] [PubMed] [Google Scholar]
- 7.von Holstein-Rathlou S, BonDurant LD, Peltekian L, et al. FGF21 mediates endocrine control of simple sugar intake and sweet taste preference by the liver. Cell Metab 2016;23:335–343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Talukdar S, Owen BM, Song P, et al. FGF21 regulates sweet and alcohol preference. Cell Metab 2016;23:344–349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Adams AC, Gimeno RE. The sweetest thing: regulation of macronutrient preference by FGF21. Cell Metab 2016;23:227–228 [DOI] [PubMed] [Google Scholar]
- 10.Degirolamo C, Sabbà C, Moschetta A. Therapeutic potential of the endocrine fibroblast growth factors FGF19, FGF21 and FGF23. Nat Rev Drug Discov 2016;15:51–69 [DOI] [PubMed] [Google Scholar]
- 11.Cantó C, Auwerx J. Cell biology. FGF21 takes a fat bite. Science 2012;336:675–676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang X, Yeung DC, Karpisek M, et al. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes 2008;57:1246–1253 [DOI] [PubMed] [Google Scholar]
- 13.Tan BK, Hallschmid M, Adya R, Kern W, Lehnert H, Randeva HS. Fibroblast growth factor 21 (FGF21) in human cerebrospinal fluid: relationship with plasma FGF21 and body adiposity. Diabetes 2011;60:2758–2762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cuevas-Ramos D, Almeda-Valdes P, Gómez-Pérez FJ, et al. Daily physical activity, fasting glucose, uric acid, and body mass index are independent factors associated with serum fibroblast growth factor 21 levels. Eur J Endocrinol 2010;163:469–477 [DOI] [PubMed] [Google Scholar]
- 15.Reinehr T, Woelfle J, Wunsch R, Roth CL. Fibroblast growth factor 21 (FGF-21) and its relation to obesity, metabolic syndrome, and nonalcoholic fatty liver in children: a longitudinal analysis. J Clin Endocrinol Metab 2012;97:2143–2150 [DOI] [PubMed] [Google Scholar]
- 16.Olszanecka-Glinianowicz M, Madej P, Wdowczyk M, Owczarek A, Chudek J. Circulating FGF21 levels are related to nutritional status and metabolic but not hormonal disturbances in polycystic ovary syndrome. Eur J Endocrinol 2015;172:173–179 [DOI] [PubMed] [Google Scholar]
- 17.Mai K, Schwarz F, Bobbert T, et al. Relation between fibroblast growth factor-21, adiposity, metabolism, and weight reduction. Metabolism 2011;60:306–311 [DOI] [PubMed] [Google Scholar]
- 18.Giannini C, Feldstein AE, Santoro N, et al. Circulating levels of FGF-21 in obese youth: associations with liver fat content and markers of liver damage. J Clin Endocrinol Metab 2013;98:2993–3000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Taniguchi H, Tanisawa K, Sun X, et al. Cardiorespiratory fitness and visceral fat are key determinants of serum fibroblast growth factor 21 concentration in Japanese men. J Clin Endocrinol Metab 2014;99:E1877–E1884 [DOI] [PubMed] [Google Scholar]
- 20.Fisher FM, Chui PC, Antonellis PJ, et al. Obesity is a fibroblast growth factor 21 (FGF21)-resistant state. Diabetes 2010;59:2781–2789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fazeli PK, Lun M, Kim SM, et al. FGF21 and the late adaptive response to starvation in humans. J Clin Invest 2015;125:4601–4611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hooper L, Abdelhamid A, Bunn D, Brown T, Summerbell CD, Skeaff CM. Effects of total fat intake on body weight. Cochrane Database Syst Rev 2015;8:CD011834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Johnston BC, Kanters S, Bandayrel K, et al. Comparison of weight loss among named diet programs in overweight and obese adults: a meta-analysis. JAMA 2014;312:923–933 [DOI] [PubMed] [Google Scholar]
- 24.Sacks FM, Bray GA, Carey VJ, et al. Comparison of weight-loss diets with different compositions of fat, protein, and carbohydrates. N Engl J Med 2009;360:859–873 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.de Souza RJ, Bray GA, Carey VJ, et al. Effects of 4 weight-loss diets differing in fat, protein, and carbohydrate on fat mass, lean mass, visceral adipose tissue, and hepatic fat: results from the POUNDS LOST trial. Am J Clin Nutr 2012;95:614–625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gimeno RE, Moller DE. FGF21-based pharmacotherapy--potential utility for metabolic disorders. Trends Endocrinol Metab 2014;25:303–311 [DOI] [PubMed] [Google Scholar]
- 27.Wall CE, Whyte J, Suh JM, et al. High-fat diet and FGF21 cooperatively promote aerobic thermogenesis in mtDNA mutator mice. Proc Natl Acad Sci U S A 2015;112:8714–8719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Müller TD, Tschöp MH. Play down protein to play up metabolism? J Clin Invest 2014;124:3691–3693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Laeger T, Henagan TM, Albarado DC, et al. FGF21 is an endocrine signal of protein restriction. J Clin Invest 2014;124:3913–3922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nygaard EB, Møller CL, Kievit P, Grove KL, Andersen B. Increased fibroblast growth factor 21 expression in high-fat diet-sensitive non-human primates (Macaca mulatta). Int J Obes 2014;38:183–191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vienberg SG, Brøns C, Nilsson E, Astrup A, Vaag A, Andersen B. Impact of short-term high-fat feeding and insulin-stimulated FGF21 levels in subjects with low birth weight and controls. Eur J Endocrinol 2012;167:49–57 [DOI] [PubMed] [Google Scholar]
- 32.Belsky J, Jonassaint C, Pluess M, Stanton M, Brummett B, Williams R. Vulnerability genes or plasticity genes? Mol Psychiatry 2009;14:746–754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Belsky J, Hartman S. Gene-environment interaction in evolutionary perspective: differential susceptibility to environmental influences. World Psychiatry 2014;13:87–89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Belsky J, Pluess M. Beyond diathesis stress: differential susceptibility to environmental influences. Psychol Bull 2009;135:885–908 [DOI] [PubMed] [Google Scholar]
- 35.Adams AC, Yang C, Coskun T, et al. The breadth of FGF21's metabolic actions are governed by FGFR1 in adipose tissue. Mol Metab 2012;2:31–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Domouzoglou EM, Naka KK, Vlahos AP, et al. Fibroblast growth factors in cardiovascular disease: the emerging role of FGF21. Am J Physiol Heart Circ Physiol 2015;309:H1029–H1038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fisher FM, Maratos-Flier E. Understanding the physiology of FGF21. Annu Rev Physiol 2016;78:223–241 [DOI] [PubMed] [Google Scholar]
- 38.Zhang J, Li Y. Fibroblast growth factor 21 analogs for treating metabolic disorders. Front Endocrinol (Lausanne) 2015;6:168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhang F, Yu L, Lin X, et al. Minireview: roles of fibroblast growth factors 19 and 21 in metabolic regulation and chronic diseases. Mol Endocrinol 2015;29:1400–1413 [DOI] [PMC free article] [PubMed] [Google Scholar]