Abstract
Rosacea is a chronic inflammatory disease with transient and non-transient redness as key characteristics. Brimonidine is a selective α2-adrenergic receptor (AR) agonist approved for persistent facial erythema of rosacea based on significant efficacy and good safety data. The majority of patients treated with brimonidine report a benefit; however, there have been sporadic reports of worsening erythema after the initial response. A group of dermatologists, receptor physiology, and neuroimmunology scientists met to explore potential mechanisms contributing to side effects as well as differences in efficacy. We propose the following could contribute to erythema after application: (1) local inflammation and perivascular inflammatory cells with abnormally functioning ARs may lead to vasodilatation; (2) abnormal saturation and cells expressing different AR subtypes with varying ligand affinity; (3) barrier dysfunction and increased skin concentrations of brimonidine with increased actions at endothelial and presynaptic receptors, resulting in increased vasodilation; and (4) genetic predisposition and receptor polymorphism(s) leading to different smooth muscle responses. Approximately 80% of patients treated with brimonidine experience a significant improvement without erythema worsening as an adverse event. Attention to optimizing skin barrier function, setting patient expectations, and strategies to minimize potential problems may possibly reduce further the number of patients who experience side effects.
Funding: Galderma International S.A.S., Paris, France.
Keywords: Adrenergic receptors, Brimonidine, Dermatology, Erythema, Rosacea, Vasoconstriction, Worsening of erythema
Introduction
Transient and non-transient erythema are key characteristics of rosacea. Up to 85% of individuals with rosacea suffer from facial redness [1]; managing this redness poses a challenge for patients and clinicians alike [2]. Rosacea-related erythema can have short or long durations, and may take many forms, including persistent underlying redness, transient erythema (flushing) that is triggered by stimuli, or redness associated with individual inflammatory lesions or telangiectasias [2–4].
Relatively little is understood about the mechanisms that cause of redness, but both vascular and inflammatory events are involved [5–9]. Neurogenic inflammation may have a role; in this case, dysregulated neurovascular communication may lead to sustained vasodilation induced by various trigger factors (e.g., spicy food, exercise, heat, etc.) and thereby prolong flushing (±stinging/burning). Neurologic factors also can influence vascular responses, and both sensory and autonomic nerves interact with immune cells in the process of flushing. Non-transient erythema of rosacea could represent a form of neurogenic inflammation, with immune mediators released by endothelial cells, keratinocytes, and immune cells resulting in chronic inflammation. Or, pathogens may activate innate immune receptors, such as toll-like receptor-2 (TLR-2) and upregulate antimicrobial peptides, such as cathelicidin LL37. LL37 can induce inflammation and vascular responses, and stimulate the release of cytokines and other mediators that secondarily activate neurovascular changes [10, 11].
Topical brimonidine gel 0.33% is approved to treat rosacea-associated redness [12, 13]. This agent selectively binds α2-adrenergic receptors (ARs) on vascular smooth muscle resulting in local transient vasoconstriction following topical application. Approximately 80% of patients treated with brimonidine report redness relief [14]. As the effects of brimonidine wear off (typically 10–12 h after application), erythema is expected to slowly return to baseline. In post-marketing experience, there were some reports of a transient worsening erythema rather than a return to baseline levels manifesting as [15–17]:
Paradoxical erythema—redness appearing within 6 h after the application of brimonidine (can be worse than baseline).
Exaggerated recurrence of erythema—redness greater than baseline that occurs as therapy wears off (10–12 h post-application).
Allergic contact dermatitis—redness (usually accompanied by other signs, such as eczema and pruritus) occurring several months after the initiation of therapy.
Side effects were not reported in publications of the pivotal clinical trials as “paradoxical erythema;” however, Holmes et al. [18] conducted an analysis of adverse events and stated 3.6% of subjects reported erythema and 1.8% flushing during the phase 3 study; in the long-term study, 9.1% of subjects had flushing and 6.5% had erythema. Tanghetti et al. [15] estimated that 10–20% of patients are at risk for worsening of redness. Jackson et al. [19] state “in our clinics, approximately 4–15% of patients have complained of worsening erythema or flushing,” adding that while this led to discontinuation of drug for some, many patients were able to successfully continue treatment, because “worsening of erythema was only temporary and not a daily issue.” Furthermore, they noted that erythema typically occurred during the initiation of therapy and subsided with regular use [19]. In response to a case report published in 2016, Tanghetti agreed, noting “Once the paradoxical erythema subsided, some patients were able to resume therapy without recurrence of this problem” [55].
We discuss physiologic mechanisms that may contribute to worsening erythema (Fig. 1). In our opinion, the most likely candidates are:
Local inflammation with perivascular inflammatory cells bearing increased or modified brimonidine-responsive ARs, leading to increased drug effect to release mediators and produce sustained vasodilatation (Fig. 1).
High concentration of brimonidine in the skin (e.g., due to skin barrier dysfunction). Brimonidine may penetrate in higher concentrations to endothelium and nerve terminals to cause vasodilatation (Figs. 1, 2).
Abnormal saturation effects and inhibited release of norepinephrine (NE) from sympathetic nerve terminals due to the action of brimonidine on α2-ARs, with a net effect of reduced NE availability.
Genetic polymorphism(s) for genes increase the affinity of brimonidine to the receptor, resulting in increased smooth muscle responses and sustained vasodilation.
Fig. 1.
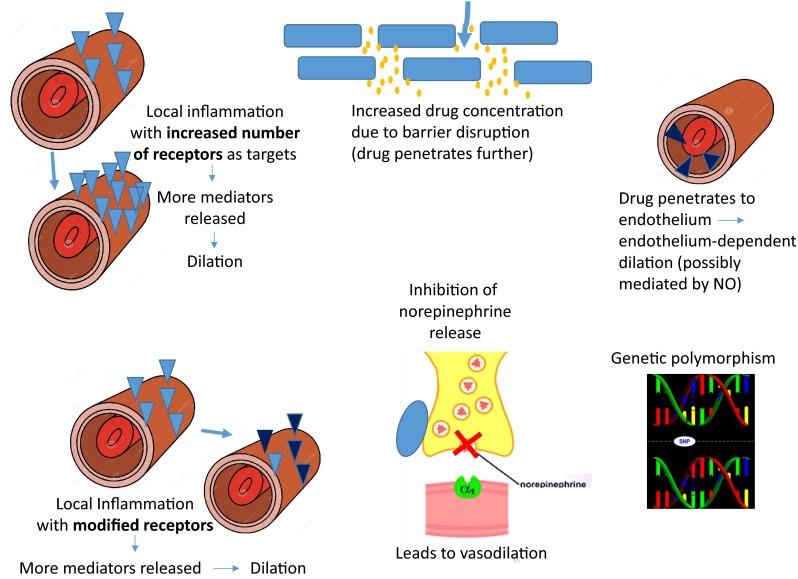
Rosacea. Schematic representation of possible mechanisms that contribute to the exacerbation of erythema during brimonidine therapy. NO nitric oxide
Fig. 2.
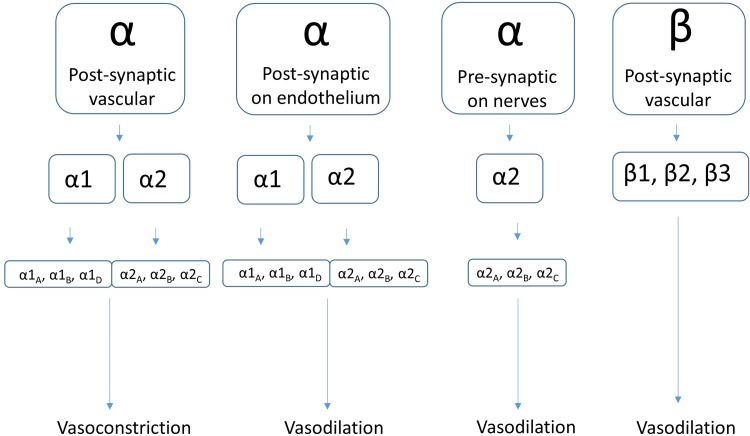
Rosacea. Possible sites of adrenergic receptors in the control of vascular tone: (1) post-synaptic α-ARs on vascular smooth muscle, (2) post-synaptic α-ARs on vascular endothelium, (3) pre-synaptic α2-ARs on nerve terminals, and (4) post-synaptic β-ARs on vascular smooth muscles
Compliance with Ethics Guidelines
This article does not contain any new studies with human or animal subjects performed by any of the authors.
Brief Review of Adrenergic Receptor Functions
Overview
Review of receptor physiology is helpful to understand what may be affecting the action of brimonidine. G protein-coupled receptors (GPCRs), including ARs, interact with catecholamines, particularly NE and epinephrine. ARs are found throughout the body, including in the vascular smooth muscle, vascular endothelium, on presynaptic nerve terminals, and on a subset of immune cells. There are a number of subtypes based on physiologic function, pharmacology, structure, and signal transduction (Fig. 2) [20]. The function of α-ARs in the skin is primarily to regulate blood flow through vasoconstriction following catecholamine binding to receptors on vascular smooth muscle, where the mobilization of calcium stores results in muscle cell contraction. Detailed molecular mechanisms of ARs are described elsewhere [21].
Factors that Affect Receptor Function
α 2 Receptors
α-ARs are divided into α1 and α2 subtypes. The relative contribution of α1 and α2 subtypes in subcutaneous vessels can vary with location, receptor density, local NE concentration, and temperature [20, 22, 23]. The density of arterial α2-ARs increases from proximal to distal location [20], and α2-ARs are thought to dominate in small vessels (<200 microns) [24].
Expression of α-ARs on non-smooth muscle cells can also mediate receptor function. Within the vasculature, α2-ARs on endothelial cells relax blood vessels by release of nitric oxide (NO) [25], an effect opposite that of ARs on vascular smooth muscle. Presynaptic nerve terminals likewise express α2-ARs and induce vasorelaxation by inhibiting release of NE and, subsequently, adrenergic neurotransmission via a negative feedback function [26].
α 1 Receptors
Sensory nerve fibers have also been shown to express α1-ARs, which generate axon-reflex vasodilation peripheral to the area of vasoconstriction [27]. The more recently described α1D-AR may be the most important receptor in terms of sympathetic nervous control of arteries, as it predominates at the sympathetic neuroeffector junction [28].
Receptor Expression on Immune Cells
Finally, several immune cell subsets present in the skin express α1- and α2-ARs. α1-ARs are expressed on cells with known pro-inflammatory activities, whereas α2-ARs appear to mediate anti-inflammatory effects [29, 30].
Adrenergic Receptors: Speculations About role in Rosacea and with Brimonidine Therapy
Rosacea is a disease of chronic, cyclic inflammation, and vasodilation. α-ARs in skin of selected rosacea patients may exhibit an altered physiological response to brimonidine that may be dependent or independent of the disease or the agonist. Possible factors that may contribute to episodes of worsening erythema are discussed below.
Impact of Concentration and/or Chronic Agonism
In rosacea patients, epidermal barrier function is disturbed and stratum corneum hydration is reduced [31, 32]. This may lead to higher drug concentrations in skin and possibly also altered drug clearance. Differences in application techniques can affect the distribution of brimonidine and local drug concentrations.
Local brimonidine concentrations may influence worsening erythema by: (1) the saturation of α2-receptors on vascular smooth muscle, resulting in the binding of additional cell types or AR subtypes and/or (2) the chronic agonism of α-ARs, resulting in the downregulation and desensitization of receptors.
Brimonidine is a selective α2A-AR agonist, but it exhibits a reduced affinity for α2C- and α1A-ARs as well [24]. Binding to the more recently characterized α1D subtype was not tested, but may also occur when brimonidine is present at high concentrations. The effect of brimonidine on other receptors or cell types might be concentration dependent, and a spillover effect after saturation of the α2A-AR on vascular smooth muscle could occur.
Increased local concentrations (‘spillover effect’) may allow brimonidine to act on at least one endothelial receptor (α2A-AR and other subtypes), thereby promoting NO-mediated vasodilation. Brimonidine may also act on presynaptic ARs of nerve terminals leading to reduced vasoconstriction (α2A- and α2C-mediated). Both effects could promote local vasodilation (α2-mediated) and worsen erythema.
α2A-ARs expressed on presynaptic nerves modulate an intense, acute release of NE that is evoked by high-frequency neuronal stimulation occurring with stress responses. In contrast, α2C-ARs on presynaptic nerve terminals regulate the basal, long-term release of NE due to low-frequency stimulation. Thus, these receptors tightly regulate neurotransmitter release from adrenergic nerves under basal and stressful conditions through their inhibitory presynaptic feedback loop.
Taken together, differences in sympathetic nerve firing rates, and thus local NE concentrations, could impact brimonidine efficacy by competing for α2-AR binding.
Neuronal activation could also lead to release of other neuromediators that induce vasodilation. For example, axon-reflex vasodilation mediated by α1-ARs is partially mediated by prostaglandins (which have been shown to be increased in rosacea skin) and the overall activity has been suggested to contribute to local vascular disturbances in acute or chronic inflammation. Furthermore, α1-ARs have been found in subpopulations of neurons in rat skin, which are immunoreactive to the TRPV1 (transient receptor potential cation channel subfamily V member 1) ion channel and the neuromediator calcitonin gene-related peptide (CGRP; glucagon-related peptide), both of which are involved in vasodilation and neurogenic inflammation in human rosacea skin [6, 33, 34].
Enhanced concentration of brimonidine may also result in the activation of α1-ARs present on immune cells, such as macrophages and mast cells, both of which are present at increased densities in rosacea-affected skin. Known effects include an increased release of vasodilatory mediators, such as prostaglandin E2, histamine, tryptase, and neuropeptides, which could in turn result in worsened erythema [35].
The ‘spillover’ effect may explain paradoxical-worsening erythema following the application of brimonidine, where an initial reduction in erythema is observable prior to the saturation of vascular α2-ARs. This may be followed by enhanced binding to other receptor subtypes on different cell types that override local vasoconstriction by vasodilatory and/or inflammatory mechanisms within a few hours of application.
Alternatively, chronic α2-AR agonism may downregulate or desensitize vasoconstrictive α2-ARs, leading to reflexive vasodilation via vasodilatatory α-ARs. This hypothesis is supported by the fact that similar feedback loops have been demonstrated with other α adrenergic agonists, e.g., rhinitis medicamentosa induced by nasally administered oxymetazoline and xylometazoline [36]. However, the adaptation of receptors with chronic ophthalmic use has not been reported, and minimal to no tachyphylaxis has been reported with chronic topical use [12].
To minimize the potential for complications related to concentration and skin barrier function, we agree with the recommendations by Tanghetti et al. [15] to set realistic patient expectations by providing clear education, to teach patients to apply the medication in a very thin layer initially, and to make the first trial of medication on a day when the patient is able to observe the effect in privacy.
Impact of Inflammation
Rosacea is characterized by abnormal inflammatory responses to regular environmental stimuli (e.g., ultraviolet light and microbes) mediated by abnormally increased neuromediators [37, 38], proteases, cytokines, vitamin D, and TLR-2 signaling [8, 9, 33, 39]. TLR-2 signaling can recruit a variety of inflammatory mediators, many of which are regulated by the transcription factor NF-kB, such as interleukin-6 (IL-6) or tumor necrosis factor-α (TNF-α) [6, 9, 40].
An NF-kB binding site in the promoter region of G protein receptor kinase (GRKs) could be involved in shifting GPCR signaling [41]. These kinases are well known to regulate GPCR cell function, including receptor internalization and desensitization, thereby leading to reduced receptor numbers (by downregulation) as well as alternate signaling via recruitment of β-arrestin [42]. Moreover, shifts in AR signaling have been linked to the induction of inflammation by increasing NF-kB levels [43–51]. This could be a possible mechanism for the adverse rebound effect observed in some patients using brimonidine.
Mast cells may contribute to the inflammatory loop in rosacea as well. Mast cell-deficient mice do not display rosacea-like inflammation [52, 53], and α adrenergic blockade increases acetylcholine release as well as subsequent pituitary adenylate cyclase-activating polypeptide (PACAP) release and protease-activated receptor 2 (PAR2) activation, contributing to inflammation and vasodilation. The stimulation of neuropeptide release by innate immune receptors, such as TLR2 and PAR2, may be critically involved in inflammatory vasodilation, as observed in all three skin subtypes of rosacea [6, 7].
COX-2 and prostaglandin production, both of which are elevated in rosacea, may increase α-receptor-mediated NO, thereby contributing to vasodilation and edema via a prostanoid-dependent pathway. Furthermore, the effects of brimonidine treatment may be different in the early versus late inflammation, as immune and vascular responses can shift with prolonged stimuli, thereby changing the repertoire and density of ARs and GPCRs.
Skin deposits of nitrogen (NO2 and NO3) are converted to NO and promote vasodilation independent of the endothelium in the presence of ultraviolet A irradiation, a common trigger in rosacea.
An inflammatory milieu and heightened neuronal activity are present in rosacea [7, 9], and it is possible that worsening erythema could represent an exaggerated compensatory neuroinflammatory mechanism of the vasculature aside from what is suggested here, such as release of neuropeptides and systemic adrenergic or other vasoactive peptides.
Best results may be obtained when rosacea-associated inflammation is managed using the traditional rosacea treatments in conjunction with brimonidine therapy. Using brimonidine to reduce the overall erythema can unmask redness from inflammatory lesions, which is unpalatable for patients. It may also be useful to educate patients about overall facial erythema, telangiectasias, and perilesional erythema [15].
Impact of Genetic Polymorphisms
Genetic polymorphism in ARs is common and may cause lack of or reduced functional receptor subtypes on vascular smooth muscle in some individuals. Polymorphisms in the receptors may produce differential binding, altered binding sensitivity, or receptor distribution. The impact of polymorphism on ARs is evidenced by the drug response to albuterol (salbutamol) in asthma, where it has been demonstrated that responses to receptor-blocking drugs can change [54].
Polymorphic differences in avidity or altered kinetics among α2A-ARs expressed on different tissues may also explain the worsening erythema phenomenon, particularly with regard to exaggerated recurrence as drug effect wanes. If the avidity of brimonidine for the α2-AR on presynaptic nerve terminals is greater than that on vascular smooth muscle, the vasoconstrictive effect on vessels may wear off as expected, but negative feedback inhibition on nerve terminals may remain for a longer period, with transiently sustained erythema.
This potential mechanism also underscores the prudence of advising patients to make the first trial of medication on the weekend or a day when the patient can be comfortable even in the unlikely event that worsening erythema occurs [15].
Summary and Conclusions
Brimonidine has proven strong vasoconstrictive effects on α2-ARs of vessels and—to a lesser extent—on α1-ARs [24]. In rosacea, ARs on other tissues—such as endothelium, presynaptic adrenergic nerve terminals, and immune cells—may also promote vasodilation. Skin reactivity to adrenergic agonists may be influenced by a complex system of activation and inhibition, systemic factors, such as epinephrine/NE levels, local tissue factors (including NO), receptor up- and downregulations, desensitization, inflammation, and exterior (e.g., temperature, ultraviolet light, microbes, and proteases) or endogenous factors (increase of local body temperature via exercising, pH changes, microbiota). In turn, worsening erythema events may occur with brimonidine use for reasons that are entirely patient dependent and can be allergic (very rarely) or non-allergic (more often) on nature.
The worsening phenomenon that occurs in some patients as the effects of brimonidine agonism wane may be explained by receptor desensitization and internalization, negative feedback loops, or activation of other compensatory mediators that lead to vasodilation. Skin barrier dysfunction can increase local concentrations of brimonidine and can be effectively treated with moisturizers prior to brimonidine therapy. In addition, the paradoxical-worsening phenomenon occurring within hours of brimonidine application may be explained by high concentrations of brimonidine within skin that leads to the saturation of receptors and ‘spillover’ effect to other α-AR subtypes with unwanted effects (vasodilation).
Brimonidine effects are predominantly beneficial. We hypothesize that certain individual characteristics may potentially override the vasoconstrictive effect transiently (e.g., barrier dysfunction) or permanently (e.g., receptor polymorphisms). Future studies are important to define those subgroups that may are most likely to benefit from brimonidine, since it is the first efficacious topical treatment against a frequent—and often challenging to treat—symptom in rosacea. It is crucial for clinicians to educate patients who should be taught to start with a very thin layer of medication and to use other strategies that can optimize brimonidine therapy [15].
Acknowledgments
Funding for the article-processing charges and open access fee for this publication were provided by Galderma International S.A.S., Paris, France. Editorial services were provided by Valerie Sanders, Sanders Medical Writing, and funded by Galderma International S.A.S. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given the final approval for the version to be published.
Disclosures
James R. Docherty, DSc, Dianne Lorton, PhD, Michael Detmar, MD, and Anna Di Nardo have served as advisory board members for Galderma Laboratories, LLP; Martin Steinhoff, MD, has served as an advisory board member, consultant, principal investigator, and speaker for Galderma International; Anna Holmes, PhD, and Gregor Schäfer, MD, are employees of Galderma.
Compliance with Ethics Guidelines
This article does not contain any new studies with human or animal subjects performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Footnotes
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/8295F060021A86F4.
References
- 1.Gupta AK, et al. J Eur Acad Dermatol Venereol. 2005;19:273–5. [DOI] [PubMed]
- 2.Del Rosso JQ, Gallo RL, Tanghetti E, Webster G, Thiboutot D. An evaluation of potential correlations between pathophysiologic mechanisms, clinical manifestations, and management of rosacea. Cutis Cutan Med Pract. 2013;91(3 Suppl):1–8. [PubMed] [Google Scholar]
- 3.Del Rosso JQ. Advances in understanding and managing rosacea: part 1: connecting the dots between pathophysiological mechanisms and common clinical features of rosacea with emphasis on vascular changes and facial erythema. J Clin Aesth Dermatol. 2012;5(3):16–25. [PMC free article] [PubMed] [Google Scholar]
- 4.Del Rosso JQ. Advances in understanding and managing rosacea: part 2: the central role, evaluation, and medical management of diffuse and persistent facial erythema of rosacea. J Clin Aesth Dermatol. 2012;5(3):26–36. [PMC free article] [PubMed] [Google Scholar]
- 5.Del Rosso JQ, Gallo RL, Kircik L, Thiboutot D, Baldwin HE, Cohen D. Why is rosacea considered to be an inflammatory disorder? The primary role, clinical relevance, and therapeutic correlations of abnormal innate immune response in rosacea-prone skin. J Drugs Dermatol JDD. 2012;11(6):694–700. [PubMed] [Google Scholar]
- 6.Steinhoff M, Schauber J, Leyden JJ. New insights into rosacea pathophysiology: a review of recent findings. J Am Acad Dermatol. 2013;69(6 Suppl 1):S15–S26. doi: 10.1016/j.jaad.2013.04.045. [DOI] [PubMed] [Google Scholar]
- 7.Steinhoff M, Buddenkotte J, Aubert J, Sulk M, Novak P, Schwab VD, et al. Clinical, cellular, and molecular aspects in the pathophysiology of rosacea. J Investig Dermatol Symp Proc Soc Investig Dermatol Inc Eur Soc Dermatol Res. 2011;15(1):2–11. doi: 10.1038/jidsymp.2011.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schwab VD, Sulk M, Seeliger S, Nowak P, Aubert J, Mess C, et al. Neurovascular and neuroimmune aspects in the pathophysiology of rosacea. J Investig Dermatol Symp Proc Soc Investig Dermatol Inc Eur Soc Dermatol Res. 2011;15(1):53–62. doi: 10.1038/jidsymp.2011.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Buhl T, Sulk M, Nowak P, Buddenkotte J, McDonald I, Aubert J, et al. Molecular and morphological characterization of inflammatory infiltrate in rosacea reveals activation of Th1/Th17 pathways. J Invest Dermatol. 2015;135(9):2198–2208. doi: 10.1038/jid.2015.141. [DOI] [PubMed] [Google Scholar]
- 10.Yamasaki K, Di Nardo A, Bardan A, Murakami M, Ohtake T, Coda A, et al. Increased serine protease activity and cathelicidin promotes skin inflammation in rosacea. Nat Med. 2007;13(8):975–980. doi: 10.1038/nm1616. [DOI] [PubMed] [Google Scholar]
- 11.Yamasaki K, Gallo RL. Rosacea as a disease of cathelicidins and skin innate immunity. J Investig Dermatol Symp Proc Soc Investig Dermatol Inc Eur Soc Dermatol Res. 2011;15(1):12–15. doi: 10.1038/jidsymp.2011.4. [DOI] [PubMed] [Google Scholar]
- 12.Moore A, Kempers S, Murakawa G, Weiss J, Tauscher A, Swinyer L, et al. Long-term safety and efficacy of once-daily topical brimonidine tartrate gel 0.5% for the treatment of moderate to severe facial erythema of rosacea: results of a 1-year open-label study. J Drugs Dermatol JDD. 2014;13(1):56–61. [PubMed] [Google Scholar]
- 13.Fowler J, Jr, Jackson M, Moore A, Jarratt M, Jones T, Meadows K, et al. Efficacy and safety of once-daily topical brimonidine tartrate gel 0.5% for the treatment of moderate to severe facial erythema of rosacea: results of two randomized, double-blind, and vehicle-controlled pivotal studies. J Drugs Dermatol JDD. 2013;12(6):650–656. [PubMed] [Google Scholar]
- 14.Layton AM, Schaller M, Homey B, Hofmann MA, Bewley AP, Lehmann P, et al. Brimonidine gel 0.33% rapidly improves patient-reported outcomes by controlling facial erythema of rosacea: a randomized, double-blind, vehicle-controlled study. J Eur Acad Dermat Venereol JEADV. 2015;29(12):2405–2410. doi: 10.1111/jdv.13305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tanghetti EA, Jackson JM, Belasco KT, Friedrichs A, Hougier F, Johnson SM, et al. Optimizing the use of topical brimonidine in rosacea management: panel recommendations. J Drugs Dermatol JDD. 2015;14(1):33–40. [PubMed] [Google Scholar]
- 16.Ilkovitch D, Pomerantz RG. Brimonidine effective but may lead to significant rebound erythema. J Am Acad Dermatol. 2014;70(5):e109–e110. doi: 10.1016/j.jaad.2014.01.853. [DOI] [PubMed] [Google Scholar]
- 17.Routt ET, Levitt JO. Rebound erythema and burning sensation from a new topical brimonidine tartrate gel 0.33% J Am Acad Dermatol. 2014;70(2):e37–e38. doi: 10.1016/j.jaad.2013.10.054. [DOI] [PubMed] [Google Scholar]
- 18.Holmes AD, Waite KA, Chen MC, Palaniswamy K, Wiser TH, Draelos ZD, et al. Dermatological adverse events associated with topical brimonidine gel 0.33% in subjects with erythema of rosacea: a retrospective review of clinical studies. J Clin Aesth Dermatol. 2015;8(8):29–35. [PMC free article] [PubMed] [Google Scholar]
- 19.Jackson JM, Knuckles M, Minni JP, Johnson SM, Belasco KT. The role of brimonidine tartrate gel in the treatment of rosacea. Clin Cosmet Investig Dermatol. 2015;8:529–538. doi: 10.2147/CCID.S58920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guimaraes S, Moura D. Vascular adrenoceptors: an update. Pharmacol Rev. 2001;53(2):319–356. [PubMed] [Google Scholar]
- 21.Minneman KP, Han C, Abel PW. Comparison of alpha 1-adrenergic receptor subtypes distinguished by chlorethylclonidine and WB 4101. Mol Pharmacol. 1988;33(5):509–514. [PubMed] [Google Scholar]
- 22.Bailey SR, Eid AH, Mitra S, Flavahan S, Flavahan NA. Rho kinase mediates cold-induced constriction of cutaneous arteries: role of alpha2C-adrenoceptor translocation. Circ Res. 2004;94(10):1367–1374. doi: 10.1161/01.RES.0000128407.45014.58. [DOI] [PubMed] [Google Scholar]
- 23.Borbujo J, Garcia-Villalon AL, Valle J, Gomez B, Diequez G. Postjunctional alpha-1 and alpha-2 adrenoceptors in human skin arteries. An in vitro study. J Pharmacol Exp Ther. 1989;249(1):284–287. [PubMed] [Google Scholar]
- 24.Piwnica D, Rosignoli C, de Menonville ST, Alvarez T, Schuppli Nollet M, Roye O, et al. Vasoconstriction and anti-inflammatory properties of the selective alpha-adrenergic receptor agonist brimonidine. J Dermatol Sci. 2014;75(1):49–54. doi: 10.1016/j.jdermsci.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 25.Matsuda H, Kuon E, Holtz J, Busse R. Endothelium-mediated dilations contribute to the polarity of the arterial wall in vasomotion induced by alpha 2-adrenergic agonists. J Cardiovasc Pharmacol. 1985;7(4):680–688. doi: 10.1097/00005344-198507000-00011. [DOI] [PubMed] [Google Scholar]
- 26.Starke K. Presynaptic alpha-autoreceptors. Rev Physiol Biochem Pharmacol. 1987;107:73–146. [PubMed] [Google Scholar]
- 27.Drummond PD. Inflammation contributes to axon reflex vasodilatation evoked by iontophoresis of an alpha-1 adrenoceptor agonist. Auton Neurosci. 2011;159(1–2):90–97. doi: 10.1016/j.autneu.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 28.Docherty JR. Vasopressor nerve responses in the pithed rat, previously identified as alpha2-adrenoceptor mediated, may be alpha1D-adrenoceptor mediated. Eur J Pharmacol. 2011;658(2–3):182–186. doi: 10.1016/j.ejphar.2011.02.029. [DOI] [PubMed] [Google Scholar]
- 29.Grisanti LA, Perez DM, Porter JE. Modulation of immune cell function by alpha(1)-adrenergic receptor activation. Curr Top Membr. 2011;67:113–138. doi: 10.1016/B978-0-12-384921-2.00006-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Averbeck B, Reeh PW, Michaelis M. Modulation of CGRP and PGE2 release from isolated rat skin by alpha-adrenoceptors and kappa-opioid-receptors. Neuroreport. 2001;12(10):2097–2100. doi: 10.1097/00001756-200107200-00011. [DOI] [PubMed] [Google Scholar]
- 31.Lonne-Rahm SB, Fischer T, Berg M. Stinging and rosacea. Acta Dermatovenereol. 1999;79(6):460–461. doi: 10.1080/000155599750009915. [DOI] [PubMed] [Google Scholar]
- 32.Dirschka T, Tronnier H, Folster-Holst R. Epithelial barrier function and atopic diathesis in rosacea and perioral dermatitis. Br J Dermatol. 2004;150(6):1136–1141. doi: 10.1111/j.1365-2133.2004.05985.x. [DOI] [PubMed] [Google Scholar]
- 33.Helfrich YR, Maier LE, Cui Y, Fisher GJ, Chubb H, Fligiel S, et al. Clinical, histologic, and molecular analysis of differences between erythematotelangiectatic rosacea and telangiectatic photoaging. JAMA Dermatol. 2015;151(8):825–836. doi: 10.1001/jamadermatol.2014.4728. [DOI] [PubMed] [Google Scholar]
- 34.Sulk M, Seeliger S, Aubert J, Schwab VD, Cevikbas F, Rivier M, et al. Distribution and expression of non-neuronal transient receptor potential (TRPV) ion channels in rosacea. J Invest Dermatol. 2012;132(4):1253–1262. doi: 10.1038/jid.2011.424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Steinhoff M, Vergnolle N, Young SH, Tognetto M, Amadesi S, Ennes HS, et al. Agonists of proteinase-activated receptor 2 induce inflammation by a neurogenic mechanism. Nat Med. 2000;6(2):151–158. doi: 10.1038/72247. [DOI] [PubMed] [Google Scholar]
- 36.Gilsbach R, Hein L. Are the pharmacology and physiology of alpha(2) adrenoceptors determined by alpha(2)-heteroreceptors and autoreceptors respectively? Br J Pharmacol. 2012;165(1):90–102. doi: 10.1111/j.1476-5381.2011.01533.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schauber J. Antimicrobial peptides, Vitamin D(3) and more. How rosacea may develop. Hautarzt. 2011;62(11):815–819. doi: 10.1007/s00105-011-2142-9. [DOI] [PubMed] [Google Scholar]
- 38.Schauber J, Gallo RL. Antimicrobial peptides and the skin immune defense system. J Allergy Clin Immunol. 2009;124(3 Suppl 2):R13–R18. doi: 10.1016/j.jaci.2009.07.014. [DOI] [PubMed] [Google Scholar]
- 39.Yamasaki K, Kanada K, Macleod DT, Borkowski AW, Morizane S, Nakatsuji T, et al. TLR2 expression is increased in rosacea and stimulates enhanced serine protease production by keratinocytes. J Invest Dermatol. 2011;131(3):688–697. doi: 10.1038/jid.2010.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Li Q, Verma IM. NF-kappaB regulation in the immune system. Nat Rev Immunol. 2002;2(10):725–734. doi: 10.1038/nri910. [DOI] [PubMed] [Google Scholar]
- 41.Lorton D, Bellinger DL. Molecular mechanisms underlying beta-adrenergic receptor-mediated cross-talk between sympathetic neurons and immune cells. Int J Mol Sci. 2015;16(3):5635–5665. doi: 10.3390/ijms16035635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Steinhoff MS, von Mentzer B, Geppetti P, Pothoulakis C, Bunnett NW. Tachykinins and their receptors: contributions to physiological control and the mechanisms of disease. Physiol Rev. 2014;94(1):265–301. doi: 10.1152/physrev.00031.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Galandrin S, Bouvier M. Distinct signaling profiles of beta1 and beta2 adrenergic receptor ligands toward adenylyl cyclase and mitogen-activated protein kinase reveals the pluridimensionality of efficacy. Mol Pharmacol. 2006;70(5):1575–1584. doi: 10.1124/mol.106.026716. [DOI] [PubMed] [Google Scholar]
- 44.Luttrell LM, Gesty-Palmer D. Beyond desensitization: physiological relevance of arrestin-dependent signaling. Pharmacol Rev. 2010;62(2):305–330. doi: 10.1124/pr.109.002436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ghanouni P, Gryczynski Z, Steenhuis JJ, Lee TW, Farrens DL, Lakowicz JR, et al. Functionally different agonists induce distinct conformations in the G protein coupling domain of the beta 2 adrenergic receptor. J Biol Chem. 2001;276(27):24433–24436. doi: 10.1074/jbc.C100162200. [DOI] [PubMed] [Google Scholar]
- 46.Reiner S, Ambrosio M, Hoffmann C, Lohse MJ. Differential signaling of the endogenous agonists at the beta2-adrenergic receptor. J Biol Chem. 2010;285(46):36188–36198. doi: 10.1074/jbc.M110.175604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kenakin T, Christopoulos A. Signalling bias in new drug discovery: detection, quantification and therapeutic impact. Nat Rev Drug Discov. 2013;12(3):205–216. doi: 10.1038/nrd3954. [DOI] [PubMed] [Google Scholar]
- 48.Small KM, Schwarb MR, Glinka C, Theiss CT, Brown KM, Seman CA, et al. Alpha2A- and alpha2C-adrenergic receptors form homo- and heterodimers: the heterodimeric state impairs agonist-promoted GRK phosphorylation and beta-arrestin recruitment. Biochemistry. 2006;45(15):4760–4767. doi: 10.1021/bi052074z. [DOI] [PubMed] [Google Scholar]
- 49.Violin JD, Lefkowitz RJ. Beta-arrestin-biased ligands at seven-transmembrane receptors. Trends Pharmacol Sci. 2007;28(8):416–422. doi: 10.1016/j.tips.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 50.Whalen EJ, Rajagopal S, Lefkowitz RJ. Therapeutic potential of beta-arrestin- and G protein-biased agonists. Trends Mol Med. 2011;17(3):126–139. doi: 10.1016/j.molmed.2010.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wisler JW, Xiao K, Thomsen AR, Lefkowitz RJ. Recent developments in biased agonism. Curr Opin Cell Biol. 2014;27:18–24. doi: 10.1016/j.ceb.2013.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Di Nardo A, Vitiello A, Gallo RL. Cutting edge: mast cell antimicrobial activity is mediated by expression of cathelicidin antimicrobial peptide. J Immunol. 2003;170(5):2274–2278. doi: 10.4049/jimmunol.170.5.2274. [DOI] [PubMed] [Google Scholar]
- 53.Moormann C, Artuc M, Pohl E, Varga G, Buddenkotte J, Vergnolle N, et al. Functional characterization and expression analysis of the proteinase-activated receptor-2 in human cutaneous mast cells. J Invest Dermatol. 2006;126(4):746–755. doi: 10.1038/sj.jid.5700169. [DOI] [PubMed] [Google Scholar]
- 54.Shahane G, Parsania C, Sengupta D, Joshi M. Molecular insights into the dynamics of pharmacogenetically important N-terminal variants of the human beta2-adrenergic receptor. PLoS Comput Biol. 2014;10(12):e1004006. doi: 10.1371/journal.pcbi.1004006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Practice Update. Available from: http://www.practiceupdate.com/content/paradoxical-erythema-reaction-to-long-term-brimonidine-gel-for-facial-erythema-of-rosacea/41069/65/4/1#collapse1. Accessed August 1, 2016.