Abstract
Transarterial chemoembolization (TACE) is a widely used standard treatment for patients with hepatocellular carcinoma (HCC) who are not suitable candidates for curative treatments. The rationale for TACE is that intra-arterial chemotherapy using lipiodol and chemotherapeutic agents, followed by selective vascular embolization, results in a strong cytotoxic effect as well as ischemia (conventional TACE). Recently, drug-eluting beads (DC Beads®) have been developed for transcatheter treatment of HCC to deliver higher doses of the chemotherapeutic agent and to prolong contact time with the tumor. DC Beads® can actively sequester doxorubicin hydrochloride from solution and release it in a controlled sustained fashion. Treatment with DC Beads® substantially reduced the amount of chemotherapeutic agent that reached the systemic circulation compared with conventional, lipiodol-based regimens, significantly reducing drug-related adverse events. In this article, we describe the treatment response, survival, and safety of TACE used with drug-eluting beads for the treatment of HCC and discuss future therapeutic possibilities.
Keywords: Hepatocellular carcinoma, Transarterial chemoembolization, Conventional TACE, Drug-eluting beads, Treatment response
Core tip: Drug eluting beads are relatively new embolic agents that allow sustained release of chemotherapeutic agents in a localized fashion to the tumor. The advantage of DC bead transarterial chemoembolization (TACE) is a better combined ischemic and cytotoxic effect locally and less system toxicity when compared with conventional TACE.
INTRODUCTION
Hepatocellular carcinoma (HCC) is the most common liver malignancy worldwide[1,2]. Unlike other malignancies, the incidence and mortality rates of HCC will continue to increase through over the next 20 years in some countries[3,4]. The incidence of HCC is influenced by major risk factors that include hepatitis B virus, hepatitis C virus, alcohol consumption and nonalcoholic steatohepatitis. The majority of patients with HCC present with advanced disease at the time of diagnosis and are not candidates for curative therapies such as surgery, transplantation, or radiofrequency ablation[5].
Transarterial chemoembolization (TACE) is the most commonly used as palliative treatment for patients with unresectable HCC[6,7]. The principle of conventional TACE (cTACE) is the synergistic effect of cytotoxic chemotherapy and ischemia. Intra-arterial chemotherapeutic agents mixed with lipiodol cause cytotoxic damage in tumor cells, and embolization of feeding vessels by gelatine or Gelform particles results in ischemia[8]. In a meta-analysis, Llovet et al[9] reported that TACE results in a significantly higher survival rate compared with best supportive care in well-selected cases.
From a pharmacokinetics perspective, for the best effect, higher doses of the intra-arterial chemotherapeutic agent should be retained within the tumor. Furthermore, a chemotherapeutic drug that is released can reduce systemic side effects. Drug-eluting beads had been developed with these objectives in mind[10]. DC Beads® (Biocompatibles, Surrey, United Kingdom) can load and release doxorubicin hydrochloride in a controlled manner[11]. TACE used with beads loaded with doxorubicin (DEBDOX) induced significantly fewer drug-related side effects compared with cTACE[10,12].
Several studies have shown promising outcomes of TACE with DEBDOX for intermediate-stage HCC, and this could be an alternative to cTACE. Here, we summarize recent results for the use of TACE with drug-eluting beads in terms of the treatment response, survival, and adverse events and discuss future therapeutic possibilities.
TECHNICAL ASPECT AND CHARACTERISTICS OF DC BEADS
There is no standard method for conducting cTACE. The procedure involves local infusion of chemotherapeutic agents with selective embolization of the HCC feeding arteries; however, the selection of the chemotherapeutic agent contrast agent, and embolization material vary from center to center and country to country[13]. From a technical perspective, cTACE should be as selective as possible, and further standardization of cTACE protocols is needed[14]. Inconsistency with using the technique is the major obstacle to standardization of the cTACE procedure. Potentially, TACE with DEBDOX could maintain sufficient consistency and repeatability during the procedure to allow its use as a standard treatment for HCC.
DC Beads are hydrogel microspheres that are biocompatible, hydrophilic, non-resorbable, precisely calibrated, and capable of loading chemotherapeutic agents. DC Beads are produced from a polyvinyl alcohol hydrogel that has been modified with sulfonate groups for the controlled loading and delivery of chemotherapeutic drugs, such as doxorubicin and irinotecan[11,15]. Positively charged doxorubicin HCl is drawn to the negatively charged sulfonate groups of the DC beads spheres by an ion-exchange mechanism. After mixing the doxorubicin, DC beads suspension, and contrast agent, only 0.2% of the doxorubicin remains free in the systemic circulation. The low rate of doxorubicin release could minimize the systemic side effects of TACE with DEBDOX compared with cTACE[12,16]. The doxorubicin is gradually sequestered inside the tumor because the drug dissociates from the DC beads only under specific ionic circumstances such as those found in tumor cells.
DOXORUBICIN LOADING AND PLANNED DOSING
DC Beads microspheres are packaged in 2 mL vials of hydrated beads in sodium phosphate solution. Each vial can be loaded with 50-75 mg doxorubicin. The maximum dose for a single treatment has been set at 150 mg, based on a study showing that this was the maximum dose for systemic infusion of doxorubicin[10].The dose of doxorubicin should depend on the tumor burden. However, absolute recommendations are difficult to determine due to individual patient- and tumor-related factors. Expert consensus hold that the doxorubicin dose should be varied based on the tumor status defined by the Milan criteria for liver transplantation (a solitary tumor ≤ 5 cm or a maximum of three tumors all ≤ 3 cm)[14].
For limited disease (within the Milan criteria), the treatment strategy needs to include escalation of the doxorubicin dose up to 75 mg per single TACE. For advanced disease (exceeding the Milan criteria), the doxorubicin dose could be increased to a maximum of 150 mg[14,17].
SELECTION OF THE BEADS DIAMETER
The DC Beads come in four sizes: 100-300, 300-500, 500-700, and 700-900 μm. HCC with an arteriovenous shunt (AV) confers an increased risk of pulmonary complications during TACE[18]. Therefore, the bead size should be chosen carefully, especially for patients with an AV shunt.
Histological analysis of HCC explants treated with DC Beads showed that 100-300 μm beads are distributed both within and at the periphery of the tumor[19]. Lewis et al[13] showed that 100-300 μm beads triggered broad necrosis of the tumor and adjacent peritumoral hepatic tissue without causing vasculopathy or inflammation[20]. As shown in Figure 1, the cytotoxic agent and ischemia induce profound liquefaction and necrosis of the tumor.
Figure 1.
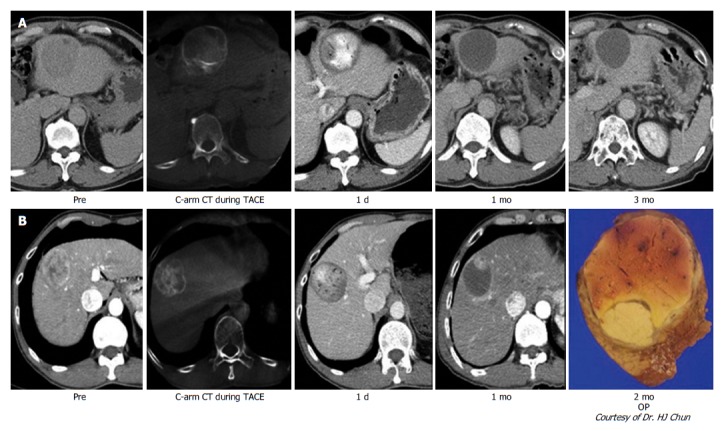
Typical imaging finding after transarterial chemoembolization with drug-eluting bead. A: Nodular hepatocellular carcinoma (HCC) with arterial enhancement showed the total necrosis of HCC through follow-up computed tomography (CT) imaging; B: Nodular HCC showed nodular arterial enhancing viable portion within the partial necrosis of HCC in follow-up CT imaging. After operation, viable HCC in resected HCC showed the matched lesion in the CT imaging.
In comparison, DC Beads with larger diameters (700-900 μm) induce limited necrosis compared with smaller beads[21]. Beads with smaller diameters can be delivered to distal vessels where they obstruct collateral channels, while, larger beads occlude more proximal vessels, allowing blood to be supplied to the tumor via collateral vessels, which are not occluded by the beads[17]. Recently, the diameters of drug-eluting microspheres are smaller than that of DC Beads are introduced in some studies[22-24]. The diameter of each microsphere is as follows: HepaSphere; 30-60 μm[22], M1 DC Beads; 70-150 μm[23], TANDEM; 40-100 μm[24], respectively.
INDICATIONS
The indications for DC Beads use with TACE (DC Beads TACE) are similar to those for cTACE. DC Beads TACE may be a better option, particularly in patients with more advanced liver disease (Child-Pugh B, Eastern Cooperative Oncology Group (ECOG) 2, Barcelona Clinic Liver Cancer (BCLC) C, bilobar, or recurrent disease) or patients with mild to moderate cardiac failure[25].
RESPONSE TO DC BEADS TACE
Varela et al[10] reported a 75% overall response rate to TACE with DEBDOX in a phase I/II clinical trial that included tumors with a mean diameter of 4.6 cm, and the majority of patients were Okuda I (Okuda I/II: 26/1). In another study, Malagari et al[16] reported a complete response (CR) was accomplished in 15.5% of 71 patients (mean tumor size 6.2 cm) who underwent DC Beads TACE. Furthermore, a 66.2%-85.5% objective response (OR) rate was achieved after an additional four cycles of treatment in the same study group. Poon et al[12] reported the treatment responses of patients with multiple larger HCCs, in whom the mean number of tumors was 3 ± 3, the maximum tumor size was 7.6 ± 4.8 cm, and the summed tumor size was 10.0 ± 5.8 cm. While treatment response rates of 16%-35% have been reported in past cTACE studies[26,27], a favorable treatment response was seen in this larger HCC group; the OR rate 1 mo after DC Beads TACE was 70% [CR = 6.7%; partial response (PR) 63.3%] using the modified Response Evaluation Criteria in Solid Tumors[12].
The PRECISION V prospective clinical trial reported a 6-mo OR rate of 52%, which is comparable to the rates of 44%-82% seen in previous phase I/II studies[24]. Although statistical significance was not achieved in terms of superior OR rates for DC Beads TACE over cTACE, the formed seemed to be associated with higher response rates in terms of CR, OR, and disease control. Moreover, DC Beads proved to be effective in those with advanced disease as shown by the improved treatment response and disease control with acceptable safety profiles. This may provide a niche for those with poorer conditions, such as patients with Child-Pugh B and ECOG 1 disease, for whom the effect of conventional treatment has been minimal.
In an Asian case-control study, Song et al[28] reported that larger tumors (> 5 cm) or multiple HCCs showed a better treatment response to DC Beads TACE than to cTACE. Another comparative study showed that the OR rate of the DC Beads group was significantly better than that of the cTACE group (81.6% vs 49.2%, P < 0.001). A subgroup analysis confirmed that intermediate-stage HCC had a significantly higher OR and time to progression when treated with DC Beads TACE compared with cTACE (75.7% vs 34.1%, P < 0.001; 11.7 vs 7.6 mo, P = 0.018, respectively). In terms of liver toxicity, the DC Beads group did not differ significantly from the cTACE group (P > 0.05)[29]. This lack of significance may be ascribable to the heterogeneous ethnicities of the patient population and various etiologies of the underlying liver disease. This suggests the importance of strict patient selection with considerate and delicate angiographic techniques when conducting TACE.
However, these significant improvements in treatment responses of DC Beads TACE as compared to those of cTACE were not proven in other studies. In two randomized controlled trials (RCTs), Golfieri et al[30] reported treatment response of 177 HCC patients involving 89 in DC Beads TACE and 88 in cTACE. OR rates at 3 mo showed 74.7% and 74.1% for DC Beads TACE and cTACE, respectively (P > 0.999). Also, Sacco et al[31] showed statistically insignificant differences in CR and PR rates at 1 mo between DC Beads TACE and cTACE (51.5% and 48.5% vs 70.6% and 29.4%, respectively, P = 0.1). Facciorusso et al[32] reported single center study with early/intermediate HCC patients (n = 249). In this study, cTACE showed better tumor response and time to progression (TTP). OR rates were 85.3% in cTACE and 74.8% in DC Beads TACE (P = 0.039), and median TTP were 17 mo in cTACE and 11 mo in DC Beads TACE, respectively (P < 0.001).
Prajapati et al[33] reported the safety and efficacy of DC Beads TACE in 121 patients with advanced HCC whose median OS was 13.5 mo. DC Beads TACE was associated with a favorable prognosis, especially in those without portal vein thrombosis (PVT) and metastasis (28.9%) compared with those with PVT and metastasis (9.9%) within the Child-Pugh A group (median survival 18.8 mo vs 4.4 mo, P = 0.001). Moreover, the few minor adverse events associated with the treatment imply that DC Beads TACE is an alternative treatment strategy with advanced disease. Nevertheless, prospective studies involving more cases are needed to evaluate the efficacy and safety of TACE used drug-eluting beads in BCLC C patients.
Han et al[34] performed a meta-analysis of three RCTs and two case-control studies. Their meta-analysis included DC Beads TACE and cTACE groups comprising 217 and 237 patients, respectively. The results showed that DC Beads TACE tended to have better results in terms of disease control, although the difference was not significant.
CLINICAL IMPACT OF MICROSPHERES WITH SMALLER DIAMETERS
As mentioned above, several beads with smaller diameters were introduced in recent studies. Malagari et al[22] performed study with HepaSphere. HepaSphere 30-60 μm is a microsphere that has a dry caliber of 30-60 μm that expands to 145-213 (148 ± 45) μm after loading with doxorubicin[35]. In this report, HepaSphere 30-60 μm showed 68.9% of OR without serious adverse events.
Spreafico et al[23] performed study using TACE with M1 DC bead for HCC patients who were not indicated to resection or ablation (n = 45). M1 DC bead is newly developed DC bead with diameter of 70-150 μm. In this study, 77.7% of patients obtained OR (CR in 33.3%) and, 78% of tumor nodules achieved OR (CR in 42%). Thirteen patients (29%) underwent surgery after achieving successful tumor downstaging by TACE with M1. (Liver transplantation = 13, Major hepatectomy = 1). Adverse events occurred in limited cases, most of which were insignificant clinical outcomes (grade 1/2).
Malagari et al[24] reported study with TANDEM microsphere. TANDEM is precisely calibrated microspheres that diameters with 40, 75, and 100 μm were used in this study. Fifty-one HCC patients who were not amenable to curative therapy were enrolled in this study (mean diameter of the tumors; 7.28 ± 2.09 cm). At 6 mo follow-up, 63.82% of patients achieved OR (CR in 21.27%). And OS at 1, 2, and 3 years were 92.3%, 88.46%, 82.6%, respectively. Majority of adverse events were mild post-embolization syndrome and 4 cases of serious adverse events (grade 3-5) were reported. Patients with high loading dose of doxorubicin (150 mg) were associated with biliary damage.
EVALUATING THE TREATMENT RESPONSE TO DC BEADS TACE
To evaluate the response to cTACE, liver dynamic computed tomography (CT) should be performed 4 wk after therapy to determine the future treatment plan[36]. Especially, the treatment response to DC Beads TACE can be evaluated more definitely than can that to cTACE, in which incomplete lipiodol uptake causes difficulty with evaluating the treatment response (Figure 2)[37].
Figure 2.
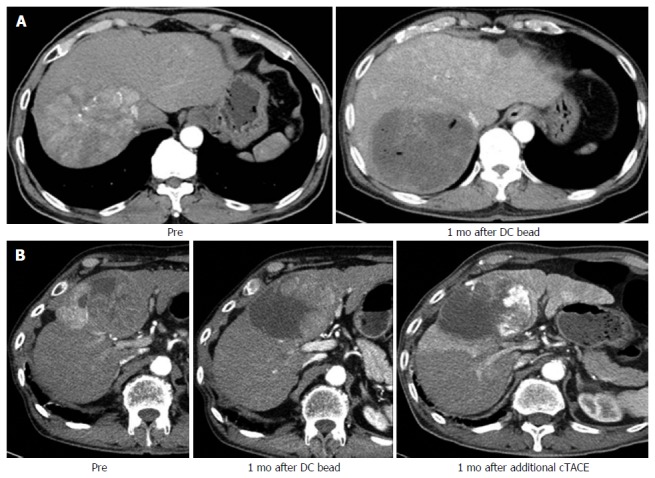
Difference of imaging finding between transarterial chemoembolization with drug-eluting bead and conventional transarterial chemoembolization. A: Two nodular hepatocellular carcinomas (HCCs) with arterial enhancement showed the total necrosis of HCC in follow-up liver dynamic CT after treating with DC bead; B: Nodular HCC showed nodular arterial enhancing viable portion within the partial necrosis of HCC in follow-up CT after treating with DC bead. After additional conventional transarterial chemoembolization (TACE), incomplete lipiodol uptake in remained HCC showed the matched lesion in the CT imaging. CT: Computed tomography.
Chung et al[37] reported that the enhancement patterns of HCC after TACE used with beads could be useful for determining the prognosis. They analyzed images of the arterial phase of dynamic liver CT 1 mo after TACE with beads and categorized the enhancement patterns as no enhancement, peripheral ring enhancement, or peripheral nodule-like enhancement. Peripheral nodule-like enhancements suggest disease progression, while no enhancement or peripheral ring enhancements indicated a CR.
Golowa et al[38] showed that DEBDOX mixed with contrast medim helps demonstrates the precise distribution and uptake of the chemotherapeutic agent without disrupting the treatment response evaluation. The contrast medium enabled visualization of the treated tumor due to the increased attenuation, similar to that of lipiodol. Unlike lipiodol, however, the contrast medium was not retained within the tumor. Noncontrast CT immediately after TACE was helpful to evaluate the treatment response.
SURVIVAL
Malagari et al[39] reported a 5-year survival analysis of patients treated with DC Beads TACE. The mean OS was 43.8 (range 1.2-64.8) mo, and the OS rates at 1, 3, and 5 years were 93.6%, 62%, and 22.5%, respectively. With respect to the Child-Pugh class, the 5-years OS, rate was higher for Child class A than Child class B (29.4% vs 12.8%). For DC Beads TACE, the number of lesions, lesion vascularity, and local response were significant independent determinants of 5-year survival.
Burret et al[40] reported the survival of 104 patients with HCC treated with DC Beads TACE. The median survival was 48.6 mo. The OS rates at 1, 3, and 5 years were 89.9%, 66.3%, and 38.3%, respectively. According to the BCLC stage, the median survival and OS rates at 1, 3, and 5 years for patients with early-stage disease was 54.2 mo and 89.7%, 67.8%, 33.9%, respectively, versus 47.7 mo and 88.2%, 64.4%, and 39.4%, respectively, for patients with intermediate-stage disease. This study reported better survival rates with DC Beads than with cTACE.
Song et al[29] compared prognostic factors that affected survival rates in patients with HCC treated with DC Beads TACE (n = 60) vs cTACE (n = 69). DC Beads TACE was a significant independent factor associated with better survival (P = 0.037). Moreover, the alpha-fetoprotein level and BCLC stage were significant predictors of superior patient survival. These results suggest that the use of DEBDOX is related to a favorable outcome in patients with unresectable HCC.
However, the survival benefits of DC Beads TACE over cTACE remain controversial. Recchia et al[41] reported retrospective study that included 35 patients of DEB-TACE and 70 patients of cTACE. There was no statistically significant difference in median OS between cTACE and DEB-TACE (11.4 mo vs 18.4 mo, respectively). Facciorusso et al[32] also reported that median survival of early/intermediate stage HCC patients (n = 249) between cTACE and DC Beads TACE showed insignificant differences (39 mo vs 32 mo, respectively, P = 0.1).
In two RCTs, Golfieri et al[30] reported the 2 year survival rates between DC Beads TACE and cTACE (56.8% vs 55.4%, respectively, P = 0.949) of 117 HCC patients. Sacco et al[31] also showed that estimated 2-year cumulative survival rates were statistically insignificant between DC Beads TACE and cTACE (86.8% vs 83.6%, respectively, P = 0.96). Furthermore, Facciorusso et al[42] reported meta-analysis consisted of four RCTs and 8 observational studies with 1449 patients who underwent 689 DC Beads TACE and 760 cTACE. In this study, statistically insignificant trends in favor of DC Beads TACE were observed for 3-year survival rates.
SAFETY AND COMPLICATIONS
The complications reported with DC Beads include pleural effusion, gastric ulcer bleeding, cholecystitis, and abscess formation, with the prevalence ranging from 4.2% to 11.4%[10,12,16,43]. These complication rates are comparable to those of cTACE. However, it is noteworthy that no systemic complicates associated with doxorubicin have been reported in the clinical studies performed so far.
Guiu et al[44] reported that biloma and liver infarct were independently related to DEB-TACE (OR = 9.78, P = 0.002) (Figure 3). Interestingly, a non-cirrhotic underlying liver was a strong independent risk factor for developing biloma and liver infarct (OR = 8.125, P = 0.04). The role of a hypertrophied peribiliary plexus as a porto-arterial shunt may account for the association observed in this study[45]. These changes seen in the cirrhotic liver reinforce collateral formation, which prevents further ischemic and chemical injury in the bile ducts.
Figure 3.
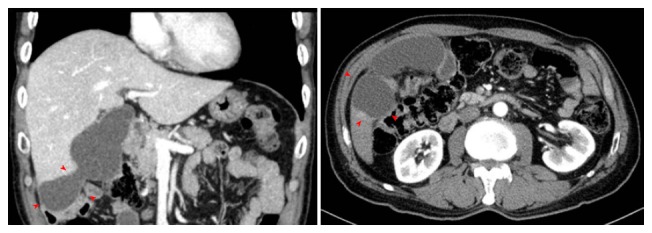
Biloma in a patient treated by transarterial chemoembolization with drug-eluting bead.
Systemic complications related to doxorubicin include neurological injury, pulmonary edema, bone marrow suppression, and gastrointestinal problem (nausea and vomiting). Especially, dose-dependent cardiomyopathy (adriamycin-induced congestive heart failure) limits its long-term use. Advanced age and left ventricular dysfunction increase the risk of this complication. At a cumulative doxorubicin dose exceeding 450 mg/m2, it is important to monitor for this high-risk of cardiovascular complication[46]. It is worth noting that no doxorubicin-related systemic complications have been observed in clinical studies of DC Beads TACE performed to date.
ASSESSMENT OF COST EFFECTIVENESS
Cucchetti et al[47] investigated the cost effectiveness of DC Beads TACE. In meta-analysis, patients with cTACE experienced significantly frequent post-TACE syndrome (P = 0.018) and longer hospitalization (P = 0.01). DC Beads TACE earned 4.0 quality-adjusted life-years (QALYs) while cTACE earned 3.3 QALYs. Total costs of DC Beads TACE were € 11656 and those of cTACE were € 10389. DC Beads TACE spent higher costs than cTACE, but, higher QALYs were achieved from the treatment. Expected cost-effectiveness for DC Beads TACE was € 3089/QALY and that of cTACE was € 3246/QALY. Improvement of quality of life could be attained by DC Beads TACE with modest increment of costs.
CONCLUSION
Intermediate- and advanced-stage HCCs remain a challenging to physicians and interventional radiologists because of suboptimal tumor control and frequent relapse even after inducing complete tumor necrosis using a catheter-based approach. The potent anticancer effect of drug-eluting beads administered through the hepatic arteries might complement standard therapeutic modalities[34,48]. DC Beads are relatively new embolic agents that allow sustained release of chemotherapeutic agents and minimize systemic side effects. The advantages of DC Beads TACE are better synergistic effects with embolization and cytotoxic effects and minimized system toxicity compared with cTACE. Especially, regarding the treatment results for unresectable single tumors or multiple tumors (intermediate stage), Child B class, and recurrent HCC, TACE used with drug-eluting Beads resulted in a better treatment response rate than did cTACE. However, cTACE may provide better outcomes than those of DEBDOX TACE, when superselective embolization of the small feeding vessels is possible.
The biliary damage caused by DC Beads TACE should be weighed against their use in selected patients. A more careful therapeutic approach to prevent of biloma or liver infarct development is needed in patients with an underlying non-cirrhotic liver. For effective TACE with DEBDOX, the duration, distribution, and dosage of the drug delivered to the tumor and surrounding non-tumor tissue are most important[19]. We must consider the doxorubicin dose, diameter of the DC Beads and tumor vascularity required for the optimal treatment response before performing TACE with DEBDOX.
DC Beads TACE may provide improved survival rates and quality of life to some extent as compared to cTACE[32,41,47,49]. However, several RCTs and meta-analysis do not demonstrate significant survival advantages of DC Beads TACE[30,31,42]. Because of relatively small numbers of prospective randomized trials, further investigations with well designed, large scaled, comparative studies searching for the long-term survival are necessary. DC Beads TACE seemed to afford better cost-effectiveness than cTACE[47]. However, previous studies were based on various conditions involving different countries and institutes in terms of costs, clinical circumstances, and technical procedures. Study with standardized technical protocols and performances will be needed to validate the cost-effectiveness of DC Beads TACE.
Indeed, it remains inconclusive as to the superiority of DC Beads TACE over cTACE. Decision to performing DC Beads TACE or cTACE needs to be tailored in each individual patient depending on his or her economic status, physician’s experiences, and expertise of institute. Future studies are warranted to determine the appropriate indications of DC Beads TACE.
Footnotes
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: South Korea
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
Conflict-of-interest statement: There is no conflict of interest.
Peer-review started: July 18, 2016
First decision: August 8, 2016
Article in press: September 14, 2016
P- Reviewer: Hoyos S, Mazzaferro V S- Editor: Qi Y L- Editor: A E- Editor: Zhang FF
References
- 1.El-Serag HB, Mason AC. Rising incidence of hepatocellular carcinoma in the United States. N Engl J Med. 1999;340:745–750. doi: 10.1056/NEJM199903113401001. [DOI] [PubMed] [Google Scholar]
- 2.Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94:153–156. doi: 10.1002/ijc.1440. [DOI] [PubMed] [Google Scholar]
- 3.Olsen AH, Parkin DM, Sasieni P. Cancer mortality in the United Kingdom: projections to the year 2025. Br J Cancer. 2008;99:1549–1554. doi: 10.1038/sj.bjc.6604710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Davis GL, Alter MJ, El-Serag H, Poynard T, Jennings LW. Aging of hepatitis C virus (HCV)-infected persons in the United States: a multiple cohort model of HCV prevalence and disease progression. Gastroenterology. 2010;138:513–521, 521.e1-6. doi: 10.1053/j.gastro.2009.09.067. [DOI] [PubMed] [Google Scholar]
- 5.Bruix J, Sherman M, Llovet JM, Beaugrand M, Lencioni R, Burroughs AK, Christensen E, Pagliaro L, Colombo M, Rodés J. Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol. 2001;35:421–430. doi: 10.1016/s0168-8278(01)00130-1. [DOI] [PubMed] [Google Scholar]
- 6.Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022. doi: 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, Sherman M, Schwartz M, Lotze M, Talwalkar J, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008;100:698–711. doi: 10.1093/jnci/djn134. [DOI] [PubMed] [Google Scholar]
- 8.Lencioni R, Crocetti L. Local-regional treatment of hepatocellular carcinoma. Radiology. 2012;262:43–58. doi: 10.1148/radiol.11110144. [DOI] [PubMed] [Google Scholar]
- 9.Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37:429–442. doi: 10.1053/jhep.2003.50047. [DOI] [PubMed] [Google Scholar]
- 10.Varela M, Real MI, Burrel M, Forner A, Sala M, Brunet M, Ayuso C, Castells L, Montañá X, Llovet JM, et al. Chemoembolization of hepatocellular carcinoma with drug eluting beads: efficacy and doxorubicin pharmacokinetics. J Hepatol. 2007;46:474–481. doi: 10.1016/j.jhep.2006.10.020. [DOI] [PubMed] [Google Scholar]
- 11.Hong K, Khwaja A, Liapi E, Torbenson MS, Georgiades CS, Geschwind JF. New intra-arterial drug delivery system for the treatment of liver cancer: preclinical assessment in a rabbit model of liver cancer. Clin Cancer Res. 2006;12:2563–2567. doi: 10.1158/1078-0432.CCR-05-2225. [DOI] [PubMed] [Google Scholar]
- 12.Poon RT, Tso WK, Pang RW, Ng KK, Woo R, Tai KS, Fan ST. A phase I/II trial of chemoembolization for hepatocellular carcinoma using a novel intra-arterial drug-eluting bead. Clin Gastroenterol Hepatol. 2007;5:1100–1108. doi: 10.1016/j.cgh.2007.04.021. [DOI] [PubMed] [Google Scholar]
- 13.Lewis AL, Taylor RR, Hall B, Gonzalez MV, Willis SL, Stratford PW. Pharmacokinetic and safety study of doxorubicin-eluting beads in a porcine model of hepatic arterial embolization. J Vasc Interv Radiol. 2006;17:1335–1343. doi: 10.1097/01.RVI.0000228416.21560.7F. [DOI] [PubMed] [Google Scholar]
- 14.Lencioni R, de Baere T, Burrel M, Caridi JG, Lammer J, Malagari K, Martin RC, O’Grady E, Real MI, Vogl TJ, et al. Transcatheter treatment of hepatocellular carcinoma with Doxorubicin-loaded DC Bead (DEBDOX): technical recommendations. Cardiovasc Intervent Radiol. 2012;35:980–985. doi: 10.1007/s00270-011-0287-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lewis AL, Gonzalez MV, Lloyd AW, Hall B, Tang Y, Willis SL, Leppard SW, Wolfenden LC, Palmer RR, Stratford PW. DC bead: in vitro characterization of a drug-delivery device for transarterial chemoembolization. J Vasc Interv Radiol. 2006;17:335–342. doi: 10.1097/01.RVI.0000195323.46152.B3. [DOI] [PubMed] [Google Scholar]
- 16.Malagari K, Alexopoulou E, Chatzimichail K, Hall B, Koskinas J, Ryan S, Gallardo E, Kelekis A, Gouliamos A, Kelekis D. Transcatheter chemoembolization in the treatment of HCC in patients not eligible for curative treatments: midterm results of doxorubicin-loaded DC bead. Abdom Imaging. 2008;33:512–519. doi: 10.1007/s00261-007-9334-x. [DOI] [PubMed] [Google Scholar]
- 17.Malagari K. Drug-eluting particles in the treatment of HCC: chemoembolization with doxorubicin-loaded DC Bead. Expert Rev Anticancer Ther. 2008;8:1643–1650. doi: 10.1586/14737140.8.10.1643. [DOI] [PubMed] [Google Scholar]
- 18.Lee JH, Won JH, Park SI, Won JY, Lee DY, Kang BC. Transcatheter arterial chemoembolization of hepatocellular carcinoma with hepatic arteriovenous shunt after temporary balloon occlusion of hepatic vein. J Vasc Interv Radiol. 2007;18:377–382. doi: 10.1016/j.jvir.2007.01.005. [DOI] [PubMed] [Google Scholar]
- 19.Namur J, Citron SJ, Sellers MT, Dupuis MH, Wassef M, Manfait M, Laurent A. Embolization of hepatocellular carcinoma with drug-eluting beads: doxorubicin tissue concentration and distribution in patient liver explants. J Hepatol. 2011;55:1332–1338. doi: 10.1016/j.jhep.2011.03.024. [DOI] [PubMed] [Google Scholar]
- 20.Lewis AL, Taylor RR, Hall B, Gonzalez MV, Willis SL, Stratford PW. Pharmacokinetic and safety study of doxorubicin-eluting beads in a porcine model of hepatic arterial embolization. J Vasc Interv Radiol. 2006;17:1335–1343. doi: 10.1097/01.RVI.0000228416.21560.7F. [DOI] [PubMed] [Google Scholar]
- 21.Gonzalez MV, Tang Y, Phillips GJ, Lloyd AW, Hall B, Stratford PW, Lewis AL. Doxorubicin eluting beads-2: methods for evaluating drug elution and in-vitro: in-vivo correlation. J Mater Sci Mater Med. 2008;19:767–775. doi: 10.1007/s10856-006-0040-y. [DOI] [PubMed] [Google Scholar]
- 22.Malagari K, Pomoni M, Moschouris H, Kelekis A, Charokopakis A, Bouma E, Spyridopoulos T, Chatziioannou A, Sotirchos V, Karampelas T, et al. Chemoembolization of hepatocellular carcinoma with HepaSphere 30-60 μm. Safety and efficacy study. Cardiovasc Intervent Radiol. 2014;37:165–175. doi: 10.1007/s00270-013-0777-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Spreafico C, Cascella T, Facciorusso A, Sposito C, Rodolfo L, Morosi C, Civelli EM, Vaiani M, Bhoori S, Pellegrinelli A, et al. Transarterial chemoembolization for hepatocellular carcinoma with a new generation of beads: clinical-radiological outcomes and safety profile. Cardiovasc Intervent Radiol. 2015;38:129–134. doi: 10.1007/s00270-014-0907-0. [DOI] [PubMed] [Google Scholar]
- 24.Malagari K, Kiakidis T, Pomoni M, Moschouris H, Emmanouil E, Spiridopoulos T, Sotirchos V, Tandeles S, Koundouras D, Kelekis A, et al. Pharmacokinetics, Safety, and Efficacy of Chemoembolization with Doxorubicin-Loaded Tightly Calibrated Small Microspheres in Patients with Hepatocellular Carcinoma. Cardiovasc Intervent Radiol. 2016;39:1379–1391. doi: 10.1007/s00270-016-1382-6. [DOI] [PubMed] [Google Scholar]
- 25.Lammer J, Malagari K, Vogl T, Pilleul F, Denys A, Watkinson A, Pitton M, Sergent G, Pfammatter T, Terraz S, et al. Prospective randomized study of doxorubicin-eluting-bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Intervent Radiol. 2010;33:41–52. doi: 10.1007/s00270-009-9711-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Raoul JL, Heresbach D, Bretagne JF, Ferrer DB, Duvauferrier R, Bourguet P, Messner M, Gosselin M. Chemoembolization of hepatocellular carcinomas. A study of the biodistribution and pharmacokinetics of doxorubicin. Cancer. 1992;70:585–590. doi: 10.1002/1097-0142(19920801)70:3<585::aid-cncr2820700308>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 27.A comparison of lipiodol chemoembolization and conservative treatment for unresectable hepatocellular carcinoma. Groupe d’Etude et de Traitement du Carcinome Hépatocellulaire. N Engl J Med. 1995;332:1256–1261. doi: 10.1056/NEJM199505113321903. [DOI] [PubMed] [Google Scholar]
- 28.Song MJ, Park CH, Kim JD, Kim HY, Bae SH, Choi JY, Yoon SK, Chun HJ, Choi BG, Lee HG. Drug-eluting bead loaded with doxorubicin versus conventional Lipiodol-based transarterial chemoembolization in the treatment of hepatocellular carcinoma: a case-control study of Asian patients. Eur J Gastroenterol Hepatol. 2011;23:521–527. doi: 10.1097/MEG.0b013e328346d505. [DOI] [PubMed] [Google Scholar]
- 29.Song MJ, Chun HJ, Song DS, Kim HY, Yoo SH, Park CH, Bae SH, Choi JY, Chang UI, Yang JM, et al. Comparative study between doxorubicin-eluting beads and conventional transarterial chemoembolization for treatment of hepatocellular carcinoma. J Hepatol. 2012;57:1244–1250. doi: 10.1016/j.jhep.2012.07.017. [DOI] [PubMed] [Google Scholar]
- 30.Golfieri R, Giampalma E, Renzulli M, Cioni R, Bargellini I, Bartolozzi C, Breatta AD, Gandini G, Nani R, Gasparini D, et al. Randomised controlled trial of doxorubicin-eluting beads vs conventional chemoembolisation for hepatocellular carcinoma. Br J Cancer. 2014;111:255–264. doi: 10.1038/bjc.2014.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sacco R, Bargellini I, Bertini M, Bozzi E, Romano A, Petruzzi P, Tumino E, Ginanni B, Federici G, Cioni R, et al. Conventional versus doxorubicin-eluting bead transarterial chemoembolization for hepatocellular carcinoma. J Vasc Interv Radiol. 2011;22:1545–1552. doi: 10.1016/j.jvir.2011.07.002. [DOI] [PubMed] [Google Scholar]
- 32.Facciorusso A, Mariani L, Sposito C, Spreafico C, Bongini M, Morosi C, Cascella T, Marchianò A, Camerini T, Bhoori S, et al. Drug-eluting beads versus conventional chemoembolization for the treatment of unresectable hepatocellular carcinoma. J Gastroenterol Hepatol. 2016;31:645–653. doi: 10.1111/jgh.13147. [DOI] [PubMed] [Google Scholar]
- 33.Prajapati HJ, Dhanasekaran R, El-Rayes BF, Kauh JS, Maithel SK, Chen Z, Kim HS. Safety and efficacy of doxorubicin drug-eluting bead transarterial chemoembolization in patients with advanced hepatocellular carcinoma. J Vasc Interv Radiol. 2013;24:307–315. doi: 10.1016/j.jvir.2012.11.026. [DOI] [PubMed] [Google Scholar]
- 34.Han S, Zhang X, Zou L, Lu C, Zhang J, Li J, Li M. Does drug-eluting bead transcatheter arterial chemoembolization improve the management of patients with hepatocellular carcinoma? A meta-analysis. PLoS One. 2014;9:e102686. doi: 10.1371/journal.pone.0102686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dinca H, Pelage J, Baylatry M, Ghegediban S, Pascale F, Manfait M, editors . Why do small size doxorubicin-eluting microspheres induce more tissue necrosis than larger ones? A comparative study in healthy pig liver (oral communication 2206-2). CIRSE Annual meeting, Lisbon; 2012 [Google Scholar]
- 36.European Association For The Study Of The Liver;; European Organisation For Research And Treatment Of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2012;56:908–943. doi: 10.1016/j.jhep.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 37.Chung WS, Lee KH, Park MS, Lee YJ, Kwon J, Baek SE, Kim MJ. Enhancement patterns of hepatocellular carcinoma after transarterial chemoembolization using drug-eluting beads on arterial phase CT images: a pilot retrospective study. AJR Am J Roentgenol. 2012;199:349–359. doi: 10.2214/AJR.11.7563. [DOI] [PubMed] [Google Scholar]
- 38.Golowa YS, Cynamon J, Reinus JF, Kinkhabwala M, Abrams M, Jagust M, Chernyak V, Kaubisch A. Value of noncontrast CT immediately after transarterial chemoembolization of hepatocellular carcinoma with drug-eluting beads. J Vasc Interv Radiol. 2012;23:1031–1035. doi: 10.1016/j.jvir.2012.04.020. [DOI] [PubMed] [Google Scholar]
- 39.Malagari K, Pomoni M, Moschouris H, Bouma E, Koskinas J, Stefaniotou A, Marinis A, Kelekis A, Alexopoulou E, Chatziioannou A, et al. Chemoembolization with doxorubicin-eluting beads for unresectable hepatocellular carcinoma: five-year survival analysis. Cardiovasc Intervent Radiol. 2012;35:1119–1128. doi: 10.1007/s00270-012-0394-0. [DOI] [PubMed] [Google Scholar]
- 40.Burrel M, Reig M, Forner A, Barrufet M, de Lope CR, Tremosini S, Ayuso C, Llovet JM, Real MI, Bruix J. Survival of patients with hepatocellular carcinoma treated by transarterial chemoembolisation (TACE) using Drug Eluting Beads. Implications for clinical practice and trial design. J Hepatol. 2012;56:1330–1335. doi: 10.1016/j.jhep.2012.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Recchia F, Passalacqua G, Filauri P, Doddi M, Boscarato P, Candeloro G, Necozione S, Desideri G, Rea S. Chemoembolization of unresectable hepatocellular carcinoma: Decreased toxicity with slow-release doxorubicineluting beads compared with lipiodol. Oncol Rep. 2012;27:1377–1383. doi: 10.3892/or.2012.1651. [DOI] [PubMed] [Google Scholar]
- 42.Facciorusso A, Di Maso M, Muscatiello N. Drug-eluting beads versus conventional chemoembolization for the treatment of unresectable hepatocellular carcinoma: A meta-analysis. Dig Liver Dis. 2016;48:571–577. doi: 10.1016/j.dld.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 43.Malagari K, Chatzimichael K, Alexopoulou E, Kelekis A, Hall B, Dourakis S, Delis S, Gouliamos A, Kelekis D. Transarterial chemoembolization of unresectable hepatocellular carcinoma with drug eluting beads: results of an open-label study of 62 patients. Cardiovasc Intervent Radiol. 2008;31:269–280. doi: 10.1007/s00270-007-9226-z. [DOI] [PubMed] [Google Scholar]
- 44.Guiu B, Deschamps F, Aho S, Munck F, Dromain C, Boige V, Malka D, Leboulleux S, Ducreux M, Schlumberger M, et al. Liver/biliary injuries following chemoembolisation of endocrine tumours and hepatocellular carcinoma: lipiodol vs. drug-eluting beads. J Hepatol. 2012;56:609–617. doi: 10.1016/j.jhep.2011.09.012. [DOI] [PubMed] [Google Scholar]
- 45.Kobayashi S, Nakanuma Y, Matsui O. Intrahepatic peribiliary vascular plexus in various hepatobiliary diseases: a histological survey. Human pathology. 1994;25:940–946. doi: 10.1016/0046-8177(94)90016-7. [DOI] [PubMed] [Google Scholar]
- 46.Chlebowski RT. Adriamycin (doxorubicin) cardiotoxicity: a review. West J Med. 1979;131:364–368. [PMC free article] [PubMed] [Google Scholar]
- 47.Cucchetti A, Trevisani F, Cappelli A, Mosconi C, Renzulli M, Pinna AD, Golfieri R. Cost-effectiveness of doxorubicin-eluting beads versus conventional trans-arterial chemo-embolization for hepatocellular carcinoma. Dig Liver Dis. 2016;48:798–805. doi: 10.1016/j.dld.2016.03.031. [DOI] [PubMed] [Google Scholar]
- 48.Green TJ, Rochon PJ, Chang S, Ray CE, Winston H, Ruef R, Kreidler SM, Glueck DH, Shulman BC, Brown AC, et al. Downstaging disease in patients with hepatocellular carcinoma outside of Milan criteria: strategies using drug-eluting bead chemoembolization. J Vasc Interv Radiol. 2013;24:1613–1622. doi: 10.1016/j.jvir.2013.07.024. [DOI] [PubMed] [Google Scholar]
- 49.Angelico M. TACE vs DEB-TACE: Who wins? Dig Liver Dis. 2016;48:796–797. doi: 10.1016/j.dld.2016.05.009. [DOI] [PubMed] [Google Scholar]


