Abstract
Objectives
The aim of this study was to assess the clinical utility of esophageal manometry among Sudanese patients presenting to the National Centre for Gastrointestinal and Liver Diseases, Ibn Sina Hospital, Khartoum, Sudan.
Methodology
Consecutive patients referred for esophageal manometry at the aforementioned center from July 2008 through January 2011 were included in the study. Manometric studies were done after stopping medicines with a known effect on esophageal motility and an overnight fast. Immediately before the manometric study, the patients’ history and clinical examination were recorded using a structured questionnaire.
Results
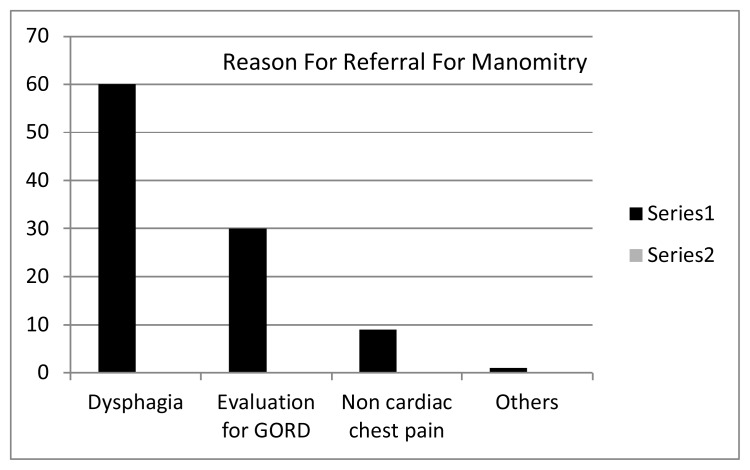
The major referral reason was the investigation of dysphagia in 78 patients (60.5%), followed by the evaluation of Gastroesophageal reflux disease (GERD) in 39 patients (30%), while 11 patients (9%) were referred because of non-cardiac chest pain. The manometric diagnosis in the 78 patients with dysphagia, where 51(65.4%) had achalasia, 13(16.7%) had nonspecific motility disorder, the remaining percentage was formed by GERD diffuse esophageal spasm, connective tissue disease, Nutcracker esophagus, hypertensive lower esophageal sphincter, patient manometry suggestive of myasthenia gravis, and normal manometry.
Conclusion
GERD and Achalasia were the commonest conditions among the study group. Patients presenting with achalasia manifest the same clinical symptoms as published in the literature. The leading abnormality predisposing to GERD was hypotensive lower esophageal sphincter and weak esophageal clearance function. GERD was main cause of non-cardiac chest pain in the study population. However, it is difficult to generalize the findings of this study for the whole country since it was a single center study.
Keywords: esophageal manometry, achalasia, GERD, non-cardiac chest pain, Sudan
Introduction
The regular use of esophageal manometry was delayed for 80 years since it was first performed in 1883, concurrently with the first published esophageal manometry atlas (1,2) Esophageal manometry is a fundamental tool during the assessment of patients with dysphagia, chest pain chest pain, and gastro esophageal reflux (GER). The usual clinical indications of esophageal manometry are: (1) To establish the diagnosis of achalasia after refuting alternative diagnoses by endoscopy or barium radiography; (2) To assist in the placement of intraluminal devices (for example a pH probe) when landmarks are necessary for optimum positioning; (3) To assess patients with symptoms of dysphagia, including those who develop symptoms post-antireflux surgery; and (4) To assess patients suffering chest pain who have been sufficiently treated for GER disease (GERD) and in whom achalasia remains to be a possibility. (3–5) Diagnostic tests has always been of great importance for the matter of making a diagnosis, however common medical practice has become increasingly dependent on them for such purpose, and thus it is highly important that these procedures be critically validated to ensure the quality of evidence they will provide. The following criteria are required in order to consider a test useful: (1) High diagnostic accuracy; (2) application of a specific treatment is supported by such test; (3) it ultimately leads to improvement in the clinical outcome for the patient. (6) Nevertheless, basic standards are seldom met on performing diagnostic studies and manometry is not an exception, carrying the jeopardy of exposing patients to unnecessary complications and risks (e.g sore throat, nasal congestion, inadvertently intubating the trachea, and esophageal rupture in extremely rare cases), (7) along with the increase in healthcare costs, and wasting of resources. Research has knocked out some routinely performed diagnostic tests and proved their poor clinical yield over the last several decades. (8, 9) On the other hand, researchers delineated the clinical usefulness of others such as endoscopic ultrasound and 1-time screening colonoscopies in the appropriate settings. (10, 11) The role of esophageal manometry in clinical practice has recently come under increased scrutiny. A number of researchers have addressed esophageal manometry clinical utility. (12, 13) Moreover, the utility of esophageal manometry in the diagnosis or confirmation of the diagnosis of GER has drawn the attention of investigators, as the case in its utilization in investigating chest pain or other esophageal symptoms as an initial investigatory tool. Furthermore, these studies as well as others question the role of esophageal manometry in the diagnosis and management of diffuse esophageal spasm, connective tissue disorders (ie, scleroderma), and during the preoperative assessment of patients with GER. (4–5) Despite its wide use, the sensitivity and specificity of esophageal manometry in diagnosing disorders with symptoms suggestive of esophageal dysfunction are questionable.(4) Moreover, literature describing clinical utility of esophageal manometry among Sudanese patients referred for such test is lacking. Thus, this study aimed at the assessment of the clinical utility of esophageal manometry among Sudanese patients presenting to the largest gastroenterology center in Sudan.
Materials and Methods
Consecutive patients referred for esophageal manometry at National Centre for Gastrointestinal and Liver Diseases Ibn Sina specialized hospital, Khartoum, Sudan from July 2008 through January 2011 were eligible for inclusion in this study. The National Centre for Gastrointestinal and Liver Diseases Ibn Sina specialized hospital provides tertiary care for Sudanese patients. Patients are referred directly to the motility laboratory at National Centre for Gastrointestinal and Liver Diseases Ibn Sina specialized hospital for esophageal manometry by a variety of providers. Manometric studies were done after stopping medicines with a known effect on esophageal motility and an overnight fast. Immediately before the manometric study, the patients’ history and clinical examination were recorded using a structured questionnaire after obtaining a written consent. The results of any previous workup e.g. endoscopy, radiological, patients’ cardiac evaluation (e.g Chest x ray, electrocardiography) and barium studies were also noted. Patients were examined using 4-Channel Stationary Pull-through catheter, utilizing water perfusion system using pneumo-hydraulic pump with external transducer type (Mui Scientific) and a computer software (Medtronic). The consecutive 5-mL water swallows were obtained separated by at least a 30 breath cycles. Patient with suspected GERD had 24 hours pH monitoring using single sensor pH catheter (zinetic 24 h, Medtronic), intubation was done with the pH sensor positioned 5 cm above the manometrically determined lower esophageal sphincter. At the time of manometry, the patient provided demographic information (age, sex, and race), age at onset of symptoms, prior tests (and results if known) performed for these symptoms. This protocol was approved by the Institutional Review Board of The National Centre for Gastrointestinal and Liver Diseases Ibn Sina specialized hospital.
Statistical Analysis
Data were entered to SPSS version 20; descriptive statistics of the patients’ demographic information including age, sex, BMI were calculated. The frequency distributions of patients’ symptoms were calculated along with the abnormal and normal esophageal manometry test. The mean, median mode and standard deviation for the various groups were obtained.
Results
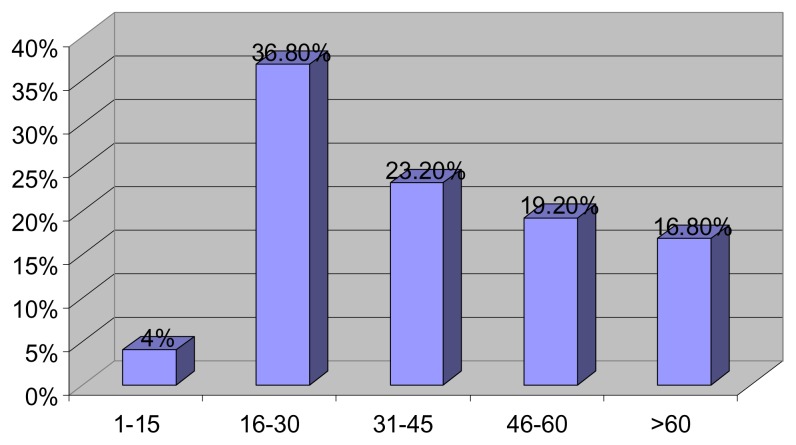
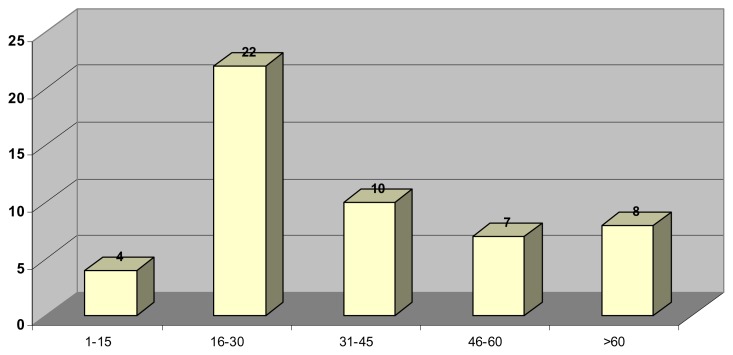
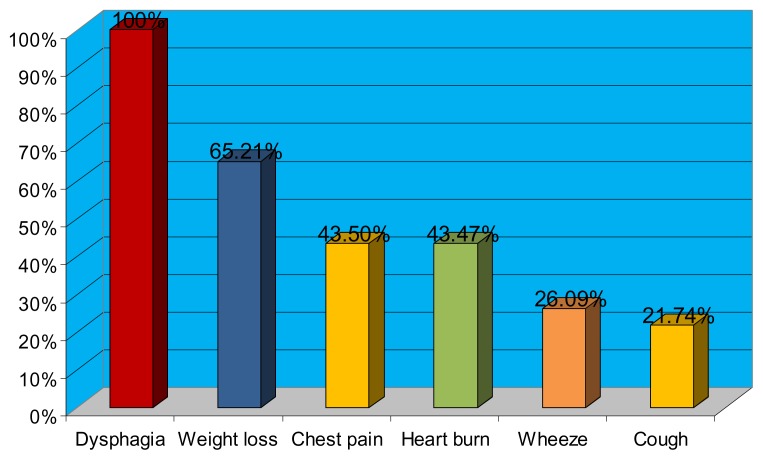
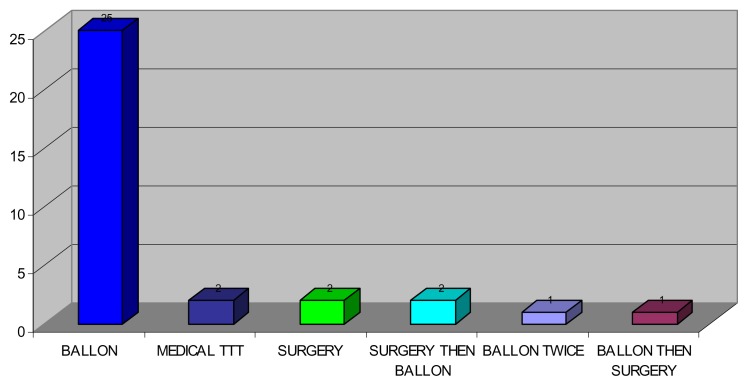
A total of 129 patients were included in the study, 71 (55%) of whom were males while 58 (45%) were females. The mean age of the group was 40.24 (1–77) years. See Fig 1. The leading cause of referral was to investigate dysphagia in 78 patients (60.5%), followed by the evaluation of GERD in 39 patients (30%), while 11 patients (9%) were referred because of non-cardiac chest pain see Fig 2. The manometric diagnosis in the 78 patients with dysphagia, where 51 (65.4%) patients had achalasia, 13 (16.7%) patients had nonspecific motility disorder, 5 (6.4%) patients had motility disorder predisposing to GERD, 2(2.3%) patients were found to have diffuse esophageal spasm, 2 (2.3%) of those studied were diagnosed with connective tissue disease, 1 (1.65%) patient had Nutcracker Oesophgus,1 (1.65%) patient had hypertensive lower esophageal sphincter, 1 (1.65%) patient had manometry suggestive of myasthenia gravis while in 2 (2.3%) patients the manometry study was absolutely normal. The Mean duration of symptoms prior to diagnosis was 28.89 (1–156) months and mean BMI (Range) 21.46 (13.6–35.49). Table (1) showed Pattern of dysphagia in 51 patients with Achalasia, where the majority of patients (82.40%) experienced dysphagia to both solids and liquids, and the majority of patients (70.37%) felt that food sticks to the distal esophagus. The clinical profile of 51 patients with achalasia, showed expectedly that dysphagia was the presenting symptom in all patients, followed by weight loss (82.40%), chest pain (43.5%), heart burn (43.47%). Respiratory symptoms, namely wheeze and cough were the presenting symptoms in (20.09%) and (21.74%) of patients respectively see Fig. 3. Regarding the endoscopic findings in 51 Achalasia patients, the endoscopic findings were consistent with Achalasia in 46 (90.20%) of those assessed while they were normal in 5 (9.80%) patients. The resting lower esophageal sphincter profile could be assessed in 39 achalasia patients as it was not possible to assess the lower esophageal sphincter in 12 patients because the manometry catheter could not be passed across the tightly closed lower esophageal sphincter. The mean resting lower esophageal sphincter pressure was 24.53 mmHg. The resting lower esophageal sphincter pressure was normal in 25 (64.1%) patients, hypertensive in 11 (28.2%) and hypotensive in 3 (7.7%) patients. Regarding the lower esophageal sphincter relaxation in these 39 achalasia patients, the mean lower esophageal sphincter relaxation was 57.52 (20–90%), in one patient the deglutitive lower esophageal sphincter relaxation was normal. In all patients contraction waves in the body of the esophagus were either: Low amplitude, aperistaltic or simultaneous, with exception of one patient who had high amplitude contractions. Fig (4) showed the therapy instituted for the 33 contactable achalasia patients, 25 patients underwent balloon dilatation without complications and they were free of symptoms at the time they were contacted, 2 were on medical treatment, 4 surgery half of which then required balloon dilatation, one patient had balloon dilatation twice and one patient required surgery after one session of balloon dilatation. Evaluation of GERD ranks the second reason for referral, this group included 51 patients of whom 33 (64.7%) were males. The vast majority of GERD patients (74.5%) presented typically i.e. with heart burn and regurgitation. The mean age of GERD patients was 48.83 years with the majority of patients (66.58%) in the age group 45 years and younger. The lower esophageal sphincter resting pressure was low in 31 patients and normal in the remaining 18 patients. 17 patients had both hypotensive lower esophageal sphincter and weak esophageal body contraction. Table (2) 11 patients were referred for the investigation of chest pain for which cardiac cause has been ruled, 7 (63.64%) out of these were males, with a mean age 43.55 years. The prevalence of GERD was high in this group of patients (63.6%). Esophageal motility disorders were responsible for chest pain in the remainder of patients (36.4%). The variants of esophageal motility disorders in non-cardiac chest pain patients were as follows: 2 patients had nonspecific motility disorder, 1 patient had hypertensive lower esophageal sphincter and 1 patient had nutcracker esophagus.
Fig. 1.
Distribution of The Study Population by Age
Fig. 2.
Table 1.
Pattern of dysphagia in 51 patients with Achalasia
| Symptom | Subdivision | Percentage |
|---|---|---|
| Dysphagia | To both solids liquid (%) | (82.40%) |
| To solids (%) | (13.70%) | |
| To liquids (%) | (3.90%) | |
| Sense of food sticking behind | Proximaloesoph | (11.12%) |
| Midoesoph | (18.51%) | |
| Distal oesoph | (70.37%) |
Fig. 3.
Age Groups for Patients with Achalasia
Fig. 4.
Clinical profile of 51 patients with Achalasia
Table 2.
Manometric profile of GERD patients
| Esophageal body contraction | Total | |||
|---|---|---|---|---|
| Normal | Weak | |||
| Resting lower esophageal sphincter Pressure | Low | 14 | 17 | 31 |
| Normal | 9 | 9 | 18 | |
| Total | 23 | 26 | 49 | |
Discussion
The current study is the first to describe the clinical utility of esophageal manometry among Sudanese patients attending the largest gastroenterology Tertiary center in Sudan. The most frequently encountered indications for manometry in the current study were the investigation of dysphagia followed by the evaluation of GERD and the diagnosis of non-cardiac chest pain. The use of manometry in the patients with dysphagia revealed that achalasia was the main underlying etiology (65.38%), followed by non-specific motility disorder (16.67%), GERD (6.41%), connective tissue disorders (2.56%) and diffuse esophageal spasm (2.56%). Manometry was reported as normal in a more or less similar proportion of patients (2.56%). A proportion of about (4%) of those presenting with dysphagia were found to have one of the followings Nutcracker esophagus, hypertensive lower esophageal sphincter and myasthenia gravis. These results differ from what was obtained by Hong et al where in their assessment of patients with esophageal motility abnormality only; ineffective esophageal motility formed the highest prevalence (49%) finding, followed by hypertensive lower esophageal sphincter (22%), classic achalasia (9%), non-specific esophageal motility disorder (7%), nutcracker esophagus (6%), diffuse esophageal spasm (4%) and finally atypical disorder of lower esophageal sphincter relaxation (4%).(14) Our data agrees with Hong et al findings as the majority of the patients (72.55%) were in the age group 45 years or younger, with a mean age at time of diagnosis was 37.45 years. The mean duration of symptoms prior to diagnosis was 28.89 (1–156) months, this contrasts with several previous studies in which the mean duration of symptoms before diagnosis was reported to fall between 4.5 and 7.6 years. (15, 16) Looking at the clinical presentation of achalasia patients it revealed that our population manifest the same clinical profile published in the literature, with the most common presenting complaints at the time of diagnosis being: Dysphagia (82–100% of patients), postprandial and/or nocturnal reflux (59–64%), weight loss(30–91%), chest pain (17–95%)and cough (11–46%). (17, 18) It is well known that Achalasia patients may experience gastro esophageal reflux symptoms and even develop esophagitis. Smart et al suggested that acid exposure time is abnormally prolonged in some achalasia patients and generates lactic acid from retained remnants within the esophagus. (19) In the aforementioned study heart burn and chest pain formed the main presenting symptoms in (43.47%) and (43.5%), respectively, such findings reflects the high prevalence GERD symptoms in these patients. Normal upper endoscopic findings were reported in a study of newly diagnosed patients with achalasia in a percentage of patients approaching 44%. (20) A barium swallow is empowered as a diagnostic tool to suggest the diagnosis of achalasia with appreciable degree of precision as suggested by Grimes et al, (17) particularly upon the existence of the combination of a dilated esophagus, retained food and barium and a smooth, gradually the constriction of the gastroesophageal junction is lessened. However, in the same study aforementioned, barium study failure to suggest the diagnosis of achalasia reached more than 64%. (17) Manometry is the test with the highest sensitivity in diagnosing achalasia. (21, 22) The diagnostic manometric hallmarks of achalasia are a peristalsis of the distal esophagus and defective or absent lower esophageal sphincter relaxation. Further supportive characteristics encompass a hypertensive lower esophageal sphincter and low amplitude contractions of the esophageal body. (21) In this series the endoscopic findings were consistent with Achalasia in 46 (90.20%) of our patients while it was normal in 5(9.80%) patients. This data supports the valuable role of manometry in the diagnosis of achalasia because in such early disease course there may be minimal dilatation of the esophagus, resulting in minimal or undetectable radiographic or endoscopic abnormalities. Our study group data in regard to contraction waves in the body of the esophagus were either: Low amplitude, a peristaltic or Simultaneous, with exception of one patient with high amplitude contractions. Domination of the manometric picture of high-amplitude esophageal body contractions may be the case a thing that fits the classification of vigorous achalasia generally defined by simultaneous esophageal contractions with amplitudes 37–60mm Hg. (23) Progression from diffuse esophageal spasm to achalasia has been described in few reports in the literature, with the subsequent emergence of a non-relaxing lower esophageal sphincter on manometry. (24) Putting in consideration the close resemblance between diffuse esophageal spasm and achalasia, such conditions also consolidate the concept that vigorous achalasia may in fact be an early stage of classic achalasia. In addition to esophageal body peristalsis, the other major criterion for the manometric diagnosis of achalasia relies on the demonstration of failure of relaxation of the lower esophageal sphincter. Among the subjects we have studied the mean lower esophageal sphincter relaxation was 57.52 Range (20–90%). Apparently complete lower esophageal sphincter relaxation occurs in about 15–30% of those diagnosed with achalasia and has been the subject of some controversy in the medical literature. (25, 26) In the current study the aforementioned statement was demonstrated in one patient where the deglutitive lower esophageal sphincter relaxation was found to be normal. An apparently normal lower esophageal sphincter relaxation in these rare patients could be explained by the likelihood that it might represents an artifact resulting from movement of the manometry catheter recording site in and out of the lower esophageal sphincter during the swallow cycle, nevertheless, such potential source of error is eliminated by resorting to a sleeve sensor for the purpose of measurement of the lower esophageal sphincter pressure. The sleeve sensor allows a more reliable recording of lower esophageal sphincter pressure over time and has been reported to be of particular use in the evaluation of achalasia. (27) The mean resting lower esophageal sphincter pressure for our patients was 24.53, the resting lower esophageal sphincter pressure was normal in 25 (64.1%) patients, hypertensive in 11 (28.2%) and hypotensive in 3 (7.7%) patients.
The management goal in achalasia is to reduce distal esophageal obstruction. Such an aim can be accomplished via variety of ways such as, medicines, instrumentation, or surgery. Calcium channel antagonists and nitrates have low efficiency, moreover the short lasting effect of the endoscopically injected botulinum toxin is well known. (28, 29) In addition to that, a number of studies have clearly shown that pneumatic balloon dilations are more effective than botulinum toxin injections. (30–32) pneumatic dilatation is regarded as the first-line of esophageal management in esophageal achalasia since remission can be achieved in the majority of patients. (33) Surgical cardiomyotomy should be resorted to in patients who fail to respond to instrumental therapy. (34) However, the long term efficacy of such a therapeutic strategy remains to be a matter of controversy since it has been reported that long-term remission rates after pneumatic dilatation drops as time passes, from 20 to 50% at 10 years, (35–37) especially in young subjects. (37) Out of the 51 achalasia patients, 33 patients could be contacted and questioned with regards to the therapy they received: 25 patients underwent balloon dilatation without complications and were free of symptoms at the time, 2 were on medical treatment, 2 had surgery, 2 had surgery then required balloon dilatation, one patient had balloon dilatation twice and one patient required surgery after one session of balloon dilatation. This data further substantiate the valuable role of balloon dilatation in the therapy of achalasia.
Esophageal manometry has valuable role in the management of GERD patients, specifically: it assesses the lower esophageal sphincter pressure, location and relaxation, assist the placement of 24 hr. pH catheter, Assess the esophageal peristalsis also it is very much crucial Prior to anti-reflux surgery. In our GERD population the lower esophageal sphincter resting pressure was low in 31 patents and normal in the remainder 18 patients. 17 patients had both hypotensive lower esophageal sphincter and weak esophageal body contraction Table (3). A proportion that approaches one third of patients referred to cardiology clinics, or even admitted to coronary care units appear to be free of ischemic heart disease. (38) In the United States ‘non-cardiac chest pain’ has surpassed all other causes of referral for esophageal manometry, while among our study population this indication constituted 11% of the referral. Some population-based studies have found undiagnosed chest pain to be associated with GERD and the prevalence of GERD among those with such chest pain is estimated to be greater than 50% (39) Our data consolidate the high prevalence of GERD among non-cardiac chest pain patients (63.6%), there by suggesting that appropriate time and cost saving strategy for managing these patients would be a trial of proton pump inhibitors before subjecting the patient to further investigations. The different types of esophageal motility disorders can be seen in patients presenting with chest pain syndromes. (40) In the current study esophageal motility disorder were found to be responsible for chest pain in (36.4%) of patients as (18.18%) patients had non-specific motility disorder, (9.09%) patient had hypertensive lower esophageal sphincter and (9.09%) patient had nutcracker esophagus. These results contrasts with what was obtained by Richter who studied esophageal manometry in non-cardiac chest pain patients and reported normal manometry in the majority of patients (75%), Nutcracker esophagus (12%), Non-specific esophageal motility disorder (9%). Achalasia, Hypertensive lower esophageal sphincter and diffuse esophageal spasm were responsible for chest pain in a small proportion of patients (4%). (41)
Conclusion
GERD and Achalasia were the commonest conditions encountered among Sudanese patients referred for Motility studies. Patients presenting with the latter condition manifest the same clinical symptoms as published in the literature. The most important motility abnormality predisposing to GERD was found to be hypotensive lower esophageal sphincter and weak esophageal clearance function. GERD was found to be an important cause of non-cardiac chest pain in the study population. However, the findings of this study cannot be generalized for the whole country since they have been carried on a single center.
Fig. 5.
Therapy for Achalasia Patients
References
- 1.Kronecker H, Meltzer SJ. Der schluckmechanismus, seineerregung und seine hemmung. Arch GesAnatPhysiol (suppl) 1983;7:328–332. [Google Scholar]
- 2.Code CF, Creamer B, Schegel JF. An Atlas of Esophageal Motility in Health and Disease. Proc R Soc Med. 1959;52:221. [Google Scholar]
- 3.Bredenoord AJ, Smout AJ. High-resolution manometry of the esophagus: more than a colorful view on esophageal motility? Expert Rev Gastroenterol Hepatol. 2007;1:61–9. doi: 10.1586/17474124.1.1.61. [DOI] [PubMed] [Google Scholar]
- 4.Ergun GA, Kahrilas PJ. Clinical applications of esophageal manometry and pH monitoring. Am J Gastroenterol. 1996;91:1077–1089. [PubMed] [Google Scholar]
- 5.Pandolfino JE, Kahrilas PJ. AGA technical review: clinical use of esophageal manometry. Gastroenterology. 2005;128:209–224. doi: 10.1053/j.gastro.2004.11.008. [DOI] [PubMed] [Google Scholar]
- 6.Sackett DL, Haynes RB, Guyatt GH. A Basic Science for Clinical Medicine. 2nd ed. Boston: Little Brown and Company; 1991. Clinical Epidemiology; pp. 51–68. [Google Scholar]
- 7.Walamies MA. Perception of esophageal manometry. Dis Esophagus. 2002;15:46–9. doi: 10.1046/j.1442-2050.2002.00224.x. [DOI] [PubMed] [Google Scholar]
- 8.Wians FH. Clinical laboratory tests: which, why, and what do the results mean? Lab Med. 2009;40:105–113. [Google Scholar]
- 9.Cancrini G, Iori A. [Traditional and innovative diagnostic tools: when and why they should be applied]. Parassitologia. 2004;46:173–6. [PubMed] [Google Scholar]
- 10.Nickl NJ, Bhutani MS, Catalano M, Hoffman B, Hawes R, Chak A, Roubein LD, et al. Clinical implications of endoscopic ultrasound: the American endosonography club study. GastrointestEndosc. 1996;44:371–378. doi: 10.1016/s0016-5107(96)70083-4. [DOI] [PubMed] [Google Scholar]
- 11.Ness RM, Holmes AM, Klein R, Dittus R. Cost-utility of one-time colonoscopic screening for colorectal cancer at various ages. Am J Gastroenterol. 2000;95:1800–1811. doi: 10.1111/j.1572-0241.2000.02172.x. [DOI] [PubMed] [Google Scholar]
- 12.Lacy BE, Paquette L, Robertson DJ, Kelley ML, Jr, Weiss JE. The clinical utility of esophageal manometry. J Clin Gastroenterol. 2009;43:809–15. doi: 10.1097/MCG.0b013e31818ddbd5. [DOI] [PubMed] [Google Scholar]
- 13.Nayar DS, Khandwala F, Achkar E, Shay SS, Richter JE, Falk GW, et al. Esophageal manometry: assessment of interpreter consistency. Clin Gastroenterol Hepatol. 2005;3:218–224. doi: 10.1016/s1542-3565(04)00617-2. [DOI] [PubMed] [Google Scholar]
- 14.Hong SN, Rhee PL, Kim JH, Lee JH, Kim YH, Kim JJ, et al. Does this patient have oesophageal motility abnormality or pathological acid reflux? Dig Liver Dis. 2005;37:475–84. doi: 10.1016/j.dld.2005.01.018. [DOI] [PubMed] [Google Scholar]
- 15.Craft RO, Aguilar BE, Flahive C, Merritt MV, Chapital AB, Schlinkert RT, et al. Outcomes of minimally invasive myotomy for the treatment of achalasia in the elderly. JSLS. 2010;14:342–7. doi: 10.4293/108680810X12924466007368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gockel I, Gith A, Drescher D, Jungmann F, Eckhard L, Lang H. Minimally invasive surgery for achalasia in patients >40 years: more favorable than anticipated. Langenbecks Arch Surg. 2012;397:69–74. doi: 10.1007/s00423-011-0832-5. [DOI] [PubMed] [Google Scholar]
- 17.Grimes OF, Stephens HB, Margulis AR. Achalasia of the esophagus. Am J Surg. 1970;120:198–20. doi: 10.1016/s0002-9610(70)80111-8. [DOI] [PubMed] [Google Scholar]
- 18.Bjelović M, Špica B, Gunjić D, Grujić D, Skrobić O, Babič T, et al. Laparoscopic myotomy in achalasia cardia treatment: experience after 36 operations. SrpArhCelokLek. 2013;141:475–81. [PubMed] [Google Scholar]
- 19.Smart HL, Foster PN, Evans DF, Slevin B, Atkinson M. Twenty four hour oesophageal acidity in achalasia before and after pneumatic dilatation. Gut. 1987;28:883–7. doi: 10.1136/gut.28.7.883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Howard PJ, Maher L, Pryde A, Cameron EW, Heading RC. Five year prospective study of the incidence, clinical features, and diagnosis of achalasia in Edinburgh. Gut. 1992;33:1011–5. doi: 10.1136/gut.33.8.1011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Todorczuk JR, Aliperti G, Staiano A, Clouse RE. Reevaluation of manometric criteria for vigorous achalasia. Is this a distinct clinical disorder? Dig Dis Sci. 1991;36:274–278. doi: 10.1007/BF01318195. [DOI] [PubMed] [Google Scholar]
- 22.Kumar P, Clark M. Clinical Medicine. 4th ed. Edinburgh: WB Saunders; 1998. pp. 229–231. [Google Scholar]
- 23.Mearin F, Malagelada JR. Complete lower esophageal sphincter relaxation observed in some achalasia patients is functionally inadequate. Am J Physiol Gastrointest Liver Physiol. 2000;278:G376–G383. doi: 10.1152/ajpgi.2000.278.3.G376. [DOI] [PubMed] [Google Scholar]
- 24.Fontes LH, Herbella FA, Rodriguez TN, Trivino T, Farah JF. Progression of diffuse esophageal spasm to achalasia: incidence and predictive factors. Dis Esophagus. 2013;26:470–4. doi: 10.1111/j.1442-2050.2012.01377.x. [DOI] [PubMed] [Google Scholar]
- 25.Shi G, Ergun GA, Manka M, Kahrilas PJ. Lower esophageal sphincter relaxation characteristics using a sleeve sensor in clinical manometry. Am J Gastroenterol. 1998;93:2373–2379. doi: 10.1111/j.1572-0241.1998.00690.x. [DOI] [PubMed] [Google Scholar]
- 26.Amaravadi R, Levine MS, Rubesin SE, Laufer I, Redfern RO, Katzka DA. Achalasia with complete relaxation of lower esophageal sphincter: radiographic-manometric correlation. Radiology. 2005;235:886–91. doi: 10.1148/radiol.2353040732. [DOI] [PubMed] [Google Scholar]
- 27.Shi G, Ergun GA, Manka M, Kahrilas PJ. Lower esophageal sphincter relaxation characteristics using a sleeve sensor in clinical manometry. Am J Gastroenterol. 1998;93:2373–2379. doi: 10.1111/j.1572-0241.1998.00690.x. [DOI] [PubMed] [Google Scholar]
- 28.Dughera L, Chiaverina M, Cacciotella L, Cisarò F. Management of achalasia. Clin Exp Gastroenterol. 2011;4:33–41. doi: 10.2147/CEG.S11593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Moawad FJ, Wong RKh. Modern management of achalasia. Curr Opin Gastroenterol. 2010;26:384–8. doi: 10.1097/MOG.0b013e32833aaf4a. [DOI] [PubMed] [Google Scholar]
- 30.Chuah S-K, Wu K-L, Hu T-H, Tai W-C, Changchien C-S. Endoscope-guided pneumatic dilation for treatment of esophageal achalasia. World Journal of Gastroenterology : WJG. 2010;16:411–417. doi: 10.3748/wjg.v16.i4.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Leyden JE, Moss AC, MacMathuna P. Endoscopic pneumatic dilation versus botulinum toxin injection in the management of primary achalasia. Cochrane Database Syst Rev. 2014;12:CD005046. doi: 10.1002/14651858.CD005046.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kumar, Schnoll-Sussman Anand R, Katz Felice H, Philip O. Botulinum toxin and pneumatic dilation in the treatment of achalasia. Techniques in Gastrointestinal Endoscopy. 2014;1:10–19. [Google Scholar]
- 33.Vaezi MF, Richter JE. Current therapies for achalasia:comparison and efficacy. J Clin Gastroenterol. 1998;27:21–35. doi: 10.1097/00004836-199807000-00006. [DOI] [PubMed] [Google Scholar]
- 34.Vantrappen G, Janssens J. To dilate or to operate? That is the question. Gut. 1983;24:1013–9. doi: 10.1136/gut.24.11.1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Karamanolis G, Sgouros S, Karatzias G, Papadopoulou E, Vasiliadis K, Stefanidis G, Mantides A. Long-term outcome of pneumatic dilation in the treatment of achalasia. Am J Gastroenterol. 2005;100:270–4. doi: 10.1111/j.1572-0241.2005.40093.x. [DOI] [PubMed] [Google Scholar]
- 36.Yaghoobi M, Mayrand S, Martel M, Roshan-Afshar I, Bijarchi R, Barkun A. Laparoscopic Heller’s myotomy versus pneumatic dilation in the treatment of idiopathic achalasia: a meta-analysis of randomized, controlled trials. Gastrointest Endosc. 2013;78:468–75. doi: 10.1016/j.gie.2013.03.1335. [DOI] [PubMed] [Google Scholar]
- 37.Eckardt VF, Gockel I, Bernhard G. Pneumatic dilation for achalasia: Late results of a prospective follow up investigation. Gut. 2004;53:629–33. doi: 10.1136/gut.2003.029298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wilcox RG, Roland JM, Hampton JR. Prognosis of patients with ‘chest pain? cause’. Br Med J. 1981;282:431–3. doi: 10.1136/bmj.282.6262.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Eslick GD, Jones MP, Talley NJ. Non-cardiac chest pain: prevalence, risk factors, impact and consulting—a population-based study. Aliment Pharmacol Ther. 2003;17:1115–24. doi: 10.1046/j.1365-2036.2003.01557.x. [DOI] [PubMed] [Google Scholar]
- 40.Ferguson CS, Hodges K, Hersh T, Jinich H. Oesophageal manometry in patients with chest pain and normal coronary arteriography. Am J Gastroenterol. 1981;75:124–7. [PubMed] [Google Scholar]
- 41.Richter JE. Esophageal testing of patients with noncardiac chest pain or dysphagia. Results of three years’ experience with 1161 patients. Ann Int Med. 1987;3 doi: 10.7326/0003-4819-106-4-593. [DOI] [PubMed] [Google Scholar]