Abstract
Background
Readmission or death following heart failure (HF) hospitalization is a consequential and closely scrutinized outcome, but risk factors may vary by population. We characterized the risk factors for post-discharge readmission/death in subjects treated for acute heart failure (AHF).
Methods and Results
A post hoc analysis was performed on data from 744 subjects enrolled in three AHF trials conducted within the Heart Failure Network (HFN): Diuretic Optimization Strategies Evaluation in Acute Heart Failure (DOSE-AHF), CARdiorenal REScue Study in Acute Decompensated Heart Failure (CARRESS-HF), and Renal Optimization Strategies Evaluation in Acute Heart Failure (ROSE-AHF). All-cause readmission/death occurred in 26% and in 38% of subjects within 30- and 60-days of discharge, respectively. Non-HF cardiovascular causes of readmission were more common in the ≤ 30 day timeframe vs. the 31-60 day timeframe (23% vs. 10%, p=0.016). In a Cox proportional hazards model adjusting a priori for LVEF < 50% and trial, the risk factors for all-cause readmission/death included: elevated baseline BUN, ACEI/ARB non-use, lower baseline sodium, non-white race, elevated baseline bicarbonate, lower SBP at discharge/day 7, depression, increased length of stay, and male sex.
Conclusions
In an AHF population with prominent congestion and prevalent renal dysfunction, early readmissions were more likely to be due to non-HF cardiovascular causes compared to later readmissions. The association between use of ACEI/ARB and lower all-cause readmission/death in Cox proportional hazards model suggests a role for these drugs to improve post-discharge outcomes in AHF.
Keywords: Cardiorenal, ACE inhibitor, RAAS
INTRODUCTION
Each year in the US, heart failure (HF) is the primary admission diagnosis for > 1 million hospitalizations and the secondary diagnosis in > 3 million hospitalizations,1 resulting in aggregate costs in excess of $10 billion.2 While inpatient mortality rates for HF hospitalization are modest and stable at ~ 3%,3 hospital readmission within 30 days of discharge is common – occurring in approximately 25% of admissions.4 For Medicare, where HF represents the most common hospital admission or readmission diagnosis, the cost of HF readmission alone is > $1.7 billion/year.5 This has led the Centers for Medicaid and Medicare Services (CMS) to attempt to mitigate costs by financially penalizing hospitals with readmission rates exceeding a model-derived risk standardized readmission rate, which has in turn focused efforts on reducing the 30-day readmission rates for those aged > 65 years.
While focus on the elderly Medicare population is timely, the scope of the issue extends beyond this group. In addition to financial consequences, HF readmission identifies patients at high risk for adverse events and mortality.6 Whether risks identified in the elderly Medicare population apply to other age and payer groups is not known. Meanwhile, the proportion of HF hospitalizations of patients under 65 years of age increased from 23% in 2000 to 29% in 2010,7 indicating a need to study readmission/death in younger patients as well. Readmission rates also differ by academic vs. non-academic hospitals. While academic hospitals may deliver higher quality of HF care in some instances,8 they may also deliver care to patients who are more likely to be younger, urban, and without prior hospitalization for HF.9 Academic hospitals also have higher risk-standardized rates of readmission.10 Thus, the purpose of this study was to identify risk factors for readmission/death in academic hospitals participating in the Heart Failure Network (HFN).
The Heart Failure Network is a National Heart, Lung, and Blood Institute (NHLBI)-sponsored research consortium established with the purpose of conducting multicenter clinical trials largely in academic hospitals across the US. We characterized the timing, causes, and risk factors for readmission in a cohort derived from three trials of acute decongestive therapy: Diuretic Optimization Strategies Evaluation in Acute Heart Failure (DOSE-AHF); CARdiorenal REScue Study in Acute Decompensated Heart Failure (CARRESS-HF); and Renal Optimization Strategies Evaluation in Acute Heart Failure (ROSE-AHF). While these trials each showed no statistically significant difference between the treatment strategy and the primary clinical outcomes, the aggregate population of the trials provides a large cohort of patients with significant congestion and renal dysfunction, detailed clinical phenotyping, and post-discharge follow-up subsequent to treatment.
METHODS
Study population
This retrospective analysis was performed using data from DOSE-AHF, CARRESS-HF and ROSE-AHF. The design and primary results of these trials have been published previously.11–16 All three trials enrolled patients hospitalized with AHF and included subjects with either preserved or reduced left ventricular ejection fraction (LVEF). AHF was defined as the presence of at least 1 symptom (dyspnea, orthopnea, or edema) AND 1 sign (rales on auscultation, peripheral edema, ascites, pulmonary vascular congestion on chest radiography) of HF in DOSE-AHF and ROSE-AHF or admission to the hospital with a primary diagnosis of HF in CARRESS-HF. DOSE-AHF and CARRESS-HF did not require a prior diagnosis of HF. Subjects were excluded if on renal replacement therapy, for elevated serum creatinine (>3.0mg/dL in DOSE-AHF, >3.5 mg/dL in CARRESS-HF), and for reduced estimated glomerular filtration rate (eGFR) < 15 mL/min/1.73 m2 in ROSE-AHF. The DOSE-AHF trial randomized 308 AHF subjects taking a baseline dose ≥ 80mg oral furosemide (or equivalent) in a 2×2 factorial fashion to intravenous high dose (2.5 times baseline oral dose) vs. low dose (equal to baseline oral dose) furosemide administered by continuous infusion vs. intermittent bolus (every 12 hours).11,12 The CARRESS-HF trial enrolled 188 patients with AHF and worsening renal function (increase in serum creatinine of ≥ 0.3 mg/dL within 12 weeks before or 10 days after admission) with persistent congestion despite intravenous diuretics or escalating doses of oral diuretics. Subjects were randomized in a 1:1 fashion to a stepped pharmacologic diuretic approach vs. ultrafiltration.13,14 The ROSE-AHF study randomized 360 patients with AHF and renal dysfunction to 1 of 3 treatment arms: low-dose nesiritide (0.005 μg/kg/min for 72 hours), low-dose dopamine (2 μg/kg/min for 72 hours), or placebo with all patients receiving intravenous loop diuretics during treatment.15–17 All study participants provided written informed consent. The studies were designed by the NHLBI-sponsored Heart Failure Clinical Research Network investigators, and were approved by protocol review and data safety monitoring committees as well as each participating site's institutional review board.
Among the three trials, 856 subjects were enrolled, however the 21 subjects enrolled in more than one trial were only analyzed for the first trial they entered, leaving a total of 835 subjects for analysis. Analysis of readmission further excluded 91 subjects: 15 died in-hospital, 2 were still hospitalized at 60 days, 6 withdrew consent in-hospital, and 68 were not followed for rehospitalization endpoints beyond discharge. A cohort of 744 subjects was available for analysis of post-discharge readmission/death.
Laboratory Values
Plasma samples from patients enrolled in DOSE-AHF, CARRESS-HF, and ROSE-AHF were collected at randomization (baseline) and at 72 hours (DOSE-AHF, ROSE-AHF) and 96 hours (CARRESS-HF). Serum chemistries were analyzed at each enrolling institution's laboratory blinded to treatment assignment. Biomarkers (including NT-proBNP, serum aldosterone, cystatin C, uric acid, and plasma renin activity [PRA]) were analyzed at The Heart Failure Clinical Research Network Core Biomarker Laboratory at the University of Vermont.
Definitions and Outcomes
Trial protocols dictated the assessment of clinical and laboratory variables at randomization (baseline) and discharge or day 7, whichever occurred first. Medications at admission and discharge were documented. Edema was categorized as absent, mild (1+), moderate (2+), or severe (3+). Based upon previous work, an “orthoedema score” was derived as a sum of edema (valued at 0 points for absent, 1 point for mild, and 2 points for moderate or greater) and orthopnea (valued at 2 points for ≥2 pillows, otherwise 0 points).18 The orthoedema score could range from 0 to 4. Depression was defined as treated by prescription medications.
The main clinical outcome of interest was re-hospitalization or death following discharge from the index hospitalization analyzed in a continuous fashion or in the intervals of 0-30 days or 31-60 days. Primary causes for hospitalization were previously determined by investigators based upon review of hospital charts and assigned to one of nineteen different diagnoses, which were identical across the three trials.. For the present analysis, these were grouped into: HF, cardiovascular non-HF (including angina, myocardial infarction, chest pain, atrial arrhythmia, ventricular arrhythmia, sudden cardiac death, cerebrovascular accident, syncope, hypotension, peripheral vascular disease, elective cardiovascular, and other cardiovascular), renal (acute renal failure, worsening renal function, hyperkalemia), and non-cardiovascular non-renal (infections, elective non-cardiac procedure, and other non-cardiovascular).
Statistical Analyses
Baseline and discharge/day 7 characteristics were compared between patients according to the timing of readmission using the Kruskal-Wallis test for non-normally distributed continuous variables, oneway ANOVA for normally distributed continuous variables, and the Pearson chi-square test for categorical variables. Causes of re-admission were compared between those readmitted within 30 days and those readmitted 31-60 days after discharge using Pearson Chi-square test and Fisher's exact test. The continuous hazard of death or all-cause hospitalization was examined by multivariable Cox proportional hazards analysis using baseline and discharge/day7 variables as inputs and a stepwise selection process. The analysis was adjusted a priori for left ventricular ejection fraction (EF) and clinical trial due to the potential impact of these factors on pharmacotherapy choices. Multiple imputation was used for missing values. Variables with > 15% missingness were not included in the models. For medications, a four-level variable was created to account for use of medication at admission, at discharge, neither, and both. Both baseline and discharge values for a given variable were candidates in the model. For collinear variables of renal function, blood urea nitrogen (BUN) was selected over GFR and Cr for its performance in other published models predicting mortality.19,20 The SAS procedure PROC MI was used to create 25 imputed datasets of size 835. Proportional hazards regression models with variable selection were run in each imputed dataset. The number of times each variable was selected using forward or backward selection and alpha 0.1 or 0.05 for retention were tabulated. Variables selected in at least 20 (80%) of models according to forward or backward selection at alpha=0.1 level were candidates for the final Cox model. PROC MIANALYZE was used in conjunction with SAS PROC PHREG to combine estimates across imputed datasets. Variables were manually removed until all remaining variables had p-value from MIANALYZE that was less than 0.050. Harrell's C index for discrimination in survival data was calculated in each imputed dataset and averaged. The relationship between baseline or discharge/day7 variables and cardiovascular hospitalization was examined in similar fashion to that described for all-cause hospitalization. A two-sided p value < 0.05 was considered significant for all analyses. All statistical analyses were performed with SAS software, version 9.2 (SAS Institute, Cary, North Carolina).
RESULTS
Baseline patient characteristics
Baseline and discharge characteristics are shown in TABLE 1 and TABLE 2. The median age was 69 years with 36% of subjects younger than 65 years of age, 75% males, and 25% non-white race. LVEF was less than 50% in 70% of subjects. Only 6 subjects (0.8%) had a recent (≤ 2 weeks) diagnosis of heart failure and the median duration of heart failure was 4 years. Renal dysfunction (median creatinine = 1.7 mg/dL; median estimated GFR = 41.3 mL/min/1.73m2) and anemia (median hemoglobin = 11.5 g/dL) were prevalent. On admission, 83% of subjects were taking beta blockers, 54% were taking angiotensin converting enzyme inhibitors or angiotensin receptor blockers (ACEI/ARB), and 28% were taking aldosterone antagonists. Subjects were congested, with orthopnea and elevated jugular venous pulse ≥ 8 cm H20 present in more than 90% of subjects.
Table 1.
Baseline Characteristics by Trial
| Characteristic | Overall (N=744) | DOSE (N=260) | CARRESS (N=147) | ROSE (N=337) |
|---|---|---|---|---|
| Demographics | ||||
| Age, years | 69 (60 - 78) | 68 (56 - 77) | 68 (58 - 78) | 70 (62 - 79) |
| Age <65 years | 271 (36.4) | 105 (40.4) | 65 (44.2) | 101 (30.0) |
| Male sex | 559 (75.1) | 196 (75.4) | 113 (76.9) | 250 (74.2) |
| White race | 554 (74.5) | 187 (71.9) | 109 (74.1) | 258 (76.6) |
| Clinical History | ||||
| Ejection fraction, % | 30 (20 - 53) | 28.5 (20.0 - 50.0) | 33 (20 - 55) | 33.5 (21.5 - 53.0) |
| Ejection fraction <50% | 516/737 (70.0) | 190/254 (74.8) | 96 (65.3) | 230/336 (68.5) |
| Hospitalization for HF in last year | 524/736 (71.2) | 192/257 (74.7) | 108/144 (75.0) | 224/335 (66.9) |
| Ischemia as cause of HF | 438 (58.9) | 150 (57.7) | 90 (61.2) | 198 (58.8) |
| Hypertension | 611 (82.1) | 207 (79.6) | 126 (85.7) | 278 (82.5) |
| Atrial fibrillation / flutter | 412 (55.4) | 136 (52.3) | 75 (51.0) | 201 (59.6) |
| Diabetes | 419 (56.3) | 135 (51.9) | 98 (66.7) | 186 (55.2) |
| ICD | 294 (39.5) | 106 (40.8) | 41 (27.9) | 147 (43.6) |
| TIA | 57 (7.7) | 25 (9.6) | 9 (6.1) | 23 (6.8) |
| Stroke | 80 (10.8) | 33 (12.7) | 19 (12.9) | 28 (8.3) |
| Hyperlipidemia | 556 (74.7) | 182 (70.0) | 113 (76.9) | 261 (77.4) |
| Peripheral vascular disease | 114 (15.3) | 39 (15.0) | 28 (19.0) | 47 (13.9) |
| COPD | 201 (27.0) | 63 (24.2) | 48 (32.7) | 90 (26.7) |
| Arrhythmia | 451 (60.6) | 149 (57.3) | 83 (56.5) | 219 (65.0) |
| Current smoking / quit <6m ago | 117 (15.7) | 45 (17.3) | 20 (13.6) | 52 (15.4) |
| Chronic alcohol use | 58 (7.8) | 24 (9.2) | 4 (2.7) | 30 (8.9) |
| Gout | 206 (27.7) | 60 (23.1) | 33 (22.4) | 113 (33.5) |
| Hepatic disease | 33 (4.4) | 7 (2.7) | 8 (5.4) | 18 (5.3) |
| Malignancy | 50 (6.7) | 19 (7.3) | 11 (7.5) | 20 (5.9) |
| Depression | 163 (21.9) | 51 (19.6) | 38 (25.9) | 74 (22.0) |
| NYHA classification | ||||
| I | 1/695 (0.1) | 1/234 (0.4) | 0/139 (0) | 0/322 (0) |
| II | 27/695 (3.9) | 11/234 (4.7) | 0/139 (0) | 16/322 (5.0) |
| III | 442/695 (63.6) | 146/234 (62.4) | 83/139 (59.7) | 213/322 (66.1) |
| IV | 225/695 (32.4) | 76/234 (32.5) | 56/139 (40.3) | 93/322 (28.9) |
| Medications | ||||
| ACE inhibitor or ARB | 400 (53.8) | 169 (65.0) | 64 (43.5) | 167 (49.6) |
| Hydralazine | 136 (18.3) | 44 (16.9) | 28 (19.0) | 64 (19.0) |
| Nitrates | 208 (28.0) | 76 (29.2) | 52 (35.4) | 80 (23.7) |
| Beta-blocker | 617 (82.9) | 218 (83.8) | 117 (79.6) | 282 (83.7) |
| Aldosterone antagonist | 208 (28.0) | 76 (29.2) | 35 (23.8) | 97 (28.8) |
| Digoxin | 187 (25.1) | 78 (30.0) | 25 (17.0) | 84 (24.9) |
| Aspirin | 497 (66.8) | 176 (67.7) | 105 (71.4) | 216 (64.1) |
| Warfarin | 320 (43.0) | 120 (46.2) | 46 (31.3) | 154 (45.7) |
| HF Clinical Assessment | ||||
| Body mass index, kg/m2 | 31.3 (26.8 - 37.7) | 31.1 (26.4 - 36.4) | 33.1 (27.3 - 41.2) | 30.6 (26.5 - 37.1) |
| Weight, lbs | 203 (173 - 251) | 203 (175 - 241) | 220 (183 - 288) | 198 (170 - 244) |
| Systolic blood pressure, mmHg | 114 (104 - 127) | 114 (104 - 130) | 115 (105 - 125) | 114 (103 - 127) |
| Diastolic blood pressure, mmHg | 66 (59 - 74) | 68 (59 - 78) | 64 (56 - 74) | 65 (59 - 72) |
| Heart rate, beats/min | 75 (66 - 84) | 76 (69 - 84) | 75 (65 - 86) | 73 (65 - 84) |
| Edema ≥ 2+ | 572/742 (77.1) | 203 (78.1) | 132 (89.8) | 237/335 (70.7) |
| JVP ≥ 8 cm water | 668/709 (94.2) | 225/247 (91.1) | 136/141 (96.5) | 307/321 (95.6) |
| Orthopnea | 645/712 (90.6) | 225/250 (90.0) | 132/140 (94.3) | 288/322 (89.4) |
| Rales | 408/740 (55.1) | 147 (56.5) | 76 (51.7) | 185/333 (55.6) |
| S3 auscultation | 160/731 (21.9) | 54/256 (21.1) | 25/146 (17.1) | 81/329 (24.6) |
| SpO2, % | 96 (94 - 98) | 97 (95 - 98) | 96 (94 - 98) | 96 (94 - 98) |
| Self-assessment | ||||
| Global VAS | 50 (30 - 67) | 47 (26 - 61) | 49 (25 - 67) | 53 (34 - 70) |
| Dyspnea VAS | 54 (33 - 77) | 50 (30 - 72) | 50 (30 - 74) | 61 (39 - 80) |
| Local Labs | ||||
| Sodium, mg/L | 138 (136 - 141) | 139 (136 - 141) | 137 (134 - 140) | 139 (136 - 141) |
| Potassium, mEq/L | 4.00 (3.70 - 4.30) | 3.95 (3.55 - 4.20) | 4.10 (3.80 - 4.40) | 4.00 (3.70 - 4.30) |
| Bicarbonate, mEq/L | 28 (25 - 30) | 28 (25 - 30) | 28.7 (25.0 - 31.0) | 27 (25 - 30) |
| Hemoglobin, g/dL | 11.3 (10.2 - 12.7) | 11.4 (10.2 - 13.1) | 11.1 (9.7 - 12.2) | 11.4 (10.3 - 12.7) |
| Blood urea nitrogen, mg/dl | 38 (26 - 53) | 31.0 (20.2 - 49.0) | 48.5 (39.0 - 64.0) | 36 (27 - 50) |
| GFR, mL/min/1.73m2 | 41.3 (31.1 - 55.2) | 49.6 (35.8 - 67.2) | 31.8 (25.5 - 41.3) | 41.9 (32.3 - 53.4) |
| Serum creatinine, mg/dl | 1.70 (1.36 - 2.15) | 1.50 (1.16 - 1.90) | 2.10 (1.70 - 2.65) | 1.69 (1.40 - 2.00) |
| Core Labs | ||||
| Creatinine, mg/dl | 1.64 (1.27 - 2.05) | 1.44 (1.08 - 1.90) | 2.03 (1.59 - 2.51) | 1.61 (1.33 - 1.97) |
| NT-proBNP, pg/ml | 4503 (2315 - 10150) | 4441 (2466 - 10517) | 4119 (1594 - 9604) | 4954 (2337 - 10120) |
| Serum aldosterone, pg/ml | 200 (108 - 356) | 183 (95 - 349) | 228 (117 - 377) | - |
| Cystatin C, mg/L | 1.70 (1.36 - 2.18) | 1.48 (1.11 - 1.92) | 2.15 (1.69 - 2.65) | 1.70 (1.42 - 2.15) |
| Uric acid, mg/dL | 9.9 (8.1 - 11.8) | 9.7 (7.9 - 11.3) | 10.8 (9.0 - 12.4) | - |
| Plasma renin activity, ng/mL/hr | 4.9 (0.9 - 15.9) | 4.1 (0.8 - 15.6) | 5.3 (1.6 - 18.1) | - |
Table 2.
Discharge / Day 7 Characteristics by Trial
| Characteristic | Overall (N=744) | DOSE (N=260) | CARRESS (N=147) | ROSE (N=337) |
|---|---|---|---|---|
| Days from randomization to discharge | 6 (4 - 9) | 5 (3 - 9) | 7 (5 - 11) | 6 (4 - 9) |
| Edema ≥ 2 | 168/710 (23.7) | 55/245 (22.4) | 48/141 (34.0) | 65/324 (20.1) |
| JVP ≥ 8 cm water | 330/636 (51.9) | 100/213 (46.9) | 81/128 (63.3) | 149/295 (50.5) |
| Heart rate | 72.5 (65.0 - 83.0) | 75.0 (66.0 - 85.5) | 73 (65 - 83) | 72 (64 - 81) |
| SBP, mmHg | 110 (100 - 125) | 110 (100 - 125) | 117 (102 - 128) | 107 (98 - 124) |
| DBP, mmHg | 61 (56 - 70) | 63.5 (56.0 - 72.0) | 62 (56 - 71) | 60 (55 - 67) |
| Weight, lbs | 190 (162 - 240) | 189 (163 - 232) | 203 (168 - 261) | 187 (159 - 233) |
| Weight change, lbs | −10.5 (−17.4, −5.51) | −10.6 (−15.9, −5.51) | −15.0 (−21.8, −7.90) | −9.26 (−15.4, −4.85) |
| BMI, kg/m2 | 29.5 (24.8 - 36.1) | 29.1 (24.6 - 35.2) | 30.7 (25.8 - 38.4) | 29.1 (24.3 - 36.0) |
| Orthopnea | 417/631 (66.1) | 132/205 (64.4) | 87/129 (67.4) | 198/297 (66.7) |
| Orthoedema score | ||||
| 0 | 405/713 (56.8) | 147/247 (59.5) | 69/142 (48.6) | 189/324 (58.3) |
| 1 | 70/713 (9.8) | 26/247 (10.5) | 20/142 (14.1) | 24/324 (7.4) |
| 2 | 148/713 (20.8) | 46/247 (18.6) | 27/142 (19.0) | 75/324 (23.1) |
| 3 | 63/713 (8.8) | 24/247 (9.7) | 18/142 (12.7) | 21/324 (6.5) |
| 4 | 27/713 (3.8) | 4/247 (1.6) | 8/142 (5.6) | 15/324 (4.6) |
| Medications | ||||
| ACEI/ARB | 334/682 (49.0) | 164 (63.1) | 56 (38.1) | 114/275 (41.5) |
| Hydralazine | 146/682 (21.4) | 54 (20.8) | 32 (21.8) | 60/275 (21.8) |
| Nitrates | 215/682 (31.5) | 86 (33.1) | 55 (37.4) | 74/275 (26.9) |
| Beta blocker | 549/682 (80.5) | 205 (78.8) | 122 (83.0) | 222/275 (80.7) |
| Aldosterone antagonist | 218/682 (32.0) | 96 (36.9) | 33 (22.4) | 89/275 (32.4) |
| Digoxin | 189/682 (27.7) | 93 (35.8) | 28 (19.0) | 68/275 (24.7) |
| Aspirin | 491/682 (72.0) | 188 (72.3) | 110 (74.8) | 193/275 (70.2) |
| Warfarin | 302/682 (44.3) | 127 (48.8) | 61 (41.5) | 114/275 (41.5) |
| Local Labs | ||||
| Sodium, mEq/L | 137 (135 - 140) | 137 (135 - 140) | 137 (133 - 139) | 137 (135 - 140) |
| Potassiumm, mEq/L | 4.0 (3.8 - 4.4) | 4.0 (3.7 - 4.3) | 4.2 (3.9 - 4.5) | 4.0 (3.7 - 4.3) |
| BUN, mg/dL | 44 (30 - 63) | 36 (25 - 53) | 60.0 (39.2 - 79.0) | 44.0 (31.0 - 58.4) |
| Bicarbonate, mEq/L | 29 (27 - 32) | 29 (27 - 32) | 29 (26 - 33) | 29 (27 - 32) |
| Creatinine, mg/dL | 1.72 (1.38 - 2.17) | 1.60 (1.20 - 2.00) | 2.00 (1.58 - 2.60) | 1.70 (1.40 - 2.13) |
| eGFR, mg/dL | 40.6 (31.4 - 53.7) | 45.3 (34.1 - 65.2) | 35.0 (25.1 - 44.3) | 40.0 (31.8 - 51.8) |
| SpO2, % | 96 (95 - 98) | 97 (95 - 98) | - | 96 (95 - 98) |
Patient characteristics at discharge/day 7
The median length of hospitalization from study randomization to hospital discharge was 6 (IQR, 4,9) days. Decongestion was incomplete with residual ≥ 2+ edema in 24% of patients, JVP ≥ 8 cm H20 in 52%, orthopnea in 66%, and full relief of congestion (orthoedema score of 0) in only 57% of patients. Median weight change from randomization to discharge/day 7 was −4.8 kg. (IQR, −7.9, −2.5).
Timing and causes of readmission
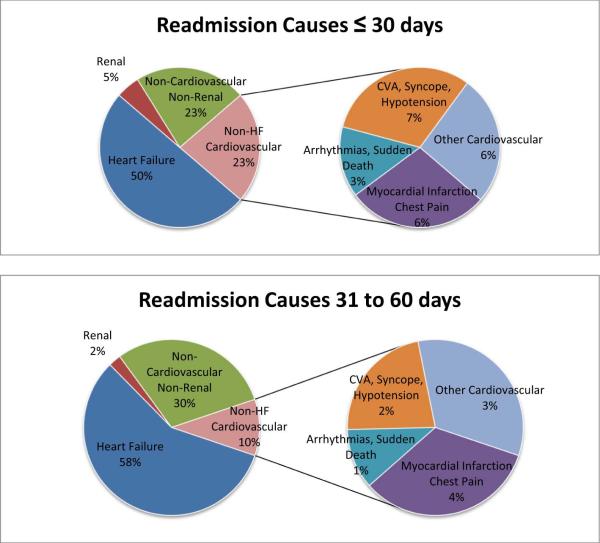
Death during the trial enrollment hospitalization occurred in 15 of the 835 subjects (1.8%) enrolled to the three trials. Complete post-discharge readmission and mortality data were available for 744 subjects. Death occurred within 60 days post-discharge in 47 of these subjects (7.3%). In time-to-event analysis, death or all-cause readmission occurred in 26% (186 readmissions, 9 deaths without readmission) within 30 days post-discharge and 38% (273 readmissions, 11 deaths without readmission) within 60 days post-discharge. Non-HF cardiovascular causes of readmission were proportionally more common in patients readmitted within 30 days vs. patients readmitted between 31 to 60 days (23% vs. 10%, p=0.016). HF (50% vs. 57%, p=0.25), renal (5% vs. 2.3%, p=0.51), and non-cardiovascular non-renal (23% vs. 30%, p=0.19) causes of readmission were not significantly different between those readmitted within 30 days and those readmitted between 31 to 60 days. Of the non-HF cardiovascular causes of readmission, the distribution among several more specific diagnoses was similar within 30 days and between 31 to 60 days. FIGURE 1.
Fig. 1.
Readmission causes within 30 days and 31–60 days after discharge from an acute heart failure (HF) hospitalization.
All-Cause Readmission/Death
Variables associated with all-cause readmission/death in a Cox proportional hazards model are shown in TABLE 3. Baseline serum chemistries that predicted greater risk of readmission/death included higher BUN, higher bicarbonate, and lower sodium. Demographic and baseline clinical predictors of readmission/death included non-white race, female sex, a history of depression, and longer length of stay. Among clinical examination variables, only lower SBP at discharge/day 7 was associated with increased readmission/death. Use of ACEI/ARB was the only medical therapy predictive of readmission/death – with a lower event rate in patients taking ACEI/ARB at either admission, discharge, or both. Those patients taking ACEI/ARB both at discharge and admission had the lowest hazard. The overall c-statistic for the multivariable model in predicting all-cause readmission/death within 60 days was 0.67.
Table 3.
Multivariable Adjusted Cox Proportional Hazards Model: All-Cause Readmission or Death†
| Variable | HR (95% CI) | p-value |
|---|---|---|
| BUN at baseline, per 1 mg/dL increase to 50 | 1.02 (1.01, 1.04) | <0.001 |
| ACEI/ARB (reference = neither at randomization nor discharge/day 7) | <0.001 | |
| Both randomization and discharge | 0.56 (0.42, 0.75) | |
| Discharge only | 0.64 (0.38, 1.06) | |
| Randomization only | 0.59 (0.40, 0.89) | |
| Sodium at baseline, per 1 mEq/L increase | 0.95 (0.92, 0.98) | <0.001 |
| White race | 0.65 (0.49, 0.86) | 0.003 |
| Bicarbonate at baseline, per 1mEq/L increase | 1.04 (1.01, 1.07) | 0.014 |
| SBP at discharge/day 7, per 10 mmHg increase | 0.93 (0.86, 0.99) | 0.030 |
| Depression | 1.33 (1.02, 1.75) | 0.038 |
| Length of stay, per 1 day increase | 1.06 (1.01, 1.11) | 0.020 |
| Male sex | 0.74 (0.57, 0.98) | 0.033 |
Adjusted for trial and ejection fraction<50% versus ≥50%
Cardiovascular Readmission/Death
Variables associated with cardiovascular readmission/death in a Cox proportional hazards model are shown in TABLE 4. Risk factors for cardiovascular readmission/death were similar to risk factors for all-cause readmission/death except for the exclusion of male sex and bicarbonate and with the addition of HF hospitalization within the last year, COPD, and degree of hypokalemia (< 3.7 mEq/L). Notably, those patients maintained or initiated on ACEI/ARB were at the lowest hazard of readmission. The overall c-statistic for the multivariable model in predicting cardiovascular readmission/death within 60 days was 0.69.
Table 4.
Multivariable Adjusted Cox Proportional Hazards Model: Cardiovascular Readmission or Death†
| Variable | HR (95% CI) | p-value |
|---|---|---|
| BUN at baseline, per 1 mg/dL increase to 50 | 1.03 (1.01, 1.04) | <0.001 |
| SBP at discharge/day 7, per 10 mmHg increase | 0.88 (0.81, 0.95) | 0.002 |
| Length of stay, per 1 day increase | 1.08 (1.02, 1.14) | 0.009 |
| White race | 0.67 (0.49, 0.93) | 0.015 |
| Chronic obstructive pulmonary disease | 1.45 (1.07, 1.96) | 0.016 |
| Sodium at baseline, per 1 mEq/L increase | 0.96 (0.93, 0.99) | 0.021 |
| Potassium at baseline, per 0.1 mEq/L increase to 3.7 | 0.93 (0.87, 0.99) | 0.023 |
| Depression | 1.43 (1.04, 1.96) | 0.030 |
| Hospitalization for heart failure in last year | 1.48 (1.04, 2.11) | 0.030 |
| ACEI/ARB (reference = neither at randomization nor discharge/day 7) | 0.049 | |
| Both randomization and discharge | 0.66 (0.47, 0.92) | |
| Discharge only | 0.55 (0.28, 1.08) | |
| Randomization only | 0.71 (0.45, 1.15) |
Adjusted for trial and ejection fraction<50% versus ≥50%
DISCUSSION
In this analysis of three AHF trials enrolling patients with prominent congestion and prevalent renal dysfunction, we found an overall rate of readmission/death of 26%, similar to contemporary reports.4 Only half of the readmissions within 30-days and 58% of readmissions within 60-days were attributed to heart failure. Readmissions for non-HF cardiovascular causes were more likely to occur within 30 days than from 31-60 days. Risk factors for all-cause readmission included renal dysfunction, hyponatremia, hypercarbia, lower blood pressure, depression, non-white race, female sex, and greater length-of-stay. The use of ACEI/ARB was associated with lower rates of readmission/death. Risk factors for cardiovascular readmission were generally similar, with COPD, HF hospitalization within one year prior to trial enrollment, hyponatremia, renal dysfunction, hypotension, and degree of hypokalemia (< 3.7 mEq/L) being particular risk factors for cardiovascular readmission. Overall, the risk of readmission/death could be predicted with modest accuracy (AUC 0.67 for all-cause, 0.69 for cardiovascular).
The present study cohort is different from previous cohorts, being younger, with more renal dysfunction, higher percentage of men, and racially more diverse. This cohort (median age = 69 years; 36% < 65 years) is younger than studies accessing Medicare-based and other administrative cohorts, however age was not a risk factor for readmission/death. As specified by trial enrollment criteria and site characteristics, renal dysfunction (median creatinine = 1.7 mg/dl; median estimated GFR = 41.3 mL/min/1.73m2) was highly prevalent. There was disproportionate representation of males (75%), and there was a relatively large proportion of non-white subjects (26%). Although patients with both preserved (HFpEF) and reduced (HFrEF) ejection fraction were eligible for enrollment, the cohort is weighted towards systolic dysfunction (70% with LVEF < 50%; median LVEF = 30%). Nearly the entire cohort was NYHA Class III or IV and > 70% had been previously hospitalized for HF within one year of trial hospitalization. While these factors might have been expected to predispose the cohort to higher readmission rates, the overall 30-day readmission rate of 25% is similar to contemporary data.4
An analysis of the large, multinational EVEREST trial demonstrated a similar proportion of HF, non-HF cardiovascular, and non-cardiovascular readmissions comparing 0-30 vs. 31-60 days post-discharge.21 Our finding of relatively more non-HF cardiovascular readmissions in the 0-30 day period may be attributable to characteristics of the subjects enrolled to these trials: treated in North American teaching hospitals, greater severity of renal dysfunction, both HFpEF and HFrEF, and a greater burden of non-HF comorbidities such as COPD, hypertension, and diabetes, which are known to be associated with preventable hospitalization.22 By focusing on the impact of HF hospitalization on the care of cardiovascular comorbidities, opportunities for preventing early readmission may be revealed.
The identified associations between serum chemistries and all-cause readmission/death reflect the central role of impaired baseline renal function (BUN) and hallmarks of aggressive diuresis (hyponatremia and hypercarbia) in the face of continued congestion. Baseline serum chemistries were selected in the models over post-treatment (discharge/day7) values, suggesting baseline values may be more predictive of readmission rates. Serum potassium was not retained in the model for all-cause readmission/death, however a “U-shaped” function for serum potassium and cardiovascular readmission/death was noted, with a statistically significant relationship between serum potassium < 3.7 mEq/L and the outcome (Supplementary appendix figure). Interestingly, forcing a linear spline for potassium > 3.7 mEq/L did not change the model performance and the relationship between hyperkalemia and cardiovascular readmission/death was not statistically significant (p=0.27), suggesting that in this population hypokalemia represents a relatively greater risk for adverse events than does hyperkalemia.
Clinical examination findings consistent with congestion were not significant in the multivariable model for all-cause or cardiovascular readmission and death. While examination findings may be valuable in HF management, serum chemistries may have performed better in predictive models because they better reflect more complex interplay of congestion, disease severity, comorbidity, and end-organ function. Among clinical variables, only lower systolic blood pressure, similar to other analyses,23 was associated with increased hazard of readmission/death.
Among demographics, clinical history, and comorbidities, the variables associated with increased all-cause readmission/death included female sex, depression, non-white race, and increased length of stay. A greater hazard for readmission/death in non-white subjects was demonstrated in previous risk-adjusted analysis of Medicare data, finding black patients and patients cared for at minority-serving hospitals to be more likely than white patients or patients cared for at non-minority-serving hospitals to be readmitted for HF, acute MI, and pneumonia.24 The proportion of non-white subjects in this analysis was notably higher than most multi-center HF readmission analyses and reflective of patient characteristics at participating hospitals. The influence of race on readmission likely represents biologic, economic, and social variables not otherwise captured in this analysis, and points towards a need to further explore and account for these factors in considering post-discharge outcomes in HF.
While population-level trends demonstrate an increased risk of readmission contemporaneous with reduced length of stay25 and countries with longer length of stay tend to have reduced readmission rates,26 we found greater length of stay was associated with an increased hazard for readmission. In this case, greater length of stay may represent greater HF disease severity, greater complexity of the AHF episode, greater overall burden of comorbidity, socioeconomic barriers to safe hospital discharge, or simply reflect practice patterns that differ by geographic location.
The high prevalence of depression in HF and its influence on increased mortality and readmission has also been previously described,27,28 and more than 20% of subjects in this cohort had a history of treated depression. Given the lack of trial data to support a role for antidepressants to improve clinical outcomes in heart failure patients,29 our analysis highlights the continued need to develop alternative strategies for addressing comorbid depression in hospitalized heart failure.
Perhaps the most intriguing finding in this analysis, one with potential application to clinical practice, is the association between ACEI/ARB use, particularly at both admission and discharge, and a lower hazard of readmission/death. As expected, patients with ACEI/ARB use had better renal function and more commonly had reduced LVEF, while comorbidities and disease severity were generally similar across groups. (Supplementary appendix table 1). Though aggressive decongestion (median 5 kg. weight loss by discharge/day 7) in the setting of baseline renal dysfunction or worsening renal function may lead clinicians to avoid RAAS-blocking medications, this multivariable analysis adjusting a priori for trial and LVEF and retaining renal function (BUN) and blood pressure in the model demonstrated that ACEI/ARB use was associated with reduced readmission/death. Management of RAAS blocking agents in the setting of AHF, particularly with associated renal dysfunction, is a current knowledge gap. Review of trial data in chronic HF suggests a benefit of RAAS-blockade that extends through CKD stage III and potentially CKD stage IV.30 In this analysis, hypokalemia more-so than hyperkalemia was associated with subsequent adverse events. Further, ACE/ARB or aldosterone antagonist use did not alter the relationship between potassium level and cardiovascular readmission/death. RAAS inhibition may in some cases be unduly abandoned in the AHF setting where modest serum creatinine and serum potassium derangements are present. While studies of chronic RAAS inhibition in the setting of renal dysfunction are in development, studies of the appropriate management of RAAS-blocking agents in the setting of AHF with renal dysfunction also warrant further prospective investigation to address the balance of benefit and risk in these patients.
Limitations
The findings of this retrospective analysis of three randomized clinical trials conducted using different therapeutic approaches to decongestion and with different enrollment criteria should be considered hypothesis-generating. Inferences from this cohort should be generalized to populations with different baseline characteristics with caution. Chiefly, these findings from subjects treated within an academic clinical trial network with defined enrollment criteria and decongestive strategies may be less applicable to community patient samples or heart failure populations with key comorbidities that would cause exclusion from trial participation. However, within populations that do mirror the study cohort and for future trials of decongestion in acute heart failure these results may be particularly useful.
Limitations to analysis imposed by the nature of trial design and conduct include the availability of a discharge/day 7 time point for certain variables as opposed to strictly discharge variables. The differences in serum chemistries following a 2-day vs. a 7-day admission are expected to be smaller and this may have contributed to the lesser performance of discharge/day7 variables in multivariable regression. Missing data may have also affected the analysis, though variables with high missing rates (PRA, aldosterone, troponin I, uric acid, and pro-collagen III NTP) were omitted from multivariable analysis. In single variable analysis, troponin I, aldosterone, and PRA had modest but significant associations with outcomes that might have been borne out in multivariable analysis. BNP, which as a value measured on discharge has been shown to improve the predictive accuracy of mortality and readmission,31 was not available at discharge/day7 in this data set. Attribution of causes for readmission was limited to diagnoses included in the trials case report forms; further retrospective detailing of multifactorial readmission causes was not possible. Data were also not available for the analysis of post-discharge and hospital-to-home care plans, a fact that may account for the modest c-statistic in this and other analyses that focus solely on acute hospitalization variables. Finally, the overall size of this cohort is modest in comparison to larger trial or registry experiences, though the degree of clinical assessment and data clarity in these trials was good, and ultimately these data may prove useful in designing and analyzing future trials in a similar population.
CONCLUSION
In this multi-trial cohort with prominent congestion and prevalent renal dysfunction, readmission after HF hospitalization was more likely to be for non-HF cardiovascular conditions in the early (0-30 day post-discharge) time-frame. Variables associated with greater cumulative all-cause readmission/death included greater non-white race, depression, lower blood pressure, and laboratory markers reflecting the interplay of decongestive therapy and renal dysfunction (elevated BUN, hyponatremia, and elevated serum bicarbonate). ACEI/ARB was the only pharmacotherapy independently associated with lesser readmission/death. The models’ performance in discriminating all-cause readmission (AUC = 0.67) or cardiovascular readmission (0.69) was modest. However, further study of the factors predictive of readmission, in particular maintaining the use of ACEI/ARB, may confirm opportunities to mitigate post-discharge readmission and death.
Supplementary Material
Highlights.
Early readmission after heart failure hospitalization was often due to cardiovascular comorbidity.
ACE/ARB use was associated with lesser risk for early readmission/death.
Hypokalemia, but not hyperkalemia, was associated with increased cardiovascular readmission/death.
Despite detailed physical examination data from a trial cohort, readmission prediction was limited.
Acknowledgments
Funding: This work was supported by grants from the National Heart, Lung, and Blood Institute (NHLBI) (coordinating center: U10 HL084904; regional clinical centers: U01 HL084861, U10 HL110312, U109 HL110337, U01 HL084889, U01 HL084890, U01 HL084891, U10 HL110342, U10 HL110262, U01 HL084931, U10 HL110297, U10 HL110302, U10 HL110309, U10 HL110336, U10 HL110338).
Disclosures: RJM receives research support from the NIH, Amgen, AstraZeneca, Bristol-Myers Squibb, GlaxoSmithKline, Gilead, Novartis, Otsuka, and ResMed; honoraria from Thoratec; and has served on an advisory board for Luitpold Pharmaceuticals, Inc. DD receives research support from the American Heart Association, Amgen, Maquet, Novartis, and Thoratec. SMD receives funding from the NIH (K23 HL116643)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Blecker S, Paul M, Taksler G, Ogedegbe G, Katz S. Heart failure–associated hospitalizations in the United States. [2014 Dec 3];J Am Coll Cardiol [Internet] 2013 61:1259–67. doi: 10.1016/j.jacc.2012.12.038. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3838728&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pfuntner A, Wier LM, Steiner C. HCUP Statistical Brief #146. Agency Healthc Res Qual; Rockville, MD: Costs for Hospital Stays in the United States, 2010. https://www.hcup us.ahrq.gov/reports/statbriefs/sb146.jsp. [PubMed] [Google Scholar]
- 3.Steiner C, Barrett M, Weiss A. HCUP Projections: Cardiovascular/ Cerebrovascular Conditions and Procedures 2001 to 2012. HCUP Proj Rep # 2012-02. 2012 https://www.hcup us.ahrq.gov/reports/projections/2012-02.pdf.
- 4.Elixhauser A, Steiner C. Readmissions to U.S. Hospitals by Diagnosis, 2010. HCUP Statistical Brief # 153. Agency Healthc Res Qual Rockville, MD. :1–19. https://www.hcup us.ahrq.gov/reports/statbriefs/sb153.jsp. [PubMed]
- 5.Hines A, Barrett M, Jiang H, Steiner C. Conditions With the Largest Number of Adult Hospital Readmissions by Payer, 2011. HCUP Statistical Brief #172. Agency Healthc Res Qual Rockville, MD. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb172-Conditions-Readmissions-Payer.pdf. [PubMed]
- 6.Chun S, Tu JV, Wijeysundera HC, Austin PC, Wang X, Levy D, Lee DS. Lifetime analysis of hospitalizations and survival of patients newly admitted with heart failure. [2015 Jan 5];Circ Heart Fail [Internet] 2012 5:414–21. doi: 10.1161/CIRCHEARTFAILURE.111.964791. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22556322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hall M, Levant S, DeFrances C. Hospitalization for congestive heart failure: United States, 2000–2010. [2015 Jan 8];NCHS data brief, no 108 Hyattsville, MD Natl Cent Heal Stat [Internet] 2012 Available from: http://stacks.cdc.gov/view/cdc/13508/cdc_13508_DS1.pdf. [PubMed]
- 8.Ayanian JZ, Weissman JS, Chasan-Taber S, Epstein a. M. Quality of care for two common illnesses in teaching and nonteaching hospitals. [2015 Jan 11];Health Aff [Internet] 1998 17:194–205. doi: 10.1377/hlthaff.17.6.194. Available from: http://content.healthaffairs.org/cgi/doi/10.1377/hlthaff.17.6.194. [DOI] [PubMed] [Google Scholar]
- 9.McAlister F a, Au AG, Majumdar SR, Youngson E, Padwal RS. Postdischarge outcomes in heart failure are better for teaching hospitals and weekday discharges. [2015 Jan 8];Circ Heart Fail [Internet] 2013 6:922–9. doi: 10.1161/CIRCHEARTFAILURE.113.000336. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23811962. [DOI] [PubMed] [Google Scholar]
- 10.Kociol RD, Peterson ED, Hammill BG, Flynn KE, Heidenreich P a, Piña IL, Lytle BL, Albert NM, Curtis LH, Fonarow GC, Hernandez AF. National survey of hospital strategies to reduce heart failure readmissions: findings from the Get With the Guidelines-Heart Failure registry. [2015 Jan 11];Circ Heart Fail [Internet] 2012 5:680–7. doi: 10.1161/CIRCHEARTFAILURE.112.967406. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22933525. [DOI] [PubMed] [Google Scholar]
- 11.Felker GM, Connor CMO, Braunwald E. Loop Diuretics in Acute Decompensated Heart Failure. Circ Heart Fail. 2009;2:56–62. doi: 10.1161/CIRCHEARTFAILURE.108.821785. 10.1161/CIRCHEARTFAILURE.108.821785.Loop. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Felker G, Lee K, Bull D. Diuretic strategies in patients with acute decompensated heart failure. New Engl J Med Engl J Med. 2011;364:797–805. doi: 10.1056/NEJMoa1005419. DOI: 10.1056/NEJMoa1005419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bart B a, Goldsmith SR, Lee KL, Redfield MM, Felker GM, O'Connor CM, Chen HH, Rouleau JL, Givertz MM, Semigran MJ, Mann D, Deswal A, Bull D a, Lewinter MM, Braunwald E. Cardiorenal rescue study in acute decompensated heart failure: rationale and design of CARRESS-HF, for the Heart Failure Clinical Research Network. [2015 Jan 8];J Card Fail [Internet] 2012 18:176–82. doi: 10.1016/j.cardfail.2011.12.009. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3503538&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bart B a, Goldsmith SR, Lee KL, Givertz MM, O'Connor CM, Bull D a, Redfield MM, Deswal A, Rouleau JL, LeWinter MM, Ofili EO, Stevenson LW, Semigran MJ, Felker GM, Chen HH, Hernandez AF, Anstrom KJ, McNulty SE, Velazquez EJ, Ibarra JC, Mascette AM, Braunwald E. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. [2014 Oct 4];N Engl J Med [Internet] 2012 367:2296–304. doi: 10.1056/NEJMoa1210357. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3690472&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen HH, Ch MBB, Ezzeddine OFA, Anstrom KJ, Givertz M, Bart BA, Felker GM, Hernandez AF, Lee KL, Braunwald E, Redfield MM, Heart F. Targeting the kidney in acute heart failure: can old drugs provide new benefit? Renal Optimization Strategies Evaluation in Acute Heart Failure (ROSE AHF) trial. Circ Hear Fail. 2014;6:1–17. doi: 10.1161/CIRCHEARTFAILURE.113.000347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen HH, Anstrom KJ, Givertz MM, Stevenson LW, Semigran MJ, Goldsmith SR, Bart B a, Bull D a, Stehlik J, LeWinter MM, Konstam M a, Huggins GS, Rouleau JL, O'Meara E, Tang WHW, Starling RC, Butler J, Deswal A, Felker GM, O'Connor CM, Bonita RE, Margulies KB, Cappola TP, Ofili EO, Mann DL, Dávila-Román VG, McNulty SE, Borlaug B a, Velazquez EJ, Lee KL, Shah MR, Hernandez AF, Braunwald E, Redfield MM. Low-dose dopamine or low-dose nesiritide in acute heart failure with renal dysfunction: the ROSE acute heart failure randomized trial. [2014 Jul 20];JAMA [Internet] 2013 310:2533–43. doi: 10.1001/jama.2013.282190. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3934929&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bekelman DB, Havranek EP, Becker DM, Kutner JS, Peterson PN, Wittstein IS, Gottlieb SH, Yamashita TE, Fairclough DL, Dy SM. Symptoms, depression, and quality of life in patients with heart failure. [2013 Jul 2];J Card Fail [Internet] 2007 13:643–8. doi: 10.1016/j.cardfail.2007.05.005. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17923356. [DOI] [PubMed] [Google Scholar]
- 18.Lala A, McNulty SE, Mentz RJ, Dunlay SM, Vader JM, AbouEzzeddine OF, DeVore AD, Khazanie P, Redfield MM, Goldsmith SR, Bart B a, Anstrom KJ, Felker GM, Hernandez AF, Stevenson LW. Relief and Recurrence of Congestion During and After Hospitalization for Acute Heart Failure: Insights From Diuretic Optimization Strategy Evaluation in Acute Decompensated Heart Failure (DOSE-AHF) and Cardiorenal Rescue Study in Acute Decompensated Heart Failure (CARESS-HF). [2016 Jan 18];Circ Heart Fail [Internet] 2015 8:741–8. doi: 10.1161/CIRCHEARTFAILURE.114.001957. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26041600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klein L, Massie BM, Leimberger JD, O'Connor CM, Piña IL, Adams KF, Califf RM, Gheorghiade M. Admission or changes in renal function during hospitalization for worsening heart failure predict postdischarge survival: results from the Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CHF). [2015 Jan 11];Circ Heart Fail [Internet] 2008 1:25–33. doi: 10.1161/CIRCHEARTFAILURE.107.746933. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19808267. [DOI] [PubMed] [Google Scholar]
- 20.Schrier RW. Blood urea nitrogen and serum creatinine: not married in heart failure. [2015 Jan 11];Circ Heart Fail [Internet] 2008 1:2–5. doi: 10.1161/CIRCHEARTFAILURE.108.770834. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19808263. [DOI] [PubMed] [Google Scholar]
- 21.O'Connor CM, Miller AB, Blair JE a, Konstam M a, Wedge P, Bahit MC, Carson P, Haass M, Hauptman PJ, Metra M, Oren RM, Patten R, Piña I, Roth S, Sackner-Bernstein JD, Traver B, Cook T, Gheorghiade M. Causes of death and rehospitalization in patients hospitalized with worsening heart failure and reduced left ventricular ejection fraction: results from Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study with Tolvaptan (EVEREST) program. [2013 Sep 27];Am Heart J [Internet] 2010 159:841–849. e1. doi: 10.1016/j.ahj.2010.02.023. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20435194. [DOI] [PubMed] [Google Scholar]
- 22.Braunstein JB, Anderson GF, Gerstenblith G, Weller W, Niefeld M, Herbert R, Wu AW. Noncardiac comorbidity increases preventable hospitalizations and mortality among medicare beneficiaries with chronic heart failure. [2014 Nov 30];J Am Coll Cardiol [Internet] 2003 42:1226–1233. doi: 10.1016/s0735-1097(03)00947-1. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0735109703009471. [DOI] [PubMed] [Google Scholar]
- 23.Ambrosy AP, Vaduganathan M, Mentz RJ, Greene SJ, Subačius H, Konstam M a, Maggioni AP, Swedberg K, Gheorghiade M. Clinical profile and prognostic value of low systolic blood pressure in patients hospitalized for heart failure with reduced ejection fraction: insights from the Efficacy of Vasopressin Antagonism in Heart Failure: Outcome Study with Tolvaptan (EVEREST) t. [2015 Jan 26];Am Heart J [Internet] 2013 165:216–25. doi: 10.1016/j.ahj.2012.11.004. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23351825. [DOI] [PubMed] [Google Scholar]
- 24.Joynt K, Orav E, Jha A. Thirty-day readmission rates for Medicare beneficiaries by race and site of care. [2016 Jan 18];JAMA J Am Med Assoc [Internet] 2011 305:675–681. doi: 10.1001/jama.2011.123. Available from: http://archsurg.jamanetwork.com/article.aspx?articleid=645647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bueno H, Ross J, Wang Y, Chen J. Trends in length of stay and short-term outcomes among Medicare patients hospitalized for heart failure, 1993-2006. [2016 Jan 18];Jama [Internet] 2010 303:2141–2147. doi: 10.1001/jama.2010.748. Available from: http://archpsyc.jamanetwork.com/article.aspx?articleid=185996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Eapen ZJ, Reed SD, Li Y, Kociol RD, Armstrong PW, Starling RC, McMurray JJ, Massie BM, Swedberg K, Ezekowitz J a, Fonarow GC, Teerlink JR, Metra M, Whellan DJ, O'Connor CM, Califf RM, Hernandez AF. Do countries or hospitals with longer hospital stays for acute heart failure have lower readmission rates?: Findings from ASCEND-HF. [2015 Jan 11];Circ Heart Fail [Internet] 2013 6:727–32. doi: 10.1161/CIRCHEARTFAILURE.112.000265. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23770519. [DOI] [PubMed] [Google Scholar]
- 27.Johnson TJ, Basu S, Pisani B a, Avery EF, Mendez JC, Calvin JE, Powell LH. Depression predicts repeated heart failure hospitalizations. [2015 Jan 18];J Card Fail [Internet] 2012 18:246–52. doi: 10.1016/j.cardfail.2011.12.005. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22385946. [DOI] [PubMed] [Google Scholar]
- 28.Jiang W, Alexander J, Christopher E, Kuchibhatla M, Califf RM, Krishnan RR, Connor CMO. Relationship of Depression to Increased Risk of Mortality and Rehospitalization in Patients With Congestive Heart Failure. Arch Intern Med. 2015;161:1849–1856. doi: 10.1001/archinte.161.15.1849. [DOI] [PubMed] [Google Scholar]
- 29.O'Connor CM, Jiang W, Kuchibhatla M, Silva SG, Cuffe MS, Callwood DD, Zakhary B, Stough WG, Arias RM, Rivelli SK, Krishnan R. Safety and efficacy of sertraline for depression in patients with heart failure: results of the SADHART-CHF (Sertraline Against Depression and Heart Disease in Chronic Heart Failure) trial. [2014 Nov 25];J Am Coll Cardiol [Internet] 2010 56:692–9. doi: 10.1016/j.jacc.2010.03.068. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3663330&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lassus J, Zannad F, Krum H, Mcmurray JJV. Current Evidence on Treatment of Patients With Chronic Systolic Heart Failure and Renal Insufficiency Practical Considerations From Published Data. J Am Coll Cardiol. 2014;63:853–871. doi: 10.1016/j.jacc.2013.11.031. [DOI] [PubMed] [Google Scholar]
- 31.Kociol RD, Horton JR, Fonarow GC, Reyes EM, Shaw LK, O'Connor CM, Felker GM, Hernandez AF. Admission, discharge, or change in B-type natriuretic peptide and long-term outcomes: data from Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE-HF) linked to Medicare claims. [2015 Jan 5];Circ Heart Fail [Internet] 2011 4:628–36. doi: 10.1161/CIRCHEARTFAILURE.111.962290. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3465672&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.