Supplemental digital content is available in the text.
Abstract
Background
Occurring frequently after solid organ and hematopoietic stem cell transplantation, cytomegalovirus (CMV) replication remains a relevant cause of mortality and morbidity in affected patients. Despite these adverse effects, an increased alloreactivity of natural killer (NK) cells after CMV infection has been assumed, but the underlying physiopathological mechanisms have remained elusive.
Methods
We used serial analyses of NK cells before and after CMV infection in kidney transplant recipients as an in vivo model for CMV primary infection to explore the imprint of CMV infection using every patient as their own control: We analyzed NK cell phenotype and function in 47 CMV seronegative recipients of CMV seropositive kidney grafts, who developed CMV primary infection posttransplant. Seronegative recipients of seronegative kidney grafts served as controls.
Results
We observed a significant increase of NKG2C expressing NK cells after CMV infection (mean increase, 17.5%; 95% confidence interval [95% CI], 10.2-24.9, P < 0.001), whereas cluster of differentiation (CD)57 expressing cells decreased (mean decrease, 14.1%; 95% CI, 8.0-20.2; P < 0.001). Analysis of killer immunoglobulin-like receptor (KIR) expression showed an increase of cells expressing KIR2DL1 as their only inhibitory KIR in patients carrying the cognate ligand HLA-C2 (mean increase, 10.0%; 95% CI, 1.7-18.3; P = 0.018). In C2-negative individuals, KIR2DL1 expression decreased (mean decrease, 3.9%; 95% CI, 1.6-6.2; P = 0.001). As for activating KIR, there was no conclusive change pattern.
Most importantly, we observed a significantly higher NK cell degranulation and IFNγ production in response to different target cells (target K562, CD107a: mean increase, 9.9%; 95% CI, 4.8-15.0; P < 0.001; IFNγ: mean increase, 6.6%; 95% CI, 1.6-11.1; P < 0.001; target MRC-5, CD107a: mean increase, 6.9%; 95% CI, 0.7-13.1; P = 0.03; IFNγ: mean increase, 4.8%; 95% CI, 1.7-7.8; P = 0.002).
Conclusions
We report evidence for an increased function of NK cells induced by CMV infection. This increased in vitro functionality was seen in NKG2C-positive and NKG2C-negative subsets, arguing for an NKG2C independent mechanism of action.
Cytomegalovirus (CMV) remains a common complication after solid organ and allogeneic hematopoietic stem cell transplantation (HSCT). Cytomegalovirus-induced multiorgan disease can cause transplantation related morbidity and mortality. Despite these adverse effects, it has been suggested that CMV triggers increased alloreactivity of natural killer (NK) cells. This hypothesis origins primarily from the HSCT setting, with several studies demonstrating a protective effect on relapse risk for myeloid malignancies in patients with early CMV reactivation.1-4 The largest study on this issue was performed by Green and colleagues,4 who were able to demonstrate an association between CMV antigenemia and reduced early relapse risk after HSCT but not overall survival among patients with acute myeloid leukemia.
Even though large efforts have been undertaken to unveil the role CMV plays in this setting, the pathophysiological principles underlying this putative protective effect remain largely unknown.
Human CMV (HCMV) infection has been shown to alter NK cells by modifying their phenotype, receptor repertoire, and function.5 Several studies have shown that CMV infection imprints NK cell subpopulations by inducing a long-lasting expansion of educated NK cells expressing the NKG2C receptor, involving several other phenotypical changes including upregulation of self-specific inhibitory killer immunoglobulin like receptors (iKIR) and the maturation marker cluster of differentiation (CD)57 as well as loss of the FcεRγ-chain.6-15 These observations and the fact that NK cells are the first directly cytotoxic lymphocyte population to recover after HSCT led to the formulation of an intriguing hypothesis, that a subset of NKG2C positive mature NK cells occurring after CMV infection mediates anti-tumor activity.
Recently, the protective effect of CMV reactivation on early relapse risk and early disease-free survival after HSCT was confirmed in a cohort of allogeneic stem cell recipients for diverse malignancies after reduced intensity conditioning but not myeloablative conditioning.16 Moreover, the authors found a preferential expansion of donor-derived CD56dimNKG2C + CD57+ NK cells in reduced intensity conditioning patients with CMV reactivation and were able to correlate expansion of these cells to a trend toward reduced 2-year relapse rate.
A major limitation of previous studies assessing the impact of CMV infection on NK cell phenotype and function is that individuals with latent chronic infection were compared to CMV naive subjects. The phenotypic and functional heterogeneity of NK cells between individuals make such comparisons notoriously complicated. Similarly, studies in stem cell transplants—undertaken to assess NK cell function during active CMV replication—faced a comparable issue, as patients without CMV replication were used as controls.7,17 To circumvent this limitation, we performed NK cell phenotype analysis in a cohort of kidney transplant recipients in a paired fashion (ie, before and after CMV infection) to eliminate interindividual heterogeneity and thus using every patient as their own control. We chose the kidney transplant setting as a model for in vivo CMV primary infection, as there are less confounding factors than in the HSCT setting. Although the imprint of CMV on NK cells has been confirmed in many different settings, the functional significance of those changes has not yet been clarified. Therefore, we investigated the response of NK cell subpopulations to a leukemia target and a CMV infected target cell.
Our data indicate a CMV driven increase in NK cell activity against target cells occurring concomitant to the specific phenotypic changes. However, this observed alteration in NK cell function is not limited to the imprinted NKG2C-positive populations.
MATERIALS AND METHODS
Patient Samples
Frozen peripheral blood mononuclear cells (PBMC) from kidney transplant recipients were provided by the Swiss Transplant Cohort Study (STCS), a collaborative approach of all solid organ transplant centers in Switzerland. Samples were obtained after written informed consent at defined timepoints, and the study was performed according to the regulations of the local ethics committees. Baseline data (age at transplant, sex, donor and recipient CMV serostatus, HLA typing, time of CMV infection, etiology of kidney failure) and data on posttransplant events (immunosuppressive medication, induction therapy, CMV infection) were prospectively collected by STCS.
For phenotypic analysis and functional assays, PBMCs were thawed and cultured overnight in Dulbecco modified eagle medium, supplemented with 10% (v/v) heat-inactivated fetal-bovine serum, penicillin (100 μ/mL), and streptomycin (10 μg/mL).
Cell Lines and Viral Strains
As targets for functional assays the fetal human lung fibroblast cell line MRC-5 (provided by A. Helenius, ETH Zürich, Switzerland) and the human HLA-deficient chronic myelogenous leukemia cell line K562 (provided by ATCC, Manassas, VA) were used. MRC-5 cells were infected with a wild-type strain of CMV (provided by H. H. Hirsch, Institute of Microbiology, University Hospital, Basel, Switzerland).
Monoclonal Antibodies and Flow Cytometry
The following monoclonal antibodies were used according to the manufacturer's instructions: anti-CD3 Brilliant Violet 785 (clone OKT3; Biolegend, San Diego, CA); anti-CD14 Brilliant Violet 785 (clone M5E2; Biolegend); anti-CD19 Brilliant Violet 785 (clone HIB19; Biolegend); anti-CD56 Brilliant Violet 605 (clone HCD56, Biolegend); anti-CD57 eF450 (clone TB01; eBioscience, San Diego, CA); anti-NKG2A APC (clone Z199; Beckman Coulter, Brea, CA), anti-NKG2C AF700 (clone 134591; R&D Systems, Minneapolis, MN); anti-KIR2DL1 FITC (clone 143211; R&D Systems), anti-KIR2DL1/S1 PE-Cy7 (clone EB6B; Beckman Coulter), biotinylated anti-KIR3DL1 (clone DX9; Biolegend), anti-KIR3DL1/S1 PE (clone Z27.3.7; Beckman Coulter), anti-KIR2DL2/L3/S2 PE-Cy5.5 (clone GL183; Beckman Coulter), anti-KIR2DL3/S2 FITC (clone 1F12, kindly provided by Etablissement Français du Sang, Nantes, not commercially available)18; anti-KIR2DL3 PE (clone 180701; R&D Systems); anti-KIR2DS4 PE-Vio770 (clone JJC11.6; Miltenyi Biotec, Bergisch-Gladbach), anti-IFNγ FITC (clone B27; BD Biosciences, Franklin Lakes, NJ); anti-CD107a PE (clone H4A3; BD Biosciences); 4′,6-diamidin-2-phenylindol and streptavidin PerCP-Cy5.5 (Biolegend).
Human FcR-blocking reagent (Miltenyi Biotec) improved staining specificity. Unstained controls were used to adjust for background fluorescence. Compensation controls were performed on PBMC. The viability dye 4′,6-diamidin-2-phenylindol was used to gate out dead cells.
Intracellular staining was performed after cell permeabilization using fixation buffer and permeabilization wash buffer (both Biolegend), according to the manufacturer's protocol. Samples were acquired with a BD LSR Fortessa 18-color flow cytometer (BD Bioscience). Results were analyzed using FlowJo version 10 software (Tree Star, Ashland, OR). All analyses of NK cell subpopulations were performed on PBMC. Natural killer cells were defined as the CD3-CD14-CD19-CD56+ population. Phenotypical analyses of NKG2C, CD57 and different KIRs after CMV infection were performed on a mature subset of CD56dimNKG2A− NK cells (see Figure 1 for gating strategy). Additionally, we analyzed phenotypical and functional changes on CD56dimNKG2A-NKG2C + CD57+ cells, since these cells have been described to represent a CMV imprinted NK cell subset.
FIGURE 1.
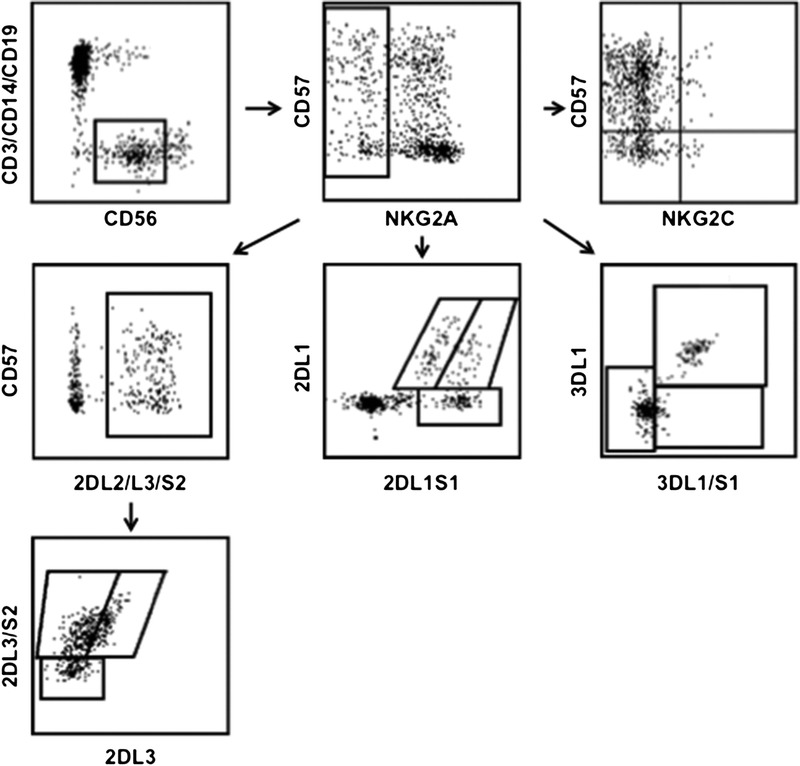
Gating strategy.
Functional Assays
To investigate virus-induced NK cell activation, wild-type HCMV infected MRC-5 cells were seeded at 104 cells/well in flat-bottom 96-well plates. Uninfected MRC-5 cells were seeded as controls. After 2 days, infection was confirmed by qualitatively assessing the cytopathic effect using light microscopy. Peripheral blood mononuclear cells were added at 3 × 105 cells per well. Coculture with uninfected MRC-5 served as a negative control. After an incubation time of 24 hours, 3 × 104 infected and uninfected MRC-5 cells, respectively, were added to boost NK cell activation. Brefeldin A (BD Bioscience) was added after 1 hour. After 6 hours, cells were harvested, stained, and analyzed by flow cytometry. Peripheral blood mononuclear cells incubated with uninfected MRC-5 cells determined the background level of activation.
To investigate leukemia-induced NK cell activation by missing-self HLA, K562 cells were seeded at 105 cells/well in round-bottom 96-well plates. Peripheral blood mononuclear cells were added at 5 × 105 per well. Brefeldin A (BD Bioscience) was added after 1 hour. After 6 hours, cells were harvested, stained, and analyzed by flow cytometry. Peripheral blood mononuclear cells incubated without target cells estimated background degranulation. All functional assays were performed with full PBMC.
HLA Typing
Serological HLA typing results for HLA-A and -B were obtained from STCS.
For typing of HLA-C, genomic DNA of PBMCs was isolated from 200ul of whole blood using a commercial kit (QIAamp DNA kit QIAGEN). The presence of HLA-C1 and -C2 public epitopes was assessed using published protocols.19,20
Statistical Analysis
Phenotypic parameters as well as degranulation and IFNγ expression in total NK cells and NK cell subsets were compared between groups by generalized linear model analysis (IBM SPSS Statistics). All P values presented are 2-sided and considered significant if less than 0.05.
RESULTS
Patient Characteristics and Sample Size
Forty-seven CMV seronegative patients receiving a CMV seropositive kidney graft and 25 CMV seronegative patients receiving a CMV seronegative kidney graft were included in the study. All 47 patients in the first group (to be further delineated as CMV group) experienced a CMV primary infection as measured by viral PCR or CMV antigenemia within 6 months posttransplant, whereas none of the 25 patients in the second group (to be further delineated as control group) had measurable CMV replication. Cytomegalovirus replication in the CMV group occurred after a median of 78 days (range, 30-172 days).
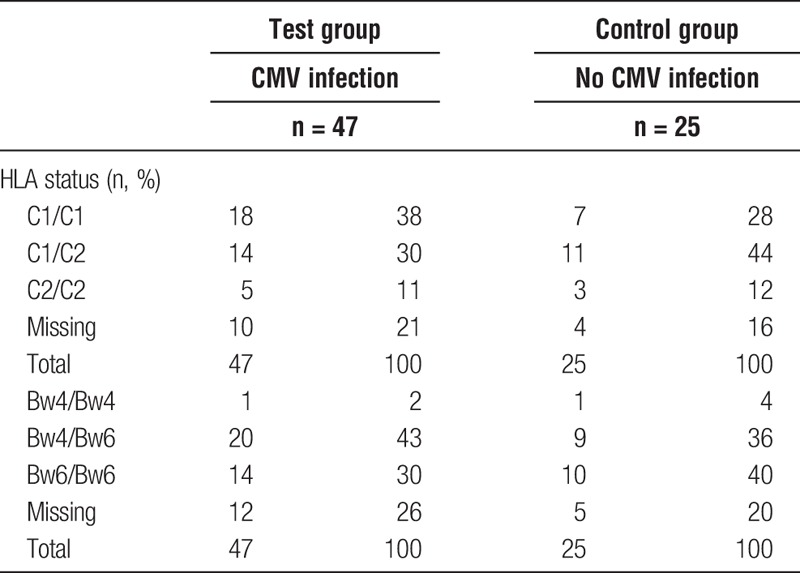
Patient pretransplant and treatment characteristics including age at transplant, sex, etiology of kidney failure, induction therapy, and immunosuppressive agents are depicted in Table 1, patient KIR ligand status is depicted in Table 2.
TABLE 1.
Patient baseline and treatment characteristics
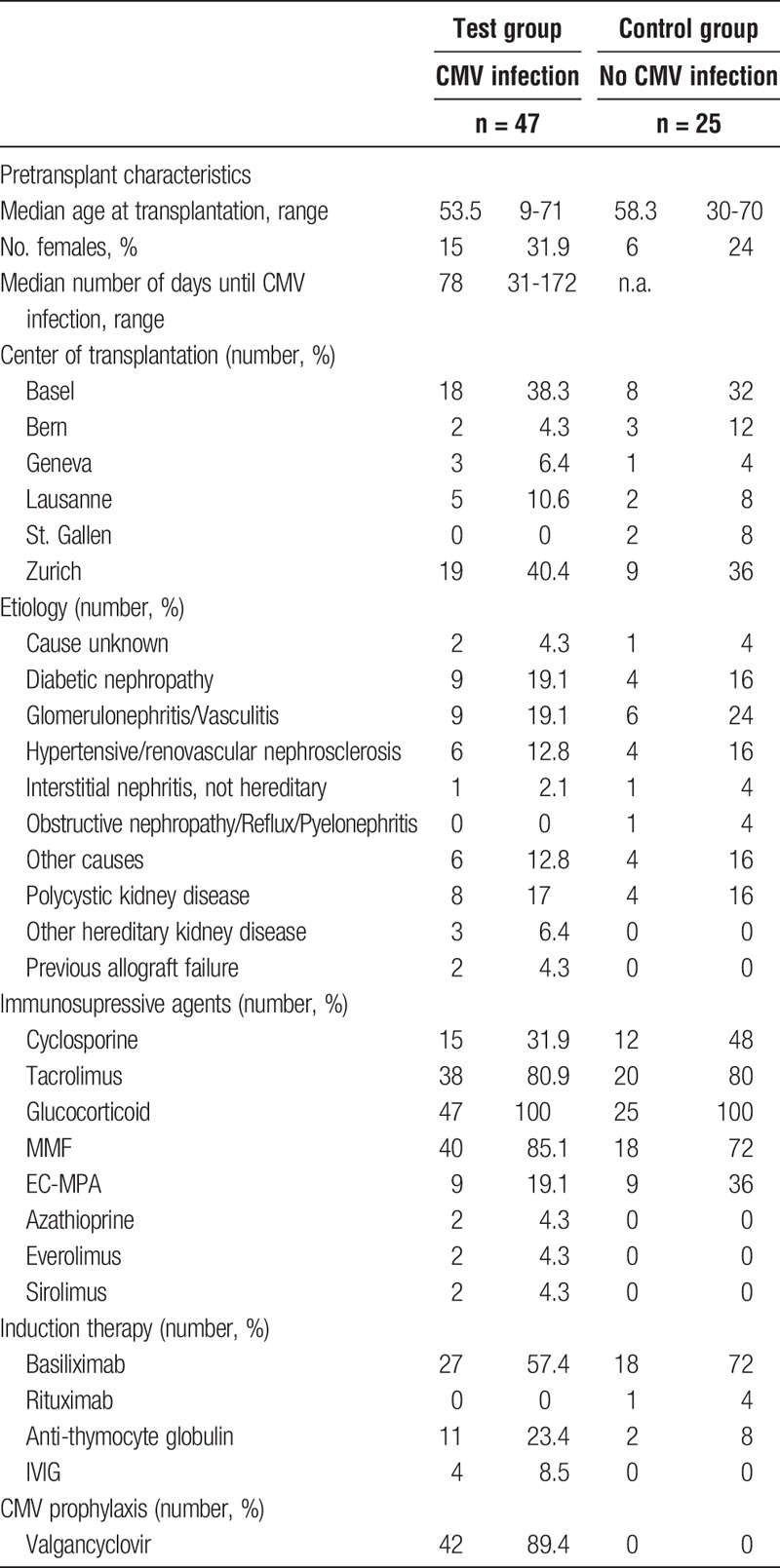
TABLE 2.
Patient HLA typing for C1/C2, Bw4/Bw6
Patient samples were obtained from all 5 kidney transplant centers in Switzerland (Basel, Bern, Geneva, Lausanne, St. Gallen, Zurich). Transplantation was performed between 2008 and 2012.
There was no routine monitoring for viral infections other than CMV.
Due to low cell numbers in some frozen PBMC samples after thawing, not all analyses could be performed on each sample.
Thus, for phenotypical analysis, 42 of 47 samples in the CMV group, and 22 of 25 samples in the control group were evaluable, for the K562 assay, 40 samples in the CMV group and 18 samples in the control group and for the MRC-5 assay, 27 samples in the CMV group and 15 samples in the control group were evaluable.
CMV Infection Promotes NKG2C But Not CD57 Expression
Expansion of NKG2C+ NK cells within the CD56dim subset after CMV infection is well established.7,21,22 CD57 defines a subset of mature NK cells,23-25 but it is less well known if there is an expansion of CD57+ NK cells driven by CMV infection.
To investigate this issue, we assessed NKG2C and CD57 expression before and after HCMV infection.
In line with published data, we observed a significant increase of NKG2C expressing NK cells after CMV primary infection compared to the control group (mean increase, 17.5%; 95% confidence interval [95% CI], 10.2-24.9; P < 0.001; Figure 2A). Interestingly, an at least 2-fold increase could be observed in a majority of patients (33 of 42, 79%). We also observed a slight but statistically nonsignificant decrease of the NKG2A expressing fraction (mean decrease, 6.6%; 95% CI, −3.0 to 16.2; P = 0.18, data not shown).
FIGURE 2.
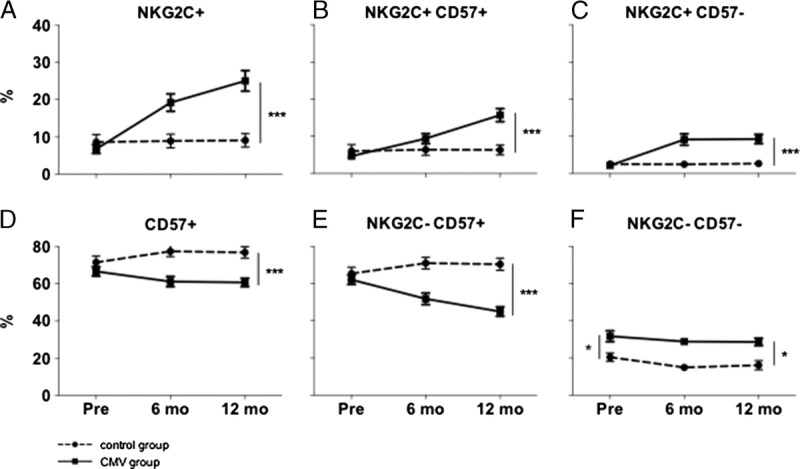
Changes in expression of different surface molecules. Full lines depict CMV group (n = 42), dotted lines depict control group (n = 22). Numbers are percent of all CD56dim NKG2A− NK cells. A, NKG2C expressing NK cells increase significantly after CMV infection. B, NKG2C and CD57 coexpressing cells increase in the CMV group, the same is true for NKG2C+CD57− cells (C). D, Decrease in CD57 expressing cells after CMV infection. E, The increase of NKG2C+ cells is going at the expense of the CD57+NKG2C− subpopulation, which decreases after CMV infection. F, A significant difference between control group and CMV group in the NKG2C−CD57− fraction is observed. Since this can already be observed before CMV infection, this difference cannot be attributed to CMV infection.
Of note, there was a relative increase of not only NKG2C+CD57+ cells (mean increase, 10.5%; 95% CI, 5.8-15.1; P < 0.001) but also of NKG2C+CD57− cells (mean increase, 7.4%; 95% CI, 4.2-10.5; P < 0.001; Figures 2B-C), speaking against the hypothesis, that expanded NKG2C+ cells preferentially express CD57. This relative increase of NKG2C+CD57+ and NKG2C+CD57− NK cell fractions came at the expense of the CD57+NKG2C− subset (mean decrease, 24.1%; 95% CI, 17.7-30.6; P < 0.001) but not the CD57−NKG2C− subpopulation (Figures 2E-F).
Surprisingly, total CD57 expression significantly decreased (mean decrease, 14.1%; 95% CI, 8.0-20.2; P < 0.001; Figure 2D).
CMV Promotes Expansion of NK Cells Carrying the Self-Ligand–Specific KIR2DL1
Next, we investigated the impact of CMV on KIR expression. According to published data, we expected CMV to trigger an expansion of mature NK cells expressing self-specific inhibitory KIR as well as activating KIR, with a concurrent decrease of KIR-negative cells.
Intriguingly, we found a significant increase of cells expressing 2DL1 as the only inhibitory KIR (negative for 2DL2, 2DL3, 3DL1) in the presence of the cognate ligand HLA-C2 after CMV infection (C2/C2 or C1/C2: mean increase, 10.0%; 95% CI, 1.7-18.3; P = 0.018; Figure 3B). In contrast, in the absence of the ligand, there was a significant decrease of 2DL1 single positive cells (C1/C1: mean decrease, 3.9%; 95% CI, 1.6-6.2; P = 0.001; Figure 3C). When looking at overall 2DL1 expression on NK cells, results were similar (C2/C2 or C1/C2: mean increase, 9.8%; 95% CI, −1.6 to 21.2; P = 0.093; C1/C1: mean decrease, 12.1%; 95% CI, 7.7-16.6; P < 0.001).
FIGURE 3.
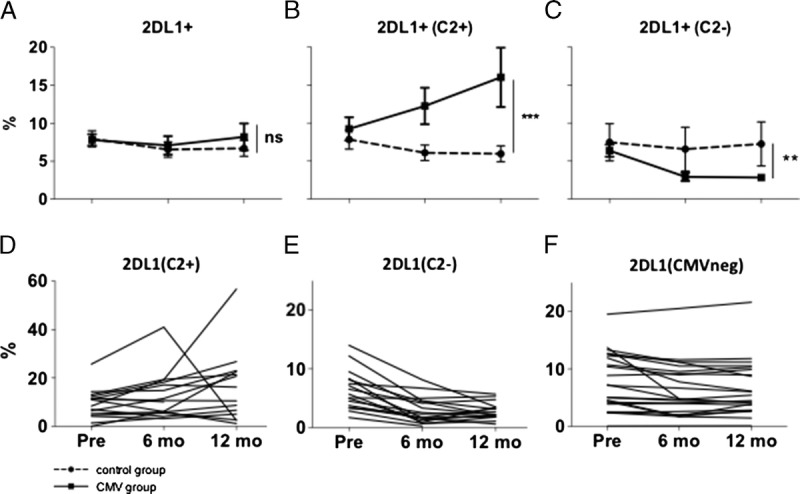
Changes in surface expression of 2DL1. Full lines depict CMV group (n = 42), dotted lines depict control group (n = 22). Numbers are percent of all CD56dim NKG2A− NK cells. A, Overall, cells expressing KIR2DL1 as the only inhibitory KIR do not change after CMV infection compared to the control group. B, In the presence of the cognate ligand HLA C2, there is a significant increase in 2DL1 expressing cells, whereas in absence of the cognate ligand, 2DL1 expressing cells decrease (C). D-F, Pattern of change in 2DL1 expression in single patients. D, CMV group, HLA-C2 positive samples (n = 16). E, CMV group, HLA-C2 negative samples (n = 17). F, Control group (n = 22).
When looking at 2DL1 single positivity on a CMV-imprinted subset of NKG2C+CD57+ cells, results were also similar with a significant increase in 2DL1 expression in the presence of the cognate ligand (mean increase, 15.9%; 95% CI, 3.4-28.3; P = 0.013).
KIR2DL2 expressing cells increased after CMV infection compared to the control population (mean increase, 6.4%; 95% CI, 0.6-12.3; P = 0.03; Figure 1, SDC, http://links.lww.com/TXD/A27). KIR2DL2 is known to be licensed by HLA-C1 as well as HLA-C2. Compatible with this characteristic, we did not find a difference in 2DL2 expression according to HLA status.
We observed no significant change in 2DL3 expressing cells after CMV infection compared with the control group (mean increase, 1.3%; 95% CI, −4.6 to 7.2, P = ns; Figure. 1, SDC, http://links.lww.com/TXD/A27), independent of the presence or absence of the cognate ligand HLA-C1. Sample size with HLA-C2/C2 status might have been too low to detect a significant difference to HLA-C1 positive individuals (compare Table 2). Again, we looked at 2DL2 and 2DL3 expressions on the CMV-imprinted population of NKG2C+CD57+ cells and found similar results to the overall NK cell population.
KIR3DL1 expression decreased upon CMV infection compared to the control group—this was true independently of the presence of the cognate ligand HLA-Bw4 (mean decrease, 8.1%; 95% CI, 4.3-11.9; P < 0.001).
Results were similar when looking at 3DL1 single positive cells (negative for 2DL1, 2DL2, 2DL3: mean decrease, 3.1%; 95% CI, 1.7-4.7; P < 0.001; Figure 1, SDC, http://links.lww.com/TXD/A27, Bw4 pos: mean decrease, 2.7%; 95% CI, −0.2 to 5.6; P = 0.069; Bw4neg: mean decrease, 4.3%; 95% CI, 2.0-6.6; P < 0.001) and when investigating the NKG2C+CD57+ population (mean decrease, 5.2%; 95% CI, 1.0-9.3; P = 0.016; Bw4pos: mean decrease, 7.7%; 95% CI, 0.6-14.7; P = 0.033; Bw4neg: mean decrease, 4.5%; 95% CI, −1.5 to 10.4; P = 0.14).
No Expansion of aKIR+ NK Cells After CMV Infection Is Observed
Previous studies found an inverse correlation between the number of activating KIR genes in the donor and the CMV replication risk after hematopoietic stem cell transplant.26-28 The same effect has been shown in kidney transplant recipients.29-31
The question has also been raised, weather CMV replication influences expression of activating KIR posttransplant. One study demonstrated an elevated expression of aKIR (ie, KIR2DS2 and KIR2DS4) after HSCT compared with donor expression before HSCT, above all in CMV viremic compared with CMV nonviremic HSCT recipients.32
In contrast to these observations, we did not find an increase in aKIR expression after CMV infection in our study setting.
We found a small statistically nonsignificant decrease of 2DS1 expressing cells upon CMV infection (mean decrease after CMV infection compared with control group, 1.1%; 95% CI, −3.3 to 5.4; P = 0.63; Figure 4A). Interestingly, this decrease was more pronounced in the presence of the cognate ligand HLA-C2, leading to a significant difference between groups (C2/C2 or C1/C2: mean decrease, 5.6%; 95% CI, 2.6-8.7; P < 0.001; C1/C1: mean decrease, 0.2%; 95% CI, −9.0 to 9.3; P = 0.97, data not shown).
FIGURE 4.
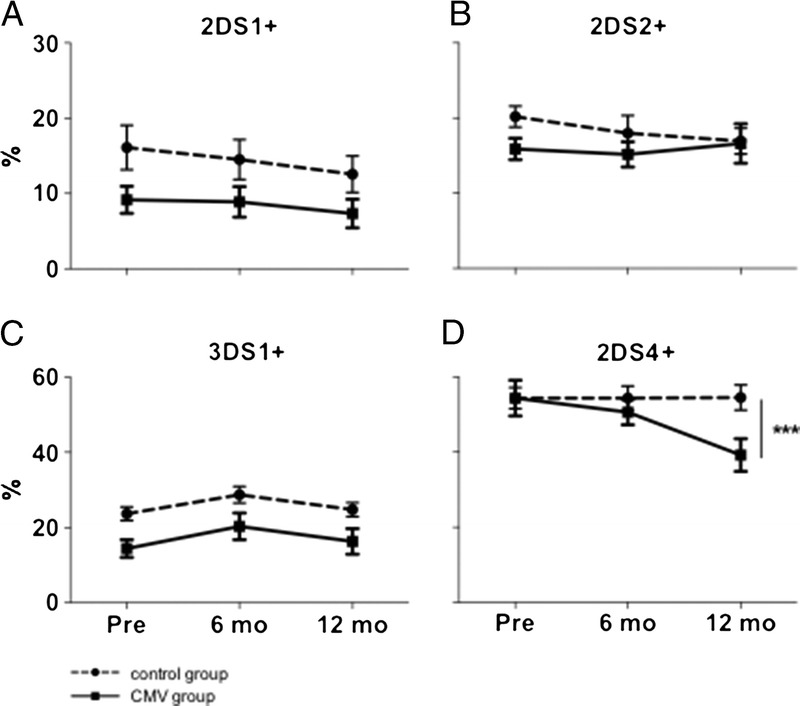
Changes in surface expression of different activating KIR. Full lines depict CMV group (n = 42), dotted lines depict control group (n = 22). A-C, No significant changes in subpopulations expressing KIR2DS1, 2DS2 or 3DS1 respectively. D, The subpopulation expressing 2DS4 decreases significantly after CMV infection.
No significant change in the expression of 3DS1 and 2DS2 on CMV infection could be observed (Figures 4B-C).
As for KIR2DS4, we found a marked decrease in 2DS4 expressing cells after CMV infection compared with the control group (mean decrease, 16.3%; 95% CI, 5.1-27.4; P = 0.004, Figure 4D).
When looking at a CMV imprinted, NKG2C+CD57+ subset, results were comparable to the overall population, with no significant change for 2DS2 and 3DS1, but a more pronounced decrease of 2DS1 expressing cells in the presence of the ligand (mean decrease, 16.4%; 95% CI, 6.3-26.4; P = 0.001) and of 2DS4-expressing cells (mean decrease, 13.6%; 95% CI, 2.2-25.1; P = 0.019).
CMV Leads to Increased In Vitro Effector Function Against Tumor Targets
To address changes in NK cell function after CMV infection, we performed coculture experiments with 2 different targets.
First, we investigated whether CMV infection leads to an increased NK cell function in vitro on stimulation with HLA-negative tumor cells. We observed a significantly higher NK cell degranulation (CD107a surface expression: mean increase, 9.9%; 95% CI, 4.8-15.0; P < 0.001) and IFNγ production (mean increase, 6.6%; 95% CI, 1.6-11.1; P < 0.001) in response to K562 after CMV infection compared with the control group. No significant differences were seen between NKG2C-positive and NKG2C-negative subsets (Figures 5 and 6).
FIGURE 5.
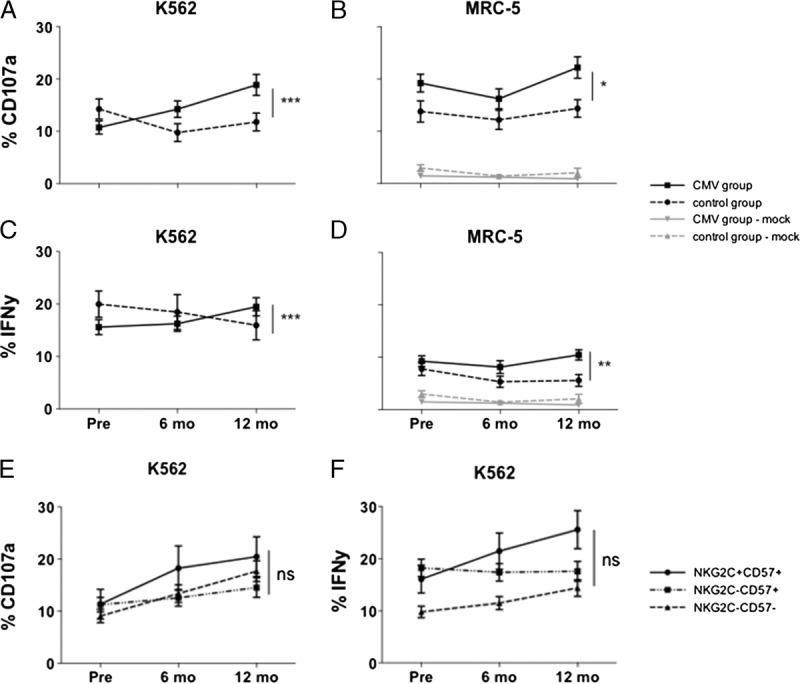
Changes in degranulation (CD107a surface expression) and IFNγ release upon coculture with different target cells. Analyses are performed on CD56dimNKG2A− NK cells. Full lines depict CMV group (n = 40/27 for K562/MRC-5, respectively), dotted lines depict control group (n = 18/15 for K562/MRC-5 respectively). A and C, Increased degranulation (A) and IFNγ release (C) on coculture with the tumor target cell line K562 after CMV infection compared with the control group. B and D, Increased degranulation (B) and IFNγ release (D) on coculture with the CMV-infected fibroblast cell line MRC-5 after CMV infection. Black lines show netto effect (activation after coculture with uninfected MRC-5 cells has been subtracted). Grey lines show NK cell activation after coculture with uninfected MRC-5 cells. E and F) No difference after CMV infection between single NK cell subpopulations in degranulation (E) and IFNγ release (F) upon coculture with the tumor target cell line K562; increased function can be observed in all subpopulations (exception: no increase in IFNγ release in the CD57+G2C− subset after CMV infection).
FIGURE 6.
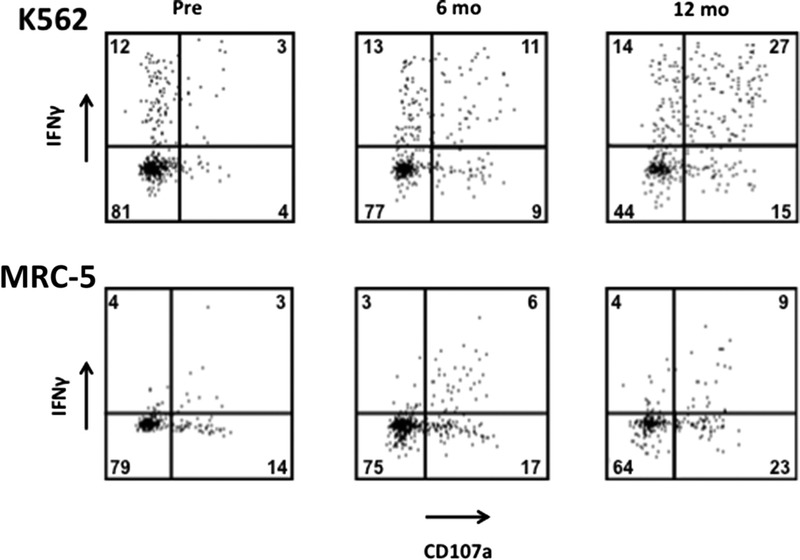
Example of a representative patient (patient 14) with increasing in vitro degranulation and IFNγ release after CMV infection. Numbers are percentages of all CD56dim NKG2A− NK cells.
CMV Leads to Increased In Vitro Effector Function Against Virus-Infected Target
On coculture with the CMV-infected target cell line MRC-5, we also observed an increased degranulation (mean increase of CD107a surface expression, 6.9%; 95% CI, 0.7-13.1; P = 0.03) and IFNγ production (mean increase, 4.8%; 95% CI, 1.7-7.8; P = 0.002), but the changes were less distinct than upon coculture with K562 cells (Figures 5 and 6).
Because induction therapy was different between groups with most patients receiving either basiliximab (57% in the CMV group, 72% in the control group) or anti-thymocyte globulin (23% in the CMV group, 8% in the control group), we analyzed NK cell phenotype and activity according to induction therapy regimen, but no difference could be detected (data not shown). The immunosuppressive regimen was identical for a far majority of patients included, with 92% in the CMV group and 100% in the control group receiving a combination of glucocorticoids, a calcineurin inhibitor, and mycophenolate.
DISCUSSION
Using serial analyses of kidney transplant recipients as a model for primary in vivo CMV infection, we observed relevant phenotypical and functional changes in the NK cell compartment. In line with previous results and with the prevalent understanding of the CMV driven imprinting process,6-9,11,21,22 we found an increase in NKG2C+ NK cells. The functional properties of these NK cells in terms of antiviral and antitumor immunity have, however, remained elusive. In the hematopoietic stem cell transplant setting, Foley and colleagues were able to demonstrate a robust IFNγ production of NKG2C + KIR+ NK cells during CMV infection, but no increase in degranulation upon stimulation with K562 cells.
In the present study, we were now able to demonstrate an enhanced degranulation as well as IFNγ production of NK cells induced by CMV infection compared to the CMV naïve situation, above all when using a HLA-negative leukemia target cell, but also—albeit to a lesser degree—when using a CMV-infected target.
Intriguingly, and in contrast to previously published data, these changes could be seen in the overall CD56dimNKG2A− NK cell population, and, when looking at subpopulations defined by NKG2C, they were not restricted to NKG2C+ subsets but could also be observed in NKG2C− NK cells. Together with the fact that the tumor target cell line K562 does not express the NKG2C ligand HLA-E, this argues for an NKG2C independent mechanism of action.
Upregulation of NKG2C on NK cells is only one of many different CMV-driven changes on the NK cell compartment, but does not seem to be causative for the increased activity we observe in our study setting. Other groups were already able to conclusively demonstrate the occurrence of CMV-induced changes on NK cell repertoire in the absence of NKG2C expression.33,34
Finally, we cannot rule out that other immune players such as T cells or monocytes interacting with NK cells might enhance these observed functional changes, to augment functional capacity of NK cells, because we performed functional analyses on PBMC and not purified NK cells.
Rölle and colleague35s were able to demonstrate in vitro that the expansion of NKG2C+ NK cells in HCMV relies on crosstalk of NK cells with monocytes, involving IL-12 and being dependent on the interaction with HLA-E on infected cells. In that study, no data on the functional capacity of NK cell subsets was given, so it can only be speculated that analogous mechanisms, involving crosstalk between NK cells and monocytes and dendritic cells might not only trigger phenotypical changes but could also play a role to exert increased functional capacity of NK cells. A recently published study by Wu and colleagues36 demonstrated that the CMV-induced enhanced antiviral activity of NK cells is dependent on IL-2 secreted by virus-specific CD4+ T cells, stressing on the importance of the adaptive immune system on NK cell-mediated immunity. Consistent with our findings, the authors provided some evidence that this increased NK cell activity was not mediated by NKG2C+ cells.
In line with previously published findings from our group,37 we observed an expansion of NK cells expressing the self-specific inhibitory KIR2DL1 on CMV infection and now could also demonstrate a decrease of 2DL1 expressing cells in absence of the ligand. The same observation could not be made for the other inhibitory KIR with known ligands KIR2DL2, 2DL3, and 3DL1, the latter was even decreasing after CMV infection independently of presence or absence of the cognate ligand. Since 2DL2 is known to bind HLA-C1 and -C2, this might explain why we did not observe a change according to ligand status. Moreover, the study included only few HLA-C2-homozygous controls, what might have precluded the detection of significant findings for 2DL3, since the C1-negative control group was too small.
Lopez-Vergès and colleagues9 described a series of solid organ transplant recipients with CMV replication, displaying an increased frequency of CD57+ NK cells within both NKG2C+ and NKG2C− subsets, with NKG2C+ NK cells being predominantly CD57− just after the detection of viremia, and over time increasing the amount of NKG2C on the cell surface and acquiring CD57 expression. In our study setting, we detected a relative increase of not only NKG2C+CD57+ cells but also of NKG2C+CD57− cells, speaking against the hypothesis of expanded NKG2C+ cells preferentially expressing CD57. Moreover, total CD57+ cells were decreasing. Because we did not dissect the evolution of NKG2C+/bright cells in the short course of acute infection, results are not directly comparable.
Similar to our findings of a decrease of 3DL1 expressing cells in the overall and NKG2C+ NK cell population, the authors stated a preferential decrease of 3DL1 on CD57+NKG2C+ cells in HLA-Bw4–positive donors and also in a minority of Bw4 negative donors. An explanation for the somewhat diverging results might be that changes in 3DL1 expression precede increase in NKG2C (and possibly CD57) expression. This theory might be reinforced by findings of De Rham and colleagues38 who also studied kidney transplantation recipients with CMV replication and found an increase in 3DL1-expressing cells right at the acute phase of the infection with a rapid fall afterward, also providing some in vitro evidence that these cells might play a role in CMV control.
It has to be pointed out, that antibody-mediated response of NK cells after CMV infection was not examined in our study setting. Costa-Garcia and colleagues39 recently reported a significant antibody-mediated increase in degranulation and cytokine production of NK cells against CMV-infected fibroblast, particularly but not only in NKG2C-positive cells. Even though we cannot comment on antibody-dependent cellular cytotoxicity, we provide evidence for increased direct, antibody-independent cellular cytotoxicity of NK cells.
In our setting, the number of patients exhibiting an at least 2-fold expansion of NKG2C-positive NK cells was substantial (33 of 42 patients, 79%) and was persisting 1 year after transplantation. When comparing CMV seropositive to CMV seronegative healthy donors, an expanded NKG2C-positive NK cell population can be found in a much lower percentage, arguing for the fact that this expanded NK cell subset does persist throughout life in many individuals, but is lost in other individuals, probably depending on the time lag from CMV primary infection/reactivation.
The fact that NKG2C expressing cells increased by a mean of 17.5% after CMV infection, NKG2A expressing cells decreased by only 6.6%, and there was no increase in double positive cells argues for a CMV driven emergence of NKG2C+ cells from the NKG2A−NKG2C–negative subset.
It finally has to be mentioned, that patients in the CMV group underwent prophylactic therapy with valganciclovir as well as ganciclovir treatment for CMV disease, while patients in the control group received none of these. These medications might have had an impact on NK cells. Ganciclovir has been shown to inhibit proliferation of lymphocytes in vitro by impairing DNA synthesis,40-42 an issue which has not been studied specifically for NK cells. Nevertheless, an antiproliferative effect can be assumed for NK cells just as for other lymphocyte types but should not be responsible for the specific changes observed in our study.
To summarize, we are able to demonstrate an increased function of NK cells induced by CMV infection, above all but not only against tumor cells and not restricted to cells expressing the activating receptor NKG2C.
Supplementary Material
ACKNOWLEDGMENTS
The authors thank Karin Schmitter, Jasmin Grählert, Lukas Christen and the Flow Team DBM for important technical assistance.
Footnotes
Published online 17 June 2016.
The members of the Swiss Transplant Cohort Study are: Rita Achermann, Patrizia Amico, John-David Aubert, Philippe Baumann, Guido Beldi, Christian Benden, Christoph Berger, Isabelle Binet, Pierre-Yves Bochud, Elsa Boely, Heiner Bucher, Leo Bühler, Thierry Carell, Emmanuelle Catana, Yves Chalandon, Sabina de Geest, Olivier de Rougemont, Michael Dickenmann, Michel Duchosal, Thomas Fehr, Sylvie Ferrari-Lacraz, Christian Garzoni, Paola Gasche Soccal, Emiliano Giostra, Déla Golshayan, Daniel Good, Karine Hadaya, Jörg Halter, Dominik Heim, Christoph Hess, Sven Hillinger, Hans H. Hirsch, Günther Hofbauer, Uyen Huynh-Do, Franz Immer, Richard Klaghofer, Michael Koller (Head of the data center), Bettina Laesser, Roger Lehmann, Christian Lovis, Oriol Manuel, Hans-Peter Marti, Pierre Yves Martin, Luca Martinolli, Pascal Meylan, (Head, Biological samples management group), Paul Mohacsi, Isabelle Morard, Philippe Morel, Ulrike Mueller, Nicolas J Mueller (Chairman Scientific Committee), Helen Mueller-McKenna (Head of local data management), Antonia Müller, Thomas Müller, Beat Müllhaupt, David Nadal, Manuel Pascual (Executive office), Jakob Passweg, Chantal Piot Ziegler, Juliane Rick, Eddy Roosnek, Anne Rosselet, Silvia Rothlin, Frank Ruschitzka, Urs Schanz, Stefan Schaub, Christian Seiler, Susanne Stampf, Jürg Steiger (Head, Executive Office), Guido Stirnimann, Christian Toso, Dimitri Tsinalis, Christian Van Delden (Executive office), Jean-Pierre Venetz, Jean Villard, Madeleine Wick (STCS coordinator), Markus Wilhelm, Patrick Yerly.
This work was supported by a grant from the University Basel (grant DMS2267 to ATP), grants from the Swiss National Science Foundation (grant PPOOP3 128461/1 to MS), furthermore by Nora van Meeuwen-Häfliger Stiftung, Freiwillige Akademische Gesellschaft Basel and Stiftung für Hämatologische Forschung Basel.
Patient data and PBMC samples were provided by the Swiss Transplant Cohort Study (STCS). This study has been conducted in the framework of the Swiss Transplant Cohort Study, supported by the Swiss National Science Foundation and the Swiss University Hospitals (G15) and transplant centers.
The authors declare no conflicts of interest.
G.T., M.S., A.T.P., and L.S. designed research. A.T.P. performed experiments and analyses and wrote the article. H.H.H., C.G., C.v.D., K.B., N.J.M., C.B., J.V., O.M., and P.M. provided patient samples and clinical data. C.R. developed and provided crucial materials. All authors edited the article.
Supplemental digital content (SDC) is available for this article. Direct URL citations appear in the printed text, and links to the digital files are provided in the HTML text of this article on the journal’s Web site (www.transplantationdirect.com).
Contributor Information
Collaborators: Swiss Transplant Cohort Study
REFERENCES
- 1.Nachbaur D, Clausen J, Kircher B. Donor cytomegalovirus seropositivity and the risk of leukemic relapse after reduced-intensity transplants. Eur J Haematol. 2006;76:414–419. [DOI] [PubMed] [Google Scholar]
- 2.Ito S, Pophali P, Co W, et al. CMV reactivation is associated with a lower incidence of relapse after allo-SCT for CML. Bone Marrow Transplant. 2013;48:1313–1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Elmaagacli AH, Steckel NK, Koldehoff M, et al. Early human cytomegalovirus replication after transplantation is associated with a decreased relapse risk: evidence for a putative virus-versus-leukemia effect in acute myeloid leukemia patients. Blood. 2011;118:1402–1412. [DOI] [PubMed] [Google Scholar]
- 4.Green ML, Leisenring WM, Xie H, et al. CMV reactivation after allogeneic HCT and relapse risk: evidence for early protection in acute myeloid leukemia. Transplantation. 2013;122:1316–1324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Muntasell A, Vilches C, Angulo A, et al. Adaptive reconfiguration of the human NK-cell compartment in response to cytomegalovirus: a different perspective of the host-pathogen interaction. Eur J Immunol. 2013;43:1133–1141. [DOI] [PubMed] [Google Scholar]
- 6.Béziat V, Dalgard O, Asselah T, et al. CMV drives clonal expansion of NKG2C + NK cells expressing self-specific KIRs in chronic hepatitis patients. Eur J Immunol. 2012;42:447–457. [DOI] [PubMed] [Google Scholar]
- 7.Foley B, Cooley S, Verneris MR, et al. Cytomegalovirus reactivation after allogeneic transplantation promotes a lasting increase in educated NKG2C + natural killer cells with potent function. Blood. 2012;119:2665–2674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Foley B, Cooley S, Verneris MR, et al. Human cytomegalovirus (CMV)-induced memory-like NKG2C(+) NK cells are transplantable and expand in vivo in response to recipient CMV antigen. J Immunol. 2012;189:5082–5088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lopez-Vergès S, Milush JM, Schwartz BS, et al. Expansion of a unique CD57+NKG2Chi natural killer cell subset during acute human cytomegalovirus infection. Proc Natl Acad Sci U S A. 2011;108:14725–14732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Petersen L, Roug AS, Skovbo A, et al. The CD94/NKG2C-expressing NK cell subset is augmented in chronic lymphocytic leukemia patients with positive human cytomegalovirus serostatus. Viral Immunol. 2009;22:333–337. [DOI] [PubMed] [Google Scholar]
- 11.Gumá M, Cabrera C, Erkizia I, et al. Human cytomegalovirus infection is associated with increased proportions of NK cells that express the CD94/NKG2C receptor in aviremic HIV-1-positive patients. J Infect Dis. 2006;194:38–41. [DOI] [PubMed] [Google Scholar]
- 12.Sáez-Borderías A, Romo N, Magri G, et al. IL-12-dependent inducible expression of the CD94/NKG2A inhibitory receptor regulates CD94/NKG2C+ NK cell function. J Immunol. 2009;182:829–836. [DOI] [PubMed] [Google Scholar]
- 13.Monsiváis-Urenda A, Noyola-Cherpitel D, Hernández-Salinas A, et al. Influence of human cytomegalovirus infection on the NK cell receptor repertoire in children. Eur J Immunol. 2010;40:1418–1427. [DOI] [PubMed] [Google Scholar]
- 14.Zhang T, Scott JM, Hwang I, et al. Cutting edge: antibody-dependent memory-like NK cells distinguished by FcR deficiency. J Immunol. 2013;190:1402–1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Béziat V, Liu LL, Malmberg JA, et al. NK cell responses to cytomegalovirus infection lead to stable imprints in the human KIR repertoire and involve activating KIRs. Blood. 2013;121:2678–2688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cichocki F, Cooley S, Davis Z, et al. CD56dimCD57 + NKG2C+ NK cell expansion is associated with reduced leukemia relapse after reduced intensity HCT. Leukemia. 2015; accepted a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Della Chiesa M, Falco M, Podestà M, et al. Phenotypic and functional heterogeneity of human NK cells developing after umbilical cord blood transplantation: a role for human cytomegalovirus? Blood. 2012;119:399–410. [DOI] [PubMed] [Google Scholar]
- 18.David G, Morvan M, Gagne K, et al. Discrimination between the main activating and inhibitory killer cell immunoglobulin-like receptor positive natural killer cell subsets using newly characterized monoclonal antibodies. Immunology. 2009;128:172–184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Frohn C, Schlenke P, Ebel B, et al. DNA typing for natural killer cell inhibiting HLA-Cw groups NK1 and NK2 by PCR-SSP. J Immunol Methods. 1998;218:155–160. [DOI] [PubMed] [Google Scholar]
- 20.Koehler RN, Walsh AM, Moqueet N, et al. High-throughput genotyping of KIR2DL2/L3, KIR3DL1/S1, and their HLA class I ligands using real-time PCR. Tissue Antigens. 2009;74:73–80. [DOI] [PubMed] [Google Scholar]
- 21.Guma M, Budt M, Saez A, et al. Expansion of CD94/NKG2C+ NK cells in response to human cytomegalovirus-infected fibroblasts. Blood. 2006;107:3624–3631. [DOI] [PubMed] [Google Scholar]
- 22.Guma M, Angulo A, Vilches C, et al. Imprint of human cytomegalovirus infection on the NK cell receptor repertoire. Blood. 2004;104:3664–3671. [DOI] [PubMed] [Google Scholar]
- 23.Béziat V, Descours B, Parizot C, et al. NK cell terminal differentiation: Correlated stepwise decrease of NKG2A and acquisition of KIRs. PLoS One. 2010;5:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Björkström NK, Riese P, Heuts F, et al. Expression patterns of NKG2A, KIR, and CD57 define a process of CD56dim NK-cell differentiation uncoupled from NK-cell education. Blood. 2010;116:3853–3864. [DOI] [PubMed] [Google Scholar]
- 25.Lopez-Vergès S, Milush JM, Pandey S, et al. CD57 defines a functionally distinct population of mature NK cells in the human CD56dimCD16+ NK-cell subset. Blood. 2010;116:3865–3874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen C, Busson M, Rocha V, et al. Activating KIR genes are associated with CMV reactivation and survival after non-T-cell depleted HLA-identical sibling bone marrow transplantation for malignant disorders. Bone Marrow Transplant. 2006;38:437–444. [DOI] [PubMed] [Google Scholar]
- 27.Cook M, Briggs D, Craddock C, et al. Donor KIR genotype has a major influence on the rate of cytomegalovirus reactivation following T-cell replete stem cell transplantation. Blood. 2006;107:1230–1232. [DOI] [PubMed] [Google Scholar]
- 28.Zaia JA, Sun JY, Gallez-Hawkins GM, et al. The effect of single and combined activating KIR genotypes on CMV infection and immunity after hematopoietic cell transplantation. Biol Blood Marrow Transpl. 2010;15:315–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stern M, Elsässer H, Hönger G, et al. The number of activating KIR genes inversely correlates with the rate of CMV infection/reactivation in kidney transplant recipients. Am J Transplant. 2008;8:1312–1317. [DOI] [PubMed] [Google Scholar]
- 30.Stern M, Hadaya K, Hönger G, et al. Telomeric rather than centromeric activating KIR genes protect from cytomegalovirus infection after kidney transplantation. Am J Transplant. 2011;11:1302–1307. [DOI] [PubMed] [Google Scholar]
- 31.Hadaya K, Avila Y, Valloton L, et al. Natural killer cell receptor—repertoire and functions after induction therapy by polyclonal rabbit anti-thymocyte globulin in unsensitized kidney transplant recipients. Clin Immunol. 2010;137:250–260. [DOI] [PubMed] [Google Scholar]
- 32.Gallez-Hawkins GM, Franck AE, Li X, et al. Expression of activating KIR2DS2 and KIR2DS4 genes after hematopoietic cell transplantation: relevance to cytomegalovirus infection. Biol Blood Marrow Transplant. 2011;17:1662–1672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Della Chiesa M, Falco M, Bertaina A, et al. Human cytomegalovirus infection promotes rapid maturation of NK cells expressing activating killer Ig-like receptor in patients transplanted with NKG2C−/− umbilical cord blood. J Immunol. 2014;192:1471–1479. [DOI] [PubMed] [Google Scholar]
- 34.Liu L, Enqvist M, Sohlberg E, et al. Redundancy of adaptive NK cell responses in the absence of NKG2C and activating KIRs. Abstr NK2015 15th Meet Soc Nat Immunity. [Google Scholar]
- 35.Rölle A, Pollmann J, Ewen E, et al. IL-12-producing monocytes and HLA-E control. J Clin Invest. 2014;124:5305–5316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wu Z, Frascaroli G, Bayer C, et al. Interleukin-2 from adaptive t cells enhances natural killer cell activity against human cytomegalovirus-infected macrophages. J Virol. 2015;89:6435–6441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Charoudeh HN, Terszowski G, Czaja K, et al. Modulation of the natural killer cell KIR repertoire by cytomegalovirus infection. Eur J Immunol. 2013;43:480–487. [DOI] [PubMed] [Google Scholar]
- 38.de Rham C, Hadaya K, Bandelier C, et al. Expression of killer cell immunoglobulin-like receptors (KIRs) by natural killer cells during acute CMV infection after kidney transplantation. Transpl Immunol. 2014;31:157–164. [DOI] [PubMed] [Google Scholar]
- 39.Costa-Garcia M, Vera A, Moraru M, et al. Antibody-mediated response of NKG2Cbright NK cells against human cytomegalovirus. J Immunol. 2015;194:2715–2724. [DOI] [PubMed] [Google Scholar]
- 40.Battiwalla M, Wu Y, Bajwa RPS, et al. Ganciclovir inhibits lymphocyte proliferation by impairing DNA synthesis. Biol Blood Marrow Transplant. 2007;13:765–770. [DOI] [PubMed] [Google Scholar]
- 41.Bowden RA, Digel J, Reed EC, et al. Immunosuppressive effects of ganciclovir on in vitro lymphocyte responses. J Infect Dis. 1987;156:899–903. [DOI] [PubMed] [Google Scholar]
- 42.Heagy W, Crumpacker C, Lopez PA, et al. Inhibition of immune function by antiviral drugs. JClinInvest. 1991;87:1916–1924. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


