Abstract
Aim
To analyse the impact of overweight and obesity on the risk of adverse maternal outcomes and fetal macrosomia in pregnancies of women treated for severe gestational diabetes.
Methods
This was a population‐based cohort study including all singleton pregnancies in Sweden without pre‐existing diabetes in the period 1998–2012. Only mothers with an early‐ pregnancy BMI of ≥ 18.5 kg/m² were included. Logistic regression analysis was used to determine odds ratios with 95% CIs for maternal outcomes and fetal growth. Analyses were stratified by maternal gestational diabetes/non‐gestational diabetes to investigate the impact of overweight/obesity in each group.
Results
Of 1 249 908 singleton births, 13 057 were diagnosed with gestational diabetes (1.0%). Overweight/obesity had the same impact on the risks of caesarean section and fetal macrosomia in pregnancies with and without gestational diabetes, but the impact of maternal BMI on the risk of preeclampsia was less pronounced in women with gestational diabetes. Normal‐weight women with gestational diabetes had an increased risk of caesarean section [odds ratio 1.26 (95% CI 1.16–1.37)], preeclampsia [odds ratio 2.03 (95% CI 1.71–2.41)] and large‐for‐gestational‐age infants [odds ratio 2.25 (95% CI 2.06–2.46)]. Risks were similar in the overweight group without gestational diabetes, caesarean section [odds ratio 1.34 (1.33–1.36)], preeclampsia odds ratio [1.76 (95% CI 1.72–1.81)], large‐for‐gestational‐age [odds ratio 1.76 (95% CI 1.74–1.79)].
Conclusions
Maternal overweight and obesity is associated with similar increments in risks of adverse maternal outcomes and delivery of large‐for‐gestational‐age infants in women with and without gestational diabetes. Obese women with gestational diabetes are defined as a high‐risk group. Normal‐weight women with gestational diabetes have similar risks of adverse outcomes to overweight women without gestational diabetes.
What's new?
Maternal overweight and obesity is associated with similar increments in risks of adverse outcomes and delivery of large‐for‐gestational‐age infants in women with and without gestational diabetes.
Overweight without gestational diabetes has a similar impact on the risk of maternal outcomes, such as preeclampsia, caesarean section and fetal macrosomia, as does gestational diabetes in women of normal weight.
Obese women without gestational diabetes have higher rates of caesarean section, preeclampsia and macrosomia than normal‐weight women with gestational diabetes.
Despite treatment, women with gestational diabetes still face excess risks of adverse pregnancy outcomes.
What's new?
Maternal overweight and obesity is associated with similar increments in risks of adverse outcomes and delivery of large‐for‐gestational‐age infants in women with and without gestational diabetes.
Overweight without gestational diabetes has a similar impact on the risk of maternal outcomes, such as preeclampsia, caesarean section and fetal macrosomia, as does gestational diabetes in women of normal weight.
Obese women without gestational diabetes have higher rates of caesarean section, preeclampsia and macrosomia than normal‐weight women with gestational diabetes.
Despite treatment, women with gestational diabetes still face excess risks of adverse pregnancy outcomes.
Introduction
The prevalence of overweight and obesity is increasing worldwide and there is evidence to suggest that the incidence of gestational diabetes (GDM) has increased from the 1990s and onwards 1, 2. According to the National Swedish Pregnancy Register, 38.7% of women giving birth in 2014 were overweight or obese. In Sweden the prevalence of gestational diabetes is low at ~1%, but rates differ between ethnic groups 3. Diabetes in pregnancy and maternal overweight are well recognized risk factors for maternal and neonatal complications, and these risks overlap 4, 5. In untreated women with a gestational diabetes diagnosis based on the International Association of Diabetes in Pregnancy Study Group (IADPSG) criteria, an independent impact of gestational diabetes and obesity on risks of adverse outcomes has been identified 5. Also, in women with treated gestational diabetes, it has been shown that obesity and gestational diabetes are independent risk factors for maternal complications and macrosomia 6, 7; however, these studies used lower glucose limits to define gestational diabetes than those used in the European Diabetic Pregnancy Study Group (DPSG) criteria 8, 9. Furthermore, only women with obesity were included, and overweight women were not analysed as a separate group 6, 7. The Hyperglycaemia and Adverse Pregnancy Outcomes study [10] showed a linear relationship between increasing maternal glycaemia and risks of adverse outcomes, such as caesarean section, preeclampsia and fetal macrosomia. The DPSG criteria use a higher limit for diagnosis of gestational diabetes than the IADPSG criteria; thus, the DPSG criteria will define a group of women with more severe hyperglycaemia and higher risk of gestational diabetes‐related complications. Because maternal overweight/obesity is a modifiable risk factor it is of clinical interest to analyse to what extent the adverse pregnancy outcomes are attributable to hyperglycaemia per se or overweight/obesity in the mentioned gestational diabetes group. In addition, it is interesting to explore if treatment for gestational diabetes modifies the risks conveyed by concomitant overweight/obesity. To optimize pregnancy surveillance it is important to identify the women who are at highest risk of complications.
The aim of the the present study was to analyse the impact of overweight and obesity on the risk of adverse maternal outcomes and fetal macrosomia in pregnancies of women treated for severe gestational diabetes.
Material and methods
The study population included women with a BMI ≥ 18.5 kg/m² and a singleton pregnancy recorded in the Swedish Medical Birth Register between 1998 and 2012. The proportion of women with missing information on BMI was 11.1%. Women with a history of Type 1 or Type 2 diabetes were excluded, as well as those with extreme values for maternal height and weight [weight < 35 or > 200 kg (n = 50) and height < 140 and > 200 cm (n = 273)]. The Medical Birth Register contains data on > 98% of all births in Sweden. Information on all hospital births is gathered prospectively and includes maternal demographic data, reproductive history and complications during pregnancy, delivery and the neonatal period. The register does not contain data on laboratory analyses such as B‐glucose. The Swedish Medical Birth Register was validated in 2002, and the quality of the variables included in the present investigation was regarded as high 11.
Definition of exposures
In Sweden there are differences among regions in screening for gestational diabetes and diagnostic criteria 12. During the study period, the main screening strategy for identifying gestational diabetes [International Classification of Diseases (ICD) code O24.4A, O24.4B], was repeated, capillary random blood glucose ≥ 8 mmol/l (plasma glucose 9.0 mmol/l) or if a traditional risk factor [first‐degree family history of diabetes, previous large‐for‐gestational‐age (LGA) infants, gestational diabetes in a previous pregnancy or obesity] was identified, the 75 g oral glucose tolerance test (OGTT). If the woman had a random blood glucose measurement above the limit, an OGTT was performed within a week, otherwise OGTT was performed between gestational weeks 28–32, if indicated 13. One region in the country has offered a simplified OGTT to all pregnant women since 1995, with determination of 2‐h blood glucose, omitting fasting glucose. This region represented 14.4% of the population during the study period 14. During the years 2010–2011, ~20% of the pregnant population has undergone an OGTT as screening for gestational diabetes 12.
During the study period, the main diagnostic criteria for gestational diabetes were fasting capillary whole‐blood glucose ≥ 6.1 mmol/l (plasma glucose 7.0 mmol/l) and/or 2‐h blood glucose ≥ 9 mmol/l (plasma glucose 10.0 mmol/l). The diagnostic criteria have changed in some regions of Sweden during the study period. Fasting values for diagnosis have not been changed, but the 2‐h values have differed over the years/counties. A national quality register during 2011–2012 showed that 19 out of 21regions used a 2‐h value of plasma glucose ≥ 10.0 mmol/l for the diagnosis of gestational diabetes, one region used plasma glucose ≥ 9 mmol/l and two regions used 12.2 mmol/l (overt diabetes) as the diagnostic criterion. More than 95% used a 2‐h value of ≥ 10.0 mmol/l for diagnosis 12.
Maternal pre‐pregnancy BMI was calculated as weight in kg divided by height in m2. Maternal weight was recorded in light indoor clothing at the first antenatal visit, which occurs before the 15th week of gestation in > 93% of pregnancies. Height was registered on recall.
Definition of outcomes
Women with a diagnosis of gestational diabetes and/or any of the studied maternal outcomes were identified based on codes according to the 10th version of the ICD (ICD‐10). There is no information in the Swedish Medical Birth Register on date of diagnoses. Chronic hypertensive disease was defined as hypertension diagnosed before pregnancy or blood pressure ≥ 140/90 mmHg before the 20th week of gestation (ICD‐10 codes O10.0, O10.2, O10.4, O10.9). Mild preeclampsia (ICD‐10 code O14.0) was defined as at least two blood pressure measurements ≥ 140/90 mmHg, combined with proteinuria (> 0.3 g/day or ≥ 1 + on a urine dipstick). Preeclampsia was regarded as severe if diastolic blood pressure was ≥ 110 mmHg and/or proteinuria was > 5 g/day (ICD‐10 codes O14.1, O14.9). Pregnancy‐induced hypertension was defined as hypertension without proteinuria after the 20th week of gestation (ICD‐10 code O13.9). In addition to absolute birth weight > 4500 g, fetal macrosomia was defined as birth weight ≥ 90th percentile according to gestational age (by week) and gender. Reference percentiles for infant size at birth were based on data from all live‐born, singletons without malformations within the dataset.
Statistical analyses
The chi‐squared test or Student's unpaired t‐test were used to analyse the differences between maternal characteristics, maternal outcomes and fetal growth in pregnancies of normal weight and overweight/obese women with and without gestational diabetes. A P value of < 0.05 was taken to indicate statistical significance. Logistic regression was used to determine odds ratios (OR) with 95% CIs for adverse maternal outcomes and fetal macrosomia. In the multivariate logistic regression models, ORs were adjusted for potential confounders, including maternal age, ethnicity, parity, chronic hypertensive disease and smoking in early pregnancy. ORs for maternal outcomes and fetal macrosomia were compared for obese (BMI ≥ 30.00 kg/m²), overweight (BMI ≥ 25.0 and < 30.0 kg/m²) and normal‐weight women (BMI ≥ 18.5 and < 25.0 kg/m²) with or without gestational diabetes. The reference group comprised women without gestational diabetes and of normal weight (BMI ≥ 18.5 and < 25.0 kg/m²).
Analyses of risks for all outcomes were also stratified by gestational diabetes/non‐gestational diabetes. Cluster analysis was carried out to adjust for repeated pregnancies and interaction analysis to evaluate any statistically significant interaction between gestational diabetes and overweight/obesity for the risk of studied outcomes. Cluster analysis and interaction analysis were carried out using stata version 12, all other statistical analysis was performed using spss statistical software, version 22.
Ethics
The study was approved by the Regional Ethics Committee in Uppsala, Sweden (2005/216).
Results
After excluding women with a history of pregestational diabetes (Type 1 or Type 2 diabetes), 1 249 908 singleton pregnancies with data on maternal BMI were identified in the Medical Birth Register for the period 1998–2012. Of these, 13 057 (1.0%) had a diagnosis of gestational diabetes. The rate of gestational diabetes in the first half of the study period was 0.93%, and in the second half it was 1.09%. The rate of obesity in 1998 within the cohort was 12.6%, and in 2012 it was 18.1%. The corresponding values for non‐Nordic women were 15.7 and 24.6%. The rate of overweight (BMI 25–29.9 kg/m²) during the study period in pregnancies without maternal gestational diabetes was 25.5%, and was 30.4% in women with gestational diabetes. Obesity was observed in 11.5% of the pregnancies without gestational diabetes and in 38.1% of the gestational diabetes pregnancies.
The distribution of maternal characteristics differed significantly between women with and without gestational diabetes (data available on request) and between women in the different BMI categories. Women with overweight and obesity in combination with gestational diabetes differed most in maternal characteristics as compared to the reference group of normal‐weight women without gestational diabetes (Table 1).
Table 1.
Maternal and neonatal characteristics for the five study groups, with normal‐weight women without gestational diabetes as the reference
| No gestational diabetes | Gestational diabetes | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| BMI > 18.5 and < 25.0 kg/m2 N = 790228 | BMI ≥ 25.0 and < 30.0 kg/m2 N = 318419 | BMI ≥ 30 kg/m2 N = 143446 | BMI > 18.5 and < 25.0 kg/m2 N = 4114 | BMI ≥ 25.0 and < 30.0 kg/m2 N = 3972 | BMI ≥30 kg/m2 N = 4971 | ||||||
| Reference | OR (95% CI) or P value | OR (95% CI) or P value | OR (95% CI) or P value | OR (95% CI) or P value | OR (95% CI) or P value | ||||||
| Age, years | 29.9 ± 5.0 | 30.3 ± 5.2 | < 0.001 | 30.2 ± 5.3 | < 0.001 | 31.7 ± 5.2 | < 0.001 | 32.4 ± 5.3 | < 0.001 | 32.6 ± 5.5 | < 0.001 |
| Multipara, % | 52.7 | 60.2 |
1.36 (1.35–1.37) |
63.8 |
1.59 (1.56–1.61) |
56.3 |
1.16 (1.09–1.23) |
66.1 |
1.75 (1.64–1.87) |
70.0 | 2.10 (1.98–2.23) |
| Non‐nordic, % | 17.7 | 20.5 |
1.20 (1.19–1.21) |
19.3 |
1.12 (1.10–1.13) |
38.5 |
2.92 (2.74–3.11) |
43.9 |
3.66 (3.43–3.89) |
36.3 | 2.66 (2.51–2.82) |
| Smoking, % | 7.3 | 9.1 |
1.27 (1.25–1.29) |
12.4 |
1.80 (1.77–1.84) |
6.3 |
0.86 (0.76–0.98) |
8.3 |
1.15 (1.03–1.29) |
12.4 | 1.80 (1.65–1.96) |
| Chronic hypertensive disease, % | 0.2 | 0.4 | 2.27 (2.10–2.45) | 0.9 | 5.69 (5.27–6.15) | 0.4 | 2.53 (1.57–4.09) | 1.2 | 7.15 (5.32–9.61) | 2.2 | 13.81 (11.34–16.81) |
| Birth weight, g | 3513 ± 536 | 3616 ± 575 | < 0.001 | 3662 ± 575 | < 0.001 | 3506 ± 586 | < 0.001 | 3620 ± 629 | < 0.001 | 3747 ± 685 | < 0.001 |
| Pregnancy duration, days |
278 ± 13 280 (273–286) |
279 ± 13 280 (273–287) |
< 0.001 |
279 ± 14 280 (272–287) |
< 0.001 |
275 ± 13 276 (269–283) |
< 0.001 |
274 ± 14 276 (268–283) |
< 0.001 |
273 ± 14 274 (267–274) |
< 0.001 |
OR, odds ratio.
ORs with 95% CIs and significance values are given in relation to the reference group. Data are given as rate (%), mean ± sd* or median (interquartile range), as appropriate.
Risks of all studied adverse outcomes were increased in the gestational diabetes group and increased with maternal overweight/obesity, in women with and without gestational diabetes (Table 2 and Fig. 1). For all outcomes, the highest risks were observed in women with both gestational diabetes and obesity. The risks of adverse outcomes were similar in normal‐weight women with gestational diabetes and in overweight women without gestational diabetes (Table 2 and Fig. 1). Obese women without gestational diabetes had significantly more adverse outcomes than normal‐weight women with gestational diabetes (Table 2 and Fig. 1). In spite of significantly shorter pregnancy duration in women with gestational diabetes, mean birth weights were similar in the offspring of women with gestational diabetes to those in the offspring of overweight women without gestational diabetes. The incidence of LGA infants differed between 7.7 and 33.5% in the different study groups and the highest risk of LGA infants was observed in women with both obesity and gestational diabetes. When analysing the impact of overweight/obesity for women with or without gestational diabetes, the risk increase for LGA infants and caesarean section were similar. For caesarean section, there was a significant interaction (P = 0.027) between obesity and gestational diabetes as well as for LGA infants (P < 0.001); i.e. risks associated with gestational diabetes were significantly modified by concomitant overweight/obesity. Interaction analysis between gestational diabetes and overweight/obesity was not significant for preeclampsia. However, the increment in risk for preeclampsia associated with maternal obesity was smaller in women with gestational diabetes compared with women without diabetes. Cluster analysis did not change risk estimates for the different outcomes, therefore, the population‐based results are reported.
Table 2.
Maternal outcomes and fetal size in the different study groups, with normal‐weight women without gestational diabetes as the reference
| No gestational diabetes | Gestational diabetes | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BMI > 18.5 and < 25.0 kg/m2 N = 790228 | BMI ≥ 25.0 and < 30.0 kg/m2 N = 318419 | BMI ≥ 30 kg/m2 N = 143446 | BMI > 18.5 and < 25.0 kg/m2 N = 4114 | BMI ≥ 25.0 and < 30.0 kg/m2 N = 3972 | BMI ≥ 30 kg/m2 N = 4971 | |||||||
| % | OR reference | % | Adjusted OR (95% CI)a | % | Adjusted OR (95% CI)a | % | Adjusted OR (95% CI)a | % | Adjusted OR (95% CI)a | % | Adjusted OR (95% CI)a | |
| Caesarean section | 13.0 | 1 | 16.5 | 1.34 (1.33–1.36) | 21.4 | 1.90 (1.87–1.93) | 17.8 | 1.26 (1.16–1.37) | 24.0 | 1.84 (1.71–1.98) | 31.1 | 2.68 (2.52–2.85) |
| Preeclampsia | 1.8 | 1 | 2.9 | 1.76 (1.72–1.81) | 5.3 | 3.39 (3.30–3.49) | 3.4 | 2.03 (1.71–2.41) | 4.8 | 3.14 (2.71–3.65) | 9.1 | 6.45 (5.82–7.14) |
| Preeclampsia mild | 1.3 | 1 | 2.2 | 1.86 (1.80–1.91) | 4.1 | 3.62 (3.50–3.74) | 2.5 | 2.13 (1.75–2.40) | 3.8 | 3.54 (3.00–4.19) | 7.3 | 7.26 (6.49–8.12) |
| Preeclampsia severe | 0.5 | 1 | 0.7 | 1.46 (1.38–1.54) | 1.1 | 2.55 (2.40–2.71) | 0.7 | 1.50 (1.04–2.17) | 0.9 | 1.98 (1.41–2.78) | 1.5 | 3.43 (2.70–4.35) |
| Pregnancy‐induced hypertensionb | 0.7 | 1 | 1.2 | 1.81 (1.73–1.88) | 2.0 | 3.24 (3.09–3.39) | 0.9 | 1.43 (1.04–1.97) | 2.0 | 3.41 (2.73–4.27) | 3.3 | 5.61 (4.78–6.58) |
| LGAc | 7.7 | 1 | 13.0 | 1.76 (1.74–1.79) | 17.8 | 2.55 (2.51–2.59) | 14.6 | 2.25 (2.06–2.46) | 23.5 | 4.07 (3.77–4.40) | 33.5 | 6.50 (6.10–6.92) |
| Macrosomia > 4500 g | 2.8 | 1 | 5.2 | 1.91 (1.87–1.95) | 7.5 | 2.84 (2.77–2.91) | 4.1 | 1.65 (1.41–1.93) | 6.9 | 2.87 (2.53–3.27) | 11.4 | 4.80 (4.40–5.27) |
LGA, large‐for‐gestational‐age; OR, odds ratio.
Adjusted for maternal age, country of birth, parity, chronic hypertensive disease, smoking.
Hypertension without proteinuria after the 20th week of gestation.
90th percentile.
Figure 1.
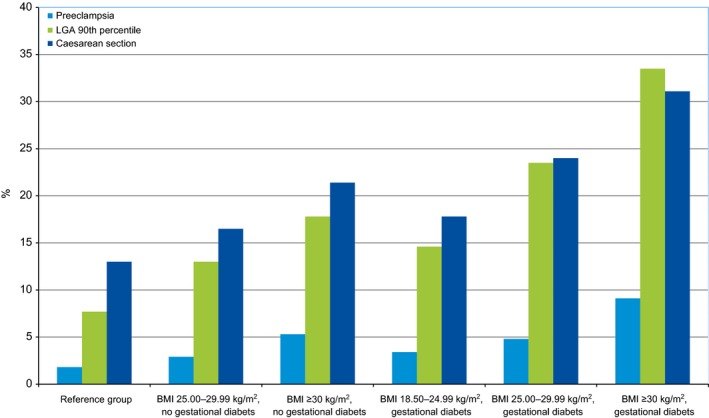
Prevalence of preeclampsia, cesarean section and LGA infants in normal weight, overweight and obese women with and without gestational diabetes. The reference group to the left. LGA, large‐for‐gestational‐age.
Discussion
The present nationwide study shows that overweight and obesity are associated with similar increments in risks of caesarean section, preeclampsia and delivery of an LGA infant in women with and without gestational diabetes. The combination of gestational diabetes and obesity conveys the greatest risks, with an independent and additive effect on risk of the adverse outcomes. In women without gestational diabetes, overweight is associated with similar risks, and obesity with higher risks, than those observed in normal‐weight women with gestational diabetes.
The strengths of the present study include its population‐based design, which included almost all pregnant women during the study period, and the fact that data were prospectively collected. BMI was registered (i.e. not self‐reported) early in pregnancy before weight gain had occurred. Furthermore, we were able to account for possible confounders. The Swedish Medical Birth Register does not include information on plasma glucose at OGTT and dates of diagnoses, which could have been of value in the present study. A limitation of the study is that, because of the mainly risk‐factor‐based screening strategies for gestational diabetes used during the study period, not all cases of gestational diabetes were detected 15. The cases missed (~50%) were mainly those with impaired glucose tolerance, which is defined as 2‐h plasma glucose values of 10–12.2 mmol/l 13. This means that the background population included undiagnosed women with gestational diabetes (impaired glucose tolerance) and differences in outcomes will probably be underestimated. The rate of gestational diabetes in Sweden is low because of the low rate of Type 2 diabetes and the relatively high thresholds used as the criteria for diagnosis of gestational diabetes. The rates of gestational diabetes and LGA infants in pregnancies of women with missing data on BMI were similar when compared with the reference group. Thus, it is unlikely that missing data on maternal BMI influenced the results. Because BMI ≥ 30 kg/m2 is used as an indication for OGTT, there could be a slight overestimation of the proportion of obese women in the gestational diabetes group, as a result of selection.
In line with previous reports, maternal overweight/obesity increased the risks of maternal complications and delivery of an LGA infant in pregnancies without gestational diabetes 16, 17.
When using the IADPSG criteria for gestational diabetes, maternal obesity and gestational diabetes were independently associated with an increased risk of LGA infants and maternal outcomes such as caesarean section and preeclampsia 5.
Treatment of gestational diabetes includes dietary advice and lifestyle changes aiming to normalize hyperglycaemia. Such treatment could result in reduced weight gain which may affect the impact of overweight and obesity on outcomes; however, the present findings show that the increment in risk associated with overweight/obesity was similar in the non‐gestational diabetes and the treated gestational diabetes group for maternal outcomes such as cesarean section, preeclampsia and fetal growth (LGA infant). This means that the treatment of gestational diabetes had no or only a minor effect on the contribution of overweight or obesity to the higher rate of adverse outcomes. This is in line with the findings of Wahabi et al. 6, who reported an independent impact of gestational diabetes and obesity on adverse outcomes. In that study, overweight women were included in the reference group, leading to an underestimation of the risks associated with increased BMI and no data on the impact of overweight on risks were presented. Ricart et al. 7 showed that pre‐pregnancy BMI and abnormal glucose tolerance were independent predictors of adverse outcome; however, they presented outcomes in quartiles of BMI which were dissimilar to the WHO classification of BMI used in the present study. The significant interaction between gestational diabetes and obesity seen for LGA infants and caesarean section was not clinically relevant in magnitude. Treatment of milder hyperglycaemia than in the present study has mainly been found to result in a reduction and normalization of the rate of LGA infants 18, 19. This is in contrast to the present study, in which a persistently elevated rate of all maternal outcomes and LGA infants in normal‐weight women with treated gestational diabetes was observed. This difference in results could be attributable to insufficient treatment or to the high level of hyperglycaemia required for the diagnosis of gestational diabetes in our cohort. To reduce rates of negative maternal outcomes and excessive fetal growth as effects of hyperglycaemia in the gestational diabetes group, different treatment strategies need to be evaluated. A recent study regarding treatment of severe hyperglycaemia with a more aggressive treatment, aiming for normoglycaemia, showed a significant reduction on fetal macrosomia 20. In that study, 67% of women required insulin treatment compared with 36% in the present study. The elevated rate of LGA infants was similar to that which could be expected as a result of a high rate of overweight/obesity.
Both Wahabi et al. and Ricart et al. concluded that, compared with obesity or high BMI, gestational diabetes has a modest effect on outcomes in pregnancies. Their findings are in contrast to the results of the present study and show that using a lower glucose limit in the diagnosis of gestational diabetes will reduce the risk estimates.
In the present study, women who were overweight or obese constituted 30% of the whole population and the corresponding proportion of women with gestational diabetes and of normal weight was 0.3%. This means that overweight and obese women without gestational diabetes account for a much higher proportion of maternal complications and LGA infants than normal‐weight women with gestational diabetes. This is consistent with the findings of Black et al. 21, who showed that pre‐pregnancy overweight accounts for a high proportion of LGA infants even in the absence of gestational diabetes. Unfortunately, evidence‐based management is still not in place to reduce the risks of maternal complications and LGA infants in overweight/obese women 22.
Because gestational diabetes is to a high degree associated with overweight/obesity, the present findings support the theory that, in addition to treating hyperglycaemia, it is important to find strategies to treat and prevent overweight and obesity.
In conclusion, maternal overweight and obesity was associated with similar increments in risks of adverse outcomes and delivery of LGA infants in women with gestational diabetes and in women without gestational diabetes. Because the majority of women with gestational diabetes were overweight/obese, this risk factor contributed greatly to adverse outcomes in the gestational diabetes group but there is a lack of effective treatment regimes. To improve maternal and neonatal outcomes in pregnancies with and without gestational diabetes, the importance of preventing overweight/obesity should be highlighted to all women of childbearing age.
Funding sources
The study was supported by grants from the Research Committee of Örebro County Council.
Competing interests
None declared.
Acknowledgements
We thank Anders Magnusson for statistical support.
Diabet. Med. 33, 1045–1051 (2016)
References
- 1. Bardenheier BH, Imperatore G, Gilboa SM, Geiss LS, Saydah SH, Devlin HM et al Trends in Gestational Diabetes Among Hospital Deliveries in 19 U.S. States, 2000‐2010. Am J Prev Med 2015; 49: 12–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ignell C, Claesson R, Anderberg E, Berntorp K. Trends in the prevalence of gestational diabetes mellitus in southern Sweden, 2003‐2012. Acta Obstet Gynecol Scand 2014; 93: 420–424. [DOI] [PubMed] [Google Scholar]
- 3. Fadl HE, Ostlund IK, Hanson US. Outcomes of gestational diabetes in Sweden depending on country of birth. Acta Obstet Gynecol Scand 2012; 91: 1326–1330. [DOI] [PubMed] [Google Scholar]
- 4. Simmons D. Diabetes and obesity in pregnancy. Best Pract Res Clin Obstet Gynaecol 2011; 25: 25–36. [DOI] [PubMed] [Google Scholar]
- 5. Catalano PM, McIntyre HD, Cruickshank JK, McCance DR, Dyer AR, Metzger BE et al The hyperglycemia and adverse pregnancy outcome study: associations of GDM and obesity with pregnancy outcomes. Diabetes Care 2012; 35: 780–786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wahabi HA, Fayed AA, Alzeidan RA, Mandil AA. The independent effects of maternal obesity and gestational diabetes on the pregnancy outcomes. BMC Endocr Disord 2014; 14: 47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ricart W, Lopez J, Mozas J, Pericot A, Sancho MA, Gonzalez N et al Body mass index has a greater impact on pregnancy outcomes than gestational hyperglycaemia. Diabetologia 2005; 48: 1736–1742. [DOI] [PubMed] [Google Scholar]
- 8. Lind T, Phillips PR. Influence of pregnancy on the 75‐g OGTT. A prospective multicenter study. The Diabetic Pregnancy Study Group of the European Association for the Study of Diabetes. Diabetes 1991; 40(Suppl. 2): 8–13. [DOI] [PubMed] [Google Scholar]
- 9. Carpenter MW, Coustan DR. Criteria for screening tests for gestational diabetes. Am J Obstet Gynecol 1982; 144: 768–773. [DOI] [PubMed] [Google Scholar]
- 10. The HAPO Study Cooperative Research Group . Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008; 358: 1991–2002. [DOI] [PubMed] [Google Scholar]
- 11. The Swedish Medical Birth Register ‐ A summary of content and quality. Available at https://www.socialstyrelsen.se/register/halsodataregister/medicinskafodelseregistret/inenglish, 2003. Last accessed 11 November 2015.
- 12. Lindqvist M, Persson M, Lindkvist M, Mogren I. No consensus on gestational diabetes mellitus screening regimes in Sweden: pregnancy outcomes in relation to different screening regimes 2011 to 2012, a cross‐sectional study. BMC Pregnancy Childbirth 2014; 14: 185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ostlund I, Hanson U. Occurrence of gestational diabetes mellitus and the value of different screening indicators for the oral glucose tolerance test. Acta Obstet Gynecol Scand 2003; 82: 103–108. [DOI] [PubMed] [Google Scholar]
- 14. Anderberg E, Kallen K, Berntorp K, Frid A, Aberg A. A simplified oral glucose tolerance test in pregnancy: compliance and results. Acta Obstet Gynecol Scand 2007; 86: 1432–1436. [DOI] [PubMed] [Google Scholar]
- 15. Fadl HE, Ostlund IK, Magnuson AF, Hanson US. Maternal and neonatal outcomes and time trends of gestational diabetes mellitus in Sweden from 1991 to 2003. Diabet Med 2010; 27: 436–441. [DOI] [PubMed] [Google Scholar]
- 16. Ovesen P, Rasmussen S, Kesmodel U. Effect of prepregnancy maternal overweight and obesity on pregnancy outcome. Obstet Gynecol 2011; 118: 305–312. [DOI] [PubMed] [Google Scholar]
- 17. Liu J, Leng J, Tang C, Liu G, Hay J, Wang J et al Maternal glucose level and body mass index measured at gestational diabetes mellitus screening and the risk of macrosomia: results from a perinatal cohort study. BMJ Open 2014; 4: e004538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Crowther CA, Hiller JE, Moss JR, McPhee AJ, Jeffries WS, Robinson JS et al Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med 2005; 352: 2477–2486. [DOI] [PubMed] [Google Scholar]
- 19. Landon MB, Mele L, Spong CY, Carpenter MW, Ramin SM, Casey B et al The relationship between maternal glycemia and perinatal outcome. Obstetr Gynecol 2011; 117: 218–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Fadl HE, Gardefors S, Hjertberg R, Nord E, Persson B, Schwarcz E et al Randomized controlled study in pregnancy on treatment of marked hyperglycemia that is short of overt diabetes. Acta Obstet Gynecol Scand 2015; 94: 1181–1187. [DOI] [PubMed] [Google Scholar]
- 21. Black MH, Sacks DA, Xiang AH, Lawrence JM. The relative contribution of prepregnancy overweight and obesity, gestational weight gain, and IADPSG‐defined gestational diabetes mellitus to fetal overgrowth. Diabetes Care 2013; 36: 56–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Thangaratinam S, Rogozinska E, Jolly K, Glinkowski S, Roseboom T, Tomlinson JW et al Effects of interventions in pregnancy on maternal weight and obstetric outcomes: meta‐analysis of randomised evidence. BMJ 2012; 344: e2088. [DOI] [PMC free article] [PubMed] [Google Scholar]


