Summary
Chemically mediated plant–herbivore interactions contribute to the diversity of terrestrial communities and the diversification of plants and insects. While our understanding of the processes affecting community structure and evolutionary diversification has grown, few studies have investigated how trait variation shapes genetic and species diversity simultaneously in a tropical ecosystem.
We investigated secondary metabolite variation among subpopulations of a single plant species, Piper kelleyi (Piperaceae), using high‐performance liquid chromatography (HPLC), to understand associations between plant phytochemistry and host‐specialized caterpillars in the genus Eois (Geometridae: Larentiinae) and associated parasitoid wasps and flies. In addition, we used a genotyping‐by‐sequencing approach to examine the genetic structure of one abundant caterpillar species, Eois encina, in relation to host phytochemical variation.
We found substantive concentration differences among three major secondary metabolites, and these differences in chemistry predicted caterpillar and parasitoid community structure among host plant populations. Furthermore, E. encina populations located at high elevations were genetically different from other populations. They fed on plants containing high concentrations of prenylated benzoic acid.
Thus, phytochemistry potentially shapes caterpillar and wasp community composition and geographic variation in species interactions, both of which can contribute to diversification of plants and insects.
Keywords: chemical interactions, community structure, Eois, multi‐trophic, phytochemical variation, Piper, population diversification
Short abstract
See also the Commentary on this article by Kessler, 212: 8–10.
Introduction
Chemically mediated interactions between plants, herbivores and natural enemies have important ecological and evolutionary impacts on biodiversity (Ehrlich & Raven, 1964; Becerra et al., 2009; Wilson et al., 2012). Despite considerable progress in understanding relationships between trophic interactions and diversity, a knowledge gap remains: specifically, how does phytochemical diversity affect multi‐trophic interactions at community and population levels? There has been progress towards addressing these questions using clonal plants to experimentally manipulate genetic diversity and quantify associated increases in arthropod richness (Crutsinger et al., 2006; Johnson et al., 2006). However, there are limitations to using clones or similar experimental approaches for understanding natural variation, especially in mega‐diverse systems; these approaches do not accurately reflect levels of interaction diversity and genetic diversity observed in nature (reviewed in Whitham et al., 2006; Hughes et al., 2008; Crutsinger, 2015). Furthermore, manipulative experiments examining multi‐trophic interactions have focused on the unidirectional influence of plant genetic diversity on arthropod richness (Fritz & Price, 1988; Johnson & Agrawal, 2005; Whitham et al., 2006), whereas only a few studies have quantified genetic and species diversity of herbivores simultaneously (Fridley & Grime, 2010; Crawford & Rudgers, 2013; Abdala‐Roberts et al., 2015). Most of these experimental examples have been limited to temperate grasslands and forests dominated by a foundation species (reviewed in Whitham et al., 2006; Hughes et al., 2008; Crutsinger, 2015), as opposed to examining these relationships in highly diverse ecosystems that lack a single dominant species. Here we focused on a model plant–herbivore–parasitoid system to examine effects of plant chemical diversity on community‐ and population‐level processes in highly diverse tropical forests.
The links between plant genetic diversity and the diversity of upper trophic levels are partially mediated via changes in phytochemical diversity (Richards et al., 2015). Although we currently lack an understanding of how population genetic and genomic variation predict phytochemical diversity, secondary metabolite concentrations are heritable phenotypes (Geber & Griffen, 2003; Johnson et al., 2009; Barbour et al., 2015) that can respond to selection and can directly and indirectly impact biotic communities (Bailey et al., 2006) and ecosystems (Driebe & Whitham, 2000; Whitham et al., 2003; Schweitzer et al., 2005). Intraspecific chemical variation between individual host plants could give rise to geographically variable selection, and contribute to shifts in herbivore preference for, or performance on, unique concentrations of individual secondary compounds (i.e. a selection mosaic; Thompson, 1999, 2005). This geographic variation in turn could affect the make‐up of herbivore and predator assemblages and might allow for greater species packing, contributing to higher alpha diversity (Rodríguez‐Castañeda et al., 2010; Richards et al., 2015). Furthermore, geographically separated herbivore populations may adapt locally to a specific chemical profile in intraspecific host plants, and this could reduce effective migration between habitats (Wilson et al., 2012). Thus, phytochemical variation across host plant populations might shape the composition of herbivore assemblages, and could give rise to geographically divergent selection on herbivore populations.
To examine such relationships, we focused on a well‐studied tropical genus of caterpillars, Eois (Geometridae), that feed exclusively on plants in the genus Piper (Piperaceae) and are host to diverse parasitoid communities (Dyer & Palmer, 2004). As recently as the Pleistocene, multiple, independent clades of Eois caterpillars have undergone geographically localized radiations, in some cases with sister species of Eois in the same geographic area, associated with the same host plant species (Wilson et al., 2012). This pattern of sympatric sister species utilizing the same resource makes it difficult to assume a framework of ecological divergence in allopatry associated with shifting host species. However, intraspecific variation in plant chemical defense could contribute to divergence (Wilson et al., 2012). Piper is chemically defended by a remarkable diversity of secondary compounds that exhibit a broad array of biological activities. These include antibacterial (Diaz et al., 2012), antitubercular (Diaz et al., 2012), antifungal (Johann et al., 2009), and anti‐herbivore effects (Dyer et al., 2003; Jeffrey et al., 2014). Piper chemical defenses are known to vary within as well as among species and have been found to function additively or synergistically to deter or poison herbivores (Dyer et al., 2003; Richards et al., 2010) and could decrease the herbivores' defenses against parasitoids (Smilanich et al., 2009). Considering the highly diverse chemistry of Piper, it is possible that sufficient intraspecific variation exists to facilitate divergence at small spatial scales. Thus, different populations of host species might provide sufficiently different niches to affect the evolution of herbivore and parasitoid communities and to facilitate local herbivore divergence.
To investigate intraspecific phytochemical variation and the consequences for both communities and populations of herbivores, we collected specialist Eois caterpillars and the leaves of Piper kelleyi Tepe they were consuming across an elevational gradient. Our goals were to understand how the abundance and composition of plant secondary metabolites within localized host plant populations might influence caterpillar and parasitoid communities along an elevational gradient, and how variation among host plant populations might influence genetic variation of one Eois species. We tested the following hypotheses: P. kelleyi populations are characterized by predictable variation in phytochemistry along an elevational gradient; phytochemical variation is a driver of caterpillar and parasitoid community structure across different microhabitats containing P. kelleyi; phytochemical variation is associated with population genetic variation in the most common Eois species.
Materials and Methods
Study system and data collection
Piper kelleyi Tepe is endemic to the eastern slopes of the Andes of Ecuador and Peru, occurring within a narrow altitudinal range between 1400 and 2400 m (Tepe et al., 2014). It is a mid‐canopy shrub characterized by broad ovate leaves and is commonly called ‘pink belly’ because of the distinct pink coloration on the ventral surface of younger leaves (Tepe et al., 2014). Piper kelleyi hosts an unusually high diversity of specialist caterpillars from the genus Eois which in turn are hosts to several species of parasitoid wasps and flies (Tepe et al., 2014). The major secondary compounds in the leaves of this species are a specific prenylated benzoic acid, chromene and dimeric chromane, which make up > 95% of the compounds present in the crude extract and are present at a high concentration of ~10% of the dry weight of the leaf material (Fig. 1a), and which all have significant negative effects on herbivores, microbes and fungi (Jeffrey et al., 2014). We collected P. kelleyi, Eois caterpillars and parasitoids from 10 sites in close proximity to Yanayacu Biological Station (00°36′S and 77°53′W) from June to August 2011 and 2012. Yanayacu, comprising disturbed habitat combined with pristine cloud forest, is located in the eastern Andes (Napo Province, Ecuador). The 10 sites were separated by elevation and consisted of four replicated plots that were randomly selected within sites. The temporary plots were 10 m in diameter. Every P. kelleyi plant was sampled for Eois caterpillars within these plots; the number of P. kelleyi plants in each plot was variable. The overall sampling resulted in a total of 125 plants and 2318 caterpillars across 40 plots. Eight of the plots were excluded because of missing leaf samples or no caterpillars being found (Supporting Information Fig. S1). Caterpillars were reared to adult moths or parasitoids to identify species and to calculate levels of parasitism. Caterpillars were identified to species level (Fig. 1b; Eois encina Dognin, Eois aff. encina Dognin, Eois ignefumata Dognin, Eois aff. pallidicosta Warren, Eois planetaria Dognin, Eois viridiflava Dognin, and Eois aff. viridiflava Dognin) based on photographs that were taken at every instar. Larval and adult images were compared to image vouchers (http://www.caterpillars.org; Dyer et al., 2010) and museum vouchers to assess species identifications. Genitalia were dissected from adult moths to confirm species determinations. Plant material was identified, and voucher specimens (Tepe & Moreno 2999 MO, QCA, QCNE; Glassmire B13 CINC, QCNE) were deposited at the Herbario Nacional del Ecuador, Quito, Ecuador (QCNE), the herbarium of the Pontificia Universidad Católica del Ecuador (QCA), the herbarium at the University of Cincinnati (CINC), and the Missouri Botanical Garden, USA (MO). In addition to P. kelleyi sampling, we sampled plant richness and total leaf abundances for all other Piper species in each plot. All collections were conducted with permission from the Museo Ecuatoriano de Ciencias Naturales in Quito, Ecuador (permit no. 001‐2011‐DPAP‐MA).
Figure 1.
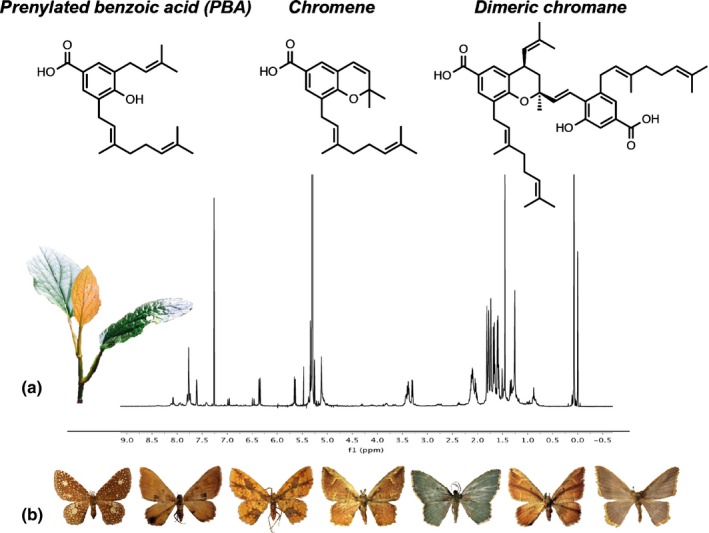
Piper kelleyi leaf chemistry and Eois caterpillar community study system. (a) NMR spectra of the crude extract containing the three major secondary compounds that have been isolated from the leaves of P. kelleyi; a specific prenylated benzoic acid, chromene and dimeric chromane. (b) Species of Eois that specialize on P. kelleyi include (from left to right): Eois planetaria Dognin, Eois aff. encina Dognin, Eois ignefumata Dognin, Eois encina Dognin, Eois viridiflava Dognin, Eois aff. pallidicosta Warren, and Eois aff. viridiflava Dognin.
HPLC analysis of P. kelleyi leaves
Young leaves were collected from 93 of the P. kelleyi shrubs that were sampled for caterpillars to quantify correlations between plant chemistry and caterpillar genetics. The leaves were dried at 25°C in a dry box at the field station. The compounds are thermally stable and incident light over relatively short periods of time is not known to cause decomposition (Jeffrey et al., 2014). We stored samples in a dark freezer to reduce light exposure during the extraction and analysis process. In the laboratory, individual leaves were ground using liquid nitrogen, mortar and pestle, and then 250 mg of leaf material was extracted with 2 ml of high‐performance liquid chromatography (HPLC)‐grade methanol for each leaf (full methods with justifications are provided in Jeffrey et al., 2014). The extract was sonicated for 15 min and the insoluble leaf material was removed by vacuum filtration. This entire extraction protocol was repeated twice. The methanol was removed under reduced pressure using rotary evaporation and placed under a high vacuum for 24 h, to remove residual solvent. The remaining crude extract was dissolved in HPLC‐grade methanol with an internal standard of 0.3 mg ml−1 methyl salicylate (Sigma‐Aldrich; product number M6752). Samples were analyzed by HPLC using a Phenomenex Luna C18 (Phenomenex Co., Torrance, CA, USA) reverse phase column (150 × 4.6 mm, 5 micron) and an Agilent Technologies (Santa Clara, CA, USA) 1200 series instrument coupled to a diode array detector (DAD) detecting at 280 nm. The solvent system employed was HPLC‐grade methanol with 0.01% trifluoroacetic acid (TFA; Sigma‐Aldrich; product no. T62200) and HPLC‐grade water with 0.01% TFA. The 5‐μl injection was eluted at a constant flow of 1 ml min−1 with a gradient of methanol and water as follows: 0–15 min, 50–100% methanol; 15–30 min, 100% methanol; 30–31 min, 100–50% methanol; 31–36 min, 50% methanol. The internal standard (methyl salicylate) was observed at retention time R t = 8.464 min. The prenylated benzoic acid was observed at R t = 6.342 min, the chromene at R t = 7.995 min, and the dimeric chromane at R t = 8.997 min relative to the internal standard. The relative abundances of prenylated benzoic acid, chromene, and dimeric chromane were quantified using the ratio of peak areas to the internal standard (methyl salicylate). The HPLC response units for area under the peak are given in mAU·s (milliabsorbance units·s).
Community analyses
We used structural equation models (Shipley, 2002) to test the hypothesis that phytochemical variation is a primary driver of Eois caterpillar assemblages based on the locations of P. kelleyi, the make‐up of the surrounding Piper community, and the diversity of parasitoids. Our focus was on P. kelleyi, but we included Piper community diversity because it is likely that some of the variance in caterpillar and parasitoid community composition could be attributable to the biology and chemistry of these other plant species (Tahvanainen & Root, 1972; Root, 1973; Barbosa et al., 2009). The community data were collected from 32 plots as described in the section ‘Study system and data collection’. From these plots, we collected 1481 Eois caterpillars that were described to species level (seven species in total), with 75% of the individuals being E. encina. We reared out 280 parasitoids that were identified to family level (Braconidae, Tachinidae, Ichneumonidae and Eulophidae). We quantified the secondary metabolites from 93 P. kelleyi plants (see the section ‘HPLC analysis of P. kelleyi leaves’). Finally, we measured richness and abundance in the plots for the 12 species of Piper plants that were found (Piper augustum Rudge, Piper sp. 1 ‘cafecito’, Piper crassinervium Kunth, Piper baezanum Sodiro ex C.DC., Piper ecuadorense Sodiro, Piper hispidum Sw., Piper lanceifolium Kunth, Piper napo‐pastazanum Trel. & Yunck., Piper perareolatum C.DC., Piper pubinervulum C.DC., Piper schuppii A.H.Gentry and Piper silvivagum C.DC.).
The relative abundances of prenylated benzoic acid, chromene, and dimeric chromane derived in relation to an internal standard from the HPLC were quantified for each individual P. kelleyi plant from which Eois caterpillars were collected. Concentrations of prenylated benzoic acid, chromene and dimeric chromane were highly correlated using Spearman rank correlations (chromene and dimeric chromane ρ = 0.91; P < 0.001; dimeric chromane and benzoic acid ρ = 0.71; P < 0.001; chromene and the benzoic acid ρ = 0.59; P < 0.001). As these compounds are hypothesized to be biosynthetically linked, a factor analysis with varimax rotation was used to create a latent variable of phytochemical defense based on shared variance of the relative abundances of each compound. As there were dominant and rare species, we used the inverse of Simpson's diversity index to calculate community entropies and species equivalents (Jost, 2006) for all Eois and parasitoid species collected from each P. kelleyi plant, and from the other Piper species located in P. kelleyi plots. Elevation was measured using GPS for each P. kelleyi plot.
For our a priori specified structural equation model, we included specific causal relationships resulting in a model with one exogenous variable (elevation) predicting three endogenous variables (Eois diversity per plant, parasitoid diversity per plant, and Piper diversity per plot); the phytochemical defense factor (from the aforementioned factor analysis) was included as a latent variable. These five variables were included in our model with hypothesized relationships (Table 1) that are context dependent and based on previous work with Piper, Eois, and parasitoids (Dyer et al., 2003, 2004; Brehm et al., 2007; Connahs et al., 2009; Smilanich et al., 2009; Rodríguez‐Castañeda et al., 2010; Wilson et al., 2012; Richards et al., 2015). For example, it was hypothesized that higher relative concentrations of secondary metabolites will decrease the diversity of specialist herbivores as a consequence of higher levels of toxicity (Poelman et al., 2009; Richards et al., 2015), and this will positively affect parasitism, because sequestered toxins impair the caterpillars' immune response when consumed (Smilanich et al., 2009; Richards et al., 2012).
Table 1.
Hypotheses and a priori predictions guiding the structural equation model
| Predictor variable | Response variable | Causal relationship | Hypothesis and prediction | Citations |
|---|---|---|---|---|
| Piper kelleyi phytochemical defense | Eois community diversity | I | The screening hypothesis posits that plants with a higher diversity of secondary metabolites have a greater probability of being toxic to a broad array of herbivores, as a result of unique mixtures and synergies that deter plant enemies. This yields the prediction that the diversity of P. kelleyi secondary metabolites will decrease Eois herbivore diversity through diverse defensive mechanisms. | Poelman et al. (2009); Smilanich et al. (2009); Firn (2010); Richards et al. (2012, 2015) |
| Piper community diversity | Piper kelleyi phytochemical defense | II | According to the associational resistance hypothesis, different plant species occurring in close proximity can decrease the likelihood of detection by herbivores. If diversity of Piper shrubs is high, then it will be harder for herbivores to detect P. kelleyi (or other species). Thus, P. kelleyi may invest less in producing defensive compounds in high Piper diversity communities because of low vulnerability to herbivores. | Root (1973); Tahvanainen & Root (1972); Barbosa et al. (2009) |
| Piper kelleyi phytochemical defense | Parasitoid community diversity | III | Plants interact with parasitoids by providing chemical cues for defense against herbivores or poisoning the immune response of caterpillars. If phytochemical defense increases, then parasitoid community diversity should increase. | Turlings & Ton (2006); Smilanich et al. (2009); Richards et al. (2012); Wäschke et al. (2014) |
| Elevation | Piper kelleyi phytochemical defense | IV | Phototoxicity occurs for many secondary metabolites when they are exposed to UV light and are metabolized to more toxic compounds or generate reactive intermediates that interfere with DNA or proteins. If UV light intensity increases with increasing elevation, then plants containing secondary metabolites that are photoactive should have a selective advantage at higher elevations. | Downum et al. (1991); Krause et al. (1999); Ruhland et al. (2013); Virjamo et al. (2014) |
| Elevation | Eois community diversity | V | Caterpillar diversity should increase as elevation increases, as a result of higher levels of specialization and lower levels of predation at higher elevations. | Brehm et al. (2007); Rodríguez‐Castañeda et al. (2010) |
| Elevation | Piper community diversity | VI | Plant diversity should decrease as elevation increases (beyond mid‐elevations) as a consequence of a variety of mechanisms, including colder temperatures, lower productivity, and smaller area. | Brehm et al. (2007); Rodríguez‐Castañeda et al. (2010) |
| Elevation | Parasitoid community diversity | VII | If herbivore specialization increases at higher elevations, then levels of parasitism and parasitoid diversity are predicted to increase as a result of preferential parasitism of specialists and higher diversities of herbivores. Increased concentrations of secondary metabolites at higher elevations may disrupt the herbivore immune response against parasitoids. | Smilanich et al. (2009); Rodríguez‐Castañeda et al. (2010); Richards et al. (2012) |
| Piper community diversity | Eois community diversity | VIII | More complex and diverse plant communities can decrease the abundance of specialist herbivores. This is because diverse plant communities decrease the detectability of preferred host plants for specialist herbivores. | Root (1973); Tahvanainen & Root (1972); Barbosa et al. (2009) |
| Piper community diversity | Parasitoid community diversity | IX | Increases in plant diversity can cause increases in parasitoid diversity by providing more host species and stronger signals for host searching and oviposition cues. Parasitoids can discriminate between their host odors and the complex odors produced by the plant community. This appears as a ‘direct’ effect because we have not measured cues or other important determinants of parasitoid diversity. | Erb et al. (2010); Wäschke et al. (2014) |
Roman numerals refer to the causal relationship depicted in the path diagram (Fig. 2). These hypotheses are context dependent and based on previous work in the Piper, Eois, parasitoid system.
We tested the fit of this model using previously established approaches (Greeney et al., 2015) and selected the formulation of the reticular action model to define alternative models. Starting values for the parameter estimates were determined by using a combination of three methods: observed moments of variables, the McDonald method, and two‐stage least squares. The estimation method for the model was maximum likelihood, and the Levenberg−Marquardt algorithm was used to iterate solutions for optimization. The χ2 for the absolute index was used to assess the fit of the model, with P > 0.1 (with 4 df) as an indication of a good fit to the data. Residuals met assumptions for the general linear model (i.e. generalized linear model with Gaussian distribution, identity link function, and fixed effects). These analyses were conducted using sas (v.9.1, proc calis; SAS Institute, Cary, NC, USA).
Caterpillar genetic variation, phytochemical variation, and elevation
We used a genotyping‐by‐sequencing (GBS) approach to generate population genetic data for E. encina. DNA was extracted from 155 individual E. encina caterpillars. DNA extractions were performed on thoraxes of dry adult specimens using Qiagen DNeasy Blood and Tissue kits (Qiagen Inc., Valencia, CA) and quantified using spectrophotometry. Reduced‐representation genomic libraries for Illumina (Illumina Biotechnology Co., San Diego, CA, USA) sequencing were constructed using a GBS approach (Gompert et al., 2012; Parchman et al., 2013). Genomic DNA was cut with two restriction enzymes, EcoRI and MseI, and a unique DNA barcode was ligated to the fragments from each individual to allow multiplexing. DNA fragment libraries were PCR amplified using standard Illumina primers and size selected for a region between 200 and 300 bases using QIAquick gel extraction kits (Qiagen). One lane of sequencing on the Illumina HiSeq at the National Center for Genome Resources (Santa Fe, NM) generated 47 million 100‐bp reads.
We used a custom Perl script to parse barcodes from sequences and to assign the correct individual ID to each read. As a reference genome is not available for E. encina, we used a two‐step assembly procedure to assemble reads into homologous genetic regions. We first performed a de novo assembly for a subset of 30 million sequences with seqman ngen 3.0.4 (dnastar, Dnastar Software Co., Madison, WI, USA), using a minimum match percentage of 92%, a gap penalty of 50, and a match size of 50 bp (full details of assembly parameterization are available upon request). We removed low‐quality contigs and sequences < 82 or > 88 bases in length to generate an artificial reference containing 481 606 contig consensus sequences. We then assembled all reads onto the reference using the aln and samse algorithms in bwa (Li & Durbin, 2009), using an edit distance of 3 (full parameters for bwa assemblies are available upon request). We used samtools and bcftools (Li et al., 2009) to identify single nucleotide polymorphisms (SNPs) in assemblies and retained SNPs where at least one read was aligned in at least 50% of the individuals. Genotype likelihoods were calculated with bcftools (Li et al., 2009), stored in variant call format (VCF), and converted to composite genotype likelihood point estimates for downstream analyses. We summarized genetic variation using principal components (PC) analysis of the genotype covariance matrix using the prcomp package in R (R Development Core Team, 2013).
We created matrices of genetic distances among caterpillar individuals using the calculated PC scores of genotype likelihoods. We created three different matrices based on: (1) all the PCs for an overall genetic representation; (2) PC1 and 2 because they explain most of the variation for population structure; and (3) just PC2 because it distinguishes the higher elevation individuals. We used a multiple regression on distance matrices (MRM; Lichstein, 2007), using the ‘mrm’ function from the ecodist package in R (Goslee & Urban, 2007), with 100 000 permutations. We conducted three models that differed in the response variable. The response variable was the genetic distances based on Euclidean distances for: (1) all the PCs, (2) PC1 and 2, and (3) only PC2, as already described. The independent variables were elevation, individual compound concentrations (i.e. prenylated benzoic acid, chromene, and dimeric chromane) and GPS location from each plant sampled, transformed into separate Euclidean distance matrices.
Results
Plant chemistry analyses
We found that prenylated benzoic acid, chromene, and dimeric chromane were present in P. kelleyi at a high concentration of ~10% of the dry weight of leaf material (Fig. 1a), which is consistent with previous results (Jeffrey et al., 2014). While these compounds were highly correlated and hypothesized to be biosynthetically linked, the relative abundance of each compound can be important for synergistic or additive effects on herbivores (Dyer et al., 2003; Richards et al., 2010). Thus, we calculated a Shannon equivalence variable (Jost, 2006) for the three compounds, which accurately captures their relative abundances in an individual plant and does not give added weight to one dominant compound as all compounds are present (i.e. the richness of compounds does not change). This compound equivalence variable was used in the structural equation models already described; higher values indicate greater abundance and evenness of the compounds.
Community analyses
The factor analysis included concentrations of the three different secondary metabolites, utilized varimax rotation, and yielded two factors. Kaiser's measure of sampling adequacy, which assesses the adequacy of correlation matrices for factor analysis, was acceptable (0.6) and the matrix was not an identity matrix. The relative concentrations of all three compounds loaded onto Factor 1 with high loadings for dimeric chromane (0.96) and chromene (0.92) and lower loadings for prenylated benzoic acid (0.73). Factor 2 had more useful loadings for separating prenylated benzoic acid (0.68) from dimeric chromane (−0.18) and chromene (−0.36), and it was utilized in path models as a latent ‘phytochemical defense’ (benzoic acid vs other compounds) variable.
For the structural equation models, the model that provided the best fit to the data is summarized by the path diagram in Fig. 2 (χ2 = 0.014; df = 1; P = 0.91; P‐values closer to 1 indicate a better fit to the data). There were several notable causal relationships supported by this model (Table 1; Figs 2b, S2). First, elevation had positive direct effects on phytochemical defense (standardized path coefficient (spc) = 0.10; slope (B) = 0.0009), Eois community diversity (spc = 0.11; B = 0.0004), and Piper community diversity (spc = 0.14; B = 0.0005). While Eulophidae parasitoids had the highest densities at 2000 m and Braconidae, Ichnuemonidae and Tachinidae parasitoids had the highest densities at 2100 m (Fig. 3), in our path model there is a linear relationship between elevation and parasitoid diversity while accounting for other factors that include Piper community diversity and high concentrations of phytochemistry (Fig. 2b; spc = 0.37; B = 0.0024). The effects of elevation on phytochemical defense also cascaded to the arthropod community; as phytochemical defense increased, Eois community diversity on P. kelleyi decreased (Fig. 2b; spc = −0.2; B = −0.12) and parasitoid diversity increased (spc = 0.26; B = 0.24) – adding to the strong direct effects of elevation on parasitoids (Fig. 2b). Increases in Piper plant community diversity also enhanced parasitoid diversity on individual P. kelleyi plants (spc = 0.11; B = 0.44) and had the strongest negative effect on Eois caterpillar diversity on individual P. kelleyi plants (Fig. 2b; spc = −0.48; B = −1.23). Finally, greater Piper plant community diversities are associated with decreased phytochemical defense in P. kelleyi (spc = −0.25; B = −1.04). Overall, the path model suggests that increases in phytochemical defense – specifically the prenylated benzoic acid – and Piper community diversity reduced Eois community diversity, while increasing parasitoid community diversity (Fig. 2b). Higher elevations are associated with greater Piper, Eois and parasitoid community diversities, as well as with greater P. kelleyi phytochemical defense.
Figure 2.
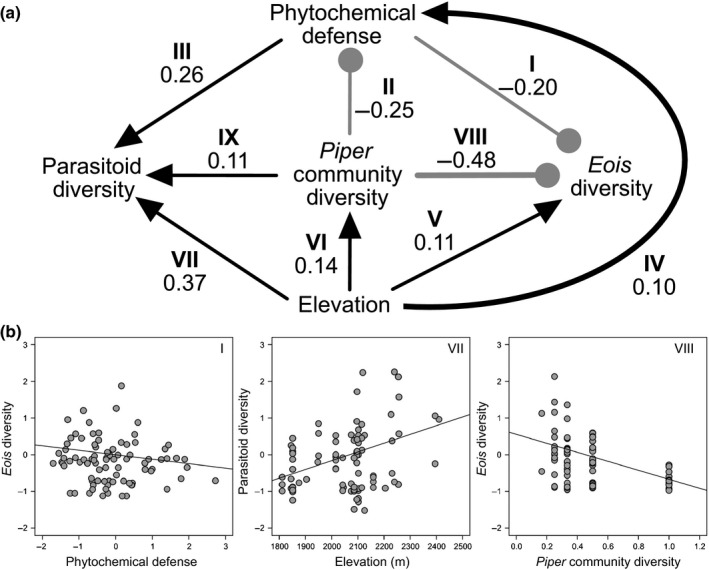
Results of a structural equation model depicting hypothesized causal relationships between: phytochemical defense (latent variable), Eois diversity per P. kelleyi plant, parasitoid diversity per P. kelleyi plant, Piper species diversity per plot, and elevation. (a) Illustration of the overall path model. The direct positive effects are indicated by black arrows, while the direct negative effects are indicated by light gray blunt‐ended lines. The numbers beside the lines are the standardized path coefficients. The Roman numerals above the path coefficients relate to Table 1, which describes specific hypotheses being tested. Piper kelleyi phytochemical variation is a latent variable, created via factor analysis on relative abundances of the three defensive compounds with varimax rotation. The path coefficients are all significant (P < 0.05) and the model is a significant fit to the data (χ2 = 0.014; df = 1; P > 0.1). (b) A subset of the partial correlation plots from paths I, VII and VIII; the remaining partial correlation plots can be found in Supporting Information Fig. S2.
Figure 3.
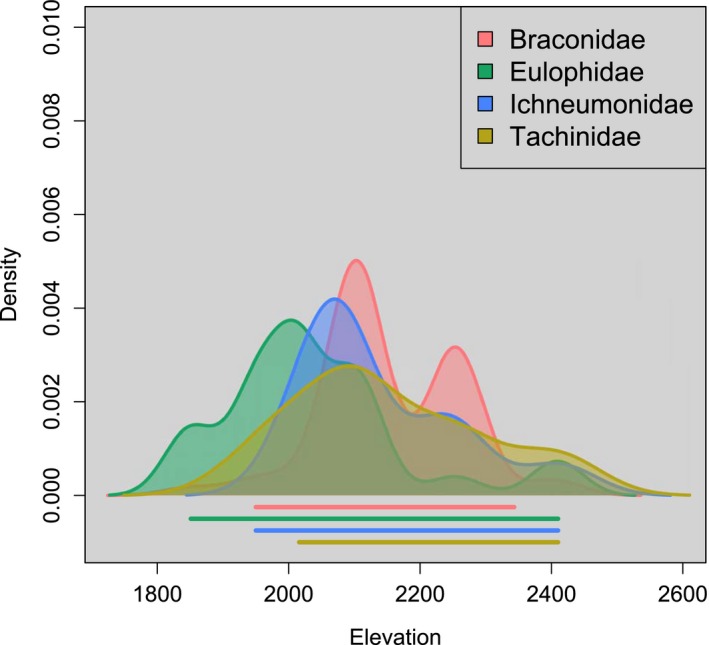
Parasitoid family density along elevation. Parasitoids reared and identified to family level included Braconidae, Eulophidae, Ichneumonidae and Tachinidae. Eulophidae had the highest densities at 2000 m and Braconidae, Ichneumonidae and Tachinidae had the highest densities at 2100 m. Lines under plots indicate 95% confidence intervals. This relationship is based on a linear model, while the path analysis includes residual variation from interacting variables.
Caterpillar genetic variation, phytochemical variation, and elevation
After executing one lane of sequencing on the Illumina HiSeq platform, and conducting assemblies and variant calling as described in the Materials and Methods section, we retained genotypes at 20 458 SNPs in 155 E. encina individuals (accessible in Dryad, doi:10.5061/dryad.d67h6). The mean coverage per locus per individual was 1.25×. We used PC analysis to summarize patterns of genetic variation across all individuals. The first two PCs explained 7% (PC1) and 1% (PC2) of the genotypic variation, and suggest that E. encina from the highest elevation is genetically differentiated from that at lower elevations (Fig. 4a). There were other differentiated individuals, but we focus discussion on the highest elevation for which we have a hypothesis about phytochemical variation. The P. kelleyi plants that this population was occurring on had higher relative concentrations of the prenylated benzoic acid (Fig. 4b). Other potential patterns suggested by the PCA were not interpretable based on our original mechanistic hypotheses.
Figure 4.
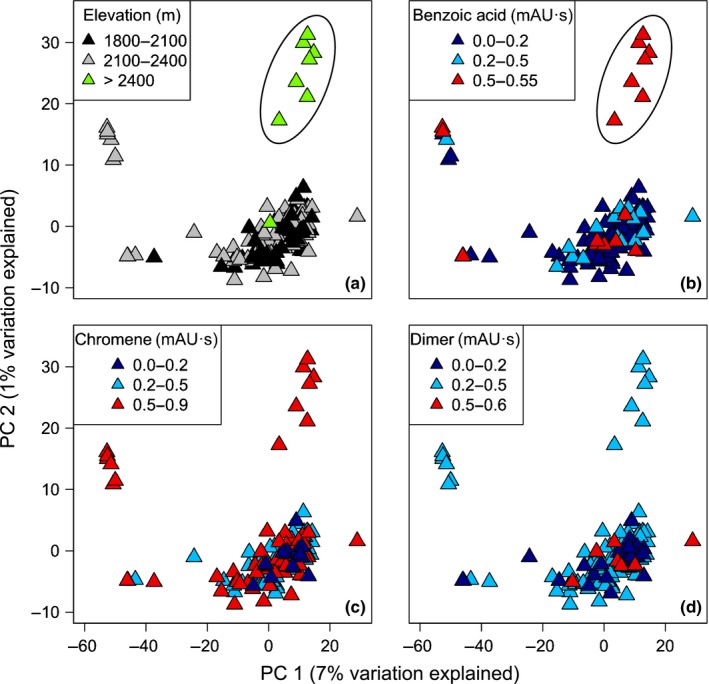
Principal components analysis illustrates genetic variation and structure across populations of Eois encina moths. Points represent genotypic data for 20 458 single nucleotide polymorphisms (SNPs) in each individual. The first two principal components (PCs) explained 7% (PC1) and 1% (PC2) of the genotypic variation across all individuals and loci and revealed previously undetected genetic differentiation in high‐elevation populations. Black circles denote individuals exhibiting genetic differentiation, which is correlated with high elevation and benzoic acid concentrations in plants. (a–d) Panels are the same principal components analysis, but differ in the overlaid gradient. Panel (a) illustrates an elevation gradient, with black being the lowest elevation and green being the highest elevation; (b–d) illustrate the prenylated benzoic acid, chromane and dimeric chromane concentration gradients, respectively. Dark blue is the lowest concentration, while red is the highest concentration.
Multiple regression using distance matrices (Lichstein, 2007) was conducted to examine whether elevation, phytochemical variation and GPS location were predictors of genetic distances among E. encina individuals. Genetic variation was estimated using the PC scores of multi‐locus genotype likelihoods of E. encina individuals transformed into a distance matrix; three matrices were created based on all the PCs, PC1 and 2, and only PC2 scores. Table 2 depicts the results from multiple Mantel tests in which models differed based on the response variable: (A) all the PCs, (B) PC1 and 2, and (C) only PC2 scores. Increases in elevation and prenylated benzoic acid significantly explained E. encina genetic variation. Geographic location was only a significant predictor with PC2 score as the response variable.
Table 2.
Results from multiple Mantel tests in which models differed based on the response variable
| Model | Elevation | PBA | Chromene | Dimeric chromane | Location | Overall model |
|---|---|---|---|---|---|---|
| A – all PCs | pMc = 0.007; P = 0.14 | pMc = 16.89; P < 0.001* | pMc = 3.74; P = 0.54 | pMc = −13.42; P = 0.18 | pMc = −1.69; P = 0.79 | R 2 = 0.05; P = 0.001* |
| B – PC1 and PC2 | pMc = 0.01; P = 0.05* | pMc = 26.94; P < 0.001* | pMc = −0.06; P = 0.99 | pMc = −12.64; P = 0.38 | pMc = −1.37; P = 0.88 | R 2 = 0.07; P < 0.001* |
| C – only PC2 | pMc = 0.02; P < 0.001* | pMc = 20.21; P < 0.001* | pMc = −3.54; P = 0.31 | pMc = −4.6; P = 0.43 | pMc = −9.82; P = 0.01* | R 2 = 0.26; P < 0.0001* |
pMc, partial Mantel coefficient.
Predictor variables for all models were elevation, prenylated benzoic acid (PBA), chromene, dimeric chromane and GPS location. For the response variable, genetic variation was estimated using the principal component (PC) scores of multi‐locus genotype likelihoods of E. encina individuals transformed into distance matrices. Three matrices were created based on: A, all the PCs; B, PC1 and 2; C, only PC2 scores. Elevation and PBA were significant predictors of genetic variation in Eois encina populations.
*Predictor variables and overall models that were significant.
Discussion
Intraspecific phytochemical variation is an underappreciated source of geographic variation in species interactions and their evolutionary outcomes (Bolnick et al., 2011; Zhang et al., 2015). At what scales might this variation shape community structure and generate divergent selection and population differentiation? We tested the hypothesis that phytochemical variation within a single species of host plant P. kelleyi drives community structure via subtle microhabitat differences in chemically mediated interactions between plants, their specialist herbivores and associated parasitoids. We also examined whether genetic structure across populations of specialist caterpillars was correlated with phytochemically variable microhabitats. We found evidence for substantial chemical variation among individual plants (Figs S3–S5; Tables S1, S2). The concentration of one important defensive compound, prenylated benzoic acid, increased with increasing elevation (Fig. S4). In turn, plant chemistry strongly predicted community structure at the herbivore and parasitoid trophic levels, with more toxic plants supporting a lower diversity of specialist herbivores, richer parasitoid communities, and higher levels of parasitism. Across populations of one of the specialist caterpillar species, our results suggest the potential for genetic differentiation between high‐elevation caterpillar populations and all others (Fig. 4), which could be mediated by high concentrations of plant secondary metabolites. Overall, the results are consistent with phytochemical variation shaping community structure, perhaps giving rise to geographic variation in the selection pressures experienced by specialist herbivores. Below, we discuss community structure and then evaluate possibilities for chemically mediated population structure.
Within our plots, associations between phytochemical defense and Eois assemblages on individual P. kelleyi plants were consistent with predictions that increased phytochemical defense causes decreased diversity of specialist caterpillars, while concurrently increasing parasitoid community diversity. Intraspecific phytochemical diversity had a negative influence on caterpillar diversity, consistent with previous studies demonstrating that increases in phytochemical variation within a host Piper species have negative effects on some Eois species but not others (Dyer et al., 2003, 2004; Richards et al., 2010, 2012). Interestingly, increased phytochemical diversity (measured as changes in relative abundance of the three defensive compounds) facilitated higher parasitoid diversity. This chemistry−parasitoid relationship could imply that increases in phytochemical variation of P. kelleyi may somehow attract more parasitoids for defense against caterpillars (Turlings & Ton, 2006; Wäschke et al., 2014) or that plant chemistry compromises the immune response of sequestering specialists (Smilanich et al., 2009) – the former is less likely as these compounds are not volatile. Could it be that volatile organic compounds are also increased and attracting richer parasitoid communities (e.g. Raguso, 2009)? It is worth pursuing this question, along with the interesting pattern of increased parasitism pressure, a result that contrasts with a previously reported trend that levels of parasitism decrease with increasing elevation in temperate zones (reviewed in Hodkinson, 2005).
Piper diversity within plots had the strongest negative effect on the diversity of Eois found on P. kelleyi, which is consistent with a hypothesis of associational resistance (Root, 1973; Barbosa et al., 2009). Alternatively, this negative relationship between Piper diversity and Eois diversity on our focal host plant could be a consequence of the highly specialized relationship these moths have with Piper combined with the fact that Eois moths are not very mobile. As a result, preferred hosts are likely to be less ‘apparent’ in high‐diversity Piper plots (Root, 1973; Barbosa et al., 2009). Higher diversity Piper communities also attracted more parasitoids that attack Eois. This result suggests that parasitoids are able to locate their preferred host even in the presence of complex cues from closely related nonhost plants (Erb et al., 2010; Wäschke et al., 2014). Alpha diversity of Piper, Eois, and associated parasitoids at a small scale (10‐m‐diameter plots) also increased with elevation, consistent with previous Piper diversity studies that demonstrated a strong effect of elevation on Piper and Eois diversity (without mid‐domain effects) in the eastern Andes (Brehm et al., 2007; Rodríguez‐Castañeda et al., 2010). Similarly, Rodríguez‐Castañeda et al. (2010) found that monophagous herbivore diversity increased with elevation, suggesting that colder temperatures and higher precipitation up the mountain are important for diversity by increasing specialization of herbivores and parasitoids on host plants. Our results contrast with studies in temperate systems. For example, Rasmann et al. (2014) found that Buprestid beetles tended to be more generalized and polyphagous at higher altitudes, and Pellissier et al. (2012) found that butterfly specialization decreased with increases in elevation; this was largely attributed to decreases in host plant abundance as elevation increased. As pointed out by Rodríguez‐Castañeda et al. (2016), there are not enough studies of chemically mediated trophic interactions across elevations to enable any generalizations to be made in tropical or temperate ecosystems, so it is not clear if the patterns reported here are part of a more general pattern of changes in plant chemistry and diversity.
Of the variables examined in our study, elevation was the main predictor of changes in phytochemical defense as well as the abundance of individual compounds. Concentrations of secondary metabolites were correlated with increasing elevation, which has been documented in a few studies with other classes of secondary metabolites, but there are exceptions (reviewed by Rodríguez‐Castañeda et al., 2016). Compounds for which there is a documented decline in concentrations of secondary metabolites with increasing elevation include terpenes (Hengxiao et al., 1999), alkaloids (Salmore & Hunter, 2001), and iridoid glycosides (Pellissier et al., 2012). As empirical data for changes in chemical defense across elevational gradients are still scarce (Rodríguez‐Castañeda et al., 2016), it is useful to examine the mechanisms that cause chemistry to change with elevation. In the case of P. kelleyi, the increase in defensive compounds at greater elevation could be attributable to the relationship between increased UVB radiation at higher elevations and the photoactive properties of the prenylated benzoic acid, chromene, and dimeric chromane (Figs S4, S5; Table S1, S2; Krause et al., 1999; Ruhland et al., 2013; Virjamo et al., 2014). UV light is known to affect concentrations of phytochemicals through a variety of mechanisms (Becker & Michl, 1966; Zangerl & Berenbaum, 1987; Downum et al., 1991; Padwa et al., 1996), and plants at higher elevations are exposed to a greater intensity of UV light compared with plants at lower elevations. The association between elevation and P. kelleyi phytochemistry was mostly attributable to changes in the relative concentration of prenylated benzoic acid in the leaves. Chromenes are particularly reactive in the presence of UV light (Becker & Michl, 1966; Padwa et al., 1996), and it is likely that increased UV radiation facilitates related biosynthetic links through oxidation of the prenylated benzoic acid and cyclization to the chromene, as well as through a photoinitiated dimerization to produce the chromane. The prenylated benzoic acid probably acts as the precursor, so an increase in one compound is usually associated with increases in the others, as we document here. Thus, Piper shrubs that produce phototoxic compounds could be more toxic at the top of elevational gradients in the Andes and less toxic towards the bottom, because of UV‐induced increases in abundance and diversity of secondary metabolites.
The possibility that changes in elevation and UV exposure modify the phytochemistry of P. kelleyi is relevant to the evidence for Eois population structure. We found evidence of subtle genetic differentiation for E. encina at the highest elevation (Fig. 4a), but little evidence for differentiation among other populations. The plants located at the highest elevation (> 2400 m) contained higher concentrations of prenylated benzoic acid (Figs S3–S5; Tables S1, S2), which could be a source of divergent selection. Local adaptation of E. encina to high‐elevation hosts with high prenylated benzoic acid concentrations could reduce gene flow by causing selection against migrants and low hybrid fitness (Zhang et al., 2015), which could explain the observed genetic differentiation among low‐ and high‐elevation caterpillars. One individual caterpillar was collected at a high‐elevation site that was not genetically grouped with the other highest elevation caterpillars (Fig. 4a). Interestingly, this one distinct high‐elevation caterpillar was feeding on plants containing a lower concentration of the prenylated benzoic acid (Fig. 4b), which could suggest that this individual was a migrant from lower elevation populations. We assume that the high‐elevation E. encina do not represent a cryptic species based on adult vouchers, genitalia dissections, and extensive photographic documentation of larvae. MRM models indicated that genetic distances were explained by chemistry while controlling for elevation (R 2 = 0.07; P < 0.001). The genetic differentiation of E. encina collected at high elevation on high‐acid plants, although subtle, is consistent with the possibility of local adaptation limiting gene flow across these populations and with the hypothesis that phytochemical variation could generate geographically divergent selection across E. encina populations. Our results are similar to those of a recent study on the elm Ulmus pumila L., which demonstrated that leaf age is a source of divergence in two sympatric sister elm leaf beetles, Pyrrhalta maculicollis and Pyrrhalta aenescens (Zhang et al., 2015). Future studies with the Piper system should include a reciprocal rearing experiment comparing high and low populations of Eois to demonstrate that Eois populations at high elevation are adapting to higher concentrations of prenlyated benzoic acid in P. kelleyi plants.
Phytochemical variation could be an ecological source of natural selection on specialist herbivores. Such variation in plant defense has performance and fitness consequences for herbivores, and this is particularly true for specialist herbivores, which may be locally adapted to detoxify specific compounds (Dyer et al., 2003; Richards et al., 2010). The biological activity of the defensive compounds in P. kelleyi included decreased development rate, lower pupal mass and decreased survival when small amounts (i.e. 3.75% of the dry weight) were fed to naïve generalist caterpillars (Jeffrey et al., 2014). This amount is less than half of the 10% dry weight typically found in the leaves of P. kelleyi (Jeffrey et al., 2014). However, because this assay was performed on generalist caterpillars, it is unknown how specialist caterpillars respond to increased concentrations of the three compounds. Other studies have shown that Piper chemical defenses have subtle effects on Eois physiology rather than direct toxic effects (Dyer et al., 2004; Smilanich et al., 2009). For example, Richards et al. (2010) found that subtle changes in mixture diets (i.e. 0.4%) of P. cenocladum resulted in decreased survival rates and increased parasitism frequency in specialist E. nympha Schaus caterpillars. It is likely that at higher elevations the intensity of UV light is greater and can enhance the toxicity of these plants by increasing the abundance and evenness of defensive compounds, leading to selection against larvae that lack adequate physiological mechanisms for detoxification or tolerance (e.g. McCloud & Berenbaum, 1999).
Conclusions
The results reported here represent a significant contribution to our understanding of the chemical processes maintaining biodiversity at different taxonomic and spatial scales. The study examined community and population structure at a fine scale by focusing on intraspecific host plant populations with distinct phytochemical profiles and associated herbivore communities. We found that variation in plant chemistry affected the caterpillar community (relative abundances of different caterpillar species) and genetic differentiation among populations of E. encina, the most common caterpillar species. The adaptive significance of phytochemical variation in response to UV light intensities, particularly whether it is plastic or genetic, as well as the potential role of phototoxicity have yet to be determined. Future studies are needed to examine whether the compounds are phototoxic and how this influences local adaptation and community assemblages of specialist and generalist consumers.
Author contributions
A.E.G. wrote the first draft of the manuscript. L.A.D., M.L.F. and T.L.P. contributed substantially to revisions. A.E.G., L.A.D., M.L.F. and L.A.R. generated hypotheses and designed experiments. L.A.D., M.L.F., C.S.J. and A.M.S. funded experiments. A.E.G., L.A.D., T.R.W., C.R.M., W.S., L.A.S., E.J.T. and S.V‐C. collected field data. A.E.G., C.S.J., L.A.R., M.D.L. and C.D.D. conducted chemical analyses. A.E.G., M.L.F., J.P.J. and J.S.W. prepared the genomic library. T.L.P., C.C.N. and M.L.F. aligned and assembled genotypes. A.E.G., M.L.F., L.A.D., T.L.P., J.P.J. and L.A.R. conducted statistical analyses. E.J.T. identified P. kelleyi plant samples. J.S.M. conducted genitalia dissections to identify Eois species.
Supporting information
Please note: Wiley Blackwell are not responsible for the content or functionality of any supporting information supplied by the authors. Any queries (other than missing material) should be directed to the New Phytologist Central Office.
Fig. S1 A map illustrating the locations of P. kelleyi plant and caterpillar samples that were collected near Yanayacu Biological Station near Cosanga, Napo Province, Ecuador in the eastern Andes (00°36′S and 77°53′W).
Fig. S2 Partial correlation plots from the structural equation model (Fig. 2a) for paths II, III, IV, V, VI and IX.
Fig. S3 Principal component analysis examining the chemical similarity between individual Piper kelleyi plants using concentrations of prenylated benzoic acid, chromene and dimeric chromane of individual plants.
Fig. S4 Linear regression examining the relationship between elevation and PC2 scores from the phytochemical PCA (Fig. S3).
Fig. S5 Linear regression examining the relationship between elevation and PC1 scores from the phytochemical PCA (Fig. S3).
Table S1 Variation explained by each component used in the principal components analysis
Table S2 The first three eigenvalues of the correlation matrix for the principal component analysis, the proportion of total variance, and the cumulative variance for each principal component
Acknowledgements
This research was funded by the National Science Foundation grants DEB 1442103, DEB 1145609 and DEB 1502059. Scott Applebee graciously created the GIS map. Special thanks are due to Dr Christopher Moore and Joshua Harrison for their help with the statistical analyses. Many hours of fieldwork were conducted by Elia Pirtle, Jessica Hogue, Kevin Salls, Danielle Salcido and Earthwatch volunteers. We thank Dr Stephen Spain for guidance with NMR and HPLC. The authors thank Dr Harold Greeney and José Simbaña for assistance at Yanayacu Biological Station. We appreciate the help of three anonymous reviewers, whose comments substantially improved the manuscript.
See also the Commentary on this article by Kessler, 212: 8–10.
References
- Abdala‐Roberts L, Mooney KA, Quijano‐Medina T, Campos‐Navarrete MJ, González‐Moreno A, Parra‐Tabla V. 2015. Comparison of tree genotypic diversity and species diversity effects on different guilds of insect herbivores. Oikos 124: 1527–1535. [Google Scholar]
- Bailey JK, Wooley SC, Lindroth RL, Whitham TG. 2006. Importance of species interactions to community heritability: a genetic basis to trophic‐level interactions. Ecology Letters 9: 78–85. [DOI] [PubMed] [Google Scholar]
- Barbosa P, Hines J, Kaplan I, Martinson H, Szczepaniec A, Szendrei Z. 2009. Associational resistance and associational susceptibility: having right or wrong neighbors. Annual Review of Ecology, Evolution, and Systematics 40: 1–20. [Google Scholar]
- Barbour MA, Rodriguez‐Cabal MA, Wu ET, Julkunen‐Tiitto R, Ritland CE, Miscampbell AE, Jules ES, Crutsinger GM. 2015. Multiple plant traits shape the genetic basis of herbivore community assembly. Functional Ecology 29: 995–1006. [Google Scholar]
- Becerra JX, Noge K, Venable DL. 2009. Macroevolutionary chemical escalation in an ancient plant–herbivore arms race. Proceedings of the National Academy of Sciences, USA 106: 18062–18066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker RS, Michl J. 1966. Photochromism of synthetic and naturally occurring 2H‐chromenes and 2H‐pyrans. Journal of the American Chemical Society 88: 5931–5933. [Google Scholar]
- Bolnick DI, Amarasekare P, Araujo MS, Burger R, Levine JM, Novak M, Rudolf VHW, Schreiber SJ, Urban MC, Vasseur DA. 2011. Why intraspecific trait variation matters in community ecology. Trends in Ecology & Evolution 26: 183–192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brehm G, Colwell RK, Kluge J. 2007. The role of environment and mid‐domain effect on moth species richness along a tropical elevational gradient. Global Ecology and Biogeography 16: 205–219. [Google Scholar]
- Connahs H, Rodríguez‐Castañeda G, Walters T, Walla T, Dyer L. 2009. Geographic variation in host‐specificity and parasitoid pressure of an herbivore (Geometridae) associated with the tropical genus Piper (Piperaceae). Journal of Insect Science 9: 28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford KM, Rudgers JA. 2013. Genetic diversity within a dominant plant outweighs plant species diversity in structuring an arthropod community. Ecology 94: 1025–1035. [DOI] [PubMed] [Google Scholar]
- Crutsinger GM. 2015. A community genetics perspective: opportunities for the coming decade. New Phytologist 210: 65–70. [DOI] [PubMed] [Google Scholar]
- Crutsinger GM, Collins MD, Fordyce JA, Gompert Z, Nice CC, Sanders NJ. 2006. Plant genotypic diversity predicts community structure and governs an ecosystem process. Science 313: 966–968. [DOI] [PubMed] [Google Scholar]
- Diaz LE, Munoz DR, Prieto RE, Cuervo SA, Gonzalez DL, Guzman JD, Bhakta S. 2012. Antioxidant, antitubercular and cytotoxic activities of Piper imperiale . Molecules 17: 4142–4157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Downum KR, Swain LA, Faleiro LJ. 1991. Influence of light on plant allelochemicals: a synergistic defense in higher plants. Archives of Insect Biochemistry and Physiology 17: 201–211. [Google Scholar]
- Driebe EM, Whitham TG. 2000. Cottonwood hybridization affects tannin and nitrogen content of leaf litter and alters decomposition. Oecologia 123: 99–107. [DOI] [PubMed] [Google Scholar]
- Dyer LA, Dodson CD, Stireman JO III, Tobler MA, Smilanich AM, Fincher RM, Letourneau DK. 2003. Synergistic effects of three Piper amides on generalist and specialist herbivores. Journal of Chemical Ecology 29: 2499–2514. [DOI] [PubMed] [Google Scholar]
- Dyer LA, Gentry GL, Greeney HF, Walla TW. 2010. Caterpillars and parasitoids of the Eastern Andes in Ecuador. [WWW document] URL http://www.caterpillars.org [accessed 8 February 2016].
- Dyer LA, Letourneau DK, Dodson CD, Tobler MA, Stireman JO III, Hsu A. 2004. Ecological causes and consequences of variation in defensive chemistry of a Neotropical shrub. Ecology 85: 2795–2803. [Google Scholar]
- Dyer LA, Palmer ADN. 2004. Piper: a model genus for studies of phytochemistry, ecology, and evolution. New York, NY, USA: Springer. [Google Scholar]
- Ehrlich PR, Raven PH. 1964. Butterflies and plants: a study in coevolution. Evolution 18: 586–608. [Google Scholar]
- Erb M, Foresti N, Turlings TCJ. 2010. A tritrophic signal that attracts parasitoids to host‐damaged plants withstands disruption by non‐host herbivores. BMC Plant Biology 10: 247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Firn R. 2010. Nature's chemicals: the natural products that shaped our world. New York, NY, USA: Oxford University Press. [Google Scholar]
- Fridley JD, Grime JP. 2010. Community and ecosystem effects of intraspecific genetic diversity in grassland microcosms of varying species diversity. Ecology 91: 2272–2283. [DOI] [PubMed] [Google Scholar]
- Fritz RS, Price PW. 1988. Genetic variation among plants and insect community structure: willows and sawflies. Ecology 69: 845–856. [Google Scholar]
- Geber MA, Griffen LR. 2003. Inheritance and natural selection on functional traits. International Journal of Plant Sciences 164(S3): S21–S42. [Google Scholar]
- Gompert Z, Lucas LK, Nice CC, Fordyce JA, Forister ML, Buerkle CA. 2012. Genomic regions with a history of divergent selection affect fitness of hybrids between two butterfly species. Evolution 66: 2167–2181. [DOI] [PubMed] [Google Scholar]
- Goslee SC, Urban DL. 2007. The ecodist package for dissimilarity‐based analysis of ecological data. Journal of Statistical Software 22: 1–19. [Google Scholar]
- Greeney HF, Meneses MR, Hamilton CE, Lichter‐Marck E, Mannan RW, Snyder N, Snyder H, Wethington SM, Dyer LA. 2015. Trait‐mediated trophic cascade creates enemy‐free space for nesting hummingbirds. Science Advances 1: e1500310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hengxiao G, McMillin JD, Wagner MR, Zhou J, Zhou Z, Xu X. 1999. Altitudinal variation in foliar chemistry and anatomy of yunnan pine, Pinus yunnanensis, and pine sawfly (Hym., Diprionidae) performance. Journal of Applied Entomology 123: 465–471. [Google Scholar]
- Hodkinson ID. 2005. Terrestrial insects along elevation gradients: species and community responses to altitude. Biological Reviews 80: 489–513. [DOI] [PubMed] [Google Scholar]
- Hughes AR, Inouye BD, Johnson MTJ, Underwood N, Vellend M. 2008. Ecological consequences of genetic diversity. Ecology Letters 11: 609–623. [DOI] [PubMed] [Google Scholar]
- Jeffrey CS, Leonard MD, Glassmire AE, Dodson CD, Richards LA, Kato MJ, Dyer LA. 2014. Antiherbivore prenylated benzoic acid derivatives from Piper kelleyi . Journal of Natural Products 77: 148–153. [DOI] [PubMed] [Google Scholar]
- Johann S, Cota BB, Souza‐Fagundes EM, Pizzolatti MG, Resende MA, Zani CL. 2009. Antifungal activities of compounds isolated from Piper abutiloides Kunth. Mycoses 52: 499–506. [DOI] [PubMed] [Google Scholar]
- Johnson MTJ, Agrawal AA. 2005. Plant genotype and environment interact to shape a diverse arthropod community on evening primrose (Oenothera biennis). Ecology 86: 874–885. [Google Scholar]
- Johnson MTJ, Agrawal AA, Maron JL, Salminen JP. 2009. Heritability, covariation and natural selection on 24 traits of common evening primrose (Oenothera biennis) from a field experiment. Journal of Evolutionary Biology 22: 1295–1307. [DOI] [PubMed] [Google Scholar]
- Johnson MTJ, Lajeunesse MJ, Agrawal AA. 2006. Additive and interactive effects of plant genotypic diversity on arthropod communities and plant fitness. Ecology Letters 9: 24–34. [DOI] [PubMed] [Google Scholar]
- Jost L. 2006. Entropy and diversity. Oikos 113: 363–375. [Google Scholar]
- Krause GH, Schmude C, Garden H, Koroleva OY, Winter K. 1999. Effects of solar ultraviolet radiation on the potential efficiency of photosystem II in leaves of tropical plants. Plant Physiology 121: 1349–1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Durbin R. 2009. Fast and accurate short read alignment with Burrows‐Wheeler transform. Bioinformatics 25: 1754–1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R. 2009. The sequence alignment/map format and SAMtools. Bioinformatics 25: 2078–2079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lichstein JW. 2007. Multiple regression on distance matrices: a multivariate spatial analysis tool. Plant Ecology 188: 117–131. [Google Scholar]
- McCloud ES, Berenbaum MR. 1999. Effects of enhanced UV‐B radiation on a weedy forb (Plantago laneolata) and its interactions with a generalist and specialist herbivore. Entomologia Experimentalis et Applicata 93: 233–247. [Google Scholar]
- Padwa A, Au A, Lee GA, Owens W. 1996. Photochemical ring‐opening reactions of substituted chromenes and isochromenes. The Journal of Organic Chemistry 61: 9072. [DOI] [PubMed] [Google Scholar]
- Parchman TL, Gompert Z, Braun MJ, Brumfield RT, McDonald DB, Uy JAC, Zhang G, Jarvis ED, Schlinger BA, Buerkle CA. 2013. The genomic consequences of adaptive divergence and reproductive isolation between species of manakins. Molecular Ecology 22: 3304–3317. [DOI] [PubMed] [Google Scholar]
- Pellissier L, Fiedler K, Ndribe C, Dubuis A, Pradervand J‐N, Guisan A, Rasmann S. 2012. Shifts in species richness, herbivore specialization, and plant resistance along elevation gradients. Ecology and Evolution 2: 1818–1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellissier L, Roger A, Bilat J, Rasmann S. 2014. High elevation Plantago lanceolata plants are less resistant to herbivory than their low elevation conspecifics: is it just temperature? Ecography 37: 950–959. [Google Scholar]
- Poelman EH, Dam NM, Loon JJA, Vet LEM, Dicke M. 2009. Chemical diversity in Brassica oleracea affects biodiversity of insect herbivores. Ecology 90: 1863–1877. [DOI] [PubMed] [Google Scholar]
- R Development Core Team . 2013. R: language and environment for statistical computing. [WWW document] URL http://www.R-project.org/. Vienna, Austria: R Foundation for Statistical Computing. [Google Scholar]
- Raguso RA. 2009. Floral scent in a whole‐plant context: moving beyond pollinator attraction. Functional Ecology 23: 837–840. [Google Scholar]
- Rasmann S, Alvarez N, Pellissier L. 2014. The altitudinal niche‐breadth hypothesis in insect‐plant interactions In: Voelckel C, Jander G, eds. Annual plant reviews, volume 47, insect–plant interactions. Chichester, UK: John Wiley & Sons Ltd, 339–359. [Google Scholar]
- Richards LA, Dyer LA, Forister ML, Smilanich AM, Dodson CD, Leonard MD, Jeffrey CS. 2015. Phytochemical diversity drives plant–insect community diversity. Proceedings of the National Academy of Sciences, USA 112: 10973–10978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards LA, Dyer LA, Smilanich AM, Dodson CD. 2010. Synergistic effects of amides from two Piper species on generalist and specialist herbivores. Journal of Chemical Ecology 36: 1105–1113. [DOI] [PubMed] [Google Scholar]
- Richards LA, Lampert EC, Bowers MD, Dodson CD, Smilanich AM, Dyer LA. 2012. Synergistic effects of iridoid glycosides on the survival, development and immune response of a specialist caterpillar, Junonia coenia (Nymphalidae). Journal of Chemical Ecology 38: 1276–1284. [DOI] [PubMed] [Google Scholar]
- Rodríguez‐Castañeda G, Brehm G, Fiedler K, Dyer LA. 2016. Ant predation on herbivores through a multitrophic lens: how effects of ants on plant herbivore defense and natural enemies vary along temperature gradients. Current Opinions in Insect Science 14: 73–80. [DOI] [PubMed] [Google Scholar]
- Rodríguez‐Castañeda G, Dyer LA, Brehm G, Connahs H, Forkner RE, Walla TR. 2010. Tropical forests are not flat: how mountains affect herbivore diversity. Ecology Letters 13: 1348–1357. [DOI] [PubMed] [Google Scholar]
- Root RB. 1973. Organization of a plant‐arthropod association in simple and diverse habitats: the fauna of collards (Brassica oleracea). Ecological Monographs 43: 95–124. [Google Scholar]
- Ruhland CT, Dyslin MJ, Krenz JD. 2013. Wyoming big sagebrush screens ultraviolet radiation more effectively at higher elevations. Journal of Arid Environments 96: 19–22. [Google Scholar]
- Salmore AK, Hunter MD. 2001. Elevational trends in defense chemistry, vegetation, and reproduction in Sanguinaria canadensis . Journal of Chemical Ecology 27: 1713–1727. [DOI] [PubMed] [Google Scholar]
- Schweitzer JA, Bailey JK, Hart SC, Whitham TG. 2005. Nonadditive effects of mixing cottonwood genotypes on litter decomposition and nutrient dynamics. Ecology 86: 2834–2840. [Google Scholar]
- Shipley B. 2002. Cause and correlation in biology: a user's guide to path analysis, structural equations and causal inference. New York, NY, USA: Cambridge University Press. [Google Scholar]
- Smilanich AM, Dyer LA, Chambers JQ, Bowers MD. 2009. Immunological cost of chemical defence and the evolution of herbivore diet breadth. Ecology Letters 12: 612–621. [DOI] [PubMed] [Google Scholar]
- Tahvanainen JO, Root RB. 1972. The influence of vegetational diversity on the population ecology of a specialized herbivore, Phyllotreta cruciferae (Coleoptera: Chrysomelidae). Oecologia 10: 321–346. [DOI] [PubMed] [Google Scholar]
- Tepe EJ, Rodríguez‐Castañeda G, Glassmire AE, Dyer LA. 2014. Piper kelleyi, a hotspot of ecological interactions and a new species from Ecuador and Peru. PhytoKeys 34: 19–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson JN. 1999. Specific hypotheses on the geographic mosaic of coevolution. The American Naturalist 153(S5): S1–S14. [Google Scholar]
- Thompson JN. 2005. The geographic mosaic of coevolution. Chicago, IL, USA: University of Chicago Press. [Google Scholar]
- Turlings TCJ, Ton J. 2006. Exploiting scents of distress: the prospect of manipulating herbivore‐induced plant odours to enhance the control of agricultural pests. Current Opinion in Plant Biology 9: 421–427. [DOI] [PubMed] [Google Scholar]
- Virjamo V, Sutinen S, Julkunen‐Tiitto R. 2014. Combined effect of elevated UVB, elevated temperature and fertilization on growth, needle structure and phytochemistry of young Norway spruce (Picea abies) seedlings. Global Change Biology 20: 2252–2260. [DOI] [PubMed] [Google Scholar]
- Wäschke N, Hardge K, Hancock C, Hilker M, Obermaier E, Meiners T. 2014. Habitats as complex odour environments: how does plant diversity affect herbivore and parasitoid orientation? PLoS ONE 9: 1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitham TG, Bailey JK, Schweitzer JA, Shuster SM, Bangert RK, LeRoy CJ, Lonsdorf EV, Allan GJ, DiFazio SP, Potts BM. 2006. A framework for community and ecosystem genetics: from genes to ecosystems. Nature Reviews Genetics 7: 510–523. [DOI] [PubMed] [Google Scholar]
- Whitham TG, Young WP, Martinsen GD, Gehring CA, Schweitzer JA, Shuster SM, Wimp GM, Fischer DG, Bailey JK, Lindroth RL. 2003. Community and ecosystem genetics: a consequence of the extended phenotype. Ecology 84: 559–573. [Google Scholar]
- Wilson JS, Forister ML, Dyer LA, O'Connor JM, Burls K, Feldman CR, Jaramillo MA, Miller JS, Rodríguez‐Castañeda G, Tepe EJ et al 2012. Host conservatism, host shifts and diversification across three trophic levels in two Neotropical forests. Journal of Evolutionary Biology 25: 532–546. [DOI] [PubMed] [Google Scholar]
- Zangerl AR, Berenbaum MR. 1987. Furanocoumarins in wild parsnip: effects of photosynthetically active radiation, ultraviolet light, and nutrients. Ecology 68: 516–520. [Google Scholar]
- Zhang B, Segraves KA, Xue H‐J, Nie R‐E, Li W‐Z, Yang X‐K. 2015. Adaptation to different host plant ages facilitates insect divergence without a host shift. Proceedings of the Royal Society of London Series B: Biological Sciences 282: 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: Wiley Blackwell are not responsible for the content or functionality of any supporting information supplied by the authors. Any queries (other than missing material) should be directed to the New Phytologist Central Office.
Fig. S1 A map illustrating the locations of P. kelleyi plant and caterpillar samples that were collected near Yanayacu Biological Station near Cosanga, Napo Province, Ecuador in the eastern Andes (00°36′S and 77°53′W).
Fig. S2 Partial correlation plots from the structural equation model (Fig. 2a) for paths II, III, IV, V, VI and IX.
Fig. S3 Principal component analysis examining the chemical similarity between individual Piper kelleyi plants using concentrations of prenylated benzoic acid, chromene and dimeric chromane of individual plants.
Fig. S4 Linear regression examining the relationship between elevation and PC2 scores from the phytochemical PCA (Fig. S3).
Fig. S5 Linear regression examining the relationship between elevation and PC1 scores from the phytochemical PCA (Fig. S3).
Table S1 Variation explained by each component used in the principal components analysis
Table S2 The first three eigenvalues of the correlation matrix for the principal component analysis, the proportion of total variance, and the cumulative variance for each principal component


