Abstract
The oculomotor system involves a large number of brain areas including parts of the basal ganglia, and various neurodegenerative diseases including Parkinson's and Huntington's can disrupt it. People with Parkinson's disease, for example, tend to have increased saccadic latencies. Consequently, the quantitative measurement of saccadic eye movements has received considerable attention as a potential biomarker for neurodegenerative conditions. A lot more can be learned about the brain in both health and disease by observing what happens to eye movements when the function of specific brain areas is perturbed. Deep brain stimulation is a surgical intervention used for the management of a range of neurological conditions including Parkinson's disease, in which stimulating electrodes are placed in specific brain areas including several sites in the basal ganglia. Eye movement measurements can then be made with the stimulator systems both off and on and the results compared. With suitable experimental design, this approach can be used to study the pathophysiology of the disease being treated, the mechanism by which DBS exerts it beneficial effects, and even aspects of normal neurophysiology.
Keywords: Medicine, Issue 113, deep brain stimulation, saccadometry, quantitative measurements, subthalamic nucleus, Parkinson's disease, neurophysiology, neuroscience
Introduction
In recent years there has been increasing interest in the use of measurements of reaction times as a quantitative and non-invasive way of gaining information about the high level mechanisms of neural decision making 1. One type of reaction time that has been studied extensively is the time taken to initiate a saccade on presentation of a visual stimulus, known as saccadic latency. Saccades are the fast eye movements that occur when we rapidly shift our gaze from one place to another. They are the commonest type of eye movements we make, occurring at a frequency of typically two or three per second. Each saccade is in effect a decision to look at one cue in the visual world rather than another 2.
The neural pathways controlling eye movements have been studied extensively and are fairly well documented 3. Using sensitive electronic equipment, aspects of oculomotor function can be precisely and objectively quantified. This facilitates the detailed study of eye movements themselves but also allows them to be used as a tool to investigate other areas of neurophysiology and pathophysiology.
Eye movement measurement can provide useful information about disease states. Saccadic eye movements have recently, for example, received much attention as potential biomarkers in neurodegenerative disorders including Huntington's 4,5 and Parkinson's diseases 6,7, and it is well established that saccadic reaction times tend to be slower than normal in these conditions. Potential uses of saccadic measurement include aids to diagnosis and disease tracking. Saccadic tasks range from the simple prosaccade (looking as quickly as possible toward a suddenly appearing visual stimulus to left or right) to more complex tasks such as the antisaccade (looking as quickly as possible to the opposite side to a visual stimulus) or memory-guided saccade (looking towards the remembered location of a target that is no longer there).
Deep brain stimulation is an effective treatment for several neurological conditions. It is most commonly used to treat the motor symptoms of Parkinson's disease including tremor, rigidity, bradykinesia, and dyskinesia. It is also used for other movement disorders including dystonia and essential tremor, and less commonly for neuropathic pain, epilepsy, and psychiatric conditions such as obsessive compulsive disorder. It is the only setting in which scientists have direct electrical access to deep structures of the human brain in vivo and thus offers a precious opportunity for experimental neurology. A variety of targets are stimulated depending on the condition being treated, including several locations in the basal ganglia, many of which are involved in oculomotor pathways. This means that a wide range of studies can be conducted using the DBS system to deliver stimulation to a given brain location and an eye tracking device to record and analyze its effects. Depending on the experimental paradigm, such studies may yield information about the physiology of the region being stimulated, the effects of the disease, or the mechanism by which DBS is working in that particular setting. This article describes a general approach to saccadic eye movement testing in Deep Brain Stimulation patients.
Several different types of eye tracking equipment are available. For the research described in this protocol a portable saccadometer was used to record horizontal saccadic eye movements. Portable saccadometers have the advantage of not requiring head restraint (see Figure 1), which means that sessions are more comfortable for patients with Parkinson's disease, especially for those suffering with severe dyskinesias. The saccadometer used here is lightweight and approximately 5 cm wide and 10 cm tall. The saccadometer measures eye movements by the use of direct infrared oculography: an infra-red source and sensor positioned in front of the medial canthus use light reflected from the cornea to establish the rotational position of the eyeball at millisecond intervals. In order to acquire good quality data for analysis the saccadometer should sample at a rate of at least 1 kHz with at least a 12 bit resolution. In the saccadometer used here the visual stimuli were three red 13 cd m-2 spots of light produced by built in low power lasers, each spot subtending some 0.1 degrees, with one spot in the midline and the other two at ± 10 degrees (i.e., to the right and left).
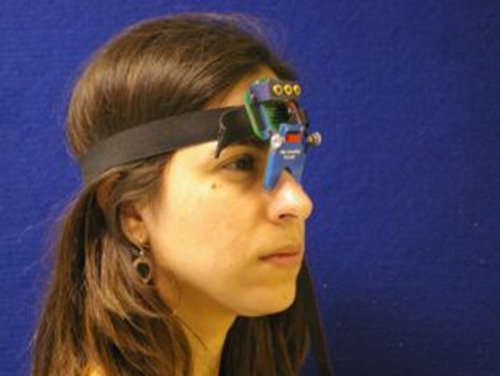 Figure 1. The Saccadometer. Head mounted saccadometer attached to an elastic band and resting on the bridge of the nose. Four miniature lasers project visual targets on to a matte surface, and the participant's eye movements are measured by differential infrared reflectance transducers on the nasal side of each eye. As the laser targets move with the head, head restraints are not required. Please click here to view a larger version of this figure.
Figure 1. The Saccadometer. Head mounted saccadometer attached to an elastic band and resting on the bridge of the nose. Four miniature lasers project visual targets on to a matte surface, and the participant's eye movements are measured by differential infrared reflectance transducers on the nasal side of each eye. As the laser targets move with the head, head restraints are not required. Please click here to view a larger version of this figure.
Protocol
The local ethics committee approved this study and informed consented was obtained from the participants as detailed below in section 1.
1. Participant Consent
Provide participants with an information sheet that explains in detail what the testing session will include.
After the participants have the opportunity to read and discuss any questions, concerns or other matters related to their taking part in the study, go through the consent form with them, explaining each point on the consent form and giving them the opportunity to ask any questions they might have. Ask the participant to complete the form.
2. Setting up the Saccadometer
Place the device on the patient's head, secured by an adjustable elastic strap and resting on the bridge of the nose. Because the stimuli move exactly with the head, no head-restraint is necessary and the device is comfortable to wear.
Ask the patient to sit at 1.5 m from a flat matte screen.
Ensure that the ambient lighting is dim so that the stimuli (red light spots) are clearly seen.
3. Recording a Saccadic Session
Note: As an example a standard protocol that tests both prosaccades and antisaccades 8 is described here. This protocol consists of five blocks: 60 prosaccades, 40 antisaccades x 3, and 60 prosaccades with a break of 1 min between blocks. The session lasts about 40 min.
Set up the saccadometer so that for each trial the central fixation target is displayed for a random fore-period of 1.0 - 2.0 sec, after which it is extinguished and one of the peripheral targets appears, randomly to right or left 9. Note: The three targets are spots of red light projected onto the screen in front of the participant (see 2.2) by low powered lasers built into the saccadometer. The saccadometer automatically switches the lasers on and off to display/extinguish the targets in the necessary sequence.
Instruct the participant that the testing consists of five blocks with a one minute break between blocks.
Prior to the first block instruct the participant to move their eyes as quickly and accurately as possible to follow the red dot jumping from the middle to one side or the other and instruct them to do this 60 times.
Set the saccadometer to generate a sequence of 60 trials. Press the button on the saccadometer to start the first block of trials (prosaccades).
After completion of the first block, leave a one minute gap before the second block. Reset the saccadometer to generate a sequence of 40 trials, and towards the end of the one minute gap, instruct the participant for the next block to move their eyes as quickly as possible in the opposite direction to the red dot, and explain that they will be required to do this 40 times. Start the second block of trials (antisaccades).
After completion of the second block, leave a further 1 min gap, repeat the instruction in step 3.5 and start the third block of trials (antisaccades).
After completion of the third block, leave a further 1 min gap, repeat the instruction in step 3.5 and start the fourth block of trials (antisaccades).
After completion of the fourth block, leave a further 1 min gap, and explain that for the final block of tests ask the participants to move their eyes as quickly and accurately as possible to follow the red dot jumping from the middle to one side or the other, exactly as they did in the first block.
Reset the saccadometer to generate a sequence of 60 trials, and start the final block of trials (prosaccades).
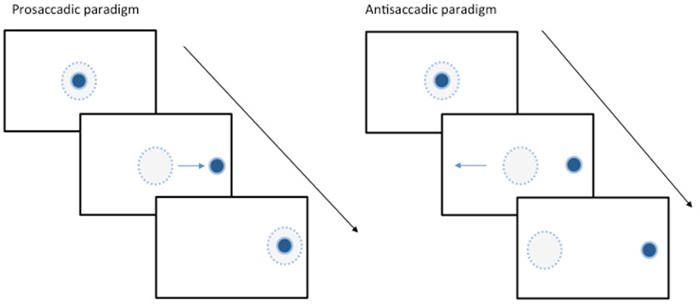 Figure 2. Eye Movement Tasks. Schematic illustration showing two examples of saccadic tasks. The solid blue spot represents the target and the dotted blue circle represents the area of fixation. LEFT shows a prosaccadic task where the subject is prompted to look towards the target. RIGHT shows an antisaccade where the subject is asked to look away from the visual stimulus. This requires inhibition of the more natural prosaccade response and generation of a saccade in the opposite direction. Please click here to view a larger version of this figure.
Figure 2. Eye Movement Tasks. Schematic illustration showing two examples of saccadic tasks. The solid blue spot represents the target and the dotted blue circle represents the area of fixation. LEFT shows a prosaccadic task where the subject is prompted to look towards the target. RIGHT shows an antisaccade where the subject is asked to look away from the visual stimulus. This requires inhibition of the more natural prosaccade response and generation of a saccade in the opposite direction. Please click here to view a larger version of this figure.
4. Deep Brain Stimulator Settings
Note: For participants with deep brain stimulators conduct the testing so far with stimulator system running as normal, i.e., an 'on stimulation' dataset has been obtained. Testing now needs to be repeated with the stimulator system turned off (for healthy control participants without DBS systems this section will not apply).
Switch the DBS system off. Do this by a trained clinical staff. Allow 30 min before testing.
Repeat the saccadic testing (steps 3.2 - 3.9) to obtain a complete 'off stimulation' dataset.
Switch the DBS system back on (again this should be done by appropriately trained clinical staff).
5. Data Analysis
Note: For participants with deep brain stimulators conduct the testing so far with stimulator system running as normal, i.e., an 'on stimulation' dataset has been obtained. Testing now needs to be repeated with the stimulator system turned off (for healthy control participants without DBS systems this section will not apply).
Download the raw data from the saccadometer to a computer for analysis.
- Use the saccadometer's software program to exclude saccades distorted by blinks and head movements, and to calculate variables including saccadic latencies, peak velocities, and amplitudes. Note: Records contaminated by excessive head movements or blinks are automatically removed by the software.
- Remove saccades with latency less than 80 msec or greater than 1,000 msec. Note: Saccadic latency is computed automatically using a saccade-detection algorithm based on velocity and acceleration. The onset of a saccade is identified as the point when the eye velocity exceeds a threshold of 5 degrees/sec.
Representative Results
Figure 3 shows an example of saccadic eye movement trajectories, from a Parkinson's disease patient with a subthalamic nucleus DBS system implanted. The two graphs plot the patient's prosaccades with the stimulator system switched off (upper graph) and switched on (lower graph). Each trace on the graphs shows the trajectory of a single saccade, i.e., how the eye position in degrees away from the midline (y axis) varies as a function of time (x axis). Both leftward and rightward saccades are shown on the graph as deflections to positive degree values. Time zero is the moment that the central target disappears and the peripheral target appears. The interval between this and the onset of the saccade (the moment when the trace deflects from zero degrees) is termed saccadic latency, and the main observation is that the distribution of saccades is altered when the stimulator is switched on, with a reduction in the number of long latency saccades and a corresponding decrease in mean latency.
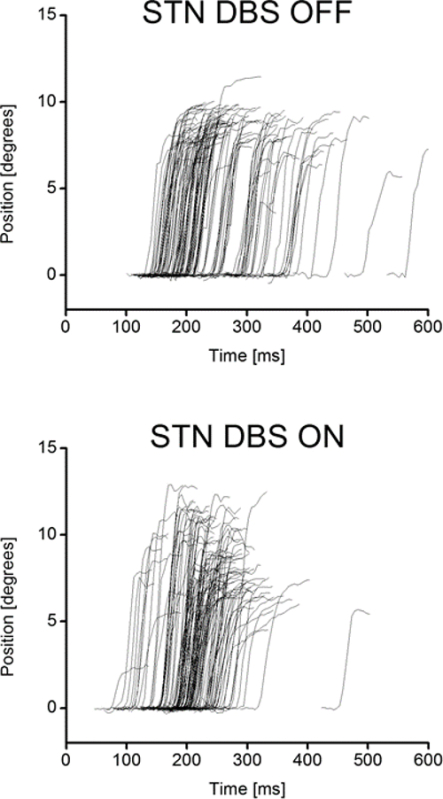 Figure 3. Prosaccadic Trajectory Results. This shows the saccadic eye movement latency results from a Parkinson's patient after undergoing Deep Brain Stimulation of the subthalamic nucleus. UPPER shows the latency profile of the patient when the stimulator is switched off LOWER shows the latency profile of the same patient when the stimulator is switched on. Please click here to view a larger version of this figure.
Figure 3. Prosaccadic Trajectory Results. This shows the saccadic eye movement latency results from a Parkinson's patient after undergoing Deep Brain Stimulation of the subthalamic nucleus. UPPER shows the latency profile of the patient when the stimulator is switched off LOWER shows the latency profile of the same patient when the stimulator is switched on. Please click here to view a larger version of this figure.
Discussion
The most critical factor in obtaining good quality saccadic data is ensuring that the instructions given to the participant are clear and precise. For example, if the instructions for the antisaccadic task are not completely clear, the participant is likely to execute prosaccades instead. Recordings may also be spoiled if the participant cannot clearly see the stimuli or the saccadometer cannot accurately gauge eye position. Thus if the data appear to be of low quality the experimenter should check that the ambient light is not too bright and that the saccadometer is sitting properly on the bridge of the nose.
Deep brain stimulation allows the direct modification of neural activity at a various locations in the basal ganglia. Coupled with equipment that can accurately and objectively quantify eye movements, DBS can be used to investigate the normal and abnormal functioning of these brain areas.
Experiments using saccadometry in conjunction with DBS must often contend with more than one unknown simultaneously. The brains under study are diseased to a variable extent, and we do not know for certain the mechanism by which DBS is achieving its effects, with hypotheses including activation of neurons in the targeted area, depolarizing blockade of neurons, stimulation of afferent and/or efferent axons, and more complex effects on network activity.
Subjects with neurodegenerative conditions are frequently on medication and this will be nearly universal for patients with PD patients who have DBS systems. Saccades are affected by antiparkinsonian medication, and in order to make a valid assessment of the effects of DBS on saccadic parameters, medication state must be similar when testing with DBS on and DBS off. This means either having the subject off all medication for the duration of testing (including a withdrawal period beforehand), or else conducting on-DBS and off-DBS tests within a reasonably short period of time. It has been shown that the complete wearing off of the effects of DBS on saccades takes several hours but that most of the change takes place within 30 min 10. The time course suggests multiple mechanisms with different time courses, from very fast (electrical effects) to much more prolonged (e.g., protein synthesis). The 30 min suggested in the protocol will therefore capture most of the DBS induced change, and is much shorter than the typical dosing interval of several hours. It does not give adequate time for the slower mechanisms to settle to steady state, and retesting at longer intervals may be useful in providing insight into these mechanisms. With longer gaps one could not assume that medication levels while testing on and off DBS were approximately similar, thus longer intervals are likely to be easier to interpret when testing off medication.
Most of the literature on the effects of DBS on eye movements has focused on high frequency stimulation of the subthalamic nucleus in PD. Prosaccadic latency is prolonged in PD and it is well established that STN DBS can substantially reduce it toward normal values (exactly how remains open to debate). However when saccadometry is performed in the early postoperative period, within hours to days of lead insertion and before electrical stimulation is switched on, it is found that latencies are actually increased 11. Electrode insertion causes edema and this will lead to temporary functional ablation of the brain immediately surrounding the electrode; this settles within a few weeks and saccadic parameters return to baseline (stimulators are commonly not turned on until a few weeks postoperatively to allow this time to settle). The oppositeness of the effects of electrode insertion and stimulation is of considerable interest, because it illustrates that one of the theories previously advanced to explain how DBS is working, by blockade of the stimulated structure (i.e., functional ablation), cannot comprehensively explain its mechanism of action. Other studies have found similar effects 12-14.
It is possible to construct experiments combining DBS and saccadometry to investigate normal physiology. Great care must be taken with experimental design and interpretation of results, because the experimental subject necessarily has some neurological condition. One approach is to compare the results of saccadic testing with DBS off and on to results from healthy control participants without DBS. If the results in DBS subjects with their stimulators turned off resemble those in controls, then it is reasonable to proceed on the basis that changes in the eye movements seen in response to DBS are likely to be similar to those which would be seen if the experiment could be done in participants without disease. For example, the performance in a prosaccade task which has been set up so that the visual stimulus is more likely to appear on one side than the other, is similar for healthy participants and PD patients with STN DBS systems in place but turned off. Latencies shorten for saccades in the direction where the target is more likely to appear, and lengthen in the less likely direction. When the systems are turned on, the lengthening of latency in the less likely direction disappears, and the interpretation was that the STN is performing a probability normalization function 15. The patients in this study had PD, but that was not directly relevant to the result. Of course this is a significant assumption and it is possible that the disease modulates the response to DBS even though it did not affect the baseline performance. Furthermore, the similarity of off-DBS and control data may be the result of compensatory mechanisms rather than a lack of effect of pathology on the parameters being measured. Results must be interpreted with this in mind.
Once the basic techniques described in the protocol have been mastered the approach can be extended to more complex paradigms. Many different saccadic tasks have been developed of which the probability based task described in the previous paragraph is just one. Further examples include memory guided saccades 16 (looking towards the remembered location of a stimulus that is no longer there) or reward-based saccadic tasks designed to invoke limbic function 17.
Here we have described one of the simplest saccadic tasks, a 10-degree visually guided horizontal step task. Many modifications of the task are possible such as inserting a temporal gap between fixation point disappearance and peripheral target appearance 18-20 or an overlap 21 where fixation point and lateral target are present simultaneously. Paradigms of this sort have been used in order to understand saccadic initiation and neural correlates have been observed in the frontal eye fields 20,22 and the superior colliculus 23. A detailed discussion of the various uses of different saccadic paradigms is beyond the scope of this article; for a review see 1.
Saccadometry has wide application outside its use in DBS. Saccadic pathways involve multiple parts of the basal ganglia and saccadic changes are a potential biomarker for any condition that damages or perturbs the function of these structures. Saccadometry has for example been investigated for use in neurodegenerative conditions including PD, Huntington's disease, frontotemporal dementia, amyotrophic lateral sclerosis as well as in head injury and in metabolic disorders including hepatic encephalopathy.
In conclusion, saccadometry is a useful quantitative tool by itself, but combining it with deep brain stimulation opens up a range of experimental approaches that may shed new light on brain function in both health and disease.
Disclosures
The authors have nothing to disclose.
Acknowledgments
Dr. Antoniades was supported by the National Institute of Health Research (NIHR) and by the Dementias and Neurodegenerative Diseases Research Network (DENDRON) and by the Wellcome Trust. Dr FitzGerald was supported by the National Institute for Health Research (NIHR) Oxford Biomedical Research Centre.
References
- Leigh RJ, Kennard C. Using saccades as a research tool in the clinical neurosciences. Brain. 2004;127:460–477. doi: 10.1093/brain/awh035. [DOI] [PubMed] [Google Scholar]
- Carpenter RH. The neural control of looking. Curr Biol. 2000;10:291–293. doi: 10.1016/s0960-9822(00)00430-9. [DOI] [PubMed] [Google Scholar]
- Leigh RJ, Zee DS. The Neurology of Eye Movements. New York: Oxford University Press; 2006. [Google Scholar]
- Antoniades CA, Xu Z, Mason SL, Carpenter RH, Barker RA. Huntington's disease: changes in saccades and hand-tapping over 3 years. Journal of Neurology. 2010;257:1890–1898. doi: 10.1007/s00415-010-5632-2. [DOI] [PubMed] [Google Scholar]
- Blekher TM, Yee RD, Kirkwood SC, Hake AM, Stout JC, Weaver MR, Foroud TM. Oculomotor control in asymptomatic and recently diagnosed individuals with the genetic marker for Huntington's disease. Vision Research. 2004;44:2729–2736. doi: 10.1016/j.visres.2004.06.006. [DOI] [PubMed] [Google Scholar]
- Chan F, Armstrong IT, Pari G, Riopelle RJ, Munoz DP. Deficits in saccadic eye-movement control in Parkinson's disease. Neuropsychologia. 2005;43:784–796. doi: 10.1016/j.neuropsychologia.2004.06.026. [DOI] [PubMed] [Google Scholar]
- Antoniades CA, Demeyere N, Kennard C, Humphreys GW, Hu MT. Antisaccades and executive dysfunction in early drug-naive Parkinson's disease: The discovery study. Mov Disord. 2015. [DOI] [PubMed]
- Antoniades C, et al. An internationally standardised antisaccade protocol. Vision Res. 2013;84:1–5. doi: 10.1016/j.visres.2013.02.007. [DOI] [PubMed] [Google Scholar]
- Ober JK, et al. Hand-Held system for ambulatory measurement of saccadic durations of neurological patients. Modelling and Measurement in Medicine. 2003.
- Temperli P, et al. How do parkinsonian signs return after discontinuation of subthalamic DBS. Neurology. 2003;60:78–81. doi: 10.1212/wnl.60.1.78. [DOI] [PubMed] [Google Scholar]
- Antoniades CA, et al. Deep brain stimulation: eye movements reveal anomalous effects of electrode placement and stimulation. PLoS ONE. 2012;7:e32830. doi: 10.1371/journal.pone.0032830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yugeta A, et al. Effects of STN stimulation on the initiation and inhibition of saccade in Parkinson disease. Neurology. 2010;74:743–748. doi: 10.1212/WNL.0b013e3181d31e0b. [DOI] [PubMed] [Google Scholar]
- Terao Y, Fukuda H, Ugawa Y, Hikosaka O. New perspectives on the pathophysiology of Parkinson's disease as assessed by saccade performance: a clinical review. Clin Neurophysiol. 2013;124:1491–1506. doi: 10.1016/j.clinph.2013.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Temel Y, Visser-Vandewalle V, Carpenter RH. Saccadic latency during electrical stimulation of the human subthalamic nucleus. Curr Biol. 2008;18:412–414. doi: 10.1016/j.cub.2008.03.008. [DOI] [PubMed] [Google Scholar]
- Antoniades CA, et al. Deep brain stimulation abolishes slowing of reactions to unlikely stimuli. J Neurosci. 2014;34:10844–10852. doi: 10.1523/JNEUROSCI.1065-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivaud-Pechoux S, et al. Improvement of memory guided saccades in parkinsonian patients by high frequency subthalamic nucleus stimulation. J Neurol Neurosurg Psychiatry. 2000;68:381–384. doi: 10.1136/jnnp.68.3.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takikawa Y, Kawagoe R, Itoh H, Nakahara H, Hikosaka O. Modulation of saccadic eye movements by predicted reward outcome. Experimental brain research. Experimentelle Hirnforschung. 2002;142:284–291. doi: 10.1007/s00221-001-0928-1. [DOI] [PubMed] [Google Scholar]
- Dorris MC, Munoz DP. A neural correlate for the gap effect on saccadic reaction times in monkey. Journal of Neurophysiology. 1995;73:2558–2562. doi: 10.1152/jn.1995.73.6.2558. [DOI] [PubMed] [Google Scholar]
- Hanes DP, Schall JD. Countermanding saccades in macaque. Visual Neuroscience. 1995;12:929–937. doi: 10.1017/s0952523800009482. [DOI] [PubMed] [Google Scholar]
- Opris I, Barborica A, Ferrera VP. On the gap effect for saccades evoked by electrical microstimulation of frontal eye fields in monkeys. Experimental brain research. Experimentelle Hirnforschung. 2001;138:1–7. doi: 10.1007/s002210100686. [DOI] [PubMed] [Google Scholar]
- Takagi M, Frohman EM, Zee DS. Gap-overlap effects on latencies of saccades, vergence and combined vergence-saccades in humans. Vision Res. 1995;35:3373–3388. doi: 10.1016/0042-6989(95)00073-n. [DOI] [PubMed] [Google Scholar]
- Schall JD. Neuronal activity related to visually guided saccades in the frontal eye fields of rhesus monkeys: comparison with supplementary eye fields. Journal of Neurophysiology. 1991;66:559–579. doi: 10.1152/jn.1991.66.2.559. [DOI] [PubMed] [Google Scholar]
- Pare M, Hanes DP. Controlled movement processing: superior colliculus activity associated with countermanded saccades. J Neurosci. 2003;23:6480–6489. doi: 10.1523/JNEUROSCI.23-16-06480.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]


