Abstract
This paper describes a fabrication protocol for a dipole-assisted solid phase extraction (SPE) microchip available for trace metal analysis in water samples. A brief overview of the evolution of chip-based SPE techniques is provided. This is followed by an introduction to specific polymeric materials and their role in SPE. To develop an innovative dipole-assisted SPE technique, a chlorine (Cl)-containing SPE functionality was implanted into a poly(methyl methacrylate) (PMMA) microchip. Herein, diverse analytical techniques including contact angle analysis, Raman spectroscopic analysis, and laser ablation-inductively coupled plasma-mass spectrometry (LA-ICP-MS) analysis were employed to validate the utility of the implantation protocol of the C-Cl moieties on the PMMA. The analytical results of the X-ray absorption near-edge structure (XANES) analysis also demonstrated the feasibility of the Cl-containing PMMA used as an extraction medium by virtue of the dipole-ion interactions between the highly electronegative C-Cl moieties and the positively charged metal ions.
Keywords: Bioengineering, Issue 114, Solid phase extraction, dipole-assisted extraction, trace metal analysis, microchip fabrication, polymer-based microchips, microchannel orientation.
Introduction
From the viewpoints of environmental management and contamination prevention, trace metals which cause serious pollution or toxicological problems are a worldwide concern. An appropriate on-chip sample pretreatment technique has been widely accepted as the key to success in processing and analyzing actual samples via chip-based platforms, because unexpected co-existing chemical species in raw samples often hamper the accurate determination of analytes present in traces amounts.1 Among the available techniques, on-chip solid phase extraction (SPE) is especially popular for trace metal analyses, because this technique permitting sample cleanup and analyte preconcentration to be performed simultaneously is extremely useful for isolation of metal ions from complicated salt matrices.2,3
The advancement of on-chip SPE techniques used for the determination of trace metals has been steadily evolving. In the early days, the SPE chips were prepared by loading commercially available resins into the microchannels to construct the resin-packed SPE units.4-7 This occasionally required the analyte to be derivatized to enable the transformation of metal ions into resin-retainable forms.4 An alternative method for the preparation of chip-based SPE devices is to utilize the chip channel as an SPE sorbent for the collection of trace metals after simple surface modification.8 Recent years have seen an emerging trend involving the incorporation of magnetic nanoparticles (MNPs) and specific chemicals that contain functional groups capable of the efficient retention of metal ions. In contrast to commercial resins, the MNPs are modified with compounds such as γ-mercaptopropyltrimethoxysilane (γ-MPTS)9 and aminobenzyl ethylenediaminetetraacetic acid (ABEDTA)10 after which they are packed into the microchannels with the aid of an external magnetic field to achieve the selective extraction of metal ions.
Although significant progress in the development of on-chip SPE techniques has been witnessed, the reported techniques typically function based on either ion exchange or chelation. The use of techniques such as these has the disadvantage of requiring unavoidable operational procedures, including those associated with conditioning, washing, or regeneration, to maintain the analytical performance. Unfortunately, the need for additional operational procedures not only extends the time required for each analysis but also risks causing high blank values and irreproducible results.11 Therefore, an alternative working strategy for on-chip SPE techniques is imperative for trace metal analyses.
In 1993, Watts and Chehimi12 found that metal ions have a retention tendency toward polymeric materials, and that most of analytes efficiently retained on a chloro (Cl)-containing polymeric material, poly(vinyl chloride) (PVC) except sodium ions. Therefore, in 2002, Eboatu et al.13 further reported on the sequestration of some toxic metal from solutions by PVC. Because this indicated that Cl-containing polymeric materials exhibited superior properties for analyte preconcentration and salt matrix elimination, chip-based devices with the Cl-containing SPE functionality were considered an attractive strategy for the development of a novel on-chip SPE technique for the determination of trace metal ions. Considering material features, such as ease of fabrication, desired chemical/mechanical properties, and optical clarity,14,15 this study took advantage of poly(methyl methacrylate) (PMMA) to fabricate a microdevice. Then, the Cl-containing SPE functionality was implanted into the fabricated device for the development of a novel on-chip SPE technique for the determination of trace metal ions.16
Remarkably, the reliance of the innovative extraction mechanism on the dipole-ion interactions between the highly electronegative C-Cl moieties in the channel interior and the positively charged metal ions makes it possible to avoid measures taken during general on-chip SPE procedures, leading to a dramatic reduction of either the contamination caused by the use of excess reagents or the labor attributed to additional steps. The protocol provided in this contribution will enable researchers from diverse backgrounds to fabricate the dipole-assisted SPE microchip for their work. Detailed characterization procedures for the fabricated microchip are described as well.
Protocol
Caution! Several chemicals (e.g., acrylamide, 1,1'-dichloroethene) used in these procedures are acutely toxic and carcinogenic. Consult all relevant material safety data sheets (MSDS) prior to use. Follow appropriate safety practices when performing the experiments.
Note: Unless otherwise stated, carry out all procedures at ambient temperature in a class 100 laminar flow hood.
1. Fabrication of the Dipole-assisted SPE Microchip
- Preparation of the PMMA MicrochipNote: The fabrication protocol of the chip was similar to that described elsewhere.8
- Draw the network pattern of the chip (Figure 1(a)) using computer aided design (CAD) software according to the manufacturer's protocol.
- Mount a PMMA sheet (350 mm (L) x 20 mm (W) x 2 mm (H)) on the working table of the laser micromachining system and then focus the laser source on the surface of the PMMA sheet.
- Select the Print in the CAD software and then set the Power, Speed, and Pen Mode as 45% (4.5 W), 13% (99.06 mm sec−1), and VECT through the control panel of the micromachining system. Note: Parameters such as Power, Speed and Pen Mode affecting the channel features were investigated in advance. The evaluation method was similar to that proposed by Yuan and Dasa.17 The parameters selected in this study were used to machine an appropriate channel for adaption to conduits without complicated academic purposes. People can select another condition for laser machining in accordance with their need.
- Print the drawn pattern and then machine the PMMA sheet by the laser micromachining system according to the manufacturer's protocol. Figure 1(b) displays the layout of the fabricated microchip. Figure 1(c) displays the photograph of the cross-section of the machined plate. Caution! Wear goggles when using the laser system to avoid severe eye damage due to exposure to the laser radiation. An appropriate exhaust system is recommended because of the production of fumes/smoke during laser machining.
- Drill three 1/16-inch diameter access holes for a sample inlet, a buffer inlet, and an eluent inlet on the bottom plate and one for a confluent outlet on the cover plate Figure 1(b). Caution! Avoid body contact with the drill bit during the machining procedures to prevent physical injury. Wearing gloves is prohibited when drilling.
- Immerse the machined plates into 1 L of 0.1% (w/v) sodium dodecyl sulfate (SDS) solution in 1-L beaker with agitation via an ultrasonic oscillator for 10 min.
- Replace the SDS solution with deionized water and agitate via an ultrasonic oscillator for 10 min.
- Replace the residual D. I. H2O with fresh one and then immerse the machined plates in 1 L of D. I. H2O with agitation via an ultrasonic oscillator for 10 min. Afterward, dry each cleaned plate with a gentle stream of nitrogen for 2 min.
- Align the two machined plates with the naked eye and then sandwich the two plates between two glass boards by using binder clips.
- Bond the two plates under compression at 105 °C for 30 min.
- Cool the sandwich to ambient temperature and then remove the binder clips and glass boards.
- Insert 1/16-inch outer diameter poly(etheretherketone) (PEEK) tubes into the access holes and then secure the conduits with a two-component epoxy-based adhesive.
- Dry the adhesive at ambient temperature for 12 hr.
- Modification of the Channel Interior of the PMMA MicrochipNote: The following partially refers to published procedures with slight modifications.8,18,19
- Deliver a saturated sodium hydroxide (NaOH) solution at a flow rate of 100 µl min-1 via a peristaltic pump into the microchip for 12 hr (72 ml total delivered volume).
- Remove the residual solution and then rinse the channel interior with D. I. H2O at a flow rate of 100 µl min-1 via a peristaltic pump for 30 min (3 ml total delivered volume).
- Remove the residual D. I. H2O and then deliver a 0.5% (v/v) nitric acid (HNO3) solution into the microchip at a flow rate of 100 µl min-1 via peristaltic pump for 30 min (3 ml total delivered volume).
- Remove the residual solution and then deliver a 50% (w/v) acrylamide solution into the microchip in the dark at a flow rate of 100 µl min-1 via peristaltic pump for 8 hr (48 ml total delivered volume).
- Remove the residual solution and then rinse the channel interior with D. I. H2O at a flow rate of 100 µl min-1 via a peristaltic pump for 30 min (3 ml total delivered volume).
- Pump air to remove the residual D. I. H2O with a peristaltic pump and then cover the microchip with an in-house-built photomask allowing the desired region of the extraction channel to be exposed to light. Note: The in-house-built photomask was made of a black paper (114 mm (L) x 22 mm (W)) that contained an open window (94 mm (L) x 2 mm (W)) allowing the desired region of the extraction channel to be exposed to light.
- Preparation of the Cl-Containing SPE Formation Solution
- Flush the inhibitor removal SPE cartridge with ethanol amounting to at least three cartridge-volumes.
- Flush the cartridge with 1,1'-dichloroethene amounting to at least three cartridge-volumes prior to use.
- Pass 1 ml of 1,1'-dichloroethene through the treated cartridge and then collect the fraction in a sample vial (20 ml) wrapped in aluminum foil.
- Add 491 μl 1,1'-dichloroethene into the solution containing 12 mg 2,2'-azobisisobutyronitrile (AIBN), 3.18 ml ethanol, and 1.65 ml hexanes in 100-ml glass bottle.
- Fill the chip channel with the Cl-containing SPE formation solution (approximately 200 μl) by syringe injection and then expose the microchip to ultraviolet (UV365) irradiation with a maximum emission wavelength of 365 nm for 10 min (light intensity ~2.65 mW cm-2). Caution! An appropriate exhaust system is recommended, because of the production of ozone during UV irradiation.
- Replace the residual solution with a fresh Cl-containing SPE formation solution (approximately 200 μl) by syringe injection and then expose the microchip to UV365 irradiation for 10 min again (light intensity ~2.65 mW cm-2).
- Repeat Step 1.2.9 18 times.
- Rinse the channel interior with ethanol at flow rate of 100 µl min-1 via peristaltic pump for 30 min (3 ml total delivered volume). After removing the residual solution with peristaltic pump, store the fabricated microchip in a zippered bag for the subsequent use.
2. Surface Verification of PMMA Modification
- Contact Angle Analysis
- Cut a PMMA sheet (350 mm (L) x 20 mm (W) x 2 mm (H)) into PMMA substrates (50 mm (L) x 20 mm (W) x 2 mm (H)) by the laser micromachining system.
- Immerse the PMMA substrates in 40 ml saturated NaOH solution in 50-ml conical tube and then agitate the resulting mixtures via a rocking shaker for 12 hr.
- Remove the residual solution and then rinse the PMMA substrates with 40 ml D. I. H2O.
- Immerse the PMMA substrates in 40 ml D. I. H2O and then agitate the resulting mixtures via a rocking shaker for 30 min.
- Remove the residual D. I. H2O. Immerse the PMMA substrates in a 40 ml of 0.5% (v/v) HNO3 solution and then agitate the resulting mixtures via a rocking shaker for 30 min.
- Remove the residual solution. Immerse the PMMA substrates in 40 ml of 50% (w/v) acrylamide solution and then agitate the resulting mixtures via a rocking shaker in the dark for 8 hr.
- Remove the residual solution and then rinse the PMMA substrates with 40 ml D. I. H2O.
- Immerse the PMMA substrates in 40 ml of D. I. H2O and then agitate the resulting mixtures via a rocking shaker for 30 min.
- Remove the residual D. I. H2O and then dry each PMMA substrate with a gentle stream of nitrogen for 2 min.
- Preparation of the Cl-Containing SPE Formation Solution
- Flush the inhibitor removal SPE cartridge with ethanol amounting to at least three cartridge-volumes.
- Flush the cartridge with 1,1'-dichloroethene amounting to at least three cartridge-volumes prior to use.
- Pass 6 ml of 1,1'-dichloroethene through the treated cartridge and then collect the fraction in a sample vial (20 ml) wrapped in aluminum foil.
- Add 4.91 ml 1,1'-dichloroethene into the solution containing 120 mg AIBN, 31.8 ml ethanol, and 16.5 ml hexanes in 100-ml glass bottle.
- Apply 2 ml of the Cl-containing SPE formation solution onto the surfaces of the PMMA substrates and then expose the substrates to UV365 irradiation for 10 min (light intensity ~2.65 mW cm-2). Caution! An appropriate exhaust system is recommended, because of the production of ozone during UV irradiation.
- Replace the residual solution with 2 ml of fresh Cl-containing SPE formation solution and then expose the substrates to UV365 irradiation for 10 min again (light intensity ~2.65 mW cm-2).
- Repeat Step 2.1.12 18 times.
- Remove the residual solution and then rinse the PMMA substrates with 40 ml ethanol in a 50-ml conical tube.
- Remove the residual solution and then rinse the PMMA substrates with 40 ml D. I. H2O.
- Remove the residual D. I. H2O and then dry each PMMA substrate with a gentle stream of nitrogen for 2 min.
- Drop 5 μl D. I. H2O onto the PMMA substrates and determine the contact angle by a contact angle meter according to manufacturer's protocol. Note: Use the average of three repeated measurements to determine the reported contact angles in each case.
- Laser Ablation (LA)-Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) Analysis
- Grind 8 g of PMMA beads into PMMA powders via a mortar and a pestle.
- Immerse the PMMA powders in 40 ml saturated NaOH solution in a 50-ml conical tube and then agitate the resulting mixtures via a rocking shaker for 12 hr.
- Remove the residual solution by a digital pipette with 5-ml tips and then rinse the PMMA powders with 40 ml D. I. H2O.
- Immerse the PMMA powders in 40 ml D. I. H2O and then agitate the resulting mixtures via a rocking shaker for 30 min.
- Remove the residual D. I. H2O. Immerse the PMMA powders in 40 ml of 0.5% (v/v) HNO3 solution and then agitate the resulting mixtures via a rocking shaker for 30 min.
- Remove the residual solution. Immerse the PMMA powders in 40 ml of 50% (w/v) acrylamide solution and then agitate the resulting mixtures via a rocking shaker in the dark for 8 hr.
- Remove the residual solution and then rinse the PMMA powders with 40 ml of D. I. H2O.
- Immerse the PMMA powders in 40 ml of D. I. H2O and then agitate the resulting mixtures via a rocking shaker for 30 min.
- Remove the residual D. I. H2O and then bake the PMMA powders at 60 °C for 8 hr.
- Preparation of the Cl-Containing SPE Formation Solution
- Flush the inhibitor removal SPE cartridge with ethanol amounting to at least three cartridge-volumes.
- Flush the cartridge with 1,1'-dichloroethene amounting to at least three cartridge-volumes prior to use.
- Pass 16 ml 1,1'-dichloroethene through the treated cartridge and then collect the fraction in a sample vial (20 ml) wrapped in aluminum foil.
- Add 14.73 ml 1,1'-dichloroethene into the solution containing 360 mg AIBN, 95.4 ml ethanol, and 49.5 ml hexanes in a 250-ml glass bottle.
- Mix the PMMA powders with 6 ml of Cl-containing SPE formation solution in a 50-ml conical tube, and equally transfer 1 ml of the mixture from conical tube into six wells of a 24-well tissue culture plate.
- Cover the tissue culture plate with a PMMA board and then expose the tissue culture plate to UV365 irradiation for 10 min (light intensity ~2.65 mW cm-2). Caution! An appropriate exhaust system is recommended, because of the production of ozone during UV irradiation.
- Replace the residual solution with 1 ml of fresh Cl-containing SPE formation solution of each well and then expose the tissue culture plate to UV365 irradiation for 10 min again (light intensity ~2.65 mW cm-2).
- Repeat Step 2.2.13 18 times.
- Remove the residual solution and then rinse the PMMA powders in each well with 1 ml ethanol.
- Remove the residual solution and then rinse the PMMA powders in each well with 1 ml D. I. H2O.
- Remove the residual D. I. H2O and then bake the PMMA powders at 60 °C for 8 hr.
- Compress the dried powders (1 g) to a pellet via a hydraulic press machine and then measure the signal for Cl by a LA-ICP-MS system. Note: The signal for Cl at m/z 35 was selected as an indicator for the implanted C-Cl moieties. A 193-nm laser was used as the ablation source. The Energy, Fluence, Spot Size, and Repetition Rate were set as 75%, 8.85 J cm-2, 100 µm, and 5 Hz. At least 7 repeated measurements for each result were required. The LA-ICP-MS analytical procedures refer to published procedures elsewhere.20
- Raman Spectroscopic Analysis
- Perform the protocol from Step 2.2.1 to Step 2.2.17.
- Compress the dried powders (1 g) to a pellet via a hydraulic press machine and then take the spectra by a Raman spectrometer. Note: Use a 780-nm laser line with maximum laser power of 100 mW as the photoexcitation source. Use the region of the Raman spectrum ranging from 550 to 900 cm-1 to investigate the attachment of the C-Cl moieties to the PMMA.
3. Characterization of the Dipole-assisted SPE Reaction
Perform the protocol from Step 2.2.1 to Step 2.2.17.
Immerse 0.5 g PMMA powders in 5 ml of 20% (w/v) manganese nitrate tetrahydrate (Mn(NO3)24H2O) solution and then equally mix the resulting mixtures with 5 ml of 40 mM maleate buffer solution.
Adjust the pH of the resulting mixtures to 8 by using a pure HNO3 solution and then agitate the mixtures via a rocking shaker for 1 hr.
Remove the residual solution and then bake the PMMA powders at 60 °C for 8 hr. Store the powders in 15-ml conical tube wrapped in aluminum foil for the X-ray absorption near-edge structure (XANES) analysis. Note: The Mn K-edge XANES spectra were collected by using 07A and 17C1 beamlines of the National Synchrotron Radiation Research Center (NSRRC, Hsinchu, Taiwan). The electron storage ring was operated with energy of 1.5 GeV and a current of 100-200 mA. A Si(111) double crystal monochromator was used for providing highly monochromatized photon beams with energy of 1 to 15 keV and resolving power (E/ΔE) of up to 5,000.The photon energy was calibrated by Mn standards using the known Mn K-edge absorption inflection point at 6539.0 eV. The Mn K-edge XANES spectra in the region between 6,530 and 6,570 eV were used to investigate the dipole-ion interactions for characterization of the proposed SPE reaction.
Representative Results
Figure 2 depicts the reaction that occurs during the channel modification procedures of the PMMA microchip. Contact angle analysis was used to monitor the surface changes during the proposed procedures. An LA-ICP-MS system and a dispersive Raman spectrometer were employed to verify the successful modification of the C-Cl moieties formation on the PMMA substrate (Figure 3(a),(b)). The proposed dipole-assisted SPE reaction was characterized by the XANES analysis (Figure 4).
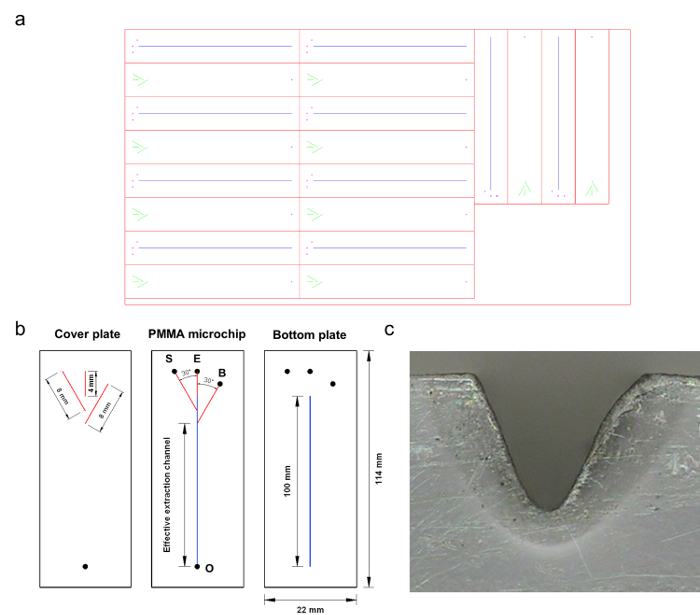 Figure 1. The PMMA microchip. (a) The snapshot of the pattern file for the fabricated microchip. (b) Layout of the fabricated microchip: S, E, and B represent the introduction ports for the sample, eluent, and buffer solutions, respectively; O represents the outlet. The black circle represents the drilled access hole for each. The channels used for the introduction of sample and buffer solutions both formed an angle of 30° with the extraction channel. The length of the effective extraction channel, which was defined as the distance from the convergence point of the flows of the sample and buffer solutions to the confluent outlet, was 94 mm. (c) The photograph of the cross-section of the machined plate. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
Figure 1. The PMMA microchip. (a) The snapshot of the pattern file for the fabricated microchip. (b) Layout of the fabricated microchip: S, E, and B represent the introduction ports for the sample, eluent, and buffer solutions, respectively; O represents the outlet. The black circle represents the drilled access hole for each. The channels used for the introduction of sample and buffer solutions both formed an angle of 30° with the extraction channel. The length of the effective extraction channel, which was defined as the distance from the convergence point of the flows of the sample and buffer solutions to the confluent outlet, was 94 mm. (c) The photograph of the cross-section of the machined plate. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
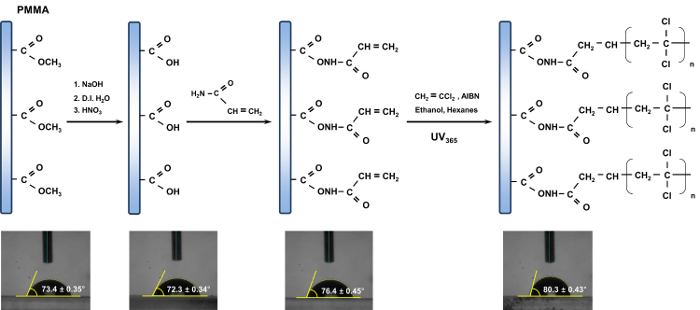 Figure 2. Scheme of the channel modification for the PMMA microchip. The inset photographs show the contact angle corresponding to the resulting product in sequence. The contact angle was determined by using an image of a water drop. The average of three repeated measurements was used for determining the reported contact angles in each case. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
Figure 2. Scheme of the channel modification for the PMMA microchip. The inset photographs show the contact angle corresponding to the resulting product in sequence. The contact angle was determined by using an image of a water drop. The average of three repeated measurements was used for determining the reported contact angles in each case. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
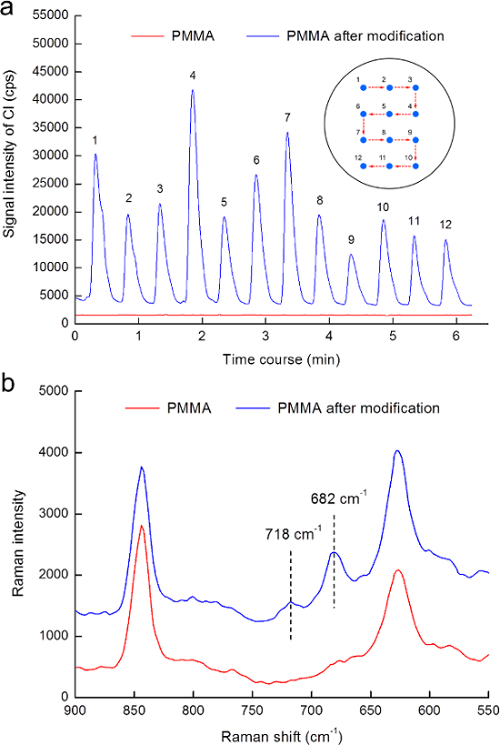 Figure 3. Surface verification of PMMA modification. (a) Signal for Cl obtained by ablating both the PMMA and PMMA modified with the C-Cl moieties. The inset shows the ablation positions corresponding to each obtaining signal. (b) Raman spectra of native and modified PMMA. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
Figure 3. Surface verification of PMMA modification. (a) Signal for Cl obtained by ablating both the PMMA and PMMA modified with the C-Cl moieties. The inset shows the ablation positions corresponding to each obtaining signal. (b) Raman spectra of native and modified PMMA. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
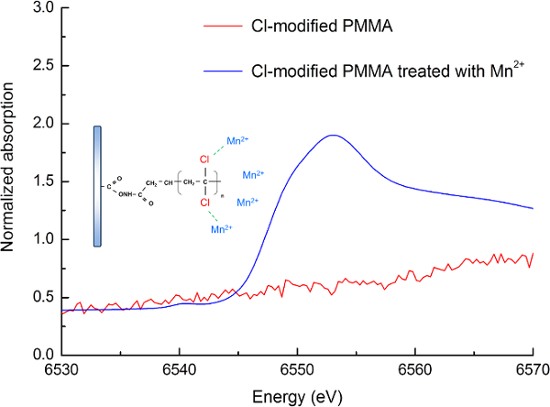 Figure 4. Mn K-edge XANES spectra of modified PMMA and modified PMMA treated with Mn2+ ions. The spectra of modified PMMA was presented as red line. The interactions between the highly electronegative C-Cl moieties of modified PMMA and the Mn2+ ions shown the absorption spectra was presented as blue line. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
Figure 4. Mn K-edge XANES spectra of modified PMMA and modified PMMA treated with Mn2+ ions. The spectra of modified PMMA was presented as red line. The interactions between the highly electronegative C-Cl moieties of modified PMMA and the Mn2+ ions shown the absorption spectra was presented as blue line. Reproduced from Ref. 16 by permission of The Royal Society of Chemistry. Please click here to view a larger version of this figure.
Discussion
The detailed procedures for the preparation of a dipole-assisted SPE microchip were presented above. In this section, the utility of the modification protocol with regard to the implantation of the C-Cl moieties on the PMMA and the feasibility of the Cl-containing PMMA, which was used as an extraction medium for the determination of trace metal ions, are evaluated step-by-step. For surface verification purposes, the sample type was selected on the basis of its compatibility with the analytical instrumentation. In other words, the types of test samples prepared via a similar process were determined in accordance with the requirements of the analytical instruments. For example, a substrate-type sample was used for the measurement of the contact angle, whereas a powder-packing-type sample was used for the LA-ICP-MS, Raman spectroscopic, and XANES analyses.
Initially, to monitor the change undergone by the chemical functionalities attached to the surface of the PMMA during the proposed procedures, a contact angle analysis for the resulting product corresponding to each step was carried out (Figure 2). As displayed in Figure 2, the variations in the contact angle clearly indicated that surface changes occurred during the modification procedures, and the contact angle of 80.3° ± 0.43° that was measured for the final product was in agreement with previously reported results.21
In addition, the existence of the C-Cl moieties on the modified PMMA was also confirmed via LA-ICP-MS analysis. Compared with the results obtained by ablating the native PMMA, distinct signals for Cl were observed expectably by ablating the PMMA modified with the C-Cl moieties (Figure 3(a)).
The Raman spectra were collected for further validating the attachment of the C-Cl moieties to the PMMA. As shown in Figure 3(b), two characteristic peaks associated with the CCl2 asymmetric stretching vibration were observed at 682 cm-1 and 718 cm-1 in the spectrum of the modified PMMA and that in reasonably good agreement with the results reported by Willis et al.22 and Hendra et al.23 In other words, the attachment of the C-Cl moieties to the PMMA could be successfully achieved after modification.
Moreover, to clarify the extraction mechanism proposed in this study, the XANES analysis was employed. As indicated in Figure 4, the interactions between the highly electronegative C-Cl moieties and the positively charged metal ions could be confirmed by the presence of the dominant absorption edge in the XANES spectrum corresponding to the modified PMMA treated with Mn2+ ions. Thus, the dipole-electrostatic interactions would be indeed applied to on-chip extraction for trace metal analyses. The detailed analytical results for water samples collected from two rivers in Taiwan have been described elsewhere.16
To the best of our knowledge, this is the first attempt to utilize an innovative working strategy in on-chip SPE reaction for the determination of trace metal ions, and that the developed device was significantly durable compared with other on-chip SPE techniques (i.e., more than 160 analytical works could be achieved without significant deterioration in terms of the extraction efficiency). Nevertheless, because such extraction mechanism was mainly relied on the interactions between the highly electronegative C-Cl moieties and the positively charged metal ions, the proposed technique was expected to be unsuited for the extraction of the negatively charged species so far.
Disclosures
The authors have nothing to disclose.
Acknowledgments
The authors would like to convey their gratitude for the technical support provided by National Synchrotron Radiation Research Center (NSRRC) (Taiwan). The authors are grateful for the financial support provided by the Ministry of Science and Technology of the Republic of China (Taiwan) and the Industrial Technology Research Institute (Taiwan).
References
- De Mello AJ, Beard N. Dealing with 'real' samples: sample pre-treatment in microfluidic systems. Lab Chip. 2003;3(1):11–19. doi: 10.1039/b301019h. [DOI] [PubMed] [Google Scholar]
- Lichtenberg J, de Rooij NF, Verpoorte E. Sample pretreatment on microfabricated devices. Talanta. 2002;56(2):233–266. doi: 10.1016/s0039-9140(01)00593-8. [DOI] [PubMed] [Google Scholar]
- Song S, Singh AK. On-chip sample preconcentration for integrated microfluidic analysis. Anal. Bioanal. Chem. 2006;384(1):41–43. doi: 10.1007/s00216-005-0206-3. [DOI] [PubMed] [Google Scholar]
- Lafleur JP, Salin ED. Pre-concentration of trace metals on centrifugal microfluidic discs with direct determination by laser ablation inductively coupled plasma mass spectrometry. J. Anal. At. Spectrom. 2009;24(11):1511–1516. [Google Scholar]
- Xue S, Liu Y, Li H-F, Uchiyama K, Lin J-M. A microscale solid-phase extraction poly(dimethylsiloxane) chip for enrichment and fluorescent detection of metal ions. Talanta. 2013;116:1005–1009. doi: 10.1016/j.talanta.2013.08.013. [DOI] [PubMed] [Google Scholar]
- Khoai DV, Kitano A, Yamamoto T, Ukita Y, Takamura Y. Development of high sensitive liquid electrode plasma-Atomic emission spectrometry (LEP-AES) integrated with solid phase pre-concentration. Microelectron. Eng. 2013;111:343–347. [Google Scholar]
- Khoai DV, Yamamoto T, Ukita Y, Takamura Y. On-chip solid phase extraction-liquid electrode plasma atomic emission spectrometry for detection of trace lead. Jpn. J. Appl. Phys. 2014;53(5S1):1–5. [Google Scholar]
- Shih T-T, Chen W-Y, Sun Y-C. Open-channel chip-based solid-phase extraction combined with inductively coupled plasma-mass spectrometry for online determination of trace elements in volume-limited saline samples. J. Chromatogr. A. 2011;1218(16):2342–2348. doi: 10.1016/j.chroma.2011.02.037. [DOI] [PubMed] [Google Scholar]
- Chen B, et al. Magnetic solid phase microextraction on a microchip combined with electrothermal vaporization-inductively coupled plasma mass spectrometry for determination of Cd, Hg and Pb in cells. J. Anal. At. Spectrom. 2010;25(12):1931–1938. [Google Scholar]
- Kim YH, Kim GY, Lim HB. Micro Pre-concentration and Separation of Metal Ions Using Microchip Column Packed with Magnetic Particles Immobilized by Aminobenzyl Ethylenediaminetetraacetic Acid. Bull. Korean Chem. Soc. 2010;31(4):905–909. [Google Scholar]
- Strabburg S, Wollenweber D, Wunsch G. Contamination caused by ion-exchange resin!? Consequences for ultra-trace analysis. Fresenius J. Anal. Chem. 1998;360(7-8):792–794. [Google Scholar]
- Watts JF, Chehimi MM. X-ray Photoelectron Spectroscopy Investigations of Acid-Base Interactions in Adhesion. J. Adhes. 1993;41(1-4):81–91. [Google Scholar]
- Eboatu AN, Diete-Spiff ST, Ezenweke LO, Omalu F. The Use of Polymers as Sequestering Agents for Toxic Metal Ions. J. Appl. Polym. Sci. 2002;85(13):2781–2786. [Google Scholar]
- Fiorini GS, Chiu DT. Disposable microfluidic devices: fabrication, function, and application. Biotechniques. 2005;38(3):429–446. doi: 10.2144/05383RV02. [DOI] [PubMed] [Google Scholar]
- Shadpour H, Musyimi H, Chen J, Soper SA. Physiochemical properties of various polymer substrates and their effects on microchip electrophoresis performance. J. Chromatogr. A. 2006;1111(2):238–251. doi: 10.1016/j.chroma.2005.08.083. [DOI] [PubMed] [Google Scholar]
- Shih T-T, et al. A dipole-assisted solid-phase extraction microchip combined with inductively coupled plasma-mass spectrometry for online determination of trace heavy metals in natural water. Analyst. 2015;140(2):600–608. doi: 10.1039/c4an01421a. [DOI] [PubMed] [Google Scholar]
- Yuan D, Das S. Experimental and theoretical analysis of direct-write laser micromachining of polymethyl methacrylate by CO2 laser ablation. J. Appl. Phys. 2007;101(2):1–6. [Google Scholar]
- Xie S, Svec F, Frechet JMJ. Preparation of Porous Hydrophilic Monoliths: Effect of the Polymerization Conditions on the Porous Properties of Poly(acrylamide-co-N,N'-methylenebisacrylamide) Monolithic Rods. J. Polym. Sci. Pol. Chem. 1997;35(6):1013–1021. [Google Scholar]
- Ericson C, Liao J-L, Nakazato K, Hjerten S. Preparation of continuous beds for electrochromatography and reversed-phase liquid chromatography of low-molecular-mass compounds. J. Chromatogr. A. 1997;767(1-2):33–41. [Google Scholar]
- Shih T-T, Lin C-H, Hsu I-H, Chen J-Y, Sun Y-C. Development of a Titanium Dioxide-Coated Microfluidic-Based Photocatalyst-Assisted Reduction Device to Couple High-Performance Liquid Chromatography with Inductively Coupled Plasma-Mass Spectrometry for Determination of Inorganic Selenium Species. Anal. Chem. 2013;85(21):10091–10098. doi: 10.1021/ac400934e. [DOI] [PubMed] [Google Scholar]
- Carre A. Polar interactions at liquid/polymer interfaces. J. Adhes. Sci. Technol. 2007;21(10):961–981. [Google Scholar]
- Willis HA, Zichy VJI, Hendra PJ. The Laser-Raman and Infra-red Spectra of Poly(Methyl Methacrylate) Polymer. 1969;10:737–746. [Google Scholar]
- Hendra PJ, Mwkenzie JR, Holliday P. The laser-Raman spectrum of polyvinylidene chloride. Spectroc. Acta Pt. A-Molec. Biomolec. Spectr. 1969;25(8):1349–1354. [Google Scholar]


