Abstract
A novel animal model involving chimeric mice with humanized livers established via human hepatocyte transplantation has been developed. These mice, in which the liver has been repopulated with functional human hepatocytes, could serve as a useful tool for investigating human hepatic cell biology, drug metabolism, and other preclinical applications. One of the key factors required for successful transplantation of human hepatocytes into mice is the elimination of the endogenous hepatocytes to prevent competition with the human cells and provide a suitable space and microenvironment for promoting human donor cell expansion and differentiation. To date, two major liver injury mouse models utilizing fumarylacetoacetate hydrolase (Fah) and uroplasminogen activator (uPA) mice have been established. However, Fah mice are used mainly with mature hepatocytes and the application of the uPA model is limited by decreased breeding. To overcome these limitations, Alb-toxin receptor mediated cell knockout (TRECK)/SCID mice were used for in vivo differentiation of immature human hepatocytes and humanized liver generation. Human hepatic stem cells (HpSCs) successfully repopulated the livers of Alb-TRECK/SCID mice that had developed lethal fulminant hepatic failure following diphtheria toxin (DT) treatment. This model of a humanized liver in Alb-TRECK/SCID mice will have functional applications in studies involving drug metabolism and drug-drug interactions and will promote other in vivo and in vitro studies.
Keywords: Medicine, Issue 114, Fulminant liver failure, transplant, diphtheria toxin, humanized liver, TRECK/SCID mice, intrasplenic transplantation, human hepatocytes, immature, fetal liver cells, hepatic stem cells, stem cell biology, albumin
Introduction
Mice are commonly used for pharmaceutical testing since biomedical research in humans is restricted1; however, these models are not always useful since they may inaccurately simulate the effects observed in humans. Most drugs in current medical use are metabolized primarily in the liver. However, the same drug can be metabolized into different metabolites in mouse and human livers because of inter-species differences. Thus, it is often difficult to determine during development whether a potential drug poses any risks for clinical applications2,3.
To address this problem, "humanized" mouse livers have been developed by growing human liver cells inside mice4-6; these models exhibit drug responses similar to those observed in the human liver. The primary mouse models currently used for humanized liver generation include uroplasminogen activator (uPA+/+) mice4,7, fumarylacetoacetate hydrolase (Fah−/−) mice6, and the recently reported thymidine kinase (TK-NOG) mice.
However, previous reports have shown that transplanted human immature cells or stem cells are less competitive than adult human hepatocytes in Alb-uPA tg(+/−)Rag2(−/−) mouse livers8-10. Moreover, Fah−/− mice provide a growth advantage only for differentiated hepatocytes and not for immature liver progenitor cells11. The transplantation of human hepatic stem cells (HpSCs) into TK-NOG mice in the lab has been unsuccessful. Hence, no useful mouse model for the efficient engraftment of human immature liver cells currently exists.
Thus, we developed a novel Alb-TRECK/SCID mouse model that could be efficiently repopulated with human immature hepatocytes. This transgenic mouse model expresses human heparin-binding EGF-like growth factor (HB-EGF) receptors under the control of a liver cell-specific albumin promoter. Following the administration of diphtheria toxin (DT), these mice develop fulminant hepatitis due to conditional ablation of hepatocytes, enabling donor cell residency and proliferation12. Although mouse hepatocytes have been successfully transplanted into Alb-TRECK/SCID mice in previous studies13,14, the generation of a humanized liver using Alb-TRECK/SCID mice has yet to be reported.
In this study, humanized livers were generated in Alb-TRECK/SCID mice via transplantation of HpSCs. This humanized liver provides an in vivo environment for universal stem cell differentiation and the ability to predict human drug metabolism patterns and drug-drug interactions.
Protocol
All animal experimental procedures were performed according to the Animal Protection Guidelines of Yokohama City University.
1. Generation of the Acute Liver Injury Mouse Model
Add 1 ml of phenolized 0.85% NaCl solution (0.6 g phenol in 100 ml of 0.85% NaCl solution) to 1 mg diphtheria toxin (DT) to make a 1 mg/ml DT stock solution. Note: DT at a concentration of 1 mg/ml can be stored for approximately 2 years at 3 °C to 8 °C.
Serially dilute the 1 mg/ml DT stock solution to 0.3 μg/ml DT in phenolized 0.85% NaCl solution and prepare an aliquot of the 0.3 μg/ml DT solution as a working solution. Note: This working solution should be freshly prepared.
- To conduct experiments using a sublethal dose (~ 50% lethality), administer 8-week-old mice a 1.5 μg/kg dose of DT.
- Perform an intraperitoneal injection of DT by holding the mouse in dorsal recumbency and insert the needle below the bend of the knees, left or right of the midline. Avoid the midline to prevent penetration of the bladder. Angle the needle at approximately 45° to the body.
- Using an insulin syringe, inject the mice with 100 µl of freshly prepared 0.3 μg/ml DT per 20 g of mouse weight. For example, an 18-g mouse should receive 90 µl of the 0.3 μg/ml DT solution.
- At 48 hr post DT injection, collect blood from the tail vein by placing the mouse in a restrainer and warming the mouse tail in a 37 °C water bath for approximately 10 min to dilate the blood vessels. Then, make a 1-mm nick in the tail 2 cm from the tip with a sharp scalpel blade and collect the blood with a microcapillary tube.
- Centrifuge the microcapillary tube containing the blood at 1,500 x g for 5 min and collect the serum by separating the supernatant and cell pellet.
Pipette 50 µl of 1:20 diluted mouse serum onto GOT/AST-PIII slides. Measure the absorbance of the reaction product (blue dye) at 650 nm using a multi-purpose automatic dry-chemistry analyzer according to manufacturer's protocol. NOTE: The read-out of aspartate aminotransferase (AST)/glutamic oxaloacetic transaminase (GOT) activity is displayed automatically. 2 days post DT injection, approximately 60% of the mice exhibited AST values between 12,000 - 16,000 IU/L and were considered to be suffering from acute liver damage. These mice were transferred to a new cage and used as cell transplant recipients.
2. Preparation of Human Hepatic Stem Cells
Isolate human hepatic stem cells from human primary fetal liver cells with a cell sorter using the CDCP1, CD90, and CD66 cell antigens to obtain a CDCP1+CD90+CD66- subpopulation, then seed the isolated cell population on collagen IV-coated culture dishes, as previously reported15. Use human primary fetal liver cells of an embryonic age between weeks 14 and 18.
Collect human hepatic stem cells cultured to 80% - 90% confluence on 100-mm culture dishes, aspirate the culture medium, wash the cells with 10 ml PBS, and then remove the PBS. Trypsinize the cells with 2 ml 0.05% trypsin/EDTA solution for 5 min at 37 °C. Note: Cells at greater than 90% confluence are not suitable for cell transplantation since cell differentiation may influence their proliferative capability in mice.
Monitor the status of cell dispersion under a microscope. When the cells appear to be floating or flowing freely, stop the enzymatic digestion by adding 8 ml of DMEM/F12 containing 10% FBS.
Suspend the cells slowly using a 10-ml serological pipette and transfer the cells into a 15-mL conical tube. Centrifuge the cells for 5 min at 100 x g and 4 °C.
Carefully resuspend the cell pellet in 10 ml DMEM/F12 containing 10% FBS and determine cell numbers using a microscope counting chamber.
Divide the cells into 1.0 x 106 cells per 50 µl of PBS aliquots for each individual mouse and store on ice until transplantation.
3. Intrasplenic Transplantation of Human Hepatic Stem Cells
Place a clean cage on a 37 °C electric heating pad.
- Anesthetize the mouse using isoflurane inhalation (1 - 1.5% (vol/vol) in 2 L of oxygen per min) by placing it underneath the nozzle. Alternatively, use tubes containing gauze soaked with isoflurane.
- Ensure that the mouse is anesthetized by lightly pinching the rear footpad. If a knee jerk is elicited, place the mouse back in the chamber. Use vet ointment on the eyes to prevent dryness while under anesthesia.
Shave the surgical site using electric clippers and apply 70% (vol/vol) ethanol and povidone-iodine to sterilize. Then make a 1-cm skin incision in the left flank just below the costal border of the ribs, followed by an incision on the abdominal wall, as well as the peritoneum.
- Carefully expose the spleen. Use a 100-μl microinjection syringe with a 32-G 1/2-inch needle to directly inject 1.0 x 106 human hepatic stem cells in 50 µl PBS into the spleen of each mouse. Tilt the needle at a 5° angle for injection.
- Ensure that the depth of injection is less than half the thickness of the spleen. The needle should enter the spleen at one end (the head) and deposit the cells at the other end (the tail).
To prevent leakage of the cells following injection, gently apply pressure using a finger for 2 min and then remove the needle.
Place the spleen back into the mouse body and close the cavity with a normal running suture for the musculature and skin.
Transfer the mice into a 37 °C pre-warmed cage immediately following transplantation. To ensure the mouse is able to breathe comfortably, place the mouse on its side in the cage, avoiding contact between the cage bedding and the site of surgery.
Monitor the mice until the anesthesia wears off to ensure the sutures remain closed and the mice return to pre-surgery conditions.
Ensure the mice are provided with normal drinking water and food. After 1 hr, return the cage to the mouse room of the animal center and monitor the mice daily.
4. Detection of Transplanted Human Hepatic Stem Cell-Derived Hepatocytes in the Mouse Liver
Note: For the following procedures, euthanize all animals using an overdose of ketamine and xylazine followed by cervical dislocation.
At 4 - 6 weeks post transplantation, euthanize the mice and open the abdominal cavity by simultaneously cutting the cutis and fascia using surgical scissors.
Dissect the connective tissue above the peritoneum, using the scissors as a spreader, and cut the peritoneum along the linea alba to open the peritoneal cavity.
Lift the sternum with forceps, puncture the diaphragm, and cut through each side of the sternum up through the cervical girdle.
Sever the vena cava on the thoracic side of the liver and pull the esophagus through the liver in the anterior direction. Remove the diaphragm in order to remove the liver. Note: Be careful to keep the tips of the scissors pointed upward in order to prevent any damage to the thoracic organs underneath. The thymus has a tendency to occasionally cling to the dorsal side of the sternum and this must be carefully avoided.
Using the diaphragm as a handle, start pulling the liver out of the abdominal cavity. The interior vena cava will be holding the liver in place. Cut the interior vena cava; be careful not to prematurely liberate the right adrenal. Note: Mice have a four-lobed liver consisting of a median lobe, left lobe, right lobe, and the caudate lobe. The gall bladder is suspended in the small bifurcation of the median lobe.
Separate the lobes from each other at the junctions and embed them in optimal cutting temperature (OCT) compound according to the manufacturer's protocol. Freeze the OCT containing the liver tissue in the metal grids on the cryostat.
Cut 5-µm-thick sample sections using a cryostat at -18 °C and mount them on RT microscope slides. Store the stock at -80 °C. Prepare the slides for hematoxylin and eosin (H&E) and immunofluorescence staining according to previously reported procedures16.
5. Detection of Human Albumin Secretion and Calculation of the Chimeric Rate
To measure human liver reconstitution, perform human albumin ELISA using serum and a human albumin ELISA kit according to the manufacturer's instructions.
Calculate the humanized liver chimeric rate by real-time PCR analysis of expression levels in the whole-humanized liver. Determine the relative expression level ratio of human-specific actin to human-mouse cross actin (ratio 1) and the ratio of mouse-specific actin to human-mouse cross actin (ratio 2). The chimeric rate is calculated as ratio 1/(ratio 1 + ratio 2).
Representative Results
Alb-TRECK/SCID mice hepatocytes express the human DT receptor HB-EGF gene under the control of an albumin promoter and exhibit cytotoxic effects following DT administration12. To evaluate the effects of DT treatment on liver injury, DT doses of 1.5 μg/kg were injected into 8-week-old Alb-TRECK/SCID mice and the pathological changes in the liver 48 hr post DT administration were histologically assessed. Compared with control mice (not treated with DT), the DT-treated mice exhibited a disorganized hepatic architecture showing a correction for congestion with increased serum AST activity. In addition, the hepatocytes of DT-treated mice had multiple, deeply stained acidophilic cytoplasmic inclusions together with dark nuclei (most probably apoptotic hepatocytes); some other hepatocytes appeared preserved or exhibited a vacuolated cytoplasm with dark nuclei with little or no portal vein inflammation (Figure 1). These findings are concordant with those of a previous study that showed that Alb-TRECK/SCID mice represent an ideal lethal fulminant hepatic failure model and can be generated using only a single DT injection12.
Human HpSCs can be used for long-term in vitro culturing and exhibit uniform cell morphology with some ALB and fewer CK19-positive cells. At 6 weeks post transplantation into the mouse livers, the human HpSCs were subjected to basic examination under a microscope and evaluated histologically; human HpSCs were observed to have reconstituted the liver structure by replacing the original mouse hepatocytes (Figure 2). Macroscopically, at 4 days post human HpSC transplantation, small human hepatic clusters that were uniformly distributed around the liver were detected; at 45 days post transplantation, these had proliferated into large clusters (Figure 3). Whole mouse livers reconstituted with human hepatocytes were designated "humanized livers." These results indicate that mice with lethal fulminant hepatic failure that undergo human HpSC transplantation may exhibit liver structures similar to those observed in normal mouse livers.
Prior to testing whether the immature human hepatocytes transplanted into mice had the potential for differentiation and could function in vivo, the presence of human hepatocytes in mouse livers was first confirmed by immunohistochemical analysis at approximately 6 weeks post human HpSC transplantation. Liver sections from mice with 60% human cell repopulation that specifically and positively co-stained with human nuclei and human cytokeratin 8/18 (CK8/18) antibodies were found to contain donor cell-derived human hepatocytes, whereas the original control mice livers were negative for these markers (Figure 4).
To assess the degree of cell differentiation in vivo, the expression of human albumin (ALB) and human cytokeratin 19 (CK19) was assessed immunohistochemically. Human HpSC-derived human hepatocytes were well differentiated in the mouse livers and exhibited upregulated human ALB expression. Human ALB-positive and CK19-negative hepatocytes resembled functional hepatocytes, whereas cells that positively co-stained with human ALB and CK19 exhibited bipotential capability for differentiating into hepatocytes and cholangiocytes (Figure 5). A large-scale scanning method used to analyze all human HpSC-derived humanized liver lobes revealed the presence of multiple round and colony-like clusters surrounding the liver lobes with clear human nuclei and CK8/18 expression (Figure 6), demonstrating the colony-forming capability of human HpSCs in the humanized livers. The repopulation level reached up to 90% one month following transplantation (Figure 7A) and human albumin secretion was detected in the repopulated mice livers (Figure 7B), indicating that human HpSCs could successfully reconstitute the Alb-TRECK/SCID mouse liver. These results demonstrate that human HpSCs have high potential for differentiation into functional hepatocytes in vivo in response to the rescue of defective mouse liver functions.
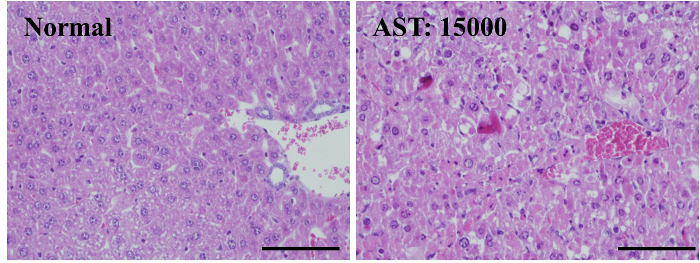 Figure 1.Liver Damage Occurs in Alb-TRECK/SCID (Toxin Receptor Mediated Cell Knockout) Mice Following Diphtheria Toxin (DT) Injection. Histology (hematoxylin-eosin staining) of mouse livers without and with DT administration after 48 hr. DT dose: 1.5 μg/kg, serum AST activity of normal liver (left panel): 30 IU/L; serum AST activity of DT-treated liver (right panel): 15000 IU/L. Scale bar = 100 µm. Please click here to view a larger version of this figure.
Figure 1.Liver Damage Occurs in Alb-TRECK/SCID (Toxin Receptor Mediated Cell Knockout) Mice Following Diphtheria Toxin (DT) Injection. Histology (hematoxylin-eosin staining) of mouse livers without and with DT administration after 48 hr. DT dose: 1.5 μg/kg, serum AST activity of normal liver (left panel): 30 IU/L; serum AST activity of DT-treated liver (right panel): 15000 IU/L. Scale bar = 100 µm. Please click here to view a larger version of this figure.
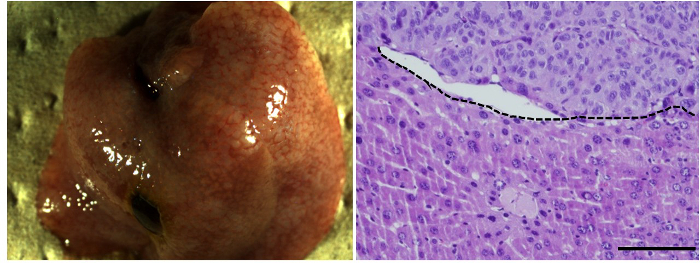 Figure 2.Transplantation of Human Hepatic Stem Cells into Alb-TRECK/SCID Mice with Fulminant Hepatic Failure. Macroscopic view (left panel) and histological analysis (hematoxylin-eosin staining, right panel) of humanized livers with human HpSCs at 6 weeks post transplantation. m, mouse liver region; h, human donor cell-derived human region. Scale bar = 100 µm. Please click here to view a larger version of this figure.
Figure 2.Transplantation of Human Hepatic Stem Cells into Alb-TRECK/SCID Mice with Fulminant Hepatic Failure. Macroscopic view (left panel) and histological analysis (hematoxylin-eosin staining, right panel) of humanized livers with human HpSCs at 6 weeks post transplantation. m, mouse liver region; h, human donor cell-derived human region. Scale bar = 100 µm. Please click here to view a larger version of this figure.
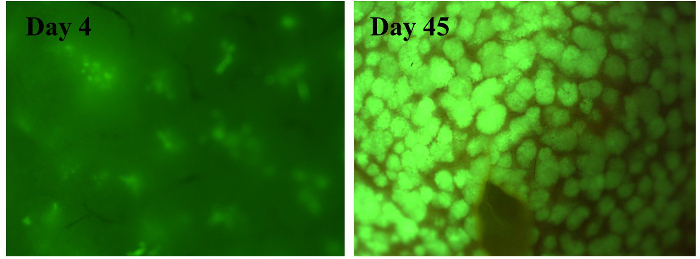 Figure 3.Macroscopic Analysis of Humanized Livers Derived from Human Hepatic Stem Cells Labeled with Green Fluorescent Protein. High-magnification images of the liver structure 4 days (left panel) and 45 days (right panel) following transplantation with human hepatic stem cells (HpSCs). Human hepatic stem cells (HpSCs) were labeled with enhanced green fluorescent protein (EGFP) using a lentivirus vector. Please click here to view a larger version of this figure.
Figure 3.Macroscopic Analysis of Humanized Livers Derived from Human Hepatic Stem Cells Labeled with Green Fluorescent Protein. High-magnification images of the liver structure 4 days (left panel) and 45 days (right panel) following transplantation with human hepatic stem cells (HpSCs). Human hepatic stem cells (HpSCs) were labeled with enhanced green fluorescent protein (EGFP) using a lentivirus vector. Please click here to view a larger version of this figure.
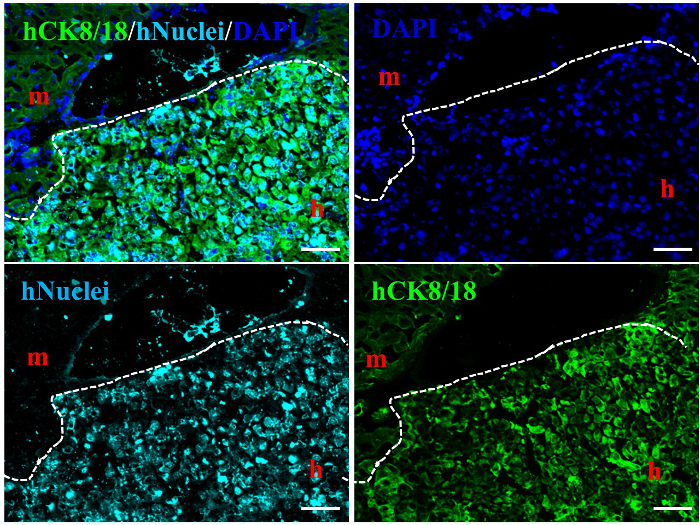 Figure 4.Characterization of Human Hepatic Stem Cell-Derived Human Hepatocytes in Alb-TRECK/SCID Mice. Immunohistochemical analysis distinguishing between human hepatocytes stained with anti-human CK8/18 (green) antigen and anti-human nuclear antigen (aqua blue) in human hepatic stem cell-derived humanized livers at 6 weeks post transplantation. m, mouse liver region; h, human donor cell-derived human liver region. White dashed line: mouse liver region vs. human liver region. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (blue). Scale bars = 100 µm. Please click here to view a larger version of this figure.
Figure 4.Characterization of Human Hepatic Stem Cell-Derived Human Hepatocytes in Alb-TRECK/SCID Mice. Immunohistochemical analysis distinguishing between human hepatocytes stained with anti-human CK8/18 (green) antigen and anti-human nuclear antigen (aqua blue) in human hepatic stem cell-derived humanized livers at 6 weeks post transplantation. m, mouse liver region; h, human donor cell-derived human liver region. White dashed line: mouse liver region vs. human liver region. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (blue). Scale bars = 100 µm. Please click here to view a larger version of this figure.
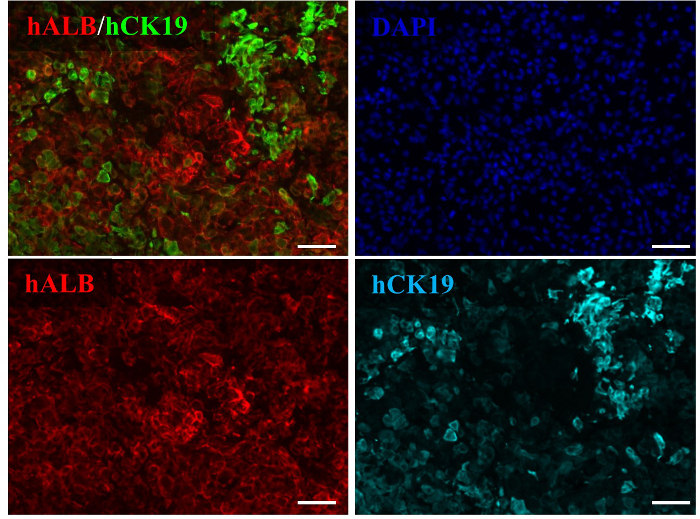 Figure 5. Human Hepatic Stem Cells Differentiation in Humanized Livers of Alb-TRECK/SCID Mice. Immunohistochemical analyses of human albumin and human CK19 in HpSC-derived livers at 6 weeks post transplantation. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (blue). Scale bars = 100 µm. Please click here to view a larger version of this figure.
Figure 5. Human Hepatic Stem Cells Differentiation in Humanized Livers of Alb-TRECK/SCID Mice. Immunohistochemical analyses of human albumin and human CK19 in HpSC-derived livers at 6 weeks post transplantation. Nuclei were counterstained with 4′,6-diamidino-2-phenylindole (blue). Scale bars = 100 µm. Please click here to view a larger version of this figure.
 Figure 6.Multiple Clusters of Human Hepatic Stem Cell-Derived Cells are Present in Alb-TRECK/SCID Mice with Humanized Livers. Multiple large clusters derived from human hepatic stem cells were identified in the liver lobes at 6 weeks post transplantation by immunohistochemical analysis for human CK8/18 (green) and human nuclei (aqua blue). Scale bars = 1,000 µm. Please click here to view a larger version of this figure.
Figure 6.Multiple Clusters of Human Hepatic Stem Cell-Derived Cells are Present in Alb-TRECK/SCID Mice with Humanized Livers. Multiple large clusters derived from human hepatic stem cells were identified in the liver lobes at 6 weeks post transplantation by immunohistochemical analysis for human CK8/18 (green) and human nuclei (aqua blue). Scale bars = 1,000 µm. Please click here to view a larger version of this figure.
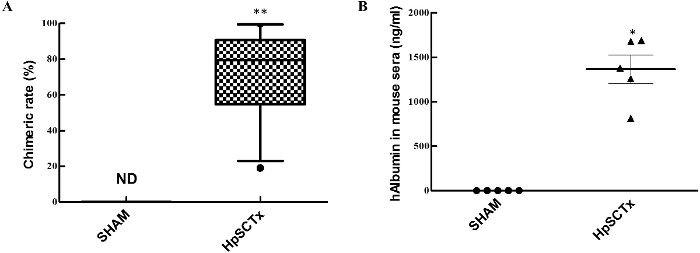 Figure 7.Humanized Liver Repopulation Rate and Human Albumin Secretion in Alb-TRECK/SCID Mice. (A) Chimeric rate in Alb-TRECK/SCID mice 1 month following human hepatic stem cell transplantation; data is presented as mean ± SEM (n = 12). Mann-Whitney test: P < 0.0005 for Sham and human HpSCs. (B) Detection of human albumin in mouse sera one month following donor cell transplantation. Data = mean ± SEM (n = 5). Mann-Whitney test: P = 0.0313 for Sham and human HpSCs. ND: undetectable; Sham: mice transplanted with PBS; HpSCTx: mice transplanted with human HpSCs. Please click here to view a larger version of this figure.
Figure 7.Humanized Liver Repopulation Rate and Human Albumin Secretion in Alb-TRECK/SCID Mice. (A) Chimeric rate in Alb-TRECK/SCID mice 1 month following human hepatic stem cell transplantation; data is presented as mean ± SEM (n = 12). Mann-Whitney test: P < 0.0005 for Sham and human HpSCs. (B) Detection of human albumin in mouse sera one month following donor cell transplantation. Data = mean ± SEM (n = 5). Mann-Whitney test: P = 0.0313 for Sham and human HpSCs. ND: undetectable; Sham: mice transplanted with PBS; HpSCTx: mice transplanted with human HpSCs. Please click here to view a larger version of this figure.
Discussion
Recent studies have shown that the mouse liver can be repopulated with human hepatocytes, including adult hepatocytes and proliferative hepatic stem cells17. These repopulated livers have been used as preclinical experimental models for drug metabolism testing and drug discovery and development18; in addition, they have provided an in vivo environment for cell maturation and differentiation19. The major aim of the present study was to generate a novel liver disease mouse model that allowed human immature hepatocyte proliferation, maturation, and differentiation, for the acquisition of drug metabolism activities.
The course of intrasplenic transplantation has been studied extensively. Cells transplanted into the spleen of rats and mice survive throughout their average life span. The intrasplenic transplantation protocol has proven to be very useful in investigating specific research questions; many laboratories worldwide have attempted to utilize this protocol as a highly reproducible and reliable model for hepatic cell transplantation because of the improved mice survival and liver regeneration. However, implanting cells into the splenic pulp poses a risk, since the majority of implanted cells will translocate to the liver where they can occlude portal branches. Even though this occlusion can be temporary and might not affect a healthy liver environment, in cases of preexisting liver damage, this procedure could further reduce liver function and increase portal hypertension and might also negatively influence cell engraftment. Although we have successfully applied hepatocyte transplantation using intrasplenic transplantation, several issues warrant further study, especially the subject of potential new cell sources. Moreover, when performing intrasplenic hepatocyte transplantation, the cell loading limit must be strictly followed in order to prevent potential disturbance by remnant hepatic microcirculation. The DT receptor has been identified as a membrane-anchored form of the heparin-binding EGF-like growth factor (HB-EGF precursor)20. Whereas HB-EGF precursors derived from toxin-sensitive animals, such as humans and monkeys, bind DT and function as toxin receptors, HB-EGF precursors from mice and rats do not bind DT21. The transgenic Alb-TRECK/SCID mouse model expresses human HB-EGF-like receptors under the control of a liver cell-specific albumin promoter; following the administration of DT, these mice develop fulminant hepatic failure due to conditional ablation of hepatocytes, providing space for donor cell residency and proliferation. Human hepatocytes, even at an immature phase, are known to possess extensive proliferative capacity in vitro, although they lose their drug metabolism functions, limiting their preclinical applications22. Histological and immunohistochemical findings obtained following the transplantation of human HpSCs into Alb-TRECK/SCID mice with lethal fulminant hepatic failure indicated that the human HpSCs could expand and reconstitute the damaged mouse liver structures; the repopulation rate in some mice was nearly 100%.
In our experiments, human HpSC transplantation promoted mouse survival and resulted in increased secretion of human ALB into mouse sera16. These cells also matured and differentiated in vivo and exhibited human drug metabolism activities, indicating that human HpSC-derived humanized mice livers are similar to mature and functional "human organs" and have potential applications in drug development. These results further confirm that the Alb-TRECK/SCID mouse is an ideal model for humanized liver generation. Despite the prospects and advantages of Alb-TRECK/SCID mice as an ideal model for human hepatic stem cell-derived humanized liver generation, they possess one unique disadvantage; when human adult hepatocytes were transplanted into three Alb-TRECK/SCID mice treated with a single dose of DT, no humanized livers were generated, possibly because human adult hepatocytes lack proliferative capacity. Additional doses of DT cannot be administered post transplantation of human adult hepatocyte; since the DT receptor (HB-EGF) in Alb-TRECK/SCID mice hepatocytes is under the control of the albumin promoter, additional DT treatment will destroy the transplanted human adult hepatocytes in the mouse liver.
To the best of our knowledge, this is the first model to generate a humanized liver from immature human cells. Cell replacement rate post-transplant is influenced by the recipient micro-environment (e.g., hepatic niche) and donor cell properties, such as mature vs. immature and proliferative and differential capability. Combining Alb-TRECK/SCID with stem cell transplantation will provide optimized conditions and advantages for the generation of humanized livers. This also constitutes a powerful tool for transplantation of fetal liver cells and potentially as well as iPS or ES-derived hepatic cells since prior to DT treatment, the Alb-TRECK/SCID animals are maintained in a healthy state, liver damage is inducible at any point, and the mice breed normally without renal damage. In contrast, uPA/SCID mice exhibit high neonatal mortality and are difficult to breed. Furthermore, overexpression of urokinase in the animals can lead to severe bleeding and renal damage and thus there is a limited window of opportunity for cell transplantation prior to weaning; Alb-TRECK/SCID can be used for cell transplantation at any age.
The successful generation of a humanized liver mouse requires the following critical steps: 1) Liver injury should be limited to sub-lethal levels (usually by using the lethal dose, 50%); 2) The human primary fetal liver cells should be maintained in a sub-confluent status no greater than 90%; 3) Cell leakage post intrasplenic-transplantation should be prevented; and 4) The levels of human albumin in mouse sera should be monitored at regular intervals. A comprehensive understanding of the critical factors involved in the process of humanized mouse liver generation might contribute to further modifications or new developments utilizing rats or other animals. Possible adaptations include: the induction of kidney injury for examining development and repair with human nephrons, their progenitors, and collecting duct cells and the self-assembly of human nephrons into three dimensional structures in the mouse body; studying the repair of islet damage in the pancreas using pancreatic stem/progenitor cells derived from directly reprogrammed, iPS, or ES cells. Examples of process modifications include: controlling tissue or organ injury with toxins or chemicals such as retrorsine, streptozotocin, or cisplatin to facilitate the competition of donor cells with resident cells; avoiding differential or decreasing proliferation in order to enhance the proliferative capability of the donor cells, thus promoting the replacement of recipient cells, such as ES or iPS- derived cells; selecting a suitable transplant sites and controlling cell infusion to ensure efficient transplantation, for example, using the portal or tail vein for entopic transplantation and the mesentery and renal capsule for ectopic transplantation; monitoring replacement status via the expression of tissue or organ secreted proteins representative of stem/progenitor or immature cells transplant and cancer cell tracing, such as alpha fetoprotein or transferrin.
In summary, Alb-TRECK/SCID mice constitute a suitable model for human hepatic stem cell transplantation, as they provide a beneficial environment that allows for differentiation into mature hepatocytes. This novel model has tremendous applicative potential, not only for ex vivo expansion of human hepatocytes but also for drug screens and testing candidate therapeutic drugs for liver toxicity and metabolism. In addition, these results will aid in the advancement of future studies examining the clinical application of this technique to human hepatic cell transplantation.
Disclosures
The authors have no competing financial interests to disclose.
Acknowledgments
We wish to thank the Mammalian Genetics Project, Tokyo Metropolitan Institute of Medical Science, for providing the mice. We also thank S. Aoyama and Y. Adachi of the ADME (Absorption, Distribution, Metabolism, Excretion) & Toxicology Research Institute, Sekisui Medical Company Ltd., Japan, and K. Kozakai and Y. Yamada for assistance with LC-MS/MS analysis. This work was supported in part by Grants-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT), Japan to Y-W.Z. (18591421, 20591531, and 23591872); by the Jiangsu innovative and entrepreneurial project for the introduction of high-level talent and the Jiangsu science and technology planning project (BE2015669); and by grants to H.T. for Strategic Promotion of Innovative Research and Development (S-innovation, 62890004) from the Japan Science and Technology Agency.
References
- Muruganandan S, Sinal C. Mice as clinically relevant models for the study of cytochrome P450-dependent metabolism. Clin. Pharmacol. Ther. 2008;83:818–828. doi: 10.1038/clpt.2008.50. [DOI] [PubMed] [Google Scholar]
- Nishimura T, et al. Using chimeric mice with humanized livers to predict human drug metabolism and a drug-drug interaction. J. Pharmacol. Exp. Ther. 2013;344:388–396. doi: 10.1124/jpet.112.198697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suemizu H, et al. Establishment of a humanized model of liver using NOD/Shi-scid IL2Rgnull mice. Biochem. Biophys. Res. Commun. 2008;377:248–252. doi: 10.1016/j.bbrc.2008.09.124. [DOI] [PubMed] [Google Scholar]
- Tateno C, et al. Near completely humanized liver in mice shows human-type metabolic responses to drugs. Am. J. Pathol. 2004;165:901–912. doi: 10.1016/S0002-9440(10)63352-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasegawa M, et al. The reconstituted 'humanized liver'in TK-NOG mice is mature and functional. Biochem. Biophys. Res. Commun. 2011;405:405–410. doi: 10.1016/j.bbrc.2011.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azuma H, et al. Robust expansion of human hepatocytes in Fah−/−/Rag2−/−/Il2rg−/− mice. Nat. Biotechnol. 2007;25:903–910. doi: 10.1038/nbt1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhim JA, Sandgren EP, Degen JL, Palmiter RD, Brinster RL. Replacement of diseased mouse liver by hepatic cell transplantation. Science. 1994;263:1149–1152. doi: 10.1126/science.8108734. [DOI] [PubMed] [Google Scholar]
- Haridass D, et al. Repopulation efficiencies of adult hepatocytes, fetal liver progenitor cells, and embryonic stem cell-derived hepatic cells in albumin-promoter-enhancer urokinase-type plasminogen activator mice. Am. J. Pathol. 2009;175:1483–1492. doi: 10.2353/ajpath.2009.090117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma AD, et al. Murine embryonic stem cell-derived hepatic progenitor cells engraft in recipient livers with limited capacity of liver tissue formation. Cell. Transplant. 2008;17:313–323. doi: 10.3727/096368908784153896. [DOI] [PubMed] [Google Scholar]
- Peltz G. Can 'humanized' mice improve drug development in the 21st century. Trends Pharmacol. Sci. 2013;34:255–260. doi: 10.1016/j.tips.2013.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu S, et al. Mouse liver repopulation with hepatocytes generated from human fibroblasts. Nature. 2014;508:93–97. doi: 10.1038/nature13020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito M, et al. Diphtheria toxin receptor-mediated conditional and targeted cell ablation in transgenic mice. Nat. Biotechnol. 2001;19:746–750. doi: 10.1038/90795. [DOI] [PubMed] [Google Scholar]
- Ishii T, et al. Transplantation of embryonic stem cell-derived endodermal cells into mice with induced lethal liver damage. Stem Cells. 2007;25:3252–3260. doi: 10.1634/stemcells.2007-0199. [DOI] [PubMed] [Google Scholar]
- Machimoto T, et al. Improvement of the survival rate by fetal liver cell transplantation in a mice lethal liver failure model. Transplantation. 2007;84:1233–1239. doi: 10.1097/01.tp.0000287967.54222.4d. [DOI] [PubMed] [Google Scholar]
- Taniguchi H, Zheng Y-W. Human hepatic stem cell, method for preparation of the same, method for induction of differentiation of the same, and method for utilization of the same. 5822287. Japan Patent. 2015
- Zhang R-R, et al. Human hepatic stem cells transplanted into a fulminant hepatic failure Alb-TRECK/SCID mouse model exhibit liver reconstitution and drug metabolism capabilities. Stem Cell Res. Ther. 2015;6:1–12. doi: 10.1186/s13287-015-0038-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strom SC, Davila J, Grompe M. Hepatocytes. Springer; 2010. Chimeric Mice with Humanized Liver: Tools for the Study of Drug Metabolism, Excretion, and Toxicity; pp. 491–509. (Methods in Molecular Biology; 640). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D, Luo G, Ding X, Lu C. Preclinical experimental models of drug metabolism and disposition in drug discovery and development. Acta Pharm. Sin. B. 2012;2:549–561. [Google Scholar]
- Nowak G, et al. Identification of expandable human hepatic progenitors which differentiate into mature hepatic cells in vivo. Gut. 2005;54:972–979. doi: 10.1136/gut.2005.064477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naglich JG, Metherall JE, Russell DW, Eidels L. Expression cloning of a diphtheria toxin receptor: identity with a heparin-binding EGF-like growth factor precursor. Cell. 1992;69:1051–1061. doi: 10.1016/0092-8674(92)90623-k. [DOI] [PubMed] [Google Scholar]
- Mitamura T, Higashiyama S, Taniguchi N, Klagsbrun M, Mekada E. Diphtheria toxin binds to the epidermal growth factor (EGF)-like domain of human heparin-binding EGF-like growth factor/diphtheria toxin receptor and inhibits specifically its mitogenic activity. J. Biol. Chem. 1995;270:1015–1019. doi: 10.1074/jbc.270.3.1015. [DOI] [PubMed] [Google Scholar]
- Liu Y, Yang R, He Z, Gao W-Q. Generation of functional organs from stem cells. Cell Regener. 2013;2:1. doi: 10.1186/2045-9769-2-1. [DOI] [PMC free article] [PubMed] [Google Scholar]


