1. Introduction
Recent studies have indicated that cholesterol metabolism is important for the immune response, with dyslipidaemia resulting in worsening inflammatory activity and disease progression in multiple sclerosis (MS) patients (Chalmin et al., 2015, Tall and Yvan-Charvet, 2015). In addition, cholesterol synthesis and transport in oligodendrocytes has been shown to be essential for optimal myelination and remyelination (Chalmin, Rochemont, 2015). Cholesterol 25-hydroxylase (CH25H) converts cholesterol into the oxysterol 25-hydroxycholesterol (25-HC), and is the rate-limiting enzyme in the conversion of cholesterol to serum oxysterols, which regulate cellular proliferation and differentiation of cells of the innate and adaptive immune system (Spann and Glass, 2013). The expression of CH25H is induced by type I interferons (IFN), potent cytokines used for the treatment of inflammatory diseases, including MS. Activation of CH25H has been shown to increase the concentration of 25-HC, reducing interleukin-1B transcription and repressing its inflammasome-mediated activation (Reboldi et al., 2014, Tall and Yvan-Charvet, 2015).
CH25H knockout mice present marked changes in their inflammatory response, with increased frequencies of IL17A+ T cells and neutrophil counts (Reboldi, Dang, 2014). Interestingly, the induction of experimental autoimmune encephalomyelitis (EAE) in these animals leads to a less severe disease course and delayed onset of symptoms, due to limited trafficking of activated lymphocytes to the central nervous system (Chalmin, Rochemont, 2015). Taken together this data suggest that the synthesis of oxysterol from cholesterol metabolism may play a role in the onset and progression of autoimmune inflammatory diseases. Since CH25H is the rate-limiting enzyme in this process, we set to assess the presence of rare coding variants in CH25H which may affect protein function in patients diagnosed with MS or neuromyelitis optica (NMO), as well as evaluate the role of common genetic variants on MS susceptibility and course in a large case-control series from Canada.
2. Material and methods
A total of 2,221 MS patients, 14 NMO patients and 882 healthy controls recruited through the Canadian Collaborative Project on the Genetic Susceptibility to Multiple Sclerosis (CCPGSMS) were characterized in this study (Sadovnick et al., 1998). With the exception of 33 patients of Asian ethnicity (19 diagnosed with MS and 14 with NMO), all subjects were of European ancestry. The Research Ethic Board (REB) at the University of British Columbia approved the study, and all participants provided informed consent.
MS and NMO patients were diagnosed using published criteria (Polman et al., 2005, Poser et al., 1983, Wingerchuk et al., 2006). The mean age at blood collection was 46.7 years (SD ± 11.7) for MS patients, 42.9 years (SD ± 13.6) for NMO patients and 67.2 years (SD ± 9.8) for controls, with a male to female ratio of 1:2.75, 1:13 and 1:0.96 respectively. The mean age at MS onset was 31.0 years (SD ± 9.7), with a median Expanded Disability Status Scale (EDSS) score of 3.5 and an average of 4.0 (SD ± 2.6) (Kurtzke, 1983). The average age at onset for NMO patients was 34.3 years (SD ± 10.1) with a median EDSS score of 3 and an average of 3.8 (SD ± 1.8). Detailed clinical course at the time of sample collection was available for 1,753 MS patients of Caucasian ancestry (79.7%); the majority presented a clinical course consistent with relapsing remitting MS (RRMS) (75.3%) and the remaining were consistent with primary progressive MS (PPMS) (24.7%).
CH25H (NM_003956.3) coding variants were identified through mining of exome sequencing data from 226 patients diagnosed with MS or NMO. Exonic regions were enriched using an Ion AmpliSeq exome kit (57.7Mb) and sequenced in an Ion Proton sequencer (Life Technologies, Carlsbad, CA, USA). Sequencing reads for CH25H encompassed its entire coding sequence with an average of 191.5 (SD ± 86.7) reads per base. Sequences with a mapping Phred quality score under 20, fewer than 5 reads or over 95% strand bias were excluded from further analysis. The Ion Torrent Server was used to map reads to NCBI Build 37.1 reference genome and identify variants differing from the reference.
Tagging SNPs (tSNPs) spanning a 12 kb region encompassing the CH25H locus were selected based on HapMap data (version 3 release 27) using Haploview software (Barrett et al., 2005). Selected tSNPs (rs17382301, rs4933497, rs4417181 and rs1074599) captured over 95 % of the genetic variability in the region with an overall r2 value of 0.949 (minor allele frequency > 5% and r2 > 0.8 in Caucasian population standards). Genotyping of variants was performed, as previously outlined, using TaqMan probes on an ABI 7900 and analyzed with SDS 2.3 software (Trinh et al., 2013). Genotypic associations were analyzed using Pearson’s chi-squared goodness-of-fit test with one degree of freedom, and p-values below 0.0125 were considered significant after Bonferroni correction for four independent tests. Allele frequencies were used to estimate genotype distribution, which was consistent with Hardy-Weinberg equilibrium (p > 0.05).
3. Results
Sequencing the coding region of CH25H in 226 patients diagnosed with MS (n=212) or NMO (n=14) identified five coding variants (Table 1). Of those, four are previously reported synonymous changes and were excluded from further analysis (rs10887932, p.L18L; rs45579235, p.H74H; rs4078488, p.A168A; rs17382301, p.N219N). One missense variant (c.51G>C, p.Q17H) was identified in two female patients of Asian ancestry diagnosed with NMO. The CH25H p.Q17H has an overall reported population frequency of 3.3×10−5 in the Exome Aggregation Consortium (ExAC) database, and 4.9×10−4 in the East Asian population. No additional NMO patients were available for screening, and genotyping of CH25H p.Q17H in 2,221 MS patients and 882 healthy controls failed to identify any additional carriers.
Table 1.
CH25H coding variants identified in MS and NMO patients. Genomic coordinates are given from NCBI Build 37.1; dbSNP IDs are provided from build 138 when available; minor allele frequencies (MAF) were obtained from The Exome Aggregation Consortium (ExAC) database. NR, not reported.
| Chromosome: Position | Nucleotide change | Protein change | dbSNP ID | ExAC MAF |
|---|---|---|---|---|
| 10:90,966,999 | C/G | p.Q17H | – | 0.00003 |
| 10:90,966,996 | C/G | p.L18L | rs10887932 | 0.26 |
| 10:90,966,828 | G/A | p.H74H | rs45579235 | 0.003 |
| 10:90,966,546 | C/T | p.A168A | rs4078488 | 0.21 |
| 10:90,966,393 | G/A | p.N219N | rs17382301 | 0.12 |
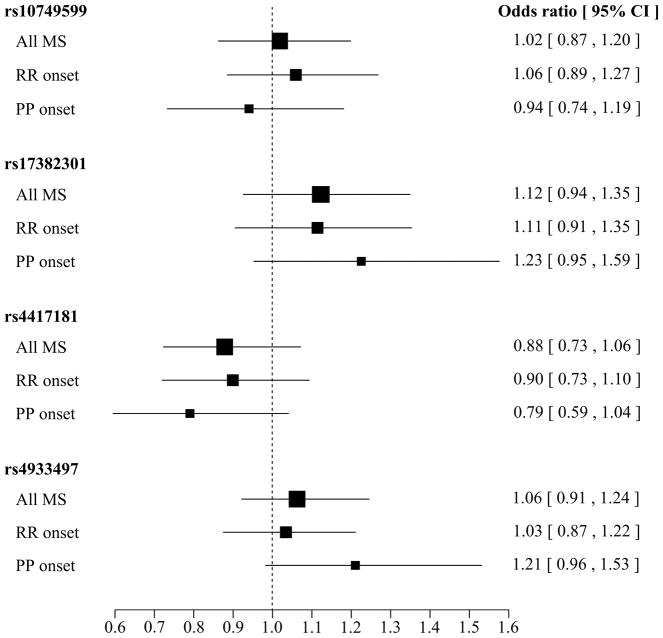
Genetic analysis of four tSNPs (rs1074599, rs17382301, rs4417181, and rs4933497) spanning the entire CH25H locus in Caucasian MS patients and controls did not identify a significant association between any of these variants and disease risk (Table 2). To assess whether variants in CH25H have an effect on disease course we performed a stratified analysis by clinical diagnosis at the onset of MS. Association analysis in 1,320 patients presenting with RRMS at the onset of disease did not identify a significant association with any of the CH25H variants genotyped; in contrast the analysis of 433 patients with PPMS identified a nominally significant association with rs17382301 and rs4417181 (Table 2). Although these associations do not withstand correction for multiple testing (p > 0.0125), this data suggests that carriers of the minor allele for rs17382301 may have an increased risk of developing PPMS (OR=1.23), whereas the minor allele for rs4417181 appears to be protective (OR=0.79) (Figure 1).
Table 2.
Genotype frequency and association analysis of CH25H tSNPs in MS patients (MS), patients with relapsing remitting (RR) disease at onset, and patient with primary progressive (PP) disease at onset.
| dbSNP | Genotypes | Groups | n (%) | p-value | ||
|---|---|---|---|---|---|---|
| rs10749599 | GG/GT/TT | Control | 328 (0.37) | 394 (0.45) | 155 (0.17) | |
| MS | 810 (0.37) | 1000 (0.45) | 389 (0.18) | 0.757 | ||
| RR | 475 (0.36) | 610 (0.46) | 234 (0.18) | 0.488 | ||
| PP | 168 (0.39) | 196 (0.45) | 68 (0.16) | 0.366 | ||
|
| ||||||
| rs17382301 | GG/GA/AA | Control | 668 (0.76) | 193 (0.22) | 18 (0.02) | |
| MS | 1623 (0.74) | 537 (0.24) | 39 (0.02) | 0.132 | ||
| RR | 978 (0.74) | 317 (0.24) | 25 (0.02) | 0.257 | ||
| PP | 312 (0.72) | 115 (0.27) | 6 (0.01) | 0.048 | ||
|
| ||||||
| rs4417181 | TT/TC/CC | Control | 662 (0.76) | 201 (0.23) | 10 (0.01) | |
| MS | 1697 (0.78) | 435 (0.20) | 40 (0.02) | 0.026 | ||
| RR | 1011 (0.78) | 269 (0.21) | 21 (0.02) | 0.123 | ||
| PP | 343 (0.80) | 77 (0.18) | 9 (0.02) | 0.015 | ||
|
| ||||||
| rs4933497 | AA/AG/GG | Control | 463 (0.53) | 347 (0.40) | 63 (0.07) | |
| MS | 1128 (0.52) | 891 (0.41) | 170 (0.08) | 0.416 | ||
| RR | 688 (0.52) | 524 (0.40) | 102 (0.08) | 0.614 | ||
| PP | 208 (0.48) | 188 (0.44) | 35 (0.08) | 0.104 | ||
Figure 1.
Forest plot of the odds ratio (OR) and 95% confidence intervals (CI) for rs1074599 (GG/GC vs. CC), rs4417181 (TT/TC vs. CC), rs4933497 (AA/AG vs. GG), rs17382301 (GG/GA vs. AA). Box dimensions are proportional to the sample size, with the horizontal line indicating the OR and corresponding 95%CI.
4. Discussion
Intermediates in cholesterol metabolism have been shown to play an important role in the innate and adaptive immune response (Spann and Glass, 2013). During cholesterol metabolism, CH25H converts cholesterol into oxysterols, promoting T-cell trafficking into the central nervous system and triggering a pro-inflammatory response, thus suggesting a potential role in the pathomechanism of MS.(Chalmin, Rochemont, 2015) This is further supported in the EAE model of MS which exhibits a reduced disease severity in CH25H deficient animals, or those exposed to cholesterol-lowering drugs.(Chalmin, Rochemont, 2015, Youssef et al., 2002) This data indicates that genetic mutations altering CH25H enzyme activity may play a role in disease susceptibility.
The genetic characterization of CH25H identified a rare variant, p.Q17H, in two patients of Asian ethnicity diagnosed with NMO (Table 1). Although evolutionarily conserved (GERP=4.58) (Davydov et al., 2010), in-silico analysis produced conflicting predictions with some algorithms considering the substitution tolerated (SIFT = 0.12) while others predict a damaging effect on protein function (Polyphen2 = 0.91) (Adzhubei et al., 2010, Kumar et al., 2009). This variant was not observed in any additional genotyped samples from controls or patient diagnosed with MS or NMO; thus further characterization, particularly in NMO patients, is necessary to elucidate whether CH25H p.Q17H plays a role in the onset of disease.
The analysis of common genetic variability in CH25H suggests a possible association in patients with PPMS (Table 2 and Figure 1). However, since these associations do not withstand correction for multiple testing, replication in a larger series of PPMS patients is necessary to confirm these findings. Extending from the identification of the rare p.Q17H variant, it would be important to also evaluate whether common genetic variability in CH25H has an effect on NMO disease susceptibility.
The functional interplay between cholesterol metabolism and the immune response has nominated CH25H as an enzyme potentially implicated in increased susceptibility to autoimmune diseases (Chalmin, Rochemont, 2015, Reboldi, Dang, 2014, Spann and Glass, 2013, Tall and Yvan-Charvet, 2015). In this study we have identified a rare CH25H p.Q17H mutation in two NMO patients and a nominal association between common variants and PPMS patients, thus further supporting a role of CH25H in autoimmune diseases and warranting further genetic evaluations in MS and NMO patients.
Acknowledgments
We are grateful to the individuals who participated in this study. We thank Kevin Atkins for data collection and extraction. This research was undertaken thanks to funding from the Canada Research Chair [950-228408] and Canada Excellence Research Chair programs [950-228408], Canadian Institute of Health Research [MOP-137051], Vancouver Coastal Health Research Institute, the Milan & Maureen Ilich Foundation [11-32095000], and the Vancouver Foundation [ADV14-1597]. Collection of clinical information and DNA samples was funded by the MS Society of Canada Scientific Research Foundation as part of the CCPGSMS.
Footnotes
Conflicts of interest
The authors declare that they have no conflict of interest.
References
- Adzhubei IA, Schmidt S, Peshkin L, Ramensky VE, Gerasimova A, Bork P, et al. A method and server for predicting damaging missense mutations. Nat Methods. 2010;7:248–9. doi: 10.1038/nmeth0410-248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005;21:263–5. doi: 10.1093/bioinformatics/bth457. [DOI] [PubMed] [Google Scholar]
- Chalmin F, Rochemont V, Lippens C, Clottu A, Sailer AW, Merkler D, et al. Oxysterols regulate encephalitogenic CD4(+) T cell trafficking during central nervous system autoimmunity. J Autoimmun. 2015;56:45–55. doi: 10.1016/j.jaut.2014.10.001. [DOI] [PubMed] [Google Scholar]
- Davydov EV, Goode DL, Sirota M, Cooper GM, Sidow A, Batzoglou S. Identifying a high fraction of the human genome to be under selective constraint using GERP++ PLoS Comput Biol. 2010;6:e1001025. doi: 10.1371/journal.pcbi.1001025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar P, Henikoff S, Ng PC. Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat Protoc. 2009;4:1073–81. doi: 10.1038/nprot.2009.86. [DOI] [PubMed] [Google Scholar]
- Kurtzke JF. Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS) Neurology. 1983;33:1444–52. doi: 10.1212/wnl.33.11.1444. [DOI] [PubMed] [Google Scholar]
- Polman CH, Reingold SC, Edan G, Filippi M, Hartung HP, Kappos L, et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol. 2005;58:840–6. doi: 10.1002/ana.20703. [DOI] [PubMed] [Google Scholar]
- Poser CM, Paty DW, Scheinberg L, McDonald WI, Davis FA, Ebers GC, et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol. 1983;13:227–31. doi: 10.1002/ana.410130302. [DOI] [PubMed] [Google Scholar]
- Reboldi A, Dang EV, McDonald JG, Liang G, Russell DW, Cyster JG. Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science. 2014;345:679–84. doi: 10.1126/science.1254790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadovnick AD, Risch NJ, Ebers GC. Canadian collaborative project on genetic susceptibility to MS, phase 2: rationale and method. Canadian Collaborative Study Group. Can J Neurol Sci. 1998;25:216–21. doi: 10.1017/s0317167100034041. [DOI] [PubMed] [Google Scholar]
- Spann NJ, Glass CK. Sterols and oxysterols in immune cell function. Nat Immunol. 2013;14:893–900. doi: 10.1038/ni.2681. [DOI] [PubMed] [Google Scholar]
- Tall AR, Yvan-Charvet L. Cholesterol, inflammation and innate immunity. Nat Rev Immunol. 2015;15:104–16. doi: 10.1038/nri3793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trinh J, Vilarino-Guell C, Donald A, Shah B, Yu I, Szu-Tu C, et al. STX6 rs1411478 is not associated with increased risk of Parkinson’s disease. Parkinsonism Relat Disord. 2013;19:563–5. doi: 10.1016/j.parkreldis.2013.01.019. [DOI] [PubMed] [Google Scholar]
- Wingerchuk DM, Lennon VA, Pittock SJ, Lucchinetti CF, Weinshenker BG. Revised diagnostic criteria for neuromyelitis optica. Neurology. 2006;66:1485–9. doi: 10.1212/01.wnl.0000216139.44259.74. [DOI] [PubMed] [Google Scholar]
- Youssef S, Stuve O, Patarroyo JC, Ruiz PJ, Radosevich JL, Hur EM, et al. The HMG-CoA reductase inhibitor, atorvastatin, promotes a Th2 bias and reverses paralysis in central nervous system autoimmune disease. Nature. 2002;420:78–84. doi: 10.1038/nature01158. [DOI] [PubMed] [Google Scholar]